08 September 2020: Clinical Research
Lower Metal Element Levels in Hypertrophic Scars: A Potential Mechanism of Aberrant Cicatrix Hyperplasia
Qifei Wang1BCE, Mi Miao1BCDF, Zelian Qin1ADEG*, Bolun Li1F, Xingtao Niu1ADOI: 10.12659/MSM.925202
Med Sci Monit 2020; 26:e925202
Abstract
BACKGROUND: We investigated levels of the metal elements Ca, Mg, Zn, Fe, and Cu in blood, normal skin (NS), and different types of scar tissue and aimed to elucidate the pathogenesis of hypertrophic scars (HS).
MATERIAL AND METHODS: Tissue specimens were excised from 3 groups of research participants: scar-free, flat scar (FS), and HS groups. Levels of the study elements were measured in blood, NS, and scar tissues with a spectrophotometer. The levels in plasma or in different types of specimens were compared among subgroups. In the FS and HS groups, levels were compared between the scar tissue and NS of each individual. In addition, element differences in exposed and unexposed areas of NS were investigated in the scar-free group. HS fibroblasts (HFB) were cultured in medium with various reduced levels of metal elements to determine the influence of metal elements on fibroblast growth.
RESULTS: Levels of trace elements, including Zn, Fe, and Cu, were significantly lower in HS than in FS. The levels of Ca, Zn, Fe, and Cu were markedly lower in HS than in the patients’ own NS, while the Cu/Zn ratio was higher. However, no such difference was observed in the FS group. No significant difference in element levels was found in either plasma or NS among the 3 groups. Reduced levels of the elements promoted HFB proliferation within 24 h while an inhibition effect was observed at 72 h.
CONCLUSIONS: Our findings indicate reduced levels of metal elements in part of the healing microenvironment, suggesting that decreased metal levels may be involved in the pathogenesis of HS.
Keywords: Pathology, Skin Diseases, Trace Elements, Adolescent, Child, Child, Preschool, Cicatrix, Cicatrix, Hypertrophic, young adult
Background
Hypertrophic scars (HS) are characterized by aberrant deposition of collagen and excessive cell proliferation and present intractable challenges in clinical treatment [1–3]. As a common type of scar, the incidence of HS is as high as 80% after burn injuries and 40% to 70% after trauma [4]. Individuals with HS experience physical and psychological problems, such as pain, pruritus, dysfunction induced by contracture, and psychological distress [2]. Although surgery, compression therapy, steroid injection, and radiotherapy are beneficial to prevent or relieve HS, each therapy has certain limitations and multiple treatments are required [5,6]. Accordingly, investigating the pathogenesis of HS to provide new therapies is highly desirable.
Metal elements, such as Ca, Mg, Zn, Fe, and Cu, play essential roles in cellular physiological activities in organisms. They are extensively involved in DNA synthesis, protein synthesis, cellular proliferation, collagen production, and crosslinking [7]. Reductions in O2, metal elements, and other nutritive factors in wound tissues, such as tissue affected by third degree burn injury, alter fibroblast function and break the balance between collagen synthesis and degradation. Overproduction of free radicals, collagen overdeposition, and hypertrophic scar formation thus occur [8,9]. In addition, all 5 elements are strongly related to collagen synthesis and degradation in skin tissue [10]. Moreover, the status of these elements has a substantial influence on wound healing and scar formation [11,12]. Changes in trace elements in the context of injuries [10,13] and beneficial effects of trace element supplementation after injuries [14] have also been widely reported.
Although alteration of metal elements has been confirmed to be significantly associated with several diseases [15–17], its association with common scars is not well investigated. A study of trace elements in keloids and HS was previously performed, but no significant difference was found between the 2 types of scars, possibly due to the limited number of participants [18]. Whether the local environment affects metal element concentrations is still unknown. In the present study, we examined the element content in plasma, skin, and scars of individuals in scar-free, flat scar (FS), and HS groups and investigated whether altered metal element content is involved in the pathogenesis of HS. Interestingly, reduced metal element levels in the healing microenvironment and not the body as a whole were demonstrated. The findings enhance the understanding of the influence of metal levels in the healing microenvironment on the pathogenesis of HS.
Material and methods
PARTICIPANTS:
All patients hospitalized in the Peking University Third Hospital were divided into 3 groups: the scar-free, FS, and HS groups (Table 1). In the scar-free group, normal skin (NS) specimens were derived from traumatic or aesthetic operations. Twelve of the specimens were taken from exposed areas (face, hand, neck), and 10 were taken from hidden areas (trunk, proximal limbs). In the FS group (n=12), scars had been present for 2 to 20 years. No patients presented conspicuous symptoms related to their scars. Operations were conducted solely for aesthetic purposes. In the HS group (n=16), scars had been present for 0.5 to 11 years. All patients had typical symptoms of redness, itch, ache, and elevation at the location of their HS, while their scars came from deep second or third degree burns. None of the patients in any of the 3 groups had occupational contacts with metals or familial predisposition to cicatrix. Moreover, all patients who had received treatments with medications or radiotherapy before surgery were excluded.
This study was approved by the institutional ethics committee of Peking University (0580) and followed the principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all participants.
COLLECTION AND PRESERVATION OF SPECIMENS:
Whole blood (1.0 mL) was collected into an anticoagulation tube prior to surgery. Plasma was obtained by removing blood cells after centrifugation (3000 rpm, 15 min) and then preserved at −80°C for determination of metal concentrations. All skin specimens were obtained during surgery, and subcutaneous tissues were discarded. The specimens (0.5×1.0 cm/epidermic square) were preserved at −80°C. All samples were analyzed within 4 weeks.
PRIMARY FIBROBLAST CULTURE:
HS fibroblasts (HFB) and NS fibroblasts (NFB) were isolated from surgically excised HS and NS as reported previously [19]. The epidermis and subcutaneous adipose tissue were removed, and the remaining tissue was washed 2 times with phosphate-buffered saline. Specimens were cut into 1 to 3 mm3 pieces and digested in 0.15% collagenase I at 37°C for 2 h. The digested suspension was filtered and centrifuged at 1000 rpm for 5 min. The fibroblast pellets were cultured in Dulbecco’s modified Eagle’s medium (DMEM; HyClone, USA) supplemented with 10% fetal bovine serum (Gibco, USA) with 1% penicillin/streptomycin in an incubator at 37°C and 5% CO2. Upon reaching 80% to 90% confluence, the fourth to sixth passages of fibroblasts were used to perform the experiments in this study.
METAL ELEMENT CONTENT DETERMINATION:
The preprocessing of blood samples was performed according to the routine steps used for soft tissues. After acidic dissolution through heating, the supernatant was diluted (1: 10) with double distilled and deionized water, and the volume was adjusted to 10 mL before detection. All scar tissue and NS samples were weighed and digested by acids, dissolved in nitric acid medium, and diluted (1: 10) with double distilled and deionized water to adjust the volume to 10 mL. Element contents were determined with GGX-5 atomic absorption spectrophotometry [20] (Beijing Geological Instruments Factory) in the Test Center of the Metallurgy Ministry at Yan Jiao (the second grade qualified unit authenticated by the National Quality Department of China). The wavelengths used for measurement were 422.7 nm for Ca, 285.2 nm for Mg, 213.9 nm for Zn, 248.3 nm for Fe, and 324.8 nm for Cu. The formula for calculation was Csp=(Asp×Csd/Wsp)×Vsp, where Csp is the element content of sample (μg/g); Asp, sample absorbance; Csd, the element content of standard (μg/mL); Wsp, sample weight (g); and Vsp, sample volume (10 mL).
PROLIFERATION ASSAY:
Briefly, HFB and NFB were seeded into 96-well plates at an initial density of 2500 cells/well with DMEM containing 0, 3.125, 12.5, 50, or 200 μmol/L ethylene diamine tetraacetic acid (EDTA) and cultured for 24, 48, and 72 h. Ten microliters of CCK8 was added to each well, and plates were then incubated at 37°C for 2 h. Absorbance was measured at 450 nm (indicating cell proliferation).
STATISTICAL ANALYSIS:
Statistical analyses were performed using SPSS version 24 (IBM, Armonk, NY, USA). All data are expressed as the mean±standard deviation (SD). One-way ANOVA was used for the comparison of data among the 3 study groups. In the FS and HS groups, a paired
Results
COMPARISON OF ELEMENT CONTENTS IN NS FROM EXPOSED AND NONEXPOSED AREAS IN THE SCAR-FREE GROUP:
To determine whether exposure to the environment affected metal element levels in skin tissue, we measured element contents in exposed and hidden skin areas of 12 and 10 subjects, respectively, in the scar-free group. We found no significant differences in element contents between exposed and hidden areas of NS in this group (Table 2).
COMPARISON OF ELEMENT CONTENTS IN BLOOD AND NS IN 3 GROUPS:
To investigate whether the element levels in blood and NS differed among the 3 groups, we detected the 5 study elements by atomic absorption spectrophotometry. Similar levels of the 5 elements were observed in NS (Figure 1A) and blood (Figure 1B) among the 3 groups.
COMPARISON OF ELEMENT CONTENTS IN SCARS AND NS IN THE HS AND FS GROUPS:
Element contents were significantly lower in HS tissues than in NS for individuals in the HS group. The average levels of Ca, Mg, Zn, Fe, and Cu were 16.37%, 12.95%, 70.94%, 65.82%, and 64.17% lower in HS tissues than in NS tissues, respectively (Table 3, Figure 2). However, no significant differences in element levels were detected between FS tissues and each participant’s own NS in the FS group (Figure 3). The average Zn, Fe, and Cu levels were lower in HS tissues than in FS tissues; the average levels were 69.04%, 67.12%, and 62.67% lower for Zn, Fe, and Cu, respectively (Table 3).
CU/ZN AND CA/MG RATIOS IN THE HS AND FS GROUPS:
In the HS group, the Cu/Zn ratio in scar tissues (0.35±0.05) was 25% higher than in NS (0.28±0.04) (P<0.01). However, the difference was not significant in the FS group (0.30±0.06 in FS vs. 0.29±0.05 in NS) (P>0.05, Figure 4A). The Ca/Mg ratio was not significantly different between each type of scar tissue and patients’ own NS (1.53±0.24 in NS of the scar-free group; 1.74±0.49 in NS and 1.64±0.44 in FS of the FS group; 1.76±0.47 in NS and 1.74±0.54 in HS of the HS group) (P>0.05, Figure 4B).
REDUCED METAL ELEMENTS AFFECTED THE PROLIFERATION OF HFB AND NFB:
To determine if altered metal elements levels affected the proliferation of HFB and NFB, we first used different concentrations of EDTA, a chelating agent that can chelate metal ions, to decrease the metal element concentrations in the medium (Table 4). Subsequently, 2500 cells/well were seeded into 96-well plates and cultured in the medium with decreased metal element levels for 24, 48, and 72 h. Interestingly, we observed that reduced metal elements promoted the proliferation of HFB and NFB within 24 h, while an inhibitory effect was obvious at 72 h (Figure 5).
Discussion
HS, a major clinical challenge owing to their high incidence and the lack of effective treatment, usually result from dermal injuries due to surgeries, deep burns, and trauma [19]. In this study, we investigated the levels of Ca, Mg, Zn, Fe, and Cu in blood, NS, and scar tissues in individuals with HS or FS. Our findings indicated that the levels of Zn, Fe, and Cu in HS tissues were lower than those in FS tissues. Importantly, all examined metal element levels were lower in HS tissues than in NS tissues.
The skin functions as an immune organ. Its microbiome and its physical, chemical, and immune barriers constitute an interactive network that protects individuals from external harm [21]. The 5 elements tested in the current study strongly influence the functions of skin, with effects on wound healing [22,23], scar formation and contraction [24], microvasculature blood flow [25,26], and immune function [27]. A deficiency of these 5 elements may cause skin lesions [28–30], while supplementation may lead to improved skin functions [21,31].
Extensive studies have confirmed the importance of micronutrients in the etiology of different types of diseases [32,33]. In this regard, metal elements seem to play a critical role in illness development, progression, and prognosis. As mentioned previously, metal elements are related to wound healing and scar formation. A previous study [18] investigated micronutrient status in keloids and FS, but no significant difference in metal levels was detected between the 2 groups. Whether external factors affect these element contents has not been reported.
In our study, we aimed to determine the influence of the external environment on the content of elements in the skin. We compared the levels of Ca, Mg, Zn, Fe, and Cu in skin from exposed and unexposed areas in a scar-free group. Our results suggested the levels were the same between the exposed and unexposed sites, which may be attributed to the barrier function and rapid renewal of skin. Considering the large individual deviations in skin element contents reported by a previous study [18], we designed an experiment to compare the element contents between scar tissues and NS in participants in the FS and HS groups with a paired
In addition, our results showed element concentrations in blood were not significantly different among the 3 groups. Similar results were also found in NS among the subgroups. These results suggest that the element contents of the whole body had only minor variations in HS patients. Herein, considering the mentioned finding of a distinguished difference in micronutrient statuses between HS tissue and NS in individuals, we can conclude that the differences in element contents in HS were localized rather than systemic.
Levels of trace elements, including Zn, Fe and Cu, were lower in HS tissues than in FS tissues, which may be explained by drastically different histological structures. The epidermis of an HS is thin, with the papillary layer disappearing and the dermal layer becoming heavily thickened. However, the whole FS structure is nearly normal except for the presence of moderately enhanced thin collagen fibers deposited in a regular manner. Obviously, HS results from a pathological process during excessive wound healing [19]. Although HS tissue has a great number of capillaries, most of them are compromised by hyperplasia and hypertrophy of endothelial cells [34]. The collagen nodes of HS are actually short of blood supply [35]. Hence, the local supply of other nutritive factors would likely also be compromised. The decrease in all element contents in HS tissues in this study confirmed this assumption.
As indicated in Figure 2, HS had markedly reduced levels of Zn, Fe, and Cu. The ratios of the levels of these metals in HS to the corresponding levels in NS were 0.29, 0.34, and 0.36, which were far lower than the ratios of 0.84 and 0.87 for Ca and Mg, respectively. Bang and Dashti [18] also reported that the level of Mn in burn-induced HS is dramatically lower than in NS, with a ratio of approximately 0.11. These elements particularly compose an important part of free radical scavenger enzymes, such as Cu/Zn-superoxide dismutase (Cu/Zn-SOD), Mn-SOD, and Fe-catalase (Fe-Cat). The decrease in Cu, Zn, and Fe would impair free radical scavenging in local tissues, leading to abnormal wound repair and scar generation [8,36]. Oxygen free radicals act in 2 ways to promote scar formation. First, they function as a necessary mediator in collagen synthesis that is catalyzed by proline hydroxylase and lysine hydroxylase. Second, oxygen free radicals nonenzymatically catalyze a reaction changing procollagen to collagen in the absence of the rate-limiting enzyme hydroxylase [37]. Consistent with this, a previous study [38] reported that eliminating free radicals in local tissue will inhibit scar formation.
Our study showed that the Cu/Zn ratio was markedly higher in individuals’ HS than in their NS. A similar phenomenon has been observed in cancer studies [39]. Although the exact reason is not yet known, it may implicate the overproliferation feature of tumor cells. Although HS do not metastasize, they demonstrate excessive local growth, which is similar to that of tumor tissues. The increase in the Cu/Zn ratio may reflect this common trait. A recent study reported that an upregulated Ca/Mg ratio increased the proliferation of prostate cancer cells [40]. Our data showed that the Ca/Mg ratio was not significantly different in HS, which may indicate that different mechanisms are involved in malignant tumors and benign hyperplasia.
Our data may provide evidence demonstrating that nutrition deficiency may be triggering and maintaining factors of HS [41]. If nutrients and oxygen could be supplied to the damaged tissue in time, the pathologic process might be reversed. Soderberg [42] applied Zn-containing adhesive tape on the surface of hypertrophic scars or keloids and reported that scars receded and symptoms of redness, pain, and itching largely diminished. Using 65Zn-containing tape, he observed the absorption of 65Zn into the scar tissue, suggesting a therapeutic effect by replenishing local Zn shortages. Similar results have been reported elsewhere [43,44].
To our knowledge, the association of metal elements with the pathology of HS has not been previously reported. Hence, investigation is urgently required regarding how the concentration of mineral elements may be involved in the pathogenesis of HS. To investigate whether altered metal elements levels affected the proliferation of HFB, we added EDTA to the culture medium of HFB and NFB to create an environment with reduced levels of metal elements. After NFB and KFB were cultured for 24, 48, and 72 h, we found that cell proliferation increased at 24 h, cells growth was inhibited at 72 h. Consequently, we can conclude that metal elements may be implicated in the formation of HS.
The present study has several limitations. No direct method for monitoring metal element contents in skin tissues was used. Tissue specimens are the only way to determine micronutrient status, but surgery is an invasive process. The study sample size was small in that the prevalence of HS and FS was low. More studies on the association between micronutrients and HS are urgently required to reveal how the concentration of mineral elements may be involved in the pathogenesis of HS.
Conclusions
Reduced levels of metal elements were confirmed in HS tissues and not in the body as a whole. The study suggests that decreased levels of these elements in HS may contribute to aberrant cicatrix hyperplasia. Reduced levels of metal elements also affected the proliferation of HFB. Studies on supplementation with metal elements, especially Ca, Mg, Zn, Fe, and Cu, are needed to demonstrate the involvement of metal elements in the pathogenesis of HS, the improvement in wound healing, and the prevention of scar formation.
Figures
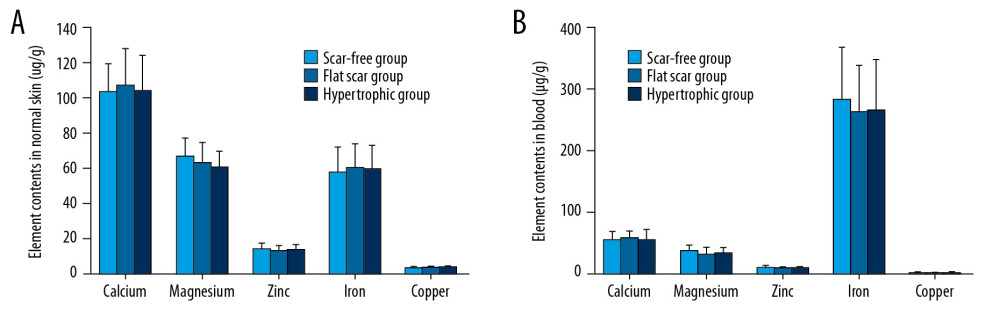 Figure 1. Comparisons of the detected metal element levels in normal skin (A) and blood (B) among the 3 groups.
Figure 1. Comparisons of the detected metal element levels in normal skin (A) and blood (B) among the 3 groups. 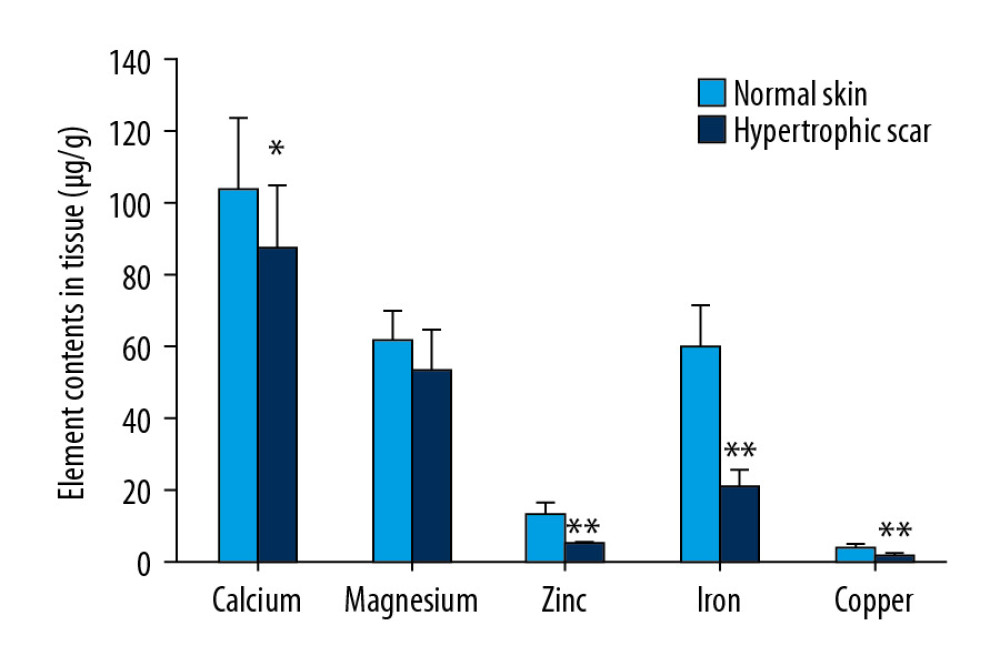 Figure 2. Comparison of element contents in HS and NS in individuals. * P<0.05, ** P<0.01, compared with NS.
Figure 2. Comparison of element contents in HS and NS in individuals. * P<0.05, ** P<0.01, compared with NS. 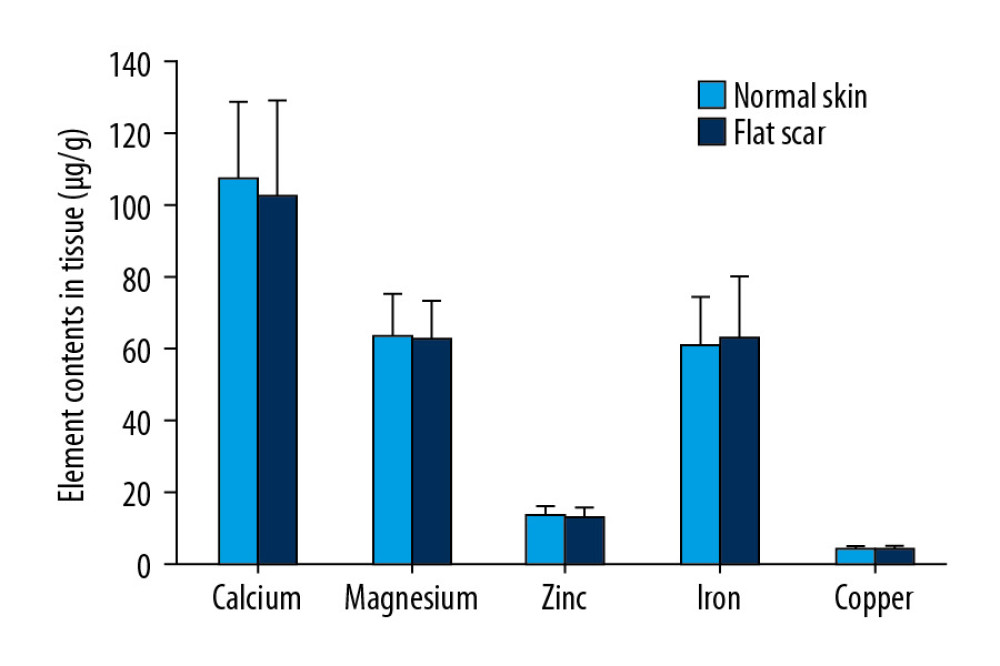 Figure 3. Comparison of element contents in FS and NS in individuals.
Figure 3. Comparison of element contents in FS and NS in individuals. 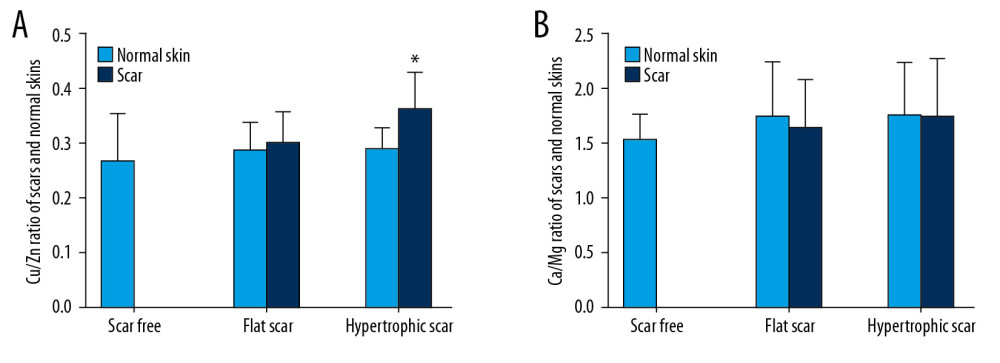 Figure 4. (A) The Cu/Zn ratio in individuals’ scar tissues compared with that in their NS. (B) The Ca/Mg ratio in individuals’ scar tissues compared with that in their NS. * P<0.05 compared with NS.
Figure 4. (A) The Cu/Zn ratio in individuals’ scar tissues compared with that in their NS. (B) The Ca/Mg ratio in individuals’ scar tissues compared with that in their NS. * P<0.05 compared with NS. 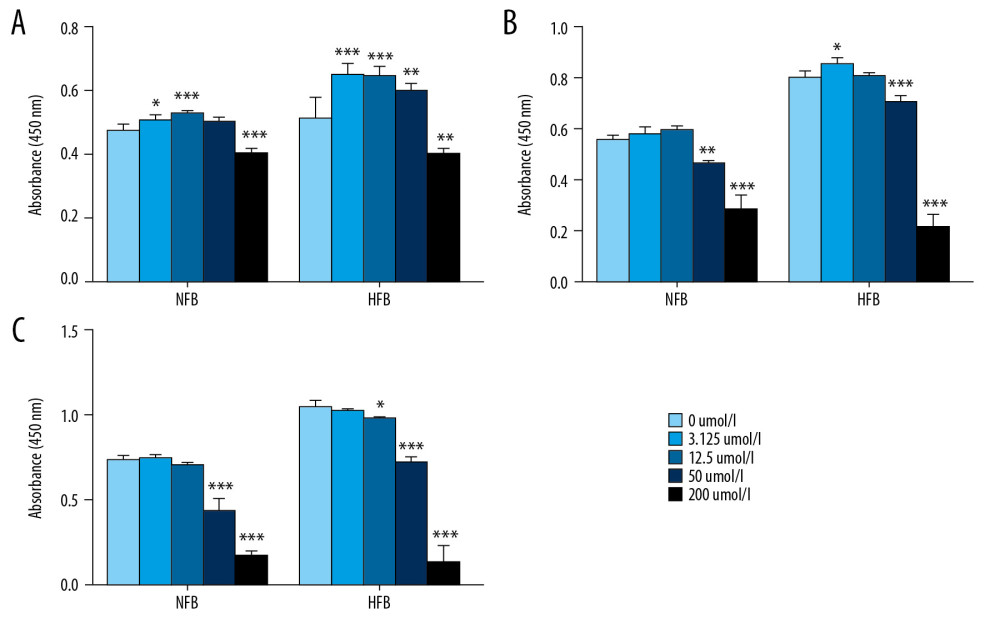 Figure 5. Effect of reduced metal element levels on HFB proliferation. NFB and HFB were conditioned in DMEM containing 0, 3.125, 12.5, 50, or 200 μmol/L EDTA and cultured for (A) 24 h, (B) 48 h, and (C) 72 h. n=4 in each group, * P<0.05, ** P<0.01, *** P<0.001 compared to 0 μmol/L group.
Figure 5. Effect of reduced metal element levels on HFB proliferation. NFB and HFB were conditioned in DMEM containing 0, 3.125, 12.5, 50, or 200 μmol/L EDTA and cultured for (A) 24 h, (B) 48 h, and (C) 72 h. n=4 in each group, * P<0.05, ** P<0.01, *** P<0.001 compared to 0 μmol/L group. Tables
Table 1. Basic characteristics of patients in the 3 groups. Table 2. Comparison of metal element contents in NS (exposed and unexposed area).
Table 2. Comparison of metal element contents in NS (exposed and unexposed area).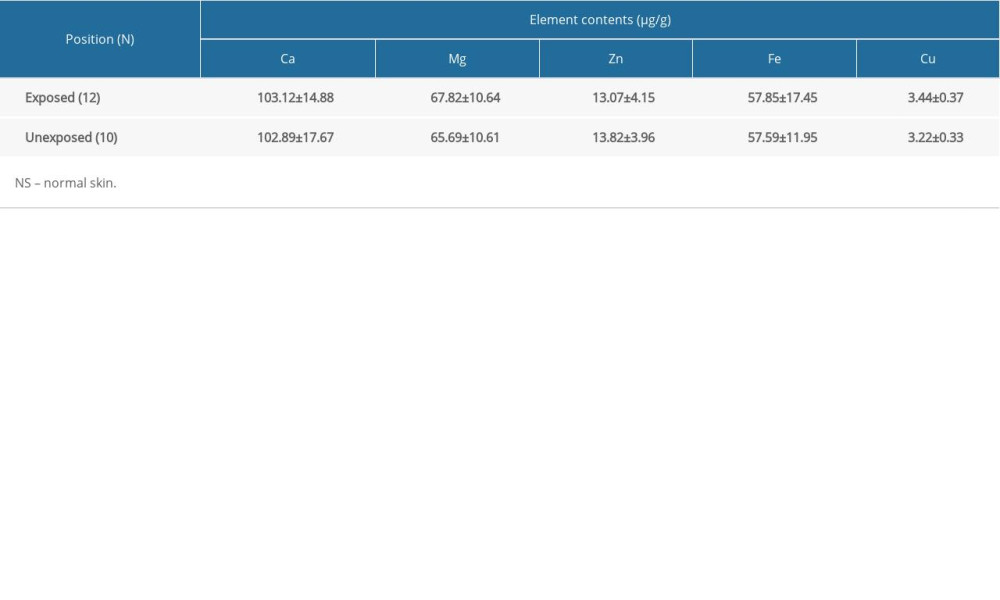 Table 3. Comparison of metal element contents among the scar-free, FS, and HS groups.
Table 3. Comparison of metal element contents among the scar-free, FS, and HS groups.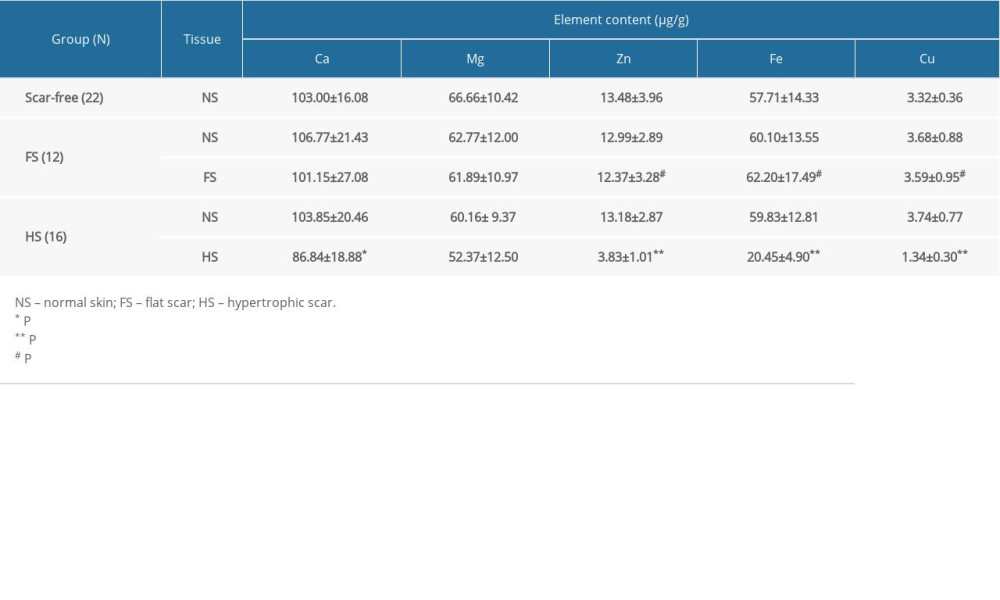 Table 4. Decreased metal element contents in DMEM containing EDTA.
Table 4. Decreased metal element contents in DMEM containing EDTA.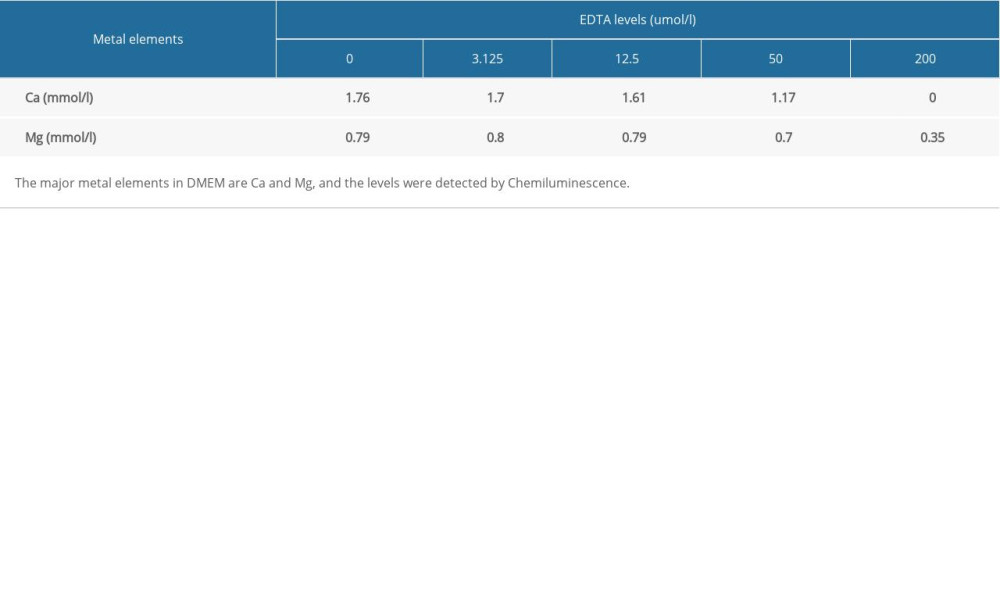
References
1. Weng W, He S, Song H, Aligned carbon nanotubes reduce hypertrophic scar via regulating cell behavior: ACS Nano, 2018; 12; 7601-12
2. Zhang Q, Guo B, Hui Q, miR-137 inhibits proliferation and metastasis of hypertrophic scar fibroblasts via targeting pleiotrophin: Cell Physiol Biochem, 2018; 49; 985-95
3. Feng Y, Wu JJ, Sun ZL, Targeted apoptosis of myofibroblasts by elesclomol inhibits hypertrophic scar formation: EBioMedicine, 2020; 54; 102715
4. Bombaro KM, Engrav LH, Carrougher GJ, What is the prevalence of hypertrophic scarring following burns?: Burns, 2003; 29; 299-302
5. Li J, Chen L, Cao C, The long non-coding RNA LncRNA8975-1 is upregulated in hypertrophic scar fibroblasts and controls collagen expression: Cell Physiol Biochem, 2016; 40; 326-34
6. Rabello FB, Souza CD, Farina JJ, Update on hypertrophic scar treatment: Clinics (Sao Paulo), 2014; 69; 565-73
7. Kavalukas SL, Barbul A, Nutrition and wound healing: an update: Plast Reconstr Surg, 2011; 127(Suppl 1); 38S-43S
8. Dil NN, Sadeghi M, Free radical synthesis of nanosilver/gelatin-poly (acrylic acid) nanocomposite hydrogels employed for antibacterial activity and removal of Cu(II) metal ions: J Hazard Mater, 2018; 351; 38-53
9. Yadav S, Jan R, Roy R, Satsangi PG, Role of metals in free radical generation and genotoxicity induced by airborne particulate matter (PM2.5) from Pune (India): Environ Sci Pollut Res Int, 2016; 23; 23854-66
10. Selmanpakoglu AN, Cetin C, Sayal A, Isimer A, Trace element (Al, Se, Zn, Cu) levels in serum, urine and tissues of burn patients: Burns, 1994; 20; 99-103
11. Barchitta M, Maugeri A, Favara G, Nutrition and wound healing: an overview focusing on the beneficial effects of curcumin: Int J Mol Sci, 2019; 20(5); 1119
12. Williams DR, Trace element starvation and wound healing problems: J Wound Care, 1996; 5; 183-84
13. Chen K, Lv J, Wang G, Changes of serum trace elements in early stage trauma and its correlation with injury severity score: Medicine (Baltimore), 2018; 97; e10077
14. Rehou S, Shahrokhi S, Natanson R, Antioxidant and trace element supplementation reduce the inflammatory response in critically ill burn patients: J Burn Care Res, 2018; 39; 1-9
15. Ni W, Yang W, Yu J, Association between selected essential trace element concentrations in umbilical cord and risk for cleft lip with or without cleft palate: A case-control study: Sci Total Environ, 2019; 661; 196-202
16. Pan CF, Lin CJ, Chen SH, Association between trace element concentrations and anemia in patients with chronic kidney disease: A cross-sectional population-based study: J Investig Med, 2019; 67; 995-1001
17. Emami A, Nazem MR, Shekarriz R, Hedayati M, Micronutrient status (calcium, zinc, vitamins D and E) in patients with medullary thyroid carcinoma: A cross-sectional study: Nutrition, 2017; 41; 86-89
18. Bang RL, Dashti H, Keloid and hypertrophic scars: Trace element alteration: Nutrition, 1995; 11; 527-31
19. Chai CY, Song J, Tan Z, Adipose tissue-derived stem cells inhibit hypertrophic scar (HS) fibrosis via p38/MAPK pathway: J Cell Biochem, 2019; 120; 4057-64
20. Azimzadeh M, Jelodar G, Trace elements homeostasis in brain exposed to 900 MHz RFW emitted from a BTS-antenna model and the protective role of vitamin E: J Anim Physiol Anim Nutr (Berl), 2020 [Online ahead of print]
21. Eyerich S, Eyerich K, Traidl-Hoffmann C, Biedermann T, Cutaneous barriers and skin immunity: Differentiating a connected network: Trends Immunol, 2018; 39; 315-27
22. Clewell A, Barnes M, Endres JR, Efficacy and tolerability assessment of a topical formulation containing copper sulfate and hypericum perforatum on patients with herpes skin lesions: a comparative, randomized controlled trial: J Drugs Dermatol, 2012; 11; 209-15
23. Coger V, Million N, Rehbock C, Tissue concentrations of zinc, iron, copper, and magnesium during the phases of full thickness wound healing in a rodent model: Biol Trace Elem Res, 2019; 191; 167-76
24. Aksoy B, Atakan N, Aksoy HM, Effectiveness of topical zinc oxide application on hypertrophic scar development in rabbits: Burns, 2010; 36; 1027-35
25. Kaptanoglu AF, Arca T, Kilinc K, Magnesium sulfate protects fetal skin from intrauterine ischemia reperfusion injury: Arch Dermatol Res, 2012; 304; 529-32
26. Heidarianpour A, Sadeghian E, Gorzi A, Nazem F, The influence of oral magnesium sulfate on skin microvasculature blood flow in diabetic rats: Biol Trace Elem Res, 2011; 143; 344-50
27. Vonderheid EC, Martinez AR, Prognostic significance of serum copper in patients with cutaneous T-cell lymphoma: Clin Lymphoma Myeloma Leuk, 2019; 19; 228-38
28. Bartosinska J, Chodorowska G, Jazienicka I, Skin lesions in a 16-month-old toddler with impaired zinc absorption: Adv Med Sci, 2011; 56; 369-72
29. Fujimori T, Yasui H, Hiromura M, Sakurai H, Suppressive effect of orally administered copper(II)-aspirinate (Cu2(asp)4) complex on the generation of reactive oxygen species in the skin of animals subjected to UVA exposure: Exp Dermatol, 2007; 16; 746-52
30. Kumar P, Lal NR, Mondal AK, Zinc and skin: a brief summary: Dermatol Online J, 2012; 18(3); 1
31. Hsu PY, Yen HH, Yang TH, Su CC, Tetrathiomolybdate, A copper chelator inhibited imiquimod-induced skin inflammation in mice: J Dermatol Sci, 2018; 92; 30-37
32. Tarwadi K, Agte V, Linkages of antioxidant, micronutrient, and socioeconomic status with the degree of oxidative stress and lens opacity in indian cataract patients: Nutrition, 2004; 20; 261-67
33. Brustolin L, Nardon C, Pettenuzzo N, Synthesis, chemical characterization and cancer cell growth-inhibitory activities of Cu(ii) and Ru(iii) aliphatic and aromatic dithiocarbamato complexes: Dalton Trans, 2018; 47; 15477-86
34. Wang XQ, Song F, Liu YK, Hypertrophic scar regression is linked to the occurrence of endothelial dysfunction: PLoS One, 2017; 12; e176681
35. Kurokawa N, Ueda K, Tsuji M, Study of microvascular structure in keloid and hypertrophic scars: Density of microvessels and the efficacy of three-dimensional vascular imaging: J Plast Surg Hand Surg, 2010; 44; 272-77
36. Wan KC, Evans JH, Free radical involvement in hypertrophic scar formation: Free Radic Biol Med, 1999; 26; 603-8
37. Chen X, Guo G, Li SA study of human keloid with electron spin resonance: Zhonghua Zheng Xing Shao Shang Wai Ke Za Zhi, 1996; 12; 9-11
38. Pal A, Singh A, Nag TC, Iron oxide nanoparticles and magnetic field exposure promote functional recovery by attenuating free radical-induced damage in rats with spinal cord transection: Int J Nanomedicine, 2013; 8; 2259-72
39. Feng JF, Lu L, Zeng P, Serum total oxidant/antioxidant status and trace element levels in breast cancer patients: Int J Clin Oncol, 2012; 17; 575-83
40. Sun Y, Selvaraj S, Varma A: J Biol Chem, 2013; 288; 255-63
41. Qin ZLThe triggering factors of hyperplasia in hypertrophic scar: Chin J Plast Surg, 2013; 19; 135 [in Chinese]
42. Soderberg T, Hallmans G, Bartholdson L, Treatment of keloids and hypertrophic scars with adhesive zinc tape: Scand J Plast Reconstr Surg, 1982; 16; 261-66
43. Mahedia M, Shah N, Amirlak B, Clinical evaluation of hyaluronic acid sponge with zinc versus placebo for scar reduction after breast surgery: Plast Reconstr Surg Glob Open, 2016; 4; e791
44. Arslan K, Karahan O, Okus A, Comparison of topical zinc oxide and silver sulfadiazine in burn wounds: an experimental study: Ulus Travma Acil Cerrahi Derg, 2012; 18; 376-83
Figures
 Figure 1. Comparisons of the detected metal element levels in normal skin (A) and blood (B) among the 3 groups.
Figure 1. Comparisons of the detected metal element levels in normal skin (A) and blood (B) among the 3 groups. Figure 2. Comparison of element contents in HS and NS in individuals. * P<0.05, ** P<0.01, compared with NS.
Figure 2. Comparison of element contents in HS and NS in individuals. * P<0.05, ** P<0.01, compared with NS. Figure 3. Comparison of element contents in FS and NS in individuals.
Figure 3. Comparison of element contents in FS and NS in individuals. Figure 4. (A) The Cu/Zn ratio in individuals’ scar tissues compared with that in their NS. (B) The Ca/Mg ratio in individuals’ scar tissues compared with that in their NS. * P<0.05 compared with NS.
Figure 4. (A) The Cu/Zn ratio in individuals’ scar tissues compared with that in their NS. (B) The Ca/Mg ratio in individuals’ scar tissues compared with that in their NS. * P<0.05 compared with NS. Figure 5. Effect of reduced metal element levels on HFB proliferation. NFB and HFB were conditioned in DMEM containing 0, 3.125, 12.5, 50, or 200 μmol/L EDTA and cultured for (A) 24 h, (B) 48 h, and (C) 72 h. n=4 in each group, * P<0.05, ** P<0.01, *** P<0.001 compared to 0 μmol/L group.
Figure 5. Effect of reduced metal element levels on HFB proliferation. NFB and HFB were conditioned in DMEM containing 0, 3.125, 12.5, 50, or 200 μmol/L EDTA and cultured for (A) 24 h, (B) 48 h, and (C) 72 h. n=4 in each group, * P<0.05, ** P<0.01, *** P<0.001 compared to 0 μmol/L group. Tables
 Table 1. Basic characteristics of patients in the 3 groups.
Table 1. Basic characteristics of patients in the 3 groups. Table 2. Comparison of metal element contents in NS (exposed and unexposed area).
Table 2. Comparison of metal element contents in NS (exposed and unexposed area). Table 3. Comparison of metal element contents among the scar-free, FS, and HS groups.
Table 3. Comparison of metal element contents among the scar-free, FS, and HS groups. Table 4. Decreased metal element contents in DMEM containing EDTA.
Table 4. Decreased metal element contents in DMEM containing EDTA. Table 1. Basic characteristics of patients in the 3 groups.
Table 1. Basic characteristics of patients in the 3 groups. Table 2. Comparison of metal element contents in NS (exposed and unexposed area).
Table 2. Comparison of metal element contents in NS (exposed and unexposed area). Table 3. Comparison of metal element contents among the scar-free, FS, and HS groups.
Table 3. Comparison of metal element contents among the scar-free, FS, and HS groups. Table 4. Decreased metal element contents in DMEM containing EDTA.
Table 4. Decreased metal element contents in DMEM containing EDTA. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








