14 July 2020: Clinical Research
Clinicopathologic Characteristics and Risk Factors of Lymph Node Metastasis in Patients with Early Gastric Cancer in the Wannan Region
Lin Li1ABE*, Pengwei Liu1CE, Jing Wang1D, Xiaoping Niu1C, Chiyi He1FGDOI: 10.12659/MSM.923525
Med Sci Monit 2020; 26:e923525
Abstract
BACKGROUND: To date, there has been no agreement on the risk factors of lymph node metastasis (LNM) for early gastric cancer (EGC). The objective of this article was to investigate the risk factors of LNM in EGC in the Wannan region of Anhui Province and then to develop a regional practice guideline to manage EGC cases in this population.
MATERIAL AND METHODS: This retrospective analysis was performed from July 1, 2014 to June 30, 2017, at First Affiliated Hospital of Wannan Medical College. We explored the independent risk factors of LNM by using univariate analysis and multivariate analysis.
RESULTS: In total, 381 patients were selected for analysis. The LNM rate of EGC was 13.65% (52 out of 381 patients). Submucosal invasion, ulcer presence, undifferentiated tumor, vascular tumor thrombus, and neural invasion were significantly associated with LNM in EGC patients. Multivariate analysis suggested that depth of invasion (odds ratio [OR], 3.50; 95% confidence interval [CI], 1.45–8.42; P=0.005), vascular tumor thrombus (OR, 6.33; 95%CI, 2.31–17.31; P=0.001), and neural invasion (OR, 10.95; 95%CI, 3.29–36.41; P=0.001) were independent predictors of LNM in EGC patients
CONCLUSIONS: In the Wannan region of Anhui Province in China, depth of invasion, vascular tumor thrombus, and neural invasion were independent predictive risk factors for LNM in EGC patients. EGC patients with these risk factors for LNM are more likely to have LNM, and radical surgical procedure was more likely to be considered. With respective to EGC with submucosal invasion, the flat type pattern had a lower risk of LNM.
Keywords: Gastroscopy, Lymph Nodes, Early Detection of Cancer, Gastrectomy, Gastric Mucosa, Multivariate Analysis, Odds Ratio, Risk Factors
Background
To date, the 5-year survival rate for gastric cancer (GC) is less than 30% [1]. However, early GC (EGC) patients with none or a low risk of lymph node metastasis (LNM) who received en bloc resection by endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD) have a much better 5-year survival rate, which is over 90% [1–3]. Nevertheless, for those patients with high risk factors for LNM, surgical treatment with lymph nodal dissection (LND) is needed. Thus, the evaluation methods for LNM are very important when choosing therapeutic intervention for patients with EGC, and also for patients’ prognosis. So far, the diagnostic accuracy of clinical investigations for LNM of EGC is still unclear, even after a combination of endoscopic ultrasonography and computed tomography.
The incidence of GC is still rising year by year in China. Therefore, early diagnosis of GC is very important [4,5]. Several research studies have revealed that tumor size, histological type, lymphovascular invasion, and depth of invasion are independent risk factors for LNM in EGC in the Chinese population [6–8]. The Wannan region, which is located in the southern area of Anhui Province in Eastern China, is where the incidence and mortality rates for GC are still high. However, the clinical features of and the risk factors for LNM in EGC cases in the Wannan region remain unclear. The main aim of our study was to describe clinicopathological characteristics of patients with EGC and to further elucidate the risk factors for LNM in the Wannan region of Anhui Province. Finally, the ultimate purpose of our study was to develop a regional practice guideline to manage EGC cases in this population.
Material and Methods
DATA COLLECTION AND VARIABLES:
We collected patient information, including name, sex, age at surgical intervention, address, family history, and so on. Symptoms data included nausea, vomit, abdominal pain, abdominal distention, hematemesis, and melena. The paraffin-embedded specimens were fixed by formalin. Two study pathologists with over 10-year clinical practice experience investigated each case independently.
The location and size of tumor, macroscopic classification, gross types, depth of invasion, lymphovascular infiltration, tumor differentiation, the total numbers of lymph nodes retrieved and involvement by carcinoma in each case were recorded in the pathology report.
STATISTICAL METHODS:
The clinical and pathological data were analyzed using SPSS (Version 19.0, IBM, USA). The χ2 test, Fisher’s exact or Mann-Whitney U test was used, when appropriate. In addition, univariate and multivariate logistic regression analysis were performed to estimate the correlation between clinicopathological variables and the LNM status. A 2-tailed value of
Results
PATIENT CHARACTERISTICS:
A total of 2194 patients who underwent radical gastrectomy and LND had their case reviewed; 1808 cases with advanced gastric cancer were excluded. There were 386 EGC patients who received gastrectomy with LND who had their cases reviewed. Another 5 patients with multiple early gastric cancers (2 or more noncontinuous gastric lesions in the same patient with GC) were excluded, including 2 males and 3 females, age range from 42 to 74 years old. Finally, a total of 381 patients with 381 lesions were enrolled in this study, of whom 52 cases had LNM; the rate of LNM was 52 out of 381 (13.65%).
A total of 7001 lymph nodes were retrieved and the number of lymph nodes retrieved range from 15 to 45 per patient. In addition, according to the anatomical drainage area of perigastric lymph nodes, we found that the rate of LNM located in the lesser curvature of the stomach was significantly higher than that in other locations (χ2=7.69,
There were 278 male cases (72.97%) and 103 female cases (27.03%), with an average age of 61.37±7.63 years in male patients and 57.13±9.39 years in female patients; the male-to-female ratio was 2.7: 1.
As shown in Table 1, of the 381 cases, the primary predicted clinical symptoms of EGC were abdominal distention (35.17%, 134 out of 381 cases), abdominal pain (32.55%, 124 out of 381 cases), and melena (7.35%, 28 out of 381 cases).
The baseline characteristics of the participants are shown in Table 2. A tumor located in the proximal stomach, middle stomach, and distal stomach was found in 63, 52, and 266 patients, respectively. There were 233 patients with tumor size over 2 cm. Mucosal lesions were found in 197 cases. Elevated, flat, and depressed type of tumors was found in 45, 98, and 238 patients, respectively. Of the 381 study patients, 81 cases had ulcer findings and 180 patients were diagnosed as having differentiated findings.
RISK FACTORS OF LYMPH NODE METASTASIS (LNM):
We carried out a univariate analysis to explore the risk factors for LNM and we found that tumors limited to the mucosal layer were less likely to have LNM than tumors with submucosal invasion. For ulcer presence, the EGC patients with ulceration findings had a significantly higher frequency of LNM than those without ulceration. The rate of LNM was significantly higher in EGC patients with undifferentiated tumors than those with differentiated tumors. As shown in Table 2, with regard to the vascular tumor thrombus and neural invasion, the EGC patients with vascular tumor thrombus and neural invasion, respectively, were more likely to have LNM than those without vascular tumor thrombus and neural invasion. We concluded that submucosal invasion (P<0.001), ulcer presence (P<0.001), undifferentiated tumor (P<0.001), vascular tumor thrombus (P<0.001), and neural invasion (P<0.001) were significantly associated with LNM in EGC (Table 2).
Furthermore, a multivariate analysis was preformed and the results identified that depth of invasion (OR, 3.50; 95%CI, 1.45–8.42; P=0.005), vascular tumor thrombus (OR, 6.33; 95%CI, 2.31–17.31; P=0.001), and neural invasion (OR, 10.95; 95%CI, 3.29–36.41; P=0.001) were independent predictors of LNM in EGC (Table 3).
SUBGROUP ANALYSIS:
Based on the aforementioned results, our multivariate analysis found that depth of invasion was an independent predictor of LNM in EGC, and the univariate analysis revealed that submucosal invasion was significantly associated with LNM in EGC. Furthermore, we carried out a univariate analysis and a multivariate analysis to explore the risk factors of LNM in EGC with submucosal invasion. As shown in Table 4, a statistically significant difference was found in ulcer presence (P=0.02), undifferentiated tumor (P=0.001), vascular tumor thrombus (P=0.001), and neural invasion (P<0.001) between patients with LNM and patients without LNM. Multivariate analysis revealed that vascular tumor thrombus with the OR of 4.15 (95%CI, 1.29–13.41; P=0.017), neural invasion with the OR of 20.56 (95%CI, 4.37–96.79; P=0.001), and flat type of EGC with the OR of 0.17 (95%CI, 0.03–0.89; P = 0.04) were the significant independent risk factors of LNM. Flat type (OR 0.17, 95%CI 0.03–0.89, P=0.04) had a lower risk of LNM in EGC with submucosal invasion in the study patients from the Wannan region (Table 5).
Discussion
EGC is known to present a low rate of LNM. However, LNM is a crucial factor for the prognosis of EGC patients, therefore, an accurate prediction of the risk of LNM is very important when considering treatment options of EGC. In our study, the rate of LNM was 13.65% for our patients in Wannan region in Anhui Province, which was a similar rate reported in other provinces in China [7,8,11].
Cardia-fundus is the most common site of gastric cancer in western countries, while the gastric antrum and body are the most common sites of gastric cancer in Asian countries [12]. Several studies showed that there was a lower risk of LNM in early gastric cardiac carcinoma compared to early gastric non-cardiac carcinoma [13–15]. Our results were parallel to these reports, as EGC was more often located in the distal stomach (69.82%), and the rate of LNM was higher in the distal stomach (15.04%) than that in the proximal stomach (9.38%).
Several studies [7,16] reported that tumor size was one of the independent risk factors for LNM. However, our study failed to find a similar result. There was no significant difference in tumor size between the EGC patients with LNM and without LNM. On the other hand, our research indicated that EGC with ulceration findings were more likely to have LNM than those without ulceration findings.
Tumor differentiation has been suggested to be an independent risk factor of LNM in several studies [7,17] which was not consistent with our results. In our study, we found that patients with differentiated tumor (10 out of 180 patients) had lower risk of LNM than those with undifferentiated tumor (42 out of 201). Zhang et al. [18] found that the lower the degree of differentiation of tumor, the higher the risk of vascular tumor thrombus and neural infiltration.
Currently, more attention has been paid to vascular tumor thrombus and neural infiltration. Lymphovascular invasion has been shown to be an important independent risk factor of LNM in most studies [19–21], which was confirmed in our study. The patients with vascular tumor thrombus have higher incidence of LNM than those without LNM. In addition, we also found that neural invasion was another vitally independent risk factor of LNM in EGC.
In our research, the depth of invasion was an independent predictive risk factor of LNM in EGC in patients from the Wannan region. The overall risk of LNM was greatly increased from 4.57% in patients with intramucosal EGC to 23.37% in patients with submucosal EGC.
Furthermore, we performed a subgroup analysis to explore the risk factors of LNM in EGC patients with submucosal invasion. And the results suggested that flat type tumors had a lower risk of LNM in EGC with submucosal invasion in our study patients from the Wannan region. However, we did not find that the tumor gross type was one of significant risk factors for LNM in EGC. Meanwhile, other research [7] showed that the endoscopic flat type was a significant risk factor for LNM in EGC in patients from the Sunan region of China. Therefore, multicenter studies in different regions of China are needed to explore the risk of LNM in EGC, and further develop regional practice guidelines for a better therapeutic strategy.
One recently published article [22] showed that the collagen signature in the tumor microenvironment was an independent indicator of LNM in EGC. Collagen, as a component of the extracellular matrix which could promote cell breakage into the basement membrane, was linked to increased tumor formation and changes in gene/protein expression. Increased density of collagen was shown to be associated with LNM in breast cancer [23], glioblastoma [24], and prostate cancer [25]. Future research is required to explore the tumor microenvironment which may more strongly predict patient outcomes.
Some limitations of this analysis should be acknowledged. Firstly, it was a retrospective study with a non-randomized design, which might generate a potential selection bias; however, we reviewed consecutive patients between July 1, 2014 and June 30, 2017 to minimize selection bias. Secondly, it was a single center study, and thus multicenter clinical trials with large-scale are required to explore the risk factors of LNM in the future.
Conclusions
In our research, our finding demonstrated that patients in Wannan region with submucous infiltration, ulcer presence, undifferentiated tumor, vascular tumor thrombus, and neural invasion are more likely to have LNM. Depth of invasion, vascular tumor thrombus, and neural invasion were independent predictive risk factors of LNM in EGC. EGC with aforementioned risk factors is more likely to have LNM and need radical gastrectomy treatment. We found that EGC with submucosal invasion, flat type tumors had a lower risk of LNM.
Tables
Table 1. Primary predicted symptoms of the 381 patients with EGC.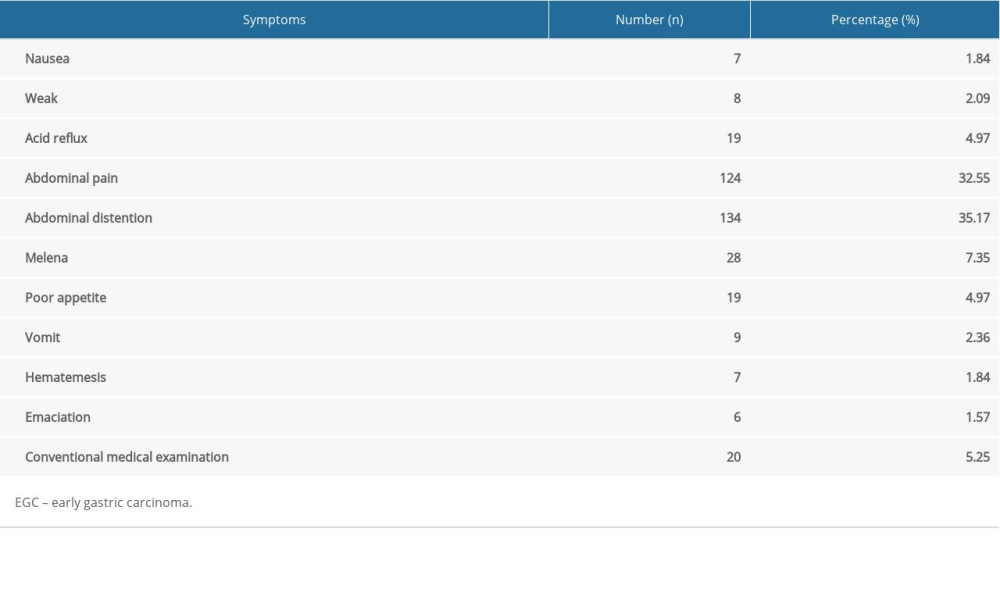 Table 2. Demographic and Clinical characteristics and univariate analysis of risk factors for LNM in 381 patients with EGC.
Table 2. Demographic and Clinical characteristics and univariate analysis of risk factors for LNM in 381 patients with EGC.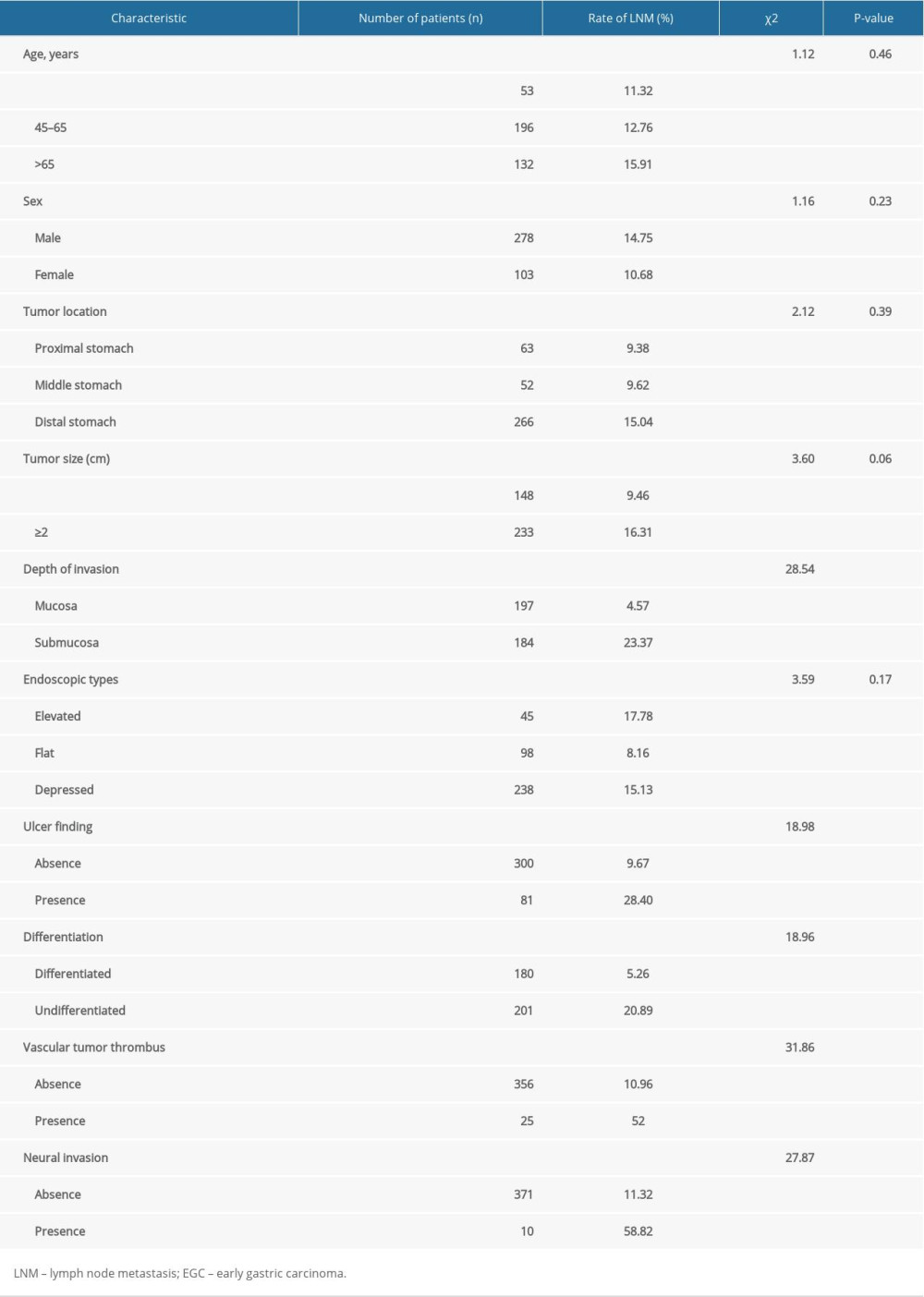 Table 3. Multivariate analysis of risk factors for LNM in 381 patients with EGC.
Table 3. Multivariate analysis of risk factors for LNM in 381 patients with EGC.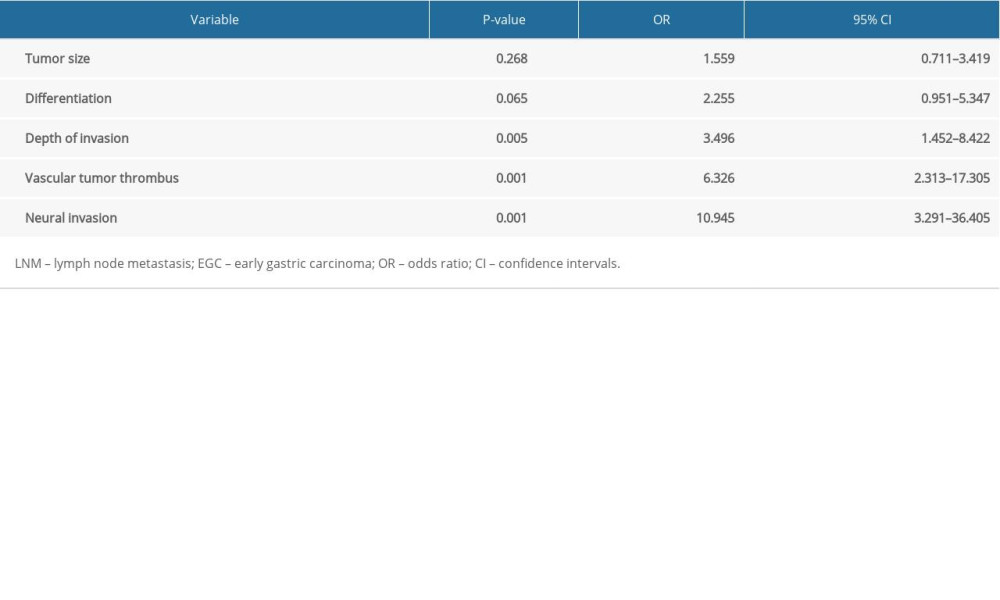 Table 4. Univariate analysis of risk factors for LNM in EGC with submucosal invasion.
Table 4. Univariate analysis of risk factors for LNM in EGC with submucosal invasion.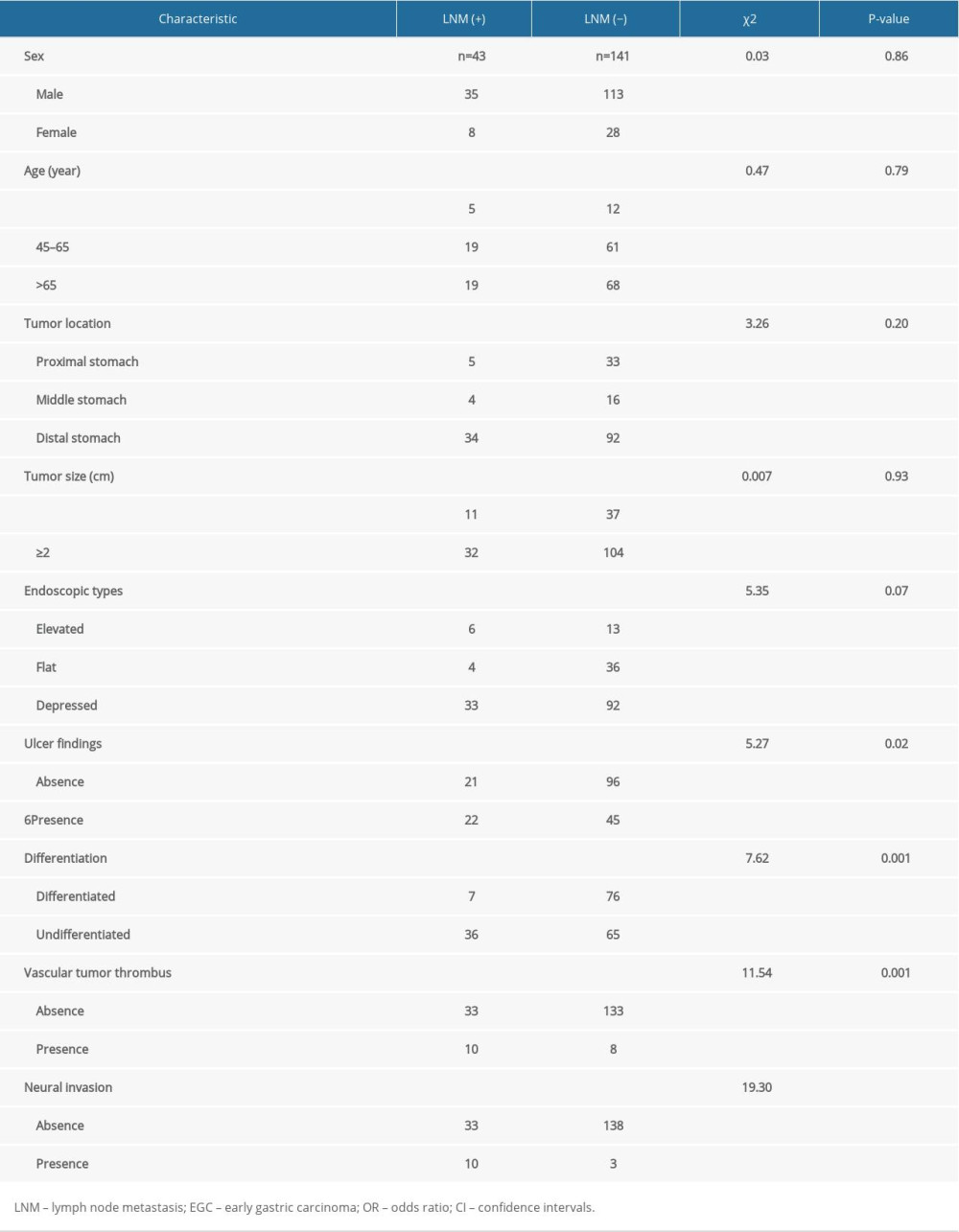 Table 5. Multivariate analysis of risk factors for LNM in EGC with submucosal invasion.
Table 5. Multivariate analysis of risk factors for LNM in EGC with submucosal invasion.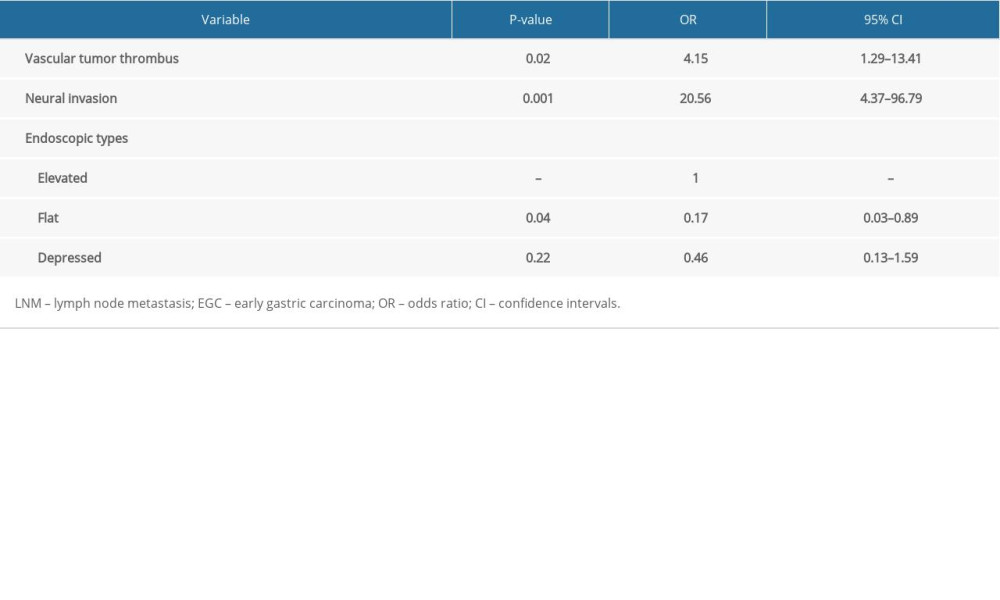
References
1. Huang Q, Fang C, Shi J, Differences in clinicopathology of early gastric carcinoma between proximal and distal location in 438 Chinese patients: Sci Rep, 2015; 5; 13439
2. Van Cutsem E, Sagaert X, Topal B, Gastric cancer: Lancet, 2016; 388(10060); 2654-64
3. Goto O, Fujishiro M, Kodashima S, Outcomes of endoscopic submucosal dissection for early gastric cancer with special reference to validation for curability criteria: Endoscopy, 2019; 41; 118-22
4. Bertuccio P, Chatenoud L, Levi F, Recent patterns in gastric cancer: A global overview: Int J Cancer, 2009; 125(3); 666-73
5. Sun X, Mu R, Zhou YAnalysis of 20-year change of mortality rate of gastric cancer in China and prediction of its development trend: Journal of Zhonghua Oncology, 2004; 26(1); 4-9 [in Chinese]
6. Fang C, Shi J, Sun Q, Risk factors of lymph node metastasis in early gastric carcinomas diagnosed by WHO criteria in 379 Chinese patients: J Dig Dis, 2016; 17; 526-37
7. Du MZ, Gan WJ, Yu J, Risk factors of lymph node metastasis in 734 early gastric carcinoma radical resections in a Chinese population: J Dig Dis, 2018; 19(10); 586-95
8. Wang Y, The predictive factors for lymph node metastasis in early gastric cancer: A clinical study: Pak J Med Sci, 2015; 31(6); 1437-40
9. Ok KS, Kim GH, Park DY, Magnifying endoscopy with narrow band imaging of early gastric cancer: Correlation with histopathology and mucin phenotype: Gut Liver, 2016; 10(4); 532-41
10. Japanese Gastric Cancer Association, Japanese gastric cancer treatment guidelines 2014 (ver. 4): Gastric Cancer, 2017; 20(1); 1-19
11. Zhang M, Zhu G, Zhang H, Clinicopathologic features of gastric carcinoma with signet ring cell histology: J Gastrointest Surg, 2010; 14; 601-6
12. Yuan J, Li Y, Tian T, Risk prediction for early-onset gastric carcinoma: A case-control study of polygenic gastric cancer in Han Chinese with hereditary background: Oncotarget, 2016; 7(23); 33608-15
13. Nesi G, Basili G, Girardi LR, Pathological predictors of lymph node involvement in submucosal gastric carcinoma: A retrospective analysis of long-term outcome: In Vivo, 2009; 23; 337-41
14. Kang HJ, Kim DH, Jeon TY, Lymph node metastasis from intestinal-type early gastric cancer: experience in a single institution and reassessment of the extended criteria for endoscopic submucosal dissection: Gastrointest Endosc, 2010; 72; 508-55
15. American Joint Committee on Cancer, Chapter 10. Esophagus and esophagogastric junction: AJCC Cancer Staging Manual, 2009; 129-44, New York, NY, Springer
16. Chen L, Wang YH, Cheng YQ, Risk factors of lymph node metastasis in 1620 early gastric carcinoma radical resections in Jiangsu Province in China: A multicenter clinicopathological study: J Dig Dis, 2017; 18(10); 556-65
17. Lai JF, Xu WN, Noh SH, Lu WQ, Effect of World Health Organization (WHO) histological classification on predicting lymph node metastasis and recurrence in early gastric cancer: Med Sci Monit, 2016; 22; 3147-53
18. Zhang D, Li Q, Yang L, Analysis of clinicopathologic factors affecting formation of lymphovascular invasion in 1 260 patients with gastric cancer: Clin J Surg, 2017; 16(3); 269-74
19. Hu Q, Dekusaah R, Cao S, Risk factors of lymph node metastasis in patients with early pure and mixed signet ring cell gastric carcinomas: J Cancer, 2019; 10(5); 1124-31
20. Jiao J, Guo P, Hu S, Laparoscopic gastrectomy for early gastric cancer and the risk factors of lymph node metastasis: J Minim Access Surg, 2019; 16(2); 138-43
21. Murai K, Takizawa K, Shimoda T, Effect of double-layer structure in intramucosal gastric signet-ring cell carcinoma on lymph node metastasis: A retrospective, single-center study: Gastric Cancer, 2019; 22(4); 751-58
22. Chen D, Chen G, Jiang W, Association of the collagen signature in the tumor microenvironment with lymph node metastasis in early gastric cancer: JAMA Surg, 2019; 154(3); e185249
23. Conklin MW, Gangnon RE, Sprague BL: Cancer Epidemiol Biomarkers Prev, 2018; 27(2); 138-45
24. Pointer KB, Clark PA, Schroeder AB, Association of collagen architecture with glioblastoma patient survival: J Neurosurg, 2017; 126(6); 1812-21
25. Penet MF, Kakkad S, Pathak AP, Structure and function of a prostate cancer dissemination-permissive extracellular matrix: Clin Cancer Res, 2017; 23(9); 2245-54
Tables
 Table 1. Primary predicted symptoms of the 381 patients with EGC.
Table 1. Primary predicted symptoms of the 381 patients with EGC. Table 2. Demographic and Clinical characteristics and univariate analysis of risk factors for LNM in 381 patients with EGC.
Table 2. Demographic and Clinical characteristics and univariate analysis of risk factors for LNM in 381 patients with EGC. Table 3. Multivariate analysis of risk factors for LNM in 381 patients with EGC.
Table 3. Multivariate analysis of risk factors for LNM in 381 patients with EGC. Table 4. Univariate analysis of risk factors for LNM in EGC with submucosal invasion.
Table 4. Univariate analysis of risk factors for LNM in EGC with submucosal invasion. Table 5. Multivariate analysis of risk factors for LNM in EGC with submucosal invasion.
Table 5. Multivariate analysis of risk factors for LNM in EGC with submucosal invasion. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








