13 August 2020: Animal Study
Expression of Fibroblast Growth Factor 4 in a Rat Model of Polydactyly of the Thumb Induced by Cytarabine
Xin Zhao1ABCDEF, Wentao Yang1ABCDEF, Guancong Li1BF, Haoran Dong1BF, Jianlun Hou1CF, Zhiwei Cao1CF, Dehong Guan1ABCDEFG*DOI: 10.12659/MSM.926953
Med Sci Monit 2020; 26:e926953
Abstract
BACKGROUND: The aim of this study was to assess the expression and mechanisms of fibroblast growth factor 4 in polydactyly of the thumb induced by cytarabine.
MATERIAL AND METHODS: Rats were intraperitoneally injected with cytarabine at different gestation periods (12.5 days, 13.5 days, and 14.5 days) to establish a polydactyly of the thumb model. Then, the expression of FGF4 in polydactyly was studied by whole-mount in situ hybridization. We used hematoxylin & eosin stain and cartilage stain to investigate the development of the skeleton and tissues in the embryo. Pictures were taken to determine the general shape of the deformity, then X-rays were taken to detect bone distortion of the rats born with a congenital malformation.
RESULTS: In the experimental group (11.5 days, 12.5 days, 13.5 days, and 14.5 days), whole-mount in situ hybridization showed that the FGF4 expression at the tip of the embryonic limb bud was significantly increased compared with the control group and FGF4 was distributed in a wider range and lasted longer than in the control group (P<0.01). HE staining and cartilage staining showed that there was an extra metacarpal bone and a phalanx in the rats with polydactyly of the thumb (P<0.01). Images of the deformed limbs showed polydactyly and syndactyly of the thumb in the rats. Further X-ray examination revealed 1 extra metacarpal bone and 1 extra phalanx.
CONCLUSIONS: Cytarabine can induce polydactyly and syndactyly of the thumb in rats. In this process, cytarabine can induce the expression of FGF4 on the tip of the embryonic limb bud, which further leads to abnormal development of the embryonic limb bud and eventually causes a congenital deformity.
Keywords: Cytarabine, Fibroblast Growth Factor 4, Polydactyly, Embryo, Mammalian, Thumb
Background
FGFs are proteins from pituitary glands in cows, which can be found in most tissues throughout the human body [1,2]. The fibroblast growth factor (FGF) family consists of 23 members. Human FGF19 is a homologue to mouse FGF15, and is grouped into 7 subfamilies based on differences in sequence homology and phylogeny [3]. Members of the FGF family are structurally similar, with approximately 14% sequence homology and a core region containing conserved amino acid sequences and structural motifs [4]. The biological activity of FGFs is achieved by binding to fibroblast growth factor receptors (FGFRs), which contain 4 members: FGFR1, FGFR2, FGFR3, and FGFR4. FGF receptors (FGFRs) exist on the surface of FGFs-sensitive cells and can be bind to FGFs to exert their effects [5]. FGF family members can promote mitosis and cell survival, and are involved in various biological processes such as cell proliferation and differentiation, embryonic development, and morphogenesis. Any irregularities in their function can lead to a range of developmental defects.
The initial stage of limb bud development is the proliferation of somatopleuric mesenchyme. Apical ectodermal ridge (AER) is a structure formed by ectodermal cells at the distal end of the limb bud and acts as the primary signaling center to ensure proper limb development.The AER and the limb mesenchyme, including the polarized active region (ZPA), communicate with each other to guide the further development of the limb [6]. In normal limb development, the expression of
Cytarabine can combine cytosine bases with arabinose sugar. It is similar enough to human deoxycytosine nucleotide to incorporate into human DNA, but not enough to kill cells. The way it works is because it is rapidly converted to cytarabine triphosphate, which destroys DNA during the S phase of the cell cycle (DNA synthesis). Thus, the mitotic phase of cells that require DNA replication is most affected [15]. It is a chemotherapy medication used to treat acute myeloid leukemia (AML), chronic myelogenous leukemia (CML), and acute lymphocytic leukemia (ALL). Cytarabine has many adverse effects and can cause harm to the fetus if used during pregnancy, and it is one of the first chemotherapeutic drugs identified with teratogenic effects [16,17]. Cytarabine is known to induce ectrodactyly in rodents, and FGF is involved in this process [18]. Therefore, we tried to use cytarabine to make a limb deformity model in rats. Fortunately, in the initial experiments, we found that cytarabine induced polydactyly of the thumb in rats and was very stable. As mentioned earlier, limb development is driven by a positive feedback loop including FGF4 [11]. Based on these studies, we hypothesized that cytarabine-induced thumb polydactyly might be related to changes of FGF4 in the limb bud, and we investigated this. The results showed that cytarabine could increase the expression of FGF4 in the limb bud, and the increase of FGF4 expression was significantly associated with thumb polydactyly in the rat embryo. The mechanism underlying congenital malformations is still unclear [19,20], and the purpose of the present study was is to explore the mechanism of congenital polydactyly and make some contributions to the further study of this deformity.
Material and Methods
ANIMAL EXPERIMENT AND EMBRYO COLLECTION:
The animals used in the experiment were provided by the Medical Experimental Animal Center. Rats were fed standard particles in the laboratory, housed at 22±2°C with a 12-h/12-h light-dark cycle. The study was approved by the Animal Experimental Ethics Committee with ethics approval number SYDW2018-007. All operations are carried out in accordance with the Guidelines for Animal Research.
We established an animal model for congenital polydactyly. First, we selected adult male and female Sprague-Dawley rats (8–10 weeks old; mean SD weight, 200–250g) and raised them in the same cage. Rats were checked for pregnancy every morning. Under a microscope, pregnancy was determined by the presence of sperm on the vaginal smear showed, and the day on which the pregnancy was confirmed was recorded as day 0. After pregnancy, the females were housed separately from males. The female rats were intraperitoneally injected with cytarabine at a dose of 100 mg/kg on the morning of the 11th day of pregnancy.
GROUPING:
In this experiment, the rats were divided into the cytarabine dose group and the control group, and the pregnant rats at 11.5, 12.5, 13.5 and 14.5 days were divided into 4 groups, with 8 pregnant rats in each group, so there were 8 groups in total. The embryos of pregnant rats were dissected and the limbs of the embryos were removed for analysis.
:
We dissected the pregnant rats under sterile conditions, cleaned the embryolemma, and then put the embryos in ice-cold DEPC-treated PBS. We transferred the cut embryos to 4% paraformaldehyde and fixed them overnight at 4°C. On the second day, we washed the sample twice with PBT at 4°C, then dehydrate it with 25%, 50%, 75%, and 100% formaldehyde gradients, then washed the sample repeatedly in reverse order to hydrate the sample, which makes the sample base low and the coloration clear. After bleaching with 6% hydrogen peroxide for 15 min and digesting the sample for 15 min at 10 μg/ml proteinase K, the digestion was then stopped with 2 mg/ml glycine and samples were washed 3 times with PBT. Prehybridization was carried out for 3 h at 65°C, followed by overnight hybridization at 70°C. On the third day, samples were washed 3 times with 2×SSC containing 50% formamide at 65°C, and then digested with 20 mg/L RNase at 37°C for 1 h. This was repeat ed4 times with 2×SSC for 20 min each time. The blocking solution was then blocked at 37°C for 3–4 h, and we then added biotinylated rat anti-digoxigen dropwise at 4°C overnight. On the fourth day, we washed the sample 6 times with PBT, and added SABC dropwise at 37°C for 30 min. After washing thoroughly, we added biotinylated peroxidase dropwise at 37°C for 30 min. We soaked the sample in the configured DAB developer to color development for 20–25 min in the dark. After the color development effect is satisfactory, we washed the embryo with PBS for 10 min or longer to terminate the reaction. We applied Re-fix with 4% paraformaldehyde and 0.2% glutaraldehyde in PBT for 20 min, followed by washing 3 times with PBS and treating with 50% glycerol for 30 min. Finally, put the sample in 80% glycerol and froze it at −20°C. All samples were photographed.
HE STAINING:
The tissue block was taken and fixed with 4% paraformaldehyde, embedded in paraffin, and sliced at 2 μm. We dewaxed the sections in xylene for 5 to 10 min, then transferred them to a mixture of xylene and pure alcohol (1: 1) for about 5 min, followed by exposure to gradient alcohol (100%, 95%, 85%, and 70%) for 2 to 5 min and transfer to dye liquor by distilled water. Sumujing dyeing solution staining was performed for 5 to 15 min, followed by washing away the excess dye on the slide with 0.5~1% hydrochloric acid (70% alcohol). Microscopic examination was performed until the chromatin in the nucleus and nucleus was clear (about 10 s). Slides were rinsed with running water for 15 to 30 min. They quickly alkalinize or turn blue in a saturated solution of lithium carbonate. Then, slides were briefly washed again with distilled water and dyed with 0.1% to 0.5% hematoxylin-eosin stain for 1 to 5 min. If coloring is difficult, added 1 to 2 drops of glacial acetic acid per 100 ml of the dye solution to make it easy to color and not easily decolorize. Dehydration was performed using 70%, 85%, 95%, and 100% alcohol for 2 to 3 min. In the concentration of alcohol below 95%, eosin is easily discolored, so the exposure time was shortened appropriately. Xylene is transparent (secondary) for about 10 min. We wiped away excess xylene around the slice, avoided drying, quickly added neutral gum, and then covered the slide with a cover slip. Staining was observed under a microscope.
CARTILAGE STAINING:
We dissolved Ainsin blue in 70% alcohol to form a 0.3% solution, dissolved alizarin red in 95% alcohol to form a 0.1% solution, added glacial acetic acid and 70% alcohol, and mixed them in a ratio of 1: 1: 1: 1. On day 22, we removed the deformed limbs of embryonic rats. The limbs were fixed in 95% alcohol for at least 1 day. We immersed the limbs in the staining solution for 2 days at 37°C, followed by bleaching in a mixture of 1% KOH and 20% glycerin for 2 days, and then limbs were stored in 100% glycerin. We observed the bone morphology of the embryonic rat limb under a microscope.
IMAGING ANALYSIS:
After the successful establishment of the rat congenital thumb polydactyly model, the deformed limbs were photographed, and then X-ray films of all the newborn rats were obtained to observe the polydactyly of the thumb on their first day of life, including analyses of the bone tissue of the excess finger. The X-ray shooting parameters were: 50 kV, 50 mA, and 125 mS. Each X-ray film was analyzed by a radiologist, a laboratory researcher, and an orthopedist. The rats were killed by cervical dislocation after the last analysis of their X-ray films. After the radiological analysis, the rats were decapitated.
STATISTICAL ANALYSIS:
All results in this study are expressed as the mean±standard deviation (χ̄±s), and processed using Statistical Product and Service Solutions (SPSS) 21.0 software (IBM, Armonk, NY, USA). Comparison of the 2 groups was performed using the
Results
EXPRESSION OF FGF4 IN LIMB BUDS OF MALFORMED EMBRYOS:
Observed under a dissecting microscope after whole rat embryo in situ hybridization, the ends of the hind limbs showed a dark brown band. Relative regions of FGF4 expression in the experimental group and the control group at 11.5 days (Figure 1A, 1B), 12.5 days (Figure 1C, 1D), 13.5 days, and 14.5 days (Figure 1E, 1F) in the cytarabine dose group (Figure 2) were 22.21%, 84.81%, 58.43%, and 47.59%, and the expression of FGF4 in each day in the blank group. The relative areas were 6.74%, 30.80%, 19.64%, and 15.44%, respectively. Using the SPSS software for chi-square test, the significance level was 7.88 at a=0.01, while the 12.5, 13.5, and 14.5-day test statistics were 73.119, 37.623, and 25.857, both of which are greater than 7.88. The overall test statistic for the 4 days was 123.380, which was also significantly greater than the standard value at the significance level of a=0.01, and the null hypothesis of invalidation was rejected. Therefore, the administration was effective (Figure 3).
We can see from the table that the relative area of embryos expressing 12.5 days in the cytarabine dose group was larger than that of the embryos at 11.5 days and 13.5 days, indicating that the peak of FGF4 expression was at 12.5 days. FGF4 began to express at 11.5 days in the blank group, and the expression increased at 12.5 days, that is, the relative area occupied by the brown band became larger, and the area at 13.5 days did not have a relative area of 12.5 days, which was similar to that of the drug-administered group. The blank group of 12.5 days is also a peak of FGF4 expression. In the embryos of 11.5 days, 12.5 days, 13.5 days, and 14.5 days with FGF4 expression on both sides of the limb, on the dorsal axis, and on the anterior-posterior axis, it can be seen that the drug-administered group is larger than the blank group and the distribution of the belts was wider.
CHANGES IN BONE TISSUE AND MORPHOLOGY:
When the rat embryos mature, HE staining (Figure 4A) and cartilage staining (Figure 4B) were used to observe the bone tissue, showing obvious bone tissue of the redundant deformed finger. The polydactyly and syndactyly of the thumb can also be observed through the appearance of the deformed limb (Figure 4C) and the X-ray image (Figure 4D).
Discussion
Congenital limb deformity is one of the most common birth defects in humans, which not only affects the appearance, shape, and motor function of the limbs, but also makes patients lose the ability to work. The etiology of congenital malformation is not completely clear, and there are no effective prevention and treatment measures. There are many causes of human birth defects, of which about 2–3% are caused by drugs [21]. In recent years, research on the molecular mechanism of congenital limb deformity has intensified. The importance of the fibroblast growth factor family in limb development is well known, but the specific role of its various factors in the family and its mechanism of action have not been fully studied [22,23]. A study showed that FGF4 plays an important role in early embryonic development. FGF4 is expressed in the AER of limb buds, and the signal from AER can maintain the viability of the mesoderm cells under it and cause its proliferation, while FGF4 indirectly affected the development of embryonic limbs [24]. Cytosine arabinoside is a well-known inhibitor of DNA synthesis, but it also inhibits DNA and RNA polymerases and nucleotide reductases required for DNA synthesis [25].
If not used properly, cytosine arabinoside will eventually lead to changes in gene expression during embryonic development, thus affecting cell proliferation, differentiation and apoptosis [26]. Further, we investigated the expression of FGF4 in the congenital thumb polydactyly caused by cytarabine. In the mesenchyme of the AER region there is a feedback loop formed by the
In the present experiments, we successfully produced a stable rat model of hind limb thumb polydactyly. The teratogenic rate of pregnant rats injected with cytarabine was 85%, and the deformation can be visualized by osteochondral staining and X-ray imaging. The results of
Therefore, it can be inferred that the increase in the expression of FGF4 causes malformation in the limbs. Use of drug treatment to inhibit the expression of FGF4 can avoid the thumb polydactyly caused by cytarabine. Our results provide a reliable guide for clinical drug use. This study also has some limitations. For example, although the results suggest that increased expression of FGF4 is associated with limb deformities caused by cytarabine, the specific molecular although FGF4 plays an important role, other factors may act together to lead to the disease, and further research is needed.
Conclusions
This experiment directly confirmed the teratogenic effect of cytarabine, which was relatively stable, and indirectly proved that cytarabine-induced congenital thumb polydactyly in rats was associated with increased expression of FGF4. In this process, cytarabine can induce the expression of FGF4 on the tip of embryonic limb bud, which further leads to abnormal development of embryonic limb bud and eventually causes congenital deformity in rats.This experiment provides an
Figures
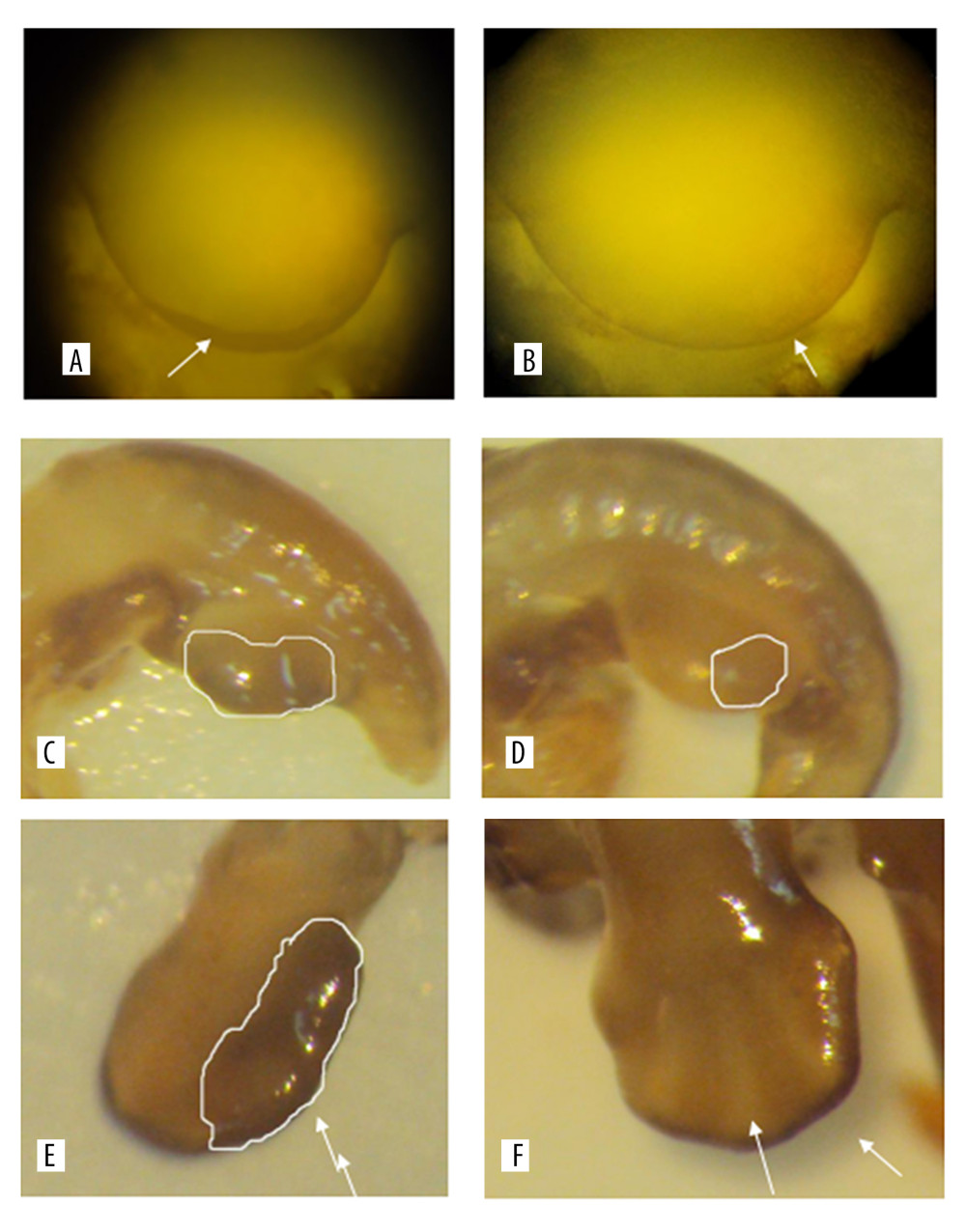 Figure 1. A and B are the results of the 11.5 days whole rat embryos in situ hybridization of the experimental group and the control group. C and D are the results of 12.5 days whole rat embryos in situ hybridization of the experimental group and the the control group. E and F are the results of the 14.5 days whole rat embryos in situ hybridization of the experimental group and the the control group.
Figure 1. A and B are the results of the 11.5 days whole rat embryos in situ hybridization of the experimental group and the control group. C and D are the results of 12.5 days whole rat embryos in situ hybridization of the experimental group and the the control group. E and F are the results of the 14.5 days whole rat embryos in situ hybridization of the experimental group and the the control group. 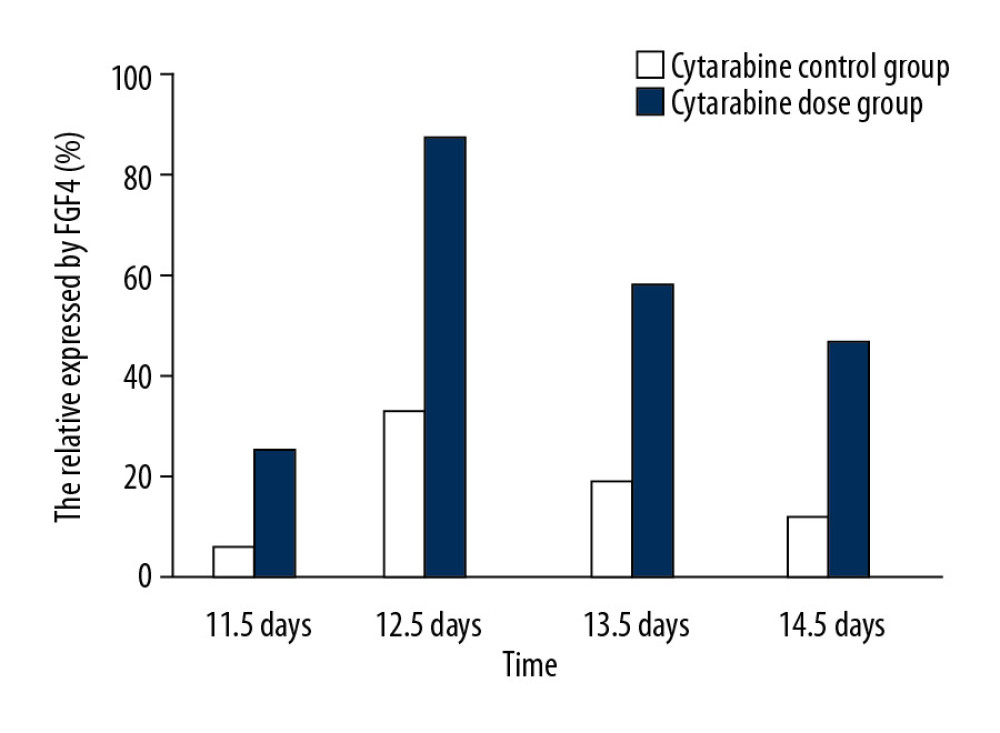 Figure 2. The relative area of FGF4 expression in embryonic limb of rats in cytarabine dose group and control group.
Figure 2. The relative area of FGF4 expression in embryonic limb of rats in cytarabine dose group and control group. 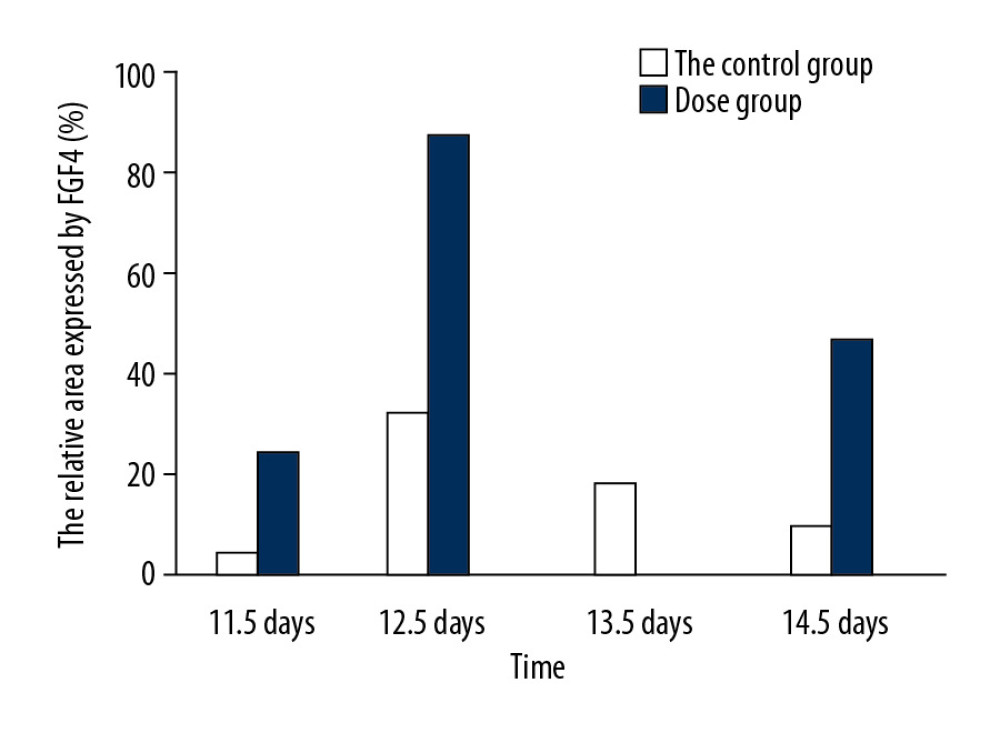 Figure 3. Statistical analysis of the expression of FGF4.
Figure 3. Statistical analysis of the expression of FGF4. 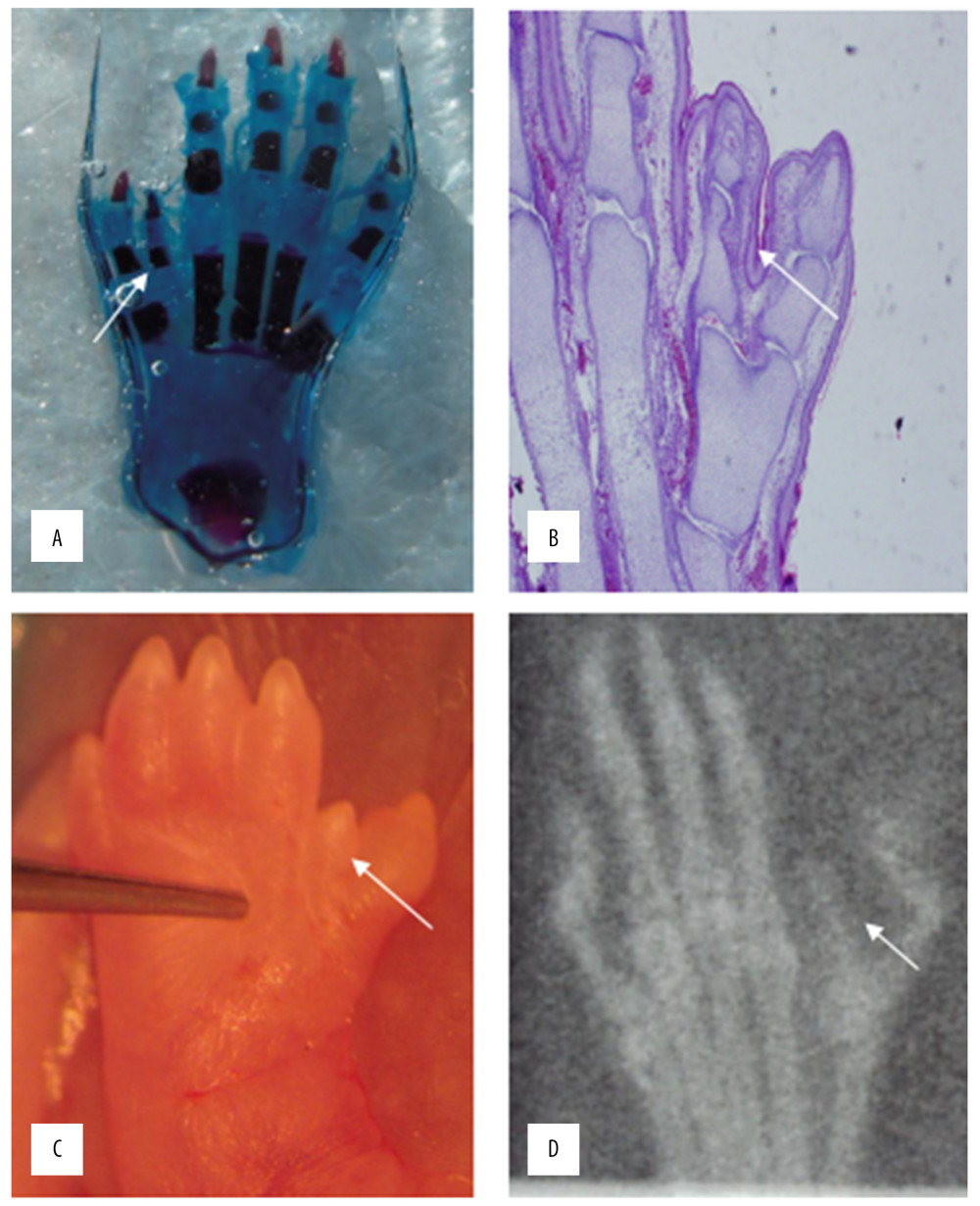 Figure 4. HE staining (A) bone cartilage staining (B) the picture of the deformed hand (C) X-ray (D). All those images of the thumb polydactyly rats were used to observe limb morphology and newborn redundant bone.
Figure 4. HE staining (A) bone cartilage staining (B) the picture of the deformed hand (C) X-ray (D). All those images of the thumb polydactyly rats were used to observe limb morphology and newborn redundant bone. References
1. Armelin HA, Pituitary extracts and steroid hormones in the control of 3T3 cell growth: Proc Natl Acad Sci USA, 1973; 70(9); 2702-6
2. Blaber M, DiSalvo J, Thomas KA, X-ray crystal structure of human acidic fibroblast growth factor: Biochemistry, 1996; 35(7); 2086-94
3. Ornitz DM, Marie PJ, Fibroblast growth factor signaling in skeletal development and disease: Genes Dev, 2015; 29(14); 1463-86
4. Goldfarb M, Fibroblast growth factor homologous factors: Evolution, structure, and function: Cytokine Growth Factor Rev, 2005; 16(2); 215-20
5. Itoh N, Ornitz DM, Evolution of the Fgf and Fgfr gene families: Trends Genet, 2004; 20(11); 563-69
6. Potuijt JW, Baas M, Sukenik-Halevy R, A point mutation in the pre-ZRS disrupts sonic hedgehog expression in the limb bud and results in triphalangeal thumb-polysyndactyly syndrome: Genet Med, 2018; 20(11); 1405-13
7. Boulet AM, Moon AM, Arenkiel BR, Capecchi MR, The roles of Fgf4 and Fgf8 in limb bud initiation and outgrowth: Dev Biol, 2004; 273(2); 361-72
8. Rodrigues AR, Yakushiji-Kaminatsui N, Atsuta Y, Integration of Shh and Fgf signaling in controlling Hox gene expression in cultured limb cells: Proc Natl Acad Sci USA, 2017; 114(12); 3139-44
9. Martin GR, The roles of FGFs in the early development of vertebrate limbs: Genes Dev, 1998; 12(11); 1571-86
10. Lu P, Minowada G, Martin GR, Increasing Fgf4 expression in the mouse limb bud causes polysyndactyly and rescues the skeletal defects that result from loss of Fgf8 function: Development, 2006; 133; 33-42
11. Scherz PJ, Harfe BD, McMahon AP, Tabin CJ, The limb bud Shh-Fgf feedback loop is terminated by expansion of former ZPA cells: Science, 2004; 305(5682); 396-99
12. Verheyden JM, Sun X, An Fgf/Gremlin inhibitory feedback loop triggers termination of limb bud outgrowth: Nature, 2008; 454(7204); 638-41
13. Knudsen TB, Kochhar DM, The Hemimelic extra toes mouse mutant: Historical perspective on unraveling mechanisms of dysmorphogenesis: Birth Defects Res C Embryo Today, 2010; 90(2); 155-62
14. Feldman B, Poueymirou W, Papaioannou VE, Requirement of FGF-4 for postimplantation mouse development: Science, 1995; 267(5195); 246-49
15. Ma J, Li X, Su Y, Mechanisms responsible for the synergistic antileukemic interactions between ATR inhibition and cytarabine in acute myeloid leukemia cells: Sci Rep, 2017; 7; 41950
16. Kochhar D, Penner JD, McDay JA, Limb development in mouse embryos. II. Reduction defects, cytotoxicity and inhibition of DNA synthesis produced by cytosine arabinoside: Teratology, 1978; 18(1); 71-92
17. Wagner V, Hill J, Weaver D, Baehner R, Congenital abnormalities in baby born to cytarabine treated mother: Lancet, 1980; 316(8185); 98-99
18. Seto ML, Nunes ME, Macarthur CA, Cunningham ML, Pathogenesis of ectrodactyly in the Dactylaplasia mouse: Aberrant cell death of the apical ectodermal ridge: Teratology, 1997; 56(4); 262-70
19. Rampal V, Giuliano F, Forefoot malformations, deformities and other congenital defects in children: Orthop Traumatol Surg Res, 2020; 106(1); S115-23
20. Guo B, Lee SK, Domph NP, Polydactyly: A review: Bull Hosp Jt Dis, 2013; 71(1); 17-23
21. Wang J, Li DY, Zhang WXTrend of mortality of congenital malformation in children aged <5 years in Beijing, 2006–2015: Zhonghua Liu Xing Bing Xue Za Zhi, 2017; 38(1); 73 [in Chinese]
22. Delgado I, López-Delgado AC, Roselló-Díez A, Proximo-distal positional information encoded by an Fgf-regulated gradient of homeodomain transcription factors in the vertebrate limb: Sci Adv, 2020; 6(23); eaaz0742
23. Guerra J, Chiodelli P, Tobia C, Long-pentraxin 3 affects primary cilium in zebrafish embryo and cancer cells via the FGF system: Cancers, 2020; 12(7); 1756
24. Moon AM, Boulet AM, Capecchi MR, Normal limb development in conditional mutants of Fgf4: Development, 2000; 127(5); 989-96
25. Harrington C, Perrino FW, The effects of cytosine arabinoside on RNA-primed DNA synthesis by DNA polymerase α-primase: J Biol Chem, 1995; 270(44); 26664-69
26. Jagtap S, Meganathan K, Gaspar J, Cytosine arabinoside induces ectoderm and inhibits mesoderm expression in human embryonic stem cells during multilineage differentiation: Br J Pharmacol, 2011; 162(8); 1743-56
27. Li X, Wang C, Xiao J, Fibroblast growth factors, old kids on the new block: Semin Cell Dev Biol, 2016; 53; 155-67
28. Vogel A, Roberts-Clarke D, Niswander L: Dev Biol, 1995; 171(2); 507-20
29. Vogel A, Tickle C: Development, 1993; 119(1); 199-206
Figures
 Figure 1. A and B are the results of the 11.5 days whole rat embryos in situ hybridization of the experimental group and the control group. C and D are the results of 12.5 days whole rat embryos in situ hybridization of the experimental group and the the control group. E and F are the results of the 14.5 days whole rat embryos in situ hybridization of the experimental group and the the control group.
Figure 1. A and B are the results of the 11.5 days whole rat embryos in situ hybridization of the experimental group and the control group. C and D are the results of 12.5 days whole rat embryos in situ hybridization of the experimental group and the the control group. E and F are the results of the 14.5 days whole rat embryos in situ hybridization of the experimental group and the the control group. Figure 2. The relative area of FGF4 expression in embryonic limb of rats in cytarabine dose group and control group.
Figure 2. The relative area of FGF4 expression in embryonic limb of rats in cytarabine dose group and control group. Figure 3. Statistical analysis of the expression of FGF4.
Figure 3. Statistical analysis of the expression of FGF4. Figure 4. HE staining (A) bone cartilage staining (B) the picture of the deformed hand (C) X-ray (D). All those images of the thumb polydactyly rats were used to observe limb morphology and newborn redundant bone.
Figure 4. HE staining (A) bone cartilage staining (B) the picture of the deformed hand (C) X-ray (D). All those images of the thumb polydactyly rats were used to observe limb morphology and newborn redundant bone. In Press
11 Mar 2024 : Clinical Research
Enhancement of Frozen-Thawed Human Sperm Quality with Zinc as a Cryoprotective AdditiveMed Sci Monit In Press; DOI: 10.12659/MSM.942946
12 Mar 2024 : Database Analysis
Risk Factors of Age-Related Macular Degeneration in a Population-Based Study: Results from SHIP-TREND-1 (St...Med Sci Monit In Press; DOI: 10.12659/MSM.943140
12 Mar 2024 : Clinical Research
Preoperative Blood Transfusion Requirements for Hemorrhoidal Severe Anemia: A Retrospective Study of 128 Pa...Med Sci Monit In Press; DOI: 10.12659/MSM.943126
12 Mar 2024 : Clinical Research
Tissue Inhibitors of Metalloproteinase 1 (TIMP-1) and 3 (TIMP-3) as New Markers of Acute Kidney Injury Afte...Med Sci Monit In Press; DOI: 10.12659/MSM.943500
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








