Abstract
BACKGROUND: Pancreatic ductal adenocarcinoma (PDAC) accounts for 85% of pancreatic carcinoma cases. Patients with PDAC have a poor prognosis. The lack of reliable prognostic biomarkers makes treatment challenging for patients with PDAC. Using a bioinformatics database, we sought to identify prognostic biomarkers for PDAC.
MATERIAL AND METHODS: Using proteomic analysis of the Clinical Proteomics Tumor Analysis Consortium (CPTAC) database, we were able to identify core differential proteins between early and advanced pancreatic ductal adenocarcinoma tissue, and then we used survival analysis, Cox regression analysis, and area under the ROC curves to screen for more significant differential proteins. Additionally, the Kaplan-Meier plotter database was utilized to determine the relationship between prognosis and immune infiltration in PDAC.
RESULTS: We identified 378 differential proteins in early (n=78) and advanced stages (n=47) of PDAC (P<0.05). PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 served as independent prognostic factors of patients with PDAC. Patients with higher COPS5 expression had shorter overall survival (OS) and recurrence-free survival, and those with higher PLG, ITGB3, and SPTA1, and lower FYN and IRF3 expression had shorter OS. More importantly, COPS5, IRF3 were negatively associated with macrophages and NK cells, but PLG, FYN, ITGB3, and SPTA1 were positively related to the expression of CD8+ T cells and B cells. COPS5 affected the prognosis of PDAC patients by acting on B cells, CD8+ T cells, macrophages, and NK cells immune infiltration, while PLG, FYN, ITGB3, IRF3, and SPTA1 affected PDAC patient prognosis through some immune cells.
CONCLUSIONS: PLG, COPS5, FYN, IRF3, ITGB3 and SPTA1 could be potential immunotherapeutic targets and valuable prognostic biomarkers of PDAC.
Keywords: Biomarkers, Immunity, Pancreatic Neoplasms, Prognosis, Proteomics, Humans, Biomarkers, Tumor, Carcinoma, Pancreatic Ductal, Adenocarcinoma
Background
According to estimates, 60 430 people in the United States will be diagnosed with pancreatic cancer in 2021, and approximately 48 220 will die of the disease. In the United States, pancreatic cancer is the fourth most common cause of cancer-related deaths among men (after lung, prostate, and colorectal cancer) and women (after lung, breast, and colorectal cancer) [1]. Over 90% of pancreatic cancer cases are pancreatic ductal adenocarcinoma (PDAC), which has a very poor prognosis [2]. It is estimated that PDAC will become the second leading cause of cancer-related death by 2030, and there is only a 12-month median survival rate for people with advanced metastatic disease [3].
The widely used biomarker, cancer antigen 19-9 (CA199), has limited specificity and sensitivity in clinical application [4]. PDAC is unable to be effectively screened using imaging-based methods due to the lack of early diagnostic criteria and the low sensitivity of CT images when compared to other cancers [5]. The current treatment options for pancreatic cancer are often ineffective. Most patients have already reached the advanced stage at the time of diagnosis [6]. Approximately 80% of patients with pancreatic cancer lose the opportunity for surgical resection [7]. As a result, the discovery of specific markers for diagnosis, prognosis, and effective treatment are crucial in improving the survival rate of patients with PDAC.
In tumor research, genes that promote the occurrence and development of cancer were often screened in gene expression profiles as the oldest functional genomics method [8–10]. Researchers have identified a variety of mRNA and lncRNA signatures as indicators of survival in the field of pancreatic cancer. Yan et al identified a four-gene signature (LYRM1, KNTC1, IGF2BP2, and CDC6) significantly associated with the progression and prognosis of pancreatic cancer [11]. More recently, Chen et al discovered a seven-gene signature (S100A16, PPP3CA, SEMA3C, PLAU, IL18, GDF11, and NR0B1) for prognosis of PDAC patients [12]. However, the prediction of gene function from co-expression may lead to inaccurate results. Because gene expression is measured at the level of mRNA, protein synthesis and degradation are ignored, resulting in fundamental differences between mRNA levels and protein expression [13,14]. Current mRNA markers have low specificity and predict outcomes incorrectly. Therefore, protein expression profiling is superior to mRNA expression profiling for the prediction of gene function [15]. In the Clinical Proteomic Tumor Analysis Consortium (CPTAC), proteomic technologies are applied to clinical samples to accelerate our understanding of the molecular basis of cancer [16,17]. As well as providing prognostic information, proteomic data can provide information about tumor characteristics, prognosis, and recurrence risk, which is critical for the development of targeted treatments [18]. More proteogenomic studies continue to emerge, increasing recognition of proteomics as a non-overlapping tool for understanding cancer [19].
In this study, we investigated the molecular subtyping of PDAC tumor samples based mainly on global proteomic expression from PDAC patients by CPTAC to identify relationships between multi-omics expression and clinical phenotypes, including tumor stage and grade, to determine potential prognostic biomarkers and investigate their immune infiltration in PDAC.
Material and Methods
PATIENT DATA ACQUISITION:
The quantitative proteomic data and clinical information of PDAC were obtained from CPTAC Data Portal (https://proteomics.cancer.gov/programs/cptac). The CPTAC used proteomics techniques by mass spectrometry to analyze tumor biological samples, quantify and identify each sample, and characterize the protein composition and the protein group [20]. Our analysis utilized genomic and proteomic data to identify and characterize the full range of proteins found in clinically staged (AJCC 8th Edition) PDAC tissues and to identify potential biomarkers of PDAC. The samples of tumor stages I–II (n=78) and tumor stages III–IV (n=47) of the CPTAC database were extracted after excluding PDAC patients with no clinical stage, tumor histological grade, survival time, and proteins with missing values of relative abundance among all samples.
DIFFERENTIALLY EXPRESSED PROTEINS (DEPS) IDENTIFICATION:
For the proteomic data from CPTAC, batch correction, quantile normalization, and batch normalization were performed using ‘Limma’ package and ‘impute’ package of R language (version 4.1.2). Differentially expressed protein cutoff criteria were set as a |log2Fold Change| >0 and an adjusted
IDENTIFICATION OF HUB GENES:
The Search Tool for the Retrieval of Interacting Genes/Proteins (version 11.5; https://string-db.org/) is a system used to identify the interactions and functional associations between DEPs. We used it to draw the protein–protein interaction (PPI) network diagram of the DEPs to analyze the specific relationship between proteins and diseases [21]. The PPI analysis result was imported into Cytoscape software (version 3.9.1) to identify and visualize hub proteins with a degree >6 in the network of key modules using the CytoHubba plug-in [22].
FUNCTIONAL ENRICHMENT ANALYSIS:
Gene Ontology (GO) analysis was performed using R packages clusterProfiler, org.Hs.eg.db, enrichplot, and ggplot2 to determine enrichment in Biological Processes (BP), Cellular Components (CC), and Molecular Functions (MF) [23]. P value <0.05 indicated a statistically significant difference.
KAPLAN-MEIER (KM) PLOTTER ANALYSIS:
Kaplan-Meier (KM) plotter was used to estimate the effect of various genes or proteins on prognosis in different cancer types (
CORRELATION ANALYSIS OF IMMUNE CELL INFILTRATION:
Through the use of the Xiantao tool (
RECEIVER OPERATING CHARACTERISTIC (ROC) ANALYSIS:
To evaluate the possibility of hub genes acting as biomarkers, we conducted ROC analysis and the ROC curve was plotted using the R package ggplot2. In addition, the area under the curve (AUC) was calculated using the R package pROC.
STATISTICAL ANALYSIS:
The analysis was performed using R language (version 4.1.2) and SPSS software (version 26.0). Our criteria for selecting differentially expressed proteins were |log2FC| >0 and an adjusted
Results
IDENTIFICATION OF DEPS IN PDAC PATIENTS AND PPI NETWORK CONSTRUCTION:
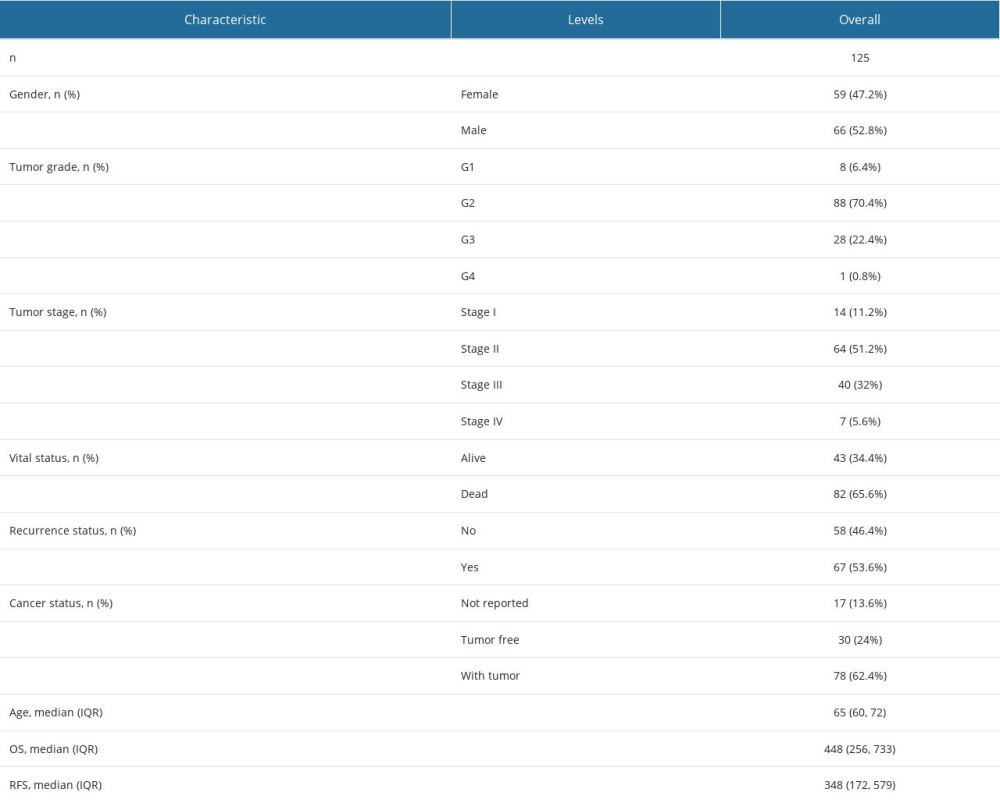
The baseline characteristics of the PDAC patients in this study were summarized (Table 1). A total of 378 differentially expressed proteins have been identified from CPTAC. We established a PPI network of differentially expressed proteins (Figure 1A). The PPI network contained 200 nodes and 324 edges. Based on the rankings of DEPs by the degree of connectivity, the cytoHubba plug-in of Cytoscape software was used to select the top 21 proteins (HRAS, STAT1, PLG, FGA, C3, COPS5, EPB42, FGG, FYN, HDAC2, ITGB3, SLC4A1, SMAD4, AHSP, IFIT3, IRF3, PRKDC, RPA2, SMAD3, SPTA1, XPA) and display the network with neighbors generated by topological analysis of degree (Figure 1B). The PPI bar chart was then generated (Figure 1C).
FUNCTIONAL ENRICHMENT ANALYSIS OF DEPS:
To investigate the role of the 378 DEPs in tumors, we performed a GO analysis. A total of 378 DEPs were identified with the following biological processes: wound healing, transition metal ion homeostasis, sterol biosynthesis, and blood coagulation regulation, and the results indicated that the DEPs were mainly associated with the following cellular contents: platelet alpha granules, cortical cytoskeletons, vesicle lumens, and secretory granules. Furthermore, these DEPs displayed molecular activities such as peptidase inhibitor activity, and endopeptidase regulator activity (Figure 2A–2C).
PLG, COPS5, EPB42, ITGB3, SLC4A1, SPTA1 WERE CORRELATED NEGATIVELY WITH OS WHILE FYN AND IRF3 WERE POSITIVELY CORRELATED WITH OS:
Survival information was collected from the CPTAC database, and we found 8 survival-related proteins (PLG, COPS5, EPB42, FYN, ITGB3, SLC4A1, IRF3, SPTA1) by Kaplan-Meier analysis (all
Additionally, low expression of FYN was associated with poor OS (HR=0.62; 95% CI, 0.39–0.98) for PDAC patients. The low expression of IRF3 was associated with poor OS (HR=0.42; 95% CI, 0.25–0.71) for PDAC patients (Figure 3A). Notably, only COPS5 was associated with OS (P<0.05) and RFS (P<0.05) (Figure 3B), while the others were associated with OS (P<0.05) but not significantly with RFS (P>0.05). Among the 8 genes, PLG, COPS5, EPB42, ITGB3, SLC4A1, and SPTA1 were negatively correlated with OS, and FYN and IRF3 were significantly associated with longer survival of patients with PDAC.
PLG, COPS5, FYN, ITGB3, IRF3, AND SPTA1 SERVED AS INDEPENDENT PROGNOSTIC AND DIAGNOSTIC FACTORS OF PATIENTS WITH PDAC:
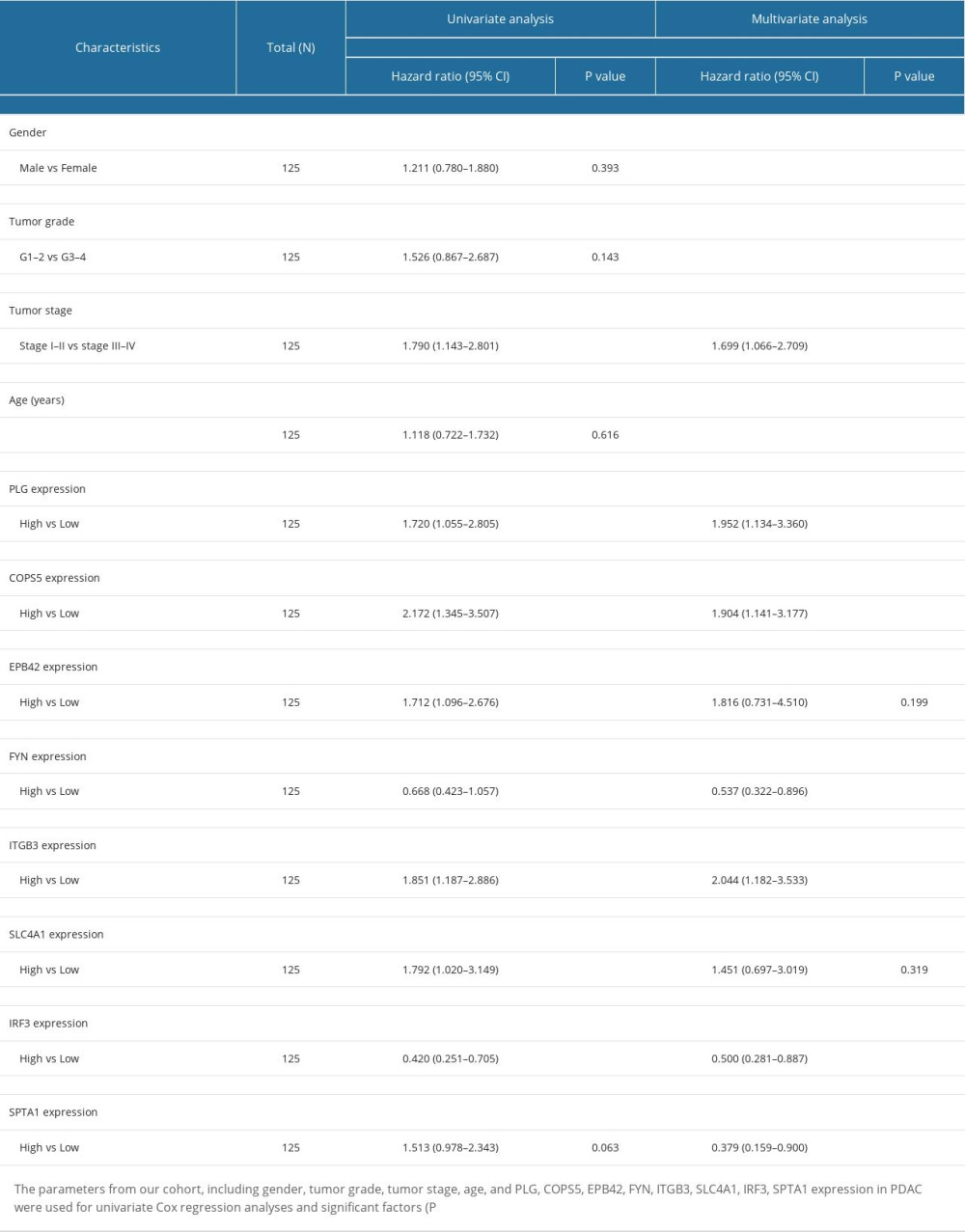
A univariate and multivariate analysis was performed to determine the effects of the 8 survival-related proteins and clinicopathological factors on survival in PDAC patients. As shown in Table 2, univariate analysis showed Tumor Stage, PLG, COPS5, EPB42, ITGB3, SLC4A1, and IRF3 expression levels were significantly correlated with OS (P=0.011, 0.030, 0.002, 0.018, 0.007, 0.042, and 0.001, respectively). Multivariate analysis was also performed to assess the parameters that significantly correlated with survival in the univariate analysis. The results showed that Tumor Stage (HR=1.699; 95% CI, 1.066–2.709; P=0.026), PLG (HR=1.952; 95% CI, 1.134–3.360; P=0.016), COPS5 (HR=1.904; 95% CI, 1.141–3.177; P=0.014), FYN (HR=0.537; 95% CI, 0.322–0.896; P=0.017), ITGB3 (HR=2.044; 95% CI, 1.182–3.533; P=0.010), IRF3 (HR=0.500; 95% CI, 0.281–0.887; P=0.018), and SPTA1 (HR=0.379; 95% CI, 0.159–0.900; P=0.028) were the independent prognostic factors for OS.
Next, the ROC analysis revealed that PLG (AUC: 0.832), COPS5 (AUC: 0.955), FYN (AUC: 0.956) (Figure 4A), ITGB3 (AUC: 0.955), IRF3 (AUC: 0.872), and SPTA1 (AUC: 0.756) had diagnostic value and could serve as biomarkers for the prognosis of PDAC (Figure 4B).
PLG, COPS5, FYN, ITGB3, IRF3, AND SPTA1 ARE ASSOCIATED WITH IMMUNE CELL INFILTRATION IN PATIENTS WITH PDAC:
To explore whether there was a correlation between immune cell infiltration into the tumors and hub differential proteins expression, we analyzed the relationships between the hub proteins signatures and 4 important immune cell types (B cells, CD8+ T cells, macrophages, and NK cells). We observed that the expression of PLG had a positive correlation with CD8+ T cells, NK cells (P<0.05) (Figure 5A), COPS5 had a negative correlation with macrophages and NK cells (P<0.05) (Figure 5B), FYN had a positive correlation with B cells, CD8+ T cells, macrophages, and NK cells (P<0.05) (Figure 5C), ITGB3 had a positive correlation with B cells, CD8+ T cells, macrophages, and NK cells (P<0.05) (Figure 5D), IRF3 had a negative correlation with macrophages and NK cells (P<0.05) (Figure 5E), and SPTA1 had a positive correlation with B cells, CD8+ T cells, and macrophages (P<0.05) (Figure 5F). These data suggest that PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 play specific roles in immune infiltration.
PLG, COPS5, FYN, ITGB3, IRF3, AND SPTA1 AFFECT THE PROGNOSIS OF PDAC PATIENTS BY ACTING ON IMMUNE CELL INFILTRATION:
Since PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 expressions are significantly correlated with immune infiltration and prognosis in PDAC, we investigated whether PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 expression affect the prognosis of PDAC because of immune infiltration. We performed prognosis analyses based on the expression levels of PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 in PDAC in related immune cell subgroups. The results demonstrated that PDAC patients with high expression of PLG and enriched infiltration of NK cells had a good prognosis (P<0.05) (Figure 6A). COPS5 up-regulation was found to be associated with poor prognosis in PDAC patients with enriched NK cells and decreased macrophages, regardless of whether B cells or CD8+ T cells were enriched or decreased (P<0.05) (Figure 6B).
A poor prognosis was observed in patients with PDAC who had low expression of FYN and decreased infiltration of B cells or CD8+ T cells, and enriched macrophages, regardless of whether NK cells had been enriched or decreased (P<0.05) (Figure 6C). Up-regulation of ITGB3 in PDAC patients with enriched CD8+ T cells, NK cells, or reduced macrophages was associated with poor outcomes (P<0.05) (Figure 6D). PDAC patients with high expression of IRF3 and enriched infiltration of macrophages or decreased CD8+ T cells had a good prognosis (P<0.05) (Figure 6E). In patients with PDAC who expressed high levels of SPTA1 and had an enriched infiltration of CD8+ T cells, the prognosis was poor (P<0.05) (Figure 6F). According to these findings, PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 can impact the prognosis of patients with PDAC through immune infiltration.
Discussion
PDAC remains the most common type of pancreatic cancer, with the highest incidence and mortality rates all over the world [24]. To the best of our knowledge, there are at present no reliable and accurate predictive tools for PDAC prognosis [25]. Recently, bioinformatics has been widely used to identify genes associated with cancer prognosis [26]. As a complex disease, cancer involves not only DNA alterations, but also protein expression and modification [27]. Furthermore, protein, as a more direct manifestation of the life activities of cells, organs, and even the human body, may be an ideal research target for the development of high-efficiency diagnostic or prognostic biomarkers [28]. Through our study, we have identified important proteins that cause and suppress the occurrence of PDAC, as well as the ability to predict its recurrence from a proteomic perspective.
Our studies identified that PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 served as prognostic biomarkers and are potential targets of immunotherapy for PADC patients. Furthermore, ROC curves showed that PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 exhibited value diagnostic efficiency for PDAC. The CPTAC has also generated comprehensive proteomic and mass spectrometry-based data for most cancers as a result of the accumulation of proteomic information in public databases [29], which provides more feasibility to do this kind of research.
PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 protein expression and its prognostic value have not been fully studied in PDAC. PLG encodes a serine protease called plasminogen, which is located on the cell surfaces and is essential for the degradation of extracellular matrices, cell migration, inflammation, oncogenesis, and metastasis [30]. PLG was found to be a prognostic protective factor in HCC [31,32] and a potential biomarker for prediction of papillary thyroid carcinoma [33], but there has been no report of it in PDAC. COPS5 (also known as CSN5) is the fifth component of the COP9 signalosome complex and affects a variety of cellular processes [34]. It is also an oncoprotein in a variety of human cancers [35,36]. CSN5 promotes the invasion and metastasis of pancreatic cancer by stabilization of FOXM1 [37]. Additionally, our study clearly showed that high COPS5 expression levels are positively correlated with poor survival rates in patients with PDAC. FYN is a non-receptor tyrosine kinase that belongs to the Src family kinases (SFKs), which under normal physiological conditions plays a role in signal transduction pathways in the nervous system as well as T lymphocyte development and activation [38]. FYN activity regulates cell proliferation and apoptosis, which affects pancreatic cancer metastasis [39]. ITGB3 is involved in intracellular communication via extracellular vesicles, which may play a critical role in cancer metastasis [40]. ITGB3 has been found to be overexpressed in many types of cancer cells, including gastric cancer, breast cancer, and colorectal cancer [40–42]. We demonstrated its essential role in metastatic progression of PDAC. Interferon regulatory factors (IRFs) play vital roles in immune response and tumor cellular biological processes [43]. It is consistent with our findings that pancreatic cancer patients with low IRF3 levels have significantly shorter overall survival [44]. SPTA1 mutations have been linked to hereditary elliptocytosis and hereditary spherocytosis, a set of congenital hemolytic syndromes, as well as being associated with hepatitis D virus infection and Mongolian liver cancer [45]. According to our data, PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 are effective in predicting the prognosis and diagnosis of patients with PDAC.
The effect of immunotherapy is closely correlated with immune infiltration, making it one of the most promising treatments for cancer [46]. A better understanding of immune infiltration into pancreatic tumors, as well as the identification of novel immune-related biomarkers, may prove useful in the development of immunotherapy. There appears to be a prognostic benefit associated with B cells or at least their subsets within PDAC tissues, suggesting a role for humoral immunity in the antitumor response by the body [47,48]. Rong et al reported cytotoxic T cells, including CD8+ T cells and NKT cells, predict prolonged OS only in samples overexpressing targets for pyroptosis or ferroptosis induction [49]. Recent studies showed that tumor-associated macrophages promote carcinogenesis and progression and metastasis of pancreatic cancer via different mechanisms [50,51]. Based our research, it appears that PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 are potential promising immunotherapeutic targets for patients with PDAC. A new prognostic signature based on DEPs in PDAC was proposed in our study. However, there are a few limitations of our study. Firstly, this study was constructed based on a public database, and more prospective clinical studies are required to verify the accuracy of this study. Furthermore, we need more in vitro and in vivo experiments to confirm the value of PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 proteins.
Conclusions
We conducted the first gene co-expression analysis using proteomic data from PDAC samples, and found that PLG, COPS5, FYN, IRF3, ITGB3, and SPTA1 had prognostic value and could be used in designing prognostic systems in the future, which may provide novel insights for the selection of immunotherapeutic targets and prognostic biomarkers for PDAC.
Figures
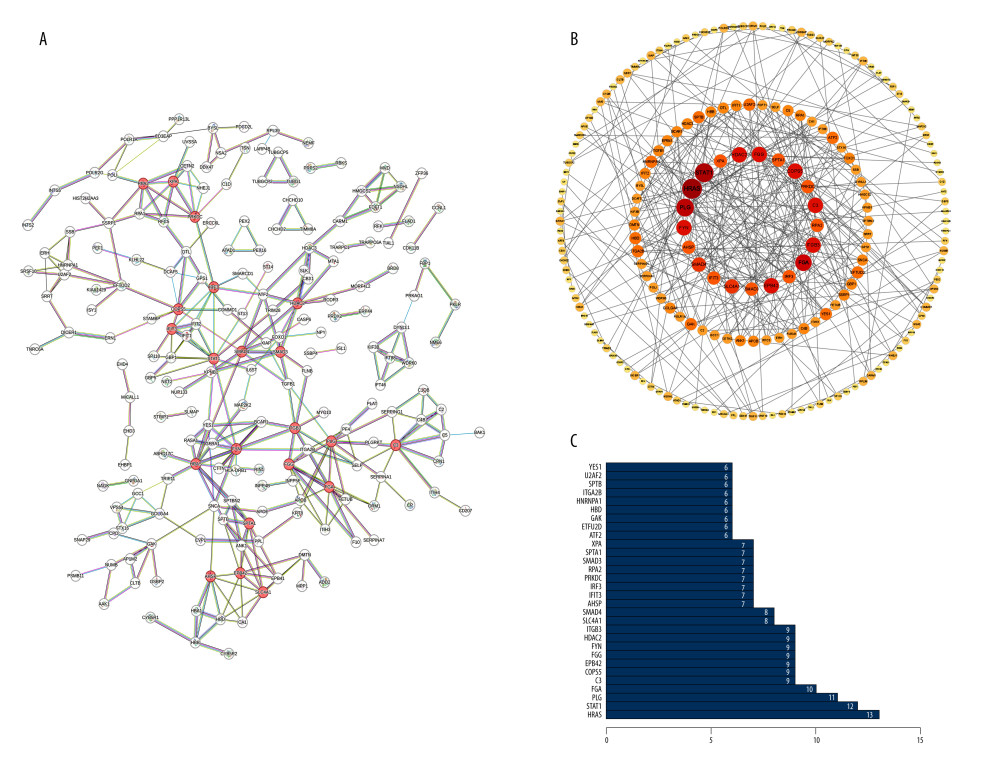 Figure 1. Differentially expressed proteins (DEPs) of PDAC network construction and Enrichment analysis of DEPs. (A) Protein–protein interaction (PPI) network of 378 DEPs, with top 21 proteins being shown in red (STRING, version 11.5, https://string-db.org/). (B) The top 21 proteins with the highest degree values were identified using CytoHubba (Cytoscape, version 3.9.1, https://cytoscape.org/). These proteins were ranked in descending degree order from red to orange to yellow. (C) The predicted association rank (from low to high) of the top 30 proteins in the PPI network (R software, version 4.1.2, USA).
Figure 1. Differentially expressed proteins (DEPs) of PDAC network construction and Enrichment analysis of DEPs. (A) Protein–protein interaction (PPI) network of 378 DEPs, with top 21 proteins being shown in red (STRING, version 11.5, https://string-db.org/). (B) The top 21 proteins with the highest degree values were identified using CytoHubba (Cytoscape, version 3.9.1, https://cytoscape.org/). These proteins were ranked in descending degree order from red to orange to yellow. (C) The predicted association rank (from low to high) of the top 30 proteins in the PPI network (R software, version 4.1.2, USA). 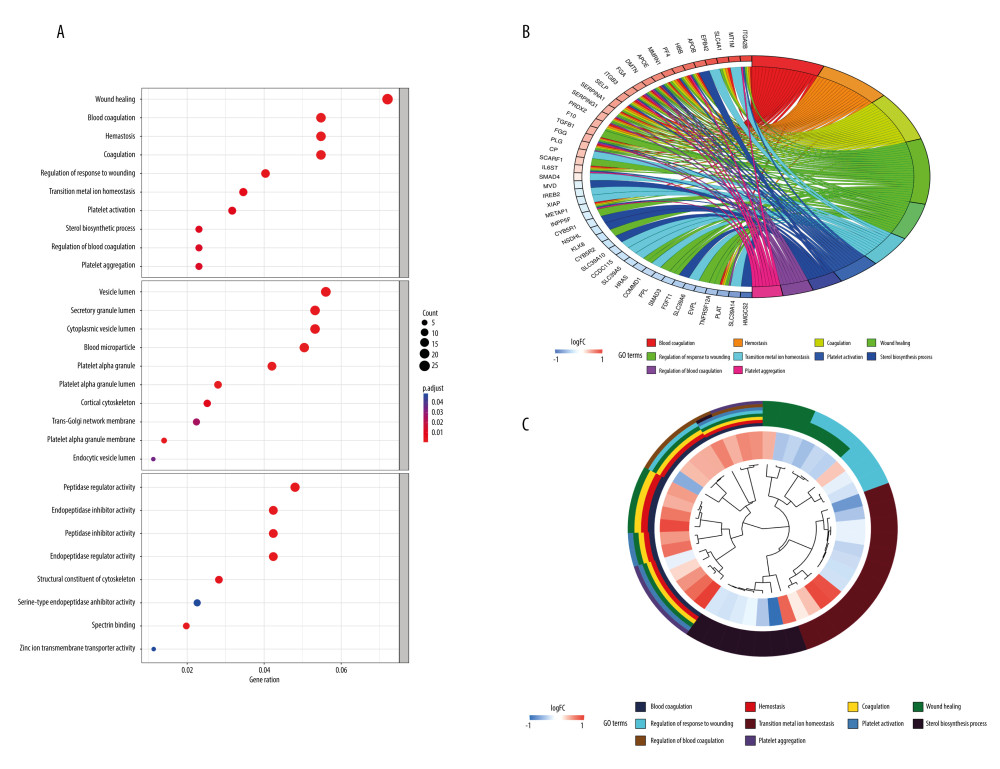 Figure 2. Functions of the identified DEPs using Gene Ontology (GO) enrichment analysis. (A) The functions of the 378 DEPs identified cover 3 main categories: biological process (BP), cellular composition (CC), and molecular function (MF). (B) Chord plot shows the top 10 most enriched GO. (C) Cluster plot shows the top 10 most enriched GO (R software, version 4.1.2, USA).
Figure 2. Functions of the identified DEPs using Gene Ontology (GO) enrichment analysis. (A) The functions of the 378 DEPs identified cover 3 main categories: biological process (BP), cellular composition (CC), and molecular function (MF). (B) Chord plot shows the top 10 most enriched GO. (C) Cluster plot shows the top 10 most enriched GO (R software, version 4.1.2, USA). 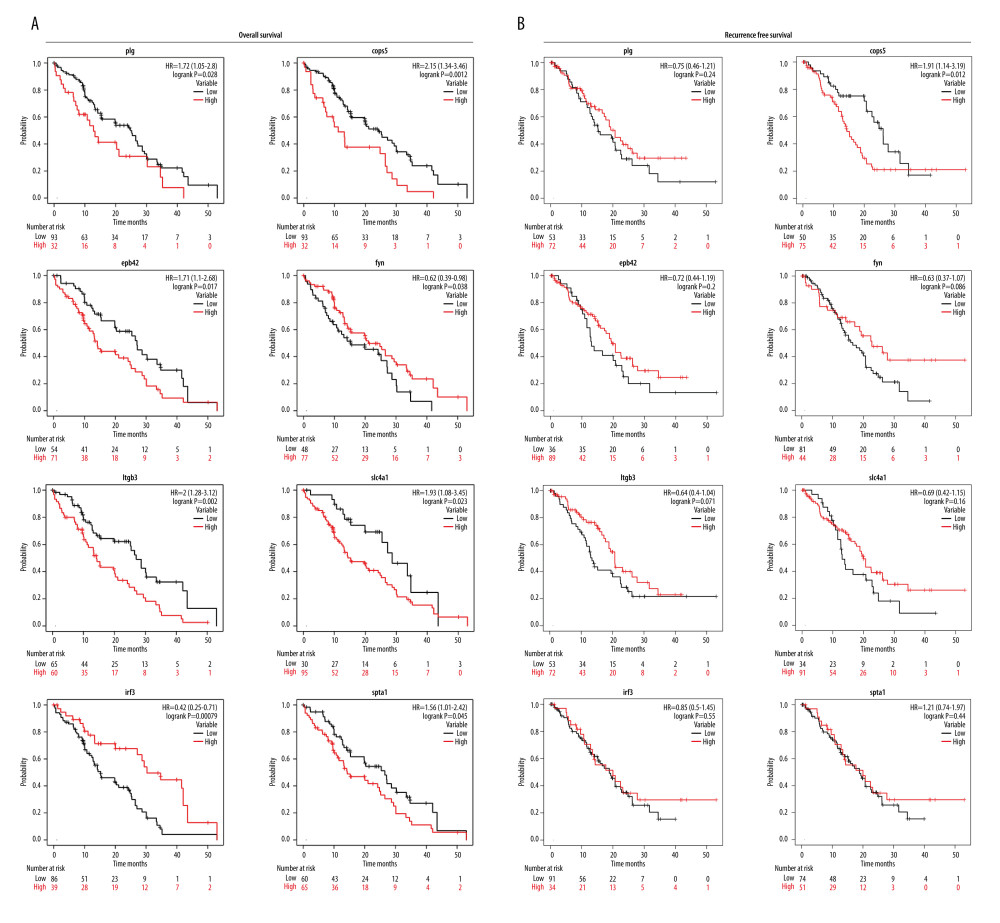 Figure 3. Kaplan-Meier (KM) survival curves analysis for hub proteins expression. (A) Based on PLG, COPS5, EPB42, FYN, ITGB3, SLC4A1, IRF3, and SPTA1 expression in PDAC samples, survival curves of the overall survival and (B) recurrence-free survival were constructed using KM plotter (KM plotter, 2022.08.23, https://kmplot.com/analysis/).
Figure 3. Kaplan-Meier (KM) survival curves analysis for hub proteins expression. (A) Based on PLG, COPS5, EPB42, FYN, ITGB3, SLC4A1, IRF3, and SPTA1 expression in PDAC samples, survival curves of the overall survival and (B) recurrence-free survival were constructed using KM plotter (KM plotter, 2022.08.23, https://kmplot.com/analysis/). 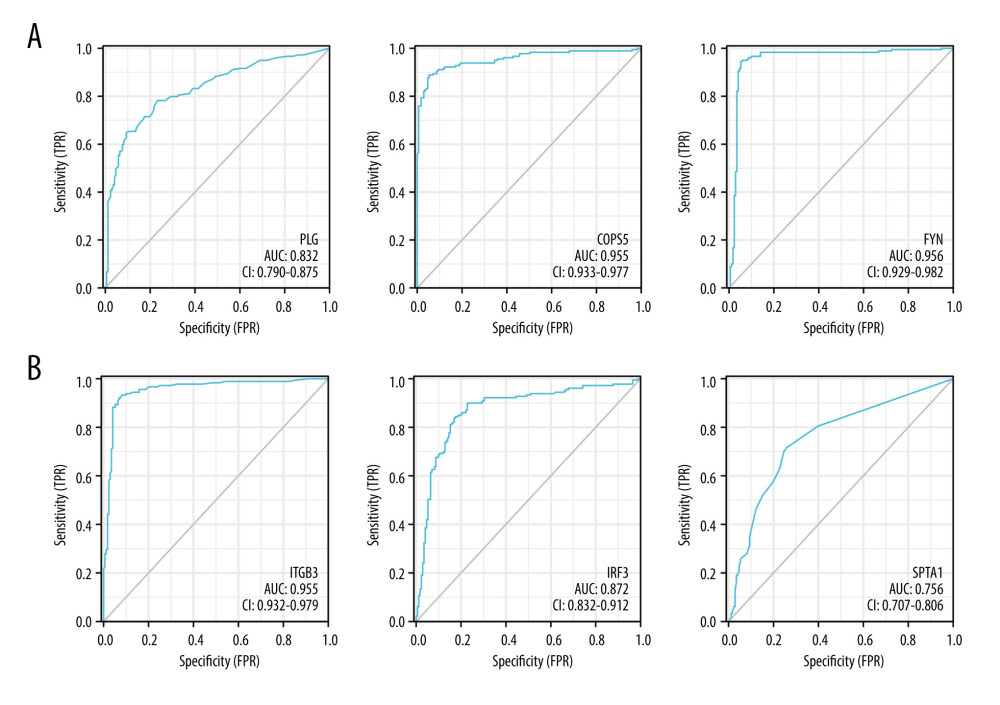 Figure 4. Receiver operating characteristics (ROC) curve analysis revealed the diagnostic value for PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 in PDAC. (A) The ROC curve showed that the AUC value of PLG, COPS5, and FYN were 0.832, 0.955, and 0.956, respectively. (B) The ROC curve showed that the AUC value of ITGB3, IRF3, SPTA1 were 0.955, 0.872, and 0.756, respectively (R software, version 4.1.2, USA).
Figure 4. Receiver operating characteristics (ROC) curve analysis revealed the diagnostic value for PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 in PDAC. (A) The ROC curve showed that the AUC value of PLG, COPS5, and FYN were 0.832, 0.955, and 0.956, respectively. (B) The ROC curve showed that the AUC value of ITGB3, IRF3, SPTA1 were 0.955, 0.872, and 0.756, respectively (R software, version 4.1.2, USA). 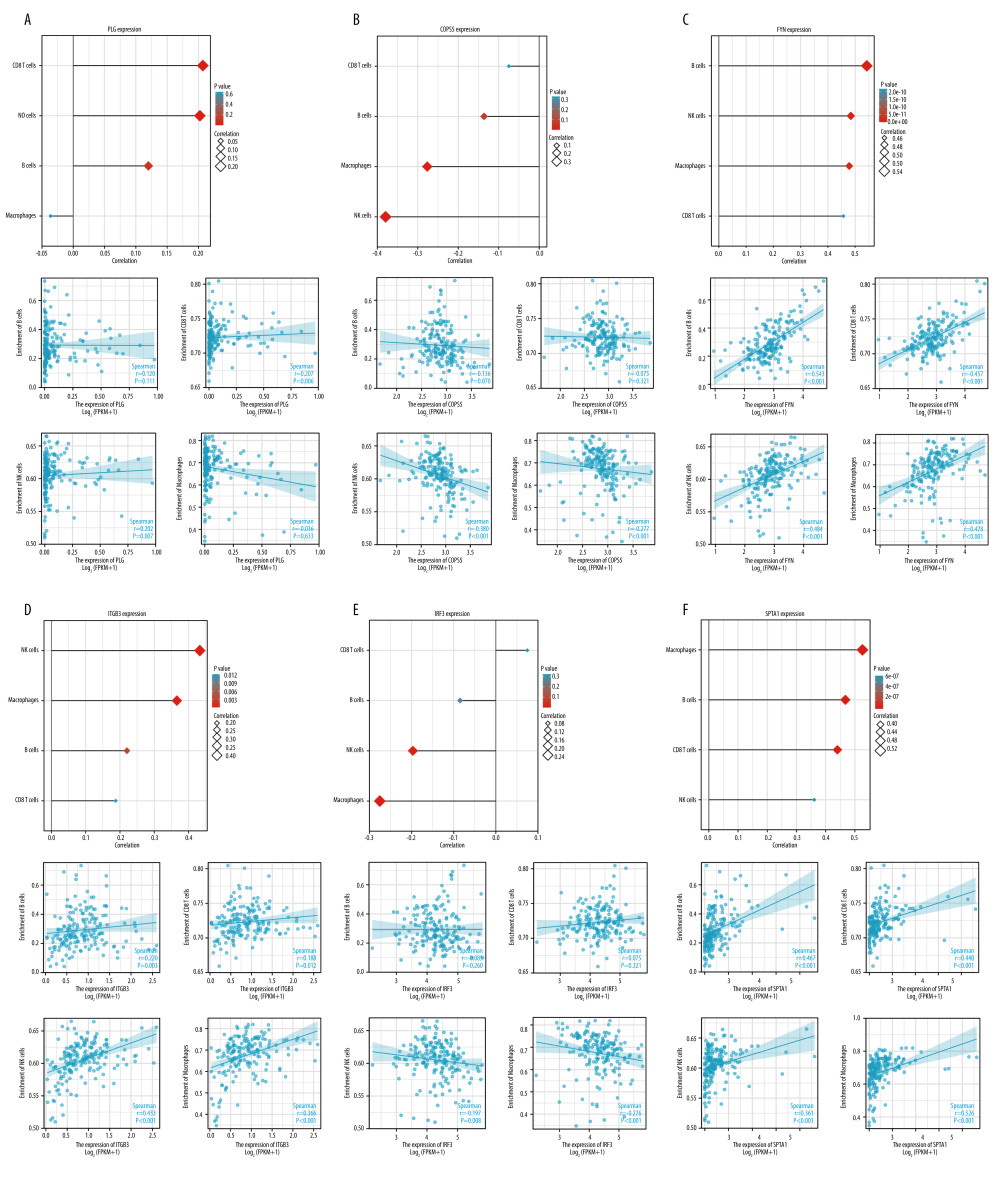 Figure 5. PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 expression and the immune level of correlation. (A) The relationships between PLG expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK), cells and macrophages) in PDAC. (B) The relationships between COPS5 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (C) The relationships between FYN expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (D) The relationships between ITGB3 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (E) The relationships between IRF3 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (F) The relationships between SPTA1 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC (Xiantao tool, version 1.0.0, https://www.xiantao.love).
Figure 5. PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 expression and the immune level of correlation. (A) The relationships between PLG expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK), cells and macrophages) in PDAC. (B) The relationships between COPS5 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (C) The relationships between FYN expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (D) The relationships between ITGB3 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (E) The relationships between IRF3 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (F) The relationships between SPTA1 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC (Xiantao tool, version 1.0.0, https://www.xiantao.love). 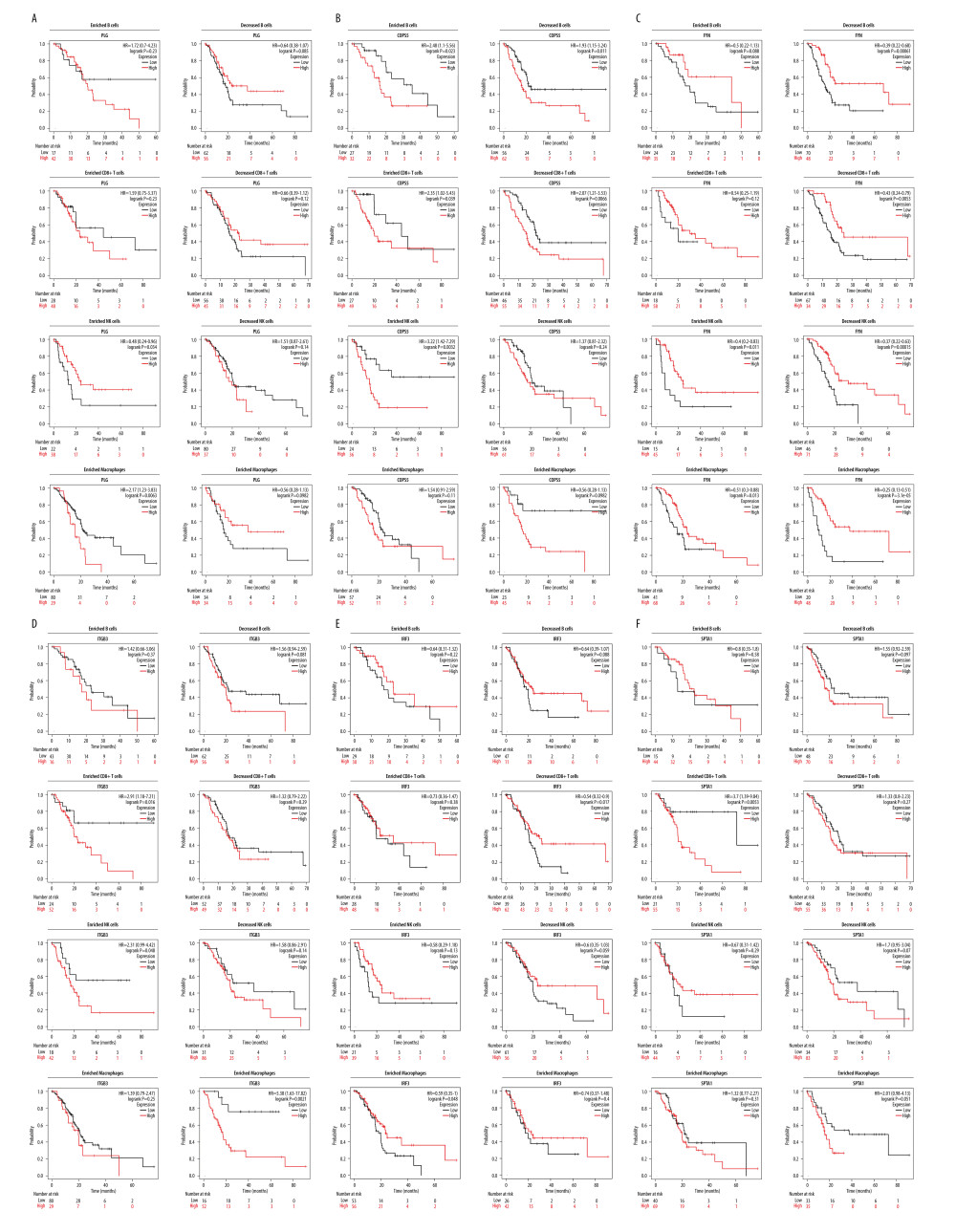 Figure 6. Kaplan-Meier survival curves according to high and low expression of PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 in immune cell subgroups in PDAC, and their diagnostic value in PDAC. (A) Kaplan-Meier survival analysis of the high and low groups of PLG expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (B) Kaplan-Meier survival analysis of the high and low groups of COPS5 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (C) Kaplan-Meier survival analysis of the high and low groups of FYN expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (D) Kaplan-Meier survival analysis of the high and low groups of ITGB3 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (E) Kaplan-Meier survival analysis of the high and low groups of IRF3 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (F) Kaplan-Meier survival analysis of the high and low groups of SPTA1 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages (KM plotter, 2022.08.23, https://kmplot.com/analysis/).
Figure 6. Kaplan-Meier survival curves according to high and low expression of PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 in immune cell subgroups in PDAC, and their diagnostic value in PDAC. (A) Kaplan-Meier survival analysis of the high and low groups of PLG expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (B) Kaplan-Meier survival analysis of the high and low groups of COPS5 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (C) Kaplan-Meier survival analysis of the high and low groups of FYN expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (D) Kaplan-Meier survival analysis of the high and low groups of ITGB3 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (E) Kaplan-Meier survival analysis of the high and low groups of IRF3 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (F) Kaplan-Meier survival analysis of the high and low groups of SPTA1 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages (KM plotter, 2022.08.23, https://kmplot.com/analysis/). References
1. Siegel RL, Miller KD, Fuchs HE, Jemal A, Cancer statistics, 2021: Cancer J Clin, 2021; 71(1); 7-33
2. Tempero MA, Malafa MP, Al-Hawary M, Pancreatic adenocarcinoma, version 2.2021, NCCN clinical practice guidelines in oncology: J Natl Compr Canc Netw, 2021; 19(4); 439-57
3. Pishvaian MJ, Blais EM, Brody JR, Overall survival in patients with pancreatic cancer receiving matched therapies following molecular profiling: A retrospective analysis of the Know Your Tumor registry trial: Lancet Oncol, 2020; 21(4); 508-18
4. Groot VP, Mosier S, Javed AA, Circulating tumor DNA as a clinical test in resected pancreatic cancer: Clin Cancer Res, 2019; 25(16); 4973-84
5. Singhi AD, Koay EJ, Chari ST, Maitra A, Early detection of pancreatic cancer: Opportunities and challenges: Gastroenterology, 2019; 156(7); 2024-40
6. He R, Yin Y, Yin W, Prevention of pancreatic acinar cell carcinoma by Roux-en-Y Gastric Bypass Surgery: Nat Commun, 2018; 9(1); 4183
7. Wang X, Li X, Wei X, PD-L1 is a direct target of cancer-FOXP3 in pancreatic ductal adenocarcinoma (PDAC), and combined immunotherapy with antibodies against PD-L1 and CCL5 is effective in the treatment of PDAC: Signal Transduct Target Ther, 2020; 5(1); 38
8. Schena M, Shalon D, Davis RW, Brown PO, Quantitative monitoring of gene expression patterns with a complementary DNA microarray: Science, 1995; 270(5235); 467-70
9. DeRisi JL, Iyer VR, Brown PO, Exploring the metabolic and genetic control of gene expression on a genomic scale: Science, 1997; 278(5338); 680-86
10. Hughes TR, Marton MJ, Jones AR, Functional discovery via a compendium of expression profiles: Cell, 2000; 102(1); 109-26
11. Yan X, Wan H, Hao X, Importance of gene expression signatures in pancreatic cancer prognosis and the establishment of a prediction model: Cancer Manag Res, 2019; 11; 273-83
12. Chen D, Huang H, Zang L, Development and verification of the hypoxia- and immune-associated prognostic signature for pancreatic ductal adenocarcinoma: Front Immunol, 2021; 12; 728062
13. Wang J, Ma Z, Carr SA, Proteome profiling outperforms transcriptome profiling for coexpression based gene function prediction: Mol Cell Proteomics, 2017; 16(1); 121-34
14. Liu Y, Beyer A, Aebersold R, On the dependency of cellular protein levels on mRNA abundance: Cell, 2016; 165(3); 535-50
15. Kustatscher G, Grabowski P, Schrader TA, Co-regulation map of the human proteome enables identification of protein functions: Nat Biotechnol, 2019; 37(11); 1361-71
16. Mertins P, Tang LC, Krug K, Reproducible workflow for multiplexed deep-scale proteome and phosphoproteome analysis of tumor tissues by liquid chromatography-mass spectrometry: Nat Protoc, 2018; 13(7); 1632-61
17. Chen F, Chandrashekar DS, Varambally S, Creighton CJ, Pan-cancer molecular subtypes revealed by mass-spectrometry-based proteomic characterization of more than 500 human cancers: Nat Commun, 2019; 10(1); 5679
18. Deutsch EW, Csordas A, Sun Z, The ProteomeXchange consortium in 2017: Supporting the cultural change in proteomics public data deposition: Nucleic Acids Res, 2017; 45(D1); D1100-6
19. Papaioannou MD, Djuric U, Kao J, Proteomic analysis of meningiomas reveals clinically distinct molecular patterns: Neuro Oncol, 2019; 21(8); 1028-38
20. Edwards NJ, Oberti M, Thangudu RR, The CPTAC Data Portal: A resource for cancer proteomics research: J Proteome Res, 2015; 14(6); 2707-13
21. Szklarczyk D, Gable AL, Lyon D, STRING v11: Protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets: Nucleic Acids Res, 2019; 47(D1); D607-13
22. Chin CH, Chen SH, Wu HH, cytoHubba: Identifying hub objects and sub-networks from complex interactome: BMC Syst Biol, 2014; 8(Suppl 4); S11
23. Walter W, Sánchez-Cabo F, Ricote M, GOplot: An R package for visually combining expression data with functional analysis: Bioinformatics, 2015; 31(17); 2912-14
24. Li YJ, Wu JY, Wang JM, Xiang DX, Emerging nanomedicine-based strategies for preventing metastasis of pancreatic cancer: J Control Release, 2020; 320; 105-11
25. Groot VP, Gemenetzis G, Blair AB, Defining and predicting early recurrence in 957 patients with resected pancreatic ductal adenocarcinoma: Ann Surg, 2019; 269(6); 1154-62
26. Wu H, Li S, Hu X, Associations of mRNA expression of DNA repair genes and genetic polymorphisms with cancer risk: A bioinformatics analysis and meta-analysis: J Cancer, 2019; 10(16); 3593-607
27. Wu P, Heins ZJ, Muller JT, Integration and analysis of CPTAC proteomics data in the context of cancer genomics in the cBioPortal: Mol Cell Proteomics, 2019; 18(9); 1893-98
28. Rodriguez H, Zenklusen JC, Staudt LM, The next horizon in precision oncology: Proteogenomics to inform cancer diagnosis and treatment: Cell, 2021; 184(7); 1661-70
29. Bradshaw RA, Hondermarck H, Rodriguez H, Cancer proteomics and the elusive diagnostic biomarkers: Proteomics, 2019; 19(21–22); e1800445
30. Law RH, Abu-Ssaydeh D, Whisstock JC, New insights into the structure and function of the plasminogen/plasmin system: Curr Opin Struct Biol, 2013; 23(6); 836-41
31. Wang J, Zhang Q, Shi F, A seven-gene signature to predict prognosis of patients with hepatocellular carcinoma: Front Genet, 2021; 12; 728476
32. Qu X, Zhang L, Li S, m(6)A-related angiogenic genes to construct prognostic signature, reveal immune and oxidative stress landscape, and screen drugs in hepatocellular carcinoma: Oxid Med Cell Longev, 2022; 2022; 8301888
33. Wang Y, Zhou S, Wang D, Complement C4-A and plasminogen as potential biomarkers for prediction of papillary thyroid carcinoma: Front Endocrinol (Lausanne), 2021; 12; 737638
34. Claret FX, Hibi M, Dhut S, A new group of conserved coactivators that increase the specificity of AP-1 transcription factors: Nature, 1996; 383(6599); 453-57
35. Yuan C, Wang D, Liu G, Pan Y, Jab1/Cops5: A promising target for cancer diagnosis and therapy: Int J Clin Oncol, 2021; 26(7); 1159-69
36. Pan Y, Yang H, Claret FX, Emerging roles of Jab1/CSN5 in DNA damage response, DNA repair, and cancer: Cancer Biol Ther, 2014; 15(3); 256-62
37. Mao L, Le S, Jin X, CSN5 promotes the invasion and metastasis of pancreatic cancer by stabilization of FOXM1: Exp Cell Res, 2019; 374(2); 274-81
38. Elias D, Ditzel HJ, Fyn is an important molecule in cancer pathogenesis and drug resistance: Pharmacol Res, 2015; 100; 250-54
39. Chen ZY, Cai L, Bie P, Roles of Fyn in pancreatic cancer metastasis: J Gastroenterol Hepatol, 2010; 25(2); 293-301
40. Fuentes P, Sesé M, Guijarro PJ, ITGB3-mediated uptake of small extracellular vesicles facilitates intercellular communication in breast cancer cells: Nat Commun, 2020; 11(1); 4261
41. Wei W, Yang Y, Cai J, MiR-30a-5p suppresses tumor metastasis of human colorectal cancer by targeting ITGB3: Cell Physiol Biochem, 2016; 39(3); 1165-76
42. You L, Wu Q, Xin Z, The long non-coding RNA HOXA11-AS activates ITGB3 expression to promote the migration and invasion of gastric cancer by sponging miR-124-3p: Cancer Cell Int, 2021; 21(1); 576
43. Tian M, Wang X, Sun J, IRF3 prevents colorectal tumorigenesis via inhibiting the nuclear translocation of β-catenin: Nat Commun, 2020; 11(1); 5762
44. Ren D, Qin G, Zhao J, Metformin activates the STING/IRF3/IFN-β pathway by inhibiting AKT phosphorylation in pancreatic cancer: Am J Cancer Res, 2020; 10(9); 2851-64
45. Candia J, Bayarsaikhan E, Tandon M, The genomic landscape of Mongolian hepatocellular carcinoma: Nat Commun, 2020; 11(1); 4383
46. Nishida N, Kudo M, Immune phenotype and immune checkpoint inhibitors for the treatment of human hepatocellular carcinoma: Cancers (Basel), 2020; 12(5); 1274
47. Delvecchio FR, Goulart MR, Fincham REA, B cells in pancreatic cancer stroma: World J Gastroenterol, 2022; 28(11); 1088-101
48. Michaud D, Mirlekar B, Steward C, B cell receptor signaling and protein kinase D2 support regulatory B cell function in pancreatic cancer: Front Immunol, 2021; 12; 745873
49. Tang R, Liu X, Liang C, Deciphering the prognostic implications of the components and signatures in the immune microenvironment of pancreatic ductal adenocarcinoma: Front Immunol, 2021; 12; 648917
50. Yang J, Li Y, Sun Z, Zhan H, Macrophages in pancreatic cancer: An immunometabolic perspective: Cancer Lett, 2021; 498; 188-200
51. Geng Y, Fan J, Chen L, A notch-dependent inflammatory feedback circuit between macrophages and cancer cells regulates pancreatic cancer metastasis: Cancer Res, 2021; 81(1); 64-76
Figures
 Figure 1. Differentially expressed proteins (DEPs) of PDAC network construction and Enrichment analysis of DEPs. (A) Protein–protein interaction (PPI) network of 378 DEPs, with top 21 proteins being shown in red (STRING, version 11.5, https://string-db.org/). (B) The top 21 proteins with the highest degree values were identified using CytoHubba (Cytoscape, version 3.9.1, https://cytoscape.org/). These proteins were ranked in descending degree order from red to orange to yellow. (C) The predicted association rank (from low to high) of the top 30 proteins in the PPI network (R software, version 4.1.2, USA).
Figure 1. Differentially expressed proteins (DEPs) of PDAC network construction and Enrichment analysis of DEPs. (A) Protein–protein interaction (PPI) network of 378 DEPs, with top 21 proteins being shown in red (STRING, version 11.5, https://string-db.org/). (B) The top 21 proteins with the highest degree values were identified using CytoHubba (Cytoscape, version 3.9.1, https://cytoscape.org/). These proteins were ranked in descending degree order from red to orange to yellow. (C) The predicted association rank (from low to high) of the top 30 proteins in the PPI network (R software, version 4.1.2, USA). Figure 2. Functions of the identified DEPs using Gene Ontology (GO) enrichment analysis. (A) The functions of the 378 DEPs identified cover 3 main categories: biological process (BP), cellular composition (CC), and molecular function (MF). (B) Chord plot shows the top 10 most enriched GO. (C) Cluster plot shows the top 10 most enriched GO (R software, version 4.1.2, USA).
Figure 2. Functions of the identified DEPs using Gene Ontology (GO) enrichment analysis. (A) The functions of the 378 DEPs identified cover 3 main categories: biological process (BP), cellular composition (CC), and molecular function (MF). (B) Chord plot shows the top 10 most enriched GO. (C) Cluster plot shows the top 10 most enriched GO (R software, version 4.1.2, USA). Figure 3. Kaplan-Meier (KM) survival curves analysis for hub proteins expression. (A) Based on PLG, COPS5, EPB42, FYN, ITGB3, SLC4A1, IRF3, and SPTA1 expression in PDAC samples, survival curves of the overall survival and (B) recurrence-free survival were constructed using KM plotter (KM plotter, 2022.08.23, https://kmplot.com/analysis/).
Figure 3. Kaplan-Meier (KM) survival curves analysis for hub proteins expression. (A) Based on PLG, COPS5, EPB42, FYN, ITGB3, SLC4A1, IRF3, and SPTA1 expression in PDAC samples, survival curves of the overall survival and (B) recurrence-free survival were constructed using KM plotter (KM plotter, 2022.08.23, https://kmplot.com/analysis/). Figure 4. Receiver operating characteristics (ROC) curve analysis revealed the diagnostic value for PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 in PDAC. (A) The ROC curve showed that the AUC value of PLG, COPS5, and FYN were 0.832, 0.955, and 0.956, respectively. (B) The ROC curve showed that the AUC value of ITGB3, IRF3, SPTA1 were 0.955, 0.872, and 0.756, respectively (R software, version 4.1.2, USA).
Figure 4. Receiver operating characteristics (ROC) curve analysis revealed the diagnostic value for PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 in PDAC. (A) The ROC curve showed that the AUC value of PLG, COPS5, and FYN were 0.832, 0.955, and 0.956, respectively. (B) The ROC curve showed that the AUC value of ITGB3, IRF3, SPTA1 were 0.955, 0.872, and 0.756, respectively (R software, version 4.1.2, USA). Figure 5. PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 expression and the immune level of correlation. (A) The relationships between PLG expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK), cells and macrophages) in PDAC. (B) The relationships between COPS5 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (C) The relationships between FYN expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (D) The relationships between ITGB3 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (E) The relationships between IRF3 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (F) The relationships between SPTA1 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC (Xiantao tool, version 1.0.0, https://www.xiantao.love).
Figure 5. PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 expression and the immune level of correlation. (A) The relationships between PLG expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK), cells and macrophages) in PDAC. (B) The relationships between COPS5 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (C) The relationships between FYN expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (D) The relationships between ITGB3 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (E) The relationships between IRF3 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC. (F) The relationships between SPTA1 expression and infiltrating immune cells (B cells, CD8+ T cells, natural killer (NK) cells, and macrophages) in PDAC (Xiantao tool, version 1.0.0, https://www.xiantao.love). Figure 6. Kaplan-Meier survival curves according to high and low expression of PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 in immune cell subgroups in PDAC, and their diagnostic value in PDAC. (A) Kaplan-Meier survival analysis of the high and low groups of PLG expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (B) Kaplan-Meier survival analysis of the high and low groups of COPS5 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (C) Kaplan-Meier survival analysis of the high and low groups of FYN expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (D) Kaplan-Meier survival analysis of the high and low groups of ITGB3 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (E) Kaplan-Meier survival analysis of the high and low groups of IRF3 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (F) Kaplan-Meier survival analysis of the high and low groups of SPTA1 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages (KM plotter, 2022.08.23, https://kmplot.com/analysis/).
Figure 6. Kaplan-Meier survival curves according to high and low expression of PLG, COPS5, FYN, ITGB3, IRF3, and SPTA1 in immune cell subgroups in PDAC, and their diagnostic value in PDAC. (A) Kaplan-Meier survival analysis of the high and low groups of PLG expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (B) Kaplan-Meier survival analysis of the high and low groups of COPS5 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (C) Kaplan-Meier survival analysis of the high and low groups of FYN expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (D) Kaplan-Meier survival analysis of the high and low groups of ITGB3 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (E) Kaplan-Meier survival analysis of the high and low groups of IRF3 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages. (F) Kaplan-Meier survival analysis of the high and low groups of SPTA1 expression in patients with PDAC based on the number of tumor-infiltrating B cells, CD8+ T cells, NK cells, and macrophages (KM plotter, 2022.08.23, https://kmplot.com/analysis/). In Press
21 Mar 2024 : Meta-Analysis
Economic Evaluation of COVID-19 Screening Tests and Surveillance Strategies in Low-Income, Middle-Income, a...Med Sci Monit In Press; DOI: 10.12659/MSM.943863
10 Apr 2024 : Clinical Research
Predicting Acute Cardiovascular Complications in COVID-19: Insights from a Specialized Cardiac Referral Dep...Med Sci Monit In Press; DOI: 10.12659/MSM.942612
06 Mar 2024 : Clinical Research
Enhanced Surgical Outcomes of Popliteal Cyst Excision: A Retrospective Study Comparing Arthroscopic Debride...Med Sci Monit In Press; DOI: 10.12659/MSM.941102
06 Mar 2024 : Clinical Research
Prevalence and Variation of Medical Comorbidities in Oral Surgery Patients: A Retrospective Study at Jazan ...Med Sci Monit In Press; DOI: 10.12659/MSM.943884
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952