14 December 2022: Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase Levels
Megė Černiauskienė BCDEF 1,2*, Loreta Bagdonaitė ABDE 1, Dovilė Karčiauskaitė A 1, Violeta Kvedarienė ABD 1
DOI: 10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
Abstract
BACKGROUND: In this paper we report on the link between increased tryptase levels and prevalence of allergen-specific immunoglobulin E (IgE) in patients with anaphylaxis. The main aim of the study was to assess the corelation between elevated tryptase levels and allergen-specific immunoglobulin E (IgE) in anaphylactic reactions, mastocytosis, and other types of allergic reactions.
MATERIAL AND METHODS: We enrolled 60 adult patients, aged 18-68 years (mean age 45±8 years). The subjects were divided into 3 groups (20 patients in each group) according to the serum tryptase changes: group 1 consisted of patients with anaphylaxis (serum tryptase level ≥11.4 µg/l), group 2 consisted of patients with mastocytosis (serum tryptase level ≥20.0 µg/l), and group 3 consisted of patients with other allergic reactions with no anaphylaxis as an atopic control group (serum tryptase level <11.4 µg/l). The test material was venous blood serum samples. The allergen-specific IgE assay was carried out by immunoblot. Tryptase concentration was determined by immunoenzymatic assay.
RESULTS: Allergen-specific IgE was found in 73% of all subjects: 75% in the anaphylactic group, 55% in the mastocytosis group, and 90% in the atopic control group. Polysensitization was common and was significantly different among groups. In the atopic group, there were more patients allergic to 4-7 allergens (P<0.05).
CONCLUSIONS: We did not find a strong correlation between allergen-specific IgE levels and elevated tryptase levels.
Keywords: Allergens, Anaphylaxis, Mastocytosis, Tryptases, adult, Humans, Middle Aged, Prevalence, Immunoglobulin E
Background
Allergic diseases have become a common and frequent occurrence in modern society, affecting the lives of more than one billion people. According to the newest data, up to 150 million people in Europe suffer from chronic allergic diseases, and projections show that by 2025, more than half of the population of all European Union countries will be sensitized to a particular allergen. Up to 20% of people with allergies develop a severe form of allergy [1].
Anaphylaxis is one of the most life-threatening systemic conditions, defined as an acute, sudden-onset, severe systemic hypersensitivity reaction [2]. Mast cells are the main effector cells involved in the pathological physiology of acute allergic reactions. The production of antigen-specific IgE and their subsequent fixation to high-affinity FceR receptors on mast cells and basophil membranes upon re-exposure to the allergen initiates the hypersensitivity reaction; therefore, the role of IgE is essential in the pathogenesis of anaphylactic reactions [3,4]. Mast cell activation results in the rapid release of inflammatory mediators from secretory granules. Tryptase is one of the pathogenetically important mediators. Serum tryptase is one of the most reliable markers for confirming anaphylactic reactions, diagnosing mastocytosis, and determining prognosis. Tryptase occurs in 2 forms: α (alpha) and β (beta). Both forms of tryptase are accumulated in mast cells, but α tryptase is continuously released, whereas β tryptase is released into the bloodstream only after mast cell activation [4], and both form the basal tryptase pool [5,6]. In mastocytosis, α tryptase levels are consistently elevated in the blood, and during anaphylactic reactions, high levels of β tryptase are released in mastocytosis patients and in healthy controls [4]. In the absence of any pathology, tryptase concentrations in the blood of healthy individuals vary from 1 μg/l to 15 μg/l (mean, 11.4 μg/l). A persistently elevated tryptase level (>20 μg/l) is one of the minor diagnostic criteria for mastocytosis. In this disease, the tryptase level is always above the normal range and varies among individuals. Unlike histamine, tryptase levels remain stable for several hours [7]. Basal tryptase levels vary among individuals and rarely change over the lifetime, so that elevated serum tryptase levels help to accurately identify anaphylactic shock and mastocytosis [8]. Identification of the allergens that cause anaphylactic reactions can help develop an individual treatment and prevention plan and in the targeted prescription of immunotherapy, but many allergic individuals are sensitized to several allergens (polysensitized). About 40% of people in Western countries have an exaggerated tendency to produce large amounts of IgE to various environmental allergens. Such excessive production of IgE is called atopy, which is genetic and determined by environmental factors. Atopy is an inherited tendency to produce IgE antibodies to common allergens, which are proteins. Atopy is genetically influenced due to the tendency to produce large amounts of IgE and become sensitized to allergens, usually in environments for which most people do not produce IgE in response to pathological allergens. Atopy involves a predisposition to diseases such as allergic rhinitis and development of asthma or atopic dermatitis (eczema) due to environmental factors and genetics. In this condition, the immune response is increased against common allergens. This syndrome determines the development of allergic hypersensitivity reactions. One allergic disorder increases the risk for development of another allergic disorder [9]. Atopic individuals have higher levels of total IgE in the bloodstream and a higher amount of eosinophils than healthy individuals. Based on type I hypersensitivity in the pathogenesis of reactions, atopic individuals and those with elevated tryptase levels have a higher risk of developing anaphylactic reactions. The present study aimed to critically assess the prevalence, variability, and correlation of allergen-specific IgE in the 3 study groups.
Material and Methods
INFORMED CONSENT STATEMENT:
Informed consent was obtained from all subjects involved in the study. The study was approved by the Vilnius Regional Biomedical Research Ethics Committee (approval No. 158200-17-926-430).
MATERIAL AND STUDY GROUP:
Venous blood serum samples from 60 patients were analyzed. The age range of the studied grup was 18–68 years and the mean age was 45±8.0 years. Blood samples of the patients were collected during visits to an allergologist when anaphylaxis was suspected. After the tryptase concentration results were obtained, patients were assigned to research groups. The subjects were divided into 3 groups (20 patients in each group) according to the serum tryptase changes. Group 1 consisted of patients with documented anaphylaxis, in whom blood serum tryptase concentrations exceeded the normal range (≥11.4 μg/l) in the first measurement only (within 1–2 h) and the tryptase concentration returned to the normal range after the second measurement more than 24 h after anaphylaxis. Group 2 consisted of subjects with mastocytosis and anaphylaxis symptoms, whose tryptase concentration remained elevated (≥20.0 μg/l). Group 3 consisted of atopic patients who were referred to an allergologist and had periodic allergic reactions to certain allergens; tryptase levels in this group were within the normal range, and they were regarded as a control group used to assess prevalence (serum tryptase level <11.4 μg/l). Changes in tryptase levels were used a markers of anaphylaxis and mastocytosis.
METHODS:
The concentration of tryptase in venous blood serum was determined by immunoassay (reagents – ImmunoCAP, ThermoScientific, Finland). For allergen analysis, OPTIGEN® innovative semi-automated AP 720STM, CLA® Pette chambers, OPTIGEN Panel Allergen-Specific IgE Assay reagents (Hitachi Chemical Diagnostics Inc., Japan) and MAST CLA-1™ luminescence analyzer were used, which use a qualitative reaction for the detection of allergen-specific IgE antibodies in serum. The Hitachi CLA® allergen-specific IgE assay is based on the immunoblot method. A qualitative reaction was performed to detect allergen-specific IgE antibodies in the patient’s serum using a palette of 36 mixed allergens. Classes 3 and 4 were considered clinically relevant in this study and were not counted separately due to their small difference and the potential for cross-reactions, which require more extensive testing of allergen components for interpretation.
STATISTICAL ANALYSIS:
Statistical analysis of the study data was performed using IBM SPSS-23.0 for Windows (Chicago, Illinois, USA) and Microsoft Office Excel 2016, Microsoft Corporation (Redmond, Washington, USA). Hypotheses about the independence of the 2 attributes were tested using the chi-squared (χ2) test and the
Results
STUDY GROUP ANALYSIS:
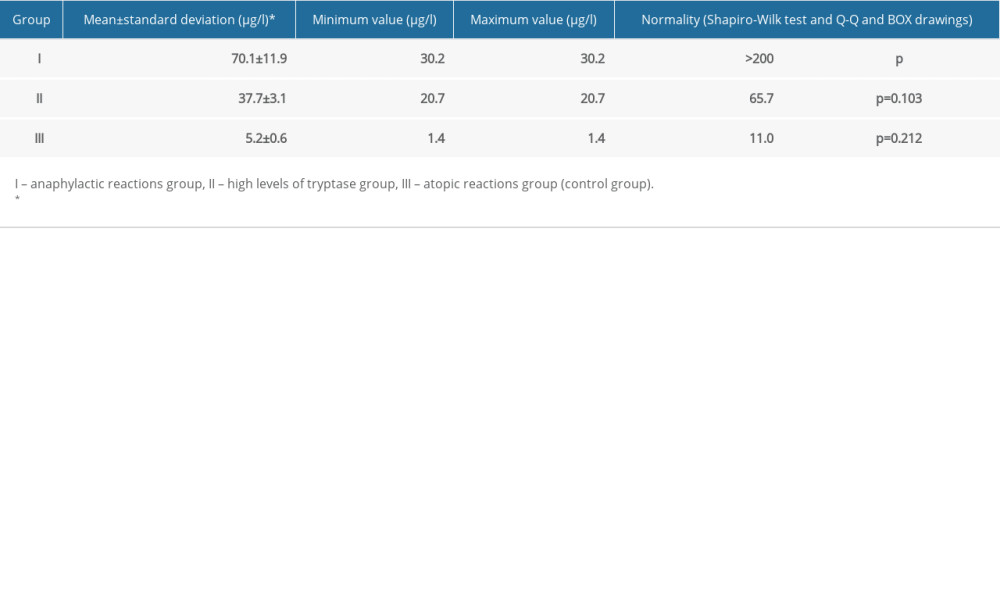
The age range of the studied grup was 18–68 years and the mean age was 45±8.0 years. Tryptase concentrations were elevated in the first 2 groups, in which subjects had elevated tryptase concentrations in anaphylaxis (episodic elevations) and mastocytosis (persistent elevations). Following group analysis by one-way ANOVA, the mean values of tryptase levels were significantly different among groups (F=23.836, P<0.05) (Table 1).
The scatter plot of tryptase concentrations by group shows that people in the anaphylactic group were significantly more likely to have higher tryptase concentrations. This distribution in the first group was due to the very high tryptase concentrations in some patients after anaphylaxis (>150 μg/l) (Figure 1).
ALLERGEN DISTRIBUTION AND ANALYSIS AMONG GROUPS:
Clinically relevant levels of allergen-specific IgE were found in 44 subjects (73.3%). The prevalence of positive reactions to at least 1 of the 36 allergens in the palette differed significantly among groups (χ2=26.250; DF=3;
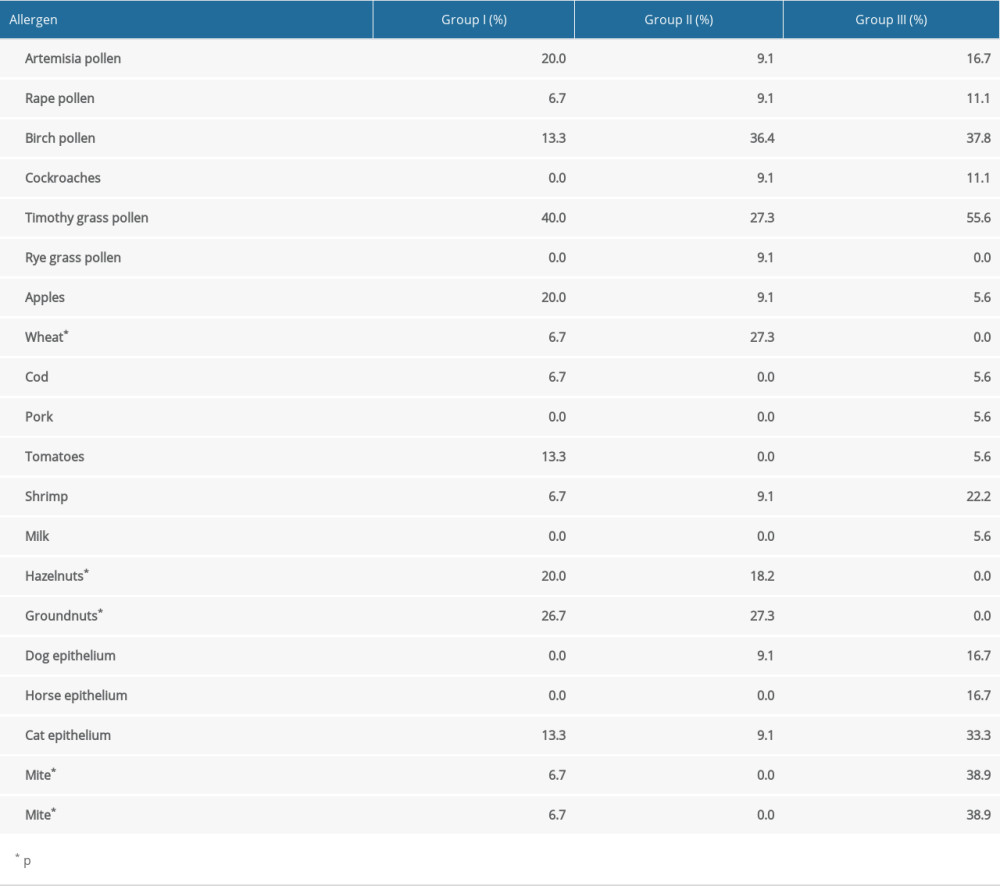
Allergies to dust mites (D. pteronyssinus and D. Farinae) were common (each 18.2%). We also found a relatively high incidence of sensitization to shrimp (13.6%), and specific IgE for shrimps was detected at a similar frequency as for mites. We found specific IgE to groundnuts and hazelnuts in the first 2 groups, and allergies to apples were also common. The least common allergies were to rye, milk, and pork (Table 2).
Most of patients were sensitized to more than 1 allergen. In the atopic group, a significantly higher number of different allergen-specific IgE per person was detected than in the other groups (χ2=16.168, DF=6, P<0.05) (Figure 2). The sensitization to any of 2 different allergens was found in all groups. Patients in the anaphylaxix group and mastocytosis group were allergic to no more than 3 allergens, in contrast to the atopic group, where 38.9% of subjects were allergic to 4–7 allergens. Allergy to 2 allergens was found in 53.3% of the anaphylaxis group and 75.0% of the mastocytosis group.
Only 20% of subjects in the anaphylaxis group and 8.3% in the mastocytosis group had a single allergen that was likely to have triggered the acute systemic reaction. Important allergen-specific IgE to peanut and hazelnut was found more common in the anaphylaxis and mastocytosis groups than in the control group (χ2=5.765, DF=2,
Of the 36 allergens in the palette, 16 allergens did not show specific IgE: nettle, Alternaria, Aspergillus, Cladosporium, Penicillium, latex, chicken, yeast, barley, yeast, oranges, beef, soybeans, peas, egg white, and hen’s feathers. In contrast, of those patients with elevated specific IgE, there were equal proportions (10 each) of inhaled and food allergens.
Analysis of our data showed that of the subjects with at least 1 allergy, inhaled allergens accounted for most of allergens identified (84.1%), while food allergens accounted for 65.9%. Specific IgE for inhalant allergens was significantly higher (90%) in the atopic group (χ2=11.140, DF=2,
A between-group prevalence analysis of allergens was performed for all 20 allergens for which allergen-specific IgE was detected. This detailed analysis showed that there were statistically significant differences in the prevalences of only 6 different allergens (Table 2).
The most common anaphylactic reactions to foods (ie, peanuts, tree nuts, and fish) were also identified in the present study. Significantly higher specific IgE for peanuts and hazelnuts (χ2=5.765, DF=2,
The atopic group had significantly higher levels of allergy to cat epithelium and both mite species (for cat epithelium: χ2=6.195, DF=2,
Discussion
Increased tryptase concentration are observed in multiple pathological conditions, but in this study, it was increased in subjects after anaphylactic reactions (episodic increase), in individuals without comorbidities, and in individuals suspected of having mastocytosis, characterized by continuously high tryptase concentrations. The aim of the study was to determine which allergens are common in subjects with elevated levels of tryptase concentration in serum. To determine their prevalence, allergen-specific IgE testing was performed. Detection and comparison of specific IgE prevalence among groups can help to determine which allergens are most common. Group analysis showed that the mean values of tryptase levels were significantly different among groups.
The severity of the anaphylactic reaction, which depends on the abundance of mediators and the physiological changes they cause in the body, as well as the clinical signs, can be measured in degrees, so that in Figure 1, the tryptase levels in the anaphylaxis and mastocytosis groups are scattered over a wide range, while in the group of atopic patients, the variation of the tryptase levels is within normal range.
Clinically relevant levels of allergen-specific IgE were found in 44 (73.3%) of the subjects. The prevalence of positive reactions to at least 1 of the 36 allergens in the palette differed significantly among groups (χ2=26.250; DF=3;
In patients with elevated allergen-specific IgE (44 or 73.3%), 6 inhaled allergens were most frequently detected, with most allergens coming from timothy grass pollen (43.2%). Birch pollen was the second (25.0%) most frequently detected allergen. The high prevalence of these inhaled allergens can be explained by cross-reactions, as both timothy grass and birch grass pollen contain many components similar to those of other allergens. Birch pollen often causes cross-reactions with fruit, nut, and vegetable allergens. Hazelnut, peanut, and apple components have the highest homology with the birch pollen component, PR-10. These proteins are very similar to the main birch pollen allergen, and cross-reactions between different IgE antibodies to PR-10 family allergens are likely, especially if serum levels of IgE to birch pollen are very high [10]. Cross-reactions between timothy grass pollen allergens usually occur in combination with other grasses, most commonly with rye pollen. The main allergens of timothy grass pollen can cross-react with pollen from various grasses, and cross-reactivity is also possible between pollen from various trees and weeds, whose components also belong to the same families [4]. Nevertheless, if the allergen causing anaphylaxis is a food product and inhalant allergens have been identified only because of cross-reactivity, they should be avoided in the same way as the true allergen, and cross-reactions are thus clinically very important. This interpretation could be applied to most of the allergens analyzed in the study but would require a separate study to show to which components specific IgE were detected [9].
Allergies to dust mites
The most common food allergens are nuts, which are one of the most common allergens that provoke an anaphylactic reaction. A person allergic to nuts is also very likely to be allergic to various inhalant allergens. Our study showed specific IgE to groundnuts and hazelnuts in the anaphylaxis and mastocytosis groups. Allergies to apples are also common due to possible cross-reactions with birch pollen and various nuts, so that a person allergic to birch pollen and nuts is very likely to also be allergic to apples. The least common allergies were to rye, milk, and pork, which were probably due to cross-reactions (Table 2). The allergy to cod was probably a non-cross-reaction. Fish is usually a strong provocative food for the development of an acute reaction [11,12].
Most of patients were sensitized to more than 1 allergen. Subjects in the anaphylaxis and mastocytosis groups were allergic to fewer allergens than subjects in the atopic group, in which most where allergic to 4–7 allergens. Most subjects in the anaphylaxis and mastocytosis groups were allergic to 2 allergens.
In 2013, Gulen et al, at the Karolinska Institute, showed that most anaphylaxis patients were sensitized to more than 1 allergen, and that one of the possible causes of polysensitization is recurrent anaphylactic reactions, which increase sensitization to allergens. It is likely that the more anaphylactic episodes there are, the more specific IgE will be detected [2].
It is clinically important to identify the main allergen that triggered the anaphylactic reaction, but the results showed that only 20% of subjects in the anaphylaxis group and 8.3% in the mastocytosis group had a single allergen that was likely to have triggered the acute systemic reaction. Allergen-specific IgE to peanut and hazelnut was more common in the anaphylaxis and mastocytosis groups than in the atopic control group (χ2=5.765, DF=2,
Of the 36 allergens in the tested palette, the allergens that did not show specific IgE were not included in statistical analysis. Analysis of our data showed that of the subjects with at least 1 allergy, inhaled allergens accounted for most of the allergens identified. Although the amount of food allergen-specific IgE was lower, the anaphylaxis cohort and the literature suggest that more attention should be paid to food allergens.
In our study, we found sensitization to certain allergens, but we do not know the exact factor that triggered the anaphylactic reaction in the clinical history, so it cannot be excluded that the finding of specific IgE for food allergens may have been a result of a cross-reaction to some extent.
Peanuts, tree nuts, and fish were identified as a most common anaphylactic reaction triggers, whereas allergy to wheat pollen, which was more common in the mastocytosis group, is not a common allergen in anaphylactic reactions, but according to the cross-reaction map, it may be involved in such reactions [14]. The wheat pollen component belongs to the group of lipid transfer proteins. Some of the peanut and hazelnut components also belong to the same group of proteins [14,15]. These groups of allergens/proteins are resistant to heating and digestion, which means that they are associated with immediate systemic reactions. Therefore, the wheat pollen-specific IgE found in the study was probably due to this. Inhaled allergens are not triger factors in anaphylactic reactions, but probably only contribute to the development of this reaction.
Gulen et al also showed that of 32 anaphylactic mastocytosis patients, 70% were allergic to pollen (birch, various grasses, and calendula), and 57% were allergic to the fur of animals (cats, dogs, and horses). Atopic subjects were sensitized mainly to inhaled allergens, but serum IgE specific to these allergens was not detected more frequently in the mastocytosis group than in the atopic group. Symptoms of allergic reactions in patients with mastocytosis may be more difficult to control when several conditions are concurrently present, such as atopy and haematologic disease [16].
Acute anaphylactic reactions can be triggered by a group of allergens rather than a single allergen, especially in patients with mastocytosis. Based on these results, a full and complete patient history should be used to assess acute reactions in patients with mastocytosis. The identification of allergens likely to cause an anaphylactic reaction should not be confirmed until it is correlated with the patient’s history [10,17,18].
To identify the causative agent of anaphylactic reactions, it is important to select the most likely allergens for testing. The identification of allergens causing acute reactions is a complex investigation, especially when individuals are at risk. For example, in our study, mastocytosis patients are such a group, which requires a more detailed analysis, as acute allergy-like reactions in their situation may be caused not only by specific IgE, but also by various exogenous or endogenous factors independent of IgE, which must be taken into account in the absence of allergen-specific IgE.
Conclusions
The prevalence of allergen-specific IgE was as follows: allergen-specific IgE was found in 73.0% of all subjects, in 90.0% of atopic patients, in 75.0% of patients with anaphylactic reactions, and in 55.0% of patients with mastocytosis. The most commonly detected specific IgE to inhaled allergens were timothy grass (43.2%), birch (25.0%), cat epithelium (20.5%), dust mites (
Regarding differences in allergen-specific IgE prevalence among study groups, sensitization was most commonly detected to 2 different allergens and was significantly different among groups: 53.3% in the anaphylactic reactions group, 75.0% in the mastocytosis group, and 33.3% in the atopic control group. The atopic group had significantly more patients (38.9%) who were allergic to 4–7 allergens.
In the group of atopic patients, specific IgE was more frequently detected to inhalable allergens (90.0%) and specific IgE to food allergens in a group of patients with anaphylactic reactions (70.0%). Specific IgE to peanuts and hazelnuts was significantly more common in the anaphylaxis and mastocytosis groups, while specific IgE to wheat was most commonly detected in patients with mastocytosis. Specific IgE to inhaled allergens (eg, cat epithelium, dust mites) was significantly more common in atopic patients.
A person who has already had an anaphylactic reaction is at risk of experiencing it again; thorough testing is needed to prevent this. The results of this study suggest that there is no strong correlation between allergen-specific IgE levels and elevated tryptase levels, and that each individual’s tryptase levels and sensitization to certain allergens are highly variable and different, so each patient should be examined individually, with detailed studies of tryptase levels, allergen-specific IgE, allergen components, and other provoking factors.
Figures
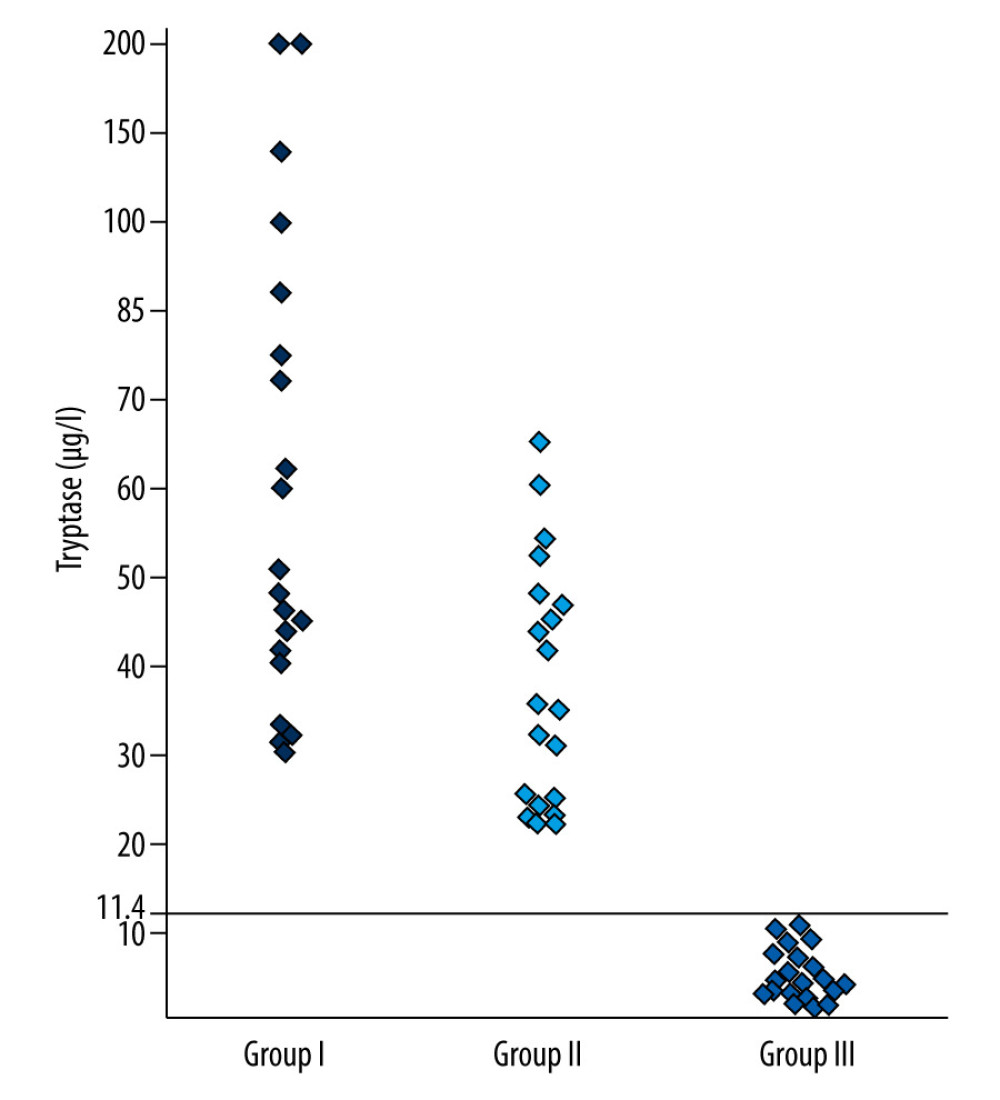 Figure 1. Distribution of tryptase concentration in patients with anaphylaxis, mastocytosis, and atopic patients (μg/L).
Figure 1. Distribution of tryptase concentration in patients with anaphylaxis, mastocytosis, and atopic patients (μg/L). 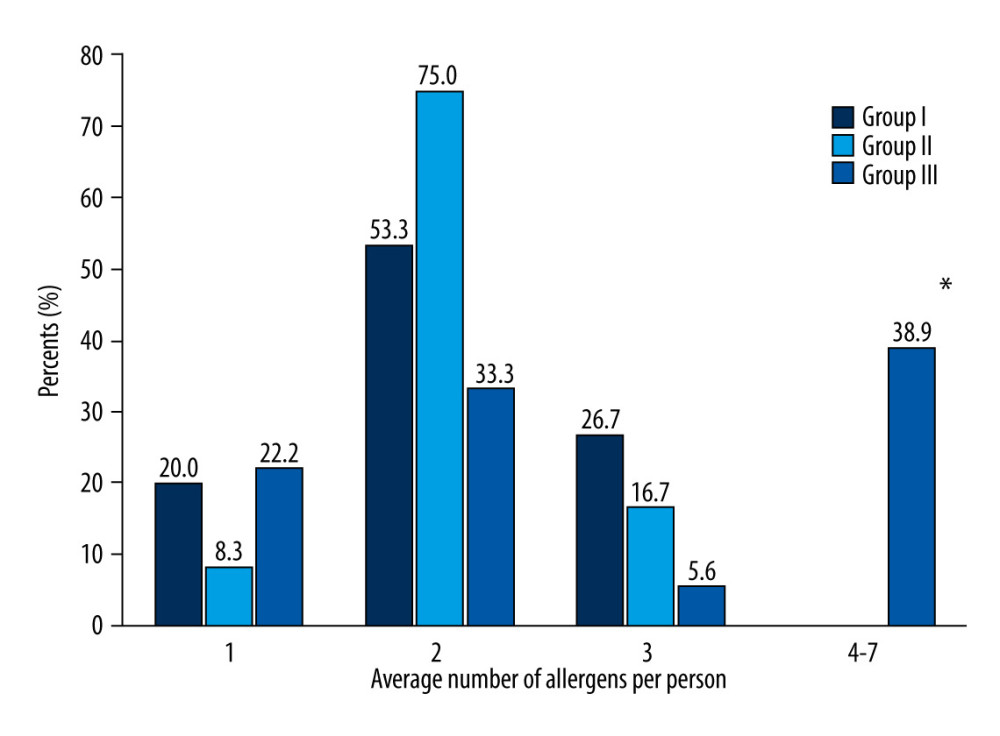 Figure 2. The distribution of patients according to the identified number of allergens per person. I – patients with anaphylasis; II – patients with mastocytosis; III – patients with atopic reactions (control group). * p<0.05 when comparing between group.
Figure 2. The distribution of patients according to the identified number of allergens per person. I – patients with anaphylasis; II – patients with mastocytosis; III – patients with atopic reactions (control group). * p<0.05 when comparing between group. References
1. The European Academy of Allergy and Clinical Immunology (EAAC): Tackling the allergy crisis in Europe concerted policy action needed, 2015, Brussels, Belgium
2. Gulen T, Hagglund H, Dahlen B, Nilsson G, High prevalence of anaphyl-axis in patients with systemic mastocytosis – a single-center experience: Clin Exp Allergy, 2014; 44(1); 121-29
3. Amaresekera M, Immunoglobulin E in health and disease: Asia Pac Allergy, 2011; 1(1); 12-15
4. Won Volcheck G: Clinical allergy. Diagnosis and management, 2009; 8-10, USA, Mayo Clinic
5. Bonadonna P, Zanotti R, Muller U, Mastocytosis and insect venom allergy: Curr Opin Allergy Clin Immunol, 2010; 10(4); 347-53
6. Vaitėnas G, Bagdonaitė L, Kvedarienė V, Serumo triptazė – anafilaksijos ir mastocitozės žymuo (žvalgomasis tyrimas): Laboratorinė Medicina, 2015; 15(1); 45-49 [in Lithuanian]
7. American Academy of Allergy, Asthma and Immunology www.aaaai.org/conditions-and-treatments/conditions-dictionary/atopy.aspx
8. Sicherer SH, Sampson HA, Food allergy: J Allergy Clin Immunol, 2010; 125(2 Suppl); S116-25
9. ImmunoCAP Cross-Reactivity Map, ThermoScientific, Phadia: International Network of Universities for Molecular Allergology and Immunology www.inunimai.org/cms/images/docs/immunocap_isac/xmap/Component_Cross-reactivity_Map_150903.pdf
10. Bonadonna P, Lombardo C, Zanotti R, Mastocytosis and allergic diseases: J Investig Allergol Clin Immunol, 2014; 24(5); 288-97
11. Asarnoj A, Glaumann S, Lilja G: IgE antibodies to allergen components, 2013, Stockholm, Karolinska University Hospital
12. Ėmužytė R, Pumputienė I, Firantienė R, Alerginio uždegimo mechanizmai ir IgE tyrimai klinikinėje praktikoje: Laboratorinė Medicina, 2011; 1(49); 31-36 [in Lithuanian]
13. Escribano L, Orfao A: Anaphylaxis in mastocytosis, 2011, Springer Science
14. , ImmunoCAP Tryptase Directions for use, 2015
15. Hitachi Chemical Diagnostics, Inc. www.hcdiagnostics.com/idc/groups/hcd/documents/
16. Broesby-Olsen SB, Dybedal I, Gulen T, Multidisciplinary management of mastocytosis: Nordic Expert Group consensus: Acta Derm Venerol, 2016; 96(5); 602-12
17. American Academy of Allergy, Asthma and Immunology: Anaphylaxis overview www.aaaai.org/conditions-and-treatments/allergies/anaphylaxis
18. ImmunoCAP (Phadia), Is it allergy?: Clinical utility of Total Tryptase
Figures
 Figure 1. Distribution of tryptase concentration in patients with anaphylaxis, mastocytosis, and atopic patients (μg/L).
Figure 1. Distribution of tryptase concentration in patients with anaphylaxis, mastocytosis, and atopic patients (μg/L). Figure 2. The distribution of patients according to the identified number of allergens per person. I – patients with anaphylasis; II – patients with mastocytosis; III – patients with atopic reactions (control group). * p<0.05 when comparing between group.
Figure 2. The distribution of patients according to the identified number of allergens per person. I – patients with anaphylasis; II – patients with mastocytosis; III – patients with atopic reactions (control group). * p<0.05 when comparing between group. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387