11 November 2020: Clinical Research
Serum Interleukin-6 Concentrations and the Severity of COVID-19 Pneumonia: A Retrospective Study at a Single Center in Bengbu City, Anhui Province, China, in January and February 2020
Hong Sun1CE, Pu Guo1BD, Lunjun Zhang1DF, Fengchao Wang1A*DOI: 10.12659/MSM.926941
Med Sci Monit 2020; 26:e926941
Abstract
BACKGROUND: At present, the relationships among COVID-19 disease progression, patient prognosis, and immune status are unclear. This single-center retrospective study evaluated the correlation between serum interleukin-6 (IL-6) levels at admission with the severity of COVID-19 pneumonia, as determined by admission to the intensive Care Unit (ICU).
MATERIAL AND METHODS: Patients admitted to The First Affiliated Hospital of Bengbu Medical College in Bengbu City, Anhui Province, China, in January and February 2020 for COVID-19 pneumonia were enrolled in this study. COVID-19 infection was confirmed by the detection of SARS-CoV-2 nucleic acid in throat swab samples using real-time fluorescent reverse transcription PCR. Serum IL-6 concentrations at admission were measured by ELISA. Correlations between serum IL-6 concentrations and ICU admission due to the development of severe COVID-19 pneumonia were evaluated.
RESULTS: This study enrolled 68 patients with novel coronavirus pneumonia. IL-6 concentrations were significantly higher in patients with more severe than less severe COVID-19 pneumonia. Eight of 40 patients with severe COVID-19 pneumonia became critically ill and required ICU admission. IL-6 concentrations were significantly higher in patients with severe COVID-19 pneumonia who were than who were not treated in the ICU. The area under the receiver operating characteristic (ROC) curve (AUC) was 0.816 (P<0.01), indicating that IL-6 was prognostic of disease severity in patients with COVID-19 pneumonia.
CONCLUSIONS: Serum IL-6 concentration is closely associated with the severity of COVID-19. Continuous monitoring of IL-6 has clinical value in evaluating patient condition.
Keywords: COVID-19, Interleukin-6, COVID-19, Critical Illness, Intensive Care Units, Pandemics, Patient Admission, ROC Curve, SARS-CoV-2
Background
The novel coronavirus, named severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) by the World Health Organization (WHO), was first observed in Wuhan, China, in December 2019 [1]. This virus belongs to the genus
Coronavirus disease-2019 (COVID-19), which is caused by SARS-CoV-2 infection, has been clinically classified into 4 main categories: mild, common, severe, and critical [4,5]. Many patients with severe COVID-19 experience acute respiratory distress, whereas critically ill patients experience respiratory failure. Although most cases are mild to moderate and can heal spontaneously, some patients develop critical illness, characterized by respiratory dysfunction and/or multiple organ failure. Identifying patients at high risk of critical illness is important, as it can provide these patients with more active treatment and reduce morbidity and mortality rates.
At present, the relationships among COVID-19 disease progression, prognosis, and immune status are unclear, and no effectively predictive biomarkers have been proposed. The progression of novel coronavirus-associated pneumonia to a critical and life-threatening illness is thought to be associated with the cytokine storm, defined as the unregulated and excessive release of pro-inflammatory factors in the body [6,7]. The serum concentrations of interleukin-6 (IL-6), granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF) [8–10], and other cytokines have been found to be significantly increased in patients with severe infections of viruses such as SARS-CoV and H1N1 influenza virus [11,12]. These inflammatory factors can recruit and activate immune cells in the lungs. Many immune system cells and tissue fluids accumulate in the lungs of these patients, blocking gas exchange between the alveoli and capillaries and leading to ARDS. Induction of a cytokine storm results in the immune system destroying large numbers of healthy lung cells while killing the virus, thus severely damaging the ventilation function of the lungs. This damage appears on computed tomography (CT) images of the lungs as large white areas, called “white lung” [13,14], and can result in respiratory failure and death.
IL-6 is an important multi-functional pro-inflammatory cytokine [15]. As an acute inflammatory factor, IL-6 mainly stimulates the proliferation and differentiation of cells involved in immune responses and improves their function, as well as participating in inflammatory and fever responses. Several retrospective studies have shown that increased levels of IL-6 are associated with death in patients with severe COVID-19 infection [16,17]. Elevated IL-6 is also associated with disease severity and course, suggesting that this cytokine may be a marker for disease monitoring in patients with severe COVID-19 [18]. Serum concentrations of IL-6 and C-reactive protein (CRP) can effectively assess disease severity, suggesting that IL-6 and CRP may be independent factors predictive of the severity of COVID-19 [19]. The present study evaluated whether serum concentration of IL-6 could predict critical illness in patients with severe COVID-19.
Material and Methods
PATIENT SELECTION:
This study included patients diagnosed with COVID-19 at the First Affiliated Hospital of Bengbu Medical College from January to February 2020. In all patients, infection with SARS-CoV-2 was confirmed by real-time fluorescent reverse transcription PCR detection of virus-specific nucleic acid in sputum and throat swab specimens, using commercially available kits specific for 2019-nCoV (Da An Gene Co., Ltd., Guangzhou, China) and an ABI 7500 thermal cycler.
The clinical severity of COVID-19 was defined according to the Chinese management guidelines for COVID-19 (version 6.0). Patients with general COVID-19 were defined as those with fever and respiratory and other symptoms, with possible pneumonia manifestations on imaging. Patients were categorized as having severe COVID 19 if they had (1) apparent shortness of breath and a respiratory rate ≥30 breaths/min, (2) oxygen saturation at rest <93%, or (3) arterial blood oxygen partial pressure (pO2)/oxygen concentration (FiO2) ≤300 mmHg (1 mmHg=133 Pa). At high altitudes (elevation above 1000 m), pO2/FiO2 was corrected using the formula: pO2/FiO2×[atmospheric pressure (mmHg)/760]. Patients were defined as critically ill if they required admission to the intensive care unit (ICU) for (1) respiratory failure requiring mechanical ventilation, (2) shock, or (3) failure of other organs.
INSTRUMENTS AND REAGENTS:
Serum concentrations of IL-6 were determined using IL-6 detection kits and the Cobas E601 electrochemiluminescence (ECL) analyzer (Roche, Basel, Switzerland). All reagents were within their period of validity, with quality control and calibration of these reagents meeting manufacturers’ requirements.
METHODS:
After a 12-h overnight fast, 2–3 mL of venous blood were drawn in the morning from patients with COVID-19 and healthy individuals and tested within 2 h. Blood samples were centrifuged at 4000 rpm for 5 min, and serum concentrations of IL-6 were measured using the ECL method and IL-6 detection kits. All tests were performed by skilled laboratory authorized personnel in accordance with the manufacturers’ instructions. The normal reference range for IL-6 was 0–7 pg/mL.
STATISTICAL ANALYSIS:
Normally distributed continuous data were expressed as mean±standard deviation (SD) and compared by
Results
BASELINE CHARACTERISTICS:
The study cohort consisted of 68 patients, 37 men and 31 women, ranging in age from 21 to 83 yr, (mean age 48.63±16.05 yr), who had been diagnosed with PCR-confirmed COVID-19. Of these 68 patients, 28 had general COVID-19 and 40 had severe COVID-19. Although there were no significant differences in gender, age, and percentages of patients with diabetes and coronary heart disease between these 2 groups, the percentage of patients with hypertension was significantly higher in patients with severe COVID-19 (P<0.05). In addition, serum IL-6 concentrations were significantly higher in patients with severe than with general COVID-19 (P<0.05; Table 1).
CLINICAL CHARACTERISTICS OF THE PROGRESSION FROM SEVERE TO CRITICAL ILLNESS:
Of the 40 patients diagnosed with severe COVID-19 pneumonia, 8 developed critical illness after 2 to 7 days (Tables 2, 3). Analysis of IL-6 concentrations at admission and at the time of diagnosis with critical illness in these 8 patients showed that 7 of these patients had elevated IL-6 levels at the time of critical illness, with a maximum 23-fold increase. In contrast, the eighth patient had lower IL-6 levels at diagnosis of critical illness than at admission.
A comparison of the 8 severely ill patients who progressed to critical illness and the 32 severely ill patients who did not show further progression showed that sex distribution did not differ in these 2 groups (P>0.05), whereas older patients and those with hypertension were more likely to progress to critical illness (P<0.05). In addition, IL-6 concentrations were significantly higher in patients who did than did not progress to critical illness (P<0.01; Figure 1).
ANALYSIS BY RECEIVER OPERATING CHARACTERISTIC CURVE:
Receiver operating characteristic (ROC) curve analysis was performed to identify the optimal IL-6 concentration predicting COVID-19 intensification. An area under the ROC curve (AUC) of 0.816 was found to predict whether COVID-19 is complicated by severe pneumonia (P<0.01). The optimum critical concentration of IL-6 in patients who progressed to critical illness was 24.3 pg/mL (Figure 2).
Discussion
To date, approximately 5 million persons worldwide have been diagnosed with novel coronavirus pneumonia, with a mortality rate over 6%. This high mortality rate has been closely associated with the induced inflammatory storm [20]. Following SARS-CoV-2 infection, the virus can enter immune cells in the lungs by binding to the angiotensin-converting enzyme-2 (ACE-2) receptor. This can result in the activation of lung immune cells, producing large amounts of inflammatory factors, which destroy lung tissue and induce respiratory failure [21,22].
IL-6 is a versatile cytokine with a wide range of functions essential for regulating immune and inflammatory responses and may therefore play a key role in the inflammatory storm [23]. During the acute phase of inflammation, IL-6 mainly stimulates the proliferation, differentiation, and functional improvement of cells involved in immune responses and participates in inflammatory and fever responses. In clinical settings, IL-6 has been used to diagnose infectious diseases, and it is highly significant in the differential diagnosis of bacterial and viral infections. Increased IL-6 is also a biomarker for the severity of hepatitis B virus (HBV) infection [24].
IL-6 may also play a key role in the development and progression of novel coronavirus pneumonia [19,25]. Circulating IL-6 concentrations have been closely associated with the clinical severity of COVID-19. For example, serum IL-6 levels were found to be significantly higher in severely ill patients than in those with mild symptoms [26], suggesting that IL-6 levels are closely associated with the occurrence of severe COVID-19 in adults and could predict the severity of illness in patients with COVID-19. IL-6 levels were also found to be significantly elevated in patients with respiratory insufficiency, suggesting that IL-6 plays an important role in lung injury due to SARS-CoV-2 infection [27]. Severe respiratory distress in patients with highly pathogenic SARS-CoV-2 was found to be caused by IL-6 elevation. Increased levels of IL-6 and CRP were found to be predictive of the need for mechanical ventilation, indicating that measurement of IL-6 can guide the escalation of treatment in patients with COVID-19 – related hyperinflammatory syndrome [28]. Taken together, these findings suggested that monitoring serum concentrations of IL-6 may be crucial for identifying disease progression in patients infected with SARS-CoV-2.
To further understand the relationship between IL-6 and the progression of novel coronavirus pneumonia, we retrospectively analyzed IL-6 levels in serum samples from 68 infected patients. We found that IL-6 levels were significantly higher in patients with severe than with mild COVID-19 symptoms. Of the 40 severely ill patients, 8 subsequently progressed to critically ill status and experienced respiratory failure. The IL-6 levels of these 8 patients were also significantly higher than those of the other 32 severely ill patients, with IL-6 level in 1 patient increasing 23-fold after progression to critical illness. In addition to confirming that IL-6 was closely associated with the severity of infection, the results suggest that IL-6 plays an important role in COVID-19-associated lung injury. During COVID-19 infection, IL-6 may rapidly activate pathogenic T cells, which produce GM-CSF, IL-6, and other factors. GM-CSF further activates CD14+ CD16+ inflammatory monocytes, producing higher quantities of IL-6 and forming a positive feedback loop, resulting in diffuse damage to alveolar and pulmonary capillary endothelial cells. The large accumulation of exudates can block the airways, leading to novel coronavirus pneumonia and ARDS [29]. Because IL-6 may play an important role in the progression of novel coronavirus pneumonia, drugs that target IL-6 receptors may show efficacy in the treatment of those who are severely ill.
IL-6 is closely associated with the degree of infection of SARS-CoV-2. We investigated whether monitoring IL-6 levels in severely ill patients with COVID-19 could predict the development of critical illness. ROC curve analysis indicated that IL-6 was a good predictor of the clinical severity of COVID-19. Serum IL-6 concentrations ≥24.3 pg/mL were associated with a greater likelihood of progression to critical illness status, indicating the need for more active interventions to prevent further deterioration to a life-threatening condition.
Conclusions
This study showed that serum IL-6 concentration was closely associated with the severity of COVID-19, and that a significant increase in IL-6 may indicate that the patient’s condition is worsening and becoming critical. IL-6 levels should be measured immediately after hospital admission of patients with COVID-19 and continuously monitored thereafter. IL-6 evidently has clinical value in evaluating patient condition and predicting its deterioration, indicating the need for active therapeutic measures.
Figures
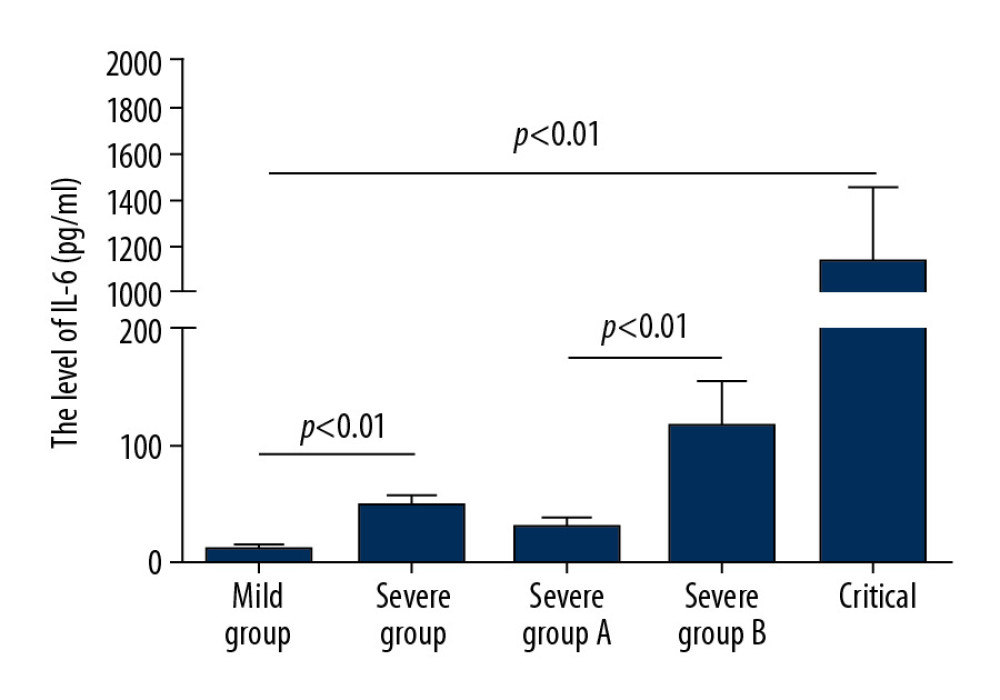 Figure 1. Serum interleukin-6 (IL-6) concentrations in patients with general, severe, severe but not critical, and critical coronavirus disease-2019 (COVID-19).
Figure 1. Serum interleukin-6 (IL-6) concentrations in patients with general, severe, severe but not critical, and critical coronavirus disease-2019 (COVID-19). 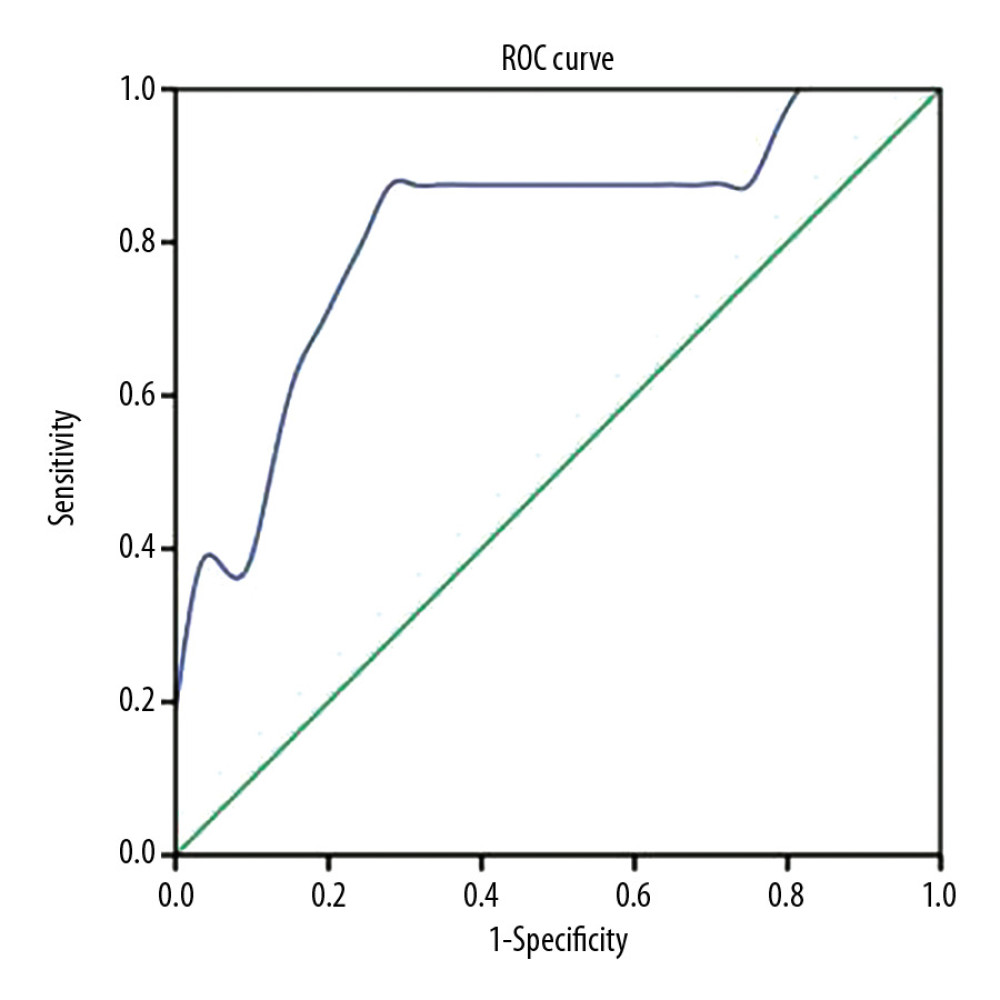 Figure 2. Receiver operating characteristic (ROC) curves of serum interleukin-6 (IL-6) concentrations in the prediction of critical illness in patients with coronavirus disease-2019 (COVID-19).
Figure 2. Receiver operating characteristic (ROC) curves of serum interleukin-6 (IL-6) concentrations in the prediction of critical illness in patients with coronavirus disease-2019 (COVID-19). Tables
Table 1. Baseline demographic and clinical characteristics of patients with coronavirus disease-2019 (COVID-19).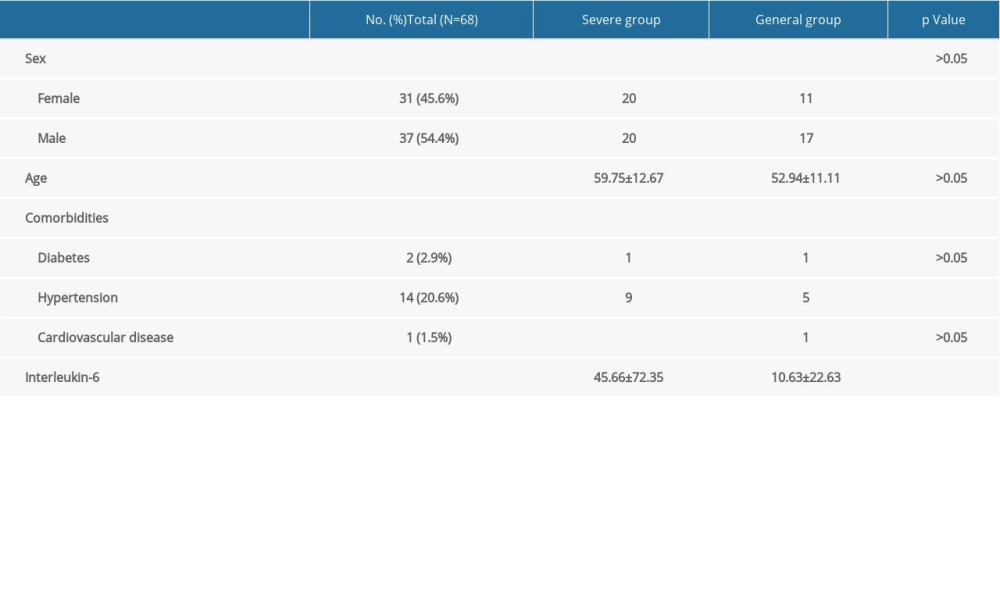 Table 2. Changes in interleukin-6 (IL-6) levels and time to progression to critical illness in patients with severe coronavirus disease-2019 (COVID-19).
Table 2. Changes in interleukin-6 (IL-6) levels and time to progression to critical illness in patients with severe coronavirus disease-2019 (COVID-19).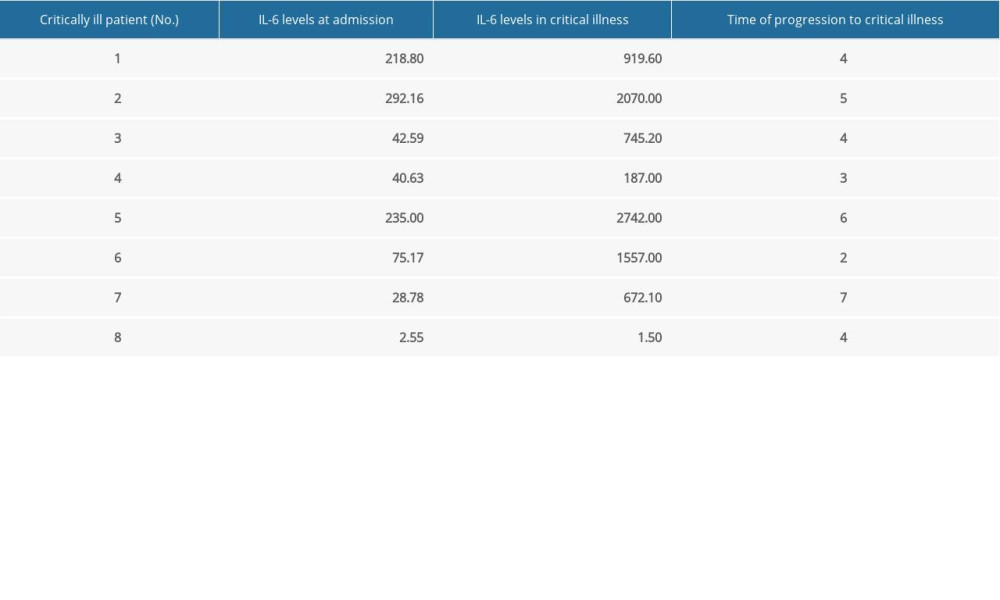 Table 3. Baseline demographic and clinical characteristics of patients with severe coronavirus disease-2019 (COVID-19).
Table 3. Baseline demographic and clinical characteristics of patients with severe coronavirus disease-2019 (COVID-19).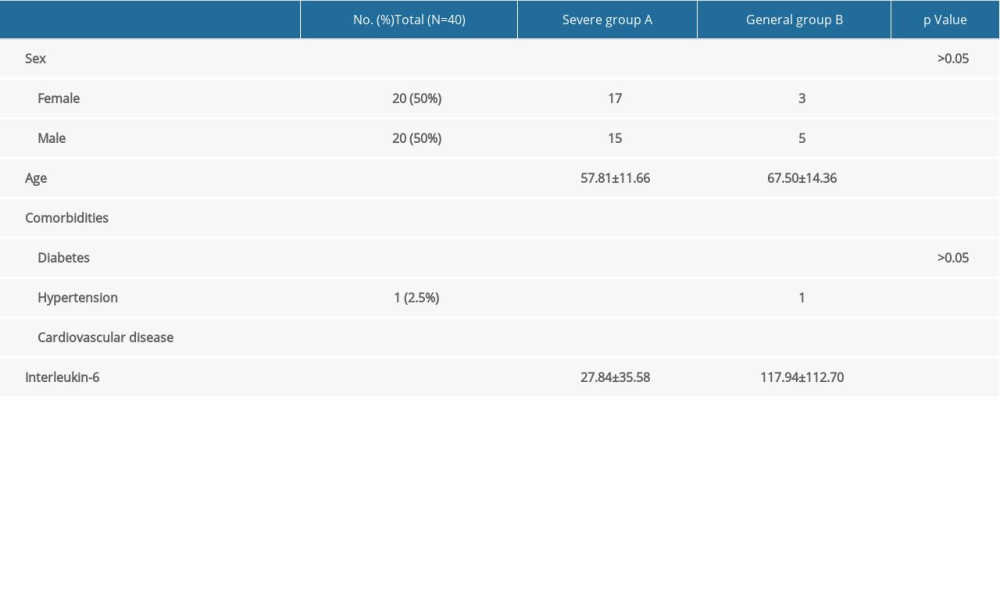
References
1. Yang X, Yu Y, Xu J, Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: A single-centered, retrospective, observational study: Lancet Respir Med, 2020; 8(5); 475-81
2. Tu YF, Chien CS, Yarmishyn AA, A review of SARS-CoV-2 and the ongoing clinical trials: Int J Mol Sci, 2020; 21(7); 2657
3. Coronaviridae Study Group of the International Committee on Taxonomy of Viruss, The species Severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-CoV-2: Nat Microbiol, 2020; 5(4); 536-44
4. Li K, Fang Y, Li W, CT image visual quantitative evaluation and clinical classification of coronavirus disease (COVID-19): Eur Radiol, 2020; 30(8); 4407-16
5. Yang F, Shi S, Zhu J, Clinical characteristics and outcomes of cancer patients with COVID-19: J Med Virol, 2020 [Online ahead of print]
6. Soy M, Keser G, Atagunduz P, Cytokine storm in COVID-19: Pathogenesis and overview of anti-inflammatory agents used in treatment: Clin Rheumatol, 2020; 39(7); 2085-94
7. Jose RJ, Manuel A, COVID-19 cytokine storm: The interplay between inflammation and coagulation: Lancet Respir Med, 2020; 8(6); e46-47
8. Reiter RJ, Sharma R, Ma Q, Melatonin inhibits COVID-19-induced cytokine storm by reversing aerobic glycolysis in immune cells: A mechanistic analysis: Med Drug Discov, 2020; 6; 100044
9. Zhao M, Cytokine storm and immunomodulatory therapy in COVID-19: Role of chloroquine and anti-IL-6 monoclonal antibodies: Int J Antimicrob Agents, 2020; 55(6); 105982
10. Arnaldez FI, O’Day SJ, Drake CG, The Society for Immunotherapy of Cancer perspective on regulation of interleukin-6 signaling in COVID-19-related systemic inflammatory response: J Immunother Cancer, 2020; 8(1); e000930
11. Lau SKP, Lau CCY, Chan KH, Delayed induction of proinflammatory cytokines and suppression of innate antiviral response by the novel Middle East respiratory syndrome coronavirus: Implications for pathogenesis and treatment: J Gen Virol, 2013; 94(Pt 12); 2679-90
12. Guo X, Zhu Z, Zhang W, Nuclear translocation of HIF-1α induced by influenza A (H1N1) infection is critical to the production of proinflammatory cytokines: Emerg Microbes Infect, 2017; 6(5); e39
13. Yasukawa K, Minami T, Point-of-care lung ultrasound findings in patients with COVID-19 pneumonia: Am J Trop Med and Hyg, 2020; 102(6); 1198-202
14. Cui N, Zou X, Xu L, Preliminary CT findings of coronavirus disease 2019 (COVID-19): Clin Imaging, 2020; 65; 124-32
15. Ulhaq ZS, Soraya GV, Interleukin-6 as a potential biomarker of COVID-19 progression: Med Maladies Infect, 2020; 50(4); 382-83
16. McGonagle D, Sharif K, O’Regan A, Bridgewood C, The role of cytokines including interleukin-6 in COVID-19 induced pneumonia and macrophage activation syndrome-like disease: Autoimmun Rev, 2020; 19(6); 102537
17. Zhang C, Wu Z, Li JW, Cytokine release syndrome in severe COVID-19: Interleukin-6 receptor antagonist tocilizumab may be the key to reduce mortality: Int J Antimicrob Agents, 2020; 55(5); 105954
18. Liu T, Zhang J, Yang Y, The role of interleukin-6 in monitoring severe case of coronavirus disease 2019: EMBO Mol Med, 2020; 12(7); e12421
19. Liu F, Li L, Xu M, Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19: J Clin Virol, 2020; 127; 104370
20. Ye Q, Wang B, Mao J, The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19: J Infect, 2020; 80(6); 607-13
21. South AM, Diz DI, Chappell MC, COVID-19, ACE2, and the cardiovascular consequences: Am J Physiol Heart Circ Physiol, 2020; 318(5); H1084-90
22. Cheng H, Wang Y, Wang GQ, Organ-protective effect of angiotensin-converting enzyme 2 and its effect on the prognosis of COVID-19: J Med Virol, 2020; 92(7); 726-30
23. Zhu Z, Cai T, Fan L, Clinical value of immune-inflammatory parameters to assess the severity of coronavirus disease 2019: Int J Infect Dis, 2020; 95; 332-39
24. Tangkijvanich P, Vimolket T, Theamboonlers A, Serum interleukin-6 and interferon-gamma levels in patients with hepatitis B-associated chronic liver disease: Asian Pac J Allergy Immunol, 2000; 18(2); 109-14
25. Chen X, Zhao B, Qu Y, Detectable serum SARS-CoV-2 viral load (RNAaemia) is closely correlated with drastically elevated interleukin 6 (IL-6) level in critically ill COVID-19 patients: Clin Infect Dis, 2020 [Online ahead of print]
26. Gao Y, Li T, Han M, Diagnostic utility of clinical laboratory data determinations for patients with the severe COVID-19: J Med Virol, 2020; 92(7); 791-96
27. Chen XY, Yan BX, Man XY, TNFα inhibitor may be effective for severe COVID-19: Learning from toxic epidermal necrolysis: Ther Adv Respir Dis, 2020; 14; 1753466620926800
28. Herold T, Jurinovic V, Arnreich C, Elevated levels of IL-6 and CRP predict the need for mechanical ventilation in COVID-19: J Allergy Clin Immunol, 2020; 146(1); 128-36.e4
29. Leng Z, Zhu R, Hou W, Transplantation of ACE2(−) mesenchymal stem cells improves the outcome of patients with COVID-19 pneumonia: Aging Dis, 2020; 11(2); 216-28
Figures
 Figure 1. Serum interleukin-6 (IL-6) concentrations in patients with general, severe, severe but not critical, and critical coronavirus disease-2019 (COVID-19).
Figure 1. Serum interleukin-6 (IL-6) concentrations in patients with general, severe, severe but not critical, and critical coronavirus disease-2019 (COVID-19). Figure 2. Receiver operating characteristic (ROC) curves of serum interleukin-6 (IL-6) concentrations in the prediction of critical illness in patients with coronavirus disease-2019 (COVID-19).
Figure 2. Receiver operating characteristic (ROC) curves of serum interleukin-6 (IL-6) concentrations in the prediction of critical illness in patients with coronavirus disease-2019 (COVID-19). Tables
 Table 1. Baseline demographic and clinical characteristics of patients with coronavirus disease-2019 (COVID-19).
Table 1. Baseline demographic and clinical characteristics of patients with coronavirus disease-2019 (COVID-19). Table 2. Changes in interleukin-6 (IL-6) levels and time to progression to critical illness in patients with severe coronavirus disease-2019 (COVID-19).
Table 2. Changes in interleukin-6 (IL-6) levels and time to progression to critical illness in patients with severe coronavirus disease-2019 (COVID-19). Table 3. Baseline demographic and clinical characteristics of patients with severe coronavirus disease-2019 (COVID-19).
Table 3. Baseline demographic and clinical characteristics of patients with severe coronavirus disease-2019 (COVID-19). Table 1. Baseline demographic and clinical characteristics of patients with coronavirus disease-2019 (COVID-19).
Table 1. Baseline demographic and clinical characteristics of patients with coronavirus disease-2019 (COVID-19). Table 2. Changes in interleukin-6 (IL-6) levels and time to progression to critical illness in patients with severe coronavirus disease-2019 (COVID-19).
Table 2. Changes in interleukin-6 (IL-6) levels and time to progression to critical illness in patients with severe coronavirus disease-2019 (COVID-19). Table 3. Baseline demographic and clinical characteristics of patients with severe coronavirus disease-2019 (COVID-19).
Table 3. Baseline demographic and clinical characteristics of patients with severe coronavirus disease-2019 (COVID-19). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








