14 November 2020: Clinical Research
A Retrospective Study of Coinfection of SARS-CoV-2 and in 11 Hospitalized Patients with Severe COVID-19 Pneumonia at a Single Center
Guillermo Rodriguez-Nava1ABCDEF*, Maria Adriana Yanez-Bello1ABDEF, Daniela Patricia Trelles-Garcia1ABDEF, Chul Won Chung1ABD, Goar Egoryan1EF, Harvey J. Friedman23ADOI: 10.12659/MSM.928754
Med Sci Monit 2020; 26:e928754
Abstract
BACKGROUND: A lethal synergism between the influenza virus and Streptococcus pneumoniae has been identified. However, bacterial coinfection is considered relatively infrequent in hospitalized patients with COVID-19, and the co-prevalence of Streptococcus pneumoniae is low.
MATERIAL AND METHODS: We retrospectively analyzed the clinical characteristics and outcomes of patients subsequently admitted to AMITA Health Saint Francis Hospital between March 1 and June 30, 2020, with documented SARS-CoV-2 and S. pneumoniae coinfection.
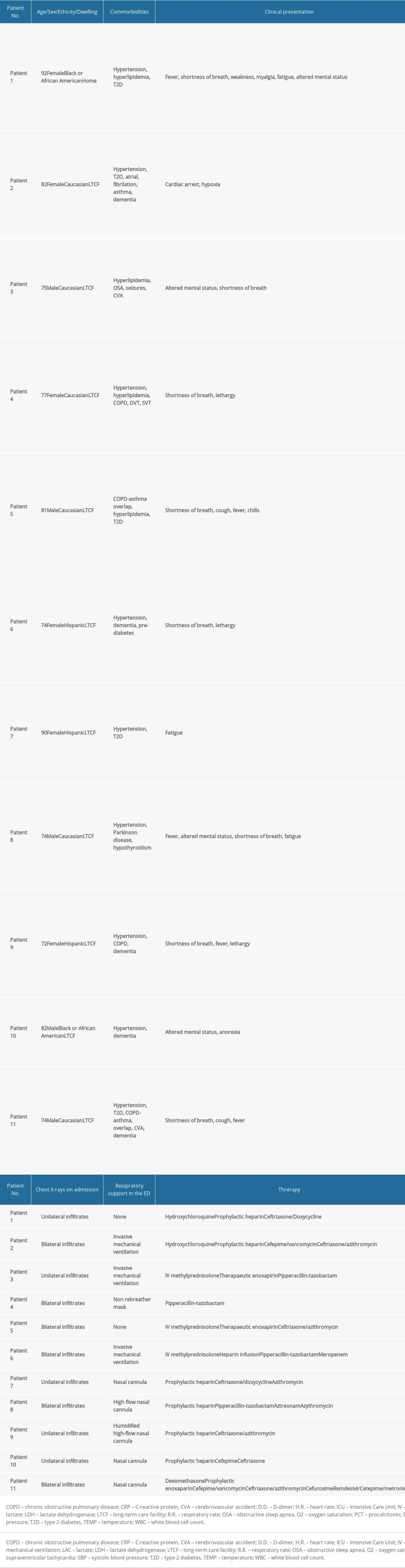
RESULTS: We identified 11 patients with S. pneumoniae coinfection. The median age was 77 years (interquartile range [IQR], 74–82 years), 45.5% (5/11) were males, 54.5% (6/11) were white, and 90.9% (10/11) were long-term care facility (LTCF) residents. The median length of stay was 7 days (IQR, 6–8 days). Among 11 patients, 4 were discharged in stable condition and 7 had died, resulting in an inpatient mortality rate of 64%.
CONCLUSIONS: At our center, 11 patients with COVID-19 pneumonia who had confirmed infection with SARS-CoV-2 were diagnosed with Streptococcus pneumoniae infection while in hospital. All patients had pneumonia confirmed on imaging and a nonspecific increase in markers of inflammation. The in-hospital mortality rate of 64% (7 patients) was higher in this group than in previous reports. This study highlights the importance of monitoring bacterial coinfection in patients with viral lung infection due to SARS-CoV-2.
Keywords: COVID-19, SARS Virus, Streptococcus pneumoniae, Aged, 80 and over, COVID-19, coinfection, Datasets as Topic, Hospital Mortality, Hospitalization, Pandemics, Pneumonia, Pneumococcal, SARS-CoV-2, Severity of Illness Index
Background
Since December 2019, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus had caused over 3 million cases of coronavirus disease 2019 (COVID-19) and more than 150 000 deaths in the U.S. [1]. As the pandemic evolves, the scientific and medical community accumulates more evidence of the disease’s clinical characteristics; however, information regarding bacterial coinfections is still minimal and sometimes overlooked [2].
Between 1918 and 1919, the “Spanish” influenza pandemic caused around 50 million deaths worldwide [3]. Preserved lung tissue sections and autopsy analyses have shown that most of these deaths likely resulted directly from secondary bacterial pneumonia by common upper-respiratory tract bacteria, with
In contrast, bacterial coinfection is considered relatively infrequent in hospitalized patients with COVID-19, and the co-prevalence of
Material and Methods
We retrospectively analyzed clinical characteristics and outcomes of the patients subsequently admitted to AMITA Health Saint Francis Hospital between March 1 and June 30, 2020, with documented SARS-CoV-2 infection by RT-PCR or isothermal nucleic acid amplification performed on nasopharyngeal throat swab specimens (Abbott™ RealTi
The Abbott™ RealTi
The study and creation of this de-identified dataset were approved by the Institutional Review Board of AMITA Health System (2020-0128-02). The Ethics Commission waived the requirement for informed consent given this research involves no more than minimal risk to subjects. Descriptive statistics were used to summarize results. The data were analyzed using SPSS Version 23.0 (IBM Corp, Armonk, NY).
Results
We identified 11 patients with
Discussion
In this retrospective study, we describe 11 hospitalized patients with a coinfection of SARS-CoV-2 and
Bacterial coinfection is considered uncommon in patients with COVID-19 who are newly admitted to the hospital [6–10]. Initial Chinese reports found a 5.1% to 38.9% incidence rate of secondary or hospital-acquired bacterial coinfections, and later reports in Western countries showed coinfection rates of 4.8% to 27.4% [2]. A retrospective cohort study that included 836 hospitalized patients with confirmed SARS-CoV-2 found that 27 (3.2%) patients had early confirmed bacterial isolates identified (0–5 days after admission), rising to 51 (6.1%) throughout the hospital stay. However, no patients with pneumococcal coinfection were identified [11]. Another large cohort study with 989 hospitalized patients identified a total of 88 non-COVID-19 infections in 72 patients (7.3%). Among those, 74 were bacterial, with
Despite the low frequency of coinfections reported in patients with COVID-19, they are associated with poor outcomes. Up to 50% of patients who have died of COVID-19 had secondary bacterial infections [8]. Furthermore, in the review mentioned above by Lansbury et al., the pooled analysis of crude odds ratios for death indicated that COVID-19 patients with a coinfection were 5.82 times more likely to die than patients who did not have a coinfection [12].
The present study has several limitations. First, this was a single-center study, and these findings may be subject to selection bias, and generalizability to larger populations of patients is limited. Second, given the situation’s novelty, our institution did not have standard practices regarding testing for other respiratory pathogens. This was subject to each attending physician’s discretion; hence, there may have been high microbiological sampling variability with only sicker patients being tested for other pathogens. Third, 90.9% of the patients studied were long-term care facility residents. These patients present a higher burden of underlying conditions and low baseline functional status, which may account for the higher mortality observed compared to other cohorts. Fourth, given the study’s observational nature, causality cannot be inferred from an uncontrolled observation. Whether there is a lethal synergism between SARS-CoV-2 and
Conclusions
At our center, 11 patients with COVID-19 pneumonia who had confirmed infection with SARS-CoV-2 were diagnosed with
References
1. Centers for Disease Control and Prevention: Coronavirus Disease 2019 (COVID-19) – Cases in US August 10, 2020
2. Antinori S, Galimberti L, Milazzo L, Ridolfo AL, Bacterial and fungal infections among patients with SARS-CoV-2 pneumonia: Infez Med, 2020; 28(Suppl 1); 29-36
3. Taubenberger JK, Morens DM, 1918 Influenza: The mother of all pandemics: Emerging Infect Dis, 2006; 12(1); 15-22
4. Morens DM, Taubenberger JK, Fauci AS, Predominant role of bacterial pneumonia as a cause of death in pandemic influenza: Implications for pandemic influenza preparedness: J Infect Dis, 2008; 198(7); 962-70
5. Rudd JM, Ashar HK, Chow VT, Teluguakula N: J Infect Pulm Dis, 2016; 2(2); 3176.114
6. Lin D, Liu L, Zhang M, Coinfections of SARS-CoV-2 with multiple common respiratory pathogens in infected patients: Sci China Life Sci, 2020; 63(4); 606-9
7. Garcia-Vidal C, Sanjuan G, Moreno-garcía E, Incidence of coinfections and superinfections in hospitalized patients with COVID-19: A retrospective cohort study: Clin Microbiol Infect, 2020 [Online ahead of print]
8. Cox MJ, Loman N, Bogaert D, O’Grady J, Co-infections: Potentially lethal and unexplored in COVID-19: Lancet Microbe, 2020; 1(1); e11
9. Rawson TM, Moore LSP, Zhu N, Bacterial and fungal coinfection in individuals with coronavirus: A rapid review to support COVID-19 antimicrobial prescribing: Clin Infect Dis, 2020 [Online ahead of print]
10. Adler H, Ball R, Fisher M, Low rate of bacterial coinfection in patients with COVID-19: Lancet Microbe, 2020; 1(2); e62
11. Hughes S, Troise O, Donaldson H, Bacterial and fungal coinfection among hospitalized patients with COVID-19: A retrospective cohort study in a U.K. secondary-care setting: Clin Microbiol Infect, 2020; 26(10); 1395-99
12. Lansbury L, Lim B, Baskaran V, Lim WS, Coinfections in people with COVID-19: A systematic review and meta-analysis: J Infect, 2020; 81(2); 266-75
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387