17 May 2021: Clinical Research
Immunogenicity of Split Inactivated Quadrivalent Influenza Vaccine in Adults with Obesity in the 2017/2018 Season
Anna M. JagielskaDOI: 10.12659/MSM.929572
Med Sci Monit 2021; 27:e929572
Abstract
BACKGROUND: Obesity is associated with susceptibility to severe influenza infection and several disturbances of the immune response to the influenza vaccine. However, the effect of obesity on the immunogenicity of the influenza vaccine is not fully understood. Our objective here was to assess the immunogenicity of the split, inactivated quadrivalent influenza vaccine (QIV) in Polish adults with obesity.
MATERIAL AND METHODS: Fifty-three subjects with obesity aged 21-69 years were vaccinated with the QIV in 2017/2018 season. Antibody titers against the 4 vaccine strains were measured using the hemagglutination inhibition (HI) assay. The mean fold antibody increase (MFI), seroprotection rate (protection rate, PR), and seroconversion rate (response rate, RR) were calculated to assess vaccine immunogenicity.
RESULTS: The vaccine elicited a significant increase in the anti-HI titers against the QIV antigens. The MFI, PR, and RR for the QIV antigens also reached the required age-specific values, indicating the QIV meets current immunogenicity criteria. Individuals with class I and class II/III obesity had similar anti-HI titers, MFI, PR, and RR to each of the vaccine strains. Adults aged <60 years had similar anti-HI titers, MFI, PR, and RR to the QIV strains to those aged ≥60 years.
CONCLUSIONS: Our results indicate that the split virion, inactivated QIV is immunogenic in adults with obesity regardless of their degree of obesity and age (ie, <60 and ≥60 years).
Keywords: Influenza Vaccines, Influenza, Human, Obesity, Vaccination, Antibodies, Viral, Immunogenicity, Vaccine, Seasons, seroconversion, young adult
Background
Influenza is a serious viral respiratory disease that impacts all countries worldwide. Each year there are an estimated 1 billion cases globally, including 3–5 million severe cases and 290 000–650 000 influenza-related deaths [1]. The impact of body weight on the immune response to influenza infection and to the influenza vaccine was recognized after the 2009 pandemic, when an excessive incidence of hospitalization and mortality rates were noted among patients with obesity [2,3]. Based on these observations, morbid obesity was recognized by the World Health Organization (WHO) as one of the priority health conditions requiring routine influenza vaccination [4,5]. However, a limited number of studies have examined the effects of obesity on the immune response to influenza vaccines, and the data remain inconclusive [6–9]. Humoral and cellular immune disturbances may be associated with the immunomodulatory properties of adipose tissue, although the precise mechanisms are not fully understood [10,11]. Unfortunately, despite the increasing prevalence of obesity, data on the efficacy and immunogenicity of the influenza vaccine in individuals with varying degrees of obesity are scarce and mainly concern the trivalent influenza vaccine (TIV). Sheridan et al showed that while the humoral response to the TIV may not initially be impaired in subjects with obesity, there is a greater decline in titers to influenza antigens after 12 months [6,12]. In addition, African Americans had slightly higher antibody responses to the TIV than did White people in Sheridan’s study, indicating ethnic-based differences may exist [6]. Meanwhile, Talbot et al studied people aged ≥50 years and found no consistent relationship between obesity and either seroprotection or seroconversion to all 3 antigens included in the TIV [9]. Finally, Callahan et al showed that a single dose of monovalent vaccine to the pandemic A/H1N1 strain induced higher hemagglutination inhibition (anti-HI) titers at day 21 in adults with obesity compared to non-obese adults, but no significant differences in responses to the A/H1N1 strain were observed in adults of various BMI following 2 vaccine doses [13].
The seasonal influenza vaccine requires annual reformulation due to rapid changes in the hemagglutinin (HA) viral surface protein, which result in new antigenically distinct virus strains. TIVs contain 2 strains of influenza A (subtypes A/H1N1 and A/H3N2) and a single type B influenza strain. However, in the 1980s, 2 antigenically different influenza B lineages capable of infecting humans appeared globally (Victoria and Yamagata). Consequently, there was a mismatch between circulating B strains and those included in the vaccine, which reduced the efficacy of TIVs [14]. Therefore, quadrivalent influenza vaccines (QIV) were formulated for better influenza prevention and control by adding a second B virus lineage (Yamagata lineage) to the already-licensed TIVs. WHO first recommended a QIV for use in the Northern Hemisphere in the 2012/2013 season [15], and a QIV became available for the first time in Poland in the 2017/2018 season.
This study aimed to assess the immunogenicity of a split virion, inactivated QIV (available for the first time on the Polish pharmaceutical market in Autumn 2017), in a population of Polish adults with obesity in the 2017/2018 flu season.
Material and Methods
STUDY POPULATION:
Fifty-three adult patients of the Polyclinic for Metabolic Diseases of the Food and Nutrition Institute in Warsaw, Poland, were recruited for the administration of the split virion, inactivated QIV (Vaxigrip Tetra®, Sanofi Pasteur, Lyon, France) from October 2017 to January 2018. Eligible subjects were aged 18–70 years, had a body mass index (BMI) of ≥30.0 kg/m2 or 25.0–29.9 kg/m2, a waist circumference of >80 cm in women and >94 cm in men, and were willing to provide informed consent. Exclusion criteria were exacerbation of chronic disease, acute infectious disease, known allergy to the vaccine component, any history of anaphylaxis after vaccination against influenza or any serious reaction after vaccination, history of Guillain-Barré syndrome up to 6 weeks after previous vaccination, or received immunosuppressive therapy.
Data on age, sex, race, and comorbidities were collected at study entry. At inclusion, patients were also subjected to anthropometric and body composition measurements.
ANTHROPOMETRIC AND BODY COMPOSITION MEASUREMENTS:
The following data were collected from the patients: height, weight (measured using a medical scale Radwag WPT 200 OW, Radom, Poland), waist circumferences (measured halfway between the last rib and the anterior superior iliac spine using a centimeter tape), non-dominant arm circumference (measured halfway between the shoulder joint and the elbow joint using a centimeter tape), skinfold thickness above the tricep muscle of the non-dominant arm (ie, at the vaccine injection site; measured halfway between the shoulder joint and the elbow joint using Harpenden Calipers, Burgess Hill, United Kingdom) [16], and body composition (measured using Bioelectrical Impedance Analyzer BioScan 920-II-S, Maltron, Rayleigh, United Kingdom, according to the manufacturer’s instructions). BMI was calculated as body weight in kilograms divided by the height in meters squared (kg/m2). Following the recommendations of the WHO and the American Association of Clinical Endocrinologist/American College of Endocrinology (AACE/ACE), BMI, waist circumferences, and obesity-related comorbidities were used to classify obesity [17,18]. The measurements of body fat percentage, abdominal obesity or total body water were carried out in accordance with the recommendations of the WHO, the International Diabetes Federation (IDF), and other published guidelines, provided with a software of Bioelectrical Impedance Analyzer BioScan 920-II-S, Maltron, Rayleigh, United Kingdom [18–20].
VACCINE AND VACCINATION:
Subunit, inactivated QIV (Vaxigrip Tetra®, Sanofi Pasteur, Lyon, France) contained the antigen composition recommended by the WHO for the Northern Hemisphere for the 2017/2018 season [21] as follows: 15 μg hemagglutinin (HA) of A/Michigan/45/2015 (A/H1N1/pdm09)-like strain (A/Michigan/45/2015, NYMC X-275), 15 μg HA of A/Hong Kong/4801/2014 (A/H3N2/)-like strain (A/Hong Kong/4801/2014, NYMC X-2638), 15 μg HA of B/Brisbane/60/2008-like strain (Victoria lineage) (B/Brisbane/60/2008 wild strain), and 15 μg HA of B/Phuket/ 3073/2013-like strain (Yamagata lineage) (B/Phuket/3073/2013 wild strain). The vaccine was provided in prefilled syringes with a 15-mm-long needle and given as a 0.5-ml dose into the deltoid muscle or deeply subcutaneously into the non-dominant arm. The cold chain was preserved, and the vaccines were stored at 2–8°C.
SEROLOGICAL TESTING:
Blood samples of 5 mL of venous peripheral blood were drawn for analyses of serum anti-hemagglutinins antibodies just before vaccination and 4–8 weeks after vaccination. Samples (3 mL) were transported at 2–4°C immediately after collection from the laboratory of the Polyclinic for Metabolic Diseases of the Food and Nutrition Institute in Warsaw, Poland, to the laboratory at the Department of Influenza Research (National Influenza Center) at the National Institute of Public Health, Warsaw, Poland. Hemagglutination inhibition (HI) assay is traditionally used to measure influenza-specific titers as a correlate of protection and it quantifies the ability of HA to block the N-acetyl-neuraminic acid-mediated viral agglutination of red blood cells [22].
ASSESSMENT OF INFLUENZA VACCINE IMMUNOGENICITY:
Immunogenicity of the vaccine was assessed using criteria established by the Committee for Proprietary Medicinal Products (CPMP) and the European Agency for the Evaluation of Medicinal Products (EMEA, now the European Medicine Agency, EMA) [23]. The HI assay was carried out in accordance with WHO recommendations [24] using turkey erythrocytes at the Department of Influenza Research (National Influenza Center) at the National Institute of Public Health, Warsaw, Poland. The anti-HI titers were expressed as the reciprocal of the highest serum dilution at which hemagglutination of erythrocytes was completely inhibited. Antibody response to virus HAs was presented as geometric mean titers (GMT) of HI antibodies, calculated at baseline (day 0) and 28–36 days after vaccination. The evaluated parameters were: 1) Mean Fold Increase (MFI) as the post- to pre-vaccination GMT ratio; a ratio of >2.5 is required for subjects aged 18–60 years and >2.0 for those aged ≥60 years); 2) seroprotection rate (protection rate, PR) as the percentage of subjects with anti-HI titers of >1: 40; PR of ≥70% is required for subjects aged 18–60 years and ≥60% for people aged ≥60 years; 3) seroconversion rate (response rate, RR) as the percentage of subjects with a pre-vaccination anti-HI titer <10 and a post-vaccination anti-HI titer ≥40 or a ≥4-fold increase in the post-vaccination anti-HI titer if the pre-vaccination anti-HI titer was ≥10 for people aged 18–60 years and a ≥2-fold increase in post-vaccination anti-HI titer for people aged ≥60 years. The required RR is ≥40% for people aged 18–60 years or ≥30% for people aged ≥60 years [23,25]. The effect of the degree of obesity and age on vaccine immunogenicity was also investigated. Regarding obesity, 2 groups of individuals were studied: those with class I obesity and those with class II or III obesity. People with class II and III obesity were analyzed together due to the small number of patients with a high degree of obesity. Furthermore, the effect of age on vaccine immunogenicity was studied in subjects aged <60 years and those aged ≥60 years.
ETHICS APPROVAL:
The study was approved by the Ethics Committee of the Medical University in Warsaw, Poland (approval number: KB/165/2017). Written informed consent was obtained from all study participants.
STATISTICAL ANALYSES:
All calculations were done using the SPSS Statistics V25 software (IBM, Armonk, NY, USA). Data distribution was tested using skewness and kurtosis. For comparisons of anti-HI titers before and after vaccination, the paired Wilcoxon rank test was applied. Associations between immunogenicity indicators and body composition variables, skinfold thickness, and the number of non-communicable diseases were assessed using the Spearman’s rank correlation test. The Mann-Whitney U test was used to assess differences in the change in anti-HI titer depending on the route of vaccine administration. The effect of the route of vaccine administration on PR and RR was evaluated using the Pearson’s chi-squared test. An analysis of variance with Sidak correction was performed to investigate the effect of obesity degree, age, or both on vaccine immunogenicity parameters. A
Results
CHARACTERISTICS OF STUDY PARTICIPANTS:
The study involved 28 (52.8%) women and 25 (47.2%) men of White race. The mean age of the subjects was 54±11.19 years (range 21–69). Thirty-two (60.4%) subjects were aged <60 years, and 21 (39.6%) were aged ≥60 years. Data on the anthropometric dimensions of the subjects, their body composition, and the number of comorbidities are presented in Table 1.
According to BMI, 4 subjects were overweight (BMI <30 kg/m2), 17 had class I obesity (BMI 30–34.9 kg/m2), 24 had class II obesity (BMI 35–39.9 kg/m2), and 8 had class III obesity (BMI ≥40 kg/m2). Body composition analysis showed that 51 (96.2%) subjects had an elevated percentage of fat mass; the remaining 2 (3.8%) had an increased waist circumference and excessive abdominal fat. According to abdominal fat content, 12 (22.6%) subjects were overweight and 41 (77.4%) had abdominal obesity. The percentage of total body water was within the normal range in 45 (86.5%) subjects. Based on the malnutrition index, 39 (75%) subjects were overweight or obese. In terms of comorbidities, 49 (92.5%) subjects had at least 1 non-communicable disease. Subjects had 2 comorbidities on average (range 0–7) (Table 1), with hypertension, dyslipidemia, type 2 diabetes mellitus, and hypothyroidism most commonly reported. According to the AACE/ACE guidelines, obesity is diagnosed when the BMI exceeds 30 kg/m2 or when the BMI exceeds 25 kg/m2 and there is at least 1 mild-to-moderate obesity-related complication (class I obesity); or at least 1 severe obesity-related complication (class II obesity). As all overweight individuals in our study had comorbidities and they were classified as having class I or class II obesity.
QIV IMMUNOGENICITY:
The QIV elicited a significant increase in the level of antibodies against each of the 4 influenza HA contained in the vaccine (Table 2). An inadequate antibody response to the vaccine (post-vaccination HI titer equal to zero) was noted in 2 subjects for the A/Michigan/45/2015 (H1N1)pdm09-like strain, 3 subjects for the A/Hong Kong/4801/2014 (A/H3N2/)-like strain, 1 subject for the B/Brisbane/60/2008 strain, and 1 subject for the B/Phuket/3073/2013 strain. Parameters of vaccine immunogenicity are shown in Table 3.
Depending on the vaccine strain, pre- and post-vaccination GMT ranged from 1.98 to 34.82 and from 59.44 to 166.53, respectively. The pre- and post-vaccination GMT were lower for the A/H1N1/pdm09-like strain and A/H3N2 strain than for both B (Victoria and Yamagata lineages) strains. The MFI against the vaccine strains ranged from 3.7 (the B Victoria lineage) to 47.3 (the A/H3N2 strain) and comparable MFI values for the A/H1N1/pdm09-like and A/H3N2 strains were observed. Pre- and post-vaccination PR to the vaccine strains ranged from 3.8% to 83.0% and from 77.4% to 96.2%, respectively. Higher post-vaccination PRs were obtained for both B lineage strains compared to the A/H1N1/pdm09-like and A/H3N2 strains. The RRs to the vaccine strains ranged from 66% to 88.7%, with the lowest value recorded for the B Yamagata strain. All parameters (MFI, PR, RR) for all 4 vaccine strains reached the levels required for the respective age range of subjects. Thus, the QIV met the immunogenicity criteria established by CPMP/EMA for influenza vaccines in the study cohort.
EFFECT OF DEGREE OF OBESITY, AGE, AND BODY COMPOSITION ON QIV IMMUNOGENICITY:
The magnitude of change in HI titers following vaccination, GMT (pre- and post-vaccination), MFI, PR, and RR to each influenza virus strain contained in the vaccine did not differ significantly between individuals with class I obesity and those with class II/III obesity, or between individuals aged <60 years and those ≥60 years. However, if both factors were considered (ie, degree of obesity and age), a greater increase in anti-HI titers against the A/H3N2 strain was observed in younger subjects (aged <60 years) with class I obesity than in those with class II/III obesity (P<0.001) (Figure 1). The opposite trend was observed in subjects aged ≥60 years. Moreover, the RR to A/H3N2 strain at the analyzed level was achieved by all individuals with class I obesity aged <60 years compared to only 67% of individuals aged ≥60 years (P<0.05) (Figure 2). However, in both age groups, the RR met the criteria recommended by the CPMP/EMA.
Although some vaccine immunogenicity parameters were correlated with body composition variables, these associations were only moderate (Table 4). Larger fat mass and fat percentage were associated with a higher average PR (rs=0.325 and 0.347, respectively, P<0.05) and a greater change in the anti-HI titer against the A/H3N2 strain (rs=0.301 and 0.307, respectively, P<0.05). However, the percentage of total body water and the percentage of fat-free mass were correlated negatively with the average PR (rs=−0.347 and −0.335, respectively, P<0.05) and the magnitude of the change in the anti-HI titer against the A/H3N2 strain (rs=−0.307 and −0.290, respectively, P<0.05). In addition, the average pre-vaccination GMT correlated positively with the percentage of fat-free mass hydration (rs=0.337, P<0.05) and the excess fluid volume (rs=0.362, P<0.01).
Subcutaneous fat deposition affects the route of vaccine administration, as intramuscular injection using the 15-mm-long needle supplied by the manufacturer may not be possible if the skinfold thickness exceeds 15 mm. In our study, regardless of whether the vaccine was given intramuscularly (in subjects with skinfold <15 mm) or deeply subcutaneously (in subjects with skinfold >15 mm), no differences were observed in the anti-HI titer rise, PR, or RR against all 4 HA (Supplementary Tables 1–4). These results indicate that the thickness of the skinfold at the vaccine injection site does not significantly influence the immunogenicity of the QIV.
Discussion
Our study provides data on the immunogenicity of the split virion, inactivated QIV in an ethnically homogeneous population of Polish adults with obesity while it was first introduced in Poland in 2017/2018 season. The QIV fulfilled the CPMP/EMA criteria for acceptance of the immunogenicity of an influenza vaccine in the studied subjects with obesity. Depending on the type of influenza virus strain, protective levels of titers were achieved in 77.4–96.2% of subjects. The PR values for the A/H3N2, B Victoria lineage, and B Yamagata lineage strains were comparable with those reported by other researchers [26,27]. Although a lower PR value than that described in the literature was observed for the A/H1N1/pdm09 strain [26–30], it still met the CPMP/EMA criteria for vaccine immunogenicity. Prior to vaccination, the GMT ranged from 1.55 (for the A/H3N2 strain) to 34.82 (for the B Yamagata lineage strain) in studied subjects with obesity, which is lower than that observed in the general adult population [28,29,31–33]. This finding is in line with other researchers, who showed reduced levels of anti-HI titers before vaccination in individuals with excessive body weight compared to people with normal weight [6,8,13]. The reason for this may be a sharp decrease in anti-HI titers against vaccine strains after 12 months following influenza vaccination, which has been observed in people with obesity [6]. The post-vaccination GMT were also lower in studied subjects compared to the general adult population [26–28,32–34]. However, it cannot be excluded that differences in GMT levels described in the literature likely reflect, at least in part, the inter-laboratory discrepancies in titer measurements.
Despite these relatively lower pre- and post-vaccination GMT, we observed significant MFIs in studied adults with obesity. The MFI for the B Victoria lineage strain was above 14, and was above 40 for both the A/H1N1/pdm09 and A/H3N2 strains. A lower MFI (3.71) was only observed for the B Yamagata lineage strain compared to other vaccine HAs. This phenomenon can be associated with the higher pre-vaccination GMT observed for the B Yamagata lineage strain compared to those for the other 3 influenza strains [35], and was also confirmed in an elderly Polish population in a study by Ganczak et al carried out 1 season later (2018/2019) [36]. Interestingly, Bart et al observed that subjects who were seronegative at baseline demonstrated substantially higher RRs than those who were seropositive at baseline [35]. Furthermore, the PRs obtained in our study were comparable to those described by Ofori-Anyinam et al [37], but the RRs were higher than those previously reported [33]. Indeed, the relatively wide ranges for the MFI, PR, and RR (3.71–47.3%, 83–96.2%, and 66–88.7%, respectively) noted in this study have already been described by others and may be associated with the simultaneous exposure to 4 influenza HAs [29,31,38].
In our study, over 90% of participants had comorbidities. However, there was no correlation between the number of non-communicable diseases that accompanied obesity and the change in anti-HI titers following QIV vaccination (data not shown). These observations are consistent with the results of a meta-analysis indicating that obesity, regardless of comorbidities, is associated with influenza-related complications [39]. Moreover, we noted that the immunogenicity of the QIV in studied adults was at least as high as in other groups with different non-communicable conditions in Poland (ie, patients with acute lymphoblastic leukemia or non-Hodgkin’s lymphoma, women with breast cancer, patients with Wegener’s granulomatosis, splenectomized patients, patients on hemodialysis, or those who are immunosuppressed) [40–47].
Further analyses of the elicited anti-HI titers, GMT, MFI, PR, or RR did not show statistically significant differences in the vaccine immunogenicity to the A/H1N1/pdm09 and both B (Victoria and Yamagata lineages) strains among patients according to the degree of obesity, age, or both of these factors. Regarding the A/H3N2 strain, a greater increase in anti-HI titers was observed in younger subjects (aged <60 years) with class I obesity than those with class II/III obesity. The interaction effect of the degree of obesity and age was also significant in the case of the RR to the A/H3N2 strain, which at the analyzed level was observed more frequently in younger adults (<60 years) than in older adults (≥60 years) with class I obesity. Although the PR and RR for the A/H3N2 and B Yamagata lineage strains calculated in this study were similar to those previously demonstrated in individuals with obesity [9,13], our RRs for the A/H1N1/pdm09 and A/H3N2 strains were twice as high as those previously described for people with obesity by Neidich et al [8]. We observed higher values of MFI, PR, and RR for A/H1N1/pdm09, B Victoria lineage, and B Yamagata lineage strains and the similar values of MFI, PR, and RR for A/H3N2 strain for adults with obesity aged ≥60 years compared to data published by Ganczak et al from a study carried out on elderly Polish people 1 season later (2018/2019) [36]. The assessment of the QIV-induced antibody response in relation to the body composition of subjects indicated that fat mass and fat percentage were positively correlated with the magnitude of the HI antibody response to the A/H3N2 strain and the average PR, while the fat-free mass was negatively correlated. However, these correlations were moderate, and their clinical implications are uncertain.
The route of vaccine administration can also influence its immunogenicity [48]. However, our results indicate that the skinfold thickness at the injection site, which determines whether the vaccine is administered intramuscularly or subcutaneously, does not significantly affect the antibody response to all virus strains contained in the QIV. Hence, a practical aspect of our work may be the indication that it is not necessary to use longer needles for vaccinating people with obesity, as considered by others [48,49].
According to WHO, CDC, and Polish guidelines for influenza vaccination, it is recommended to use standard-dose inactivated (TIV or QIV) or live attenuated influenza vaccines (LAIVs) [5,50,51]. LAIVs are recommended for naïve populations like children, as they are able to induce a more protective and longer-lasting immune response [5,52]. TIV or QIV are recommended for the general population, especially for pregnant women, children aged 6 months to 2 years, people with chronic diseases, or adults with certain comorbidities such as obesity, diabetes, or liver disease [5,50,51]. Our results indicate the immunogenicity of the standard-dose QIV in adults with obesity fulfilled the CPMP/EMA criteria, which might result in the observed effectiveness of traditional seasonal influenza vaccines [52]. The observed lower increase in anti-HI titers or RR to the A/H3N2 strain of standard-dose QIV in older adults (≥60 years) with obesity suggests use of high-dose influenza vaccines in this age group of patients with obesity, which was described in the ECDC Technical Report as being more effective in prevention of influenza-like illness, respiratory illness, and respiratory-related or pneumonia-related hospital admissions [52].
The limitation of our study is the relatively small number of participants, but groups of similar numbers of subjects have already been investigated in studies assessing the humoral response to an influenza vaccine [7,45,46].
Conclusions
In conclusion, this is one of the first reports on the immunogenicity of the QIV in a Polish population with obesity. Our results indicate that the split virion, inactivated QIV is immunogenic in adults with obesity regardless of their degree of obesity and age (ie, <60 and ≥60 years). The effectiveness of the QIV measured as an epidemiological effect in observational studies of vaccinated and unvaccinated adults with obesity remains an interesting issue for evaluation in future research.
Supplementary Data
SUPPLEMENTARY TABLES 1–4. THE EFFECT OF ROUTE OF ADMINISTRATION ON QIV IMMUNOGENICITY:
Subjects were grouped according to the thickness of skinfold at the injection site: with a skinfold <15 mm enabling intramuscular injection and with a skinfold >15 mm which determines subcutaneous injection. Vaccine immunogenicity by route of administration is presented (Supplementary Tables 1–4).
Figures
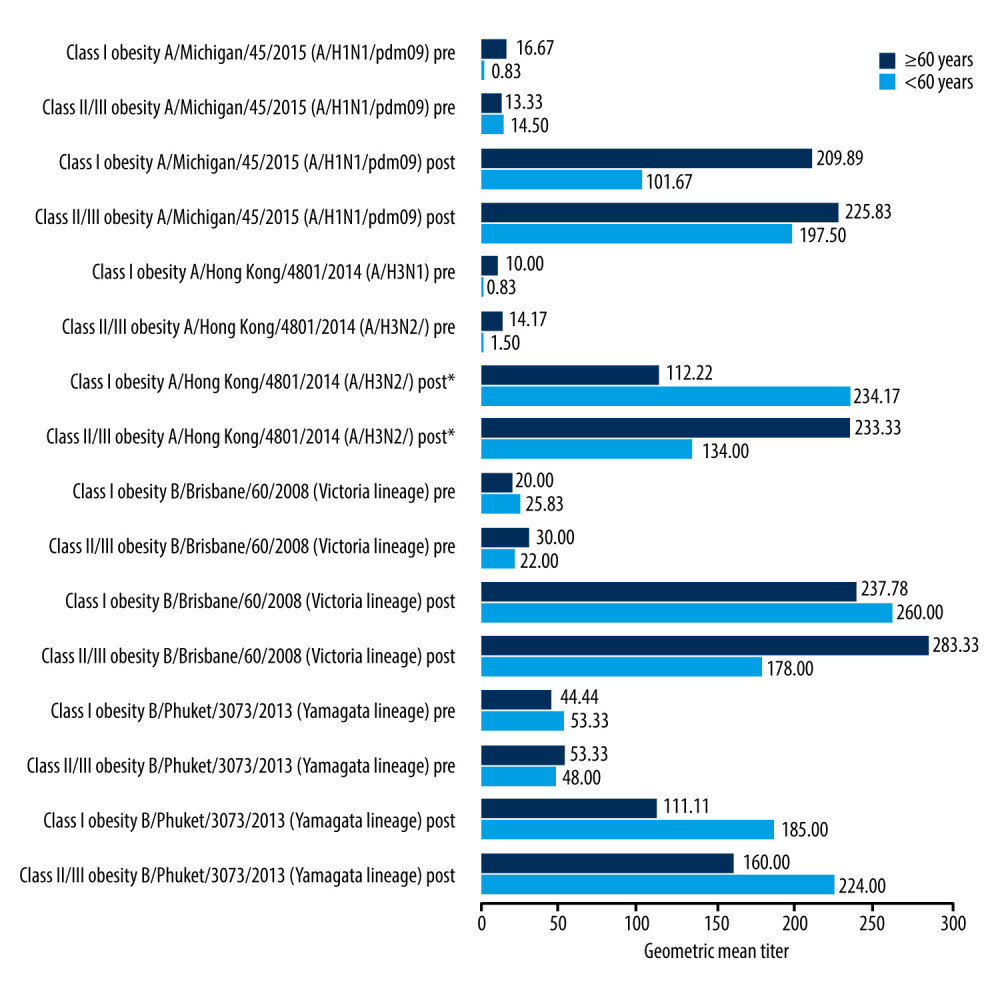 Figure 1. Pre-vaccination and post-vaccination geometric mean anti-hemagglutinins titers in subjects grouped by obesity class and age (N=53). Asterisks indicate groups that differ significantly (P<0.001). Pre – pre-vaccination; Post – post-vaccination; GMT – Geometric Mean antibody Titers.
Figure 1. Pre-vaccination and post-vaccination geometric mean anti-hemagglutinins titers in subjects grouped by obesity class and age (N=53). Asterisks indicate groups that differ significantly (P<0.001). Pre – pre-vaccination; Post – post-vaccination; GMT – Geometric Mean antibody Titers. 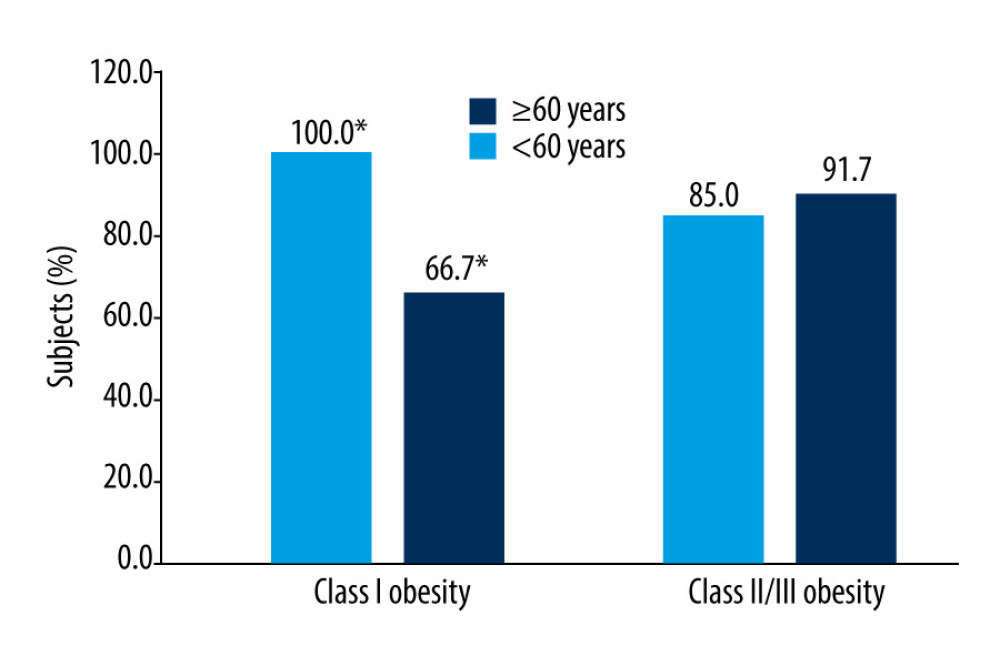 Figure 2. The effect of the degree of obesity and age on the response rate to the A/Hong Kong/4801/2014 (A/H3N2/)-like strain (N=53). Asterisks indicate groups that differ significantly (P<0.05).
Figure 2. The effect of the degree of obesity and age on the response rate to the A/Hong Kong/4801/2014 (A/H3N2/)-like strain (N=53). Asterisks indicate groups that differ significantly (P<0.05). Tables
Table 1. Data on height, weight, BMI, body composition of subjects and the number of comorbidities (N=53).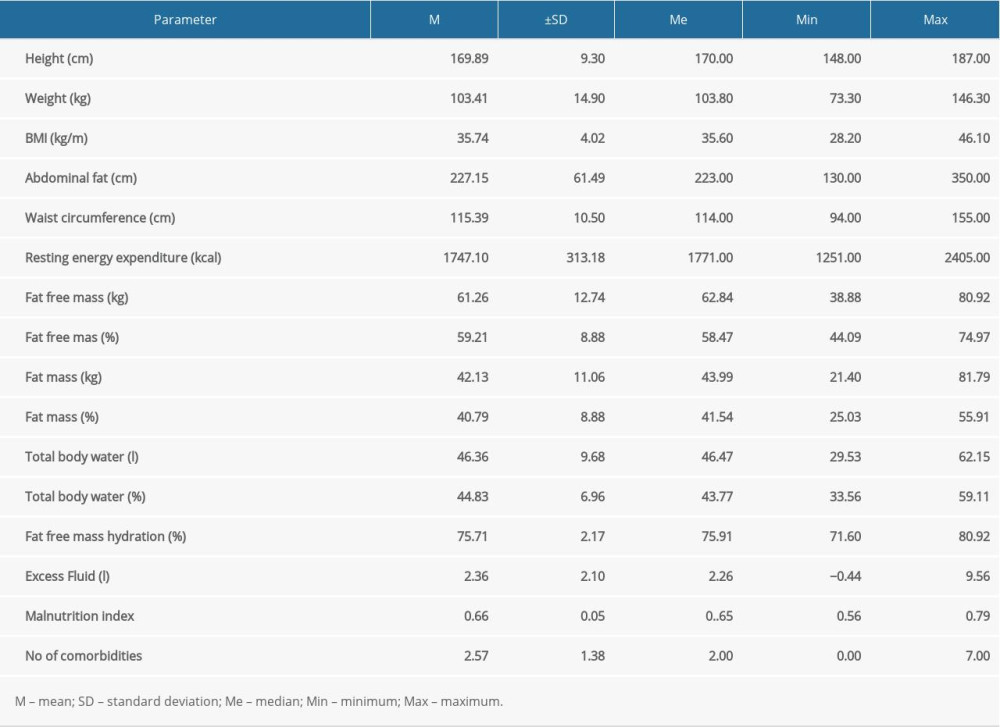 Table 2. Pre-vaccination and post-vaccination anti-HI titers against influenza virus strains (N=53).
Table 2. Pre-vaccination and post-vaccination anti-HI titers against influenza virus strains (N=53).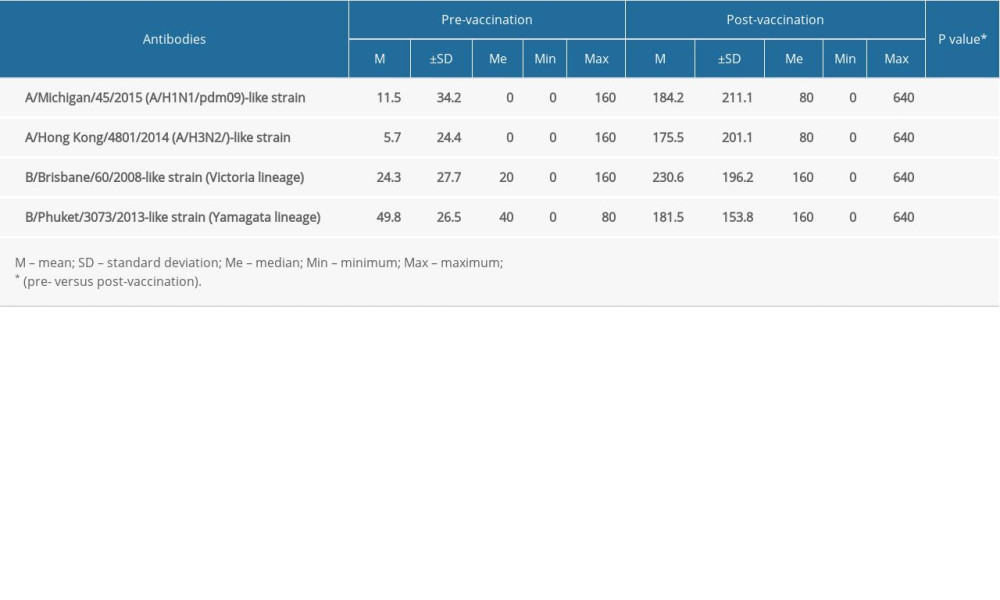 Table 3. Parameters of vaccine immunogenicity (N=53).
Table 3. Parameters of vaccine immunogenicity (N=53).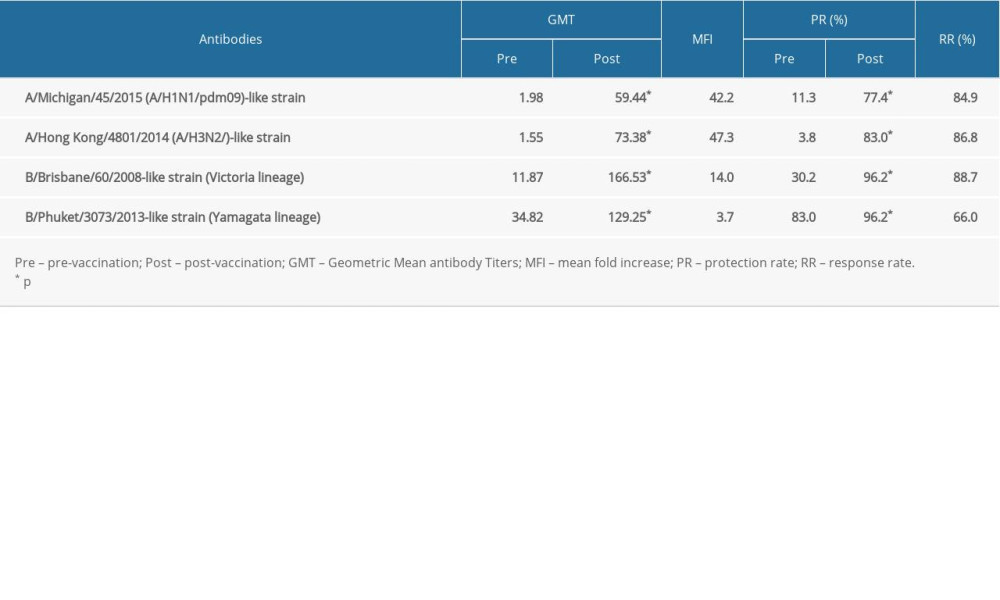 Table 4. Relations between anthropometric dimensions, BMI, and body composition variables and antibody response – Spearman’s correlation coefficient (N=53).
Table 4. Relations between anthropometric dimensions, BMI, and body composition variables and antibody response – Spearman’s correlation coefficient (N=53).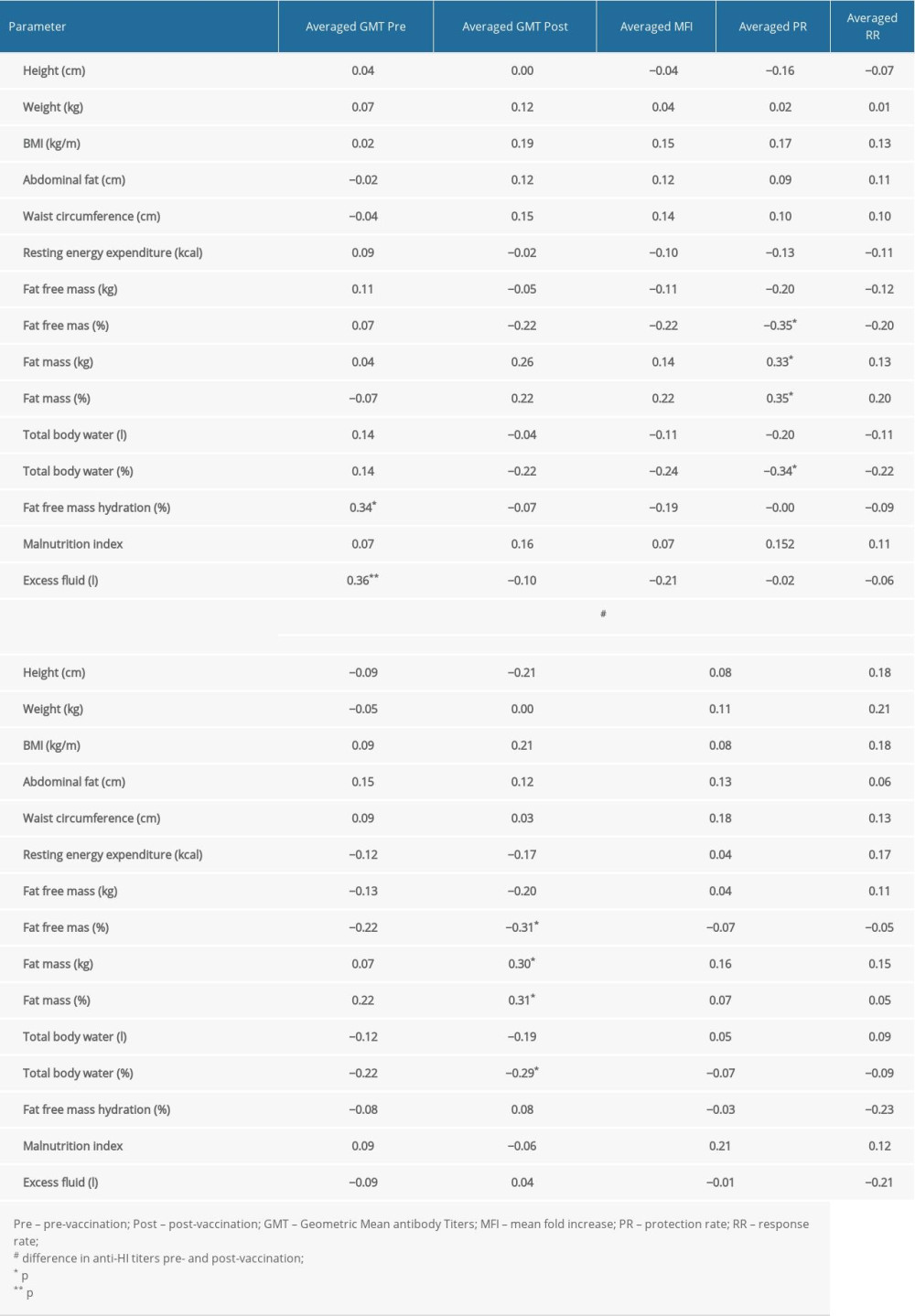
References
1. World Health Organization: Global Influenza Strategy 2019–2030 February 1, 2021 https://www.who.int/influenza/Global_Influenza_Strategy_2019_2030_Summary_English.pdf?ua=1
2. Van Kerkhove MD, Vandemaele KA, Shinde V, Risk factors for severe outcomes following 2009 influenza A (H1N1) infection: A global pooled analysis: PLoS Med, 2011; 8; e1001053
3. Louie JK, Acosta M, Samuel MC, A novel risk factor for a novel virus: Obesity and 2009 pandemic influenza A (H1N1): Clin Infect Dis, 2011; 52; 301-12
4. Fiore AE, Uyeki TM, Broder KCenters for Disease Control and Prevention (CDC), Prevention and control of influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2010: MMWR Recomm Rep, 2010; 6(RR-8); 1-62
5. : Seasonal influenza vaccines: An overview for decision-makers, 2020, Geneva, World Health Organization Licence: CC BY-NC-SA 3.0 IGO
6. Sheridan PA, Paich HA, Handy J, Obesity is associated with impaired immune response to influenza vaccination in humans: Int J Obes (Lond), 2012; 36; 1072-77
7. Frasca D, Ferracci F, Diaz A, Obesity decreases B cell responses in young and elderly individuals: Obesity (Silver Spring), 2016; 24; 615-25
8. Neidich SD, Green WD, Rebeles J, Increased risk of influenza among vaccinated adults who are obese: Int J Obes (Lond), 2017; 41; 1324-30
9. Talbot HK, Coleman LA, Crimin K, Association between obesity and vulnerability and serologic response to influenza vaccination in older adults: Vaccine, 2012; 30; 3937-43
10. Huttunen R, Syrjanen J, Obesity and the risk and outcome of infection: Int J Obes (Lond), 2013; 37; 333-40
11. Weisberg SP, McCann D, Desai M, Obesity is associated with macrophage accumulation in adipose tissue: J Clin Invest, 2003; 112; 1796-808
12. World Health Organization: Prevalence of obesity among adults March 1, 2019 https://www.who.int/gho/ncd/risk_factors/overweight_obesity/obesity_adults/en/
13. Callahan ST, Wolff M, Hill HR, Edwards KM, Impact of body mass index on immunogenicity of pandemic H1N1 vaccine in children and adults: J Infect Dis, 2014; 210; 1270-74
14. Tisa V, Barberis I, Faccio V, Quadrivalent influenza vaccine: A new opportunity to reduce the influenza burden: J Prev Med Hyg, 2016; 57(1); E28-33
15. World Health Organization: Recommended composition of influenza virus vaccines for use in the 2012–2013 northern hemisphere influenza season February 21, 2020 https://www.who.int/influenza/vaccines/virus/recommendations/201202_recommendation.pdf
16. Talbot LA, Lister Z, Assessing body composition: the skinfold method: AAOHN J, 1995; 43(12); 605-13
17. World Health Organization: Europe. BMI-body mass index October 1, 2020 http//www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi
18. Garvey WT, Mechanick JI, Brett EM, American Association of Clinical Endocrinologists and American College of Endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity: Endocr Pract, 2016; 22(Suppl 3); 1-203
19. Tatoń J, Czech A, Bernas M: Otyłość. Zespół metaboliczny. PZWL, wyd.1. Warszawa, 2006; 97-115 [in Polish]
20. Duren DL, Sherwood RJ, Czerwinski SA, Body composition methods: Comparisons and interpretation: J Diabetes Sci Technol, 2008; 2; 1139-46
21. World Health Organization: Recommended composition of influenza virus vaccines for use in the 2017–2018 northern hemisphere influenza season January 15, 2019 https://www.who.int/influenza/vaccines/virus/recommendations/2017_18_north/en/
22. Fonville JM, Wilks SH, James SL, Antibody landscapes after influenza virus infection or vaccination: Science, 2014; 346; 996-1000
23. : European Medicines Agency Harmonisation of requirements for influenza vaccines (Adopted guideline) March 12, 97 https://www.ema.europa.eu/en/harmonisation-requirements-influenza-vaccines
24. World Health Organization Global Influenza Surveillance Network, Identification of the haemagglutinin subtype of viral isolates by haemagglutination inhibition testing: Manual for the laboratory diagnosis and virological surveillance of influenza, 2011; 43-62, WHO
25. Beyer WE, Palache AM, Lüchters G, Seroprotection rate, mean fold increase, seroconversion rate: Which parameter adequately expresses seroresponse to influenza vaccination?: Virus Res, 2004; 103; 125-32
26. Greenberg DP, Robertson CA, Noss MJ, Safety and immunogenicity of a quadrivalent inactivated influenza vaccine compared to licensed trivalent inactivated influenza vaccines in adults: Vaccine, 2013; 31; 770-76
27. Tinoco JC, Pavia-Ruz N, Cruz-Valdez A, Immunogenicity, reactogenicity, and safety of inactivated quadrivalent influenza vaccine candidate versus inactivated trivalent influenza vaccine in healthy adults aged ≥18 years: A phase III, randomized trial: Vaccine, 2014; 32; 1480-87
28. Kieninger D, Sheldon E, Lin WY, Immunogenicity, reactogenicity and safety of an inactivated quadrivalent influenza vaccine candidate versus inactivated trivalent influenza vaccine: A phase III, randomized trial in adults aged ≥18 years: BMC Infect Dis, 2013; 13; 343
29. Beran J, Peeters M, Dewe W, Immunogenicity and safety of quadrivalent versus trivalent inactivated influenza vaccine: A randomized, controlled trial in adults: BMC Infect Dis, 2013; 13; 224
30. Pepin S, Donazzolo Y, Jambrecina A, Safety and immunogenicity of a quadrivalent inactivated influenza vaccine in adults: Vaccine, 2013; 31; 5572-78
31. Levin MJ, Buchwald UK, Gardner J, Immunogenicity and safety of zoster vaccine live administered with quadrivalent influenza virus vaccine: Vaccine, 2018; 36; 179-85
32. Montalban C, Montellano MB, Santos J, Lavis N, Immunogenicity and safety of the 2015 Southern Hemisphere formulation of a split-virion inactivated quadrivalent vaccine: Hum Vaccin Immunother, 2018; 14; 593-95
33. Sesay S, Brzostek J, Meyer I, Safety, immunogenicity, and lot-to-lot consistency of a split-virion quadrivalent influenza vaccine in younger and older adults: A phase III randomized, double-blind clinical trial: Hum Vaccin Immunother, 2018; 14; 596-608
34. Zerbini CA, Ribeiro Dos Santos R, Jose Nunes M, Immunogenicity and safety of Southern Hemisphere inactivated quadrivalent influenza vaccine: A Phase III, open-label study of adults in Brazil: Braz J Infect Dis, 2017; 21; 63-70
35. Bart S, Cannon K, Herrington D, Mills R, Immunogenicity and safety of a cell culture-based quadrivalent influenza vaccine in adults: A Phase III, double-blind, multicenter, randomized, non-inferiority study: Hum Vaccin Immunother, 2016; 12; 2278-88
36. Ganczak M, Dubiel P, Drozd-Dąbrowska M, Quadrivalent influenza vaccine-induced antibody response and influencing determinants in patients ≥ 55 years of age in the 2018/2019 season: Int J Environ Res Public Health, 2019; 16(22); 4489
37. Ofori-Anyinam O, Leroux-Roels G, Drame M, Immunogenicity and safety of an inactivated quadrivalent influenza vaccine co-administered with a 23-valent pneumococcal polysaccharide vaccine versus separate administration, in adults ≥50years of age: Results from a phase III, randomized, non-inferiority trial: Vaccine, 2017; 35; 6321-28
38. Tsurudome Y, Kimachi K, Okada Y, Immunogenicity and safety of an inactivated quadrivalent influenza vaccine in healthy adults: A phase II, open-label, uncontrolled trial in Japan: Microbiol Immunol, 2015; 59; 597-604
39. Fezeu L, Julia C, Henegar A, Obesity is associated with higher risk of intensive care unit admission and death in influenza A (H1N1) patients: A systematic review and meta-analysis: Obes Rev, 2011; 12; 653-59
40. Brydak LB, Calbecka M, Immunogenicity of influenza vaccine in patients with hemato-oncological disorders: Leuk Lymphoma, 1999; 32; 369-74
41. Brydak LB, Guzy J, Starzyk J, Humoral immune response after vaccination against influenza in patients with breast cancer: Support Care Cancer, 2001; 9; 65-68
42. Wyzgal J, Brydak LB, Zygier D, Study on efficacy of influenza vaccination in renal allograft recipients: Transplant Proc, 2002; 34; 572-75
43. Brydak LB, Machala M, Laguna P, Rokicka-Milewska R, Antibody response to influenza vaccination in splenectomized patients in Poland: J Clin Immunol, 2004; 24; 225-36
44. Brydak LB, Skwarczynski T, Machaka M, Antibody response to influenza vaccination in healthy adults: Viral Immunol, 2004; 17; 609-15
45. Zycinska K, Romanowska M, Nowak I, Antibody response to inactivated subunit influenza vaccine in patients with Wegener–s granulomatosis: J Physiol Pharmacol, 2007; 58(Suppl 5); 819-28
46. Brydak LB, Machala M, Centkowski P, Humoral response to hemagglutinin components of influenza vaccine in patients with non-Hodgkin malignant lymphoma: Vaccine, 2006; 24; 6620-23
47. Mastalerz-Migas A, Steciwko A, Brydak LB, Immune response to influenza vaccine in hemodialysis patients with chronic renal failure: Adv Exp Med Biol, 2013; 756; 285-90
48. Boonnak K, Dhitavat J, Thantamnu N, Immune responses to intradermal and intramuscular inactivated influenza vaccine among older age group: Vaccine, 2017; 35; 7339-46
49. Gorse GJ, Falsey AR, Fling JA, Intradermally-administered influenza virus vaccine is safe and immunogenic in healthy adults 18–64 years of age: Vaccine, 2013; 31; 2358-65
50. Grohskopf LA, Alyanak E, Broder KR, Prevention and control of seasonal influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices-United States, 2020-21 influenza season: MMWR Recomm Rep, 2020; 69(RR-8); 1-24
51.
52. European Centre for Disease Prevention and Control: Systematic review of the efficacy, effectiveness and safety of newer and enhanced seasonal influenza vaccines for the prevention of laboratory confirmed influenza in individuals aged 18 years and over, 2020, Stockholm, ECDC
Figures
 Figure 1. Pre-vaccination and post-vaccination geometric mean anti-hemagglutinins titers in subjects grouped by obesity class and age (N=53). Asterisks indicate groups that differ significantly (P<0.001). Pre – pre-vaccination; Post – post-vaccination; GMT – Geometric Mean antibody Titers.
Figure 1. Pre-vaccination and post-vaccination geometric mean anti-hemagglutinins titers in subjects grouped by obesity class and age (N=53). Asterisks indicate groups that differ significantly (P<0.001). Pre – pre-vaccination; Post – post-vaccination; GMT – Geometric Mean antibody Titers. Figure 2. The effect of the degree of obesity and age on the response rate to the A/Hong Kong/4801/2014 (A/H3N2/)-like strain (N=53). Asterisks indicate groups that differ significantly (P<0.05).
Figure 2. The effect of the degree of obesity and age on the response rate to the A/Hong Kong/4801/2014 (A/H3N2/)-like strain (N=53). Asterisks indicate groups that differ significantly (P<0.05). Tables
 Table 1. Data on height, weight, BMI, body composition of subjects and the number of comorbidities (N=53).
Table 1. Data on height, weight, BMI, body composition of subjects and the number of comorbidities (N=53). Table 2. Pre-vaccination and post-vaccination anti-HI titers against influenza virus strains (N=53).
Table 2. Pre-vaccination and post-vaccination anti-HI titers against influenza virus strains (N=53). Table 3. Parameters of vaccine immunogenicity (N=53).
Table 3. Parameters of vaccine immunogenicity (N=53). Table 4. Relations between anthropometric dimensions, BMI, and body composition variables and antibody response – Spearman’s correlation coefficient (N=53).
Table 4. Relations between anthropometric dimensions, BMI, and body composition variables and antibody response – Spearman’s correlation coefficient (N=53). Table 1. Data on height, weight, BMI, body composition of subjects and the number of comorbidities (N=53).
Table 1. Data on height, weight, BMI, body composition of subjects and the number of comorbidities (N=53). Table 2. Pre-vaccination and post-vaccination anti-HI titers against influenza virus strains (N=53).
Table 2. Pre-vaccination and post-vaccination anti-HI titers against influenza virus strains (N=53). Table 3. Parameters of vaccine immunogenicity (N=53).
Table 3. Parameters of vaccine immunogenicity (N=53). Table 4. Relations between anthropometric dimensions, BMI, and body composition variables and antibody response – Spearman’s correlation coefficient (N=53).
Table 4. Relations between anthropometric dimensions, BMI, and body composition variables and antibody response – Spearman’s correlation coefficient (N=53). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








