20 October 2022: Clinical Research
Latency and Interpeak Interval Values of Auditory Brainstem Response in 73 Individuals with Normal Hearing
Milaine Dominici SanfinsDOI: 10.12659/MSM.937847
Med Sci Monit 2022; 28:e937847
Abstract
BACKGROUND: Auditory brainstem response (ABR) potential is important for audiological diagnosis, reflecting the integrity of the structures of the auditory system up to the brainstem. The click stimulus is the best known and is most used in clinical practice. However, different devices and examiners may yield distinct results, and each institution tends to use its own parameters. We aimed to analyze the latency values of wave I, III, V, and interpeak intervals I-III, III-V, I-V values obtained in assessing ABR using a new device.
MATERIAL AND METHODS: We performed a cross-sectional study of 73 participants with normal hearing thresholds and no hearing problems. All underwent basic audiological (air and bone conduction, Speech Recognition Threshold, Speech Recognition Index, acoustic reflex, and tympanometry) and electrophysiological evaluation (ABR assessment).
RESULTS: Absolute latency and interpeak values from ABR showed earlier responses in women, faster than international standards suggest. The responses were similar to other studies carried out previously, with the exception of wave I values, which were a little earlier in females.
CONCLUSIONS: We assessed normative data from measurement of latency values of wave I, III, V, and interpeak intervals I-III, III-V, and I-V applying 2 standard deviations in the assessment of ABR using the new Neuro-Audio/ABR device created by Neurosoft.
Keywords: Audiology, Electrophysiology, Evoked Potentials, Auditory, Brain Stem, Hearing, Humans, Female, Cross-Sectional Studies, Acoustic Impedance Tests, Bone Conduction
Background
The brainstem auditory evoked potential (ABR) is an important instrument for audiological diagnosis, allowing the integrity of the structures of the auditory system up to the brainstem to be tested [1]. A variety of verbal and non-verbal stimuli can be used in the test [2], although a click is the best known and most used in clinical practice [2,3].
Assessment via ABR has been shown to be able to diagnose hearing disorders; the technique plays a central role in the differential diagnosis of cochlear, retrocochlear, or conductive hearing loss [4,5]. Assessments are made by measuring the absolute latencies of waves I, III, and V, together with the values of the interpeak intervals I–III, III–V, and I–V. Hearing deficits can be identified through analysis of these values [1–3].
According to Esteves et al [6], different devices and examiners will yield distinct results; thus, each institution should have its own standard set of parameters. The normative values of latency and interpeak intervals are based on population studies. Equipment known and marketed for a long time has well-established normative standards.
But with new equipment, an important task is to provide normative data for that brand and model. A new model of equipment for performing auditory evoked potentials has recently become available, the Neuro-Audio/ABR device by Neurosoft (Ivanovo, Russia).
The objective of the present study is to latency values of wave I, III, V and interpeak intervals I–III, III–V, I–V values obtained in ABR evaluation using a newly equipment of ABR, the Neuro-Audio/ABR device by Neurosoft (Ivanovo, Russia). Thus, it possible to present a normative data for this equipment.
Material and Methods
ETHICS STATEMENT:
This retrospective, cross-sectional study was approved by the Research Ethics Committee (number 2.831.741). Written informed consent was obtained from all participants. Data were collected at the Audiology and Electrophysiology Laboratory of Clinica Ouvire.
PARTICIPANTS:
Based on results from a previous study, a mean minimum sample size of 25 individuals was calculated (error of ±0.3 on average and a significance level of 5%). However, the present study, to ensure accuracy, was performed with a total of 73 participants (33 men and 40 women, aged 3–79 years at the time of assessment) were recruited.
Inclusion criteria were age above 3 years, normal otoscopy bilaterally, normal hearing and immittance at the time of assessment (defined as pure tone audiometry thresholds below 20 dB HL from 250 to 8000 Hz and Type A tympanograms with peak compliance within 0.3 to 1.3 mmhos at −100 to +200 daPa pressure), and the presence of 1 kHz ipsi- and contralateral acoustic reflexes in both ears at the time of assessment [7]. Exclusion criteria were lack of behavioral or neurological disorders and/or genetic syndromes and use of psychoactive medication.
PROCEDURES AND MEASURES:
The audiometric evaluation involved 2 types of thresholds. First, air conduction thresholds were tested at 0.25, 0.5, 1, 2, 3, 4, 6, and 8 kHz. Secondly, bone conduction thresholds were examined at 0.5, 1, 2, and 4 kHz. According to the classification of Davis and Silverman, the normal values of the auditory threshold were set down until 15 dB for air conduction and set down until 20 dB for bone conduction [8]. The assessment was performed in an acoustic booth using an Interacoustics AC40 audiometer with TDH39 headset calibrated in accordance with ISO-389 and IEC-645 norms. For the speech Recognition Threshold, a list of disyllables was used. The final result was the intensity when the participant obtained 50% of the words presented. For the Speech Recognition Index, the list of monosyllabic words 40 dB above the mean tonal threshold of 0.5, 1, and 2 kHz was used for the test. The normal values were obtained if the number of correct answers was 88–100%.
For immittance audiometry (acoustic reflex and tympanometry), we used the Interacoustics 235 h clinical tympanometer through with a 226 Hz probe and the pressure measured in daPa. Investigation of ipsilateral and contralateral acoustic reflexes was carried out with a volume of 0.3–1.3 ml [7]. Immittance audiometry was performed prior to electrophysiological evaluation.
Electrophysiological evaluation was conducted using the Neuro-Audio device from Neurosoft (Ivanovo, Russia). Electrophysiological responses were recorded while the participant was sitting passively in a reclining chair in a comfortable position in a sound-attenuating electrically shielded room.
Responses were recorded with the active electrode positioned on the vertex (Fz), the reference electrode on the ipsilateral mastoid, and the ground on the contralateral mastoid [9]. Impedance was maintained below 5 kΩ and inter-electrode impedance below 3 kΩ. Participants were asked to keep their eyes closed to avoid eye movement artifacts. The order in which the ears were tested was randomized across subjects – in 50% of patients the right ear was recorded first, and in the other 50% the left ear was recorded first. A click stimulus of 0.1 ms duration and rarefaction polarity was presented monaurally to the right and left ears using insert earphones (ER-3C; Etymotic Research, Inc.), with a repetition rate of 19.3/s at 80 dB HL. Two collections of 2000 artifact-free stimuli were collected. The band-pass filter was set at 100–3000 Hz.
The data were analyzed and wave peaks were visually identified and marked as waves I, III, and V. The interpeak intervals I–III, III–V, and I–V were then calculated.
STATISTICAL ANALYSIS:
Statistical analysis was performed using IBM Statistics SPSS version 24. A statistical test for paired samples was conducted. First, the assumption of normality was checked using the Kolmogorov-Smirnov test. This assumption was not fully met, so for further analysis the Wilcoxon signed rank test was used. The level of statistical significance was set at
Results
AGE AND SEX DISTRIBUTION:
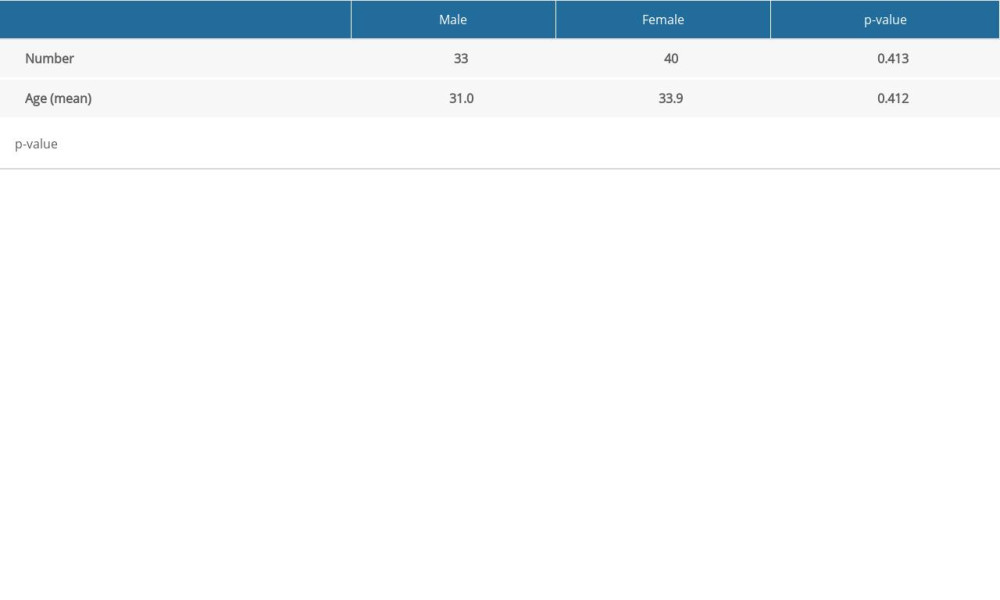
Table 1 presents the age distribution of males and females and shows that the sample was homogeneous in both analyses (P=0.41).
ABSOLUTE LATENCY AND INTERPEAK INTERVAL VALUES:
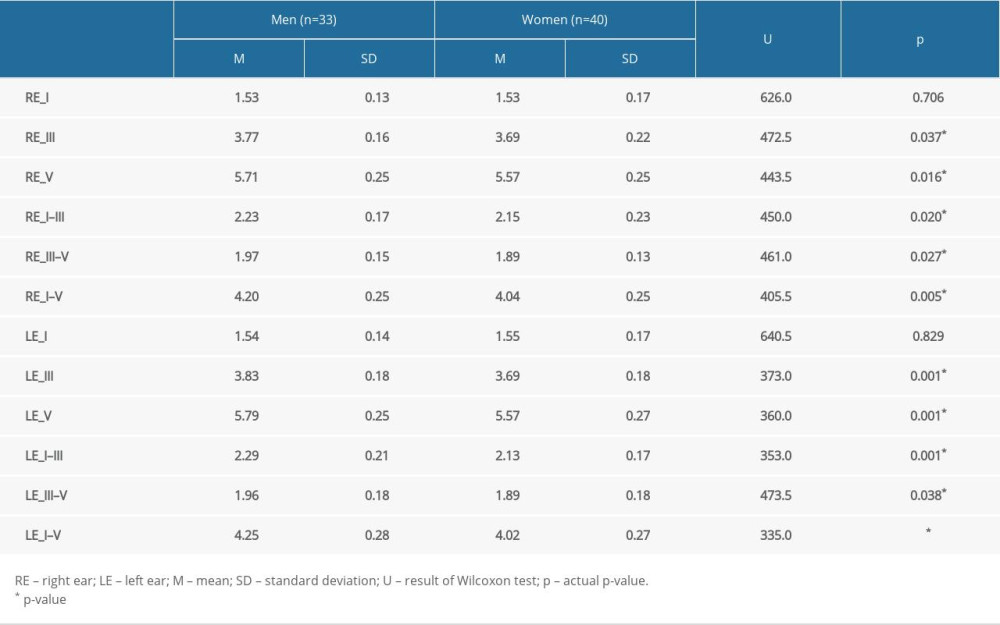
Table 2 presents the absolute latency values of waves I, III, and V and values of the interpeak intervals I–III, III–V, and I–V in the right and left ears at an intensity of 80 dB HL for males and females. Statistically significant differences between sexes were found for waves III and V (in right and left ears) and for interpeak intervals I–III, III–V, and I–V (in right and left ears). In general, women had lower values than men. Only wave I had the same result for both sexes in right and left ears.
EXPECTED NORMATIVE DATA VALUES OF ABR WITH 2 STANDARD DEVIATIONS:
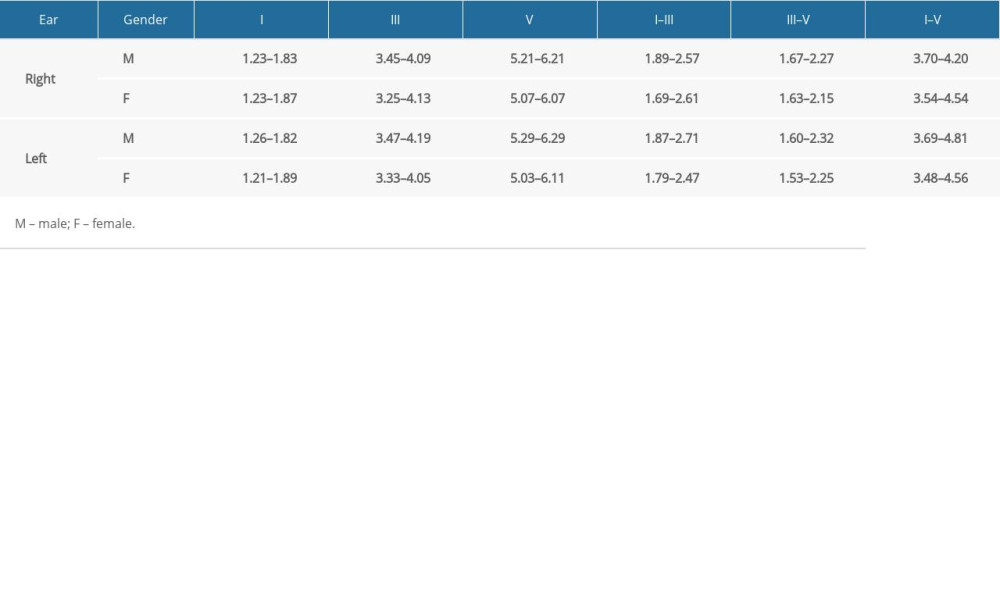
Absolute latencies of waves I, III, and V and of the interpeak values I–III, III–V, and I–V in the right and left ears for females and males are set out in terms of 2 standard deviations (SD) from the mean (Table 3).
Discussion
The present study sets out results found with the ABR responses in individuals who had normal auditory thresholds using the newly commercialized device. In the literature, there are several documents with guidelines on good practice for carrying out ABR assessments, such as the Newborn Hearing Screening Program (NHSP, 2013) [10] and the British Columbia Early Hearing Program (2012) [11]. According to these works, by applying suitable norms when analyzing electrophysiological responses, it is possible to use any brand of equipment available on the market [10,11]. However, the use of normative values is essential for interpretation of ABR responses. According to Esteves et al [10], different devices and examiners will yield distinct results; thus, each institution should have its own standard set of parameters. The present study used parameters following international guidelines, thus giving the evaluator confidence in the quality of the data and the results obtained, and minimizing possible clinician biases and errors.
Our results were based on a Gaussian probabilistic model considering the expected normative values (±2 standard deviations) for the latencies of waves I, III, and V and the interpeak intervals I–III, III–V, and I–V [12]. It is important to note that use of ±2 standard deviations represents 95.5% of the population, and this should result in only a small number of inappropriate diagnoses. The ABR assessment is one of several clinical tests used by healthcare professionals to test for the presence of hearing loss [13,14]. Neurodiagnostics aims to detect retrocochlear pathologies present in the auditory nervous system that impair proper functioning of anatomical structures such as the proximal and distal portions of the auditory nerve, cochlear nuclei, superior olivary nuclei, lateral lemnisci, and inferior colliculi [15,16].
The sensitivity of the ABR derives from the normative values of latency and the interpeak intervals. Hence, these values must be well established so that it is possible to identify alterations in the auditory nervous system. When considering the normative values established for each device, researchers report a sensitivity of 89% for the ABR to detect a lesion smaller than 1.0 cm; if a lesion is 1.0–1.2 cm, the sensitivity of the test increases to 98%, whereas the sensitivity reaches 100% if the lesion is larger than 2.0 cm [17].
We analyzed ABR response of individuals with normal hearing at high intensity (80 dB nHL), rate of 19.3/s, and rarefaction polarity. These parameters were chosen because a neurodiagnostic evaluation must be performed at high intensity (80dB nHL) so that the neural synchrony of the XVIII nerve and the brainstem responses are optimized and robust. At higher intensities, the noise level must be controlled [18,19]. The rate values of 19.3/s provides robust responses with less electrical interference and is often used in evaluations aimed at neurodiagnosis [19–21]. The responses need to be replicable, and if there is any uncertainty after collecting 2 waveforms, a third wave should be collected, which should be like the first 2. If there is a persistent difference between the waves, it is possible there is excessive noise [19]. Finally, in terms of polarity, previous studies have reported that rarefaction polarity is more appropriate because it provides better and robust responses [22].
Another interesting finding is the earlier ABR response values in women than in men. These findings agree with previous studies in the literature showing that in differences in females are due to anatomical issues such as smaller head size and subcortical neural differences [23–27]. Latency values are directly related to the speed of neural responses following a sound stimulation. In cases of neurological impairment, due, for example to the presence of a mass (or tumor) or a reduced number of neurons in a certain structure within the auditory trajectory, latency values will be compromised. Therefore, reference values are essential for an accurate diagnosis to be made, and from there appropriate interventions and treatments may be possible. Assessments of the ABR are the criterion standard for investigating the maturation and integrity of the auditory pathway from the inner ear to the brainstem [28–31].
Our study used parameters following international guidelines and thus gives the evaluator confidence in the quality of the data and the results obtained, minimizing possible clinician biases and errors. In the present work, the responses were very similar to those of previous studies, except for wave I values, which were a little earlier, with values of 1.23 ms (right ear) and 1.21 ms (left ear) for females. Therefore, we strongly recommend that a survey be carried out with a larger number of female subjects to understand whether this earlier wave I response characteristic is specific to this new device. It is worth noting that this response model was not found for men in any of the evaluated ears. Below, we discuss some previous studies performed in patients with normal hearing and with different equipment.
Schwartz et al [32] reported ABR responses at 80 dBnHL and found the following results with ±2.5 SD: Wave I (1.29–1.79 ms), Wave III (3.32–4.08 ms), Wave V (5.12–6.08 ms), I–III (1.60–2.80 ms), III–V (1.42–2.26 ms), and I–V (3.59–4.49 ms). Joseph et al [33] reported the ABR responses at 80 dBnHL and found the following results considering the absolute mean of the values: Wave I (1.65 ms), Wave III (3.80 ms), Wave V (5.64 ms), I–III (2.15 ms), III–V (1.84 ms) and I–V (3.99 ms). Hall [23] presented the ABR responses at 80 dBnHL and the following results were found with ±2.0 SD: Wave I (1.37–1.93 ms), Wave III (3.4–4.16 ms), Wave V (5.18–6.10 ms), I–III (1.87–2.43 ms), III–V (1.56–2.12 ms), and I–V (3.59–4.39 ms). Hood (1998) [34] reported the ABR responses in women with normal hearing at 80 dBnHL and found the following results with ±2 SD: Wave I (1.37–1.93 ms), Wave III (3.44–4.16 ms), Wave V (5.18–6.10 ms), I–III (1.87–2.43 ms), III–V (1.56–2.12 ms), and I–V (3.59–4.39 ms). Musiek et al [35] analyzed only the responses of the interpeak intervals of the ABR at 80 dBnHL and found the following results with ±2SD: I–III (1.80–2.30 ms), III–V (1.40–2.30 ms), and I–V (3.36–4.40 ms).
Our study showed the following responses at 80 dBnHL in normal hearing with ±2SD: Right ear (male) – [Wave I (1.23–1.83 ms), Wave III (3.45–4.09 ms), Wave V (5.21–6.21 ms), I–III (1.89–2.57 ms), III–V (1.67–2.27 ms), and I–V (3.70–4.20 ms)]; Right ear (woman) – [Wave I (1.23–1.87 ms), Wave III (3.45–4.13 ms), Wave V (5.07–6.07 ms), I–III (1.69–2.61 ms), III–V (1.63–2.15 ms), and I–V (3.54–4.54 ms)]; Left ear (male) – [Wave I (1.26–1.82 ms), Wave III (3.47–4.19 ms), Wave V (5.29–6.29 ms), I–III (1.87–2.71 ms), III–V (1.60–2.32 ms) and I–V (3.69–4.81 ms)] and Left ear (woman) – [Wave I (1.21–1.89 ms), Wave III (3.33–4.05 ms), Wave V (5.03–6.11 ms), I–III (1.79–2.47 ms), III–V (1.53–2.25 ms), and I–V (3.48–4.56 ms)].
The participants who took part in this study were all at least 3 years old, since research has concluded that there is stabilization of latency at age 24–36 months, after which maturation in the brainstem is complete and the responses of children and adults become similar [22]. Therefore, the values described in the present study should not be applied to patients under 3 years of age. For this portion of the pediatric population, further studies will be required to verify the ABR responses. The next stage of this research will be to compare the values obtained with the new device with those obtained using standard equipment.
Conclusions
We assessed normative data through of measurement of latency values of wave I, III, V, and interpeak intervals I–III, III–V and I–V values applying 2 standard deviations in the assessment of ABR using the new Neuro-Audio device by Neurosoft. Normative values are set out in Table 3.
References
1. Picton TW: Human auditory evoked potentials, 2011, San Diego (CA), Plural Publishing
2. Sanfins MD, Donadon C, Skarzynski PH, Colella-Santos MF, Electrophysiology and Auditory Training: Auditory system – function and disorders, 2021, London, IntechOpen
3. Moller AR, Janetta PJ, Auditory evoked potentials recorded intracranially from the brain in man: Exp Neurol, 1982; 78; 144-57
4. Hecox K, Galambos R, Brain stem auditory evoked responses in human infants and adults: Arch Otolaryngol, 1974; 99; 30-33
5. House J, Brackmann D, Brainstem audiometry in neurologic diagnosis: Arch Otolaryngol, 1979; 105; 305-9
6. Esteves MCBN, Dell’Aringa AHB, Arruda GV, Brainstem evoked response audiometry in normal hearing subjects: Braz J Otorhinolaryngol, 2009; 75; 420-25
7. Jerger J, Clinical experience with impedance audiometry: Arch Otolaryngol, 1970; 92; 311-24
8. Davis H, Silverman RS: Hearing and deafness, 1970, New York (NY), Rinehart & Wiston
9. Klem GH, Lüders HO, Jasper HH, The ten-twenty electrode system of the International Federation. The International Federation of Clinical Neurophysiology: Electroencephalogr Clin Neurophysiol Suppl, 1999; 52; 3-6
10. England NHS: Public health functions to be exercised by NHS England service specification no20: newborn hearing screening, 2013, London, Department of Health
11. British Columbia, Early Hearing Program (BCEHP): Audiology Assessment Protocol, 2012, British Columbia, BC Children’s Hospital http://www.phsa.ca/Documents/bcehpaudiologyassessmentprotocol.pdf
12. Reynolds D, Gaussian mixture models: Encyclopedia of biometrics, 2009; 659-63, Boston, Springer
13. Norrix LW, Velenovsky D, Unraveling the mystery of auditory brainstem response corrections: The need for universal standards: J Am Acad Audiol, 2017; 28; 950-60
14. Norrix LW, Velenovsky D, Clinicians’ guide to obtaining a valid auditory brainstem response to determine hearing status: Signal, noise, and cross-checks: Am J Audiol, 2018; 27; 25-36
15. Burkard RF, Don M, Eggermont JJ: Auditory evoked potentials: Basic principles and clinical applications, 2007, Philadelphia (PA), Lippincott Williams and Wilkins
16. Song JH, Banai K, Russo NM, Kraus N, On the relationship between speech- and nonspeech-evoked auditory brainstem responses: Audiol Neurootol, 2006; 11; 233-41
17. Zappia JJ, O’Connor CA, Wiet RJ, Dinces EA, Rethinking the use of auditory brainstem response in acoustic neuroma screening: Laryngoscope, 1997; 107; 1388-92
18. Seewald R, Tharpe AM: Comprehensive handbook of pediatric audiology, 2011, San Diego (CA), Plural Publishing
19. Smith JT, Wolfe J, Sound advice for ABR analysis: Heart J, 2014; 67; 14-18
20. Hall JW: New handbook of auditory evoked responses, 2006, Boston (MA), Allyn and Bacon
21. ASHA: American Speech-Language-Hearing Association [Internet]: Guidelines for audiologists providing informational and adjustment counseling to families of infants and young children with hearing loss birth to 5 years of age, 2008, Rockville, ASHA
22. Gorga MP, Kaminski JK, Beaucahine KL, Effects of stimulus phase on the latency of the auditory brainstem response: J Am Acad Audiol, 1991; 2; 1-6
23. Jerger J, Hall J, Effects of age and sex on auditory brainstem response: Arch Otolaryngol, 1980; 106; 387-91
24. Chan YW, Woo EKW, Hammond SR, The interaction between sex and click polarity in brain-stem auditory potentials evoked from control subjects of Oriental and Caucasian origin: Electroencephalog Clin Neurophysiol, 1988; 71; 77-80
25. McFadden D, Champlin CA, Comparison of auditory evoked potentials in heterosexual, homosexual, and bisexual males and females: J Assoc Res Otolaryngol, 2000; 1; 89-99
26. Trune DR, Mitchell C, Phillips DS, The relative importance of head size, gender and age on the auditory brainstem response: Hear Res, 1988; 32; 165-74
27. Don M, Ponton CW, Eggermont JJ, Masuda A, Gender differences in cochlear response time: An explanation for gender amplitude differences in the unmasked auditory brain-stem response: J Acoust Soc Am, 1993; 94; 2135-48
28. Angrisani RMG, Azevedo MFD, Carvallo RMM, Electrophysiological study of hearing in small-for-gestational-age full-term newborns: J Soc Bras Fonoaudiol, 2012; 24; 162-67
29. Esteves MCBN, Dell’Aringa AHB, Arruda GV, Brainstem evoked response audiometry in normal hearing subjects: Braz J Otorhinolaryngol, 2009; 75; 420-25
30. Jiang ZD, Brosi DM, Wu YY, Wilkinson AR, Relative maturation of peripheral and central regions of the human brainstem from preterm to term and the influence of preterm birth: Pediatr Res, 1991; 65; 657-62
31. Casali RL, Santos MFC, Auditory brainstem evoked response: Response patterns of full-term and premature infants: Braz J Otorhinolaryngol, 2010; 76; 729-38
32. Schwartz DM, Pratt RE, Schwartz JA, Auditory brain stem responses in preterm infants: Evidence of peripheral maturity: Ear Hear, 1989; 10; 14-22
33. Joseph JM, West CA, Thornton AR, Herrmann BS, Improved decision criteria for evaluation of clinical ABR’s
34. Hood LJ: Clinical applications of the auditory brainstem response, 1998, San Diego (CA), Singular Publishing Group
35. Musiek FE, Josey AF, Glasscock ME, Auditory brain stem response: Interwave measurement in acoustic neuromas: Ear Hear, 1986; 7; 100-5
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387