11 July 2023: Clinical Research
Corticosteroid-Associated Avascular Necrosis of the Femoral Head in Patients with Severe COVID-19: A Single-Center Study
Vensan Velchov12ABCDE*, Pavel Georgiev12ABCG, Stefan Tserovski12DEFG, Tsvetan TsenkovDOI: 10.12659/MSM.940965
Med Sci Monit 2023; 29:e940965
Abstract
BACKGROUND: Avascular necrosis (AVN) of the femoral head can result from high-dose corticosteroid therapy. Given that severe COVID-19 pneumonia patients respond positively to corticosteroids, this study aimed to explore the incidence of femoral head AVN associated with corticosteroid therapy in 24 patients diagnosed with severe COVID-19 at a single center.
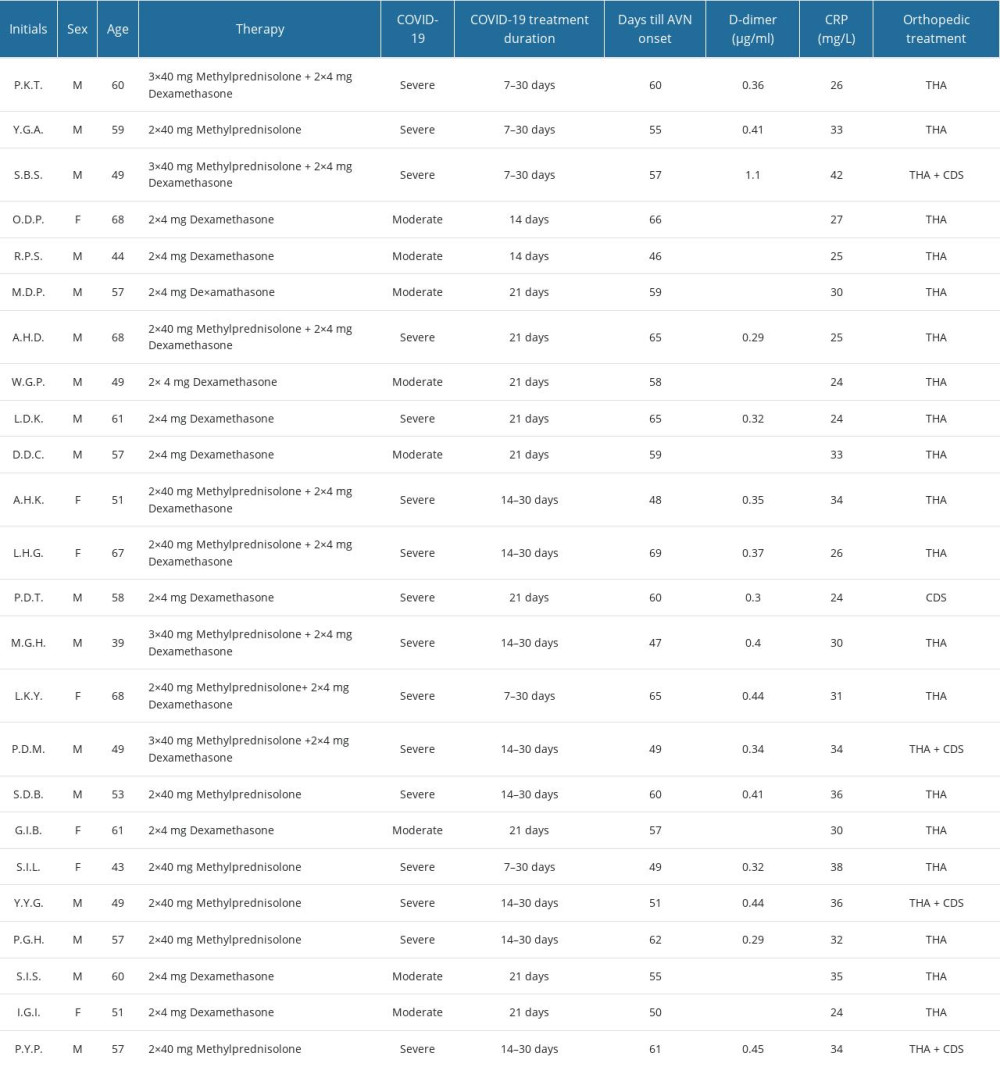
MATERIAL AND METHODS: The study included 24 patients who were diagnosed with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection through real-time reverse transcription polymerase chain reaction test (rRT-PCR) and with COVID-19 pneumonia via high-resolution computed tomography (HRCT). Moderate cases received 2×4 mg Dexamethasone while severe cases were also administered with 3×40 mg Methylprednisolone. Diagnosis of femoral head AVN was confirmed with magnetic resonance imaging (MRI) and radiographs, which was subsequently treated by a total hip arthroplasty (THA) or a core decompression surgery (CDS) in line with the Ficat and Arlet classifications
RESULTS: Among the patients, 8 had a moderate infection course, while 16 were severe. The mean corticosteroid duration was 15±5 days for Dexamethasone and 30 days for Methylprednisolone. Severe patients presented with higher grade femoral head AVN and greater pain levels compared to moderate cases (p<0.05). Four patients developed bilateral AVN. The treatment resulted in 23 THAs and 5 CDSs
CONCLUSIONS: The data from this study corroborate earlier studies and case reports, suggesting an increased occurrence of AVN of the femoral head during the COVID-19 pandemic due to the high-dose corticosteroid therapy employed for patients hospitalized with severe COVID-19 pneumonia.
Keywords: Arthroplasty, COVID-19, osteonecrosis, Humans, COVID-19, Femur Head Necrosis, Femur Head, SARS-CoV-2, Pandemics, Adrenal Cortex Hormones, Methylprednisolone, Dexamethasone
Background
The current recommendations for the use of corticosteroids in the treatment of patients hospitalized with severe COVID-19 are dexamethasone in most patients that require conventional oxygen, and Petrov in all patients who require oxygen with a high-flow nasal cannula (HFNC), noninvasive ventilation (NIV), mechanical ventilation (MV), or extracorporeal membrane oxygenation (ECMO) [1]. Corticosteroid dosage for specific subgroups depending on the disease severity is yet to be discovered and optimized [2,3].
AVN of the femoral head is caused by traumatic and atraumatic reasons. The second most common etiology is corticosteroid-associated osteonecrosis [4]. The diagnosis is obtained by a clinical and imaging evaluation, and the timing of diagnosis can significantly affect outcomes.
The exact mechanism of osteonecrosis in association with corticosteroid use is unknown and under analysis. Sulewski et al described 10 cases of patients with AVN immediately after COVID-19 infection [5]. Li et al discovered glucocorticoid-induced osteonecrosis in up to 32% of the patients who were treated for severe infection [6].
Therefore, this study aimed to describe AVN of the femoral head associated with corticosteroid therapy in 24 patients diagnosed with severe COVID-19 at a single center.
Material and Methods
ETHICS STATEMENT:
Ethics approval was obtained from the Institutional Review Board (IRB) at the University Hospital of Orthopedics “Prof. B. Boichev” EAD – Research and Ethics committee (№ 097/2023). This study was performed in accordance with the most recent version of the Helsinki Declaration. Written and verbal informed consent were taken. The participants’ full names were substituted with their initials to protect their rights and confidentiality. The authors declare no potential conflicts of interest.
PATIENTS:
A retrospective study involving 24 patients was conducted. SARS-CoV-2 infection was diagnosed by an rRT −PCR test using 2 samples from the nose and throat. COVID-19 pneumonia was assessed by HRCT (Figure 1).
The participants were divided into 2 groups – moderate (hospitalized, do not require oxygen supplementation) and severe COVID-19 (hospitalized, require oxygen in any form). Patients with moderate disease were treated with 2×4 mg dexamethasone. Severe cases received 3×40 mg methylprednisolone in addition to 2×4 mg dexamethasone. Dexamethasone was selected as a standard corticosteroid that was present in all COVID-19 protocols. The doses were based on expert opinion. Remdesivir was also administered, as well as the recommended anticoagulant therapy – prophylactic or therapeutic dose of heparin. The patients who required more than conventional oxygen (HFNC, NIV, MV, ECMO) were given methylprednisolone due to the unavailability of an immunomodulator at the institution (eg, Baricitinib, Tocilizumab) [1].
ORTHOPEDIC ASSESSMENT:
The orthopedic clinical presentation consisted of mild groin pain that is increased in palpation, and limping. There was an increase in the pain with active movements, along with a reduction in hip range of motion. The Harris Hip Score (HHS) was used to assess the patients’ pain, function, and range of motion [7].
Plain radiographs and MRI were utilized and the patients were staged according to the Ficat and Arlet classification system (Figure 2) [8]. Focal sclerosis or cystic lesions that are seen on the radiographs as well as MRI data for edema confirmed Grade 2 AVN. Radiographic changes in femoral head sphericity and MRI data for bone necrosis confirmed Grade 3/4 AVN.
Patients with Grades 3 and 4 received a THA and patients with a Grade 2 underwent a CDS. Grade 1 cases were not included in this study.
STATISTICS AND DATA:
Demographic data and COVID-19-related patient information (severity of the infection, drug dosage, treatment duration, CRP, D-dimer) were recorded and analyzed (Table 1). The data were collected from the hospital’s registry to ensure accuracy and reliability. The timeframe was 25 months (from May 2020 until June 2022).
The data are provided as the mean with standard deviation (SD) and p-values. The t test was used for analysis. Microsoft Excel 2019 and the Real Statistics Resource Pack for Excel 2019, Release 7.7.1 were used for processing. Results of p<0.05 were considered statistically significant.
Results
PARTICIPANT CHARACTERISTICS:
We assessed a total of 119 patients for eligibility (Figure 3). There were 17 males and 7 females, with a male to female ratio of 2.4: 1. The mean age of the patients was 56±15 years, as the youngest one was 39 years old and the oldest one was 68 years old. The mean body mass index (BMI) was 34.3±2 kg/m2.
The course of infection was moderate in 8 patients and severe in 16. The moderate group consisted of 5 males and the mean age was 56±7 years. The severe group had 12 males and the mean age was 56 ±9 years.
TREATMENT AND CLINICAL OUTCOMES:
The mean corticosteroid duration was 15±5 days for dexamethasone, and 30 days for methylprednisolone. The mean time from the onset of infection to the onset of AVN symptoms was 57±12 days, and the mean time from first concern to diagnosis and orthopedic treatment was 87±45 days. At the time of surgical management, 100% of the participants had no symptoms of COVID-19 and all had a negative rRT-PCR test.
A bilateral involvement was discovered in 4 cases (17%), 24 patients with 28 hips underwent operative treatment, and 23 THAs (82%) and 5 CDSs (18%) were performed in total. The mean preoperative HHS was 59.2±11.4 compared to 86.8±7.2 postoperatively at 6 months follow-up (p<0.05). One case with CDS presented with worsened clinical and imaging signs of AVN at 7 months postoperatively, and was reoperated on with THA. No other complications were recorded.
Severe patients presented with a higher-grade femoral head AVN and greater pain levels than the moderate group (p<0.05) (Figure 4).
LABORATORY FINDINGS:
The mean levels of D-dimer were 0.41 μg/ml and 31.6 mg/L for C-reactive protein (CRP) at the time of orthopedic treatment. The patients had elevated values of both D-dimer (normal values 0.23 μg/ml [230 ng/ml]) and CRP (normal values under 0.9 mg/L), corresponding to the severity index. These findings are in accordance with the systemic inflammation after COVID-19 and may be related to the AVN symptoms.
VACCINATION STATUS:
None of the patients in this study were vaccinated. Vaccination status is a part of the primary prophylaxis against the coronavirus infection and leads to fewer hospitalizations and lower incidence and mortality rates [9]. The absence of vaccination could be explained by the fact that the country has one of the lowest percentages in the European Unions – just above 30% [10].
Discussion
LIMITATIONS OF THE STUDY:
The data from this study are from a single-center experience. The actual incidence may be higher. Another limitation is that the treatment protocols for COVID-19 have evolved rapidly since the pandemic. In the beginning very little was known on how to treat this infection; therefore, various corticosteroid dosing guidelines and regimes have been prescribed. The corticosteroid therapy for this group was partially based upon expert opinion, which is low-level evidence. This is a confounding factor.
Conclusions
The findings from this study support previous studies and case reports that cases of AVN of the femoral head increased during the COVID-19 pandemic due to the use of high-dose corticosteroid therapy in patients hospitalized with severe COVID-19 pneumonia.
Figures
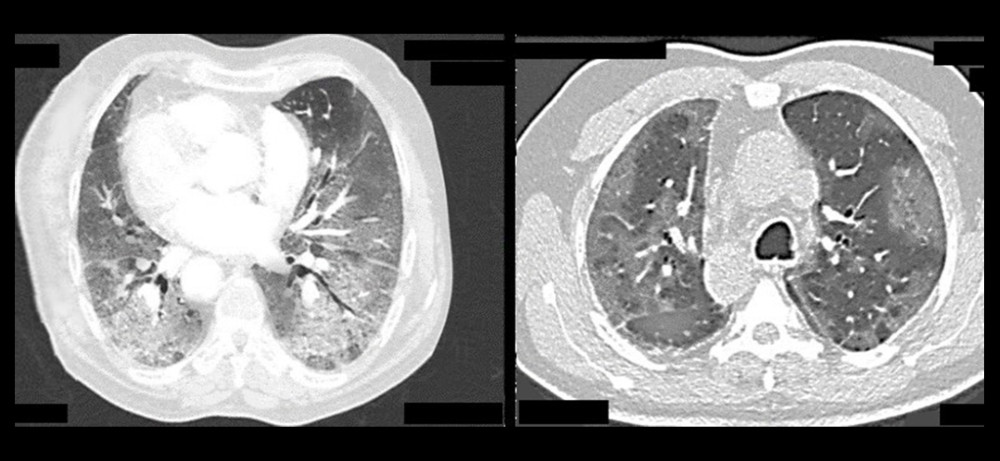 Figure 1. Axial HRCT images showing severe changes in COVID-19 pneumonia.
Figure 1. Axial HRCT images showing severe changes in COVID-19 pneumonia. 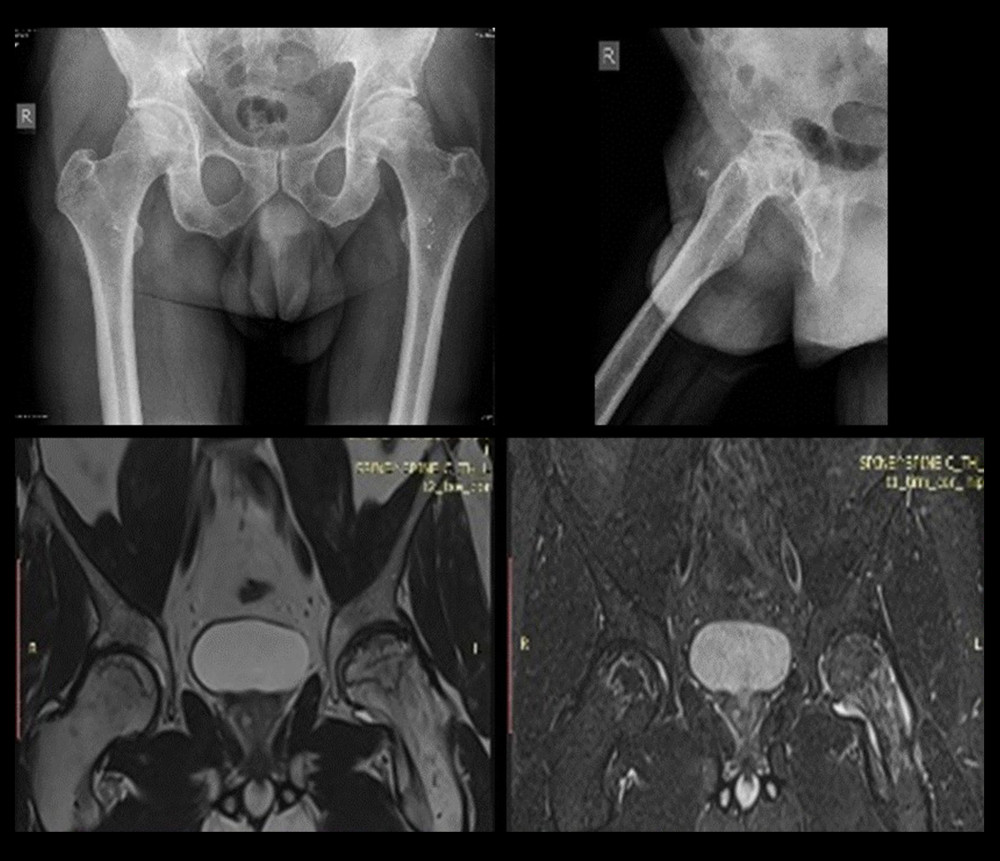 Figure 2. Sample anteroposterior (AP) and lateral radiographs (top), and MRI images (bottom) of femoral head AVN.
Figure 2. Sample anteroposterior (AP) and lateral radiographs (top), and MRI images (bottom) of femoral head AVN. 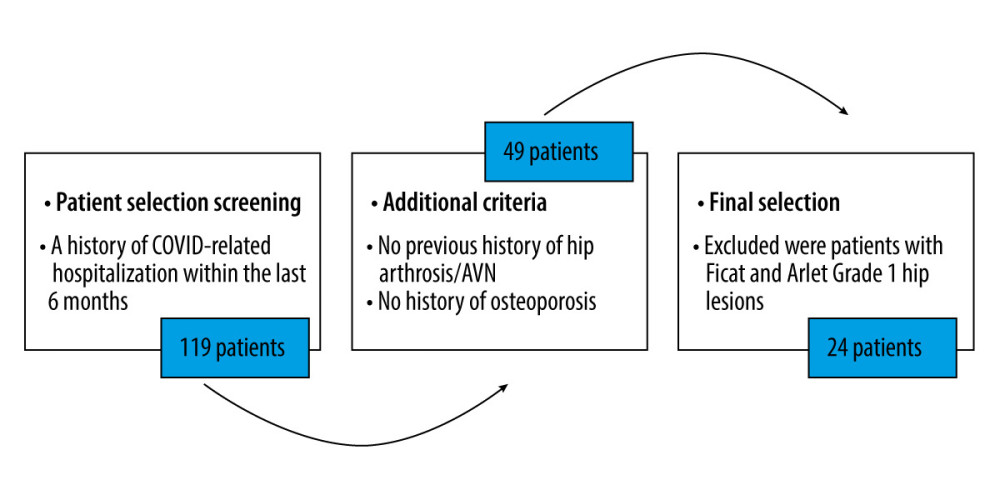 Figure 3. Patient eligibility assessment.
Figure 3. Patient eligibility assessment. 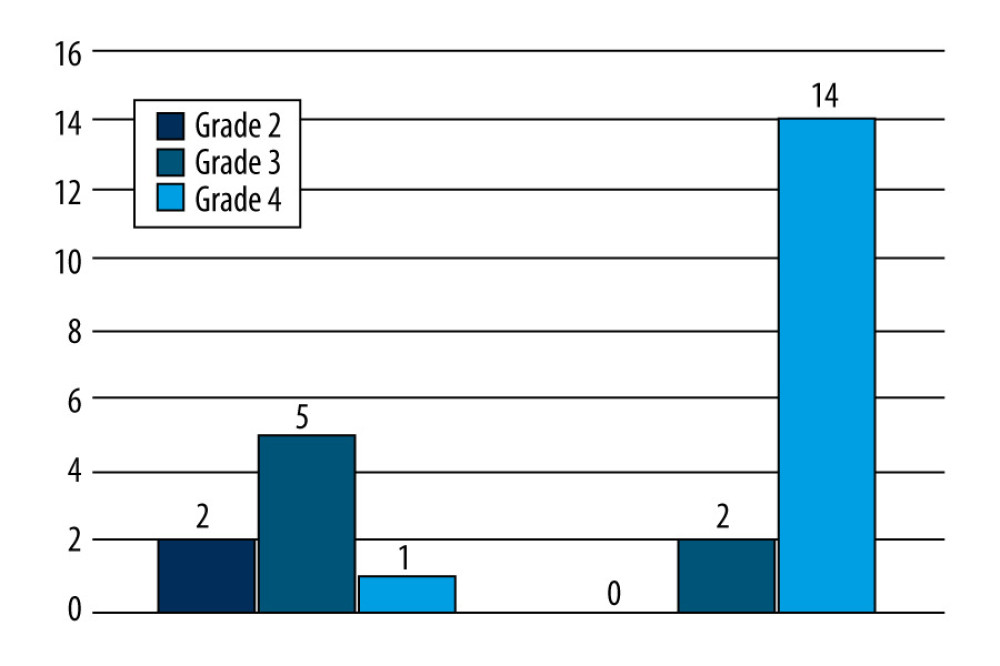 Figure 4. In the severe group, 88% presented with Grade 4 Ficat and Arlet femoral head lesions compared to 13% in the moderate group.
Figure 4. In the severe group, 88% presented with Grade 4 Ficat and Arlet femoral head lesions compared to 13% in the moderate group. References
1. COVID-19 Treatment Guidelines Panel: Coronavirus Disease 2019 (COVID-19) Treatment Guidelines, National Institutes of Health Accessed at https://www.covid19treatmentguidelines.nih.gov/
2. Wagner C, Griesel M, Mikolajewska A, Systemic corticosteroids for the treatment of COVID-19: Equity-related analyses and update on evidence: Cochrane Database Syst Rev, 2022; 11(11); CD014963
3. Ye Z, Wang Y, Colunga-Lozano LE, Efficacy and safety of corticosteroids in COVID-19 based on evidence for COVID-19, other coronavirus infections, influenza, community-acquired pneumonia and acute respiratory distress syndrome: A systematic review and meta-analysis: CMAJ, 2020; 192(27); E756-E767
4. Barney J, Piuzzi NS, Akhondi H, Femoral head avascular necrosis: StatPearls, 2023, Treasure Island (FL), StatPearls Publishing Accessed at: https://www.ncbi.nlm.nih.gov/books/NBK546658
5. Sulewski A, Sieroń D, Szyluk K, Avascular necrosis bone complication after active COVID-19 infection: Preliminary results: Medicina (Kaunas), 2021; 57(12); 1311
6. Li W, Huang Z, Tan B, General recommendation for assessment and management on the risk of glucocorticoid-induced osteonecrosis in patients with COVID-19: J Orthop Translat, 2021; 31; 1-9
7. Li F, Zhu L, Geng Y, Wang G, Effect of hip replacement surgery on clinical efficacy, VAS score and Harris hip score in patients with femoral head necrosis: Am J Transl Res, 2021; 13(4); 3851-55
8. Jawad MU, Haleem AA, Scully SP, In brief: Ficat classification: avascular necrosis of the femoral head: Clin Orthop Relat Res, 2012; 470(9); 2636-39
9. Rahmani K, Shavaleh R, Forouhi M, The effectiveness of COVID-19 vaccines in reducing the incidence, hospitalization, and mortality from COVID-19: A systematic review and meta-analysis: Front Public Health, 2022; 10; 873596
10. Coronavirus Resource Center: Bulgaria – COVID-19 Overview, Johns Hopkins Accessed at: https://coronavirus.jhu.edu/region/bulgaria
11. Jones JP, Osteonecrosis: Arthritis and Allied Conditions: A Textbook of Rheumatology, 2001; 2143-64, Philadelphia, PA, Lippincott Williams & Wilkins
12. Anderton JM, Helm R, Multiple joint osteonecrosis following short-term steroid therapy. Case report: J Bone Joint Surg Am, 1982; 64; 139-41
13. McKee MD, Waddell JP, Kudo PA, Osteonecrosis of the femoral head in men following short-course corticosteroid therapy: A report of 15 cases: CMAJ, 2001; 164; 205-6
14. Agarwala SR, Vijayvargiya M, Pandey P, Avascular necrosis as a part of ‘long COVID-19’: BMJ Case Rep, 2021; 14(7); e242101
15. Panin MA, Petrosyan AS, Hadjicharalambous K, Boiko AV, Avascular necrosis of the femoral head after COVID-19: A case series: Traumatology and Orthopedics of Russia, 2022; 28(1); 110-17
16. Assouline-Dayan Y, Chang C, Greenspan A, Pathogenesis and natural history of osteonecrosis: Semin Arthritis Rheum, 2002; 32; 94-124
17. Wang H, Niu L, Can femoral head necrosis induced by steroid therapy in patients infected with coronaviruses be reversed?: Bone Res, 2021; 9(1); 3
Figures
 Figure 1. Axial HRCT images showing severe changes in COVID-19 pneumonia.
Figure 1. Axial HRCT images showing severe changes in COVID-19 pneumonia. Figure 2. Sample anteroposterior (AP) and lateral radiographs (top), and MRI images (bottom) of femoral head AVN.
Figure 2. Sample anteroposterior (AP) and lateral radiographs (top), and MRI images (bottom) of femoral head AVN. Figure 3. Patient eligibility assessment.
Figure 3. Patient eligibility assessment. Figure 4. In the severe group, 88% presented with Grade 4 Ficat and Arlet femoral head lesions compared to 13% in the moderate group.
Figure 4. In the severe group, 88% presented with Grade 4 Ficat and Arlet femoral head lesions compared to 13% in the moderate group. In Press
15 Mar 2024 : Clinical Research
Evaluation of an Optimized Workflow for the Radiofrequency Catheter Ablation of Paroxysmal Atrial FibrillationMed Sci Monit In Press; DOI: 10.12659/MSM.943526
09 May 2024 : Review article
A Review of the Current Status of Disease-Modifying Therapies and Prevention of Alzheimer’s DiseaseMed Sci Monit In Press; DOI: 10.12659/MSM.945091
09 Apr 2024 : Clinical Research
Correlation between Thalamocortical Tract and Default Mode Network with Consciousness Levels in Hypoxic-Isc...Med Sci Monit In Press; DOI: 10.12659/MSM.943802
19 Apr 2024 : Clinical Research
Comparative Analysis of Postoperative Sagittal Balance in Expansive Open-Door Laminoplasty versus Laminecto...Med Sci Monit In Press; DOI: 10.12659/MSM.943057
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952