07 May 2021: Clinical Research
Real-World Efficacy of Proprotein Convertase Subtilisin/Kexin Type 9 Inhibitors (PCSK9i) in Heterozygous Familial Hypercholesterolemia Patients Referred for Lipoprotein Apheresis
Anthony Matta123ABCDEF*, Vanina Bongard12ABCDEF, Frédéric Bouisset12ABCDEF, Dorota Taraszkiewicz12ABCDEF, Jean-Pierre Rabès4BDEF, Jean Ferrières12ABCDEFGDOI: 10.12659/MSM.928784
Med Sci Monit 2021; 27:e928784
Abstract
BACKGROUND: A small proportion of familial hypercholesterolemia (FH) patients can adequately control this condition, although achieving the recommended targets for low-density lipoprotein cholesterol (LDL-c) levels remains a challenge. Proprotein convertase subtilisin/kexin type 9 inhibitors (PCSK9i) are new and potent lipid-lowering drugs. However, there is scarce literature on real-world data about their use in patients with FH.
MATERIAL AND METHODS: We examined the reduction in LDL-c levels from the baseline, after PCSK9i initiation in heterozygous familial hypercholesterolemia patients referred for lipoprotein apheresis in our regional lipid clinic. The study was conducted from March 2018 to September 2019, the period immediately after PCSK9i reimbursement was available in France. PCSK9i was added on top of the patients’ maximal tolerated lipid-lowering regimens.
RESULTS: The study had 123 patients with heterozygous FH. The mean age of the patients was 59±11 years. The mean baseline LDL-c for all the participants was 277±78 mg/dl. It was 283±81 mg/dl in the PCSK9i monotherapy group (n=83), 247±68 mg/dl in the PCSK9i plus ezetimibe group (n=12), and 264±78 mg/dl in the PCSK9i plus statin and ezetimibe group (n=28). The mean decrease observed in the LDL-c level from baseline was 136±70 mg/dl (n=123), 125±60 mg/dl (n=83), 103±77 mg/dl (n=12), and 175±70 mg/dl (n=28), respectively.
CONCLUSIONS: An overall reduction of 49.1% from the baseline LDL-c was observed in the heterozygous FH population after PCSK9i initiation in a real-world experience. The group treated with PCSK9i ezetimibe plus statin showed further reduction of their LDL-c levels with a better responder rate, achieving the target 50% reduction in LDL-c from the baseline.
Keywords: Cholesterol, LDL, dyslipidemias, Proprotein Convertases, Blood Component Removal, Cohort Studies, Hydroxymethylglutaryl-CoA Reductase Inhibitors, Hyperlipoproteinemia Type II, Hypolipidemic Agents, Lipid Metabolism, Lipids, Lipoproteins, PCSK9 Inhibitors, Subtilisins
Background
Familial hypercholesterolemia (FH) is a common, autosomal dominant hereditary disease caused by mutations in the genes encoding key proteins involved in the low-density lipoprotein (LDL) receptor’s (LDLR) endocytic and recycling pathways. The LDLR gene encodes the LDLR, the apolipoprotein B gene encodes apolipoprotein B (the ligand of the LDL receptor), and the PCSK9 gene encodes a serine protease (proprotein convertase subtilisin/kexin type 9 or PCSK9), which plays a role in the degradation of the LDLR [1]. Mutations in the LDLR adaptor protein 1 gene are responsible for the recessive form of the disease [2]. However, FH is an underdiagnosed and undertreated genetic disorder that results in premature mortality and morbidity due to atherosclerotic disease. Myocardial infarction, stroke, or other cardiovascular events could be the first clinical presentation of FH [3]. European guidelines classify FH patients at high cardiovascular risk and the optimal target for low-density lipoprotein cholesterol (LDL-c) is ≤70 mg/dl with ≥50% reduction from the level at baseline [4]. Reaching these LDL-c goals in clinical practice remains a challenge for physicians.
All the statins have proven their effectiveness in significantly lowering LDL-c levels (upregulating the LDL receptor in the liver and peripheral tissues) and are recommended as first-line therapy. Nevertheless, muscle and joint pain are common adverse effects associated with statin therapy, which lead to an interruption of medical treatment in real-life practice. PCSK9 inhibitors (PCSK9i) have recently emerged as novel medications indicated for patients who cannot tolerate statins, and cannot achieve the required optimal LDL-c levels [5].
Clinical trials have shown that PCSK9i can effectively reduce the baseline LDL-c levels by 60% on average, when used alone or combined with other lipid-lowering agents [4]. Current trials are investigating PCSK9i as an alternative therapeutic option for patients undergoing lipoprotein apheresis, as it shows a synergistic effect leading to a drop in extracorporeal apheresis sessions and lipoprotein apheresis withdrawal (in some cases) [6]. Concerns regarding the inter-individual variability, FH mutations, LDLR abnormalities, and the importance of pretreatment with statins in patients on PCSK9i have been raised, as all these factors are well-known predictors of the efficacy of lipid-lowering drugs. In the Trial Evaluating PCSK9 Antibody in Subjects With LDL Receptor Abnormalities (TESLA) [7], neither a single autosomal recessive hypercholesterolemia patient nor a single null/null LDLR homozygote patient responded to evolocumab. Similar results were observed in larger numbers of patients for both these patient categories in the Trial Assessing Long-Term Use of PCSK9 Inhibition in Subjects With Genetic LDL Disorders (TAUSSIG) study [8]. Robinson et al reported that PCSK9i is more effective in patients with higher levels of circulating PCSK9 stimulated by previous statin therapy [9]. In addition, the failure to respond to PCSK9i occurs more frequently, as PSCK9i is more commonly prescribed [10].
The real-world efficacy of PCSK9i regarding patients’ characteristics and the severity of LDL-c levels are not known since the vast majority of randomized controlled trials have been conducted on patients receiving combination therapy (PCSK9i and statin), and patients with low LDL-c before treatment [11–14]. Therefore, we aimed to investigate the variation in LDL-c levels after initiation of PCSK9i, either as monotherapy, or in combination with previously prescribed ezetimibe or statin plus ezetimibe in a real-life cohort study of severely dyslipidemic heterozygous FH patients referred for lipoprotein apheresis. In this study population, the baseline LDL-c level was one of the highest reported in literature.
Material and Methods
STUDY DESIGN AND POPULATION:
An observational single-center cohort study was conducted on patients referred for lipoprotein apheresis at our Lipid, Atherosclerosis, and Preventive Cardiology Clinic at Rangueil University Hospital, Toulouse, France between March 2018 and September 2019. Patients were eligible for participating in the study if they were diagnosed with FH according to the Dutch Lipid Clinic Network score [15]. In France, alirocumab is reimbursed only for patients treated for primary prevention (LDL-c ≥300 mg/dl) or secondary prevention (LDL-c ≥200 mg/dl) in the setting of an experienced lipoprotein apheresis center. Therefore, patients with these baseline LDL-c levels were requested to participate in the study. The patients were divided into 3 groups based on their real-world prescriptions in this observational study: those treated with PCSK9i only, those treated with PCSK9i in addition to the preexisting ezetimibe therapy, and those treated with PCSK9i in addition to the preexisting statin plus ezetimibe therapy. Alirocumab monotherapy was initiated in the statin-intolerant patients. Statin intolerance was defined as an inability in the past to tolerate 2 or more statins (1 statin at the lowest daily starting dose). Alirocumab was added on top of a maximal tolerated lipid-lowering regimen in patients taking either ezetimibe or a statin plus ezetimibe. The alirocumab was prescribed as a 150 mg subcutaneous injection administered every 2 weeks.
DATA COLLECTION AND END POINTS:
During the initial visit, a physician performed a detailed physical examination, and recorded family and personal medical histories of the participants. The issues of incorrect injection techniques and non-adherence to treatment were eliminated, as the medical team supervised the injections. Full lipid panels at the initiation of the PCSK9i therapy (baseline LDL-c) and 6 months after the initiation of the PCSK9i treatment were collected. Based on the 2019 European Society of Cardiology (ESC) guidelines for FH management, ≥50% reduction from the LDL-c baseline was considered a satisfactory response to PCSK9i in this study. The main outcome was the variation in LDL-c levels, 6 months from the baseline levels after initiation of the PCSK9i treatment in the 3 groups of patients. None of the patients had received a statin without ezetimibe before inclusion in the study. The secondary outcome was the percentage of responders at 6 months (ie, ≥50% drop in LDL-c).
STATISTICAL ANALYSIS:
Continuous data were summarized as means and standard deviations, and categorical variables were summarized as counts and percentages. The continuous variables were compared between the 3 groups through an analysis of variance (ANOVA). The bivariate associations with the categorical variables were studied with the chi-square test or Fisher’s exact test as appropriate.
ANOVA was used to compare the mean change in LDL-c levels between the groups. A multivariate linear regression analysis was conducted for studying percentage variations in the LDL-c levels from baseline in the 3 groups. The patients treated with PCSK9i alone were considered the reference group. A multivariate logistic regression analysis was conducted to study the satisfactory response to treatment as a dependent variable. The multivariate analyses were adjusted for age, sex, type of prevention (primary or secondary), and the baseline lipid panel. A
The patients were seen at our clinic within a regular follow-up; therefore, approval from the Ethics Committee was not needed. The patients gave oral consent for participation in the study, which followed the institution’s ethical rules.
Results
A total of 123 patients with heterozygous FH were included in the study (Supplementary Online Table 1). They were divided into 3 groups. The baseline characteristics of these participants are shown in Table 1. Their mean age was 59±11 years; there were 66 (53.7%) men and 57 (46.3%) women. Four (3.3%) patients were smokers, 30 (24.4%) patients had systemic hypertension, 9 (7.3%) patients had diabetes mellitus, 28 (22.8%) patients were taking statin and ezetimibe prior to the PCSK9i initiation, and 95 (77.2%) patients were statin-intolerant. Of the statin-intolerant patients, 12 (9.8%) patients were taking ezetimibe prior to the PCSK9i initiation and 83 (67.5%) patients did not receive any lipid-lowering agents before the PCSK9i initiation. Twenty-nine (23.6%) patients were being treated for primary prevention. Among the 94 (76.4%) patients treated for secondary prevention, 82 (87.2%) patients had coronary artery disease, 3 (3.2%) patients had a history of ischemic stroke, and 12 (12.8%) patients had peripheral artery disease. The overall mean total cholesterol, LDL-c, high-density lipoprotein cholesterol (HDL-c), and triglyceride baseline levels were 361±83 mg/dl, 277±78 mg/dl, 52±13 mg/dl, and 168±69 mg/dl, respectively.
The mean baseline LDL-c level dropped from 277±78 mg/dl to 141±84 mg/dl in the entire study population, accounting for a 49.1% reduction. The mean baseline LDL-c level showed a 44.2% reduction (from 283±81 mg/dl to 158±83 mg/dl) in the PCSK9i monotherapy group (n=83), a 41.7% reduction in LDL-c (from 247±68 mg/dl to 144±100 mg/dl) in the PCSK9i plus ezetimibe group (n=12), and a 66.3% reduction in LDL-c (from 264±78 mg/dl to 89±57 mg/dl) in the PCSK9i plus statin and ezetimibe group (n=28) (Figures 1, 2). Statistical analysis (ANOVA) showed a statistically significant difference in the mean LDL-c reduction between the PCSK9i plus statin and ezetimibe group compared to the other 2 groups (PCSK9i only,
There were 61/123 (49.6%) responders in the study population, who achieved the target of at least 50% reduction in their LDL-c level from the baseline. They were 32/83 (38.6%) patients from the PCSK9i only group, 7/12 (58.3%) patients from the PCSK9i plus ezetimibe group, and 22/28 (78.6%) patients from the PCSK9i plus statin and ezetimibe group. Statistical analysis showed a positive statistically significant relationship between the PCSK9i plus statin and ezetimibe group and the responder rate (OR=6, CI [2–17.6],
Discussion
LIMITATIONS:
This study was a single-center experience reflecting the findings for PCSK9i use in a regional lipid clinic. Only alirocumab was used and its effects are reported in this study. The restricted inclusion criteria were defined according to the conditions for PCSK9i-reimbursement approval by the French health authorities. The 6-month follow-up is a short period to monitor the long-term LDL-c efficacy in FH patients.
Conclusions
We presented a single-center study showing an overall efficacy of PCSK9i in real life for the heterozygous FH population referred for lipoprotein apheresis. In these patients, the association of statin is essential for a greater reduction in LDL-c and an improved chance to achieve the target goals as a significant difference has been shown between PCSK9i monotherapy and the statin-associated regimen. Further research is needed to disentangle the best predictive parameters of PCSK9i efficacy in this severe FH population.
Figures
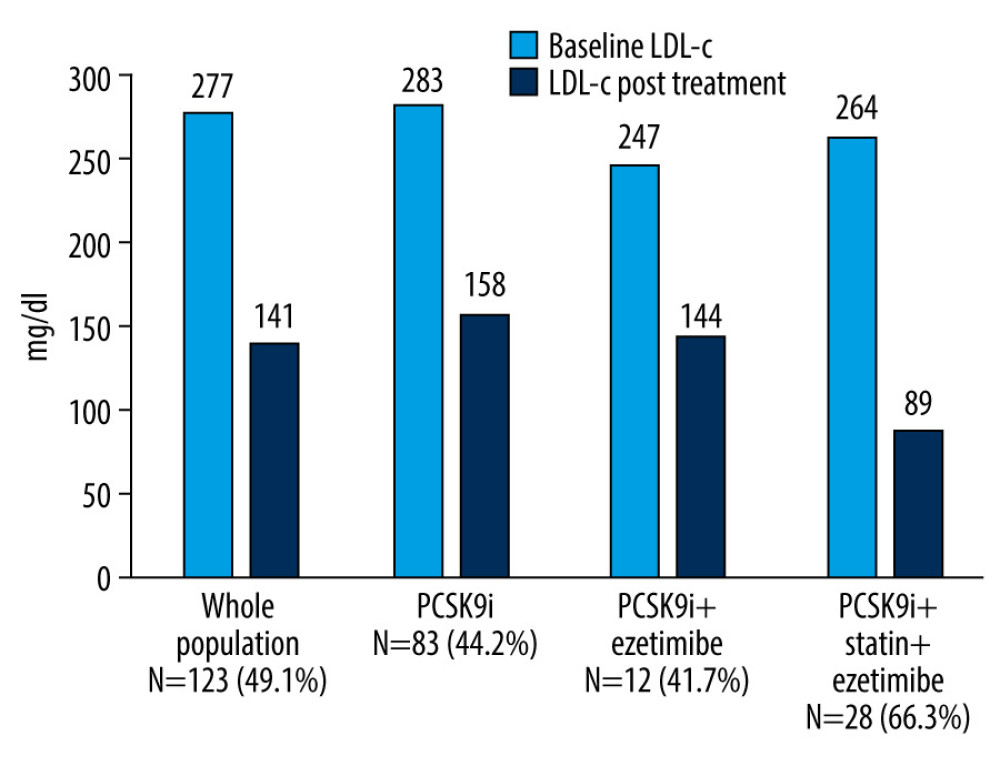 Figure 1. Low-density lipoprotein cholesterol reduction (%) from the baseline after PCSK9i initiation. PCSK9i – proprotein convertase subtilisin/kexin type 9 inhibitor.
Figure 1. Low-density lipoprotein cholesterol reduction (%) from the baseline after PCSK9i initiation. PCSK9i – proprotein convertase subtilisin/kexin type 9 inhibitor. 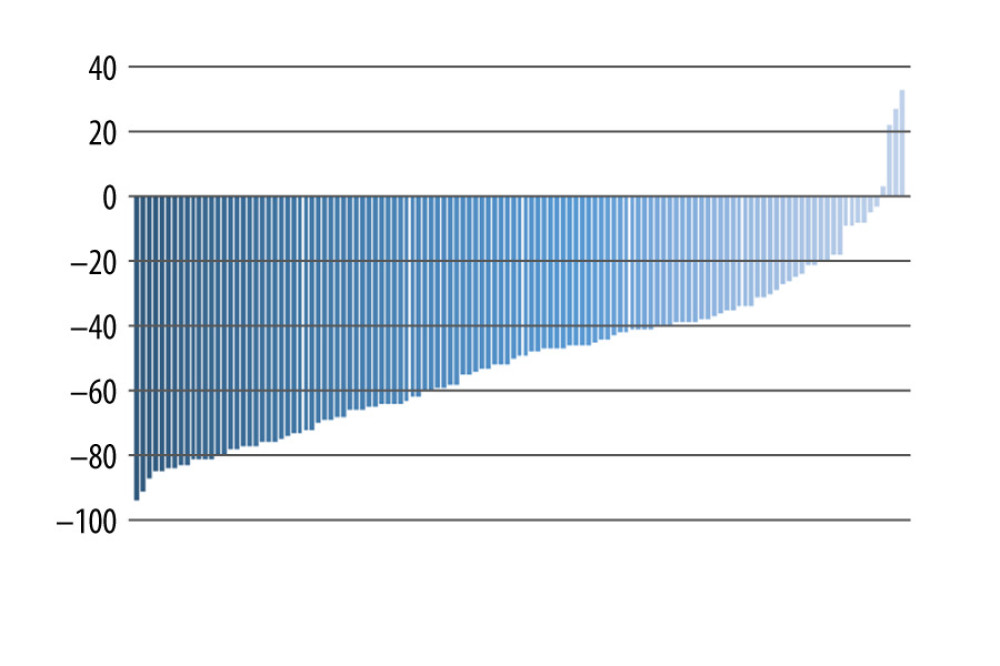 Figure 2. Waterfall plot of the variation (%) in low-density lipoprotein cholesterol of 123 heterozygous familial hypercholesterolemia patients after initiation of PCSK9i. PCSK9i – proprotein convertase subtilisin/kexin type 9 inhibitor.
Figure 2. Waterfall plot of the variation (%) in low-density lipoprotein cholesterol of 123 heterozygous familial hypercholesterolemia patients after initiation of PCSK9i. PCSK9i – proprotein convertase subtilisin/kexin type 9 inhibitor. References
1. Rabès JP, Béliard S, Carrié A, Familial hypercholesterolemia: Experience from France: Curr Opin Lipidol, 2018; 29; 65-71
2. Garcia CK, Wilund K, Arca M, Autosomal recessive hypercholesterolemia caused by mutations in a putative LDL receptor adaptor protein: Science, 2001; 292(5520); 1394-98
3. Gidding SS, Champagne MA, de Ferranti SD, American Heart Association Atherosclerosis Hypertension and Obesity in Young Committee of Council on Cardiovascular Disease in Young, Council on Cardiovascular and Stroke Nursing, Council on Functional Genomics and Translational Biology, and Council on Lifestyle and Cardiometabolic Health: The agenda for familial hypercholesterolemia: A scientific statement from the American Heart Association. Circulation, 2015; 132; 2167-92
4. Mach F, Baigent C, Catapano AL, 2019 ESC/EAS Guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk: The Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and European Atherosclerosis Society (EAS): Eur Heart J, 2020; 41(1); 111-88
5. Blanchard V, Khantalin I, Ramin-Mangata S, PCSK9: From biology to clinical applications: Pathology, 2019; 51(2); 177-83
6. Bláha V, Bláha M, Lánská M, The role of PCSK9-inhibitors and of lipoprotein apheresis in the treatment of homozygous and severe heterozygous familial hypercholesterolemia: A rivalry, or are things quite different?: Vnitr Lek (Winter), 2018; 64(1); 43-50
7. Raal FJ, Honarpour N, Blom DJ, Inhibition of PCSK9 with evolocumab in homozygous familial hypercholesterolemia (TESLA Part B) : A randomised, double blind, placebo-controlled trial: Lancet, 2014; 385(9965); 341-50
8. Raal FJ, Hovingh JK, Blom D, Long-term treatment with evolocumab added to conventional drug therapy, with or without apheresis, in patients with homozygous familial hypercholesterolaemia: An interim subset analysis of the open-label TAUSSIG study: Lancet Diabetes Endocrinol, 2017; 5(4); 280-90
9. Robinson JG, Farnier M, Kastelein JJP, Relationship between alirocumab, PCSK9, and LDL-C levels in four phase 3 ODYSSEY trials using 75 and 150 mg doses: J Clin Lipidol, 2019; 13(6); 979-88
10. Saeed A, Virani SS, Jones PH, Case reports of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibition nonresponse: J Clin Lipidol, 2018; 12(5); 1141-45
11. Matta A, Taraszkiewicz D, Bongard V, Ineffective Subtilisin/Kexin type 9 inhibitors monotherapy in dyslipidemia with low density lipoprotein cholesterol (LDL-c) receptor abnormalities: A report of 2 cases: Am J Case Rep, 2020; 21; e923722
12. Bays HE, Rosenson RS, Baccara-Dinet MT, Assessment of the 1% of patients with consistent <15% reduction in low-density lipoprotein cholesterol: Pooled analysis of 10 phase 3 ODYSSEY alirocumab trials: Cardiovasc Drugs Ther, 2018; 32(2); 175-80
13. Rymer JA, Mues KE, Monda KL, Use of low density lipoprotein lowering therapies before and after PCSK9 inhibitor initiation: J Am Heart Assoc, 2020; 9(9); e014347
14. Hollstein T, Kassner U, Grenkowitz T, PCSK9 inhibitors in a German single center clinical practice: Real-world treatment of patients at high cardiovascular risk over 68 weeks: Am J Cardiovasc Drugs, 2020; 21(1); 83-92
15. Watts GF, Gidding AS, Toth PP, Integrated guidance on the care of familial hypercholesterolaemia from the International FH foundation: Int J Cardiol, 2014; 171; 309-25
16. Landmesser U, Chapman MJ, Farnier M, European Society of Cardiology/European Atherosclerosis Society Task Force consensus statement on proprotein convertase subtilisin/kexin type 9 inhibitors: Practical guidance for use in patients at very high cardio-vascular risk: Eur Heart J, 2017; 38; 2245-55
17. Ridker PM, Revkin J, Amarenco P, Cardiovascular efficacy and safety of bococizumab in high-risk patients: N Engl J Med, 2017; 376; 1527-39
18. Razek O, Cermakova L, Armani H, Attainment of recommended lipid targets in patients with familial hypercholesterolemia: Real-world experience with PCSK9 inhibitors: Can J Cardiol, 2018; 34(8); 1004-9
19. Sarsam S, Berry A, Degheim G, Real-world use of PCSK9 inhibitors: A single-center experience: J Int Med Res, 2019; 47(1); 265-70
20. Parhofer KG, Stritzky BV, Pietschmann N, PEARL: A non interventional study of real-world alirocumab use in German clinical practice: Drugs- Real World Outcomes, 2019; 6(3); 115-23
21. Graesdal A, Dybvig A, Real-life PCSK9 experience: Efficacy, compliance and side effects after one year treatment in familial hypercholesterolemia patients: J Am Coll Cardiol, 2018; 71(11S); A1754
22. Kaufman TM, Warden BA, Minnier J, Application of PCSK9 inhibitors in practice: part 2: The patient experience: Circ Res, 2019; 124(1); 32-37
23. Deshmukh H, Narayanan D, Papageorgiou M, PCSK9 inhibitors in familial hypercholesterolaemia: A real-world experience and a meta-analysis: Br J Cardiol, 2019; 26; 137-40
24. Zafrir B, Jubran A, Lipid-lowering therapy with PCSK9-inhibitors in the real-world setting: Two-year experience of a regional lipid-clinic: Cardiovasc Ther, 2018; 36; e12439
25. Stoekenbroek RM, Hartgers ML, Rutte R, PCSK9 inhibitors in clinical practice: Delivering on the promise?: Atherosclerosis, 2018; 270; 205-10
26. Galema-Boers AM, Lenzen MJ, Sijbrands EJ, Proprotein convertase subtilisin/kexin 9 inhibition in patients with familial hypercholesterolemia: Initial clinical experience: J Clin Lipidol, 2017; 11(3); 674-81
27. Rallidis LS, Skoumas I, Liberopoulos EN, PCSK9 inhibitors in clinical practice: Novel directions and new experiences: Hellenic J Cardiol, 2020; 61(4); 241-45
28. Nozue T, Lipid lowering therapy and circulating PCSK9 concentration: J Atheroscler Thromb, 2017; 24(9); 895-907
29. Kastelein JJ, Ginsberg HN, Langslet G, ODYSSEY FHI and FHII: 78 week results with alirocumab treatment in 735 patients with heterozygous familial hypercholesterolaemia: Eur Heart J, 2015; 36(43); 2996-3003
30. Raal FJ, Stein EA, Dufour R, PCSK9 inhibition with evolocumab (AMG 145) in heterozygous familial hypercholesterolaemia (RUTHEREFORD-2) : A randomised, double-blind, placebo-controlled trial: Lancet, 2015; 385(9965); 331-40
31. Kastelein JJ, Hovingh GK, Langslet G, Efficacy and safety of the proprotein convertase subtilisin/kexin type 9 monoclonal antibody alirocumab vs placebo in patients with heterozygous familial hypercholesterolemia: J Clin Lipidol, 2017; 11(1); 195-203
Figures
 Figure 1. Low-density lipoprotein cholesterol reduction (%) from the baseline after PCSK9i initiation. PCSK9i – proprotein convertase subtilisin/kexin type 9 inhibitor.
Figure 1. Low-density lipoprotein cholesterol reduction (%) from the baseline after PCSK9i initiation. PCSK9i – proprotein convertase subtilisin/kexin type 9 inhibitor. Figure 2. Waterfall plot of the variation (%) in low-density lipoprotein cholesterol of 123 heterozygous familial hypercholesterolemia patients after initiation of PCSK9i. PCSK9i – proprotein convertase subtilisin/kexin type 9 inhibitor.
Figure 2. Waterfall plot of the variation (%) in low-density lipoprotein cholesterol of 123 heterozygous familial hypercholesterolemia patients after initiation of PCSK9i. PCSK9i – proprotein convertase subtilisin/kexin type 9 inhibitor. Tables
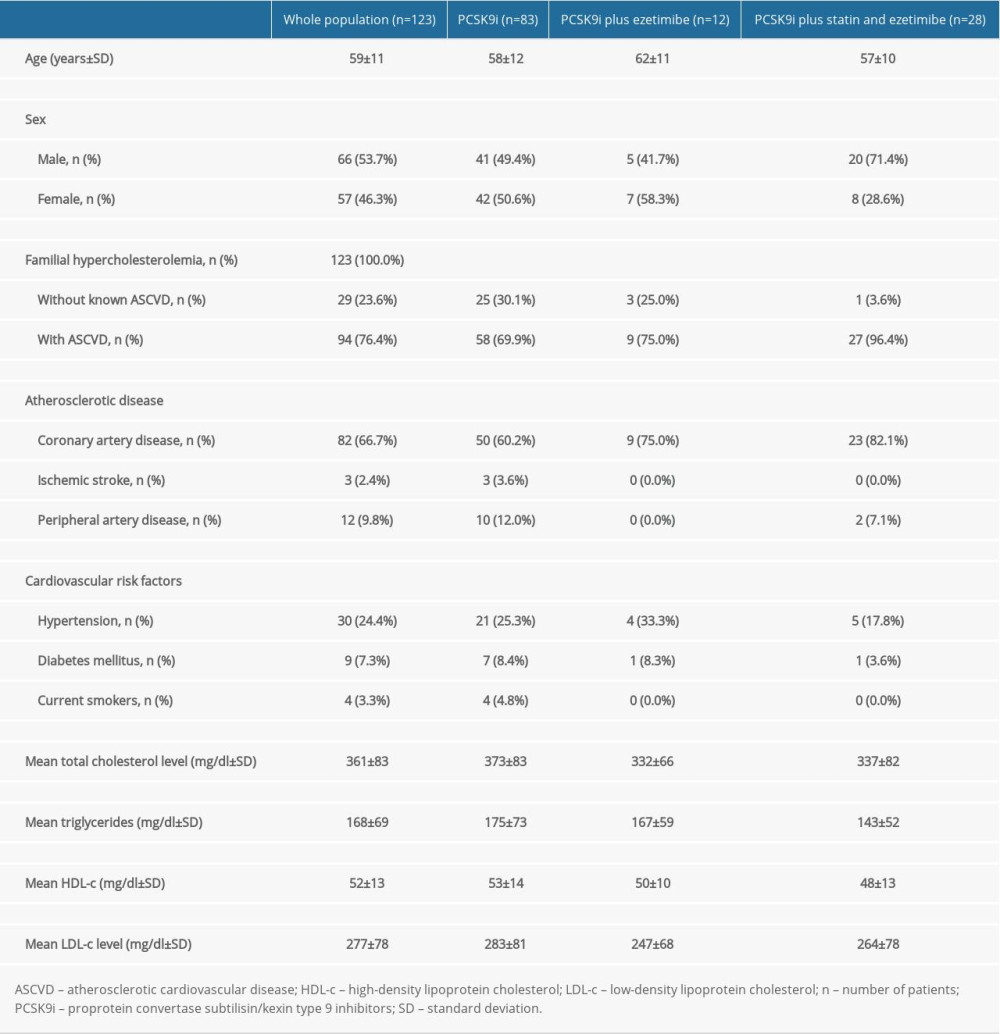 Table 1. Baseline characteristics of the study population.
Table 1. Baseline characteristics of the study population.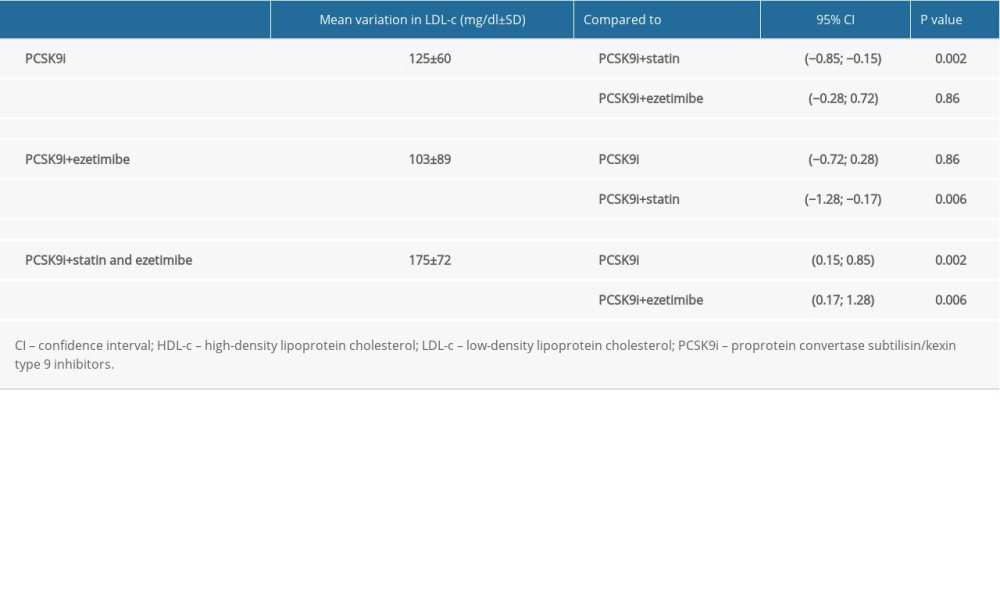 Table 2. Analysis of variance (ANOVA) comparing the reduction in mean low-density lipoprotein cholesterol between the 3 groups after adding PCSK9i.
Table 2. Analysis of variance (ANOVA) comparing the reduction in mean low-density lipoprotein cholesterol between the 3 groups after adding PCSK9i.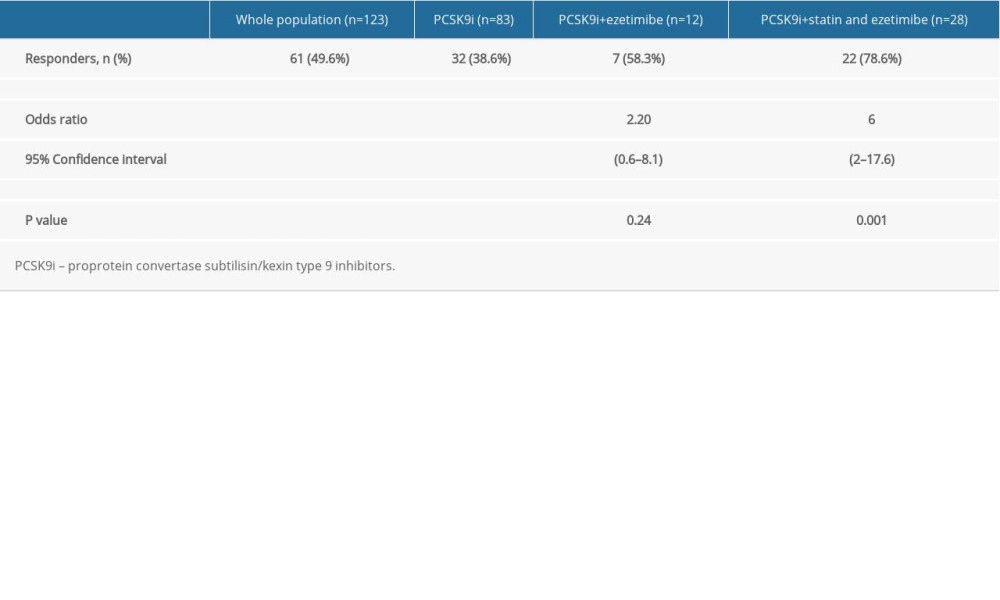 Table 3. A comparison of the responder rate between the 3 groups.
Table 3. A comparison of the responder rate between the 3 groups. Table 1. Baseline characteristics of the study population.
Table 1. Baseline characteristics of the study population. Table 2. Analysis of variance (ANOVA) comparing the reduction in mean low-density lipoprotein cholesterol between the 3 groups after adding PCSK9i.
Table 2. Analysis of variance (ANOVA) comparing the reduction in mean low-density lipoprotein cholesterol between the 3 groups after adding PCSK9i. Table 3. A comparison of the responder rate between the 3 groups.
Table 3. A comparison of the responder rate between the 3 groups. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








