17 April 2022: Meta-Analysis
Pharmacological Therapies for Osteoporosis: A Bayesian Network Meta-Analysis
Jiping Shen1ACDE, Zheng Ke2D, Shuangshuang Dong2DF, Minzhi Lv3CD, Ying Yuan1BF, Le Song1BF, Kefen WuDOI: 10.12659/MSM.935491
Med Sci Monit 2022; 28:e935491
Abstract
BACKGROUND: Numerous randomized controlled trials (RCTs) have evaluated pharmacological therapies for osteoporosis. The aim of this Bayesian network meta-analysis was to compare the efficacy and safety of pharmacological therapies for osteoporosis patients.
MATERIAL AND METHODS: The electronic databases of PubMed, Embase, and Cochrane Library were systematically searched for eligible RCTs from their inception up to January 2021. The primary endpoints were all fractures, vertebral fractures, and non-vertebral fractures, while the secondary endpoints were fractures at hip or peripheral locations, bone mineral density (BMD) at various sites, and potential adverse events.
RESULTS: We included 79 RCTs reporting a total of 108 797 individuals in the final quantitative analysis. The results of network analysis indicated that romosozumab (92.1%) was the most effective in reducing the risk for all fractures, with the best therapeutic effects on vertebral fracture (97.2%) and non-vertebral fracture (88.0%). Romosozumab (92.5%) provided better therapeutic effects for the reduction of hip fracture. The best treatment agents for improving whole-body BMD (100.0%), spine BMD (95.7%), hip BMD (92.4%), femoral neck BMD (86.7%), and trochanter BMD (95.5%) were alendronate, strontium ranelate, ibandronate, risedronate, and ibandronate, respectively. Finally, the use of bazedoxifene was associated with the highest incidence of any upper-gastrointestinal event, nasopharyngitis, and back pain, while risedronate was associated with higher incidence of abdominal pain and dyspepsia.
CONCLUSIONS: This study found that romosozumab yielded the best effects for preventing fracture risk, while abaloparatide was the most effective in reducing the risk of vertebral fracture and non-vertebral fracture.
Keywords: Bone Density, Fractures, Bone, Network Meta-Analysis, Pharmacological and Toxicological Phenomena, Bone Density Conservation Agents, Female, Hip Fractures, Humans, Ibandronic Acid, Osteoporosis, Osteoporosis, Postmenopausal, Risedronic Acid, Spinal Fractures
Background
Osteoporosis is a chronic metabolic bone disease that is highly prevalent in the elderly population, especially in postmenopausal women [1]. Osteoporosis morbidity is rapidly increasing with the increase in the aging population, with an estimated 200 million people affected worldwide [2]. Changes in bone mineral density (BMD) in postmenopausal women are significantly associated with hormonal changes related to ovarian function after menopause [3]. Moreover, nearly half of women and one-third of men with osteoporosis present with bone fractures during their lives [4]. The crude estimated prevalence of osteoporotic fractures is 9 million worldwide, which has a direct impact on morbidity, mortality, quality of life, and treatment cost [5]. Although numerous pharmacological therapies remain to be developed for reducing the risk of fractures, fractures mainly occur in individuals with osteoporosis who are receiving treatment but show inadequate responses to therapy.
Currently, bisphosphonates are widely used to prevent and treat osteoporosis; however, the long-term use of bisphosphonates can induce bone micro-damage accumulation, excessive acceleration of mineralization, and atypical insufficiency fractures in the skeletal system [6]. Several systematic reviews have been conducted to compare various drugs for treating osteoporosis [7–9]. Murad et al included 116 randomized controlled trials (RCTs) and found that teriparatide, bisphosphonates, and denosumab are the most effective agents for preventing fragility fracture risk, but the differences in effectiveness among the investigated drugs were not significant [7]. Freemantle et al suggested that osteoporotic patients who received denosumab, risedronate, and zoledronate have a significantly lower risk of non-vertebral and hip fractures, while alendronate, strontium ranelate, and teriparatide yield beneficial effects for non-vertebral fractures. Moreover, mixed comparison results indicated that denosumab was associated with a greater reduction in new vertebral fractures than strontium ranelate, raloxifene, alendronate, and risedronate [8]. Barrionuevo et al enrolled 107 RCTs and comprehensively examined the effectiveness of various agents on the risk of fragility fractures in postmenopausal women [9].
Recently, the effectiveness of various pharmacological therapies to reduce the risk of fractures and improve BMD at various sites in osteoporotic patients has been studied in numerous RCTs, and the therapeutic effects of these drugs should be re-evaluated and updated. Moreover, prior meta-analyses did not address safety outcomes [7–9]. Therefore, our study systematically analyzed the current existing treatment options for osteoporosis, and a Bayesian network meta-analysis was performed to summarize the evidence through direct and indirect comparisons of different pharmacological therapies.
Material and Methods
SEARCH STRATEGY AND SELECTION CRITERIA:
We identified the studies published in English through a systematic search of PubMed, Embase, and the Cochrane Library from inception to January 2021, using the following search terms: “osteoporosis,” “therapeutic,” and “random*.” The details of the search strategy in PubMed are presented in the
DATA COLLECTION AND QUALITY ASSESSMENT:
The data collected included the first author’s surname, publication year, country, sample size, mean age, sex, interventions and controls, co-calcium, co-vitamin D, follow-up duration, and investigated outcomes. The Jadad scale was used to evaluate methodological quality, as it is quite comprehensive and has been validated for assessing the quality of RCTs in meta-analyses [12]. Data extraction and quality assessment were conducted independently by 2 authors. Information was examined and adjudicated independently by another author referring to the original studies.
STATISTICAL ANALYSES:
We initially performed a pairwise meta-analysis using a random-effects model because it is likely the most appropriate and conservative methodology to account for between-trial heterogeneity within each comparison [13]. We estimated the relative treatment effects of the competing interventions using odds ratios for dichotomous outcomes and standardized mean differences for continuous outcomes. For indirect and mixed comparisons, we used Bayesian network meta-analysis to compare different drugs [14]. To check for the presence of inconsistency, we used the loop-specific approach to assess the difference between direct and indirect estimates for a specific comparison in the loop [15]. To check the assumption of consistency in the entire network, we used the design-by-treatment interaction inconsistency model [14]. Because of the heterogeneity in patients, we still used the inconsistent model to analyze data. To rank the treatments for each outcome, we used surface under the cumulative ranking (SUCRA) probabilities [16]. Comparison-adjusted funnel plots were used to determine whether small-study effects were present in our analysis [17]. The summary results for adverse events for each drug were calculated using the random-effects model [13,18], and heterogeneity was assessed using I2 and Q statistics [19,20]. All tests were two-tailed, and a P value of <0.05 was considered statistically significant. Data analyses were performed using Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Results
LITERATURE SEARCH:
The results of the study selection process are shown in Figure 1. The initial electronic searches provided 5434 articles after removal of duplicates. After reviewing the titles and abstracts, 5077 of these articles were excluded owing to irrelevant topics. The remaining 357 articles were retrieved for full-text evaluations, and 278 were excluded for the following reasons: other drugs (n=85), affiliate studies (n=81), review (n=75), and insufficient data (n=37). A manual search of the reference lists of these studies did not yield any new eligible studies. Finally, 79 RCTs that assessed a total of 108 797 patients were included in our systematic review. The baseline characteristics of these studies are summarized in the Supplementary Material.
STUDY CHARACTERISTICS:
These studies were published from 1984 to 2020, with 39 to 9331 patients included in each trial, and the follow-up period was 0.5–5.0 years. Eleven RCTs included men only, 56 RCTs included women only, and the remaining 12 RCTs included both males and females. Sixty-nine trials reported patients using calcium supplementation, and 57 trials reported patients using vitamin D supplementation in intervention and control groups. Study quality was evaluated using the Jadad scale, in which 36 trials scored 4, 30 trials scored 3, 9 trials scored 2, and the remaining 4 trials scored 1.
PRIMARY ENDPOINTS:
In the network meta-analysis, eligible comparisons of outcomes are presented in the network plot (Figure 2). In the figure, the nodes are weighted according to the number of studies that evaluated each treatment, and the edges were weighted according to the precision of the direct estimate for each pairwise comparison. An inconsistency plot was produced to assume the loop-specific heterogeneity estimate, exp (IF), which showed no significant differences among the studies. We ranked the comparative effects of the drugs with SUCRA probabilities (%). The results indicated that romosozumab (92.1%) and zoledronate (90.6%) were more effective in preventing all fracture risks (Figure 3). The details of the pairwise comparisons agents are presented in Figure 4 and the Supplementary Material. A comparison-adjusted funnel plot used to assess publication bias and determine the presence of small-study effects did not suggest that there was any publication bias (Figure 5).
The network meta-analysis comparing the effectiveness of various drugs to reduce the risk of vertebral and non-vertebral fracture is presented in Figure 6. The loop-specific heterogeneity inconsistency plot showed no significant differences. The SUCRA rank showed that abaloparatide (97.2%), denosumab (85.5%), and romosozumab (91.2%) were more effective for reducing the risk of vertebral fractures (Figure 7A), while abaloparatide (88.0%), and zoledronate (85.0%) were most likely to prevent the risk of non-vertebral fractures (Figure 7B). The details of the pairwise comparisons agents for the risk of vertebral and non-vertebral fractures are shown in Figure 8 and Supplementary Material. There was no significant publication bias for the risk of vertebral and non-vertebral fracture (Figure 9).
SECONDARY ENDPOINTS:
The results of the network meta-analysis comparing the effectiveness of various drugs to reduce the risk of hip fracture and peripheral fracture are shown in the
The network meta-analysis showing the effects of various drugs on BMD at various sites are shown in the
The safety profiles for each drug were also pooled and listed in the
Discussion
In this study, we performed a network meta-analysis to compare the effectiveness of various pharmacological therapies for osteoporotic patients. This large quantitative study included 108 797 individuals from 79 RCTs with a broad range of baseline characteristics. This meta-analysis indicated that romosozumab and zoledronate yielded better pharmacological outcomes for all fractures. Abaloparatide, denosumab, and romosozumab were found to be effective for preventing vertebral fractures. Abaloparatide and zoledronate were the most effective for preventing non-vertebral fractures. Romosozumab was the most effective for preventing hip fracture, while alendronate (61.0%), calcitonin (64.9%), and zoledronate provided similar reduction of the risk of peripheral fracture. Furthermore, the best therapeutic effects for improving whole-body BMD, spine BMD, hip BMD, femoral neck BMD, and trochanter BMD were alendronate, strontium ranelate, ibandronate, risedronate, and ibandronate, respectively. Finally, several adverse events should be addressed in clinical practice, including bazedoxifene-related to upper-gastrointestinal events, nasopharyngitis, headache, and back pain, denosumab related to musculoskeletal pain, and calcitonin related to nausea.
Several systematic reviews and meta-analyses have previously compared various pharmacological therapies for osteoporosis [7–9]. Moreover, a recent network meta-analysis conducted by Kataoka et al to compare the recommendation pharmacological therapies for postmenopausal osteoporosis and found no apparent discrepancy between guideline recommendations and drug prescribing rankings [21]. Deng et al identified 56 RCTs and found that bisphosphonates, teriparatide, and denosumab were associated with a reduced risk of fracture in patients undergoing glucocorticoids, while the anti-fracture efficacy of vitamin D metabolites and analogs were superior to plain vitamin D [22]. A network meta-analysis performed by Liu et al found teriparatide and ibandronate had the best effect in reducing vertebral and non-vertebral fractures in patients with glucocorticoid-induced osteoporosis [23]. Lin et al identified 94 RCTs and indicated that parathyroid hormone had the best effect in reducing hip fractures, while strontium ranelate, fluoride, and hormone replacement therapy provided the best efficacy in increasing BMD at total hip, lumbar spine, and distal radius [24]. Migliorini et al identified 64 RCTs and found denosumab had the best effect in increasing BMD at spine, hip, and femur in selected women with postmenopausal osteoporosis [25]. However, these studies focused on fractures at vertebral, non-vertebral, or hip, and BMD at various sites, while several other important outcomes, including all fractures, peripheral fractures, and safety profiles, were not addressed. Moreover, the analyses in prior meta-analyses contained several abandoned drugs, and the results might be overestimates based on network analysis. Furthermore, recently published RCTs should be entered into meta-analysis, and the pooled conclusions require re-evaluation. Therefore, we performed a Bayesian network meta-analysis to provide evidence regarding better pharmacological therapies for osteoporosis treatment.
The study results indicated that romosozumab and zoledronate provided enhanced effectiveness for preventing all fractures. Several factors might explain these results: romosozumab provides a dual effect on bones owing to the changes in bone formation and bone resorption through binding and inhibiting sclerostin [26–30]; and the beneficial effects of zoledronate could be explained by the high potency of bisphosphonates and high compliance rate of patients [31]. Additionally, the relatively better agents for preventing vertebral and non-vertebral fractures were abaloparatide, denosumab, or romosozumab; and abaloparatide and zoledronate, respectively. Abaloparatide selectively binds to the RG versus R0 conformation of the parathyroid hormone type 1 receptor [32–37], subsequently affecting BMD, restoration of bone microarchitecture, and increased bone strength [38–40].
Our study also suggests the best agents for improving whole-body BMD, spine BMD, hip BMD, femoral neck BMD, and trochanter BMD were alendronate, strontium ranelate, ibandronate, risedronate, and ibandronate, respectively, which had already been illustrated in numerous studies [41–49]. Although our study found several other agents that more effectively improved BMD at various sites, these results might not be stable due to the smaller number of trials reporting these data. Further large-scale RCTs are required to verify these data. Finally, although numerous traditional systematic reviews and meta-analyses have already illustrated the effectiveness of pharmacological treatment for osteoporotic patients, direct or indirect comparisons among various agents remain inconclusive. The present comprehensive network meta-analysis studied various medicinal treatments for osteoporotic patients to further clarify the effectiveness of pharmacological therapies for osteoporosis. In addition, our study calculated the pooled incidences for adverse events related to each drug. However, Bayesian network meta-analysis was not conducted for specific adverse events because they were relatively uncommon.
Several limitations in our study should be acknowledged: (1) the use of vitamin D and calcium supplements by patients may have introduced heterogeneity across included trials; (2) the inconsistent results regarding the Bayesian network meta-analyses and pairwise comparisons should be verified by further direct comparison RCT; (3) nearly half of included studies had low to moderate quality, and the outcomes of this study should be interpreted cautiously; (4) although no significant publication bias was observed, the potential publication bias was inevitable owing to the analysis of our study based on published articles; and (5) there are inherent limitations for meta-analyses based on study level, and more detailed analyses are needed.
Conclusions
This network meta-analysis presents the optimal pharmacological therapies for preventing fracture, improving BMD at various sites, and potential adverse events in osteoporosis patients. These findings could be recommended in clinical practice for individuals at high risk of fractures. Further comprehensive network meta-analyses should be conducted to compare the cost-effectiveness of pharmacological therapies for osteoporosis.
Supplementary Material
4. THE THERAPEUTIC EFFECTS OF PAIRWISE COMPARISONS AGENTS ON ALL FRACTURES:
Treatment/comparator Alendronate Alfacalcidol Calcitonin Calcitriol Denosumab Eldecalcitol Ibandronate Lasofoxifene Placebo Raloxifene Risedronate Romosozumab Strontiumranelate Zoledronate – 1.58 (0.81, 3.06) 1.11 (0.82, 1.51) 19.27 (2.12, 174.74) 1.21 (0.66, 2.22) 1.51 (0.60, 3.82) 1.05 (0.81, 1.36) 2.62 (0.22, 31.01) 1.31 (1.14, 1.51) 0.95 (0.77, 1.17) 1.03 (0.51, 2.08) 0.75 (0.62, 0.91) 1.12 (0.88, 1.43) 0.77 (0.62, 0.95) 0.63 (0.33, 1.23) – 0.71 (0.34, 1.47) 12.23 (1.22, 122.37) 0.77 (0.31, 1.89) 0.96 (0.50, 1.83) 0.67 (0.33, 1.37) 1.66 (0.13, 21.49) 0.83 (0.42, 1.64) 0.60 (0.30, 1.21) 0.66 (0.25, 1.72) 0.48 (0.24, 0.95) 0.71 (0.35, 1.44) 0.49 (0.24, 0.98) 0.90 (0.66, 1.22) 1.42 (0.68, 2.94) – 17.32 (1.88, 159.26) 1.09 (0.56, 2.10) 1.36 (0.51, 3.60) 0.95 (0.66, 1.36) 2.35 (0.20, 28.22) 1.18 (0.88, 1.57) 0.85 (0.61, 1.19) 0.93 (0.44, 1.96) 0.67 (0.47, 0.96) 1.00 (0.71, 1.42) 0.69 (0.50, 0.96) 0.05 (0.01, 0.47) 0.08 (0.01, 0.82) 0.06 (0.01, 0.53) – 0.06 (0.01, 0.61) 0.08 (0.01, 0.86) 0.05 (0.01, 0.50) 0.14 (0.00, 3.71) 0.07 (0.01, 0.61) 0.05 (0.01, 0.45) 0.05 (0.01, 0.54) 0.04 (0.00, 0.35) 0.06 (0.01, 0.53) 0.04 (0.00, 0.36) 0.83 (0.45, 1.52) 1.30 (0.53, 3.22) 0.92 (0.48, 1.78) 15.95 (1.63, 155.82) – 1.25 (0.41, 3.80) 0.87 (0.48, 1.57) 2.17 (0.17, 27.44) 1.08 (0.60, 1.96) 0.78 (0.42, 1.45) 0.85 (0.50, 1.46) 0.62 (0.33, 1.16) 0.93 (0.49, 1.73) 0.64 (0.49, 1.73) 0.66 (0.26, 1.68) 1.04 (0.55, 1.99) 0.74 (0.28, 1.96) 12.76 (1.17, 139.69) 0.80 (0.26, 2.43) – 0.70 (0.27, 1.83) 1.73 (0.12, 24.32) 0.87 (0.34, 2.22) 0.63 (0.24, 1.63) 0.68 (0.21, 2.19) 0.50 (0.19, 1.28) 0.74 (0.28, 1.94) 0.51 (0.20, 1.32) 0.95 (0.73, 1.23) 1.50 (0.73, 3.05) 1.06 (0.73, 1.52) 18.29 (2.00, 166.93) 1.15 (0.64, 2.06) 1.43 (0.55, 3.77) – 2.48 (0.21, 29.60) 1.24 (1.00, 1.55) 0.90 (0.69, 1.18) 0.98 (0.49, 1.96) 0.71 (0.53, 0.96) 1.06 (0.79, 1.43) 0.73 (0.55, 0.96) 0.38 (0.03, 4.53) 0.60 (0.05, 7.79) 0.43 (0.04, 5.10) 7.36 (0.27, 200.97) 0.46 (0.04, 5.85) 0.58 (0.04, 8.10) 0.40 (0.03, 4.80) – 0.50 (0.04, 5.90) 0.36 (0.03, 4.29) 0.39 (0.03, 5.12) 0.29 (0.02, 3.41) 0.43 (0.04, 5.08) 0.29 (0.02, 3.49) 0.76 (0.66, 0.88) 1.20 (0.61, 2.38) 0.85 (0.64, 1.13) 14.73 (1.63, 132.95) 0.92 (0.51, 1.67) 1.15 (0.45, 2.96) 0.81 (0.65, 1.00) 2.00 (0.17, 23.60) – 0.72 (0.62, 0.85) 0.79 (0.40, 1.57) 0.57 (0.47, 0.70) 0.85 (0.70, 1.04) 0.59 (0.50, 0.69) 1.06 (0.86, 1.30) 1.66 (0.83, 3.35) 1.18 (0.84, 1.64) 20.35 (2.24, 184.77) 1.28 (0.69, 2.36) 1.59 (0.61, 4.15) 1.11 (0.85, 1.46) 2.76 (0.23, 32.79) 1.38 (1.18, 1.62) – 1.09 (0.54, 2.21) 0.79 (0.61, 1.02) 1.18 (0.92, 1.52) 0.81 (0.65, 1.02) 0.97 (0.48, 1.95) 1.53 (0.58, 4.02) 1.08 (0.51, 2.27) 18.66 (1.86, 187.15) 1.17 (0.69, 1.99) 1.46 (0.46, 4.69) 1.02 (0.51, 2.04) 2.53 (0.20, 32.87) 1.27 (0.64, 2.52) 0.92 (0.45, 1.86) – 0.73 (0.35, 1.49) 1.08 (0.53, 2.22) 0.74 (0.37, 1.51) 1.33 (1.10, 1.62) 2.10 (1.05, 4.21) 1.48 (1.04, 2.11) 25.72 (2.82, 234.43) 1.61 (0.86, 3.02) 2.02 (0.78, 5.21) 1.41 (1.04, 1.90) 3.49 (0.29, 41.58) 1.75 (1.42, 2.15) 1.26 (0.98, 1.64) 1.38 (0.67, 2.83) – 1.49 (1.12, 1.99) 1.03 (0.79, 1.34) 0.89 (0.70, 1.14) 1.41 (0.69, 2.87) 1.00 (0.70, 1.41) 17.24 (1.89, 157.05) 1.08 (0.58, 2.02) 1.35 (0.52, 3.54) 0.94 (0.70, 1.27) 2.34 (0.20, 27.86) 1.17 (0.96, 1.43) 0.85 (0.66, 1.09) 0.92 (0.45, 1.89) 0.67 (0.50, 0.89) – 0.69 (0.53, 0.89) 1.30 (0.53, 0.89) 2.05 (1.02, 4.13) 1.45 (1.04, 2.01) 25.06 (2.76, 227.63) 1.57 (0.85, 2.90) 1.96 (0.76, 5.10) 1.37 (1.04, 1.80) 3.40 (0.29, 40.39) 1.70 (1.45, 2.00) 1.23 (0.98, 1.55) 1.34 (0.66, 2.72) 0.97 (0.75, 1.27) 1.45 (1.12, 1.88) –
Figures
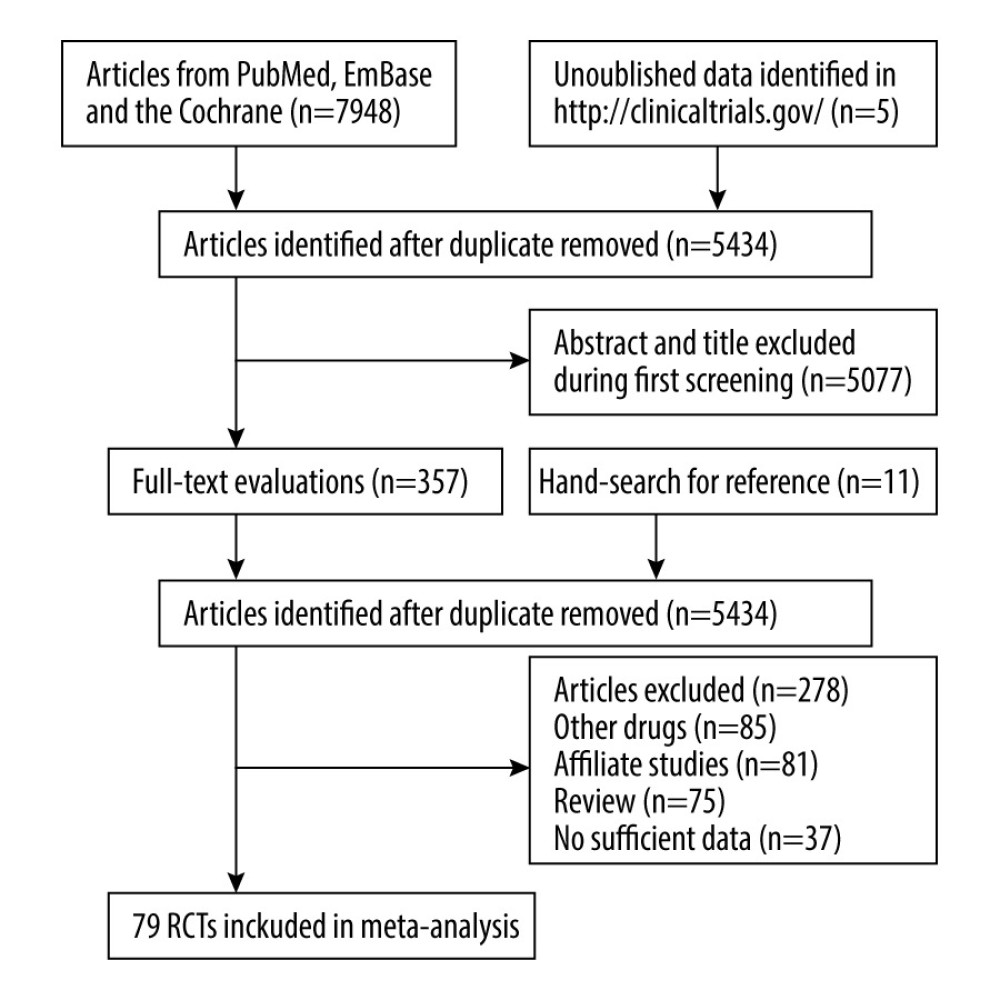 Figure 1. PRISMA flowchart for the literature search and study selection.
Figure 1. PRISMA flowchart for the literature search and study selection. 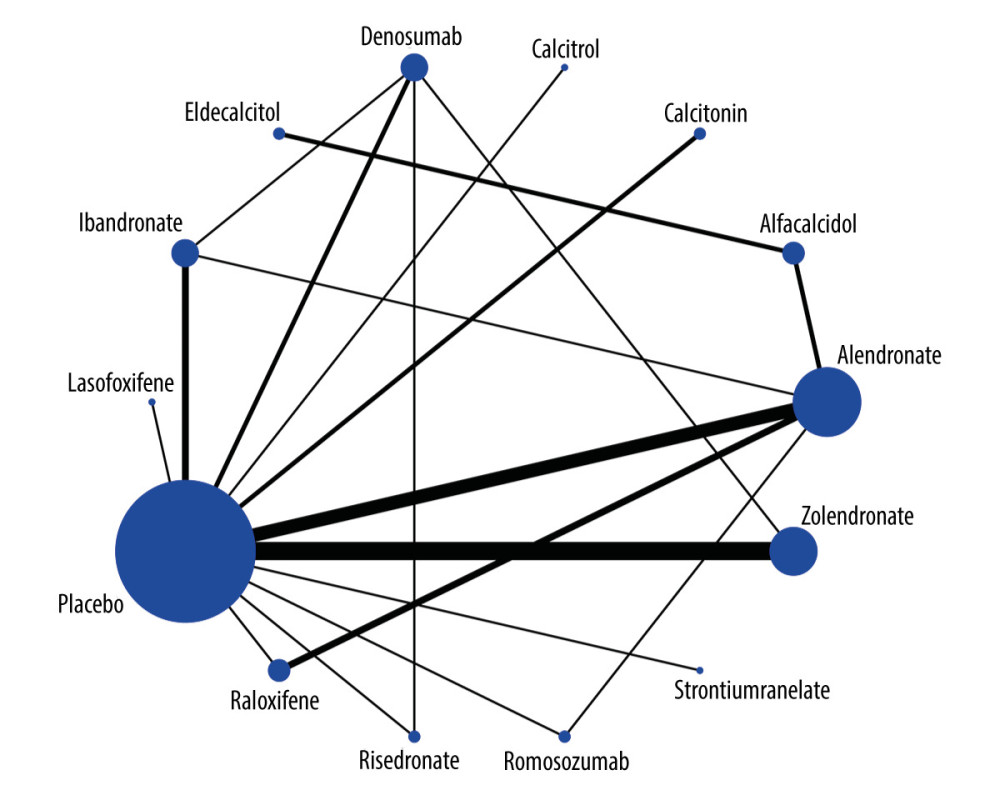 Figure 2. Network of comparisons for all fracture included in the analysis, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 2. Network of comparisons for all fracture included in the analysis, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). 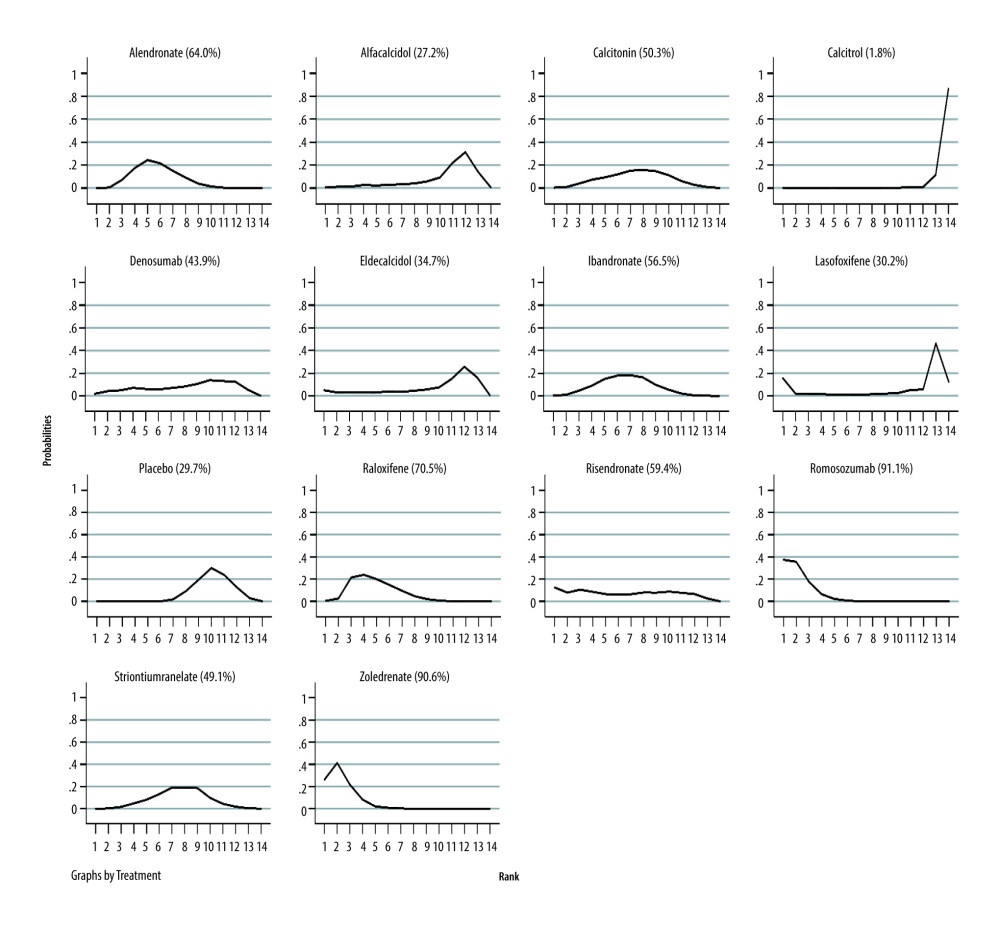 Figure 3. The SUCRA rank test for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 3. The SUCRA rank test for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). 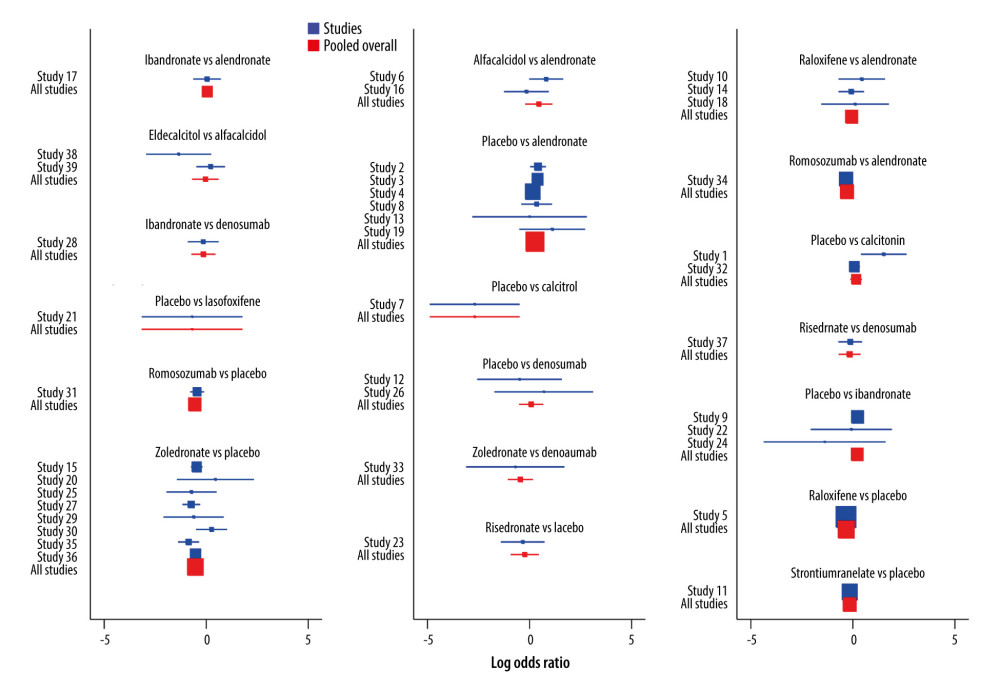 Figure 4. The pairwise comparisons agents for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 4. The pairwise comparisons agents for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). 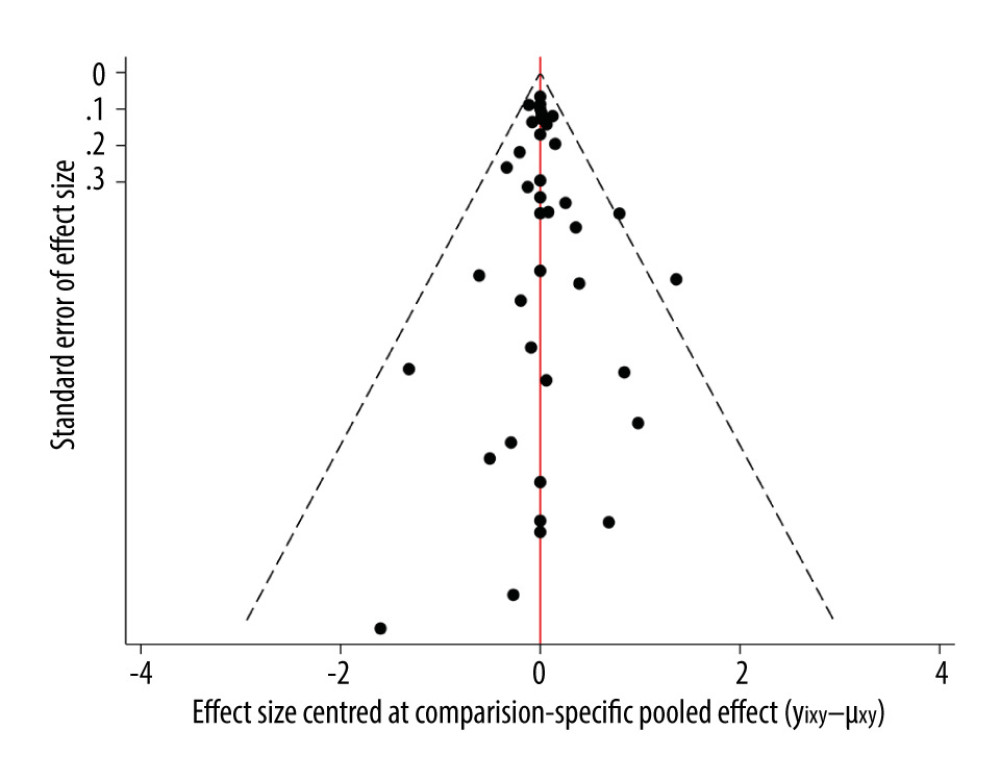 Figure 5. Funnel plot for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 5. Funnel plot for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). 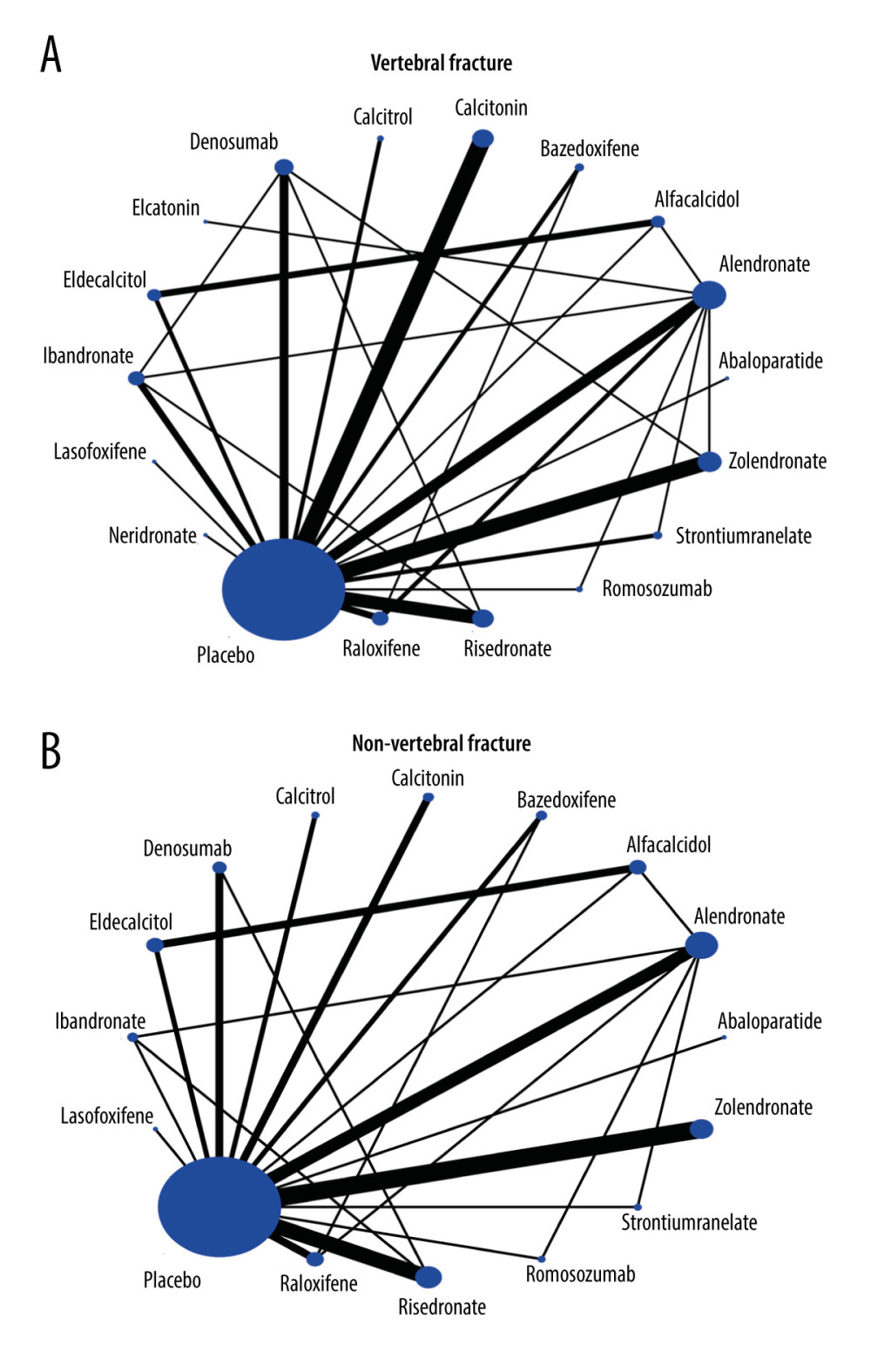 Figure 6. Network of comparisons for vertebral fracture (A) and non-vertebral fracture (B) included in the analysis, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 6. Network of comparisons for vertebral fracture (A) and non-vertebral fracture (B) included in the analysis, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). 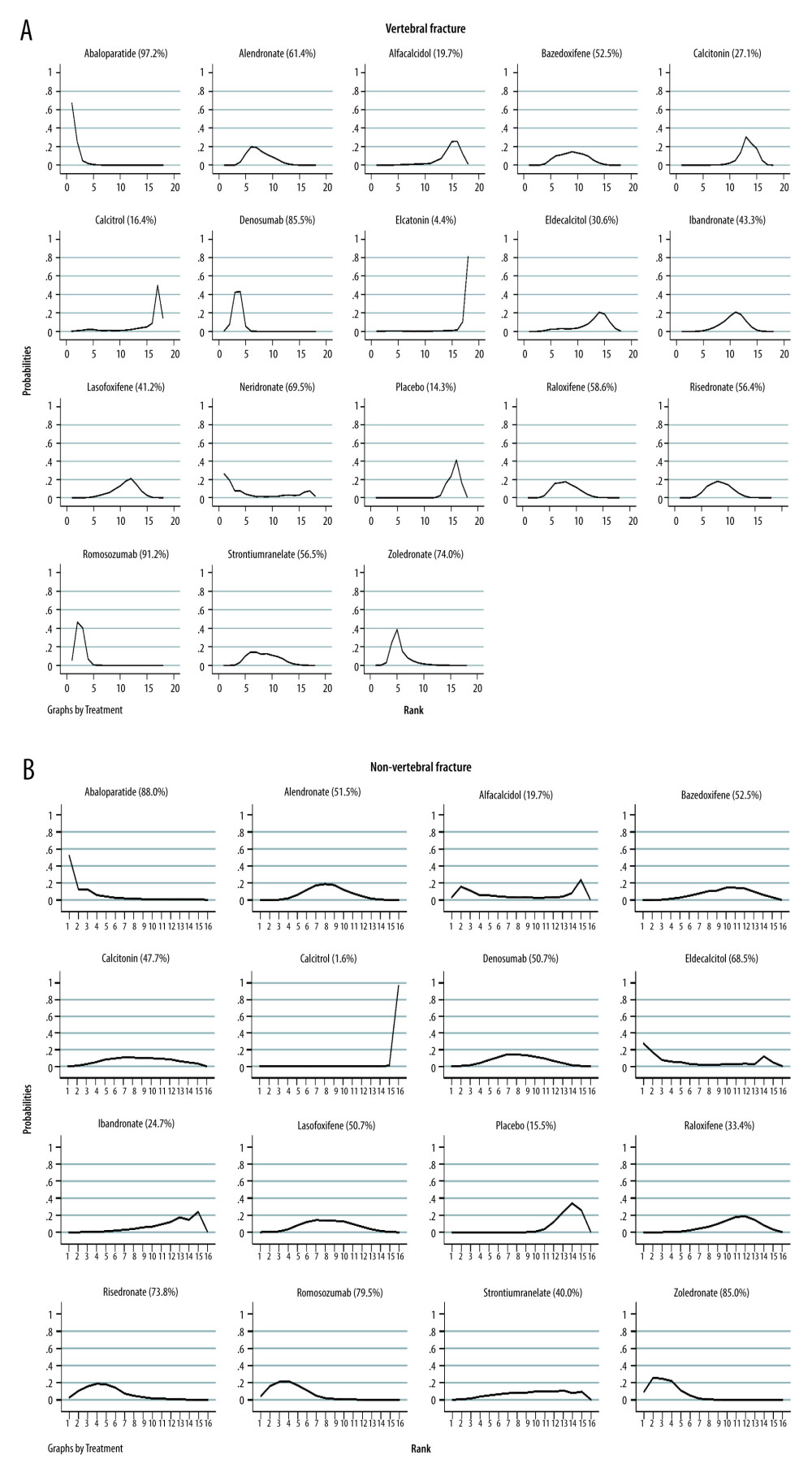 Figure 7. The SUCRA rank test for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 7. The SUCRA rank test for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA). 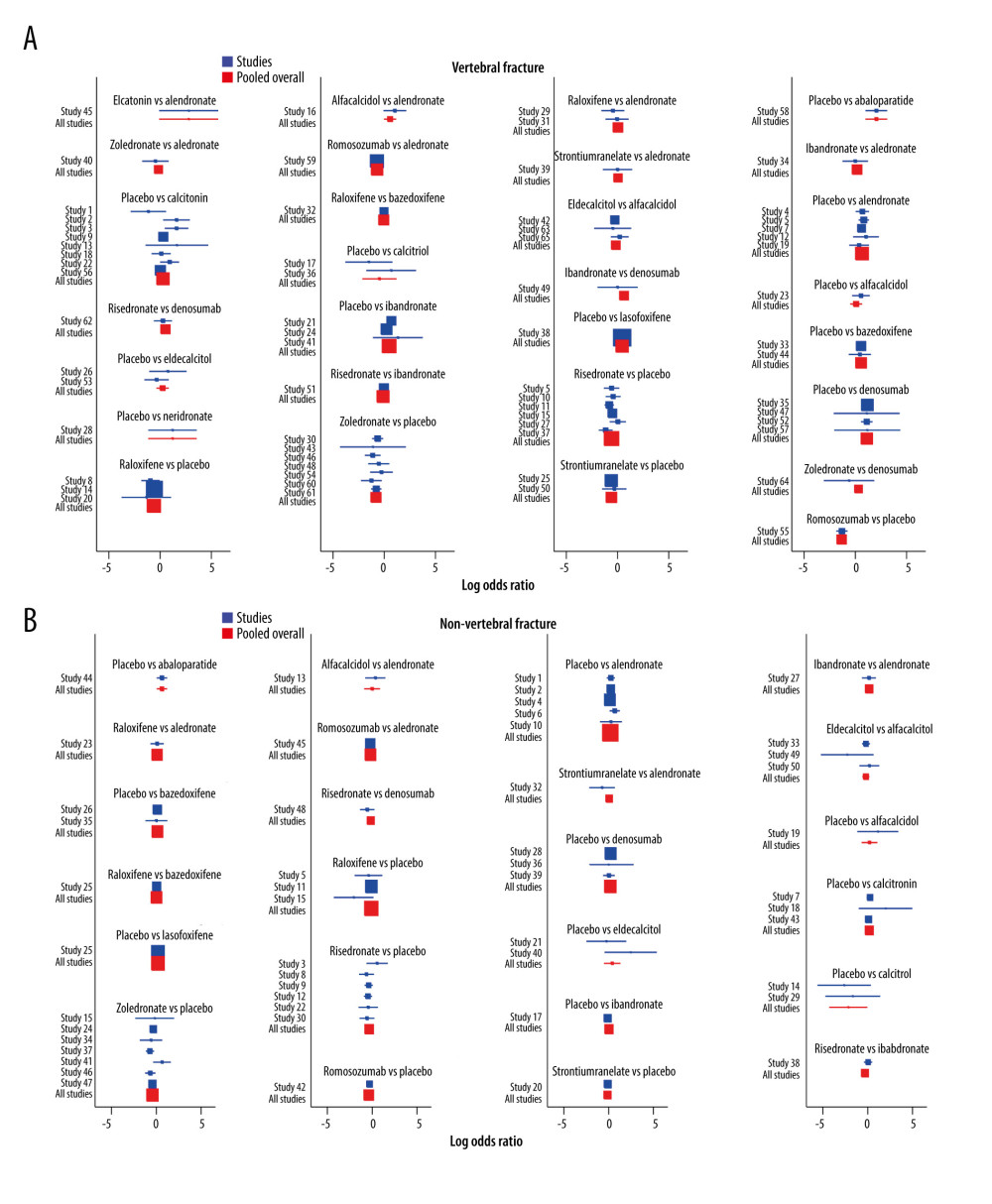 Figure 8. The pairwise comparisons agents for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 8. The pairwise comparisons agents for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA). 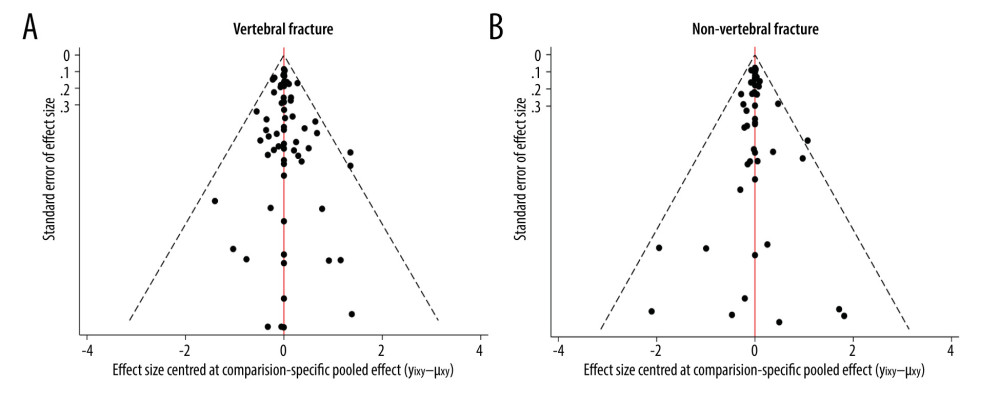 Figure 9. Funnel plots for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 9. Funnel plots for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA). References
1. Chesnut CH, Silverman S, Andriano K, A randomized trial of nasal spray salmon calcitonin in postmenopausal women with established osteoporosis: The prevent recurrence of osteoporotic fractures study. PROOF Study Group: Am J Med, 2000; 109(4); 267-76
2. Kanis JAon behalf of the World Health Organization Scientific Group, Assessment of osteoporosis at the primary health-care level: Technical Report, 2007, University of Sheffield, UK: World Health Organization Collaborating Centre for Metabolic Bone Diseases. University of Sheffield
3. Briot K, Cortet B, Thomas T, 2012 update of French guidelines for the pharmacological treatment of postmenopausal osteoporosis: Joint Bone Spine, 2012; 79(3); 304-13
4. Ross PD, Osteoporosis. Frequency, consequences, and risk factors: Arch Intern Med, 1996; 156(13); 1399-411
5. El-Hajj Fuleihan G, Chakhtoura M, Cauley JA, Chamoun N, Worldwide fracture prediction: J Clin Densitom, 2017; 20(3); 397-424
6. Whyte MP, Wenkert D, Clements KL, Bisphosphonate-induced osteopetrosis: N Engl J Med, 2003; 349(5); 457-63
7. Murad MH, Drake MT, Mullan RJ, Clinical review. Comparative effectiveness of drug treatments to prevent fragility fractures: A systematic review and network meta-analysis: J Clin Endocrinol Metab, 2012; 97(6); 1871-80
8. Freemantle N, Cooper C, Diez-Perez A, Results of indirect and mixed treatment comparison of fracture efficacy for osteoporosis treatments: a meta-analysis: Osteoporos Int, 2013; 24(1); 209-17
9. Barrionuevo P, Kapoor E, Asi N, Efficacy of pharmacological therapies for the prevention of fractures in postmenopausal women: A network meta-analysis: J Clin Endocrinol Metab, 2019; 104(5); 1623-30
10. Hu Y, Ke Z, Han J, Effectiveness of pharmacological therapies on bone mineral density and fracture at various sites: A network meta-analysis of randomized controlled trials: PROSPERO, 2020 CRD42020158203 Available from: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42020158203
11. Hutton B, Salanti G, Caldwell DM, The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: Checklist and explanations: Ann Intern Med, 2015; 162(11); 777-84
12. Jadad AR, Moore RA, Carroll D, Assessing the quality of reports of randomized clinical trials: is blinding necessary?: Control Clin Trials, 1996; 17(1); 1-12
13. Ades AE, Lu G, Higgins JPT, The interpretation of random-effects meta-analysis in decision models: Med Decis Making, 2005; 25(6); 646-54
14. White IR, Barrett JK, Jackson D, Higgins JPT, Consistency and inconsistency in network meta-analysis: Model estimation using multivariate meta-regression: Res Synth Methods, 2012; 3(2); 111-25
15. Bucher HC, Guyatt GH, Griffith LE, Walter SD, The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials: J Clin Epidemiol, 1997; 50(6); 683-91
16. Li D, Wang T, Shen S, Effects of fluroquinolones in newly diagnosed, sputum-positive tuberculosis therapy: A systematic review and network meta-analysis: PLoS One, 2015; 10(12); e0145066
17. Trinquart L, Chatellier G, Ravaud P, Adjustment for reporting bias in network meta-analysis of antidepressant trials: BMC Med Res Methodol, 2012; 12; 150
18. DerSimonian R, Laird N, Meta-analysis in clinical trials: Control Clin Trials, 1986; 7(3); 177-88
19. Deeks JJ, Higgins JPT, Altman DG, Analyzing data and undertaking meta-analyses: Cochrane Handbook for Systematic Reviews of Interventions 5.0.1, 2008, Oxford UK, The Cochrane Collaboration
20. Higgins JPT, Thompson SG, Deeks JJ, Altman DG, Measuring inconsistency in meta-analyses: BMJ, 2003; 327(7414); 557-60
21. Kataoka Y, Luo Y, Chaimani A, Cumulative network meta-analyses, practice guidelines, and actual prescriptions for postmenopausal osteoporosis: A meta-epidemiological study: Arch Osteoporos, 2020; 15(1); 21
22. Deng J, Silver Z, Huang E, Pharmacological prevention of fractures in patients undergoing glucocorticoid therapies: A systematic review and network meta-analysis: Rheumatology (Oxford), 2021; 60(2); 649-57
23. Liu Z, Zhang M, Shen Z, Efficacy and safety of 18 anti-osteoporotic drugs in the treatment of patients with osteoporosis caused by glucocorticoid: A network meta-analysis of randomized controlled trials: PLoS One, 2020; 15(12); e0243851
24. Lin SY, Hung MC, Chang SF, Efficacy and safety of postmenopausal osteoporosis treatments: A systematic review and network meta-analysis of randomized controlled trials: J Clin Med, 2021; 10(14); 3043
25. Migliorini F, Maffulli N, Colarossi G, Effect of drugs on bone mineral density in postmenopausal osteoporosis: A Bayesian network meta-analysis: J Orthop Surg Res, 2021; 16(1); 533
26. Liu Y, Cao Y, Zhang S, Romosozumab treatment in postmenopausal women with osteoporosis: A meta-analysis of randomized controlled trials: Climacteric, 2018; 21(2); 189-95
27. Lim SY, Bolster MB, Profile of romosozumab and its potential in the management of osteoporosis: Drug Des Devel Ther, 2017; 11; 1221-31
28. Ominsky MS, Boyd SK, Varela A, Romosozumab improves bone mass and strength while maintaining bone quality in ovariectomized cynomolgus monkeys: J Bone Miner Res, 2017; 32(4); 788-801
29. Kendler DL, Bone HG, Massari F, Bone mineral density gains with a second 12-month course of romosozumab therapy following placebo or denosumab: Osteoporos Int, 2019; 30(12); 2437-48
30. Geusens P, Oates M, Miyauchi A, The effect of 1 year of romosozumab on the incidence of clinical vertebral fractures in postmenopausal women with osteoporosis: Results from the FRAME study: JBMR Plus, 2019; 3(10); e10211
31. Ruza I, Mirfakhraee S, Orwoll E, Gruntmanis U, Clinical experience with intravenous zoledronic acid in the treatment of male osteoporosis: Evidence and opinions: Ther Adv Musculoskelet Dis, 2013; 5(4); 182-98
32. Hattersley G, Dean T, Corbin BA, Binding selectivity of abaloparatide for PTH-type-1-receptor conformations and effects on downstream signaling: Endocrinology, 2016; 157(1); 141-49
33. Leder BZ, O’Dea LSL, Zanchetta JR, Effects of abaloparatide, a human parathyroid hormone-related peptide analog, on bone mineral density in postmenopausal women with osteoporosis: J Clin Endocrinol Metab, 2015; 100(2); 697-706
34. Deal CL, Mitlak BH, Wang Y, Response rates for hip, femoral neck, and lumbar spine bone mineral density in patients treated with abaloparatide followed by alendronate: Results from phase 3 ACTIVExtend: Bone Rep, 2019; 11; 100230
35. Chandler H, Lanske B, Varela A, Abaloparatide, a novel osteoanabolic PTHrP analog, increases cortical and trabecular bone mass and architecture in orchiectomized rats by increasing bone formation without increasing bone resorption: Bone, 2019; 120; 148-55
36. Miller PD, Hattersley G, Lau E, Bone mineral density response rates are greater in patients treated with abaloparatide compared with those treated with placebo or teriparatide: Results from the ACTIVE phase 3 trial: Bone, 2019; 120; 137-40
37. McCloskey EV, Fitzpatrick LA, Hu MY, Effect of abaloparatide on vertebral, nonvertebral, major osteoporotic, and clinical fractures in a subset of postmenopausal women at increased risk of fracture by FRAX probability: Arch Osteoporos, 2019; 14(1); 15
38. Bahar H, Gallacher K, Downall J, Six weeks of daily abaloparatide treatment increased vertebral and femoral bone mineral density, microarchitecture and strength in ovariectomized osteopenic rats: Calcif Tissue Int, 2016; 99(5); 489-99
39. Doyle N, Varela A, Haile S, Abaloparatide, a novel PTH receptor agonist, increased bone mass and strength in ovariectomized cynomolgus monkeys by increasing bone formation without increasing bone resorption: Osteoporos Int, 2018; 29(3); 685-97
40. Varela A, Chouinard L, Lesage E, One Year of abaloparatide, a selective activator of the PTH1 receptor, increased bone formation and bone mass in osteopenic ovariectomized rats without increasing bone resorption: J Bone Miner Res, 2017; 32(1); 24-33
41. Cranney A, Wells G, Willan A, Meta-analyses of therapies for postmenopausal osteoporosis. II. Meta-analysis of alendronate for the treatment of postmenopausal women: Endocr Rev, 2002; 23(4); 508-16
42. Cranney A, Tugwell P, Wells GThe Osteoporosis Research Advisory Group, Meta-analyses of therapies for postmenopausal osteoporosis. I. Systematic reviews of randomized trials in osteoporosis: introduction and methodology: Endocr Rev, 2002; 23(4); 496-507
43. Cranney A, Guyatt G, Griffith L, Meta-analyses of therapies for postmenopausal osteoporosis. IX: Summary of meta-analyses of therapies for postmenopausal osteoporosis: Endocr Rev, 2002; 23(4); 570-78
44. Zhu S-Y, Deng Y, Wang Y-F, Bone protection for early menopausal women in China: Standard or half-dose estrogen with progestin? A one-year prospective randomized trail: Gynecol Endocrinol, 2019; 35(2); 165-69
45. Sullivan SD, Lehman A, Nathan NK, Age of menopause and fracture risk in postmenopausal women randomized to calcium + vitamin D, hormone therapy, or the combination: Results from the Women’s Health Initiative Clinical Trials: Menopause, 2017; 24(4); 371-78
46. Watts NB, Cauley JA, Jackson RD, No increase in fractures after stopping hormone therapy: Results from the women’s health initiative: J Clin Endocrinol Metab, 2017; 102(1); 302-8
47. Saarelainen J, Hassi S, Honkanen R, Bone loss and wrist fractures after withdrawal of hormone therapy: The 15-year follow-up of the OSTPRE cohort: Maturitas, 2016; 85; 49-55
48. Cauley JA, Estrogen and bone health in men and women: Steroids, 2015; 99; 11-15
49. Gambacciani M, Levancini M, Hormone replacement therapy and the prevention of postmenopausal osteoporosis: Menopause Review, 2014; 13; 213-20
Figures
 Figure 1. PRISMA flowchart for the literature search and study selection.
Figure 1. PRISMA flowchart for the literature search and study selection. Figure 2. Network of comparisons for all fracture included in the analysis, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 2. Network of comparisons for all fracture included in the analysis, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). Figure 3. The SUCRA rank test for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 3. The SUCRA rank test for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). Figure 4. The pairwise comparisons agents for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 4. The pairwise comparisons agents for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). Figure 5. Funnel plot for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 5. Funnel plot for all fracture, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). Figure 6. Network of comparisons for vertebral fracture (A) and non-vertebral fracture (B) included in the analysis, Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 6. Network of comparisons for vertebral fracture (A) and non-vertebral fracture (B) included in the analysis, Stata software (version 12.0; Stata Corporation, College Station, TX, USA). Figure 7. The SUCRA rank test for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 7. The SUCRA rank test for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA). Figure 8. The pairwise comparisons agents for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 8. The pairwise comparisons agents for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA). Figure 9. Funnel plots for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA).
Figure 9. Funnel plots for vertebral fracture (A) and non-vertebral fracture (B), Stata software (version 12.0; Stata Corporation, College Station, TX, USA). In Press
08 Mar 2024 : Clinical Research
Evaluation of Foot Structure in Preschool Children Based on Body MassMed Sci Monit In Press; DOI: 10.12659/MSM.943765
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








