28 July 2020: Clinical Research
Reliability of Myotonometric Measurement of Stiffness in Patients with Spinal Cord Injury
Jun-Sheng Ge1BDF, Tian-Tian Chang2BDE, Zhi-Jie Zhang3ACG*DOI: 10.12659/MSM.924811
Med Sci Monit 2020; 26:e924811
Abstract
BACKGROUND: Contracture is related to modulation of passive stiffness in muscle and tendon after spinal cord injury (SCI). Current clinical assessments of stiffness in muscles and tendons are subjective in patients with spinal cord injury. We proposed a quantitative method to evaluate stiffness of the gastrocnemius and Achilles tendon (AT) with a portable device, the MyotonPRO. The purpose of this study was to investigate the intraoperator and interoperator reliability of the MyotonPRO when used in patients after spinal cord injury.
MATERIAL AND METHODS: Fourteen patients with SCI participated in this study. Gastrocnemius stiffness and AT stiffness were measured with the MyotonPRO.
RESULTS: In participants with SCI, the intraclass correlation coefficient (ICC) values for intraoperator and interoperator reliability of stiffness measurements in the gastrocnemius and AT were excellent (all ICC >0.87), with relatively low values for standard error measurement (SEM) and minimal detectable change (MDC).
CONCLUSIONS: Our findings suggest that use of the MyotonPRO is feasible for evaluating stiffness of the gastrocnemius and AT in the lower limbs of patients with spinal cord injury.
Keywords: Achilles Tendon, Elastic Tissue, Spinal Cord Injuries, Adolescent, Ankle Joint, Biomechanical Phenomena, muscle strength, Muscle Tonus, Muscle, Skeletal, Range of Motion, Articular, Reproducibility of Results
Background
Spinal cord injury (SCI) is the second most serious and frequent traumatic event after craniocerebral injury, and can lead to a high rate of disability [1]. It leads to physical, psychological, social, and economic dysfunction for patients and their families [2]. Muscle dysfunction and contracture are common complications in patients with SCI and can lead to pain, deformity, and failure of a nerve recovery plan [3]. The contracture always occurs in the ankle joint, because of the variation in passive stiffness in muscles and tendons [4]. Thus, it is vital to quantify the stiffness of muscles and tendons to prevent secondary diseases after SCI and to offer targeted treatments.
There are various methods of evaluating the stiffness of muscles and tendons. Passive joint stiffness can be accurately evaluated by monitoring joint torque and using surface electromyography (EMG), but clinical application is impractical [5]. The modified Ashworth Scale, palpation, and the pendulum test are usually used to evaluate joint stiffness clinically [6,7]. Although these methods have been used globally by physiotherapists because they are low cost, they have been criticized for subjective limitations and non-repeatability [6,8,9]. Shear wave elastography (SWE) also has been used to quantitatively assess muscle and tendon stiffness [10,11], but it requires more expensive equipment and more professional operating skills.
The MyotonPRO is a noninvasive and portable device that has recently been used to quantify modulations in muscle and tendon stiffness. Our previous studies have demonstrated the feasibility of using the MyotonPRO to evaluate stiffness in the gastrocnemius [12], upper trapezius [13] and Achilles tendon (AT) [14] in healthy populations. Furthermore, our previous study also found a significant correlation between shear elastic modulus values determined by SWE and stiffness obtained by the MyotonPRO. Several studies also have confirmed that the MyotonPRO is a reliable method of assessing soft-tissue stiffness in patients with tendinopathy [15], stroke [16], Parkinson’s disease [17], and dementia [18]. It is unclear, however, whether the MyotonPRO is reliable for quantifying stiffness of muscles and tendons in patients with SCI.
Therefore, the objectives of this study were to: 1) establish intraoperator and interoperator reliability of the MyotonPRO for quantifying muscle and AT stiffness in patients with SCI; and 2) calculate the minimal detectable change (MDC) and standard error measurement (SEM) of measurements to provide a reference for clinical evaluation in the future. We hypothesized that the MyotonPRO would be a reliable method of quantifying stiffness of muscles and tendons in patients with SCI.
Material and Methods
ETHICS:
This study received institutional approval by the Human Subjects Ethics committee of Luoyang Orthopaedic Hospital of Henan Province (No. 2019-001-01) and followed the recommendations of the Declaration of Helsinki. Before participating in the experiment, all subjects understood the experiment procedures and signed written informed consent.
PARTICIPANTS:
Fourteen patients with SCI (traumatic or non-traumatic) were recruited from the inpatient rehabilitation department of Henan Province Orthopaedic Hospital. The sample size was determined through the method described by Walter et al. 1998. It was decided that for three repeated measurements in each subject, based on the assumptions of α=0.05 and 80% power, P0 (ICC)=0.5. When P1 (ICC)=0.8, the sample size was 14.4, and P1 (ICC)=0.9, the sample size was 5.9. Therefore, 14 patients were enough for both interoperator and intrapoperator reliability analyses [19]. Basic information for each subject was recorded before participating in the trial, such as age, height, weight, injury level and injury time (Table 1). All consecutively admitted patients with SCI were screened by clinical team members. The level of injury ranged from C5 to S2. Data collection was completed from 14: 00 to 16: 00. Inclusion criteria included: 1) SCI within the last 12 months; 2) history of stable spasticity over 2 weeks before the test; 3) age over 18 years; 4) Mini-Mental State Examination score >25; and 5) no change in spasticity treatment during the study. Exclusion criteria included: 1) SCI within the last month; 2) multiple central nervous system lesions; 3) medical instability; 4) significant complications, such as decubitus ulcers, heterotopic ossification, urinary tract infections, and any other infections; 5) musculoskeletal impairments that could confound results of the experiment; 6) skin lesions at the measuring region; and 7) inability to give informed consent.
EQUIPMENT:
The machine used for measuring muscle and tendon stiffness was the MyotonPRO (MyotonAS, Tallinn, Estonia). The probe of the MyotonPRO sends out short impulses to soft tissue after precompression of the skin in region to be measured at 0.8-sec intervals. These impulses cause oscillations in the measured soft tissue. Then, the oscillation wave form is recorded by a triaxial accelerometer, and five mechanical parameters of soft tissue are calculated by the MyotonPRO. Stiffness is one of the parameters, and the higher the stiffness value, the greater the capability of tissue to resist deforming forces. Any data with a coefficient of variation greater than 3% in any measurement of quintuple scanning mode were remeasured.
PROCEDURES:
Methods to quantify the gastrocnemius and AT stiffness by MyontonPRO were adopted from our previous studies [14,20]. Stiffness of the medial head of the gastrocnemius (MG) was measured at near 30% of the length between the popliteal fossa and the lateral malleolus, where the cross-sectional area of the muscle is the largest [20,21]. The lateral head of the gastrocnemius (LG) was measured at one-third of the length between the small head of the fibula and the heel [20,22]. AT stiffness was measured 4 cm above the calcaneal tuberosity [14].
All measurements were made in the ward with a room temperature of 25°C. All stiffness measurements in the right limb were performed with the subject in the prone position, and the subject was asked to completely relax their upper and lower limbs. The measurement regions were located by a physical therapist when the ankle was in a relaxed, resting position [23–25]. The probe of the MyotonPRO was placed on and perpendicular to the measured region when evaluating stiffness of muscle and tendon. Rater A measured stiffness of the LG, MG and AT first; then, rater B repeated the procedure using MyotonPRO. The order was as follows: LG, MG, AT. The order of the raters was always rater A performing a measurement first. Both raters were blinded to all measurement results. In addition, rater A measured again after 5 days, following the same steps.
STATISTICAL ANALYSES:
SPSS software (SPSS version 22.0, IBM, United States) was used for data analysis. Normality distribution of the data was assessed using the Shapiro-Wilk test. Demographic data from participants including age, weight, and height were evaluated by descriptive statistics. All stiffness data were presented as mean±standard deviation. The significance level was set to 0.05 for all stiffness analyses. The interclass correlation coefficient (ICC) (2,2) and ICC (3,1) models were used to evaluate the interoperator and intraoperator reliability of the test. Strength of correlations was interpreted as low (0.00–0.25), fair (0.25–0.50), moderate to good (0.50–0.75), and good to excellent (>0.75) [26].The Bland-Altman diagram drawn by medcalc 18 (Software bvba, Ostend, Belgium), shows the reliability of the intraoperator and interoperator. The calculation formula of standard error measurement (SEM) was SEM=standard deviation×√1-ICC. The formula of minimal detectable change (MDC) was MDC=1.96×SEM×√2.
Results
DEMOGRAPHIC DATA:
Fourteen subjects with SCI were enrolled (age: 42.14±13.38 y; height: 169.50±6.28 cm; weight: 63.25±8.07 kg) (Table 1). All participants were able to successfully complete the experiment without any discomfort.
INTRAOPERATOR AND INTEROPERATOR RELIABILITY:
ICCs of the intraoperator and interoperator reliability for the three studied muscles and tendon are shown in Tables 2, 3. All intraoperator and interoperator reliabilities were 0.87 and above. The ICC values for LG reliabilities ranged from 0.91 to 0.98. The ICC values for intraoperator reliability [ICC=0.91; 95% confidence interval (CI)=0.74–0.97; SEM <12.18 N/m; and MDC <33.76 N/m] and interoperator reliability(ICC=0.98; 95% CI=0.93–0.99; SEM <12.74 N/m; and MDC <35.31 N/m) were excellent. The ICC values for MG reliabilities ranged from 0.87 to 0.98. The ICC values for intraoperator reliability (ICC=0.87; 95% CI=0.61–0.96; SEM <9.83 N/m; and MDC <27.25 N/m) and interoperator reliability (ICC=0.98; 95% CI=0.95–1.00; SEM <6.73 N/m; and MDC <18.65 N/m) were excellent. The ICC values for AT reliabilities ranged from 0.89 to 0.98. The ICC values for intraoperator reliability (ICC=0.89; 95% CI=0.68–0.97; SEM <37.44 N/m; and MDC <103.78 N/m) and interoperator reliability (ICC=0.98; 95% CI=0.93–0.99; SEM <31.00 N/m; and MDC <85.93 N/m) were excellent. In brief, all the intraoperator and interoperator reliabilities were excellent for the gastrocnemius and the AT of the lower limbs.
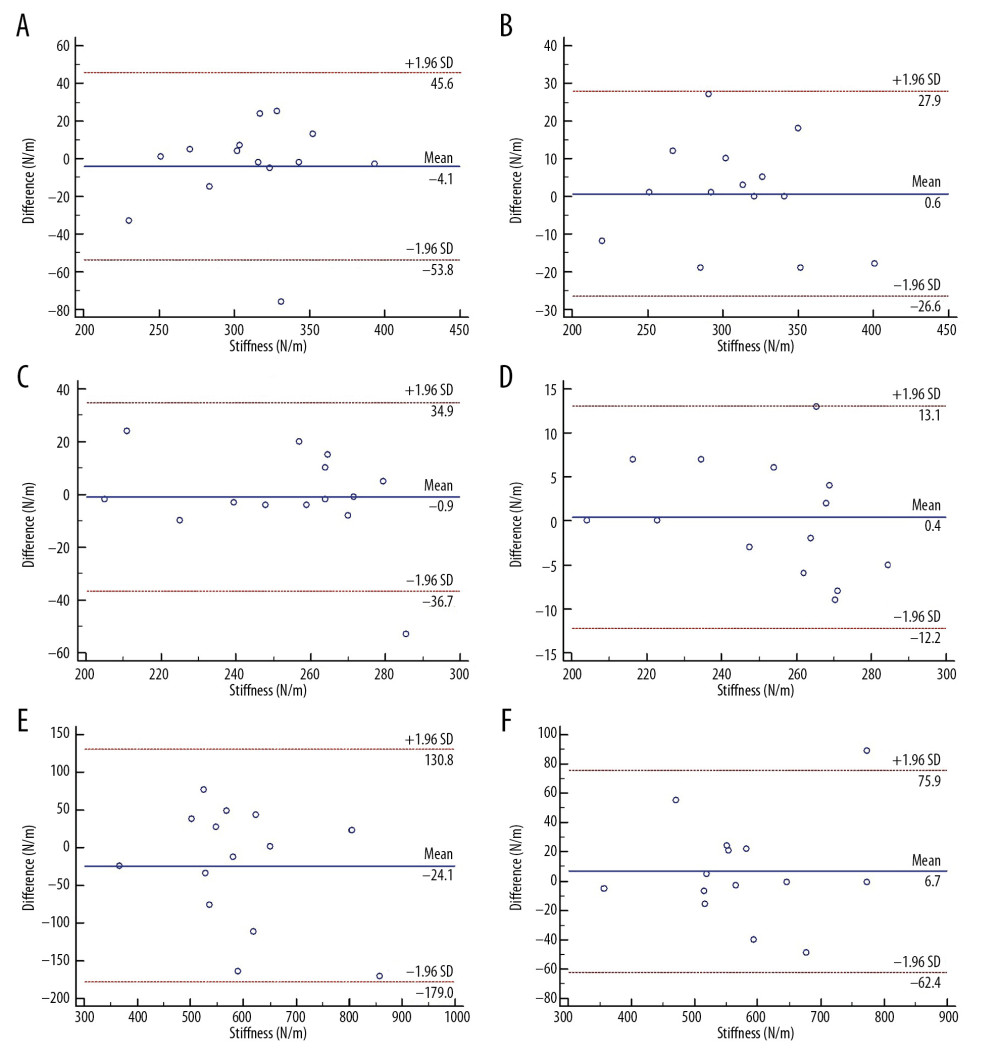
BLAND-ALTMAN ANALYSIS:
In Figure 1A–1F are the Bland-Altman plots of intraoperator and interoperator reliability of the LG, MG and AT, respectively. Almost all points are evenly distributed above and below the mean difference value in A, B, C and D, which shows no or less systematic bias in these measurements. The 95% limits of agreement (LoA) of A and Bare −53.8 to 45.6 and −26.6 to 27.9; the 95% LoA of C and D are −36.7 to 34.9 and −12.2 to 13.1; and, the 95% LoA of E and F are −179.0 to 130.8 and −62.4 to 75.9. There was only one outlier in the Bland-Altman plots of intraoperator reliability of the LG and MG, and no outliers in the Bland-Altman plots of interoperator reliability of the LG and MG. By contrast, there was no outlier in the Bland-Altman plots of intraoperator reliability of the AT, and only one outlier in the Bland-Altman plots of interoperator reliability of the AT.
Discussion
LIMITATION:
There were several limitations to this study. Although the measurement regions were marked by the same experienced therapist, the same position cannot be accurately determined by two measurements without using a waterproof marker. Only two of the subjects were women, thus, sex-based differences in the muscle and tendon stiffness could not be evaluated. Further studies should concentrate on comparing gender differences in muscle and tendon stiffness in patients after SCI.
Conclusions
This study assessed intraoperator and interoperator reliability of the MyotonPRO when used for patients with SCI, and it confirmed the reliability of the MyotonPRO for measuring muscle and tendon stiffness in this population. The standard error measurement (SEM) and the minimal detectable change (MDC) of stiffness measurements should be used in future clinical evaluations.
References
1. Van Den Berg M, Castellote JM, Mahillo-Fernandez I, Incidence of traumatic spinal cord injury in Aragón, Spain (1972–2008): Neurotrauma, 2011; 28; 469-77
2. Enrique BB, Méndez-Suárez JL, Alemán-Sánchez C, Change in the profile of traumatic spinal cord injury over 15 years in Spain: Scand J Trauma Resuscit Emerg Med, 2018; 26; 27
3. Diong J, Harvey LA, Kwah LK, Gastrocnemius muscle contracture after spinal cord injury: Am J Phys Med Rehabil, 2013; 92; 565-74
4. Diong JH, Herbert RD, Harvey LA, Passive mechanical properties of the gastrocnemius after spinal cord injury: Muscle Nerve, 2012; 46; 237-45
5. Eby S, Zhao H, Song P, Quantitative evaluation of passive muscle stiffness in chronic stroke: Am J Physical Med Rehabil, 2016; 95; 899-916
6. Fröhlich-Zwahlen AK, Casartelli NC, Item-Glatthorn JF, Validity of resting myotonometric assessment of lower extremity muscles in chronic stroke patients with limited hypertonia: A preliminary study: Electromyogr Kinesiol, 2014; 24; 762-69
7. Joghtaei M, Arab AM, Hashemi-Nasl H, Assessment of passive knee stiffness and viscosity in individuals with spinal cord injury using pendulum test: J Spinal Cord Med, 2015; 38; 170-77
8. Davidson MJ, Nielsen PMF, Taberner AJ, Kruger JA, Is it time to rethink using digital palpation for assessment of muscle stiffness?: Neurourol Urodyn, 2020; 39; 279-85
9. Fleuren JFM, Voerman GE, Erren-Wolters CV, Stop using the Ashworth Scale for the assessment of spasticity: J Neurology Neurosurg Psych, 2010; 81; 46-52
10. Siu WL, Chan CH, Lam CH, Sonographic evaluation of the effect of long-term exercise on Achilles tendon stiffness using shear wave elastography: J Sci Med Sport, 2016; 19; 883-87
11. Brandenburg JE, Eby SF, Song P, Feasibility and reliability of quantifying passive muscle stiffness in young children by using shear wave ultrasound elastography: J Ultrasound Med, 2015; 34; 663-70
12. Feng YN, Li YP, Liu CL, Assessing the elastic properties of skeletal muscle and tendon using shear wave ultrasound elastography and MyotonPRO: Sci Rep, 2018; 8; 17064
13. Liu CL, Feng YN, Zhang HQ, Assessing the viscoelastic properties of upper trapezius muscle: Intra- and inter-tester reliability and the effect of shoulder elevation: J Electromyogr Kinesiol, 2018; 43; 226-29
14. Liu CL, Li YP, Wang XQ, Quantifying the stiffness of achilles tendon: Intra- and inter-operator reliability and the effect of ankle joint motion: Med Sci Monit, 2018; 24; 4876-81
15. Ericson Morgan G, Martin R, Williams L, Objective assessment of stiffness in Achilles tendinopathy: A novel approach using the MyotonPRO: BMJ Open Sport Exercise Med, 2018; 4(1); e000446
16. Ambrose Lo WL, Zhao JL, Li L, Relative and absolute interrater reliabilities of a hand-held myotonometer to quantify mechanical muscle properties in patients with acute stroke in an inpatient ward: BioMed Res Int, 2017; 2017 4294028
17. Marusiak J, Jaskólska A, Koszewicz M, Myometry revealed medication–induced decrease in resting skeletal muscle stiffness in Parkinso’s disease patients: Clin Biomech, 2012; 27; 932-35
18. Drenth H, Zuidema SU, Krijnen WP, Psychometric properties of the myotonpro in dementia patients with paratonia: Gerontology, 2018; 64; 401-12
19. Walter SD, Eliasziw M, Donner A, Sample size and optimal designs for reliability studies: Stats Med, 1998; 17; 101-10
20. Huang JP, Qin K, Tang CZ, Assessment of passive stiffness of medial and lateral heads of gastrocnemius muscle, Achilles tendon, and plantar fascia at different ankle and knee positions using the MyotonPRO: Med Sci Monit, 2018; 24; 7570-76
21. Hirata K, Kanehisa H, Miyamoto N, Acute effect of static stretching on passive stiffness of the human gastrocnemius fascicle measured by ultrasound shear wave elastography: Eur J Appl Physiol, 2017; 117(3); 1-7
22. Masood T, Bojsen-Møller J, Kalliokoski K, Differential contributions of ankle plantarflexors during submaximal isometric muscle action: A PET and EMG study: J ElectromyogrKines, 2014; 24; 367-74
23. Zhou J, Yu J, Liu C, Regional elastic properties of the Achilles tendon is heterogeneously influenced by individual muscle of the gastrocnemius: Appl Bionics Biomech, 2019; 2019 845271
24. Dewall RJ, Slane LC, Lee KS, Spatial variations in Achilles tendon shear wave speed: J Biomech, 2014; 47; 2685-92
25. Aubry S, Nueffer JP, Tanter M, Viscoelasticity in Achilles tendonopathy: Quantitative assessment by using real-time shear-wave Elastography: Radiology, 2015; 274; 821-29
26. Portney LG, Watins MP, Foundations of clinical research: Applications to practice: J Hand Therapy, 2001; 14; 221
27. Bizzini M, Mannion AF, Reliability of a new, hand-held device for assessing skeletal muscle stiffness: Clin Biomech (Bristol, Avon), 2003; 18; 459-61
28. Taş S, Salkın Y, An investigation of the sex-related differences in the stiffness of the Achilles tendon and gastrocnemius muscle: Inter-observer reliability and inter-day repeatability and the effect of ankle joint motion: Foot (Edinb), 2019; 41; 44-50
29. Saeki J, Ikezoe T, Nakamura M, The reliability of shear elastic modulus measurement of the ankle plantar flexion muscles is higher at dorsiflexed position of the ankle: J Foot Ankle Res, 2017; 10; 18
30. Dubois G, Kheireddine W, Vergari C, Reliable protocol for shear wave elastography of lower limb muscles at rest and during passive stretching: Ultrasound Med Biol, 2015; 41; 2284-91
31. Lexell JE, Downham DY, How to assess the reliability of measurements in rehabilitation: Am J Phys Med Rehab, 2005; 84; 719-23
32. Hopkins WG, Measures of reliability in sports medicine and science: Sports Med, 2000; 30; 1-15
33. Leonardo F, Annette S, The applicability of standard error of measurement and minimal detectable change to motor learning research – a behavioral study: Front Hum Neurosci, 2018; 12; 95
34. Sohirad S, Wilosn D, Waugh C, Feasibility of using a hand-held device to characterize tendon tissue biomechanics: PLoS One, 2017; 12; e0184463
35. Nawarathna LS, Choudhary PK, Measuring agreement in method comparison studies with heteroscedastic measurements: Stat Med, 2013; 32; 5156-71
36. Ludbrook J, Confidence in Altman-Bland plots: A critical review of the method of differences: Clin Experiment Pharmacol Physiol, 2010; 37; 143-49
Tables
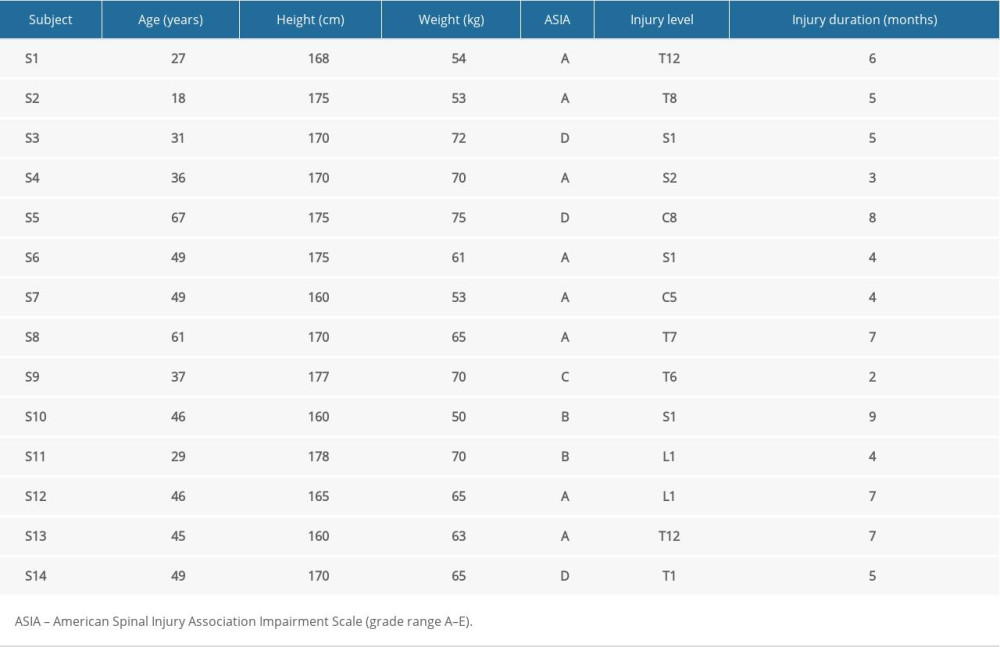 Table 1. Demographic data on subjects.
Table 1. Demographic data on subjects.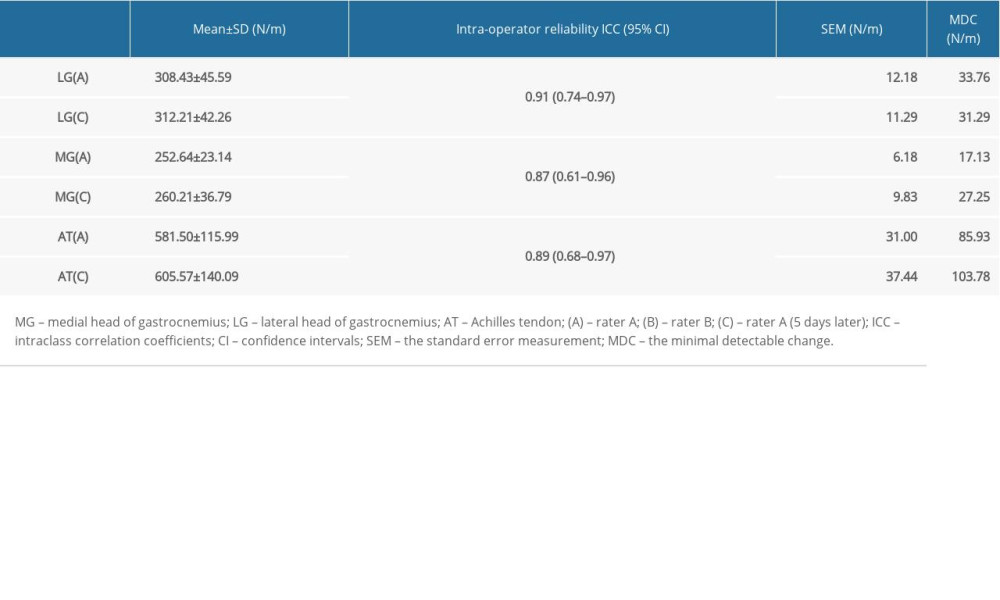 Table 2. Intraoperator reliability of muscle and tendon.
Table 2. Intraoperator reliability of muscle and tendon.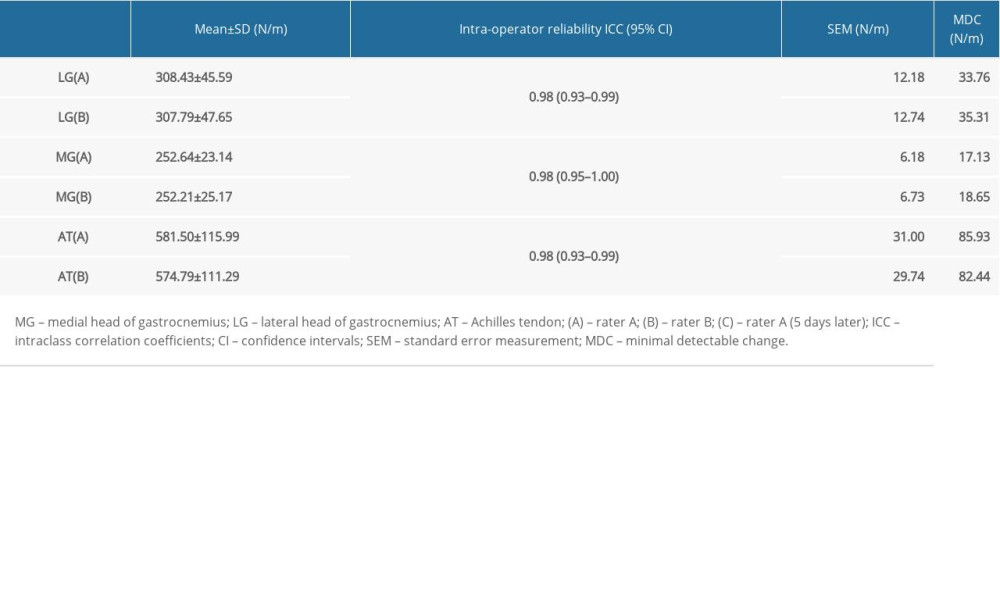 Table 3. Interoperator reliability of muscle and tendon.
Table 3. Interoperator reliability of muscle and tendon. Table 1. Demographic data on subjects.
Table 1. Demographic data on subjects. Table 2. Intraoperator reliability of muscle and tendon.
Table 2. Intraoperator reliability of muscle and tendon. Table 3. Interoperator reliability of muscle and tendon.
Table 3. Interoperator reliability of muscle and tendon. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387