27 November 2020: Review Articles
Role of Exosomes in the Progression, Diagnosis, and Treatment of Gliomas
Ji Shi1AEFG, Ye Zhang1BC, Bing Yao1BD, Peixin Sun1CD, Yuanyuan Hao1CF, Haozhe Piao1FG*, Xi Zhao2BFDOI: 10.12659/MSM.924023
Med Sci Monit 2020; 26:e924023
Abstract
ABSTRACT: Gliomas are the most common primary malignant brain tumors associated with a low survival rate. Even after surgery, radiotherapy, and chemotherapy, gliomas still have a poor prognosis. Extracellular vesicles are a heterogeneous group of cell-derived membranous structures. Exosomes are a type of extracellular vesicles, their size ranges from 30 nm to 100 nm. Recent studies have proved that glioma cells could release numerous exosomes; therefore, exosomes have gained increasing attention in glioma-related research. Recent studies have confirmed the importance of extracellular vesicles, particularly exosomes, in the development of brain tumors, including gliomas. Exosomes mediate intercellular communication in the tumor microenvironment by transporting biomolecules (proteins, lipids, deoxyribonucleic acid, and ribonucleic acid); thereby playing a prominent role in tumor proliferation, differentiation, metastasis, and resistance to chemotherapy or radiation. Given their nanoscale size, exosomes can traverse the blood-brain barrier and promote tumor progression by modifying the tumor microenvironment. Based on their structural and functional characteristics, exosomes are demonstrating their value not only as diagnostic and prognostic markers, but also as tools in therapies specifically targeting glioma cells. Therefore, exosomes are a promising therapeutic target for the diagnosis, prognosis, and treatment of malignant gliomas. More research will be needed before exosomes can be used in clinical applications. Here, we describe the exosomes, their morphology, and their roles in the diagnosis and progression of gliomas. In addition, we discuss the potential of exosomes as a therapeutic target/drug delivery system for patients with gliomas.
Keywords: Biological Markers, exosomes, Glioma, Disease Progression, Immunotherapy
Background
A glioma is the most common primary tumor of the central nervous system (CNS); it is characterized by high aggression, relapse, and mortality [1]. Gliomas account for 24.7% of all primary brain tumors and 74.6% of malignant brain tumors [2]. Approximately half of all the gliomas are glioblastoma multiforme (GBM). Patients with gliomas generally undergo conventional treatment, including surgical resection combined with radiotherapy and temozolomide (TMZ)-based chemotherapy. Although the molecular etiology of gliomas has been elucidated and advances have been made in glioma diagnosis and treatment, the prognosis of these patients undergoing treatment remains poor [3].
Extracellular vesicles (EVs) are membrane-bound subcellular organelles that bud from the cell surface and participate in transferring cytoplasmic or membrane-bound biomolecules to the neighboring cells, or the extracellular space [4]. EVs contain proteins, lipids, deoxyribonucleic acid (DNA), messenger ribonucleic acid (mRNA), and multiple noncoding RNAs [4]. EVs are released by several cells and play a key role in intercellular communication by transmitting their molecular contents to different cell types [5]. EVs are currently divided into 4 types: exosomes, microvesicles (microparticles), membrane particles, and apoptotic vesicles; while the terms “ectosomes” and “exosome-like vesicles” have been abandoned because there was insufficient evidence to support their existence [6]. Given their potential as a therapeutic target or a drug delivery system, this review describes the exosomes, their morphology, and their role in the diagnosis and progression of gliomas.
In 1983, Harding et al. [7] and Pan et al. [8] first described peptide-containing vesicles capable of mediating intercellular communication in sheep reticulocytes. These vesicles were termed “exosomes” by Johnstone et al. in 1989. They were eventually isolated and purified from reticulocytes [9]. For a long time, exosomes were considered to be cellular waste [9]. However, they are nanovesicles that are 30 nm to 100 nm in diameter and originate from endosomes. Exosomes are formed from multivesicular bodies and released extracellularly after fusing with the cell membrane. They exist in reticulocytes and other cell types, and can be found in various body fluids, including blood, cerebrospinal fluid (CSF), lymphatic fluid, urine, and pleural and abdominal effusions [10]. They recognize target cell(s), participate in signal transduction, and can transfer their contents into cells by direct fusion with the recipient cell membrane [11]. The biological functions exerted by exosomes vary according to the physiological conditions and their cellular origin [12]. The contents of some exosomes are similar, whereas exosomes from different subclasses specialize in specific biological functions [13]. As exosomes are rich and diverse in their biomolecular contents, can be obtained by minimally invasive techniques, and monitored dynamically, the detection of exosomes and their contents in the peripheral fluids (including blood and cerebrospinal fluid) of patients is an efficient way to diagnose tumors using a fluid biopsy [14]. Tumor cells secrete more exosomes than normal cells [14]. Recent studies have revealed that exosomes bind to receptors on the surfaces of target tumor cells via surface markers [15,16]. Moreover, given their nanoscale size, exosomes can freely pass through the blood-brain barrier (BBB). Therefore, exosomes have gained increasing attention in the diagnosis and treatment of malignant gliomas. In this review, we describe the contents of exosomes and their role in the diagnosis, progression, and potential therapeutic intervention (target/delivery system) of gliomas.
Exosomes
FORMATION AND SECRETION OF EXOSOMES:
The formation of exosomes begins with cell membrane invagination forming vesicles that contain cytoplasmic material called early endosomes [17]. Early endosomes mature into late endosomes when the microsomal membrane “sprouts” inward to form intraluminal vesicles that coalesce to form the multivesicular endosome [17]. Subsequently, the multivesicular endosomes employ multiple mechanisms to fuse with the cell membrane after which they are released into the extracellular matrix as exosomes [17]. The secretion of exosomes requires the concerted action of numerous families of proteins. Rab proteins, including Rab11, Rab27, and Rab35, and a soluble N-ethylmaleimide-sensitive-factor (NSF) attachment protein receptor play a key role in exosome secretion [18]. There are other factors, including changes in the intracellular pH and potassium concentration that regulate the formation and secretion of exosomes [19].
EXOSOME CONTENTS:
Exosomes are small vesicles (30 nm to 100 nm) that contain lipids, proteins, mRNAs, and micro ribonucleic acids (miRNAs) [20]. The commonly found exosomal proteins include cytoskeletal proteins (tubulin and microfilament-binding proteins), membrane fusion proteins (Rab proteins, ALIX, and flotillin), metabolic enzymes, and channel proteins. Exosomes also contain unique proteins depending on their biological source [21]. Some proteins that are overexpressed in tumor cells can be present in exosomes too, including Fas ligand proteins, tumor necrosis factor (TNF)-related apoptosis-inducing ligands, tumor antigens, and immunosuppressive proteins (transforming growth factor [TGF]-β) [22]. The lipids contained in exosomes include lysophosphatidic acid, cholesterol, and ceramide [21]. Exosomes encapsulate a large amount of RNA and are effective vectors for RNA transport into target cells; thereby, playing a regulatory role. This encapsulation ensures that the RNA molecules are protected from degradation by the intra- and extracellular RNases. Although numerous studies have shown the presence of protein and RNA in exosomes, few studies report the presence of DNA. However, mitochondrial and chromosomal DNA have been found in exosomes [23]. Exosomes have been demonstrated to contain 100 bases to 17 kilobases of double-stranded DNA [24]. Thakur et al. demonstrated that the majority of exosomes from tumor cells contain genomic double-stranded DNA that reflects the tumor mutation status [24]. However, the mechanism by which DNA is transported into the exosomes remains unclear. Some studies have suggested that a deregulated DNA repair pathway in tumor cells allows DNA to accumulate in the cytoplasm (as compared to healthy cells); thus making DNA available for entry into the exosomes [25].
MORPHOLOGY AND PHYSIOLOGICAL FUNCTIONS OF EXOSOMES:
The exosomes are circular or cup-shaped vesicles, primarily related to the metabolism and exchange of biomolecules between the cells and body fluids [26]. The donor exosomes can fuse with recipient cells and regulate their intracellular metabolism. Exosomes are considered to be signaling mediators. Specific enzymes within exosomes are responsible for converting their contents into signaling molecules that bind to the target-cell receptors and mediate the intercellular transfer of materials. The internalization of exosomes by target cells involves fusion with the plasma membrane, endocytosis, micropinocytosis, phagocytosis, and a lipid raft-mediated internalization [27]. Exosomes play a vital role in the differentiation and development of neurons, and in tumor growth, invasion, metastasis, angiogenesis, and immunity [26]. Therefore, biomarkers within the exosomes can be used in the diagnosis and treatment of certain diseases, such as miR-21, miR-222, miR-124-3p [28,29].
ISOLATION OF EXOSOMES:
Exosomes are found in multiple biofluids [30]. The current methods for exosome isolation are mostly based on their physical properties, including particle size and density; and include ultracentrifugation, density-based separation, precipitation, ultrafiltration, and immunoaffinity. Ultracentrifugation is regarded as the criterion standard for exosome separation because of its large extraction capacity, reliable purity, and its ability to completely satisfy the needs for subsequent biochemical and cellular experiments. However, it is difficult to differentiate between exosomes and other EVs [17]. Recently, Ibsen et al. [31] developed an alternating current-based, electrokinetic microarray chip device that requires only 30 μL to 50 μL of a plasma sample that can be processed in 15 minutes. Cumba Garcia et al. isolated exosomes from the plasma of GBM patients using convenient density gradient-based ultracentrifugation to analyze the presence of biomarkers [32]. The presence and purity of exosome preparations can be further assessed by western blotting for exosome-specific markers (CD9, CD63, ALIX, or TSG-101) [32].
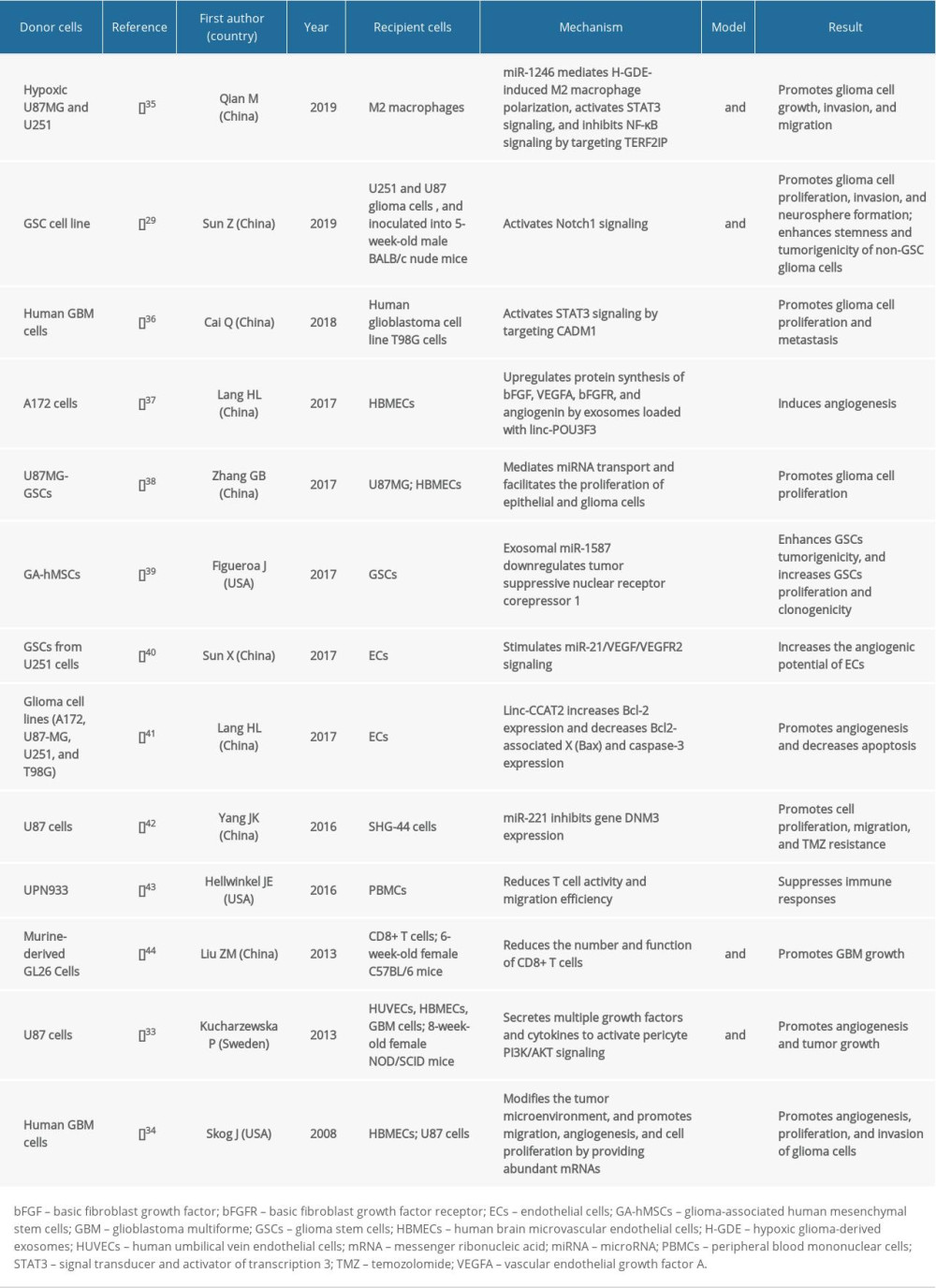
Role of Exosomes in the Progression of a Glioma
EXOSOMES AFFECT THE MICROENVIRONMENT AROUND GLIOMAS:
The resistance of malignant gliomas to various treatments is primarily attributed to their immunosuppressive tumor microenvironment (TME) that comprises tumor cells, fibroblasts, and immune cells [45]. Cellular communication between the tumor and its environment is mainly mediated by the shedding of membranous microbubbles that can fuse with other cells within the TME [46]. Exosomes play an important role in immunosuppression, stimulation of tumor progression, invasion, metastasis, and multidrug resistance [47,48].
EXOSOME-MEDIATED ANGIOGENESIS:
Angiogenesis is a critical event in glioma progression and numerous angiogenic factors in GBM-derived exosomes stimulate angiogenesis [49,50]. The tetraspanins selectively supply exosomes with proteins and mRNA to mediate the exchange of information between the exosomes and the vascular endothelial cells, thus promoting angiogenesis [51]. The GDEs carry proteases associated with malignancies, including MMP-9 and active MMP-2 along with a tissue plasminogen activator, and a high molecular weight urokinase-plasminogen activator, which are essential for angiogenesis. A variety of angiogenic factors (mainly epidermal growth factor receptor variant III [EGFRvIII]) are secreted by gliomas. The EGFRvIII can be “shared” between glioma cells by the intercellular transfer of exosomes via a phosphatidylserine-dependent mechanism [52]. When human astrocytes were incubated with exosomes containing EGFRvIII for 24 hours, the EGFRvIII bound the cell membrane and the phosphorylated extracellular protein kinases [52]. The angiogenesis-related genes and proteins play a vital role in pro-angiogenesis [37]. The U251 human glioma cells secrete exosomes containing various proangiogenic factors, which promote the proliferation, migration, and lumen formation of endothelial cells (ECs) [47]. Moreover, the GDEs regulate the angiogenic capacity of the ECs by up- or downregulating miRNA expression. Xu et al. reported that the glioma stem cell (GSC)-derived exosomes activate the angiogenic potential of ECs by increasing the levels of miR-21 and the proangiogenic growth factor, vascular endothelial growth factor (VEGF) [40]. Lang et al. reported that glioma cells stimulate angiogenesis in ECs mainly via the linc-POU3F3-containing exosomes [37]. The GDEs contain miR-296, miR-29a, and miR-30e to regulate the ECs-mediated angiogenesis. The exosomes derived from GSCs or glioma cells enhance tube formation and migration of the ECs, indicating that the malignant cells affect the function of the surrounding cells [40]. Skog et al. reported that the tubule length of the human brain microvascular ECs (hBMECs) doubles within 16 hours in the presence of the exosomes derived from GBM, and the ECs in the brain microvasculature internalize the GDEs that are responsible for tubule formation and angiogenesis [34]. Lang et al. found that glioma cells stimulate angiogenesis by transferring the long intergenic noncoding-RNA CCAT2 (linc-CCAT2) to the ECs via the exosomes, suggesting that they are the potential therapeutic targets in gliomas [41]. Monteforte et al. reported that the exosomes from GBM induce angiogenesis during peripheral ischemia, and contain proteoglycans and small RNAs that promote revascularization [53]. Thus, exosomes can be the potential therapeutic targets for multiple cancers associated with enhanced capillary formation.
EXOSOMES AND GLIOMA HYPOXIA:
Hypoxia is a hallmark of the GBM microenvironment; it regulates the transcriptome and proteome of the tumor cells [50]. The exosomes play a prominent role in the adaptation of tumor cells to hypoxia, which is related to angiogenesis, tumor progression, and metastasis. Hypoxic stress modulates the physiological activity of the surrounding or distant cells; thus, qualitatively and quantitatively altering the protein content of the GBM cell-derived exosomes. A recent study has revealed that hypoxic exosomes regulate the glioma cells by upregulating the transcription of small nucleolar RNA, C/D box 116-21, and downregulating the potassium voltage-gated channel subfamily J member 3 [50]. The protein cargo in exosomes differentially regulates the expression of certain genes. The hypoxic status of the glioma cells is mirrored in their cargo [33]. King et al. reported that in response to hypoxia, cancer cells improve their survival and invasiveness by enhancing the secretion of exosomes into the microenvironment [54]. In addition, the exosomes secreted by the hypoxic cancer cells promote tumor progression in the normoxic areas [48]. The hypoxia regulates the exosomal miRNA content and release [54–56]. A tube formation assay with endothelial progenitor cells indicated that compared to normoxic exosomes, the hypoxic exosomes enhanced more features associated with angiogenesis [50]. The GDEs contain various mRNAs and proteins that regulate hypoxia; this is often associated with poor prognosis, and the resistance of gliomas to radiotherapy and chemotherapy [57]. Kucharzewska et al. revealed that the GBM cell-derived hypoxic exosomes regulate ECs by secreting various growth factors and cytokines that stimulate pericyte PI3K/AKT signaling and migration [33]. The GBM-derived exosomes show enhanced autocrine and promigratory functions under hypoxic conditions. Thus, the hypoxic exosomes play a vital role in tumor vascularization, pericyte vessel coverage, and GBM cell proliferation [33]. Qian et al. found that hypoxic GDEs (H-GDEs) promote glioma progression and facilitate the formation of an immunosuppressive microenvironment by inducing M2 macrophage polarization [35]. The H-GDEs are potent stimulants of myeloid-derived suppressor cells that regulate the formation of an immunosuppressive environment while evading host immunity against tumors. Guo et al. demonstrated that hypoxia upregulates the miR-10a and miR-21 contents in the GDEs, and activates myeloid-derived suppressor cells via the RORα/IκBα/NF-κB and PTEN/PI3K/AKT pathways [58]. miR-29a and miR-92a in the H-GDEs mediate the differentiation of functional myeloid-derived suppressor cells [59]. Thus, the H-GDEs play an important role in the development of gliomas.
ROLE OF EXOSOMES IN THE PROLIFERATION AND INVASIVENESS OF GLIOMAS:
Proliferation and invasion are regulated by the exosomes and are important for glioma cell survival and glioma recurrence [60]. The GDEs are indispensable in the regulation of tumor proliferation, survival, and invasion due to their coding and noncoding RNA contents. Cai et al. [36] reported that the GBM progression is promoted by exosomal miR-148a (which targets CAMD1) and increased signal transducer and activator of transcription 3 (STAT3) activation. The exosomes derived from glioma-associated-human mesenchymal stem cells (GA-hMSCs) contain miR-1587, which targets the tumor suppressor gene NCOR1 in GSCs; thereby altering the biological functions of the GSCs and increasing tumor cell proliferation and colony formation [39]. The GSC-derived exosome-mediated miRNA transport facilitates the proliferation of epithelial and glioma cells [39]. Further, exosomes secreted by the GSCs mediate the dedifferentiation of non-GSC glioma cells into GSCs via the Notch signaling pathway, and enhance the stemness and tumorigenicity of non-GSC glioma cells [29,38].
Integrins, adhesion molecules, and other extracellular matrix factors play a prominent role in regulating cell migration. The GDEs contain the adhesion/recognition molecule L1CAM. A disintegrin and metalloprotease 10 (ADAM10) catalyze the proteolysis of L1CAM to release its extracellular domain, which allows an interaction with the fibroblast growth factor receptor, and integrins β1 and α5 on the target cells. This results in the activation of the integrin ligase, focal adhesion kinase (FAK); upregulation of PI3K/AKT, and NF-κB signaling to induce cell migration [61]. The NEDD4 family interacting protein 1 (NDFIP1) promotes the cellular uptake of PTEN-containing exosomes; thereby downregulating the PI3K/AKT signaling. The NDFIP1 expression is suppressed in a variety of tumors, including gliomas, resulting in a decrease in the uptake of PTEN-containing exosomes to maintain PI3K/AKT signaling, and promoting tumor survival and proliferation [62]. A study by Pinet et al. revealed that silencing the chitinase protein (YKL-40) led to downregulation of the tyrosine kinase receptor B (TrkB), sortilin, and p75NTR, resulting in low invasiveness [63]. The migration and activation of the YKL-40-inactivated cells could be rescued by the TrkB released from the exosomes secreted by the glioma cells. Arscott et al. reported that therapeutic radiation affects the production of exosomes in a dose- and time-dependent manner; radiation-induced exosomes are more likely to be absorbed in cocultured cells [64]. Moreover, the activation of TrkA and FAK signaling enhanced cell migration by changing the molecular composition of the exosomes. Thus, exosomes regulate intercellular signaling upon stimulation by radiation.
ROLE OF EXOSOMES IN RESISTANCE TO GLIOMA TREATMENT:
Exosomes play an important role in the treatment resistance exhibited by gliomas as they can export therapeutics from tumor cells [65]. Exosomes can promote the formation of fibroblasts causing fibroblastic reactions, which form a barrier to antitumor drugs. However, exosomes can also transform drug-sensitive tumor cells into drug-resistant tumor cells via biomolecules, such as the miRNA [66,67]. Hypoxia promotes the resistance of gliomas to chemotherapy by inducing specific signaling pathways depending on the uptake of exosomes [68]. Moreover, it has been suggested that the GDEs regulate the surrounding cells and create an optimal environment for glioma cell growth [69]. O-6-methylguanine-DNA methyltransferase (MGMT) and alkylpurine-DNA-N-glycosylase (APNG) are essential for repairing TMZ-induced DNA damage; their expression negatively correlates with therapeutic efficacy [70]. Shao et al. reported that in TMZ-resistant cell lines, the exosomal mRNA levels of MGMT and APNG increased rapidly upon TMZ treatment; whereas in TMZ-sensitive cell lines, they decreased, indicating that the exosomal transcript level of MGMT can predict the efficacy of TMZ in GBM patients [70]. The APNG promotes chemoresistance to TMZ in patients with GBM, and the co-expression of APNG with MGMT further enhances drug resistance [71]. Garnier et al. reported that exosome-transcribed MGMT mRNA is released by the GSCs and reflects the resistance to TMZ [72]. Moreover, Yu demonstrated that the exosomal MGMT mRNA levels of reactive astrocytes are significantly higher than those of nonreactive astrocytes. The reactive normal human astrocytes enhance recipient-cell chemoresistance to TMZ by transferring exosomal MGMT mRNA to the surrounding glioma cells; thereby suppressing apoptosis [68]. Zeng et al. reported that the protein tyrosine phosphatase receptor type Z1 (PTPRZ1)- receptor tyrosine kinase (MET) fusion-containing GDEs increase the resistance of GBM to TMZ and regulate the microenvironment by targeting multiple pro-oncogenic effectors in the recipient cells. Thus, the PTPRZ1-MET and gene fusions in exosomes play a crucial role in GBM invasiveness [73]. The exosomal miRNA-221 in the blood of glioma patients positively correlates with the malignant grade and resistance of gliomas to TMZ [42,74].
EXOSOMES MEDIATE IMMUNE RESPONSES:
Exosomes play an important role in the communication between tumor and immune cells. The exosomes secreted by tumor cells carry tumor-specific antigens that activate and inhibit the immune system (under specific conditions) and promote GBM proliferation, invasiveness, and chemoresistance. The differentiation and function of the immune cells can be regulated by miRNAs released from the tumor-derived exosomes [75]. These tumor-derived exosomes have multiple physiopathological roles and act on various immune cells, including effector T cells, naturally occurring Treg cells, and natural killer (NK) cells, which are involved in immune suppression and tumor progression [51,76]. Numerous immunomodulatory molecules can be found in the serum exosomes isolated from GBM patients, including antigen-presenting molecules, TGF-β, tumor antigens, and immune intracellular adhesion molecules [77]; indicating that tumors exert immunosuppressive activity across the CNS barrier. Moreover, Harshyne et al. reported that the serum exosomes from GBM patients drive M2 polarization in normal monocytes, indicating a bias towards the reactivity of T helper 2 cells (Th2). The Th2 responses are considered undesirable in tumor immunotherapy since they alleviate cytotoxic antitumor immune mechanisms and help escape cell-mediated immunity [78]. Moreover, microglia-derived exosomes mediate immune responses important for inflammation, degeneration, and tumorigenesis in the CNS [79]. Dendritic cells (DCs) are professional antigen-presenting cells. Upon incubation with the GDEs, they induce T cell activation and mediate cytotoxicity against gliomas in vitro [80]. Hellwinkel et al. [43] demonstrated that immunosuppressive phenotypes and high cytokine concentrations in the tumor-derived exosomes lead to a decreased expression of other cytokines, including interleukin 2 (IL-2) and CD69, and T cell activity; consequently hindering lymphocyte migration and inhibiting tumor immunity. The exosomes derived from the GBM GL26 cells reduced the number and function of cytotoxic CD8+ T cells, stimulating tumor growth [44]. The GDEs help differentiate peripheral blood-derived monocytes into alternately activated M2 tumor-supporting macrophages [81], and regulate the cytokine output and migratory ability of mitogen-stimulated, healthy, peripheral blood mononuclear cells [43].
An immune system evasion is the hallmark of glioma development and progression. The GSC-derived exosomes promote evasion from tumor immunity via intercellular communication. Yu et al. [82] demonstrated that the GSC-derived exosomes induce peripheral T cell immunosuppression. This involves active monocytes and the conversion of peripheral T cells to the monocytic, myeloid-derived suppressor cell-induced tumorigenic phenotype. Since exosomes promote tumor progression by mediating intercellular communication, they can be used as a tool to program anticancer immunity and restore the effector function of immunosuppressive cells. Konrad et al. demonstrated that mononuclear cells regulate the M2 immunosuppressive phenotype and the expression of the programmed death ligand 1 by specific and rapid uptake and release of the GSC-derived exosomal biomolecules. This includes STAT3; a vital molecule in the tumor-mediated immune suppression [83]. In conclusion, the exosomes derived from GSCs are important regulators of the immunosuppressive TME in patients with GBM [83].
Exosomes in the Diagnosis and Prognosis of Gliomas
MRNA:
Tumor-specific RNA in serum exosomes can be used to detect disease progression, and predict the prognosis of tumors in the CNS [20]. Chen et al. [85] detected mutant IDH1 mRNA transcripts in exosomes from the CSF of glioma patients, which also revealed that the exosomes from the CSF of glioma patients contain mutant IDH1 transcripts. Exosomal mRNA levels of MGMT and APNG can be used to monitor the therapeutic efficacy of TMZ in glioma treatment [70]. Yang et al. found that the genes p65 and dynamin 3 (DNM3) were upregulated in exosomes from the brain and blood of patients with primary and recurrent GBM by a xenograft orthotopic mouse model, suggesting that the exosomal levels of specific transcripts may be potential clinical diagnostic markers for GBM [30].
MIRNAS:
miRNAs are small (~22 nucleotide long) noncoding single-stranded RNAs involved in the regulation of post-transcriptional gene expression. The dysregulated miRNAs affect cellular physiology and development, leading to disease states [86]. Wei et al. showed that the exosomes isolated from GBM patients contain a large pool of miRNAs [87]. Since miRNAs are relatively stable within the exosomes and exhibit differential expression based on the tumor type, they can be used as biomarkers for the diagnosis and prognosis of tumors [88]. Santangelo et al. reported that compared with healthy controls and low-grade glioma patients, the serum exosomes from high-grade glioma patients contained significantly higher levels of miR-21, miR-222, and miR-124-3p, which decreased rapidly after neurosurgical therapy [89]. The GDE contents of miR-21, miR-222, and miR-124-3p can be determined using minimally invasive methods that can help diagnose brain tumors, predict the pathological grading of gliomas, and preoperative metastases. However, there is a need for more prospective studies to confirm whether the changes in serum exosomal-miRNA concentrations in glioma patients reflect tumor recurrence after surgery, and help devise personalized treatment options.
In 2014, Manterola et al. [90] analyzed the miR-574-3p, miR-320, and RNU6-1 contents in the serum exosomes isolated from GBM patients and healthy individuals; RNU6-1 alone or in combination with numerous miRNAs could specifically diagnose GBM. The expression of miR-301a was markedly increased in the serum exosomes from GBM patients when compared with those of healthy individuals, and correlated with the pathological grade and Karnofsky performance score of GBM patients [91]. The postsurgical levels of miR-301a secreted from the serum exosomes of glioma cells were remarkably decreased and associated with an increase in tumor recurrence. This indicates that exosomal miR-301a levels reflect the actual status and pathological changes of the glioma. Transcription polymerase I and releasing factor (PTRF, also known as Cavin-1) is stably detected in glioma tissues, serum and CSF samples. PTRF overexpression induced malignancy in the surrounding cells by regulating the TME and leading to a poor prognosis for the glioma patients; whereas downregulation of PTRF reversed the malignant phenotype of GBM, indicating that PTRF is an ideal diagnostic and prognostic indicator, and a promising therapeutic target against GBM [92]. Murgoci et al. reported high levels of miR-221 in the exosomes derived from the serum and CSF of GBM patients, suggesting its potential as a GBM biomarker [79]. miR-21 is the most consistently overexpressed miRNA in malignant gliomas and regulates glioma cell invasiveness, migration, and the grades of gliomas [55,93,94]. The amount of miR-21 in the exosomes isolated from the CSF of GBM patients was 10-times higher when compared with that in the exosomes from healthy subjects [93].The miR-21 levels in the serum exosomes from GBM patients were 40-times higher than those from the serum exosomes of healthy individuals [34]. The receiver-operating characteristic curve analysis revealed the high efficacy (area under the curve: 0.927, 95% confidence interval, 0.865 to 0.985) of using exosomal miR-21 in the diagnosis of gliomas [94]. Together, these findings indicate that only a small amount of serum or CSF is required for the extraction of exosomes to determine the diagnosis, grading, and prognosis of gliomas based on exosomal miR-21. Recent research has shown that miR-1246, the most abundant miRNA in H-GDEs mediates H-GDE-induced M2 macrophage polarization, and the miR-1246 levels decreased after surgery. The exosomal miR-1246 in the CSF of GBM patients activates STAT3 signaling and inhibits NF-κB signaling by targeting the telomeric repeat-binding factor 2-interacting protein (TERF2IP), and serves as an innovative biomarker for the diagnosis of GBM [35]. The exosomal levels of miR-10b and miR-143/145 also correlate with the prognosis of gliomas [95]. Some miRNAs, such as miR-128 and miR-34a, negatively correlate with the increasing malignancy of gliomas [96]. Therefore, the exosomal miRNAs are excellent candidates as biomarkers because of their stability, ease of collection and detection, and tissue and cell specificity. They can be used for early diagnosis of malignant gliomas with accurate stratification. These features make exosomal miRNAs suitable biomarkers for patients who cannot undergo surgery or are suspected of relapse. Fluid biopsies using exosomes can be a promising approach for the development of precision medicine in the future.
PROTEIN:
The protein contents in exosomes correlated with the malignancy of gliomas [97], indicating their importance in the diagnosis and prognosis of gliomas. Shao et al. [98] reported that 90% of patients with GBM had at least 1 protein from either EGFR, EGFRvIII, podoplanin, and IDH1 differentially expressed in their exosomes. EGFRvIII exists in 25% of glioma patients and can be detected with sensitively and specificity [34]. However, EGFRvIII cannot distinguish between the different grades of gliomas, since it is found only in high-grade gliomas. The sensitivity of EGFRvIII detection depends on the tumor size, location, serum or CSF content, and method of RNA extraction [34]. Setti et al. reported that the chloride intracellular channel 1 detected in exosomes promotes the proliferation and invasiveness of GBM, and is related with poor prognosis [99]. Phillip et al. found that glioma patients release CD9+/GFAP+/SVN+- and CD9+/SVN+-containing exosomes into circulation; the quantities decrease significantly following anti-survivin immunotherapy. This could be related to longer progression-free survival [100]. Currently, the detection of changes in the protein contents of body fluids or tissues is the most commonly used diagnostic method for the diagnosis, treatment, and prognosis of gliomas.
DNA:
DNA has been detected in the exosomes obtained from the serum of pancreatic, and prostate cancer patients. García-Romero et al. successfully detected mutations in gene IDH1 (R132H) in the peripheral blood exosomes of patients with high-grade gliomas; consistent findings were obtained from the immunohistochemistry of surgical specimens [101]. However, there are only a few reports on the presence of DNA in exosomes from the body fluids of glioma patients, and the accuracy of exosomal DNA detection in disease prognosis remains to be determined [101].
Exosomes in the Treatment of Gliomas
EXOSOMES AS THERAPEUTIC AGENTS:
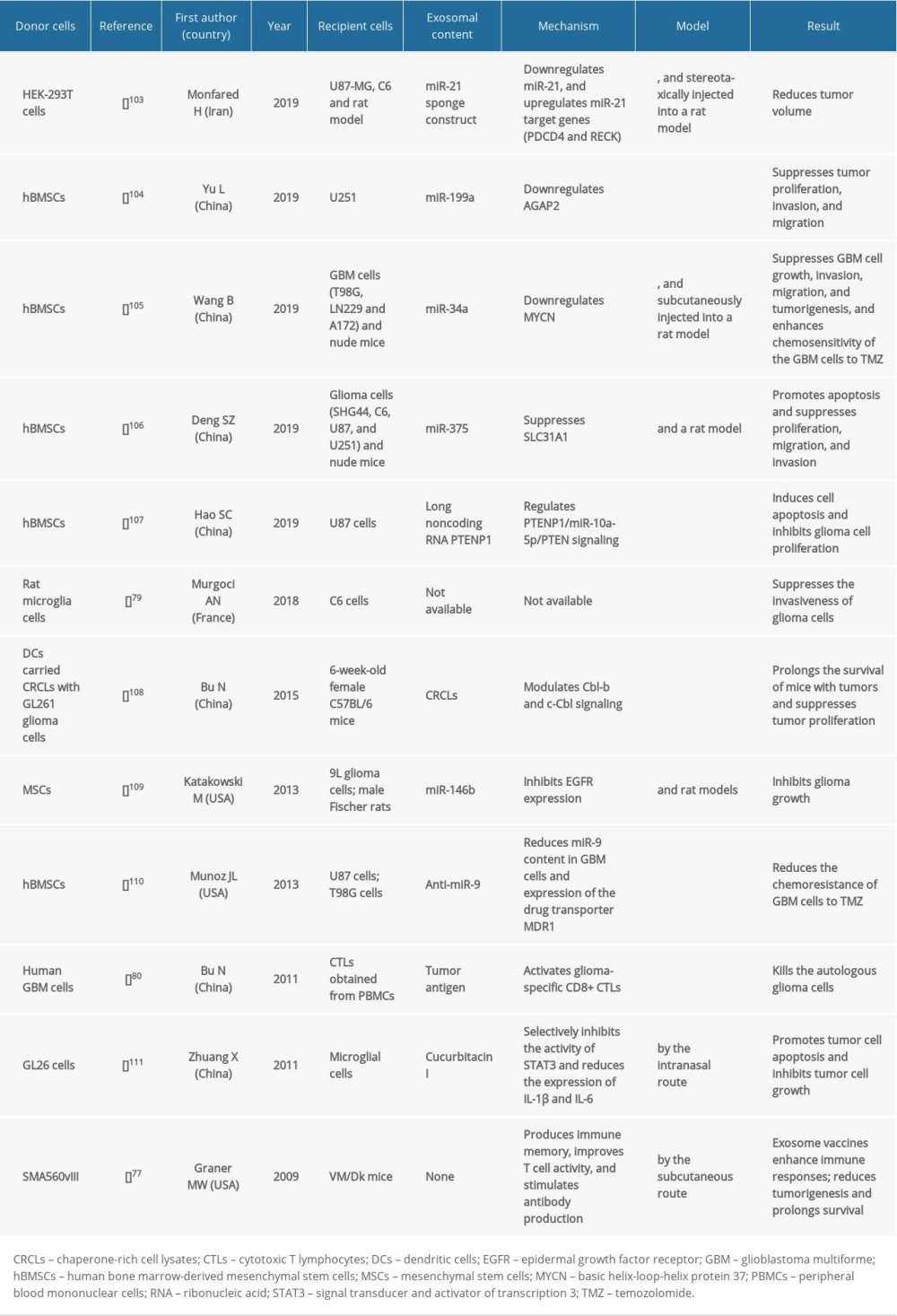
Multiple studies have shown that cell-derived exosomes have potential in disease treatment. NK cell-based immunotherapy is a low-toxicity alternative to chemotherapy and radiotherapy. The NK cell-derived exosomes target GBM cells and play an antitumor role. However, the underlying mechanism remains unclear [104,112]. Hao et al. [107] found that exosomes from the human umbilical cord-derived mesenchymal stem cells (hUC-MSCs) exert partial antitumor activity by regulation of the miR-10a-5p/PTEN signaling. Therefore, this axis may be a possible target for early diagnosis and treatment of gliomas. Combining gene-engineered exosomes with other drugs, including temozolomide, can improve the therapy against GBM cells and make it more effective. The exosomes derived from the embryonic stem cells that were conjugated with a tumor-targeting c(RGDyK) peptide and loaded with paclitaxel showed enhanced targeting and better curative efficacy of the paclitaxel towards the GBM cells [113]. To obtain glioma-targeting exosomes, Gang et al. loaded superparamagnetic iron oxide nanoparticles with curcumin and conjugated them with a neuropilin-1-targeted peptide by click chemistry for enhanced stability and biocompatibility [114]. This approach yielded good exosomal targeting towards glioma cells, and could be used in the early diagnosis and estimation of drug efficacy against GBM. Numerous studies are being conducted on the chemical modification and genetic engineering of exosomes to improve the BBB permeation; however, the practicality of this approach remains unclear.
Focused ultrasound (FUS) is a noninvasive technique that transiently and reversibly disrupts the stability of the BBB. Chen et al. demonstrated that the BBB could be “opened” by FUS to increase the delivery of IL-12 to brain tumors [115]. Given the lack of specific targeting, the number of natural exosomes that can cross the BBB are inadequate. Bai et al. developed a natural and secure transportation system using FUS to increase the targeted delivery of exosomes for glioma treatment [116]. FUS can be used with blood serum-derived exosomes to inhibit the growth of a glioma without obvious side effects, indicating that this combination is potent for developing glioma therapeutics. Murgoci et al. used a 3D spheroid glioma model to show that microglia-derived exosomes suppress tumor invasion in a time-dependent manner, and suggested their potential as nanotherapeutic agents against glioma cells [79].
EXOSOME-MEDIATED DRUG OR GENE DELIVERY:
To exploit the exosome potential in clinical applications, an increasing number of studies have focused on utilizing the nanoscale characteristics of exosomes to package and deliver bioactive substances, such as small molecules (anticancer drugs), proteins, and miRNAs [117].
DRUG DELIVERY: As exosomes are EVs that originate from multivesicular bodies, they do not trigger adverse immune responses, and are not recognized by the complement system. They are regarded as an innovative delivery system because of their properties of low toxicity, low immunogenicity, excellent biocompatibility, and transport of specific molecules and surface markers [118]. Previous studies have demonstrated the exosome-mediated transfers of lipophilic and hydrophilic drugs, including curcumin [119] and doxorubicin [120]. The current obstacles seem to be the lack of understanding about penetration of the BBB for delivering drugs into the CNS and striking a balance between the blood concentration of the brain parenchyma, and effective therapeutic drug concentration. As exosomes are nanoscale in size, they can efficiently penetrate the BBB via surface proteins that have a donor cell signature [121]. This has been achieved using the exosomes containing metalloproteinases in combination with tumor necrosis factor alpha (TNF-α), interferon γ (IFN-γ), and IL1-β, which promoted the degradation of the extracellular matrix and nitric oxide production, participated in leukocyte or T cell-mediated permeation, and disrupted the BBB [122]. Grapp et al. showed that fusion of the exosomal surface folate receptor-α with the receptor cell membrane helped exosomes permeate the BBB allowing exosome entry into the brain parenchyma from the CSF [123].
The BBB permeability poses a critical obstacle for the delivery of chemotherapeutic drugs. The properties of exosomes make them suitable for the treatment of CNS tumors, and exosome-based combined drugs can overcome the issues of current treatments for GBM [124,125]. There are 3 techniques used to conjugate exosomes with drugs: incubation, the easiest method involves co-incubation [126]; electroporation, which involves the penetration of exosomes using short, high-voltage pulses [127]; and sonication, drugs (such as paclitaxel) can be loaded into exosomes by effective sonication [126]. Alvarez-Erviti et al. [128] injected rabies viral glycoprotein-targeted exosomes intravenously into wild mice; the exosomes permeated the BBB and released glyceraldehyde-3-phosphate dehydrogenase (GADPH)-targeting carrier small interfering RNAs (siRNAs) to the neurons to specifically knock down neuronal GAPDH. The ECs are an important part of the BBB. The exosomes derived from ECs contain various receptors, including the transferrin receptor (TFRC), low density lipoprotein receptor (LDLR), insulin receptor (INSR), and transmembrane protein 30A (TMEM30A). The EC-derived exosomes possess transmembrane activity that could transport substances across the BBB, and enable signaling between the nerve cells and the extracellular material [129]. The exosomal surface derived from glioma cells comprises the pro-permeability protein semaphorin 3A. It disrupts the endothelial barrier in an Nrp1-dependent manner [130]. Consequently, the exosomes can mediate communication across the BBB. Yang et al. [124] demonstrated a decrease in the fluorescent intensity of xenotransplanted zebrafish cancer models and tumor growth markers upon the delivery of anticancer drugs by the brain ECs-derived exosomes. STAT3 is an important protein in multiple malignant tumors, including GBM. Zhuang et al. [111] intranasally administered exosomes containing the STAT3 inhibitor cucurbitacin I into mice with GBM, which significantly prolonged the survival of these mice from 20 days to 44.5 days without adverse reactions or behavioral abnormalities. Salarpour reported that the loading capacity of exosomes depends on the technique used, and chemotherapeutics exhibit a stronger cytotoxicity when encapsulated into exosomes [118]. Therefore, exosomes are promising agents for delivering drugs against brain tumors.
GENE DELIVERY: miRNAs are promising candidates for GBM therapy. However, the challenges include identifying the most effective miRNAs and the delivery method [131]. Exosomes are good carriers of miRNAs that exert anticancer activity against multiple cancer types. The exosomes secreted by the miRNA-transfected mesenchymal stem cells (MSCs) have potent efficacy in treating gliomas [132]. Lang et al. [133] isolated the exosomes derived from miR-124a-expressing (via lentiviral expression) MSCs that released miR-124a, which could be used as therapeutic agents against gliomas. A study by Yu et al. revealed that the exosomes derived from MSCs transport miR-199a to the glioma cells to downregulate AGAP2 and inhibit glioma cell proliferation, invasion, and metastasis [104].The overexpression of miR-199a in MSCs improved the chemosensitivity to TMZ, and suppressed glioma cell proliferation. Similarly, Munoz et al. demonstrated that the anti-miR-9-delivering exosomes increased the multidrug transporter expression and sensitivity to TMZ in drug-resistant GBM cells, resulting in higher rates of cell death and caspase activity [110]. The miRNA-146b-containing exosomes derived from MSCs inhibited GBM cell growth [109]. miR-34a released by the exosomes derived from hBMSCs suppressed MYCN expression inhibiting glioma cell growth, invasion, and metastasis, and improving the sensitivity of GBM to TMZ [105]. miR-375 secreted by the hMSC-derived exosomes suppressed SLC31A1 expression and increased apoptosis, while inhibiting glioma cell proliferation, migration, and invasion [106]. Ma et al. showed that the GDEs increased glycolysis and tumor-like transformation of the hBMSCs, indicating that interrupting the interaction between exosomes and MSCs in the TME shows promise as therapy against gliomas [134]. miRNA inhibition using sponge-like constructs is also used as a treatment strategy. Sponge constructs are designed to bind corresponding miRNA(s) or their seed sequences, thus preventing miRNAs from binding to their biological targets [135]. Hamideh et al. reported that exosomes containing an miR-21 sponge decreased the tumor volume in a rat model of GBM; thus, miR-21 sponges seem to offer a promising therapeutic approach [103]. Exosome-mediated intercellular signaling during tumor proliferation is a potential therapeutic target [38]. The exosomal long noncoding-RNA antisense transcript of the hypoxia-inducible factor-1α promoted GBM progression and radioresistance by downregulating the HIF-1α expression; hence, it is a potential therapeutic target in GBM [136]. Immature mouse DCs show promise in the production of exosomes with low toxicity and immunogenicity. Tian et al. found that exosomes modified by targeting ligands improved the tumor targeting of doxorubicin, resulting in a stronger suppression of tumor proliferation without toxicity [120]. Sugahara et al. fused an αv integrin-specific iRGD peptide to the surface of the exosomes derived from engineered immature mouse DCs, which facilitated tumor targeting and the absorption of anticancer drugs [137].
Numerous techniques have been applied to producer cells to secrete exosomes with specific cargo. However, low exosome abundance is one of the main obstacles in developing exosome-based treatment. Li et al. reported that small noncytotoxic pharmacological molecules, including MOPIPP and vacuolin-1, promote the production of exosomes and can be used to treat GBM [138]. Other current challenges associated with exosome-based treatment are donor-cell selection, drug encasement into exosomes, and drug resistance [14]. Therefore, miRNAs can be efficacious treatment agents for multiple diseases, and for the delivery of different synthetic molecules and biomolecules in cell therapy.
EXOSOME-BASED IMMUNOTHERAPY:
Lymphocytes could enter the CNS when the BBB was damaged by brain tumors [139]. Immunotherapy as a treatment for gliomas has recently gained importance. Exosome-mediated immunotherapy is an efficient treatment strategy [108]. The DCs are the most efficient antigen-presenting cells that stimulate a strong immune response. The DC-derived exosomes can enhance antitumor immunity in vitro and in vivo [140]; tumor vaccines can be designed based on this information [141]. Immunotherapy for gliomas has recently gained importance as malignant glioma cell growth is influenced by the immune system. The GDEs are antigen transporters that once loaded into the DCs activate the CD8+ cytotoxic T lymphocytes (CTLs) and kill autologous glioma cells [80]. Positive results for the DC-based vaccine immunotherapy for GBM have been obtained in vitro, in vivo, and in early-phase clinical trials [142,143]. However, the overall efficacy of DC vaccines remains unclear. Therefore, there remains an urgent need to improve immunotherapy [144,145]. Ning et al. found that the DC-derived exosomes containing chaperone-rich cell lysates stimulate stronger and more effective antitumor T cell immune responses by regulating Cbl-b and c-Cbl signaling [108]. Liu et al. demonstrated that the α-Galcer-containing tumor-derived exosomes constitute efficacious DC-based vaccines. Alpha-Galcer-activated invariant NK-T cells were used as a cellular adjuvant, thus breaking immune tolerance and inducing an antigen-specific CTL response against the GBM cells [146]. The GDEs can be used as a storage pool for tumor antigens, which can overcome the limitations associated with immunotherapy against malignant gliomas to some extent. For example, phenotypic changes can be observed in patients with progressing gliomas by extracting exosomes, which helped to develop a vaccine that stimulates antitumor immunity in the host [147]. Using animal experiments, Graner et al. [77] showed that the exosome-based vaccines induced strong antitumor immunity via humoral and cellular pathways, and prolonged the survival of mice with gliomas. Changes in the total exosomal protein and mRNA contents in glioma patients treated with antitumor vaccines can be used to monitor their immune and clinical responses. Muller et al. [148] showed that the exosomal immune-related protein content positively correlates with the different grades of gliomas, indicating the significance of immune-related gene expression in serum exosomes in the response of glioma patients to these vaccines.
Conclusions
Since the discovery of exosomes, their composition, functions, and correlations with various diseases have been widely studied. Their involvement in gliomas has gained attention over the years. Exosomes are involved in the immune evasion, tumor hemodynamic remodeling, and progression of gliomas. Exosomes contain a variety of biomolecules that can serve as biomarkers for the diagnosis [149]. Since exosomes are detected in body fluids such as serum or CSF, they allow for minimally invasive diagnosis and treatment of gliomas. Malignant gliomas secrete an abundance of exosomes that enter the peripheral fluid through the BBB. They contain various bioactive antigens that are tumor-specific and participate in the onset and development of gliomas. Therefore, exosomes have been speculated to be promising candidates for the diagnosis and treatment of gliomas. Exosomes are nanovesicles that exhibit targeted homing, stability, biocompatibility, selective transmissivity, low toxicity, and low immunogenicity, and they can traverse the BBB as a carrier for (immuno) therapeutic or diagnostic drugs.
However, there remain many challenges in the clinical application of exosomes. Their isolation and purification has not been standardized; miRNA quantification remains a limitation; the mechanisms involved in the loading and transport of siRNAs and miRNAs in exosomes remain to be unraveled; the multitarget nature of miRNA and the characteristics of exosomes with immune targets may result in side effects and toxicity
References
1. Lapointe S, Perry A, Butowski NA, Primary brain tumors in adults: Lancet, 2018; 392; 432-46
2. Ji S, Peixin S, Ye Z, The antitumor effects of Newcastle disease virus on glioma: Biocell, 2019; 43(3); 119-28
3. Cheng T, Xu Y, Effects of Enhancer of Zeste Homolog 2 (EZH2) expression on brain glioma cell proliferation and tumorigenesis: Med Sci Monit, 2018; 24; 7249-55
4. Tian J, Casella G, Zhang Y, Potential roles of extracellular vesicles in the pathophysiology, diagnosis, and treatment of autoimmune diseases: Int J Biol Sci, 2020; 16(4); 620-32
5. Sun Y, Li L, Wang L, Liu J, Body fluids-derived exosomes: Paving the novel road to lung cancer diagnosis and therapy: Anticancer Agents Med Chem, 2017; 17(13); 1734-40
6. van der Pol E, Böing AN, Harrison P, Classification, functions, and clinical relevance of extracellular vesicles: Pharmacol Rev, 2012; 64; 676-705
7. Harding C, Heuser J, Stahl P, Endocytosis and intracellular processing of transferrin and colloidal gold-transferrin in rat reticulocytes: Demonstration of a pathway for receptor shedding: Eur J Cell Biol, 1984; 35; 256-63
8. Pan BT, Johnstone RM: Cell, 1983; 33; 967-78
9. Johnstone RM, Adam M, Hammond JR, Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes): J Biol Chem, 1987; 262; 9412-20
10. Pascual M, Ibáñez F, Guerri C, Exosomes as mediators of neuron-glia communication in neuroinflammation: Neural Regen Res, 2020; 15; 796-801
11. Yu X, Odenthal M, Fries JW, Exosomes as miRNA carriers: Formation-function-future: Int J Mol Sci, 2016; 17; E2028
12. Chen R, Xu X, Qian Z, The biological functions and clinical applications of exosomes in lung cancer: Cell Mol Life Sci, 2019; 76; 4613-33
13. Rufino-Ramos D, Albuquerque PR, Carmona V, Extracellular vesicles. Novel promising delivery systems for therapy of brain diseases: J Control Release, 2017; 262; 247-58
14. Nalamolu KR, Venkatesh I, Mohandass A, Exosomes secreted by the cocultures of normal and oxygen-glucose-deprived stem cells improve post-stroke outcome: Neuromolecular Med, 2019; 21; 529-39
15. Wang J, Zheng Y, Zhao M, Exosome-based cancer therapy: Implication for targeting cancer stem cells: Front Pharmacol, 2016; 7; 533
16. Cooper JM, Wiklander PB, Nordin JZ, Systemic exosomal siRNA delivery reduced alpha-synuclein aggregates in brains of transgenic mice: Mov Disord, 2014; 29; 1476-85
17. Raposo G, Stoorvogel W, Extracellular vesicles: Exosomes, microvesicles, and friends: J Cell Biol, 2013; 200; 373-83
18. Bobrie A, Colombo M, Raposo G, Théry C, Exosome secretion: Molecular mechanisms and roles in immune responses: Traffic, 2011; 12; 1659-68
19. Fauré J, Lachenal G, Court M, Exosomes are released by cultured cortical neurones: Mol Cell Neurosci, 2006; 31; 642-48
20. Li Z, Ye L, Wang L, Identification of miRNA signatures in serum exosomes as a potential biomarker after radiotherapy treatment in glioma patients: Ann Diagn Pathol, 2020; 44; 151436
21. Mathivanan S, Ji H, Simpson RJ, Exosomes: Extracellular organelles important in intercellular communication: J Proteomics, 2010; 73; 1907-20
22. Languino LR, Singh A, Prisco M, Exosome-mediated transfer from the tumor microenvironment increases TGFβ signaling in squamous cell carcinoma: Am J Transl Res, 2016; 8; 2432-37
23. Sharma A, Johnson A, Exosome DNA: Critical regulator of tumor immunity and a diagnostic biomarker: J Cell Physiol, 2020; 235; 1921-32
24. Thakur BK, Zhang H, Becker A, Double-stranded DNA in exosomes: A novel biomarker in cancer detection: Cell Res, 2014; 24; 766-69
25. Kalluri R, LeBleu VS, Discovery of double-stranded genomic DNA in circulating exosomes: Cold Spring Harb Symp Quant Biol, 2016; 81; 275-80
26. Al-Nedawi K, Meehan B, Rak J, Microvesicles: Messengers and mediators of tumor progression: Cell Cycle, 2009; 8; 2014-18
27. Mulcahy LA, Pink RC, Carter DR, Routes and mechanisms of extracellular vesicle uptake: J Extracell Vesicles, 2014; 3; 24641
28. Jiang L, Dong H, Cao H, Exosomes in pathogenesis, diagnosis, and treatment of Alzheimer’s disease: Med Sci Monit, 2019; 25; 3329-35
29. Sun Z, Wang L, Zhou Y, Glioblastoma stem cell-derived exosomes enhance stemness and tumorigenicity of glioma cells by transferring Notch1 protein: Cell Mol Neurobiol, 2020; 40; 767-84
30. Yang JK, Song J, Huo HR, DNM3, p65 and p53 from exosomes represent potential clinical diagnosis markers for glioblastoma multiforme: Ther Adv Med Oncol, 2017; 9; 741-54
31. Ibsen SD, Wright J, Lewis JM, Rapid isolation and detection of exosomes and associated biomarkers from plasma: ACS Nano, 2017; 11; 6641-51
32. Cumba Garcia LM, Peterson TE, Cepeda MA, Isolation and analysis of plasma-derived exosomes in patients with glioma: Front Oncol, 2019; 9; 651
33. Kucharzewska P, Christianson HC, Welch JE, Exosomes reflect the hypoxic status of glioma cells and mediate hypoxia-dependent activation of vascular cells during tumor development: Proc Natl Acad Sci USA, 2013; 110; 7312-17
34. Skog J, Würdinger T, van Rijn S, Glioblastoma microvesicles transport RNA and proteins that promote tumour growth and provide diagnostic biomarkers: Nat Cell Biol, 2008; 10; 1470-76
35. Qian M, Wang S, Guo X, Hypoxic glioma-derived exosomes deliver microRNA-1246 to induce M2 macrophage polarization by targeting TERF2IP via the STAT3 and NF-κB pathways: Oncogene, 2020; 39; 428-42
36. Cai Q, Zhu A, Gong L, Exosomes of glioma cells deliver miR-148a to promote proliferation and metastasis of glioblastoma via targeting CADM1: Bull Cancer, 2018; 105; 643-51
37. Lang HL, Hu GW, Chen Y, Glioma cells promote angiogenesis through the release of exosomes containing long non-coding RNA POU3F3: Eur Rev Med Pharmacol Sci, 2017; 21; 959-72
38. Bălaşa A, Şerban G, Chinezu R, The involvement of exosomes in glioblastoma development, diagnosis, prognosis, and treatment: Brain Sci, 2020; 10; 553
39. Figueroa J, Phillips LM, Shahar T, Exosomes from glioma-associated mesenchymal stem cells increase the tumorigenicity of glioma stem-like cells via transfer of miR-1587: Cancer Res, 2017; 77; 5808-19
40. Sun X, Ma X, Wang J, Glioma stem cells-derived exosomes promote the angiogenic ability of endothelial cells through miR-21/VEGF signal: Oncotarget, 2017; 8; 36137-48
41. Lang HL, Hu GW, Zhang B, Glioma cells enhance angiogenesis and inhibit endothelial cell apoptosis through the release of exosomes that contain long non-coding RNA CCAT2: Oncol Rep, 2017; 38; 785-98
42. Yang JK, Yang JP, Tong J, Exosomal miR-221 targets DNM3 to induce tumor progression and temozolomide resistance in glioma: J Neurooncol, 2017; 131; 255-65
43. Hellwinkel JE, Redzic JS, Harland TA, Glioma-derived extracellular vesicles selectively suppress immune responses: Neuro Oncol, 2016; 18; 497-506
44. Liu ZM, Wang YB, Yuan XH, Exosomes from murine-derived GL26 cells promote glioblastoma tumor growth by reducing number and function of CD8+T cells: Asian Pac J Cancer Prev, 2013; 14; 309-14
45. Chen J, Hou C, Wang P, Grade II/III glioma microenvironment mining and its prognostic merit: World Neurosurg, 2019; 132; e76-88
46. Ratajczak J, Wysoczynski M, Hayek F, Membrane-derived microvesicles: Important and underappreciated mediators of cell-to-cell communication: Leukemia, 2006; 20; 1487-95
47. Giusti I, Delle Monache S, Di Francesco M, From glioblastoma to endothelial cells through extracellular vesicles: Messages for angiogenesis: Tumour Biol, 2016; 37; 12743-53
48. Roma-Rodrigues C, Fernandes AR, Baptista PV, Exosome in tumour microenvironment: Overview of the crosstalk between normal and cancer cells: Biomed Res Int, 2014; 2014 179486
49. Sanner T, Formation of transient complexes in the glutamate dehydrogenase catalyzed reaction: Biochemistry, 1975; 14; 5094-98
50. Kore RA, Edmondson JL, Jenkins SV, Hypoxia-derived exosomes induce putative altered pathways in biosynthesis and ion regulatory channels in glioblastoma cells: Biochem Biophys Rep, 2018; 14; 104-13
51. Filipazzi P, Bürdek M, Villa A, Recent advances on the role of tumor exosomes in immunosuppression and disease progression: Semin Cancer Biol, 2012; 22; 342-49
52. Al-Nedawi K, Meehan B, Micallef J, Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells: Nat Cell Biol, 2008; 10; 619-24
53. Monteforte A, Lam B, Sherman MB, Glioblastoma exosomes for therapeutic angiogenesis in peripheral ischemia: Tissue Eng Part A, 2017; 23; 1251-61
54. King HW, Michael MZ, Gleadle JM, Hypoxic enhancement of exosome release by breast cancer cells: BMC Cancer, 2012; 12; 421
55. Li L, Li C, Wang S, Exosomes derived from hypoxic oral squamous cell carcinoma cells deliver miR-21 to normoxic cells to elicit a prometastatic phenotype: Cancer Res, 2016; 76; 1770-80
56. Shaimardanova AA, Solovyeva VV, Chulpanova DS, Extracellular vesicles in the diagnosis and treatment of central nervous system diseases: Neural Regen Res, 2020; 15; 586-96
57. Toustrup K, Sørensen BS, Nordsmark M, Development of a hypoxia gene expression classifier with predictive impact for hypoxic modification of radiotherapy in head and neck cancer: Cancer Res, 2011; 71; 5923-31
58. Guo X, Qiu W, Liu Q, Immunosuppressive effects of hypoxia-induced glioma exosomes through myeloid-derived suppressor cells via the miR-10a/Rora and miR-21/Pten Pathways: Oncogene, 2018; 37; 4239-59
59. Guo X, Qiu W, Wang J, Glioma exosomes mediate the expansion and function of myeloid-derived suppressor cells through microRNA-29a/Hbp1 and microRNA-92a/Prkar1a pathways: Int J Cancer, 2019; 144; 3111-26
60. Basu B, Ghosh MK, Extracellular vesicles in glioma: From diagnosis to therapy: Bioessays, 2019; 41; e1800245
61. Mohanan V, Temburni MK, Kappes JC, Galileo DS, L1CAM stimulates glioma cell motility and proliferation through the fibroblast growth factor receptor: Clin Exp Metastasis, 2013; 30; 507-20
62. Howitt , Low LH, Putz U, Ndfip1 represses cell proliferation by controlling Pten localization and signaling specificity: J Mol Cell Biol, 2015; 7; 119-31
63. Pinet S, Bessette B, Vedrenne N, TrkB-containing exosomes promote the transfer of glioblastoma aggressiveness to YKL-40-inactivated glioblastoma cells: Oncotarget, 2016; 7; 50349-64
64. Arscott WT, Tandle AT, Zhao S, Ionizing radiation and glioblastoma exosomes: Implications in tumor biology and cell migration: Transl Oncol, 2013; 6; 638-48
65. Yue X, Lan F, Xia T, Hypoxic glioma cell-secreted exosomal miR-301a activates Wnt/β-catenin signaling and promotes radiation resistance by targeting TCEAL7: Mol Ther, 2019; 27; 1939-49
66. Corcoran C, Rani S, O’Brien K, Docetaxel-resistance in prostate cancer: Evaluating associated phenotypic changes and potential for resistance transfer via exosomes: PLoS One, 2012; 7; e50999
67. Azmi AS, Bao B, Sarkar FH, Exosomes in cancer development, metastasis, and drug resistance: A comprehensive review: Cancer Metastasis Rev, 2013; 32; 623-42
68. André-Grégoire G, Gavard J, Spitting out the demons: Extracellular vesicles in glioblastoma: Cell Adh Migr, 2017; 11; 164-72
69. Treps L, Perret R, Edmond S, Glioblastoma stem-like cells secrete the pro-angiogenic VEGF-A factor in extracellular vesicles: J Extracell Vesicles, 2017; 6; 1359479
70. Shao H, Chung J, Lee K, Chip-based analysis of exosomal mRNA mediating drug resistance in glioblastoma: Nat Commun, 2015; 6; 6999
71. Agnihotri S, Gajadhar AS, Ternamian C, Alkylpurine-DNA-N-glycosylase confers resistance to temozolomide in xenograft models of glioblastoma multiforme and is associated with poor survival in patients: J Clin Invest, 2012; 122; 253-66
72. Ashley J, Cordy B, Lucia D, Retrovirus-like Gag protein Arc1 binds RNA and traffics across synaptic boutons: Cell, 2018; 172; 262-74
73. Zeng AL, Yan W, Liu YW, Tumour exosomes from cells harbouring PTPRZ1-MET fusion contribute to a malignant phenotype and temozolomide chemoresistance in glioblastoma: Oncogene, 2017; 36; 5369-81
74. Federici C, Petrucci F, Caimi S, Exosome release and low pH belong to a framework of resistance of human melanoma cells to cisplatin: PLoS One, 2014; 9; e88193
75. van der Vos KE, Abels ER, Zhang X, Directly visualized glioblastoma-derived extracellular vesicles transfer RNA to microglia/macrophages in the brain: Neuro Oncol, 2016; 18; 58-69
76. Clayton A, Mitchell JP, Court J, Human tumor-derived exosomes selectively impair lymphocyte responses to interleukin-2: Cancer Res, 2007; 67; 7458-66
77. Graner MW, Alzate O, Dechkovskaia AM, Proteomic and immunologic analyses of brain tumor exosomes: FASEB J, 2009; 23; 1541-57
78. Harshyne LA, Nasca BJ, Kenyon LC, Serum exosomes and cytokines promote a T-helper cell type 2 environment in the peripheral blood of glioblastoma patients: Neuro Oncol, 2016; 18; 206-15
79. Murgoci AN, Cizkova D, Majerova P, Brain-cortex microglia-derived exosomes: Nanoparticles for glioma therapy: Chemphyschem, 2018; 19; 1205-14
80. Bu N, Wu H, Sun B, Exosome-loaded dendritic cells elicit tumor-specific CD8+ cytotoxic T cells in patients with glioma: J Neurooncol, 2011; 104; 659-67
81. de Vrij J, Maas SL, Kwappenberg KM, Glioblastoma-derived extracellular vesicles modify the phenotype of monocytic cells: Int J Cancer, 2015; 137; 1630-42
82. Yu S, Liu C, Su K, Tumor exosomes inhibit differentiation of bone marrow dendritic cells: J Immunol, 2007; 178; 6867-75
83. Gabrusiewicz K, Li X, Wei J, Glioblastoma stem cell-derived exosomes induce M2 macrophages and PD-L1 expression on human monocytes: Oncoimmunology, 2018; 7; e1412909
84. Westphal M, Lamszus K, Circulating biomarkers for gliomas: Nat Rev Neurol, 2015; 11; 556-66
85. Chen WW, Balaj L, Liau LM, BEAMing and droplet digital PCR analysis of mutant IDH1 mRNA in glioma patient serum and cerebrospinal fluid extracellular vesicles: Mol Ther Nucleic Acids, 2013; 2; e109
86. Zare M, Bastami M, Solali S, Alivand MR, Aberrant miRNA promoter methylation and EMT-involving miRNAs in breast cancer metastasis: Diagnosis and therapeutic implications: J Cell Physiol, 2018; 233; 3729-44
87. Wei Z, Batagov AO, Schinelli S, Coding and noncoding landscape of extracellular RNA released by human glioma stem cells: Nat Commun, 2017; 8; 1145
88. Müller S, Janke F, Dietz S, Sültmann H, Circulating MicroRNAs as potential biomarkers for lung cancer: Recent Results Cancer Res, 2020; 215; 299-318
89. Santangelo A, Imbrucè P, Gardenghi B, A microRNA signature from serum exosomes of patients with glioma as complementary diagnostic biomarker: J Neurooncol, 2018; 136; 51-62
90. Manterola L, Guruceaga E, Gállego Pérez-Larraya J, A small noncoding RNA signature found in exosomes of GBM patient serum as a diagnostic tool: Neuro Oncol, 2014; 16; 520-27
91. Lan F, Qing Q, Pan Q, Serum exosomal miR-301a as a potential diagnostic and prognostic biomarker for human glioma: Cell Oncol, 2018; 41; 25-33
92. Huang K, Fang C, Yi K, The role of PTRF/Cavin1 as a biomarker in both glioma and serum exosomes: Theranostics, 2018; 8; 1540-57
93. Akers JC, Ramakrishnan V, Kim R, MiR-21 in the extracellular vesicles (EVs) of cerebrospinal fluid (CSF): a platform for glioblastoma biomarker development: PLoS One, 2013; 8; e78115
94. Shi R, Wang PY, Li XY, Exosomal levels of miRNA-21 from cerebrospinal fluids associated with poor prognosis and tumor recurrence of glioma patients: Oncotarget, 2015; 6; 26971-81
95. Koo S, Martin GS, Schulz KJ, Serial selection for invasiveness increases expression of miR-143/miR-145 in glioblastoma cell lines: BMC Cancer, 2012; 12; 143
96. Li Y, Guessous F, Zhang Y, MicroRNA-34a inhibits glioblastoma growth by targeting multiple oncogenes: Cancer Res, 2009; 69; 7569-76
97. Fraser K, Jo A, Giedt J, Characterization of single microvesicles in plasma from glioblastoma patients: Neuro Oncol, 2019; 21; 606-15
98. Shao H, Chung J, Balaj L, Protein typing of circulating microvesicles allows real-time monitoring of glioblastoma therapy: Nat Med, 2012; 18; 1835-40
99. Setti M, Osti D, Richichi C, Extracellular vesicle-mediated transfer of CLIC1 protein is a novel mechanism for the regulation of glioblastoma growth: Oncotarget, 2015; 6; 31413-27
100. Galbo PM, Ciesielski MJ, Figel S, Circulating CD9+/GFAP+/survivin+ exosomes in malignant glioma patients following survivin vaccination: Oncotarget, 2017; 8; 114722-35
101. García-Romero N, Carrión-Navarro J, Esteban-Rubio S, DNA sequences within glioma-derived extracellular vesicles can cross the intact blood-brain barrier and be detected in peripheral blood of patients: Oncotarget, 2017; 8; 1416-28
102. Kooijmans SA, Vader P, van Dommelen SM, Exosome mimetics: A novel class of drug delivery systems: Int J Nanomedicine, 2012; 7; 1525-41
103. Monfared H, Jahangard Y, Nikkhah M, Potential therapeutic effects of exosomes packed with a miR-21-sponge construct in a rat model of glioblastoma: Front Oncol, 2019; 9; 782
104. Yu L, Gui S, Liu Y, Exosomes derived from microRNA-199a-overexpressing mesenchymal stem cells inhibit glioma progression by down-regulating AGAP2: Aging, 2019; 11; 5300-18
105. Wang B, Wu ZH, Lou PY, Human bone marrow-derived mesenchymal stem cell-secreted exosomes overexpressing microRNA-34a ameliorate glioblastoma development via down-regulating MYCN: Cell Oncol, 2019; 42; 783-99
106. Deng SZ, Lai MF, Li YP, Human marrow stromal cells secrete microRNA-375-containing exosomes to regulate glioma progression: Cancer Gene Therapy, 2019; 27; 203-15
107. Hao SC, Ma H, Niu ZF, hUC-MSCs secreted exosomes inhibit the glioma cell progression through PTENP1/miR-10a-5p/PTEN pathway: Eur Rev Med Pharmacol Sci, 2019; 23; 10013-23
108. Bu N, Wu H, Zhang G, Exosomes from dendritic cells loaded with chaperone-rich cell lysates elicit a potent T cell immune response against intracranial glioma in mice: J Mol Neurosci, 2015; 56; 631-43
109. Katakowski M, Buller B, Zheng X, Exosomes from marrow stromal cells expressing miR-146b inhibit glioma growth: Cancer Lett, 2013; 335; 201-4
110. Munoz JL, Bliss SA, Greco SJ, Delivery of functional anti-miR-9 by mesenchymal stem cell-derived exosomes to glioblastoma multiforme cells conferred chemosensitivity: Mol Ther Nucleic Acids, 2013; 2; e126
111. Zhuang X, Xiang X, Grizzle W, Treatment of brain inflammatory diseases by delivering exosome encapsulated anti-inflammatory drugs from the nasal region to the brain: Mol Ther, 2011; 19; 1769-79
112. Federici C, Shahaj E, Cecchetti S, Natural-killer-derived extracellular vesicles: Immune sensors and interactors: Front Immunol, 2020; 11; 262
113. Zhu Q, Ling X, Yang Y, Embryonic stem cells-derived exosomes endowed with targeting properties as chemotherapeutics delivery vehicles for glioblastoma therapy: Adv Sci, 2019; 6; 1801899
114. Jia G, Han Y, An Y: Biomaterials, 2018; 178; 302-16
115. Lin Q, Mao KL, Tian FR, Brain tumor-targeted delivery and therapy by focused ultrasound introduced doxorubicin-loaded cationic liposomes: Cancer Chemother Pharmacol, 2016; 77; 269-80
116. Bai L, Liu Y, Guo K, Ultrasound facilitates naturally equipped exosomes derived from macrophages and blood serum for orthotopic glioma treatment: ACS Appl Mater Interfaces, 2019; 11; 14576-87
117. Ha D, Yang N, Nadithe V, Exosomes as therapeutic drug carriers and delivery vehicles across biological membranes: Current perspectives and future challenges: Acta Pharm Sin B, 2016; 6; 287-96
118. Salarpour S, Forootanfar H, Pournamdari M, Paclitaxel incorporated exosomes derived from glioblastoma cells: comparative study of two loading techniques: Daru, 2019; 27; 533-39
119. Sun D, Zhuang X, Xiang X, A novel nanoparticle drug delivery system: The anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes: Mol Ther, 2010; 18; 1606-14
120. Tian Y, Li S, Song J, A doxorubicin delivery platform using engineered natural membrane vesicle exosomes for targeted tumor therapy: Biomaterials, 2014; 35; 2383-90
121. Haney MJ, Klyachko NL, Zhao Y, Exosomes as drug delivery vehicles for Parkinson’s disease therapy: J Control Release, 2015; 207; 18-30
122. Sáenz-Cuesta M, Osorio-Querejeta I, Otaegui D, Extracellular vesicles in multiple sclerosis: What are they telling us?: Front Cell Neurosci, 2014; 8; 100
123. Grapp M, Wrede A, Schweizer M, Choroid plexus transcytosis and exosome shuttling deliver folate into brain parenchyma: Nat Commun, 2013; 4; 2123
124. Yang T, Martin P, Fogarty B: Pharm Res, 2015; 32; 2003-14
125. Jiang XC, Gao JQ, Exosomes as novel bio-carriers for gene and drug delivery: Int J Pharm, 2017; 521; 167-75
126. Kim MS, Haney MJ, Zhao Y, Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells: Nanomedicine, 2016; 12; 655-64
127. Gehl J, Electroporation: theory and methods, perspectives for drug delivery, gene therapy and research: Acta Physiol Scand, 2003; 177; 437-47
128. Alvarez-Erviti L, Seow Y, Yin H, Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes: Nat Biotechnol, 2011; 29; 341-45
129. Haqqani AS, Delaney CE, Tremblay TL, Method for isolation and molecular characterization of extracellular microvesicles released from brain endothelial cells: Fluids Barriers CNS, 2013; 10; 4
130. Treps L, Edmond S, Harford-Wright E, Extracellular vesicle-transported Semaphorin 3A promotes vascular permeability in glioblastoma: Oncogene, 2016; 35; 2615-23
131. Wang H, Wang F, Wang X, Friend or foe: A cancer suppressor microRNA-34 potentially plays an adverse role in vascular diseases by regulating cell apoptosis and extracellular matrix degradation: Med Sci Monit, 2019; 25; 1952-59
132. Kim R, Lee S, Lee J, Exosomes derived from microRNA-584 transfected mesenchymal stem cells: Novel alternative therapeutic vehicles for cancer therapy: BMB Rep, 2018; 51; 406-11
133. Lang FM, Hossain A, Gumin J, Mesenchymal stem cells as natural biofactories for exosomes carrying miR-124a in the treatment of gliomas: Neuro Oncol, 2018; 20; 380-90
134. Ma Z, Cui X, Lu L, Exosomes from glioma cells induce a tumor-like phenotype in mesenchymal stem cells by activating glycolysis: Stem Cell Res Ther, 2019; 10; 60
135. Bak RO, Hollensen AK, Mikkelsen JG, Managing microRNAs with vector-encoded decoy-type inhibitors: Mol Ther, 2013; 21; 1478-85
136. Dai X, Liao K, Zhuang Z, AHIF promotes glioblastoma progression and radioresistance via exosomes: Int J Oncol, 2019; 54; 261-70
137. Sugahara KN, Teesalu T, Karmali PP, Coadministration of a tumor-penetrating peptide enhances the efficacy of cancer drugs: Science, 2010; 328; 1031-35
138. Li Z, Mbah NE, Maltese WA, Vacuole-inducing compounds that disrupt endolysosomal trafficking stimulate production of exosomes by glioblastoma cells: Mol Cell Biochem, 2018; 439; 1-9
139. Parajuli P, Mathupala S, Mittal S, Sloan AE, Dendritic cell-based active specific immunotherapy for malignant glioma: Expert Opin Biol Ther, 2007; 7; 439-48
140. Besse B, Charrier M, Lapierre V, Dendritic cell-derived exosomes as maintenance immunotherapy after first line chemotherapy in NSCLC: Oncoimmunology, 2016; 5; e1071008
141. Escudier B, Dorval T, Chaput N, Vaccination of metastatic melanoma patients with autologous dendritic cell (DC) derived-exosomes: results of the first phase I clinical trial: J Transl Med, 2005; 3; 10
142. Jie X, Hua L, Jiang W, Clinical application of a dendritic cell vaccine raised against heat-shocked glioblastoma: Cell Biochem Biophys, 2012; 62; 91-99
143. Ji B, Chen Q, Liu B, Glioma stem cell-targeted dendritic cells as a tumor vaccine against malignant glioma: Yonsei Med J, 2013; 54; 92-100
144. Mitchell DA, Batich KA, Gunn MD, Tetanus toxoid and CCL3 improve dendritic cell vaccines in mice and glioblastoma patients: Nature, 2015; 519; 366-69
145. Antonios JP, Soto H, Everson RG, PD-1 blockade enhances the vaccination-induced immune response in glioma: JCI Insight, 2016; 1; e87059
146. Liu H, Chen L, Liu J, Co-delivery of tumor-derived exosomes with alpha-galactosylceramide on dendritic cell-based immunotherapy for glioblastoma: Cancer Lett, 2017; 411; 182-90
147. Sayegh ET, Oh T, Fakurnejad S, Vaccine therapies for patients with glioblastoma: J Neurooncol, 2014; 119; 531-46
148. Muller L, Muller-Haegele S, Mitsuhashi M, Exosomes isolated from plasma of glioma patients enrolled in a vaccination trial reflect antitumor immune activity and might predict survival: Oncoimmunology, 2015; 4; e1008347
149. Li D, Huang S, Zhu J: Med Sci Monit, 2019; 25; 1871-85
In Press
05 Mar 2024 : Clinical Research
Muscular Function Recovery from General Anesthesia in 132 Patients Undergoing Surgery with Acceleromyograph...Med Sci Monit In Press; DOI: 10.12659/MSM.942780
05 Mar 2024 : Clinical Research
Effects of Thermal Insulation on Recovery and Comfort of Patients Undergoing Holmium Laser LithotripsyMed Sci Monit In Press; DOI: 10.12659/MSM.942836
05 Mar 2024 : Clinical Research
Role of Critical Shoulder Angle in Degenerative Type Rotator Cuff Tears: A Turkish Cohort StudyMed Sci Monit In Press; DOI: 10.12659/MSM.943703
06 Mar 2024 : Clinical Research
Comparison of Outcomes between Single-Level and Double-Level Corpectomy in Thoracolumbar Reconstruction: A ...Med Sci Monit In Press; DOI: 10.12659/MSM.943797
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952