23 July 2020: Clinical Research
Red Blood Cell Distribution Width is Associated with Glomerulonephritis in Diabetic Patients with Albuminuria
Tao Chen1ABDEF*, Xuchu Wang2AC, Qihua Bi1ACGDOI: 10.12659/MSM.924923
Med Sci Monit 2020; 26:e924923
Abstract
BACKGROUND: The aim of this study was to explore predictive factors to inform accurate diagnosis of glomerulonephritis (GNs) in patients with diabetes.
MATERIAL AND METHODS: Clinical characteristics and laboratory data were retrospectively analyzed from 200 patients with diabetes including 115 patients who had undergone a renal biopsy. Eligible patients were categorized into three groups: pure type 2 diabetes mellitus (T2DM), isolated diabetic nephropathy (DN), and GN. Odds ratios (ORs) were calculated to evaluate the contributions of predictive factors for GN. A receiver operating characteristic curve (ROC) was created to obtain cut-off values for predictive factors for GNs and investigate their corresponding predictive accuracy.
RESULTS: Red cell distribution width (RDW) was significantly higher in the GN group than in the DN group. Multivariate regression analysis revealed that baseline RDW level (OR=1.988, 95% CI=1.237~3.194, P=0.005) was an independent predictive factor for development of GNs.
CONCLUSIONS: Increased RDW levels are independently associated with a greater risk of GN in patients with diabetes who have albuminuria, and may be an additional valuable and noninvasive predictive tool for differentiating GNs and DN.
Keywords: Erythrocyte Indices, Glomerulonephritis, Albuminuria, Diabetes Complications, Diabetes Mellitus, Erythrocytes, Odds Ratio, ROC Curve, Risk Factors
Background
With the rising prevalence of diabetes mellitus (DM), diabetic nephropathy (DN) is one of the most severe complications of DM and a primary cause of end-stage renal disease (ESRD) [1], and has become a heavy economic burden on the health care system worldwide [2]. While albuminuria is an independent risk factor for end-stage diabetic nephropathy (DN) [3], it also occurs in patients with diabetes and individuals with non-diabetic renal diseases (NDRDs). Some studies suggest that prevalence of DN is only less than 50% in patients with DM undergoing renal biopsy [4,5]. Among individuals diagnosed with NDRD, the most common pathologic type is glomerulonephritis (GN) [4,5]. Generally, the treatment strategy and prognosis for DN and glomerulonephritis are quite different. For instance, steroids are recommended in the Kidney Disease: Improving Global Outcomes (KDIGO) Clinical Practice Guideline for glomerulonephritis, but they may exacerbate diabetic nephropathy [6,7]. Thus, accurate differential diagnosis of glomerulonephritis from DN is of prime importance.
Renal biopsy remains the diagnostic gold-standard and enables reliable diagnosis of glomerulonephritis, but it is invasive. Therefore, a way to use available clinical and laboratory data prior to biopsy to identify glomerulonephritis is urgently needed. Typically, absence of clinical features such as diabetic retinopathy, diabetic neuropathy or short duration of DM suggest a lower likelihood of DN in patients with diabetes, and provide a stronger clinical rationale for renal biopsy. Multivariant predictive models based on such clinical parameters have been constructed to facilitate differentiation of GN from DN [4,8,9]. The results, however, have not been uniform, mainly due to differences in the study populations or selection criteria. Presence of microhematuria has been suggested as an indicator for renal biopsy in patients with diabetes who have albuminuria [10,11]. However, a comprehensive analysis by Li and coworkers [12] showed that the diagnostic sensitivity of hematuria for NDRDs in type 2 diabetes (T2DM) with kidney disease was only 42%, indicating a lack of adequate evidence for either to predicting GN or exclude isolated DN.
In this study, we aimed to investigate the association between red cell distribution width (RDW) levels and presence of GN in patients with diabetes who have albuminuria, and to explore potential predictive roles of RDW in identifying GN in patients with diabetes who are diagnosed with albuminuria.
Material and Methods
PATIENTS RECRUITMENT:
Adult patients with T2DM from Zhejiang hospital were enrolled from February 2013 to December 2019. Exclusion criteria were the following: (1) incomplete medical history or clinical examination results; and (2) serious infectious disease, tumor or other serious conditions. Two hundred patients with diabetes were finally enrolled, including 130 with albuminuria and 70 without albuminuria. This retrospective observational study was based on anonymized patient data. The study protocol was approved by the Human Ethics Review Committee of Zhejiang Hospital. The requirement for informed consent was waived due to the observational, retrospective nature of the study.
DIAGNOSTIC CRITERIA:
Diagnosis of T2DM was based on the standard from the World Health Organization [13], and diagnosis of diabetic nephropathy was based on the standards of the 2010 Renal Pathology Society (RPS) [14]. GN is a group of kidney diseases and defined on the basis of The 2012 KDIGO practice guideline [6]. Albuminuria was defined based on urinary albumin/creatinine ratio (ACR) higher than 2.5 mg/mmol (male) or 3.5 mg/mmol (female) [15].
CLINICAL AND LABORATORY DATA:
Clinical information and laboratory data were extracted within 1 month before renal biopsy, including gender, age, medical history of DM, diabetes duration, medical history of hypertension, pharmacotherapy, serum glucose, glycated hemoglobin (HbA1c), estimated glomerular filtration rate (eGFR; calculated using the CKD epidemiology formula), urinary albumin, urinary creatinine, hypersensitive C-reaction protein (Hs-CRP) and hematological data such as white blood cell (WBC) count, red blood cell (RBC) count, hemoglobin (Hb), mean corpuscular volume (MCV), and red blood cell distribution width (RDW).
STATISTICAL ANALYSIS:
Continuous clinical data were presented as mean±standard deviation (SD) or median (interquartile range) (IQR). The intergroup comparison was assessed by student’s
Results
BASELINE CHARACTERISTICS AND LABORATORY DATA:
Clinical and laboratory data on all patients with diabetes are shown in Table 1. Of 200 patients with T2DM enrolled in our study, 65% were diagnosed with albuminuria. No statistically significant relationship was observed between disease duration and incidence of albuminuria. Notably, Hs-CRP was significantly higher in patients with albuminuria than in patients without albuminuria, and patients with albuminuria suffered worse renal function. Of the 130 patients with albuminuria, 115 patients have undergone renal biopsy including 57 patients with isolated diabetic nephropathy and 44 patients with GN. Mean duration of diabetes in all patients was 11.66 years. Patients diagnosed with GN had a shorter duration of diabetes than did patients with DN (Table 2). Patients with GN were less likely to have hypertension than were patients with DN (P=0.023), which may be due to the longer duration of diabetes in the latter patients (r=0.518, P<0.001) (data not shown). Nevertheless, no significant difference in urinary albumin was observed between patients with different stages of CKD (data not shown).
HEMATOLOGICAL FEATURES IN DIABETIC PATIENTS WITH ALBUMINURIA:
Much higher RDW levels were observed in GN compared with isolated DN in patients with diabetes who had albuminuria (Table 2, Figure 1A). A univariate model showed that RDW had significant predictive value for GN (OR=1.829, 95% CI=1.274~2.626, P<0.001). It has been reported that eGFR may have an influence on RDW, making eGFR a confounding factor in the context of higher RDW levels in patients with GN compared with patients with DN who have albuminuria. We also found a significant correlation between GFR and levels of Hb and RDW values, with correlation coefficients of 0.499 (95% CI=0.354~0.632, P<0.001) and −0.187 (95% CI=−0.328~−0.017, P=0.046), respectively (Figure 1B, 1C). That may be a result of less reduced erythropoietic activity secondary to impaired renal function [16]. Therefore, we constructed multivariate model to rule out the possibility that the associations between hematological features and presence of GN in patients with diabetes might be affected by confounding factors. Accordingly, when adjusted for age, gender, eGFR, hypertensive status, and diabetic duration, the association between RDW values and presence of GN remained statistically significant (OR=1.988, 95% CI=1.237~3.194, P=0.005).
A receiver operating curve (ROC) analysis was performed to determine the cutoff value of RDW for predicting GN versus DN in patients with diabetes who have albuminuria. The area under the ROC curve (AUC) for RDW was 0.697 (95% CI: 0.592~0.801, P<0.001), and an RDW higher than 13.95 predicted GN in patients with diabetes with a sensitivity of 61.36% and specificity of 70.18% (Figure 2A). Furthermore, a decision curve analysis (DCA) based on the net benefit for RDW-based tests showed that using RDW levels to predict GN in patients with diabetes who had albuminuria might add more clinical benefit than treating or omitting all patients with albuminuria (Figure 2B).
Discussion
GN is a group of complex and heterogeneous diseases, caused by different underlying pathological conditions, that include primary forms such as IgA nephropathy and membranous GN [17], or occur as a consequence of systemic diseases such as lupus nephritis and infections [18,19]. In patients with diabetes who have albuminuria, GN has been shown to account for a great proportion of renal biopsies [4,5]. Most patients with diabetes are unwilling to undergo renal biopsy, and do so only if their physicians strongly recommend further diagnostic examination when albuminuria is suspected and is unlikely to be caused by diabetic nephropathy. Therefore, a noninvasive diagnostic indicator for GN prior to renal biopsy is desirable for patients with diabetes who are diagnosed with albuminuria. A number of studies show that misdirected infiltration of inflammatory cells in renal glomerulus and tubulointerstitium is found in most forms of GN regardless of their etiologies [20–22]. Thus, GN has been regarded as resulting from inflammation, such that various inflammatory factors are reported as possible predictors of disease activity and for diagnosis in patients with GN [23]. In our study, Hs-CRP higher than 1.89 mg/L was found to positively contribute to incidence of GN in patients with diabetes who had albuminuria, with an OR of 3.695 (95% CI: 1.000–10.486, P<0.05).
Red cell distribution width (RDW), part of complete blood count, is a common hematological parameter reflecting the heterogeneity of circulating RBC size. While RDW serves as a differential indicator for anemias, especially for diagnosis of iron deficiency [24], it has also been revealed to be altered under many clinical conditions associated with inflammation other than anemia [25,26]. An elevated RDW can be observed in patients who have diseases representing inflammatory states, such as systemic lupus erythematosus [27], rheumatoid arthritis [28] and cardiovascular disease [29]. Our study also suggests that high RDW is significantly associated with inflammatory factors (such as Hs-CRP) in patients with diabetes who have GN, with a correlation coefficient of 0.285 (95% CI: 0.075–0.466, P<0.01). In addition, Hs-CRP positively contributed to higher RDW (>13.95) with an OR of 3.444 (95% CI: 1.322–8.972, P<0.05) in patients with diabetes who had albuminuria, and combining Hs-CRP with RDW could significantly increase the predictive ability, with an AUC of 0.735 (95% CI: 0.593~0.877, P<0.005) compared with RDW only (Figure 2A). Even though other circulating cytokines such as TNF-α or interleukin 6 were not involved in the study, we believe that the molecular mechanism of the above-mentioned relationships between RDW and incidence of GN may be mainly due to the roles of RDW in patients with inflammatory status and reliably reflect increasing levels of circulating cytokines [25,30].
This is the first study investigating the relationship between baseline RDW and GN incidence in patients with diabetes. The main finding is that when a patient with diabetes is diagnosed with albuminuria, increased RDW level at baseline before renal biopsy are significantly associated with increased risk of GN rather than of DN. This is of prime clinical importance as physicians may be inclined to proceed with renal biopsy in patients with diabetes who have albuminuria if a higher RDW is observed, while considering that for the remainder of patients, renal biopsy would represent a high unnecessary burden, both physical and mental. Results of our study also indicate that the predictive value of RDW mainly may be attributable to the inflammatory microenvironment in patients with diabetes who have GN. Another study has suggested that baseline RDW value is also a promising novel biomarker for predicting response to treatment in adult patients with nephrotic syndrome due to primary glomerulonephritides [31], which further underscores the clinical significance of RDW.
It is important to note that some studies have revealed that inflammatory conditions also exist in pure DN [32,33], and multiple inflammatory factors can be rationally applied to identify DN [34–36]. Similarly, our study showed statistically significant higher levels of Hs-CRP in patients with DN compared with patients who had DM without nephropathy. RDW levels higher than 13.4% also significantly contributed to DN in patients with T2DM (P<0.05), which may be associated with decreased RBC deformability that is caused by increased inflammation and decreased levels of antioxidants [37–39]. Nevertheless, as an exploration of noninvasive differentiation of nephropathy associated with DM versus that caused by other factors in patients with DM, our results showed that RDW yielded high predictive capacities for GN.
Conclusions
In conclusion, our study suggests that a high level of RDW is independently associated with increased risk of GN patients with diabetes who have albuminuria. RBC distribution width may predict possible existence of GN when a patient with diabetes is found to have albuminuria, and thus, to some extent, prevent unnecessary renal biopsies.
Figures
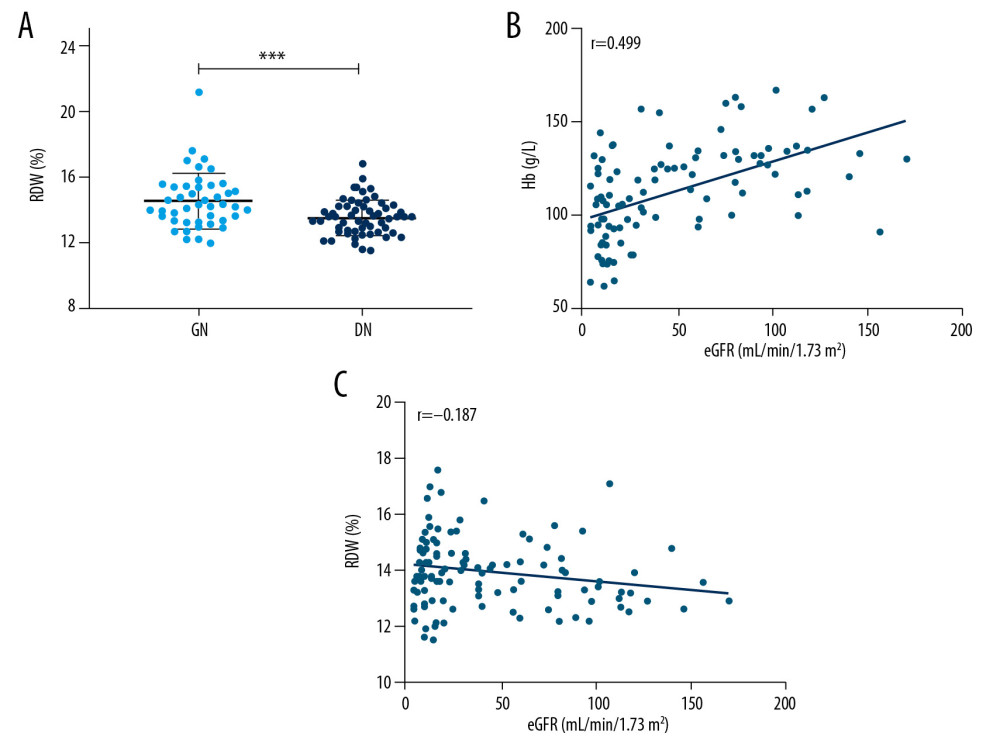 Figure 1. (A) Comparison of RBC distribution width (RDW) levels between the GNs and DN. (B) Correlation between eGFR and Hb. (C) Correlation between eGFR and RDW. *** P<0.001.
Figure 1. (A) Comparison of RBC distribution width (RDW) levels between the GNs and DN. (B) Correlation between eGFR and Hb. (C) Correlation between eGFR and RDW. *** P<0.001. 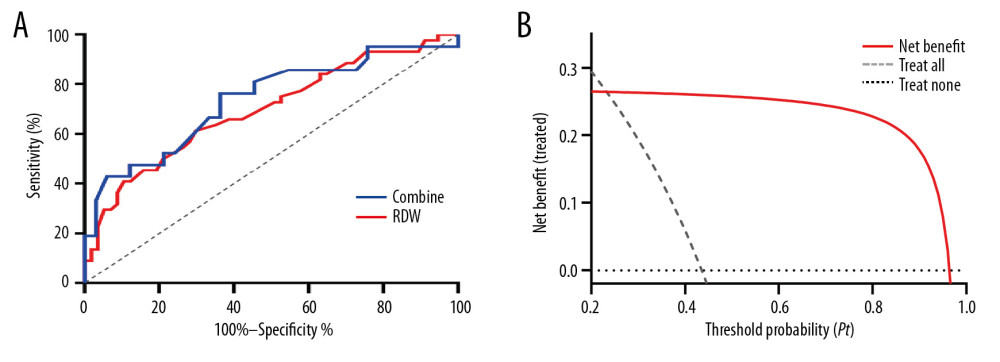 Figure 2. (A) Receiver operating characteristic curve (ROC) of RDW and combined indicator (RDW and Hs-CRP) for prediction of GN in patients with diabetes who have albuminuria. (B) Decision curve analysis: The net benefit when using RDW as predictor for GN.
Figure 2. (A) Receiver operating characteristic curve (ROC) of RDW and combined indicator (RDW and Hs-CRP) for prediction of GN in patients with diabetes who have albuminuria. (B) Decision curve analysis: The net benefit when using RDW as predictor for GN. References
1. Zimmet P, Alberti KG, Shaw J, Global and societal implications of the diabetes epidemic: Nature, 2001; 414; 782-87
2. Thomas B, The global burden of diabetic kidney disease: Time trends and gender gaps: Curr Diab Rep, 2019; 19; 18
3. Atkins RC, Briganti EM, Lewis JB, Proteinuria reduction and progression to renal failure in patients with type 2 diabetes mellitus and overt nephropathy: Am J Kidney Dis, 2005; 45; 281-87
4. Dong Z, Wang Y, Qiu Q, Clinical predictors differentiating non-diabetic renal diseases from diabetic nephropathy in a large population of type 2 diabetes patients: Diabetes Res Clin Pract, 2016; 121; 112-18
5. Zhuo L, Ren W, Li W, Evaluation of renal biopsies in type 2 diabetic patients with kidney disease: A clinicopathological study of 216 cases: Int Urol Nephrol, 2013; 45; 173-79
6. Radhakrishnan J, Cattran DC, The KDIGO practice guideline on glomerulonephritis: Reading between the (guide)lines – application to the individual patient: Kidney Int, 2012; 82; 840-56
7. Winocour P, Bain SC, Chowdhury TA, Managing hyperglycaemia in patients with diabetes and diabetic nephropathy – chronic kidney disease: Summary of recommendations, 2018; 2018
8. Liang S, Zhang XG, Cai GY, Identifying parameters to distinguish non-diabetic renal diseases from diabetic nephropathy in patients with Type 2 diabetes mellitus: A meta-analysis: PLoS One, 2013; 8; e64184
9. Liu MY, Chen XM, Sun XF, Validation of a differential diagnostic model of diabetic nephropathy and non-diabetic renal diseases and the establishment of a new diagnostic model: J Diabetes, 2014; 6; 519-26
10. Hommel E, Carstensen H, Skott P, Prevalence and causes of microscopic haematuria in type 1 (insulin-dependent) diabetic patients with persistent proteinuria: Diabetologia, 1987; 30; 627-30
11. American Diabetes Association, Clinical practice recommendations 2005: Diabetes Care, 2005; 28(Suppl 1); S1-79
12. Jiang S, Wang Y, Zhang Z, Accuracy of hematuria for predicting non-diabetic renal disease in patients with diabetes and kidney disease: A systematic review and meta-analysis: Diabetes Res Clin Pract, 2018; 143; 288-300
13. Alberti KG, Zimmet PZ, Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus provisional report of a WHO consultation: Diabet Med, 1998; 15; 539-53
14. Tervaert TW, Mooyaart AL, Amann K, Pathologic classification of diabetic nephropathy: J Am Soc Nephrol, 2010; 21; 556-63
15. Johnson DW, Global proteinuria guidelines: Are we nearly there yet?: Clin Biochem Rev, 2011; 32; 89-95
16. Ferrucci L, Guralnik JM, Woodman RC, Proinflammatory state and circulating erythropoietin in persons with and without anemia: Am J Med, 2005; 118; 1288
17. Floege J, Amann K, Primary glomerulonephritides: Lancet, 2016; 387; 2036-48
18. Flores-Mendoza G, Sanson SP, Rodriguez-Castro S, Mechanisms of tissue injury in lupus nephritis: Trends Mol Med, 2018; 24; 364-78
19. Cacoub P, Desbois AC, Isnard-Bagnis C, Hepatitis C virus infection and chronic kidney disease: Time for reappraisal: J Hepatol, 2016; 65; S82-94
20. Kuriakose J, Redecke V, Guy C, Patrolling monocytes promote the pathogenesis of early lupus-like glomerulonephritis: J Clin Invest, 2019; 130; 2251-65
21. Huang J, Filipe A, Rahuel C, Lutheran/basal cell adhesion molecule accelerates progression of crescentic glomerulonephritis in mice: Kidney Int, 2014; 85; 1123-36
22. Andersen K, Eltrich N, Lichtnekert J, The NLRP3/ASC inflammasome promotes T-cell-dependent immune complex glomerulonephritis by canonical and noncanonical mechanisms: Kidney Int, 2014; 86; 965-78
23. Aizawa T, Imaizumi T, Tsuruga K, Urinary fractalkine and monocyte chemoattractant protein-1 as possible predictors of disease activity of childhood glomerulonephritis: Tohoku J Exp Med, 2013; 231; 265-70
24. Bessman JD, More on the RDW: Am J Clin Pathol, 1985; 84; 773
25. Lippi G, Targher G, Montagnana M, Relation between red blood cell distribution width and inflammatory biomarkers in a large cohort of unselected outpatients: Arch Pathol Lab Med, 2009; 133; 628-32
26. Fornal M, Wizner B, Cwynar M, Association of red blood cell distribution width, inflammation markers and morphological as well as rheological erythrocyte parameters with target organ damage in hypertension: Clin Hemorheol Microcirc, 2014; 56; 325-35
27. Zou XL, Lin XJ, Ni X, Baseline red blood cell distribution width correlates with disease activity and therapeutic outcomes in patients with systemic lupus erythematosus, irrespective of anemia status: Clin Lab, 2016; 62; 1841-50
28. Lee WS, Kim TY, Relation between red blood cell distribution width and inflammatory biomarkers in rheumatoid arthritis: Arch Pathol Lab Med, 2010; 134; 505-6
29. Lippi G, Filippozzi L, Montagnana M, Clinical usefulness of measuring red blood cell distribution width on admission in patients with acute coronary syndromes: Clin Chem Lab Med, 2009; 47; 353-57
30. Forhecz Z, Gombos T, Borgulya G, Red cell distribution width in heart failure: prediction of clinical events and relationship with markers of ineffective erythropoiesis, inflammation, renal function, and nutritional state: Am Heart J, 2009; 158; 659-66
31. Turgutalp K, Kıykım A, Bardak S, Is the red cell distribution width strong predictor for treatment response in primary glomerulonephritides?: Renal Fail, 2014; 36; 1083-89
32. Gurley SB, Ghosh S, Johnson SA, Inflammation and immunity pathways regulate genetic susceptibility to diabetic nephropathy: Diabetes, 2018; 67; 2096-106
33. Wada J, Makino H, Inflammation and the pathogenesis of diabetic nephropathy: Clin Sci (Lond), 2013; 124; 139-52
34. Zhang D, Ye S, Pan T, The role of serum and urinary biomarkers in the diagnosis of early diabetic nephropathy in patients with type 2 diabetes: Peer J, 2019; 7; e7079
35. Liu Q, Jiang CY, Chen BX, The association between high-sensitivity C-reactive protein concentration and diabetic nephropathy: A meta-analysis: Eur Rev Med Pharmacol Sci, 2015; 19; 4558-68
36. Li L, Jiang XG, Hu JY, The association between interleukin-19 concentration and diabetic nephropathy: BMC Nephrol, 2017; 18; 65
37. Patel KV, Mohanty JG, Kanaparu B, Association of the red cell distribution width with red blood cell deformability: Adv Exp Med Biol, 2013; 765; 211-16
38. Magri CJ, Fava S, Red blood cell distribution width and diabetes-associated complications: Diabetes Metab Syndr, 2014; 8; 13-17
39. Semba RD, Patel KV, Ferrucci L, Serum antioxidants and inflammation predict red cell distribution width in older women: The Women’s Health and Aging Study I: Clin Nutr, 2010; 29; 600-4
Figures
 Figure 1. (A) Comparison of RBC distribution width (RDW) levels between the GNs and DN. (B) Correlation between eGFR and Hb. (C) Correlation between eGFR and RDW. *** P<0.001.
Figure 1. (A) Comparison of RBC distribution width (RDW) levels between the GNs and DN. (B) Correlation between eGFR and Hb. (C) Correlation between eGFR and RDW. *** P<0.001. Figure 2. (A) Receiver operating characteristic curve (ROC) of RDW and combined indicator (RDW and Hs-CRP) for prediction of GN in patients with diabetes who have albuminuria. (B) Decision curve analysis: The net benefit when using RDW as predictor for GN.
Figure 2. (A) Receiver operating characteristic curve (ROC) of RDW and combined indicator (RDW and Hs-CRP) for prediction of GN in patients with diabetes who have albuminuria. (B) Decision curve analysis: The net benefit when using RDW as predictor for GN. Tables
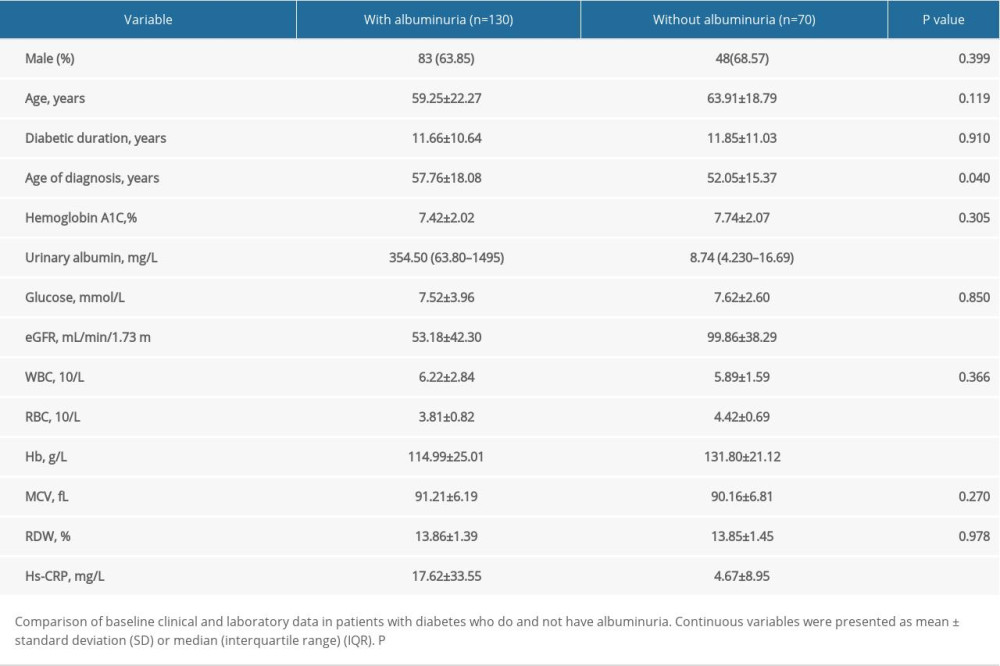 Table 1. Comparison of baseline data in diabetic patients with and without albuminuria.
Table 1. Comparison of baseline data in diabetic patients with and without albuminuria.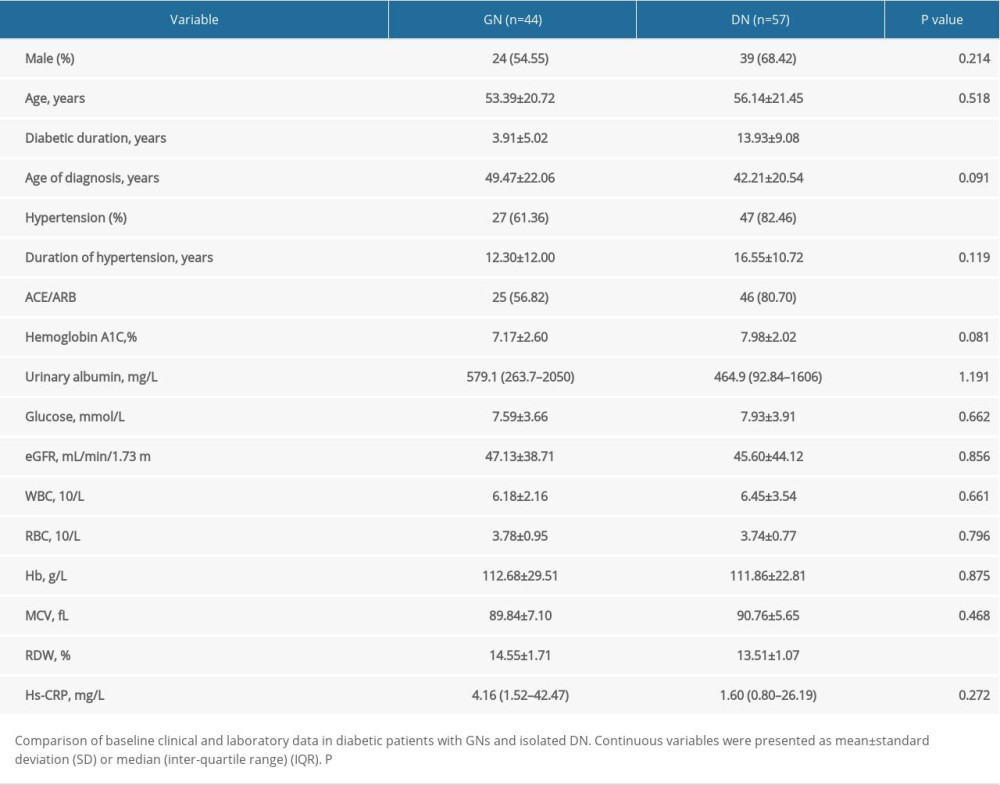 Table 2. Comparison of baseline data in diabetic patients with GNs and isolated DN.
Table 2. Comparison of baseline data in diabetic patients with GNs and isolated DN. Table 1. Comparison of baseline data in diabetic patients with and without albuminuria.
Table 1. Comparison of baseline data in diabetic patients with and without albuminuria. Table 2. Comparison of baseline data in diabetic patients with GNs and isolated DN.
Table 2. Comparison of baseline data in diabetic patients with GNs and isolated DN. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








