14 August 2020: Clinical Research
Characteristics and Outcome of 69 Cases of Coronavirus Disease 2019 (COVID-19) in Lu’an City, China Between January and February 2020
Zhenghao Jiang1ABCD, Jie Sun1ABCE, Xiude Li1ABCF, Yong Lyu1CEFG*DOI: 10.12659/MSM.925442
Med Sci Monit 2020; 26:e925442
Abstract
BACKGROUND: This population study aimed to investigate the demographic and clinical characteristics and outcome of cases of coronavirus disease 2019 in Lu’an City, China between January 22, 2020 and February 18, 2020 identified from the China Information System for Disease Control and Prevention (CISDCP).
MATERIAL AND METHODS: Laboratory-confirmed cases of COVID-19 reported in the CISDCP were included in this study. The distribution of cases, exposure history, clustered epidemic situation, and clinical manifestations, disease severity, and key time nodes were analyzed. Once the throat swab or sputum sample was positive for SARS-CoV-2 by real-time reverse transcriptase-polymerase chain reaction testing as confirmed cases.
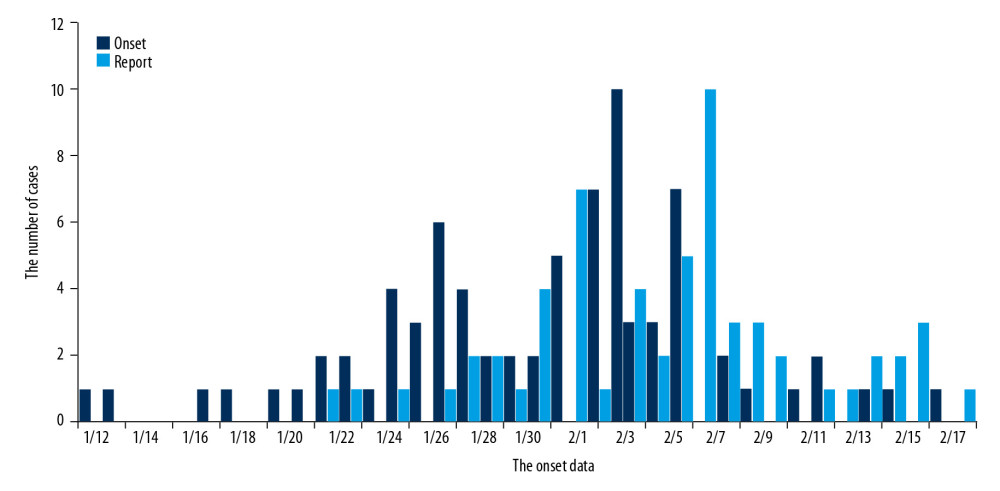
RESULTS: There were 69 cases of COVID-19 that were confirmed between January 22 and February 18, 2020 reported. The onset time was concentrated on January 25, 2020 solstice to February 6, 2020 (71.0%), and the reporting dates were concentrated on January 31, 2020 and February 9, 2020 (69.6%). Nineteen cases (27.5%) had a history of sojourn in Hubei Province, and none of the cases reported after February 6, 2020 had a history of exposure in Hubei Province. There were 14 cluster outbreaks, and human-to-human transmission was the most common (78.6%). The most common symptoms were fever (56.5%), cough (37.7%), and self-conscious discomfort (14.5%). Besides, there were 9 severe cases (13.0%).
CONCLUSIONS: The epidemic prevention and control in Lu’an City has achieved phased results. Yet, new strict control measures need to be implemented to prevent a further outbreak, especially for those who will return to Lu’an City.
Keywords: Cluster Analysis, Coronavirus Infections, Epidemiologic Methods, Betacoronavirus, COVID-19, COVID-19 Testing, COVID-19 Vaccines, Cities, Clinical Laboratory Techniques, Contact Tracing, Geography, Medical, Nasopharynx, Oropharynx, Pandemics, Pneumonia, Viral, Real-Time Polymerase Chain Reaction, SARS-CoV-2, Sputum, Symptom Assessment
Background
A cluster of unexplained pneumonia cases was reported in Wuhan in late December 2019. On February 11, 2020, the World Health Organization (WHO) named the disease coronavirus disease 2019 (COVID-19). Up till now (April 16, 2020), the outbreak of COVID-19 has spread to 6 continents, infecting 3 million people and killing nearly 137 000 [1,2]. COVID-19 outbreak is considered one of the deadliest pandemics in human history. On February 28, 2020 the WHO raised the pandemic level to the highest level [3,4].
COVID-19 is a new infectious disease, whose etiological characteristics, pathogenesis, and epidemic characteristics are still not well understood. Early studies have shown that the disease is highly contagious and has a high risk of human-to-human and inter-city transmission, with an early basic regeneration index (R0) of about 2.2 [5]. This paper summarizes and analyzes the epidemic characteristics of confirmed cases in Lu’an City (located 300 km from Wuhan City), China between January 22, 2020 and February 18, 2020 identified from the China Information System for Disease Control and Prevention (CISDCP) [6], so as to facilitate the formulation and adjustment of prevention and future control strategies.
Material and Methods
STUDY PARTICIPANTS:
Laboratory-confirmed cases with COVID-19 reported in the China Information System for Disease Control and Prevention (CISDCP) between January 22, 2020 and February 18, 2020 were included in this study.
LABORATORY TESTING:
Cases with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection were confirmed by the real-time reverse transcriptase-polymerase chain reaction (RT-PCR) of nasopharyngeal and oropharyngeal swabs [7]. The RT-PCR kit was manufactured by the Sun Yat-sen University Da’an Gene Co., Ltd, China (20203400063). According to the test results of the clinical samples, the ROC curve was used to determine that the ORF1 ab and N reference values exhibited Ct values equal to 40.
DEFINITION OF STUDY OUTCOMES:
Once the throat swab or sputum sample was positive for SARS-CoV-2 by RT-PCR testing as confirmed cases [8,9]. Positive cases were confirmed based on clinical data or epidemiological history, and nucleic acid testing. The clinical classification of COVID-19 included mild cases, ordinary cases, severe cases, and critical cases [10]. A cluster outbreak was defined as 2 or more confirmed cases in a small area (such as a family) within 2 weeks; this usually suggests a human-to-human transmission through close contact or joint exposure [11].
POPULATION INFECTION CONTROL MEASURES:
All close contacts received 14 days of intensive medical observation and nucleic acid testing. On January 25, 2020, the government issued a notice to cancel all gatherings and asked residents to spend their holidays at home as much as possible. From February 5, 2020 all communities, villages and units will be placed under closed management to restrict the movement of people.
RESEARCH METHODS:
The onset date, report date, epidemiological exposure history, clinical characteristics, treatment history, and general demographic characteristics of the COVID-19 confirmed cases were collected and further analyzed. Clinical data of the COVID-19 confirmed cases were obtained from patient records; epidemiological and socio-demographic data were obtained from epidemiological surveys. EXCEL 2013 was used to draw the epidemiological curve of the disease, while ArcGIS 10.3 was used to draw the regional distribution map of the disease
STATISTICAL ANALYSIS:
Statistical analyses were performed using SPSS 20.0. Significance level was set at
ETHICAL STATEMENT:
The medical ethics committee of Lu’an Center for Disease Control approved this study. Informed consent was obtained from all patients.
Results
GENERAL DATA:
A total of 69 cases of COVID-19 that were confirmed between January 22, 2020 and February 18, 2020 were reported in Lu’an City. All COVID-19 patients were successfully cured, and, consequently, discharged (before March 3, 2020).
TIME DISTRIBUTION:
The onset date of all cases (69 cases) was between January 12, 2020 and February 17, 2020; while the onset date of 49 cases (71%) was between January 25, 2020 and February 6, 2020. The number of reported cases has gradually increased since the first case was reported on January 12, 2020, and the onset date of 48 cases (69.6%) was mainly concentrated between January 31, 2020 and February 9, 2020. The number of new confirmed cases per day took a downward trend since February 9, 2020, while no new cases were reported after February 18, 2020 (Figure 1).
REGIONAL DISTRIBUTION:
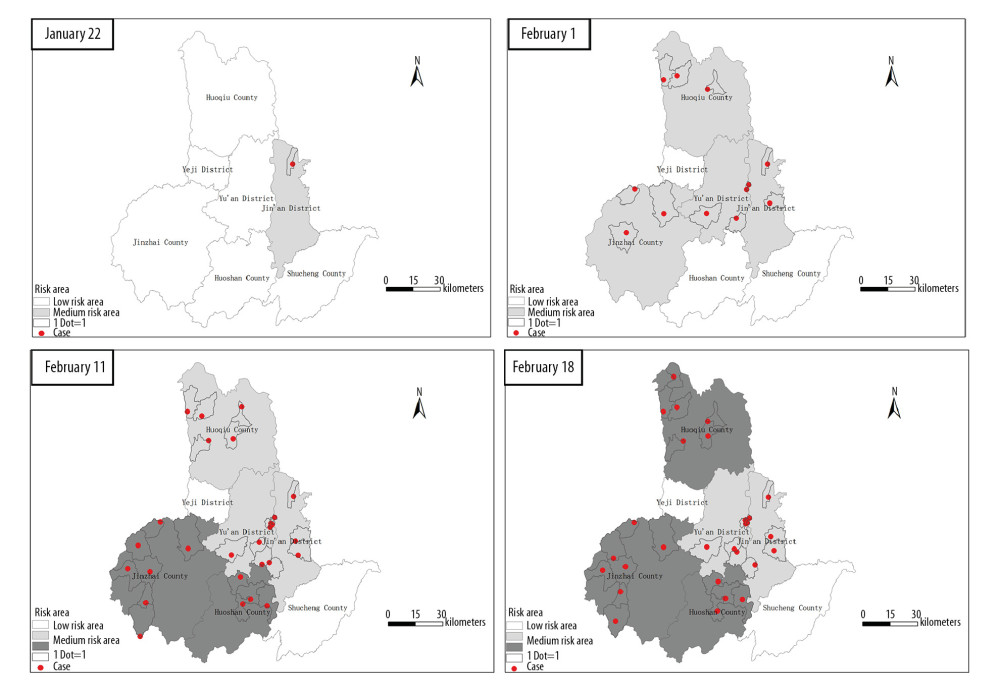
The regional distribution of the 69 cases involved 5 counties/areas, including Jinzhai county (23 cases), Huoshan county (17 cases), Huoqiu county (13 cases), Yu ‘an area (8 cases) and Jin ‘an area (8 cases). Twenty-two cases (31.9%) were from urban areas (including county) and 47 cases (68.1%) from rural areas.
On January 22, 2020, only one suburb area reported positive cases. Consequently, (from January 22, 2020 to February 18, 2020) infection was reported in the other 5 counties/areas in Lu’an City: Tangjiahui Town of Jinzhai County (14 cases), Hengshan Town of Huoshan County (12 cases), and Zhouji Town of Huoqiu County (6 cases), accounting for 46.4% of the total cases (Figure 2).
POPULATION DISTRIBUTION:
Among the 69 cases, the male (44 cases) to female (25 cases) ratio was 1.8: 1. The patients’ age ranged from 10 months to 78 years old, with a median age of 41.0 years. The age group 20–49 years had the largest number of cases (68.1%). In the occupational distribution, those making up the largest proportion were farmers (44.9%), business people (23.2%), domestic workers or unemployed (7.2%), food service personnel (4.3%), government employees (4.3%), workers (4.3%), medical staff (2.9%), children (2.9%), and other occupations (5.8%).
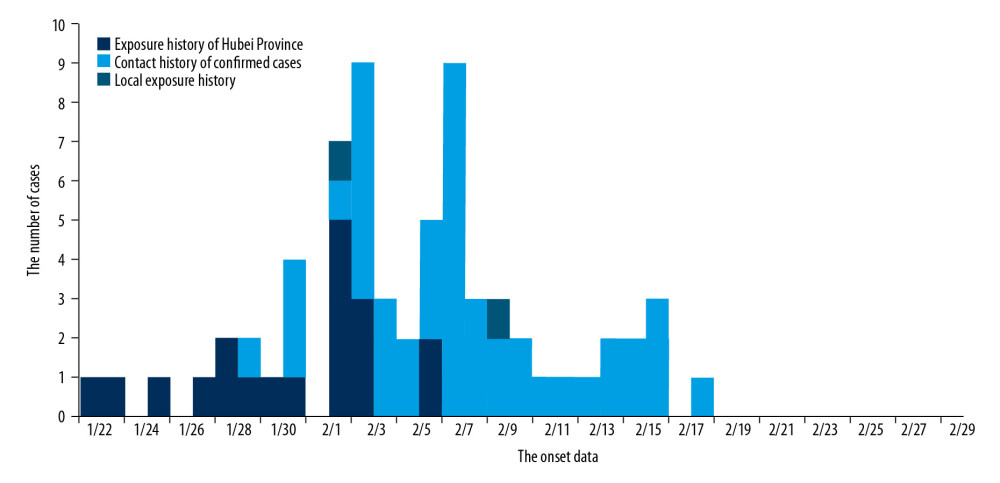
EXPOSURE HISTORY IN HUBEI PROVINCE:
Of the total cases, 19 cases (27.5%) had a history of sojourn in Hubei Province, while the other 50 cases stated not having lived or traveled in Hubei province in the 2 weeks before the onset of the disease. The cases reported before January 29, 2020 had a history of exposure to Hubei province, and none of the cases reported after February 6, 2020 had a history of exposure to Hubei province (Figure 3).
CLUSTER EPIDEMIC SITUATION ANALYSIS:
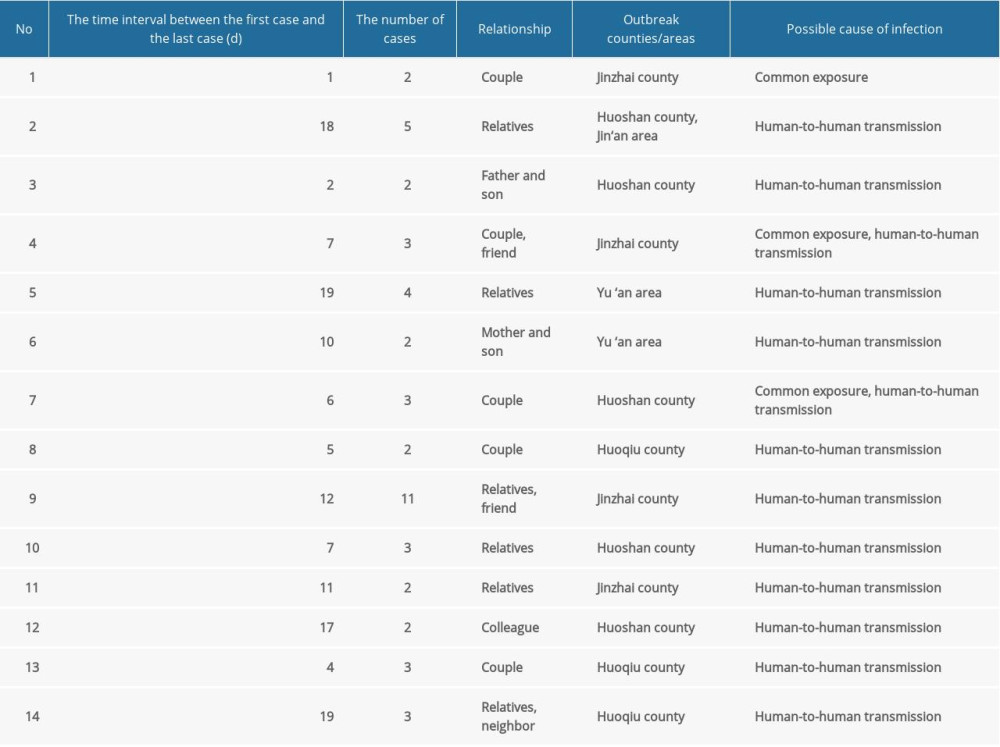
A total of 14 clustered outbreaks occurred, with 47 cases, accounting for 68.1% of the total cases. The main counties/areas where the outbreak occurred were Huoshan county (5 outbreaks), Jinzhai county (4 outbreaks), and Jin ‘an area (1 outbreak). The shortest interval between the first case and the last case was 1 day, and the longest interval between the first case and the last case was 19 days. Eleven clustered outbreaks were reported; human-to-human transmission was the most common route of transmission, accounting for 78.6% of the total cluster outbreaks (Table 1).
CLINICAL MANIFESTATIONS:
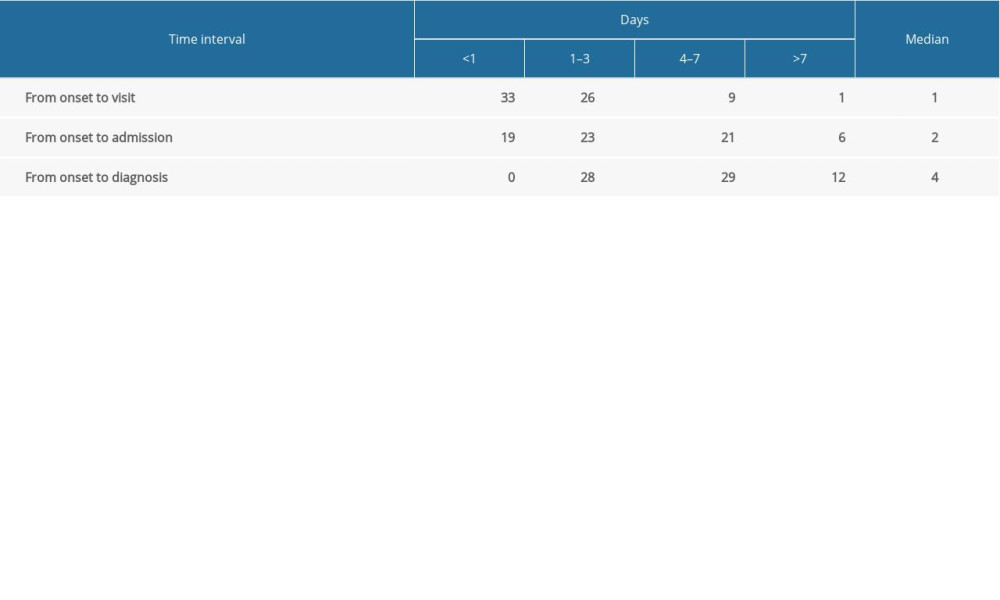
The first symptoms of the COVID-19 were fever (56.5%), cough (37.7%), self-conscious discomfort (14.5%), chills (8.7%), fatigue (8.7%), and other symptoms (43.5%). Among the 69 cases, 5 cases (7.2%) had chronic underlying diseases, mainly including diabetes, uremia, chronic bronchitis, duodenal ulcer, and heart disease. There were 60 mild cases (87.0%) and 9 severe cases (13.0%). The time from onset to doctor visit was <24 hours (33 cases; 47.8%), 1–3 days (23 cases; 33.3%), and 4–7 days (29 cases; 42.0) (Table 2). The median age of severe cases (56.0 years) was significantly higher than that of mild cases (41.0 years), and the difference was statistically significant (P=0.015).
Discussion
The transmission mode of COVID-19 changed from small-scale exposure in the early stage to human-to-human transmission in the later stage. The change of transmission mode led to the rapid outbreak of COVID-19 in the whole country from January 2020 [12]. The prevention and control of COVID-19 in Lu’an City is a challenging task considering the city’s geographical location and the proximity to the Wuhan area (the focal point of infection). The number of COVID-19 cases in Lu’an City reached the peak in the period between February 3, 2020 and February 7, 2020 [12], after which strict control measures were implemented, eventually reducing the number of reported positive cases in the city [13,14]. After February 7, 2020, newly reported cases were mainly local cases, and the causes of infection were mostly family clusters, which were similar to the epidemic characteristics of Jinan City [15].
Lu’an City is composed of 3 areas and 4 counties. Although strict control measures have been implemented to control the outbreak from and between counties/areas, there were still many close contacts of imported cases or returnees from Wuhan, which have not been fully controlled, thus leading to the second wave of infections in some areas [15].
According to this study, the 69 confirmed cases of COVID-19 in Lu’an City were more likely to be male, middle-aged, and farmers; according to statistics, Lu’an City had the highest rate of infection among farmers compared to other Chinese cities [8,12–16]. This may be because Lu’an is an economically underdeveloped area with a large number of young adults working outside the city.
The most common clinical features were fever and cough. Some cases presented with self-conscious discomfort and chills, which was similar to the results reported by Hunan province [17]. The atypical clinical manifestations of COVID-19 bring great difficulties to the early differential diagnosis; thus, the screening capabilities should be improved to detect suspicious cases as early as possible [18].
All cases in Lu’an City were cured, and, consequently, discharged from the hospital. The incidence rate of severe diseases was 13.0%, which was significantly lower than the national rate of 25.5%, and no death was reported, which may be related to the timely diagnosis and treatment of diseases in Lu’an City. Besides, the time interval from the onset to treatment, from the onset to the admission, and from the onset to the diagnosis in Lu’an was obviously lower than the data reported in earlier reports nationwide [19].
Conclusions
At present (April 16, 2020), no new suspected or confirmed cases have been reported in Lu’an. All the cases have been successfully cured and discharged from the hospital, indicating that the early transmission chain has been blocked. Yet, new strict control measures need to be implemented to prevent a further outbreak, especially for those who plan to return to Lu’an City.
This study only carried out descriptive epidemiological observation on the cases in Lu’an City, China between January 2020 and February 2020, but lacked systematic analysis. In the future, it is necessary to carry out special studies on clustered epidemic situations.
References
1. Wang C, Horby PW, Hayden FG, A novel coronavirus outbreak of global health concern: Lancet, 2020; 395(10223); 470-73
2. National Center for Immunization and Respiratory Diseases (NCIRD), Division of Viral Diseases: Coronavirus Disease 2019 (COVID-19) in the U.S. [EB/OL] 02 21, 2020 available from:https://www.cdc.gov/coronavirus/2019-ncov/cases-in-us.html
3. World Health Organization: Naming the coronavirus disease (COVID-19) and the virus that causes it. [EB/OL] 02 11, 2020 available from: www.who.int/emergencies/diseases/novel-coronavirus-2019/technical-guidance/naming-the-coronavirus-disease-(covid-2019)-and-the-virus-that-causes-it
4. World Health Organization: Coronavirus disease 2019 (COVID-19) Situation Report – 39. [EB/OL] 02 28, 2020 available from: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200228-sitrep-39-covid-19.pdf?sfvrsn=5bbf3e7d_2
5. Li Q, Guan X, Wu P, Early transmission dynamics in Wuhan, China, of novel coronavirus-infected Pneumonia: N Engl J Med, 2020; 382; 1199-207
6. Ren M, Dong JS, Zhou MEffect analysis of Chinese information system for disease control and prevention: Journal of Preventive Medical Information, 2012; 28(7); 558-59 [in Chinese]
7. Babiker A, Myers CW, Hill CE, SARS-CoV-2 testing: Am J Clin Pathol, 2020; 153(6); 706-8
8. WHO, Coronavirus disease 2019 (COVID-19): Situation Report-99. [EB/OL] 04 28, 2020 available from: https://www.who.int/docs/default-source/coronaviruse/situation-reports/20200428-sitrep-99-covid-19.pdf?sfvrsn=119fc381_2
9. WHO: Global Surveillance for human infection with coronavirus disease (COVID-19). [EB/OL] 03 20, 2020 available from: https://www.who.int/publications-detail/global-surveillance-for-human-infection-with-novel-coronavirus-(2019-ncov)
10. National Health Commission of the People’ Republic of China: Notice on the issuance of COVID-19 diagnosis and treatment scheme (trial seventh edition) [EB/OL] 03 04, 2020 available from: http://www.nhc.gov.cn/yzygj/s7653p/202003/46c9294a7dfe4cef80dc7f5912eb1989.shtml
11. National Health Commission of the People’ Republic of China: Notice of the general office of the national health commission on the issuance of COVID-19 prevention and control scheme (sixth edition) [EB/OL] 03 07, 2020 available from: http://www.nhc.gov.cn/jkj/s3577/202003/4856d5b0458141fa9f376853224d41d7.shtml
12. Analysis of epidemiological characteristics of COVID-19: Zhong Hua Liu Xing Bing Xue Za Zhi, 2020; 41(2); 145-51 [in Chinese]
13. Wang XZ, Liao CH, Li ZHPreliminary analysis of early prevalence and spatial and temporal distribution of COVID-19 in Guangdong province: Journal of Tropical Medicine available from: [in Chinese]http://kns.cnki.net/kcms/detail/44.1503.R.20200213.0806.002.html
14. Lin JF, Wu MN, Wu HCEpidemiological characteristics of COVID-19 cases in Zhejiang province: Preventive Medicine available from: [in Chinese]http://kns.cnki.net/kcms/detail/33.1400.R.20200302.0955.002.html
15. Cui LL, Geng XY, Zhao XDCurrent characteristics and thoughts of COVID-19 in Jinan city: Journal of Shandong University (Health Sciences) available from: [in Chinese]http://kns.cnki.net/kcms/detail/37.1390.r.20200304.1426.004.html
16. Chen N, Zhou M, Dong X, Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study: Lancet, 2020 S0140-6736(20): 30211-17
17. Dai ZH, Gao LD, Luo KWAnalysis of clinical characteristics of COVID-19 in Hunan province: Practical Preventive Medicine available from: [in Chinese]http://kns.cnki.net/kcms/detail/43.1223.R.20200305.1537.005.html
18. Liu ZY, Gao LD, Hu SXAnalysis of diagnosis of 697 confirmed COVID-19 cases in Hunan province: Practical Preventive Medicine available from: [in Chinese]http://kns.cnki.net/kcms/detail/43.1223.R.20200305.1921.008.html
19. Recent understanding of the epidemiological characteristics of COVID-19: Chinese Journal of Viral Diseases available from: [in Chinese]https://doi.org/10.16505/j.2095-0136.2020.0015
Figures
Tables
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387