21 August 2021: Clinical Research
Sevoflurane-Remifentanil Versus Propofol-Remifentanil Anesthesia During Noncardiac Surgery for Patients with Coronary Artery Disease – A Prospective Study Between 2016 and 2017 at a Single Center
Zhongliang Dai1ACDE, Miao Lin1BDE, Yali Li1BE, Wenli Gao1BDE, Ping Wang1BDE, Juan Lin1CE, Zhenzhen Wan2AE, Yuanxu Jiang1CE*DOI: 10.12659/MSM.929835
Med Sci Monit 2021; 27:e929835
Abstract
BACKGROUND: Volatile anesthesia possesses cardioprotective properties, and it is widely used in patients undergoing coronary artery bypass surgery, but no randomized controlled trials (RCTs) are available on the use of sevoflurane-remifentanil versus propofol-remifentanil anesthesia for patients with coronary artery disease (CAD) during noncardiac surgery. This study was designed to compare the 2 different types of general anesthesia in patients with CAD undergoing noncardiac surgery at a single center.
MATERIAL AND METHODS: Patients with CAD undergoing noncardiac surgery were enrolled in an RCT conducted between March 2016 and December 2017. The participants were randomized to receive either sevoflurane-remifentanil or propofol-remifentanil anesthesia. The primary endpoint was occurrence of in-hospital cardiovascular events. The secondary endpoints included delirium, postoperative nausea and vomiting (PONV), Intensive Care Unit (ICU) length of stay (LOS), in-hospital morbidity and mortality, and hospital LOS.
RESULTS: A total of 164 participants completed the study (sevoflurane: 81; propofol: 83). The occurrence of in-hospital cardiovascular events did not differ between the 2 groups (42.6% vs 39.4%, P=0.86). The occurrence of delirium did not differ between the 2 groups after the operation. PONV had a higher frequency after sevoflurane anesthesia at 48 h compared with propofol. In-hospital morbidity and mortality, ICU LOS, and hospital LOS were similar between the 2 groups (all P>0.05). At 30 days after surgery, no between-group differences in cardiac morbidity and mortality were observed.
CONCLUSIONS: In this study, anesthesia using sevoflurane-remifentanil did not provide additional postoperative cardioprotection in comparison with propofol-remifentanil in patients with CAD undergoing noncardiac surgery.
Keywords: acute coronary syndrome, Coronary Artery Disease, Propofol, Aged, 80 and over, Anesthesia, General, Clinical Decision-Making, Disease Management, Follow-Up Studies, Outcome Assessment, Health Care, Perioperative Care, remifentanil, Surgical Procedures, Operative
Background
Coronary artery disease (CAD) refers to the atherosclerotic narrowing of coronary arteries. It is often asymptomatic early in the course of the disease, but it may lead to stable or unstable angina, myocardial infarction, or both owing to progressive thickening of the wall of the coronary arteries or to plaque rupture [1-4]. The prevalence of CAD is 6.2% in people ≥20 years of age [5]. Based on the type of intervention, the patient’s clinical condition, and the skill of the surgeons, the global perioperative mortality due to CAD was 0.4%–2% in recent years [6]. Common risk factors for CAD include dyslipidemia, tobacco use, hypertension, family history of CAD, diabetes mellitus, and obesity [1–4]. The complications include acute coronary syndromes, ST-elevation myocardial infarction, acute heart failure, arrhythmias, and sudden death [1–4].
For patients with CAD, the occurrence of cardiovascular events, especially acute myocardial infarction (AMI) and arrhythmias, may increase during noncardiac surgery [7]. Anesthesia was first demonstrated to have an effect on myocardial protection from ischemia in 1974 [8]. Subsequently, many in vitro studies and in vivo animal experiments have indicated that halogenated volatile anesthetics have a protective effect on the ischemic myocardium [9–11]. In addition, clinical studies have addressed the effect of volatile anesthetics on postoperative cardiac protection [12–14]. The anesthetic propofol is associated with a decrease in arterial blood pressure compared with volatile anesthetics [15]. Propofol is generally acknowledged to increase the cardiac risk for patients with CAD during noncardiac surgery [16,17]. However, few randomized controlled trials (RCTs) have compared the effects of volatile halogenated anesthesia versus intravenous anesthesia for noncardiac surgery in patients with CAD.
Therefore, this study was conducted at a single center between 2016 and 2017 and was designed to compare sevoflurane and remifentanil with propofol and remifentanil for general anesthesia in patients with CAD who were undergoing noncardiac surgery. We hypothesized that the administration of sevoflurane would have cardioprotective effects, especially by reducing the postoperative occurrence of cardiovascular events, compared with propofol.
Material and Methods
STUDY DESIGN AND PARTICIPANTS:
This study was an RCT that enrolled patients with CAD scheduled to undergo noncardiac surgery at Shenzhen People’s Hospital. This study was approved by the Shenzhen People’s Hospital Ethics Committee, Guangdong, China (Ethics Committee No. LL-KT-2015304), on May 28, 2015. From March 2016 to December 2017, 200 consecutive patients were enrolled from the Department of Anesthesiology of Shenzhen People’s Hospital. All study participants signed an informed consent form. The study was registered in the Chinese Clinical Trial Registry (ChiCTR1900020502), and the analysis plan was uploaded on April 19, 2018, before any data extraction or analysis occurred.
The inclusion criteria were as follows: (1) patient had a known diagnosis of CAD, confirmed by prior AMI or coronary angiography, or the occurrence of representative angina pectoris, and at least 2 of the following risk factors: age >65 years, active smoking, hypertension, hyperlipidemia, and diabetes mellitus; and (2) patient was scheduled for major noncardiac surgery (vascular surgery of the abdominal aorta or the lower extremities, open surgery of the peritoneal or thoracic cavity, or major orthopedic surgery of the hip or spine). The exclusion criteria were (1) signs of acute cardiac failure; (2) unstable angina; (3) recent onset (<6 months) myocardial infarction; (4) percutaneous coronary intervention within 2 months; (5) emergency surgery; (6) combined or repeated procedures; or (7) participation in other research that might interfere with the current study endpoint.
RANDOMIZATION AND BLINDING:
The participants were randomly allocated to receive either sevoflurane-remifentanil or propofol-remifentanil anesthesia. A series of 200 unmarked, sealed, opaque envelopes were prepared by an independent statistician using a random number table. Randomization was performed by picking an envelope randomly at the time of operation by one of the investigators. Secrecy was maintained until the patient arrived in the operation room, after which the attending anesthesiologist opened the envelope. The data collector, patient (who was sedated when anesthesia was started), and data analyzer were blinded to the type of anesthetic used.
ANESTHESIA AND MONITORING:
On the day of their surgery, each patient came to the operation room and was provided with standard monitoring, including noninvasive arterial pressure (Datex Ohmeda, Helsinki, Finland), electrocardiogram (ECG), peripheral oxygen saturation, and bispectral index (BIS) (Aspect Medical Systems, Inc., Newton, MA, USA). Pre-oxygenation was provided using a suitable face mask, and an infusion of fentanyl 20 μg/kg was started. Three minutes later, the general anesthesia was induced using etomidate 0.2 mg/kg and cisatracurium 0.2 mg/kg intravenously. After tracheal intubation, mechanical ventilation was maintained with a tidal volume of 10 mL/kg and an end-tidal pCO2 kept at 35–45 mm Hg by adjusting the ventilatory frequency through a closed-circuit anesthesia machine (PhysioFlex, Dräger, Lubeck, Germany), with a fraction of inspiration O2 (FiO2) 50% of 2.0 L/min. In the sevoflurane group, anesthesia was maintained with sevoflurane 1.0–2.0% minimum alveolar concentration end-tidal and remifentanil 0.1–0.3 μg/kg/min. In the propofol group, anesthesia was maintained with continuous infusions of propofol 2–4 mg/kg/h and remifentanil 0.1–0.3 μg/kg/min. Sevoflurane or propofol was adjusted to keep the BIS between 40 and 60. The remifentanil infusion was continued to the end of the operation, and the dose was increased by 0.05 μg/kg/min if arterial pressure or heart rate suddenly increased by >20% or if spontaneous movements of the patient occurred. If the surgical procedure required muscle relaxation, additional 0.1 mg/kg boluses of cisatracurium were given. No other anesthetics were administered until the termination of operation. After surgery, all patients received patient-controlled intravenous analgesia for 2 days using fentanyl 5 μg/mL, tramadol 5 mg/mL, and tropisetron 50 μg/mL in 100 mL. The patient-controlled intravenous analgesia was programmed with a background infusion of 2 mL/h and a bolus of 2 mL in 15 min.
BIOCHEMISTRY:
Blood samples were drawn in sterile tubes with lithium heparin at the following times: before the induction of anesthesia, at the end of the operation, and at 24 and 72 h after the end of the surgery. All samples were sent to hospital laboratory for the analysis of troponin T (cTnT). Normal values for cTnT were considered to be in the range of 0–0.3 ng/mL.
DATA COLLECTION:
Age, sex, body mass index, New York Heart Association (NYHA) classification, degree and extent of CAD, American Society of Anesthesiologists (ASA) classification, and preoperative medication were recorded. Type of fluid, amount of fluid loss, time of operation, sevoflurane inhalation concentration, propofol infusion rate, total remifentanil infused perioperatively, and BIS values were recorded. Myocardial ischemia on ECG, the occurrence of AMI, clinically relevant arrhythmia (atrial fibrillation or bundle branch block), Intensive Care Unit (ICU) length of stay (LOS), hospital LOS, reoperation during hospital stay, and any complications were recorded. AMI was diagnosed according to cTnT >0.3 ng/mL postoperatively and at least 1 of the following: new ECG changes indicative of ischemia (ST and/or T changes and/or left bundle branch block), symptoms of ischemia, and development of pathological Q waves [18].
FOLLOW-UP:
At 30 days after surgery, a telephone interview was conducted with the patients participating in the study and their families. Data on mortality, AMI, arrhythmias, cardiac dysfunction, readmissions, and any complications were collected.
ENDPOINTS:
The primary endpoint was the occurrence of in-hospital cardiovascular events (including myocardial ischemia detected by ECG or cTnT, cardiac death, acute coronary syndromes, congestive heart failure, or arrhythmia requiring treatment) in an intent-to-treat analysis. The secondary endpoints included delirium, postoperative nausea and vomiting (PONV), ICU LOS, in-hospital morbidity and mortality, and hospital LOS.
STATISTICAL ANALYSIS:
Statistical analyses were performed using SPSS 13.0 for Windows (SPSS, Inc., Chicago, IL, USA). The rate of myocardial ischemia was used for sample size calculation. A 10% difference between groups was reported on the basis of other studies [14,17]. With α=5% and power of 0.8, such a difference would be detected with a sample size of n=160 (or 80 per group). The target was fixed at 200 patients to account for the loss of participants for any reason.
Continuous variables were expressed as means±standard deviations. Nonparametric continuous variables were expressed as medians (25th–75th percentiles). Categorical variables were expressed as numbers (percentage). Fisher’s exact test was used to compare dichotomous variables, and Mann-Whitney
Results
RECRUITMENT:
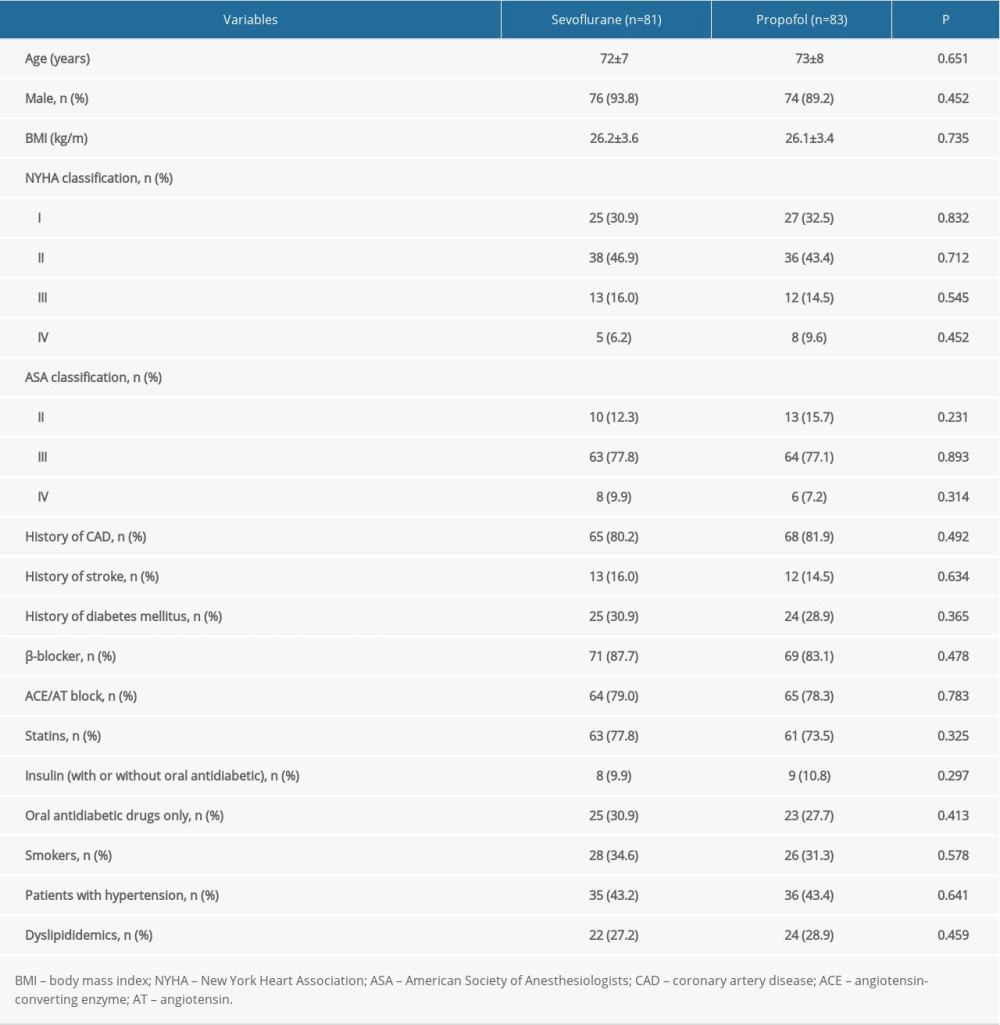
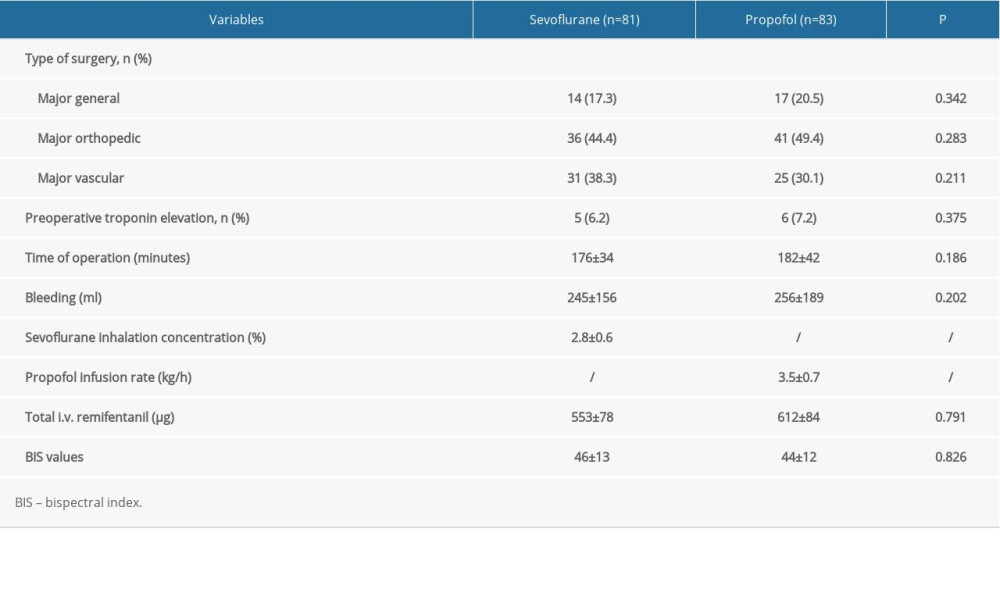
A total of 200 participants were screened for eligibility; 11 declined to participate and 25 were excluded. Eventually, 164 participants completed the protocol, with 81 in the sevoflurane group and 83 in the propofol group (Figure 1). The baseline characteristics of the participants are shown in Table 1. There were no significant differences in age, sex, body mass index, NYHA classification, ASA classification, comorbidities, medications, time of operation, and BIS values between the 2 groups.
IN-HOSPITAL CARDIOVASCULAR EVENTS:
There were no significant differences in the occurrence of cardiovascular events between the 2 groups (P=0.86) (Table 2). Based on traditional diagnostic criteria, myocardial ischemia (myocardial ischemia detected by ECG or troponin elevation) during the first 3 postoperative days was observed in 68 participants (sevoflurane: 36; propofol: 32). No participants experienced cardiac death during the postoperative period. Twenty-two participants had new acute coronary events (sevoflurane: 12; propofol: 10). Fifteen participants had episodes of new heart failure (sevoflurane: 7; propofol: 8). Arrhythmia requiring treatment was diagnosed in 37 participants (sevoflurane: 18; propofol: 19).
Preoperative cTnT levels were similar in both groups, and the postoperative levels of cTnT increased by 72 h after surgery in both groups. Five participants in the sevoflurane group and 6 in the propofol group had postoperative cTnT >0.3 ng/mL (P=0.68; Table 2).
SECONDARY ENDPOINTS:
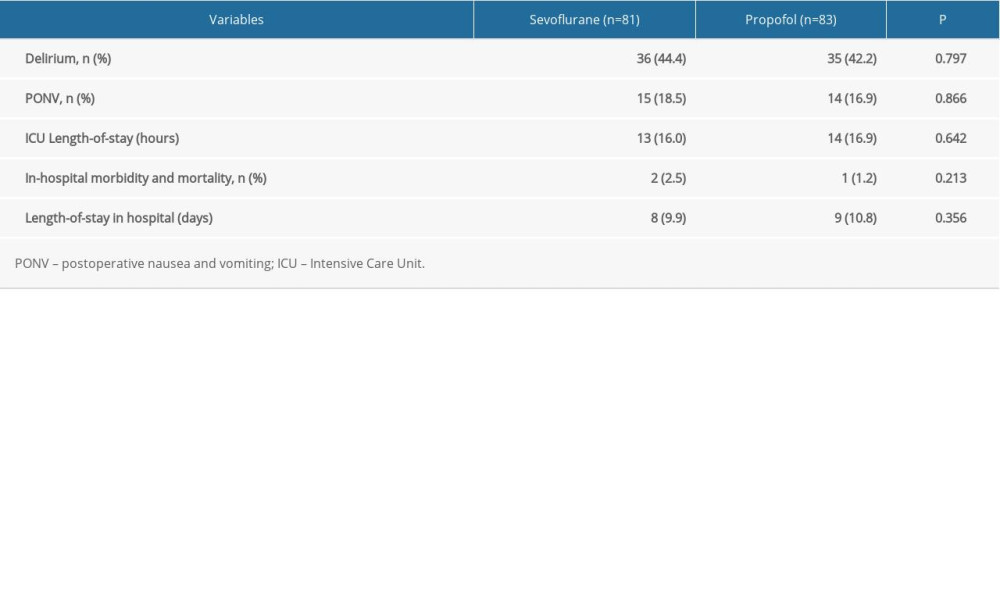
The occurrence of delirium did not differ between groups (Table 3). PONV at 48 h was more frequent after sevoflurane anesthesia than after propofol (Table 3). In-hospital morbidity and mortality, ICU LOS, and hospital LOS were similar between the 2 groups.
FOLLOW-UP AT 30 DAYS:
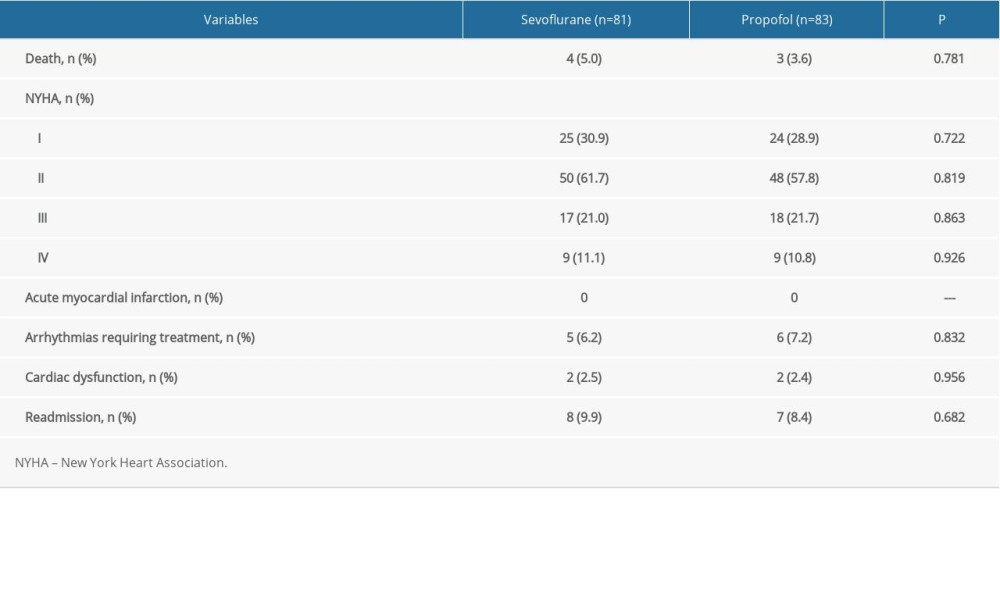
At 30 days, 7 patients had died of noncardiac causes. The NYHA classification improved compared with baseline in both groups (P 0.01), but did not differ between the 2 groups (P=0.56). No differences in cardiovascular events were observed (Table 4). No patients had AMI after surgery. Eleven patients had arrhythmias requiring treatment (5 in the sevoflurane group vs 6 in the propofol group; P=0.83). Cardiac dysfunction was diagnosed in 4 patients (2 in each group; P=0.95). Fifteen patients were readmitted because of wound infection and pneumonia (8 in the sevoflurane group vs 7 in the propofol group; P=0.68).
Discussion
The prevalence of patients with CAD is high [19,20]. For patients with CAD, the occurrence of intraoperative cardiovascular events, especially AMI and arrhythmias, may increase the operation risk [21]. Propofol can increase cardiac risk for patients with CAD during noncardiac surgery compared with volatile halogenated anesthetics [22]. Halogenated agents appear to possess cardioprotective properties, but the mechanism of action has been little studied [9–11]. These agents are widely used in patients undergoing coronary artery bypass operation, but no RCTs are available on the use of sevoflurane-remifentanil versus propofol-remifentanil anesthesia for patients with CAD during noncardiac surgery. Ours results suggest that anesthesia using sevoflurane-remifentanil does not provide additional postoperative cardioprotection in comparison with propofol-remifentanil in patients with CAD undergoing noncardiac surgery.
Propofol and sevoflurane are associated with different outcomes in different patients. Compared with patients receiving sevoflurane, patients who receive propofol display better intestinal motility after surgery [23] and less delayed neurocognitive recovery [24] but have lower renal function [25] and lower brain oxygenation [26]. The observed lack of cardiovascular events is inconsistent with previous experimental and clinical studies. Some trials demonstrated that sevoflurane anesthesia was associated with more myocardial protection than propofol anesthesia [12,27,28]. Yang et al [27] compared the myocardial protective effects of sevoflurane and propofol in patients undergoing cardiac valve replacement operation with cardiopulmonary bypass. They observed that sevoflurane anesthesia had a stronger protective effect on the myocardium than propofol and that the inflammatory response was weaker, thereby shortening ICU and hospital LOS. Jovic et al [28] studied 22 patients randomized into 2 groups (sevoflurane or propofol) and observed that sevoflurane regulated cytochrome c oxidase and ATP synthase, while propofol regulated cytochrome c, Cx43, mtDNA transcription, and UCP2. Myocardial oxidative stress plays a crucial role in the pathogenesis of ischemia-reperfusion injury associated with coronary heart disease. Ballester et al [12] studied patients undergoing off-pump coronary artery bypass grafting and showed that sevoflurane had better antioxidant performance than propofol. Guerrero Orriach et al [25] showed in 90 patients who underwent off-pump myocardial revascularization surgery that sevoflurane achieved better cardioprotective effects than propofol. Overall, these data suggest that sevoflurane anesthesia produces more clinically relevant myocardial protection than propofol anesthesia in patients with coronary heart disease. Therefore, the American College of Cardiology/American Heart Association guidelines recommend that volatile anesthetics should be used to maintain general anesthesia during noncardiac surgery in patients with a risk of perioperative myocardial ischemia [29].
Until now, only 3 RCTs have compared the myocardial protection of volatile anesthesia vs venous anesthesia throughout noncardiac surgery in patients with CAD [30–32]. These trials demonstrated that compared with propofol, sevoflurane did not reduce the incidence of myocardial ischemia in high-risk patients undergoing major noncardiac surgery. In the RCT by Zangrillo et al [32], the proportion of patients with an increase in postoperative troponin I in the sevoflurane group (12/44, 27.3%) was similar to that in the propofol group (9/44, 20.5%;
The present study supports the conclusions of the above trials. In comparison with propofol-remifentanil anesthesia, sevoflurane-remifentanil did not provide additional postoperative cardioprotection. Nevertheless, there were some differences between the trials. First, we studied sevoflurane combined with remifentanil, rather than sevoflurane anesthesia alone, reducing the interference of opioids. Second, the primary endpoint was the occurrence of in-hospital cardiovascular events, which can better reflect the protective effect to the myocardium.
The results of the present study suggest some myocardial protective effects of propofol-remifentanil anesthesia in patients with coronary heart disease. The NYHA classification at 30 days was better compared with the preoperative classification. There is experimental evidence on the cardioprotective effect of propofol on isolated animal hearts [33,34]. The mechanism includes antioxidant action, which reduces lipid peroxidation; sodium ion-hydrogen ion exchange channel inhibition; and anti-muscle calcium current inhibition to reduce calcium overload in the heart [33,35,36]. Some clinical studies have also indicated that a therapeutic dose of propofol reduces the lipid peroxide formation in ischemia-reperfusion injury caused by the surrounding tourniquet. Xia et al [37] found lower postoperative cTnT values for higher-dose propofol in cardiac ischemia-reperfusion injury. Therefore, although the mechanisms remain unclear, propofol may also have cardioprotective effects. In the future, we should clarify the mechanism of this cardioprotection.
One of the possible limitations of this study is the problem of “baseline” anesthesia and the depth of anesthesia. We cannot prove that all patients achieved the same depth of anesthesia because there are currently no accepted methods to precisely determine the depth of anesthesia. BIS has not been validated for the depth of anesthesia and its use with the halogenated volatile anesthetics has been questioned [38]. A second limitation of our trial is its small sample size. We used the ratio of myocardial ischemia for sample size calculations and then used myocardial ischemia as the primary target criterion. For the incidence of myocardial ischemia, according to other studies, the difference between the 2 groups was 10%. However, 19 patients in the sevoflurane group and 17 patients in the propofol group were excluded from analysis. This reduced the statistical power of the study. The third limitation is the comparability of heart risks. Even though all patients met the inclusion criteria for typical angina, along with more than 1 risk factor, this included multiple different cardiovascular statuses. On the basis of Lee’s revised Cardiac Risk Index, most of the patients in both groups in this study showed moderate risk (Class III), so we considered both groups to be comparable in this regard. However, each patient may actually have had a different cardiac risk. The third limitation is that this study ended in 2017. The latest clinical guidelines propose that propofol can cause dose-dependent vasodilatation with a decrease in systemic blood pressure levels, but no conclusive evidence supports the use of one type of anesthetic over another in preventing adverse cardiovascular effects. Accordingly, guidelines recommend that the choice of anesthetic agent should be based on factors other than prevention of myocardial ischemia [6].
Conclusions
In conclusion, sevoflurane and remifentanil did not provide additional postoperative cardioprotection in comparison with propofol and remifentanil for patients with known CAD who required general anesthesia for noncardiac surgery at a single center between 2016 and 2017. This study had a small population, however, and to prove that there was indeed no difference between the 2 groups, a larger number of patients need to be studied.
Figures
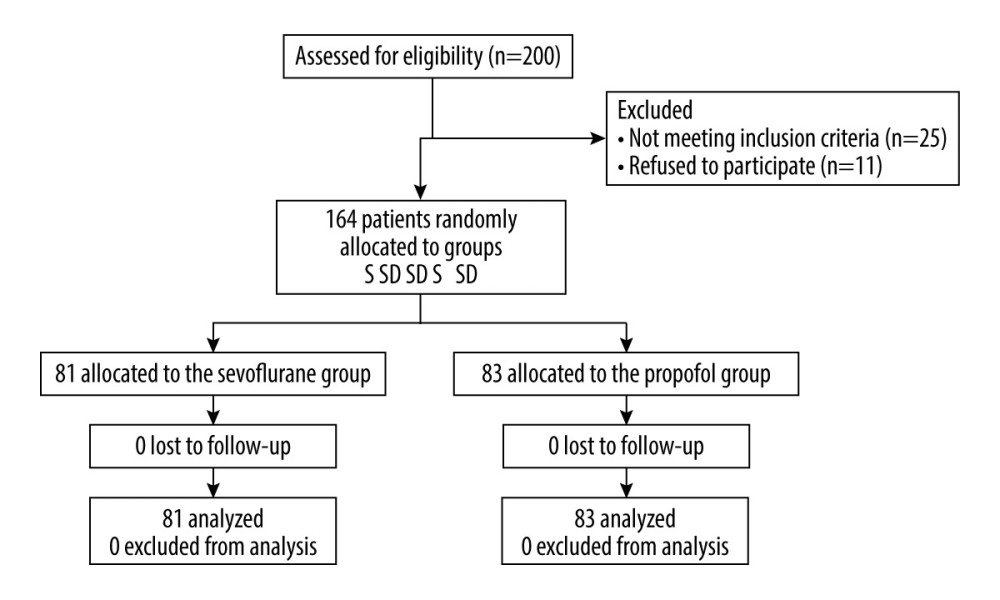 Figure 1. Study flow chart.
Figure 1. Study flow chart. References
1. Lawton JS, Sex and gender differences in coronary artery disease: Semin Thorac Cardiovasc Surg, 2011; 23; 126-30
2. Fihn SD, Blankenship JC, Alexander KP, 2014 ACC/AHA/AATS/PCNA/SCAI/STS focused update of the guideline for the diagnosis and management of patients with stable ischemic heart disease: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines, and the American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons: Circulation, 2014; 130; 1749-67
3. Fihn SD, Gardin JM, Abrams J, 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the diagnosis and management of patients with stable ischemic heart disease:A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons: J Am Coll Cardiol, 2012; 60; e44-164
4. Poon M, Lesser JR, Biga C, Current evidence and recommendations for coronary CTA first in evaluation of stable coronary artery disease: J Am Coll Cardiol, 2020; 76; 1358-62
5. Mozaffarian D, Benjamin EJ, Go AS, Heart disease and stroke statistics – 2015 update:A report from the American Heart Association: Circulation, 2015; 131; e29-322
6. Cao D, Chandiramani R, Capodanno D, Non-cardiac surgery in patients with coronary artery disease:Risk evaluation and periprocedural management: Nat Rev Cardiol, 2021; 18; 37-57
7. Smilowitz NR, Gupta N, Guo Y, Trends in cardiovascular risk factor and disease prevalence in patients undergoing non-cardiac surgery: Heart, 2018; 104; 1180-86
8. Flier S, Post J, Concepcion AN, Influence of propofol-opioid vs isoflurane-opioid anaesthesia on postoperative troponin release in patients undergoing coronary artery bypass grafting: Br J Anaesth, 2010; 105; 122-30
9. Warltier DC, Classic papers revisited:an early study of cardioprotection by volatile anesthetics:A behind-the-scenes look: Anesthesiology, 2018; 129; 576-79
10. Tsutsumi YM, Tsutsumi R, Horikawa YT, Geranylgeranylacetone and volatile anesthetic-induced cardiac protection synergism is dependent on caveolae and caveolin-3: J Anesth, 2014; 28; 733-39
11. Ramirez-Aliaga M, Escalona-Belmonte JJ, Mechanisms of cardioprotection of halogenated agents during extracorporeal circulation in cardiac surgery: Curr Vasc Pharmacol, 2018; 16; 319-28
12. Ballester M, Llorens J, Garcia-de-la-Asuncion J, Myocardial oxidative stress protection by sevoflurane vs propofol:A randomised controlled study in patients undergoing off-pump coronary artery bypass graft surgery: Eur J Anaesthesiol, 2011; 28; 874-81
13. Mellidis K, Ordodi V, Galatou E, Activation of prosurvival signaling pathways during the memory phase of volatile anesthetic preconditioning in human myocardium:A pilot study: Mol Cell Biochem, 2014; 388; 195-201
14. Yang B, Fung A, Pac-Soo C, Ma D, Vascular surgery-related organ injury and protective strategies: Update and future prospects: Br J Anaesth, 2016; 117(Suppl 2); ii32-43
15. Van Rompaey N, Barvais L, Clinical application of the cardioprotective effects of volatile anaesthetics:CON – total intravenous anaesthesia or not total intravenous anaesthesia to anaesthetise a cardiac patient?: Eur J Anaesthesiol, 2011; 28; 623-27
16. Besch G, Liu N, Samain E, Occurrence of and risk factors for electroencephalogram burst suppression during propofol-remifentanil anaesthesia: Br J Anaesth, 2011; 107; 749-56
17. An R, Pang QY, Chen B, Effect of anesthesia methods on postoperative major adverse cardiac events and mortality after non-cardiac surgeries:A systematic review and meta-analysis: Minerva Anestesiol, 2017; 83(7); 749-61
18. Ibanez B, James S, Agewall S, 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation:The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC): Eur Heart J, 2018; 39; 119-77
19. Fishman SL, Sonmez H, Basman C, The role of advanced glycation end-products in the development of coronary artery disease in patients with and without diabetes mellitus:A review: Mol Med, 2018; 24; 59
20. Kim OY, Lee SM, An WS, Impact of blood or erythrocyte membrane fatty acids for disease risk prediction:Focusing on cardiovascular disease and chronic kidney disease: Nutrients, 2018; 10(10); 1454
21. Feng B, Lin J, Jin J, The effect of previous coronary artery revascularization on the adverse cardiac events ninety days after total joint arthroplasty: J Arthroplasty, 2018; 33; 235-40
22. Cromheecke S, ten Broecke PW, Hendrickx E, Incidence of atrial fibrillation early after cardiac surgery:Can choice of the anesthetic regimen influence the incidence?: Acta Anaesthesiol Belg, 2005; 56; 147-54
23. Desmet M, Vander Cruyssen P, Pottel H, The influence of propofol and sevoflurane on intestinal motility during laparoscopic surgery: Acta Anaesthesiol Scand, 2016; 60; 335-42
24. Zhang Y, Shan GJ, Zhang YX, Propofol compared with sevoflurane general anaesthesia is associated with decreased delayed neurocognitive recovery in older adults: Br J Anaesth, 2018; 121; 595-604
25. Guerrero Orriach JL, Galan Ortega M, Ramirez Fernandez A, Cardioprotective efficacy of sevoflurane vs propofol during induction and/or maintenance in patients undergoing coronary artery revascularization surgery without pump:A randomized trial: Int J Cardiol, 2017; 243; 73-80
26. Doe A, Kumagai M, Tamura Y, A comparative analysis of the effects of sevoflurane and propofol on cerebral oxygenation during steep Trendelenburg position and pneumoperitoneum for robotic-assisted laparoscopic prostatectomy: J Anesth, 2016; 30; 949-55
27. Yang XL, Wang D, Zhang GY, Guo XL, Comparison of the myocardial protective effect of sevoflurane versus propofol in patients undergoing heart valve replacement surgery with cardiopulmonary bypass: BMC Anesthesiol, 2017; 17; 37
28. Jovic M, Stancic A, Nenadic D, Mitochondrial molecular basis of sevoflurane and propofol cardioprotection in patients undergoing aortic valve replacement with cardiopulmonary bypass: Cell Physiol Biochem, 2012; 29; 131-42
29. American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery); American Society of Echocardiography; American Society of Nuclear Cardiology, et al, ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery:executive summary:A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery): Anesth Analg, 2008; 106; 685-712
30. Zhang Y, Lin W, Shen S, Randomized comparison of sevoflurane versus propofol-remifentanil on the cardioprotective effects in elderly patients with coronary heart disease: BMC Anesthesiol, 2017; 17; 104
31. Lurati Buse GA, Schumacher P, Seeberger E, Randomized comparison of sevoflurane versus propofol to reduce perioperative myocardial ischemia in patients undergoing noncardiac surgery: Circulation, 2012; 126; 2696-704
32. Zangrillo A, Testa V, Aldrovandi V, Volatile agents for cardiac protection in noncardiac surgery:A randomized controlled study: J Cardiothorac Vasc Anesth, 2011; 25; 902-7
33. Kamada N, Kanaya N, Hirata N, Cardioprotective effects of propofol in isolated ischemia-reperfused guinea pig hearts:Role of KATP channels and GSK-3beta: Can J Anaesth, 2008; 55; 595-605
34. King N, Al Shaama M, Suleiman MS, Propofol improves recovery of the isolated working hypertrophic heart from ischaemia-reperfusion: Pflugers Arch, 2012; 464; 513-22
35. Zhang Q, Cai S, Guo L, Zhao G, Propofol induces mitochondrial-associated protein LRPPRC and protects mitochondria against hypoxia in cardiac cells: PLoS One, 2020; 15; e0238857
36. Cho YJ, Nam K, Kim TK, Sevoflurane, propofol and carvedilol block myocardial protection by limb remote ischemic preconditioning: Int J Mol Sci, 2019; 20(2); 269
37. Xia Z, Huang Z, Ansley DM, Large-dose propofol during cardiopulmonary bypass decreases biochemical markers of myocardial injury in coronary surgery patients:A comparison with isoflurane: Anesth Analg, 2006; 103; 527-32
38. Whitlock EL, Villafranca AJ, Lin N, Relationship between bispectral index values and volatile anesthetic concentrations during the maintenance phase of anesthesia in the B-Unaware trial: Anesthesiology, 2011; 115; 1209-18
Figures
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387