28 September 2022: Clinical Research
Factors Predictive of Varicella Zoster Virus Encephalitis/Meningitis: A Single-Center, Retrospective Study
Yanrong Yuan1ABCE, Jun Wang1BCF, Yan Zhang1BC, Huili Liu1BC, Yongxing Yan1ACDFG*DOI: 10.12659/MSM.938057
Med Sci Monit 2022; 28:e938057
Abstract
BACKGROUND: The outcomes of varicella zoster virus (VZV) encephalitis/meningitis vary from complete recovery to severe complications. This study aimed to investigate the predictive factors of encephalitis and meningitis caused by VZV reactivation.
MATERIAL AND METHODS: VZV encephalitis/meningitis patients (n=39) who were treated between January 2019 and December 2021 were included. Patients were followed up for 3 months after discharge and divided into a favorable outcome group (FO, n=18; 46.2%) and an unfavorable outcome group (UO, n=21; 53.8%) according to whether it affects quality of life. The clinical data were retrospectively analyzed and compared between groups.
RESULTS: As compared to the FO group, patients in the UO group were more likely to have higher body temperature (>38°C) at admission, longer interval from onset of CNS symptoms to initial of antiviral therapy, higher white blood cells (WBC) and adenosine deaminase (ADA) in the cerebrospinal fluid (CSF) and higher CRP in the blood (P<0.05 or P<0.01). Univariate logistic regression analysis showed CSF ADA (OR=1.279, 95% CI: 0.996~1.642) and interval from onset of CNS symptoms to initial of antiviral therapy (OR=1.299, 95% CI: 1.011~1.669) were independent risk factors for unfavorable outcomes (P<0.05). The sensitivity and specificity of combined CSF ADA and time interval from onset of CNS symptoms to initial of antiviral therapy were 78.8% and 95.2%, respectively, in predicting outcomes.
CONCLUSIONS: Higher CSF ADA and longer interval from onset of CNS symptoms to initial of antiviral therapy predict an unfavorable outcome, and the combination of both factors can achieve better performance.
Keywords: Encephalitis, Meningitis, Treatment Outcome, Varicella Zoster Virus Infection, Adenosine Deaminase, Antiviral Agents, Encephalitis, Varicella Zoster, Herpesvirus 3, Human, Humans, Quality of Life
Background
Herpes zoster (HZ) is caused by reactivation of latent varicella zoster virus (VZV) in the human body, and is characterized by clustered papules and hyaline blisters with strip-like distribution, which is often accompanied by pain [1]. Epidemiological studies have shown that the incidence of HZ is gradually increasing [2–6]. Generally, HZ in most patients can be completely cured without complications, but some patients, especially the elderly and immunocompromised patients, develop various complications (eg, postherpetic neuralgia, herpes zoster ophthalmicus). Severe complication can cause central nervous system infection and even death. Among central nervous system (CNS) complications of HZ, meningitis and encephalitis are most common [7–9].
With the advancement of polymerase chain reaction (PCR) technology, the diagnosis of CNS infections such as VZV encephalitis/meningitis has become more convenient, especially for intracranial VZV infection without skin lesions. Some case series and retrospective studies on CNS VZV infection have been reported [10–14]. Corral et al [12] analyzed the clinical spectrum and outcome of 98 patients with neurological complications of reactivated VZV infection, finding the clinical spectrum of neurological complications in VZV reactivation is wide. Mortality was low (3%) and 24% of patients had persistent symptoms, although these were usually mild. Shorter latency between herpes zoster and neurologic symptoms was the risk factor associated with unfavorable outcome. Yan et al [13] summarized the clinical data of 74 patients with VZV meningitis/meningoencephalitis, and found that fever, headache, cognitive changes or cranial nerve involvement, nausea and vomiting or meningeal irritation, and Ramsay-Hunt syndrome (RHS) were the most common clinical manifestations of VZV meningitis/meningoencephalitis, and there were no characteristic abnormal manifestations on brain MRI. Early intravenous antiviral treatment for VZV meningitis/meningoencephalitis mostly has a favorable outcome. The clinical information on 92 VZV encephalitis patients in Denmark from 2015 to 2019 was analyzed by Herlin et al [14], who found the diagnosis of VZV encephalitis was often delayed. Predominant symptoms were confusion (76%), headache (56%), nausea (45%), gait disturbance (42%), and personality changes (41%). The mortality rates in-hospital and at 1 and 3 months after since discharge were 4%, 9%, and 11%, respectively. Unfavorable outcome was found in 69% at discharge. The risk factors for unfavorable outcome were age, cerebral vasculitis, and GCS <15. Some study results indicate that CNS VZV infection usually has no specific symptoms and signs at the early stage, the outcome varies from complete recovery to severe disability and death, and the factors affecting the prognosis are different. Some retrospective studies included cases with a long span, even more than 10 years, which causes inconvenience in clinical practice. Further studies are warranted to assess the outcome of VZV encephalitis/meningitis because the understanding of risk factors affecting outcome of CNS VZV infection and early intervention is crucial to reduce the incidence of VZV encephalitis/meningitis and the disability caused by VZV encephalitis/meningitis. The present study aimed to investigate the clinical characteristics of VZV encephalitis/meningitis patients who were treated in our center, and to explore the clinical features and risk factors of VZV encephalitis/meningitis with poor outcome, which may provide reference for the clinical diagnosis and early treatment of CNS VZV infection, thereby reducing the incidence of sequelae.
Material and Methods
PATIENTS AND GROUPING:
Thirty-nine patients with VZV encephalitis/meningitis who were treated in our hospital between January 2019 and December 2021 were included in the present study. They were diagnosed by using CSF VZV DNA detection. The inclusion criteria were as follows: (1) diagnosed with VZV encephalitis/meningitis according to the diagnostic criteria from European consensus-based guideline on the management of the herpes zoster [15]; (2) herpes zoster diagnosis within 4 weeks; (3) CSF was negative bacterial, fungus culture; (4) patients older than 18 years; (5) clinical data (symptoms, signs, auxiliary examinations, and treatments) were complete; (6) patients were followed up for more than 3 months after discharge. The exclusion criteria were as follows: (1) patients had incomplete clinical data; (2) patients had central infections caused by other pathogens such as other viruses, Treponema pallidum or fungi; (3) encephalitis/meningitis caused by other diseases.
OUTCOME JUDGMENT:
Based on whether it affects the patient’s quality of life within 3 months after discharge, the patients were divided into a favorable outcome group (FO; patients had absence of symptoms or had only mild pain [NRS score less than 3 points] or modified Rankin Scale (mRS) <2) or an unfavorable outcome group (UO; patients had pain [NRS score higher than 3], or mRS ≥2). The outcome and NRS/mRS score were determined by the specialists in our center. Of the 39 patients included, there were 18 in the FO group and 21 in the UO group.
METHODS:
The clinical data of patients were collected, including general characteristics (age, gender, comorbidities, VZV vaccination), clinical characteristics at baseline (site of herpes, clinical manifestations, body temperature at admission, time interval from HZ to the onset of neurological symptoms, and time interval from the onset of neurological symptoms to the initiation of antiviral treatment, hospital stay, duration of antiviral treatment, glucocorticoids therapy) and laboratory findings. The results of first routine blood test, C-reactive protein (CRP), blood biochemical test, first routine CSF test, CSF biochemical test, and pathogen culture were also recorded for further analysis. The findings were compared between the FO group and UO group.
ANTIPYRETIC PROTOCOL:
Patients with body temperature below 38.5°C were given physical cooling (eg, alcohol wiping or ice bag cold compress). Patients with temperature above 38.5°C were given indomethacin suppository 50 mg to plug the anus in addition to physical cooling.
ANTIVIRAL THERAPY:
Thirty-seven patients were injected with acyclovir at a dose of 10mg/kg, once every 8 h for 10–21 days. Two patients with chronic childhood disease were treated with foscarnet sodium injection (40 mg/kg, Q8H) for 10–14 days.
STATISTICAL ANALYSIS:
Statistical analysis was performed with SPSS version 25.0 for Windows. Qualitative data are expressed as rates and compared with the chi-square test. Quantitative data with normal distribution are expressed as mean±standard deviation (SD) and those without normal distribution as median (interquartile); these data were compared with the
ETHICS CONSIDERATION:
All procedures were conducted in accordance with the Helsinki Declaration revised in 1983. This study was approved by the Ethics Committee of our hospital before study initiation (NO. 2021KA013).
Results
GENERAL CHARACTERISTICS:
A total of 39 patients were included in the present study. Of them, 9 were diagnosed with VZV encephalitis and 30 with VZV meningitis. None of the patients received VZV vaccination. There were 28 males and 11 females in our study and the mean age was 54.8±16.6 years (range: 21–81 years). According to whether it affects the patient’s quality of life in the follow up period, 18 patients had favorable outcome (18/39, 46.2%), including 13 males and 5 females with the mean age of 52.7±16.9 years (range: 24–76 years, while 21 patients had unfavorable outcome (21/39, 53.8%) and there were 15 males and 6 females with the mean age of 56.6±16.5 years (range: 21–81 years). There were no significant differences in gender and age between groups (P>0.05) (Table 1).
CLINICAL CHARACTERISTICS OF PATIENTS IN THE 2 GROUPS:
The proportion of patients with fever at admission (body temperature higher than 38°C) (33.3% vs 71.4%) and the time interval from the onset of CNS symptoms to the initiation of antiviral therapy (1 [1–2] days vs 4 [2–6] days) were significantly different between groups. However, there were no significant differences in the comorbidities, site of herpes, other clinical manifestations, and time interval from the HZ to the onset of CNS symptoms (P>0.05) (Table 2).
LABORATORY FINDINGS OF PATIENTS IN THE 2 GROUPS:
In the UO group, the CSF WBC count was markedly higher (18 [4–50.8] vs 75 [8–202.5]) and CSF ADA increased significantly (1.3±0.9 vs 2.5±1.5) as compared to the FO group. The CSF protein content increased in the UO group (61.5±23.4 vs 82.4±40.5), but there was no marked difference between groups (P=0.06). In the UO group, the blood CRP was significantly higher than in the FO group, but there were no marked differences in the blood WBC count, neutrophil proportion, lymphocyte proportion, CSF chlorine, CSF glucose, and CSF LDH between the 2 groups (P>0.05) (Table 3).
TREATMENTS IN 2 GROUPS:
All the patients received intravenous antiviral therapy. Of them, 28 patients (28/39, 71.8%) received intravenous glucocorticoids therapy for 3–5 days (methylprednisolone, 40–80 mg/d). In the FO group, the hospital stay was 17.6±3.6 days, the duration of antiviral therapy was 15.9±3.6 days, and 12 patients received glucocorticoids therapy (12/18, 66.7%). In the UO group, the hospital stay was 18.7±8.4 days, the duration of antiviral therapy was 15.7±5.7 days, and 16 pateints received glucocorticoids therapy (16/21, 76.2%). There were no marked differences in the hospital stay, duration of antiviral therapy, and glucocorticoids therapy (P>0.05) between groups (Table 2).
INDEPENDENT RISK FACTORS FOR UNFAVORABLE OUTCOME:
The outcome within 3 months after discharge served as the dependent variable, and the above factors with significant difference (fever [temperature >38°C]; time interval from onset of CNS symptoms to initiation of antiviral therapy; CSF WBC count; CSF ADA; blood CRP) served as independent variables in the multivariate logistic regression analysis. Results showed that the CSF ADA (OR=1.279, 95% CI: 0.996–1.642) and time interval from the onset of CNS symptoms to the initiation of antiviral therapy (OR=1.299, 95% CI: 1.011–1.669) were the independent risk factors for unfavorable outcome of patients with VZV encephalitis/meningitis (P<0.05) (Table 4). Correlation analysis indicated that there was a positive correlation between CSF WBC count and ADA content (r=0.32, P=0.04) (Figure 1).
When the CSF ADA levels was 1.6 u/L, the AUC, sensitivity and specificity of CSF ADA in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 0.802, 77.8%, and 81.0%, respectively. When the time interval from the onset of CNS symptoms to the initiation of antiviral therapy was 2.5 days, the AUC, sensitivity, and specificity of the time interval from the onset of CNS symptoms to the initiation of antiviral therapy in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 0.839, 88.9%, and 66.7%, respectively. With the combined use of CSF ADA and time interval from the onset of CNS symptoms to the initiation of antiviral therapy, the AUC, sensitivity and specificity were 0.919, 78.8%, and 95.4%, respectively, in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge (Figure 2).
Discussion
Acute encephalitis/meningitis is the most common complication of CNS VZV infection. The identification of VZV DNA or/and anti-VZV antibody in the CSF is an important clue in the diagnosis of CNS VZV infection. In the present study, all the patients had a history of HZ, had clear clinical symptoms, and were positive for VZV DNA, abnormal white blood cell count and protein content in the CSF. Therefore, they were diagnosed with CNS VZV infection. Previous literature reported that the outcome of VZV encephalitis/meningitis patients was varies from complete recovery to severe disability and death. In the present study, the incidence of unfavorable outcome was 53.8% at 3 months after discharge, which was significantly higher than previously reported [12,13,16,17]. This might be ascribed to the difference in the criteria for outcome assessment. Some investigators [16,17] used the GOS score for the outcome assessment. Generally, GOS is mainly used for the assessment of outcome after brain trauma, but it is less sensitive in patients with mild symptoms and no consciousness disturbance. Some VZV encephalitis/meningitis patients had no noticeable symptoms or had only mild symptoms, which may have led to deviation if GOS scores were used to evaluate the outcome of those patients. Corral et al [12] employed mRS for the assessment of prognosis, but mRS is mostly used for evaluation of outcomes after stroke. Their prognostic criteria are different from ours. Therefore, in the present study, based on whether it affects the patient’s quality of life, the combination of NRS score and mRS score, but not the functional disability alone, was employed for the assessment of outcomes after VZV encephalitis/meningitis. In addition, PHN is one of main complications of HZ and its incidence is higher than 5% or even up to 30% [18,19], An NRS score of more than 3 is classified as moderate pain, which can significantly affect quality of life. Therefore, patients with an NRS score of more than 3 were assigned to the UO group in this study, which may have led to higher incidence of unfavorable outcomes compared with previous reports.
There are many factors influencing the outcome of VZV encephalitis/meningitis. Our study indicated that the time interval from the onset of CNS symptoms to the initiation of antiviral therapy was an independent risk factor for poor outcome, and the longer the time interval, the worse the outcome was, which was similar to the findings in previous reports [8,13]. This suggests that delayed treatment worsens VZV encephalitis/meningitis, resulting in a poor outcome. Therefore, clinicians should pay more attention to HZ patients with CNS symptoms. Early diagnosis and early intravenous antiviral treatment are crucial for the patients with suspected CNS VZV infection, thereby reducing the occurrence of poor prognosis.
CSF adenosine deaminase (ADA) levels in the UO group were significantly higher than in the FO group, and it was also one of the independent risk factors for poor prognosis. ADA is an important enzyme in purine nucleoside metabolism. It is a hydrolase acting in the purine nucleoside decomposition and is widely distributed in various tissues of the human body. There is evidence showing that CSF ADA levels in patients with tuberculous meningitis are significantly higher than in those with non-tuberculous meningitis, and CSF ADA has a better predictive performance for tuberculous meningitis [20]. Elevated CSF ADA level has also been reported in other neurological disorders such as lymphoma, sarcoidosis, neurobrucellosis, neurocytomegalovirus disease, cryptococcal meningitis, candidal meningitis, and subarachnoid bleeding [21–23]. Fujiwara et al [24] reported that CSF ADA levels were higher in patients with autoimmune encephalitis. Some studies have confirmed that ADA is an enzyme related to immunity. It is closely related to the immune response and disease progression, and is involved in proliferation and differentiation of lymphocytes, especially T lymphocytes. In the presence of antigens, T cells are activated to release ADA. Our study showed the CSF ADA level in the UO group was significantly higher than in the FO group, and it was one of the independent risk factors for unfavorable outcome. Although CSF WBC count was not an independent risk factor for unfavorable outcome, univariate analysis showed the CSF WBC count in the UO group was significantly higher than in the FO group (
In this study, we found that the time interval from onset of CNS symptoms to the initiation of antiviral therapy and the content of ADA in CSF were independent risk factors for unfavorable outcome of VZV encephalitis/meningitis patients. CSF ADA and time interval from the onset of CNS symptoms to the initiation of antiviral therapy, the AUC, sensitivity, and specificity were 0.919, 78.8%, and 95.4%, respectively, in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge. The combined predictive ability of the 2 factors is higher than with use of a single factor. This suggests that clinicians should pay attention to the level of ADA in CSF of patients with VZV encephalitis/meningitis while using antiviral drugs as early as possible to provide early prediction of patient outcome. CSF is often collected for diagnosis of CNS diseases. The detection of CSF-related parameters is helpful for the clinical diagnosis of CNS infection and is also crucial for assessment of disease severity and response to therapy. In the present study, the CSF WBC count and protein content was significantly higher in patients with VZV encephalitis/meningitis, which was consistent with previous reports. This further highlights the important of CSF detection for patients with suspected intracranial infection.
There are several limitations of the present study. First, this was a single-center, retrospective study, and the sample size was small after exclusion of patients with incomplete clinical data, which may bias our results to a certain extent. Second, the duration of follow-up was short (only 3 months). Third, the parameters were not dynamically monitored in these patients. Moreover, due to incomplete clinical data, other factors (such as the extent and duration of herpes lesions) that may affect the prognosis were not included in the analysis.
Conclusions
In summary, early diagnosis and early treatment of CNS VZV infection are extremely important for patients with VZV encephalitis/meningitis. Patients with VZV encephalitis/meningitis are more likely to have complications that affect quality of life. In addition, the higher the CSF ADA level and the longer the time interval from the onset of CNS symptoms to the initiation of antiviral therapy, the worse the outcome within 3 months after discharge. The combination of CSF ADA level and time interval from the onset of CNS symptoms to initiation of antiviral therapy may be employed for prediction of outcomes in patients with VZV encephalitis/meningitis. Prospective, multicenter studies with larger sample sizes are warranted to confirm our findings.
Figures
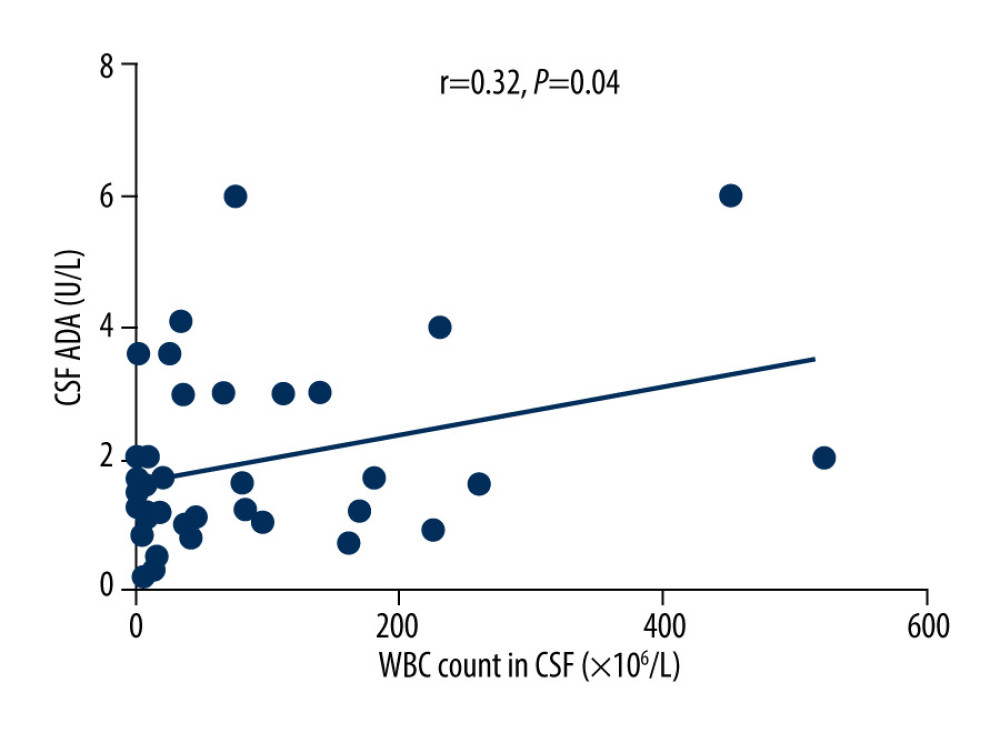 Figure 1. There was a positive correlation between CSF WBC count and ADA content (r=0.32, P=0.04).
Figure 1. There was a positive correlation between CSF WBC count and ADA content (r=0.32, P=0.04). 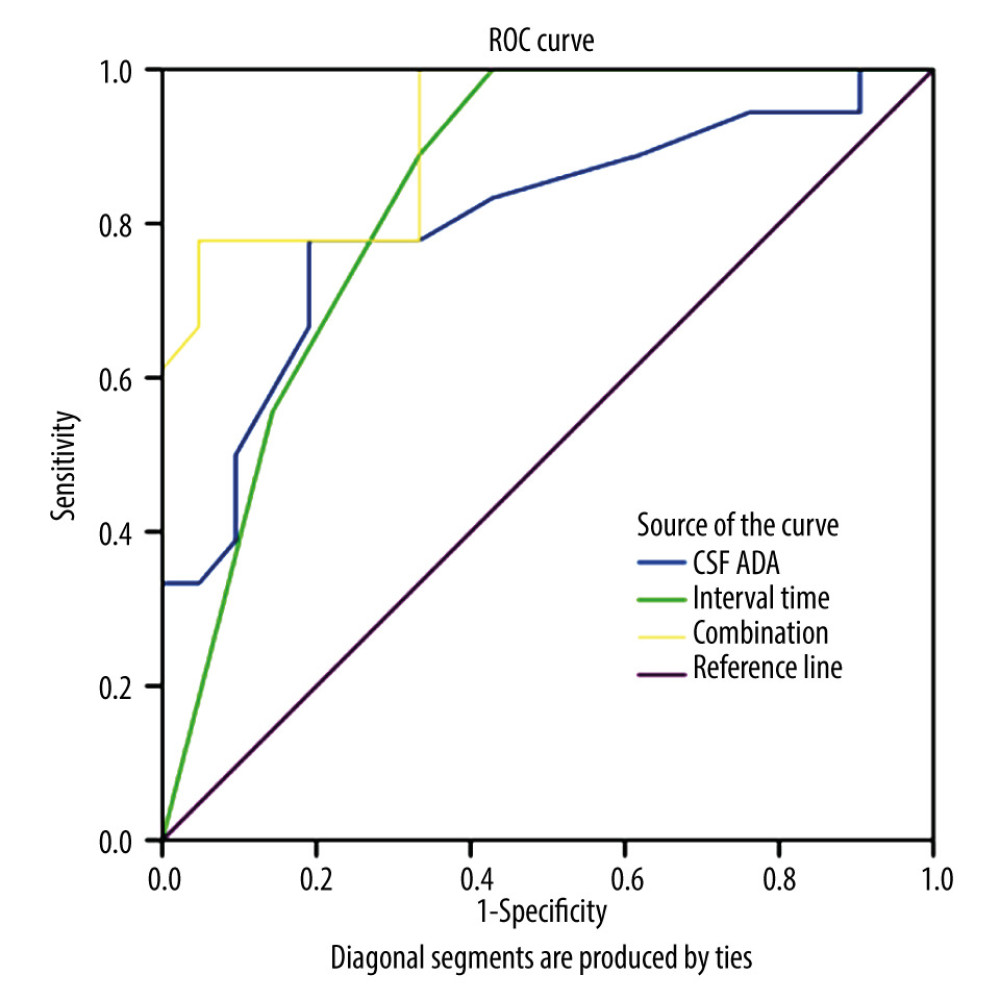 Figure 2. The sensitivity and specificity of CSF ADA in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 77.8% and 81.0%, respectively. The sensitivity and specificity of the time interval from the onset of CNS symptoms to the initiation of antiviral therapy in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 88.9% and 66.7%, respectively. With the combined use of CSF ADA and time interval from the onset of CNS symptoms to the initiation of antiviral therapy, the sensitivity and specificity were 78.8% and 95.4%, respectively, in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge.
Figure 2. The sensitivity and specificity of CSF ADA in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 77.8% and 81.0%, respectively. The sensitivity and specificity of the time interval from the onset of CNS symptoms to the initiation of antiviral therapy in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 88.9% and 66.7%, respectively. With the combined use of CSF ADA and time interval from the onset of CNS symptoms to the initiation of antiviral therapy, the sensitivity and specificity were 78.8% and 95.4%, respectively, in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge. References
1. Cohen JI, Clinical practice: Herpes zoster: N Engl J Med, 2013; 369(3); 255-63
2. Chao DY, Chien YZ, Yeh YP, The incidence of varicella and herpes zoster in Taiwan during a period of increasing varicella vaccine coverage, 2000–2008: Epidemiol Infect, 2012; 140(6); 1131-40
3. Leung J, Harpaz R, Molinari NA, Herpes zoster incidence among insured persons in the United States, 1993–2006: Evaluation of impact of varicella vaccination: Clin Infect Dis, 2011; 52(3); 332-40
4. Nelson MR, Britt HC, Harrison CM, Evidence of increasing frequency of herpes zoster management in Australian general practice since the introduction of a varicella vaccine: Med J Aust, 2010; 193(2); 110-13
5. Pierik JG, Gumbs PD, Fortanier SA, Epidemiological characteristics and societal burden of varicella zoster virus in the Netherlands: BMC Infect Dis, 2012; 12; 110
6. Esteban-Vasallo MD, Gil-Prieto R, Domínguez-Berjón MF, Temporal trends in incidence rates of herpes zoster among patients treated in primary care centers in Madrid (Spain), 2005–2012: J Infect, 2014; 68(4); 378-86
7. Nagel MA, Niemeyer CS, Bubak AN, Central nervous system infections produced by varicella zoster virus: Curr Opin Infect Dis, 2020; 33(3); 273-78
8. Arruti M, Piñeiro LD, Salicio Y, Incidence of varicella zoster virus infections of the central nervous system in the elderly: A large tertiary hospital-based series (2007–2014): J Neurovirol, 2017; 23(3); 451-59
9. Halling G, Giannini C, Britton JW, Focal encephalitis following varicella zoster virus reactivation without rash in a healthy immunized young adult: J Infect Dis, 2014; 210(5); 713-16
10. Persson A, Bergström T, Lindh M, Varicella-zoster virus CNS disease-viral load, clinical manifestations and sequels: J Clin Virol, 2009; 46(3); 249-53
11. Becerra JC, Sieber R, Martinetti G, Infection of the central nervous system caused by varicella zoster virus reactivation: A retrospective case series study: Int J Infect Dis, 2013; 17(7); e529-34
12. Corral C, Quereda C, Muriel A, Clinical spectrum and prognosis of neurological complications of reactivated varicella-zoster infection: The role of immunosuppression: J Neurovirol, 2020; 26(5); 696-703
13. Yan Y, Yuan Y, Wang J, Meningitis/meningoencephalitis caused by varicella zoster virus reactivation: A retrospective single-center case series study: Am J Transl Res, 2022; 14(1); 491-500
14. Herlin LK, Hansen KS, Bodilsen J, Varicella zoster virus encephalitis in Denmark from 2015 to 2019 – a nationwide prospective cohort study: Clin Infect Dis, 2021; 72(7); 1192-99
15. Werner RN, Nikkels AF, Marinović B, European consensus-based (S2k) guideline on the management of herpes zoster-guided by the European Dermatology Forum (EDF) in cooperation with the European Academy of Dermatology and Venereology (EADV), Part 1: Diagnosis: J Eur Acad Dermatol Venereol, 2017; 31(1); 9-19
16. Granerod J, Ambrose HE, Davies NW, Causes of encephalitis multicentre, population-based prospective study: Lancet Infect Dis, 2010; 10(12); 835-44
17. Mailles A, De Broucker T, Costanzo P, Long-term outcome of patients presenting with acute infectious encephalitis of various causes in France: Clin Infect Dis, 2012; 54(10); 1455-64
18. Kawai K, Gebremeskel BG, Acosta CJ, Systematic review of incidence and complications of herpes zoster: Towards a global perspective: BMJ Open, 2014; 4(6); e004833
19. Yang F, Yu S, Fan B, The epidemiology of herpes zoster and postherpetic neuralgia in china: Results from a cross-sectional study: Pain Ther, 2019; 8(2); 249-59
20. Pormohammad A, Riahi SM, Nasiri MJ, Diagnostic test accuracy of adenosine deaminase for tuberculous meningitis: A systematic review and meta-analysis: J Infect, 2017; 74(6); 545-54
21. He Z, Li X, Jiang L, Clinical analysis on 430 cases of infantile purulent meningitis: Springerplus, 2016; 5(1); 1994
22. Cho BH, Kim BC, Yoon GJ, Adenosine deaminase activity in cerebrospinal fluid and serum for the diagnosis of tuberculous meningitis: Clin Neurol Neurosurg, 2013; 115(9); 1831-36
23. Shinde A, A study of cerebrospinal fluid adenosine deaminase and C-reactive protein in Bacterial, Tubercular and Viral meningitis: Asian J Biomed Pharm Sci, 2015; 5(44); 15-18
24. Fujiwara S, Mimura N, Yoshimura H, Elevated adenosine deaminase levels in the cerebrospinal fluid in immune checkpoint inhibitor-induced autoimmune encephalitis: Intern Med, 2019; 58(19); 2871-74
Figures
 Figure 1. There was a positive correlation between CSF WBC count and ADA content (r=0.32, P=0.04).
Figure 1. There was a positive correlation between CSF WBC count and ADA content (r=0.32, P=0.04). Figure 2. The sensitivity and specificity of CSF ADA in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 77.8% and 81.0%, respectively. The sensitivity and specificity of the time interval from the onset of CNS symptoms to the initiation of antiviral therapy in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 88.9% and 66.7%, respectively. With the combined use of CSF ADA and time interval from the onset of CNS symptoms to the initiation of antiviral therapy, the sensitivity and specificity were 78.8% and 95.4%, respectively, in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge.
Figure 2. The sensitivity and specificity of CSF ADA in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 77.8% and 81.0%, respectively. The sensitivity and specificity of the time interval from the onset of CNS symptoms to the initiation of antiviral therapy in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge were 88.9% and 66.7%, respectively. With the combined use of CSF ADA and time interval from the onset of CNS symptoms to the initiation of antiviral therapy, the sensitivity and specificity were 78.8% and 95.4%, respectively, in predicting the outcome of VZV encephalitis/meningitis at 3 months after discharge. Tables
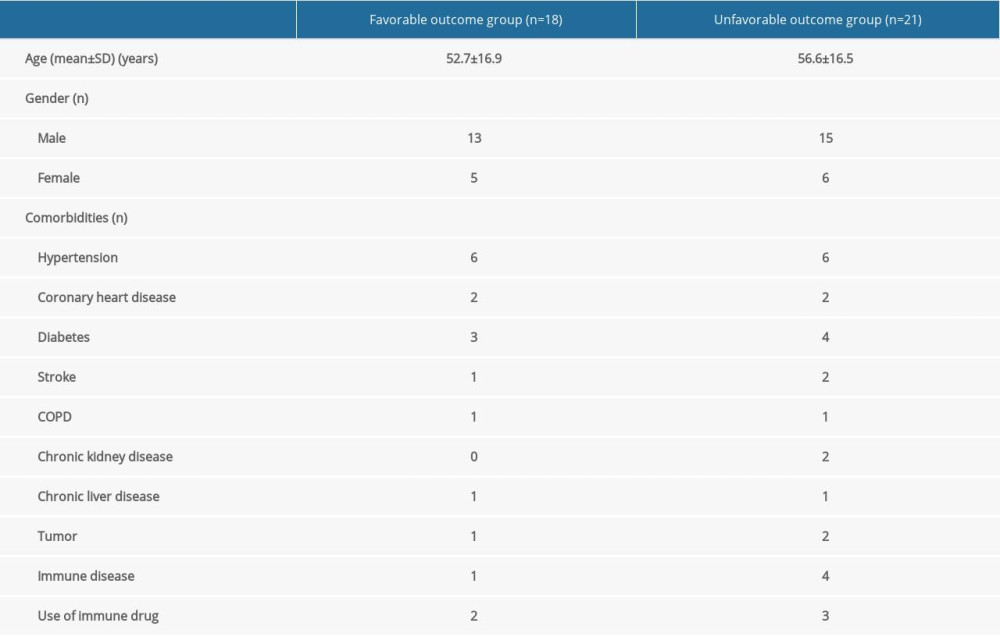 Table 1. Baseline characteristics of patients in 2 groups.
Table 1. Baseline characteristics of patients in 2 groups.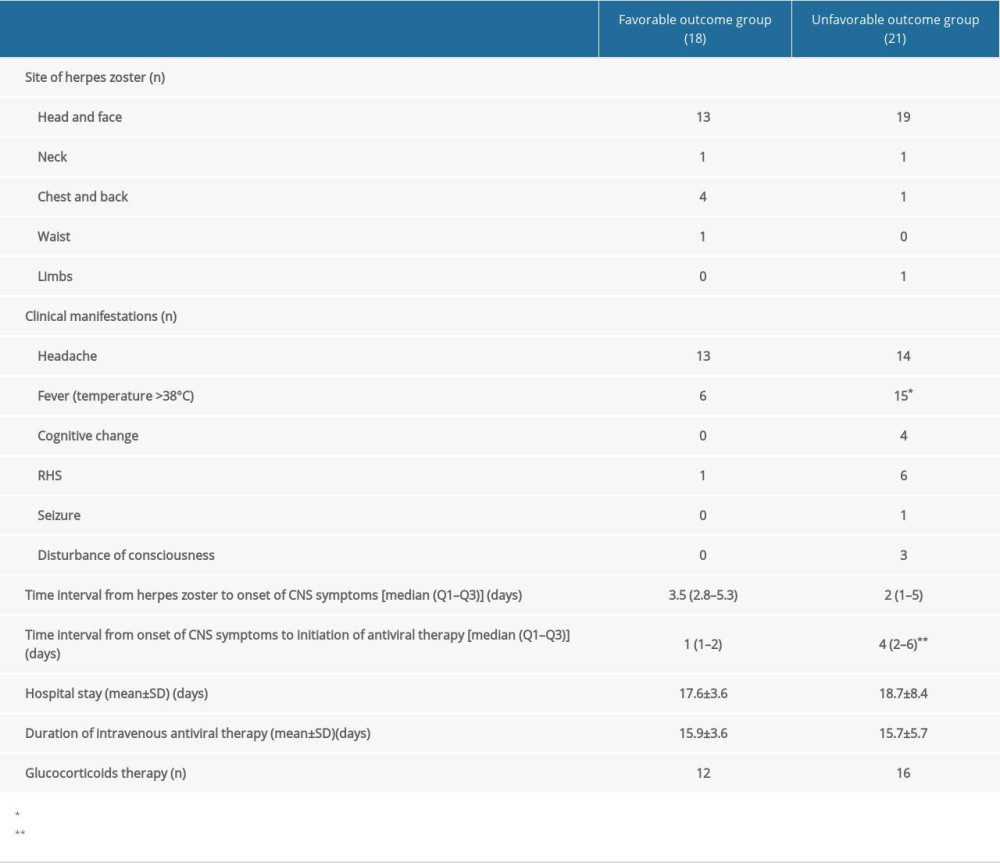 Table 2. Clinical characteristics of patients in 2 groups.
Table 2. Clinical characteristics of patients in 2 groups.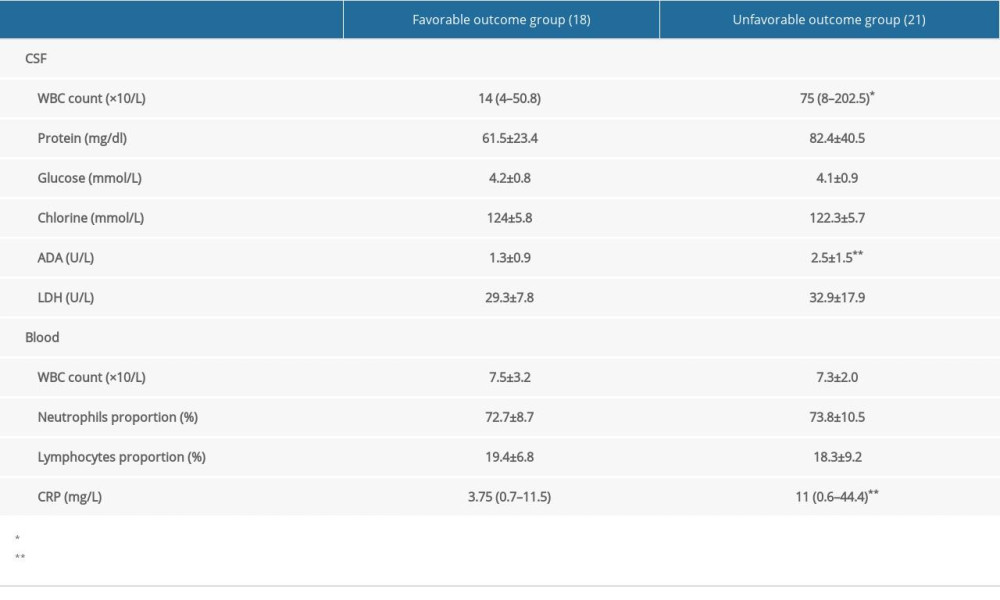 Table 3. Laboratory findings of patients in 2 groups.
Table 3. Laboratory findings of patients in 2 groups.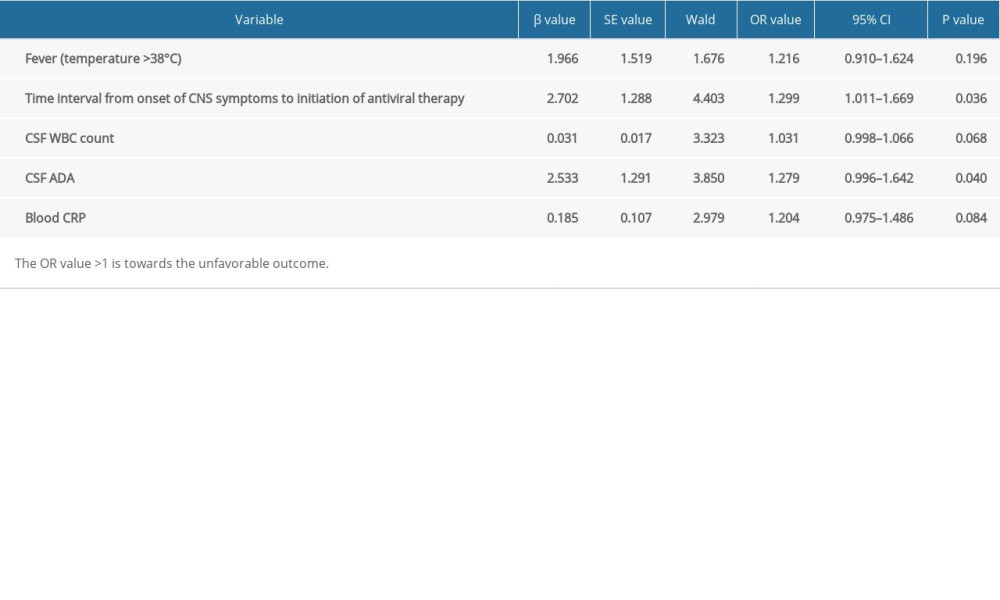 Table 4. Logistic regression analysis of factors affecting outcomes of VZV encephalitis/meningitis.
Table 4. Logistic regression analysis of factors affecting outcomes of VZV encephalitis/meningitis. Table 1. Baseline characteristics of patients in 2 groups.
Table 1. Baseline characteristics of patients in 2 groups. Table 2. Clinical characteristics of patients in 2 groups.
Table 2. Clinical characteristics of patients in 2 groups. Table 3. Laboratory findings of patients in 2 groups.
Table 3. Laboratory findings of patients in 2 groups. Table 4. Logistic regression analysis of factors affecting outcomes of VZV encephalitis/meningitis.
Table 4. Logistic regression analysis of factors affecting outcomes of VZV encephalitis/meningitis. In Press
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
08 Mar 2024 : Clinical Research
Evaluation of Foot Structure in Preschool Children Based on Body MassMed Sci Monit In Press; DOI: 10.12659/MSM.943765
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








