08 February 2021: Clinical Research
Diagnosis of Prostate Cancer in Patients with Prostate-Specific Antigen (PSA) in the Gray Area: Construction of 2 Predictive Models
Ming Chen1BCE, Ting Ma1CDE, Jun Li1AEG*, Hai-Jun Zhang2D, Qiang Li3F, Jia-Jia Wang1F, Tian Sang1B, Chun-Li Cao1D, Xin-Wu Cui4AEFDOI: 10.12659/MSM.929913
Med Sci Monit 2021; 27:e929913
Abstract
BACKGROUND: Two diagnostic models of prostate cancer (PCa) and clinically significant prostate cancer (CS-PCa) were established using clinical data of among patients whose prostate-specific antigen (PSA) levels are in the gray area (4.0-10.0 ng/ml).
MATERIAL AND METHODS: Data from 181 patients whose PSA levels were in the gray area were retrospectively analyzed, and the following data were collected: age, digital rectal examination, total PSA, PSA density (PSAD), free/total PSA (f/t PSA), transrectal ultrasound, multiparametric magnetic resonance imaging (mpMRI), and pathological reports. Patients were diagnosed with benign prostatic hyperplasia (BPH) and PCa by pathology reports, and PCa patients were separated into non-clinically significant PCa (NCS-PCa) and CS-PCa by Gleason score. Afterward, predictor models constructed by above parameters were researched to diagnose PCa and CS-PCa, respectively.
RESULTS: According to the analysis of included clinical data, there were 109 patients with BPH, 44 patients with NCS-PCa, and 28 patients with CS-PCa. Regression analysis showed PCa was correlated with f/t PSA, PSAD, and mpMRI (P<0.01), and CS-PCa was correlated with PSAD and mpMRI (P<0.01). The area under the receiver operating characteristic curves of 2 models for PCa (sensitivity=73.64%, specificity=64.23%) and for CS-PCa (sensitivity=71.41%, specificity=81.82%) were 0.79 and 0.87, respectively.
CONCLUSIONS: The prediction models had satisfactory diagnostic value for PCa and CS-PCa among patients with PSA in the gray area, and use of these models may help reduce overdiagnosis.
Keywords: Logistic Models, Magnetic Resonance Imaging, Prostate-Specific Antigen, Prostatic Neoplasms, Age Factors, Biopsy, Diagnosis, Differential, digital rectal examination, Kallikreins, Medical Overuse, Models, Statistical, Multiparametric Magnetic Resonance Imaging, Neoplasm Grading, Prostate, Prostatic Hyperplasia, ROC Curve, Reference Values, Risk Assessment, Ultrasonography
Background
Prostatic cancer (PCa) is a common malignant tumor of the male urinary system, the morbidity of which ranks fifth in overall global malignancies [1]. In China, the incidence rate of PCa is lower than that in Western countries[2]. However, with the change of lifestyle and the prolongation of life expectancy, the incidence rate of PCa has been increasing rapidly [2]. Epidemiological surveys showed the incidence rate of PCa in 2014 was nearly 1.5 times higher than that in 2012 (6.10/10 000 vs 4.39/10 000), ranking sixth in male cancer mortality [3,4].
The major prognostic benefit of PCa is early diagnosis and hence treatment [5]. Prostate-specific antigen (PSA) as a serum marker is the usual diagnostic method for biopsy-driven PCa. In the European Association of Urology Guidelines, the higher levels of PSA indicate likelihood of PCa [6]. However, PSA levels between 4.0 ng/ml and 10.0 ng/ml are often called the “gray area”, within which it is controversial to diagnose PCa [7]. According to a survey, when PSA level is within this range, the positive rate of prostate biopsy was only 15.9% to 26.0%, so it is often necessary to combine it with other clinical parameters to improve the detection rate [8].
Traditionally, the digital rectal examination (DRE), which is based on the perceived diversities in the stiffness of normal prostatic tissue and neoplasm, is always used as a primary method for detecting PCa [5]. Total PSA (tPSA) is a commonly used diagnostic indicator in prostatic disease, whose derivates, including free PSA (fPSA), PSA density (PSAD), and free/total PSA (f/t PSA), have reference value for PCa with the PSA in the gray area. Huang et al reported the summary sensitivity of fPSA was 70% in patients with PSA in the gray area [9]. In addition, another 2 studies indicated the indication for prostate biopsy is that f/t PSA is ≤15% and PSAD is >0.15 ng/ml [10,11].
TRUS is a commonly used imaging method in clinical practice, and it is non-invasive and simple. Some scholars pointed out that the sensitivity and specificity of transrectal ultrasound (TRUS) measurements in diagnosing the PCa were 17% to 57% and 40% to 63%, respectively [12]. Multiparametric magnetic resonance imaging (mpMRI) is well-established as an important aide for diagnosis of PCa [13]. The diagnostic rate of mpMRI for PCa was 73%, which is helpful in the staging of PCa [14].
Therefore, the diagnosis of PCa with PSA in the gray area is not satisfactory using a single diagnostic mode. The present study aimed to improve the diagnostic rate of PCa with PSA in the gray area by analyzing the comprehensive clinical parameters, including age, DRE, tPSA, f/t PSA, PSAD, TRUS, mpMRI, and construct modes, based on the significant parameters.
Material and Methods
PATIENTS:
This retrospective research was approved by the Ethics Review Board of the First Affiliated Hospital of Medical College of Shihezi University (No. 2019-123-01), with waiver of patient consent. There were 1243 patients who underwent prostate biopsy from August 2013 to October 2019 at the First Affiliated Hospital of Medical College, Shihezi University. Inclusion criteria were: 1) patient accepted prostate biopsy and pathology reports could be consulted; and 2) patient had complete electronic patient records and clinical data, including age, DRE, tPSA, f/t PSA, PSAD, TRUS, and mpMRI. The exclusion criterion was patients with contraindications for prostate biopsy. Of these patients, 181 (11.73%) had PSA levels between 4.0 ng/ml and 10.0 ng/ml, and all patients had either benign prostatic hyperplasia (BPH) or PCa. The collected medical records were divided into a BPH group and a PCa group according to pathology results. The patients in the PCa group were divided into a clinically significant PCa (CS-PCa) group and a non-clinically significant PCa (NCS-PCa) group by Gleason score (GS) modified 2005 International Society of Urological Pathology (ISUP) [15]. The PCa patients with GS <7 were assigned to the NCS-PCa group, while other patients were assigned to the CS-PCa group.
EQUIPMENT AND METHODS:
Clinical parameters of all enrolled patients were collected, including age at diagnosis and treatment, DRE, serum tPSA, f/t PSA, PSAD, TRUS, and mpMRI in the medical records system. DRE was performed by a deputy senior urologist. The tPSA and f/t PSA was collected by immunofluorescence assay. From ultrasound prostate scanning reports, we collected data on prostatic volume (PV) using the exact prolate ellipsoid formula (PV=anteroposterior diameter×left-right diameter×vertical diameter×π/6), then PSAD was calculated by tPSA/PV [16].
All mpMRI of prostate examinations were performed with a Discovery MR 750 3.0-tesla (General Electric Medical Systems, Wisconsin, USA). The T1-weighted (T1WI), T2-weighted (T2WI), diffusion-weighted imaging (DWI), and dynamic contrast-enhanced (DCE) perfusion imaging were obtained, as described previously. The parameters of diffusion sequences were the following: (
The criterion standard for diagnosing enrolled patients was TRUS-guided 12-point systematic prostate biopsy, and the diagnosis of specimens was performed using GS. The pathology was diagnosed by an experienced pathologist, who was not aware of clinical information about these patients.
STATISTICAL ANALYSES:
SPSS version 21.0 (IBM, Armonk, NY, USA) and MedCalc version 15.2 (MedCalc Software, Ltd, Acacialaan 22, Ostend, Belgium) were used for statistical analysis. For normally distributed continuous variables, means and standard deviations are used in descriptive analysis, and the 2-sample
Results
CHARACTERISTICS OF CLINICAL PARAMETERS OF ENROLLED PATIENTS:
The characteristics of included patients are shown in Table 1. Comparing the BPH group with the PCa group, clinical parameters consisted of f/t PSA (0.21±0.12 vs 0.15±0.06; P<0.01), PSAD (0.14 ng/ml·cm3, IQR: 0.07, 0.19 vs 0.19 ng/ml·cm−3, IQR: 0.12, 0.25; P<0.01) and mpMRI (P<0.01) had statistically significant differences, while age (74.00 years, IQR: 66.00–77.00 vs 76.00 years, IQR: 69.00–79.00; P=0.07), tPSA (6.91±1.35 ng/ml vs 7.25±1.47 ng/ml; P=0.12), DRE (P=0.09), and TRUS (P=0.72) results did not have significant differences. The PSAD (0.15±0.08 ng/ml·cm3 vs 0.24±0.05 ng/ml·cm3; P<0.01) and mpMRI (P=0.01) results were significantly different between the NCS-PCa group and CS-PCa group. Conversely, the differences for age (76.00 years, IQR: 69.00–78.00 vs 75.50 years, IQR: 66.50–79.00; P=0.86), tPSA (7.14±1.64 ng/ml vs 7.42±1.18 ng/ml; P=0.44), f/t PSA (0.14±0.07 vs 0.15±0.04; P=0.57), DRE (P=0.53), and TRUS (P=0.82) were not significant between the NCS-PCa group and CS-PCa group.
In correlation analysis, the pathology and mpMRI had a moderate correlation (r=0.36,
VERIFYING THE DIAGNOSTIC VALUE OF CLINICAL PARAMETERS BY USING UNIVARIATE AND MULTIVARIATE LOGISTIC REGRESSION ANALYSES IN PREDICTING BPH VS PCA:
In univariate analysis, the risk of PCa was positively correlated with log-transformed PSAD (OR=2.52, 95% CI: 1.53–4.15, P<0.01) and mpMRI (P<0.01) classification, and was negatively correlated with f/t PSA (OR=0.01, 95% CI: 0.00–0.06, P<0.01) (Table 2). In analysis of diagnostic value, the univariate parameters including f/t PSA (AUC=0.68, 95% CI: 0.61–0.75, P<0.01), PSAD (AUC=0.68, 95% CI: 0.62–0.75, P<0.01), and grade of mpMRI (AUC=0.69, 95% CI: 0.62–0.77, P<0.01) had predictive ability, and log-transformed PSAD (AUC=0.82, 95% CI: 0.76–0.87, P<0.01) showed the best predictive power for PCa (Table 3).
In binary logistic regression analysis with stepwise regression, f/t PSA (P<0.01), log-transformed PSAD (P<0.01), and grade of mpMRI (P<0.01) were brought into the prediction model. Finally, the logistic model (AUC=0.79, 95% CI: 0.73–0.84, P<0.01) for predicting PCa was significantly better than other clinical parameters, and it also had good sensitivity (73.64%) and specificity (64.23%) when its cut-off value was >0.36 (Figure 1).
VERIFYING THE DIAGNOSTIC VALUE OF CLINICAL PARAMETERS BY USING UNIVARIATE AND MULTIVARIATE LOGISTIC REGRESSION ANALYSES IN PREDICTING NCS-PCA AND CS-PCA:
The risk of CS-PCa increased with log-transformed PSAD (OR=66.46, 95% CI: 6.93–637.02) and grade of mpMRI (P<0.01) in univariate analysis. In multivariate analysis, regression analysis also verified the relationship between log-transformed PSAD (OR=85.73, 95% CI: 6.95–1058.08), mpMRI (P<0.01) classification, and risk of CS-PCa increasing (Table 4). In evaluating the diagnostic efficacy, the logistic model (AUC=0.87, 95% CI: 0.77–0.93, P<0.01) for predicting CS-PCa was significantly higher than the separate mpMRI (AUC=0.67, 95% CI: 0.54–0.77, P<0.01) (Figure 2). When choosing cut-off value >0.50, the sensitivity and specificity of the logistic model were 71.41% and 81.82%, respectively.
Discussion
PSA has a revolutionary significance for diagnosing PCa, particularly in the diagnosis of early-stage asymptomatic PCa. Notably, when PSA level is between 4 ng/ml and 10 ng/ml, the number of patients with BPH and PCa significantly overlapped [17].
The systematic prostate biopsy usually has been considered as the criterion standard for diagnosing PCa. Nevertheless, this invasive method can cause psychological distress in patients with PSA in the gray area, so it was essential to combine multiple parameters to diagnose these patients [7]. On this issue, many scholars have tried to use regression analysis to construct models to predict PCa. Liu et al used PSA and PSA derivatives to build a prediction model of PCa in 197 patients with PSA in the gray area, which predicted the sensitivity and specificity of PCa to be 75.4% and 75.8%, respectively [18]. However, that study did not consider imageology examination. Liu et al constructed a prediction model of PCa by using PSA derivatives and mpMRI, and the multivariate models performed significantly better for detection of PCa (AUC=0.79) and CS-PCa (AUC=0.84), but the authors did not assess the sensitivity and specificity of these 2 regression models in the article [19].
In this study, we used logistic regression to analyze not only relevant biochemical indexes PSA including f/t PSA, PSAD, but also some frequently-used clinical examinations such as age, DRE, TRUS, and mpMRI. We also explored which clinical indicators were more valuable in differentiating between NCS-PCa and CS-PCa. Finally, binary logistic regression analysis, log-transformed PSAD, f/t PSA, and grade of mpMRI have identified the relevance of rising of PCa, and the log-transformed PSAD and grade of mpMRI helped identify NCS-PCa and CS-PCa. The diagnostic models of PCa (sensitivity=73.64%; specificity=64.23%; AUC=0.79), and CS-PCa (sensitivity=71.41%; specificity=81.82%; AUC=0.87) both exhibited good diagnostic utility.
Nowadays, screening and detection PCa primarily depend on DRE, tPSA, f/t PSA, PSAD, TRUS, and mpMRI. Several studies have linked an abnormal DRE and a high GS [20,21]. However, our results did not confirm the statistical significance of this association. The China Prostate Cancer Coalition (CPCC) reported that Chinese patients with PCa have the characteristics of smaller PV, higher tPSA level, and higher GS score than their counterparts in Europe and America [22]. Therefore, the main reason why the correlation between DRE and PCa was inconspicuous in the present study is that the enrolled patients were all Chinese. Whether there is racial diversity in the relationship between abnormal DRE and high GS score may require large-scale and multicenter studies.
PSA derivatives, including f/t PSA and PSAD, usually are recommended to assist detection of PCa among patients with a PSA level in the gray area in clinical practice. The free PSA (fPSA) is the fraction of tPSA that does not bind in serum, whose concentration is lower in malignancy patients than in BPH patients, so detection of f/t PSA is most valuable for clinical application when PSA level are in the gray area [23,24]. Our results also show that increased f/t PSA level (OR=0.01,
PSAD is the ratio of tPSA concentration to PV, which offers good guidance on whether to perform a prostate biopsy in patients with a PSA level in the gray zone [26,27]. A previous article reported PSAD value >0.15 ng/ml·cm3 necessitated prostate biopsy [28]. Our results showed PSAD was an independent clinical parameter for predicting of PCa and CS-PCa. However, the cut-off value of PSAD in distinguish BPH from PCa was 0.18 ng/ml·cm3 in our study. The reason why the cut-off value of PSAD is higher than the previous reference value may also be that Chinese patients with PCa have smaller PV and higher tPSA levels [22].
TRUS and mpMRI are both frequently used imaging examinations in prostate disease. Generally, gray-scale TRUS has a certain value in the diagnosis of BPH, but it is not reliable for detecting PCa [6,29]. Our results also verified TRUS had little value for detection of PCa with PSA level in the gray area, while mpMRI including T1WI, T2WI, DWI, and DCE technologies was confirmed to have a perfect diagnostic value, which was similar to other studies [30–32]. Liu et al just reported the AUCs of mpMRI for PCa and CS-PCa were 0.69 and 0.79, respectively [19]. In our study, the sensitivities of mpMRI for predicting PCa and CS-PCa were 0.63 and 0.82, respectively, which is similar to the results of other scholars. Notably, the Prostate Imaging Reporting and Data System (PI-RADS) is a standardized evaluation pattern for prostate mpMRI, whose second version was published in 2015 [33]. PI-RADS second version (PI-RADS V2) showed a better diagnostic value (AUC for PCa=0.79; AUC for CS-PCa=0.86) for evaluating PCa in PSA with gray area [34]. Besides, some scholars reported the PI-RADS score was correlated with the pathological results of PCa, but was not correlated with the levels of PSA [35,36]. Nevertheless, because this retrospective study lacks popularization of PI-RADS, the results of mpMRI for patients were only classified as “positive”, “suspicious”, and “negative”, which resulted in low diagnostic values of predicting PCa and CS-PCa (AUC for PCa=0.69; AUC for CS-PCa=0.67). Therefore, PI-RADS V2 could be used as a dependable standardized evaluation pattern for further promotion in prostate mpMRI for PSA level in the gray area.
Binary logistic regression models also were used to predict and evaluate PCa and CS-PCa among patients with PSA in the gray area. We brought clinical parameters consisting of age, DRE, tPSA, f/t PSA, log-transformed PSAD, TRUS, and mpMRI into regression analysis to construct the prediction model. Finally, the 2 prediction models both showed better diagnostic value than all single parameters. When selecting cut-off value >0.36, the sensitivity, specificity, and AUC of predicting PCa model were 73.64%, 64.23%, and 0.79, respectively, which is a more reliable way to predict PCa in clinical practice.
According to GS, PCa patients were divided into the NCS-PCa group (GS <7) and the CS-PCa group (GS ≥7), and different groups received different treatments. NCS-PCa could be monitored closely without surgical intervention, which has a good prognosis and a low mortality rate, while high-risk PCa patients must be actively treated [37,38]. Therefore, accurate identification of low-risk PCa patients could reduce the risk of performing unnecessary invasive biopsies, without increasing the risk to patient health [18]. In the present study, we developed a satisfactory diagnostic model for predicting NCS-PCa and CS-PCa, whose sensitivity and specificity were 71.41% and 81.82%, respectively. Compared with the model of Jun Liu et al, who just analyzed PSA and its derivatives for CS-PCa, our prediction model also included mpMRI and had a better detection value (AUC: 0.82 vs 0.87) [18].
Our study was subject to several limitations. Firstly, this was a retrospective study with single-center samples, with drawbacks inherent in the study design. Furthermore, adding more clinical parameters perhaps augment the accuracy of the prediction model, some scholars considered some PCa genomic biomarkers could improve the diagnostic value and avoid unnecessary biopsy [39]. Selvi et al reported that arginine and its metabolites, including plasma arginine, ornithine, arginine to ornithine ratio, and urinary diacetylspermine, could be used as molecular markers to predict PCa when the PSA level is in the gray area, and these biomarkers could be an important research focus in diagnosing PCa [40]. Third, active surveillance (AS), a strategy of management of patients with low-risk prostate cancer, could reduce unnecessary over-treatment [41]. A meta-analysis found the mpMRI-targeted biopsy is a useful addition to systematic confirmation in patients on AS with low-risk PCa [42]. However, the present research has not follow-up this field because AS was carried out late in our hospital. We may consider performing more relevant studies in the future. Nonetheless, the models we built are convenient, and the required parameters are accessible and valuable.
Conclusions
We developed 2 models to predict PCa and CS-PCa. When the best cut-off value is 0.36 in the regression model for the PCa group and 0.50 for the CS-PCa group, the 2 prediction models had satisfactory diagnostic value for PCa and CS-PCa among patients with PSA in the gray area, which could provide an important reference value for clinical treatment. Prospective studies are needed for further confirmation.
Figures
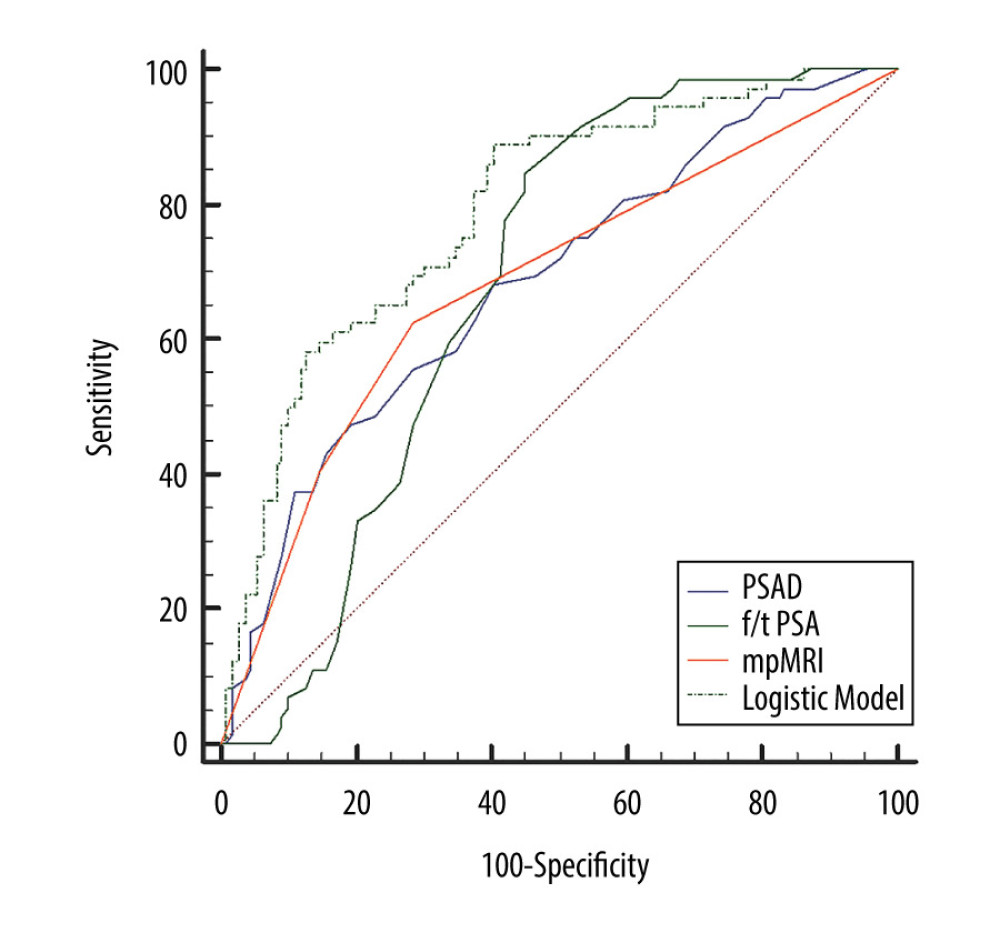 Figure 1. The ROC curve of univariate and multivariate analysis for predicting PCa. PSAD – prostate-specific antigen density; f/t PSA – free/total prostate-specific antigen; mpMRI – multiparametric magnetic resonance imaging.
Figure 1. The ROC curve of univariate and multivariate analysis for predicting PCa. PSAD – prostate-specific antigen density; f/t PSA – free/total prostate-specific antigen; mpMRI – multiparametric magnetic resonance imaging. 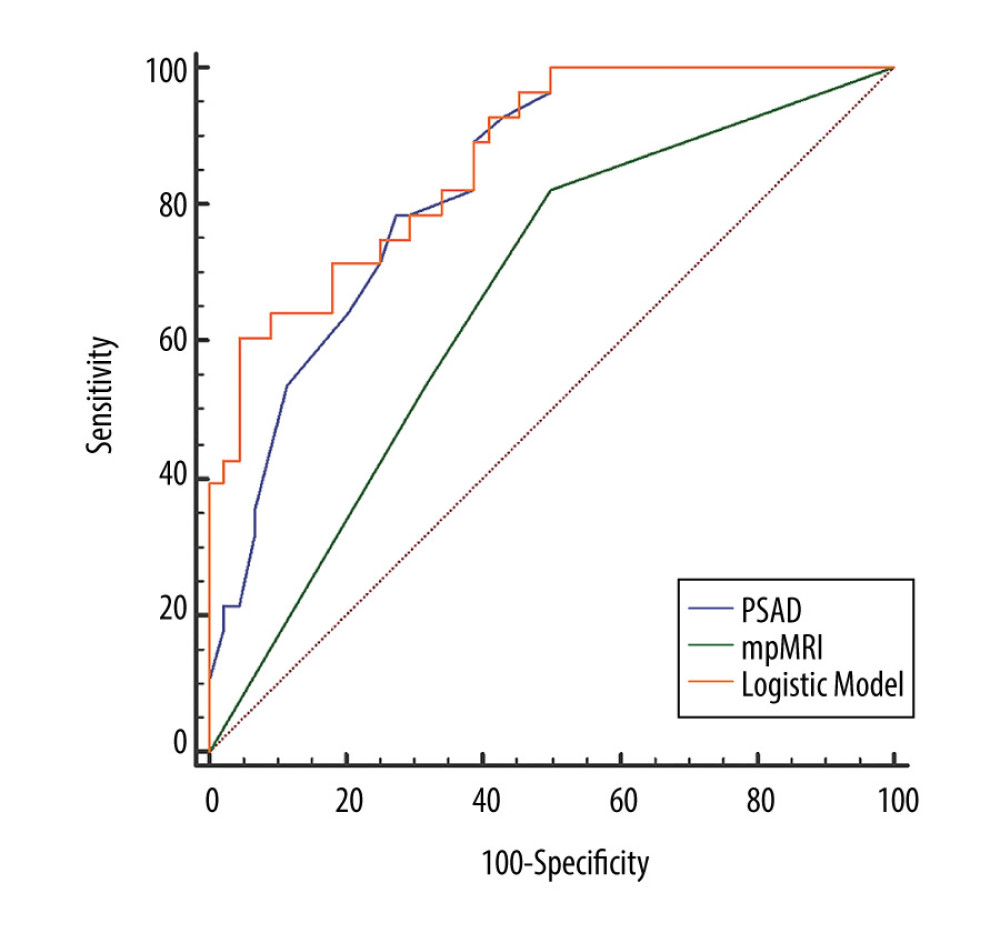 Figure 2. The ROC curve of univariate and multivariate analysis for predicting CS-PCa. PSAD – prostate-specific antigen density; mpMRI – multiparametric magnetic resonance imaging.
Figure 2. The ROC curve of univariate and multivariate analysis for predicting CS-PCa. PSAD – prostate-specific antigen density; mpMRI – multiparametric magnetic resonance imaging. Tables
Table 1. Characteristics of clinical information for enrolled patient.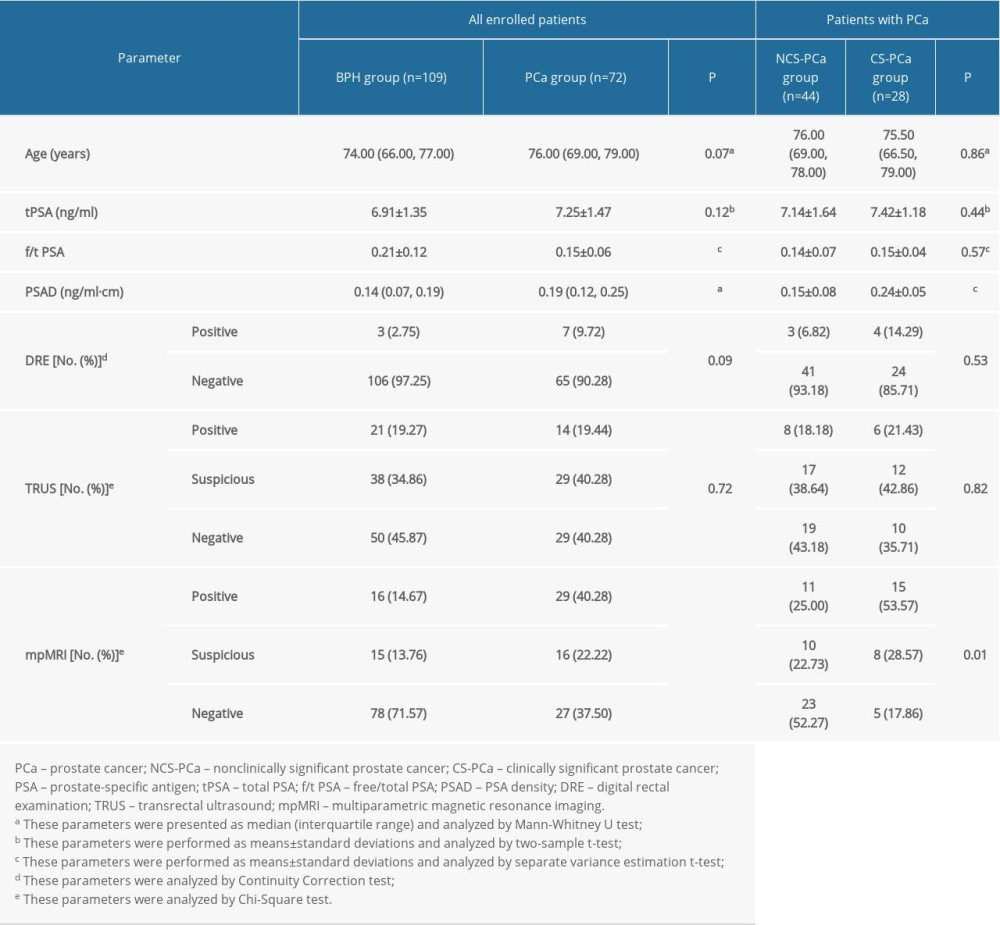 Table 2. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting BPH vs PCa.
Table 2. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting BPH vs PCa.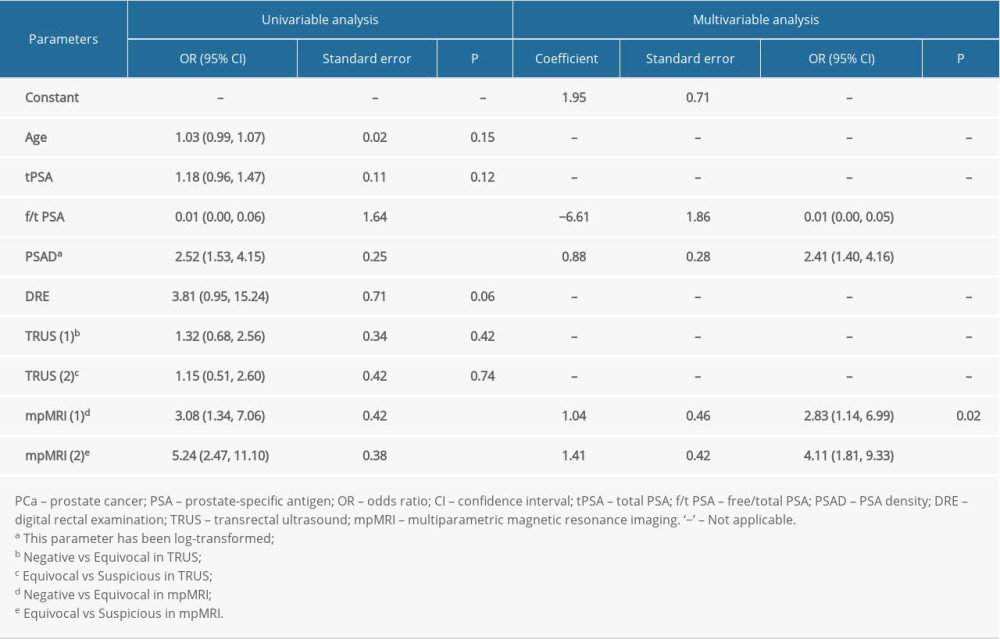 Table 3. Diagnostic value of clinical parameters and logistic regression models in predicting PCa and CS-PCa.
Table 3. Diagnostic value of clinical parameters and logistic regression models in predicting PCa and CS-PCa.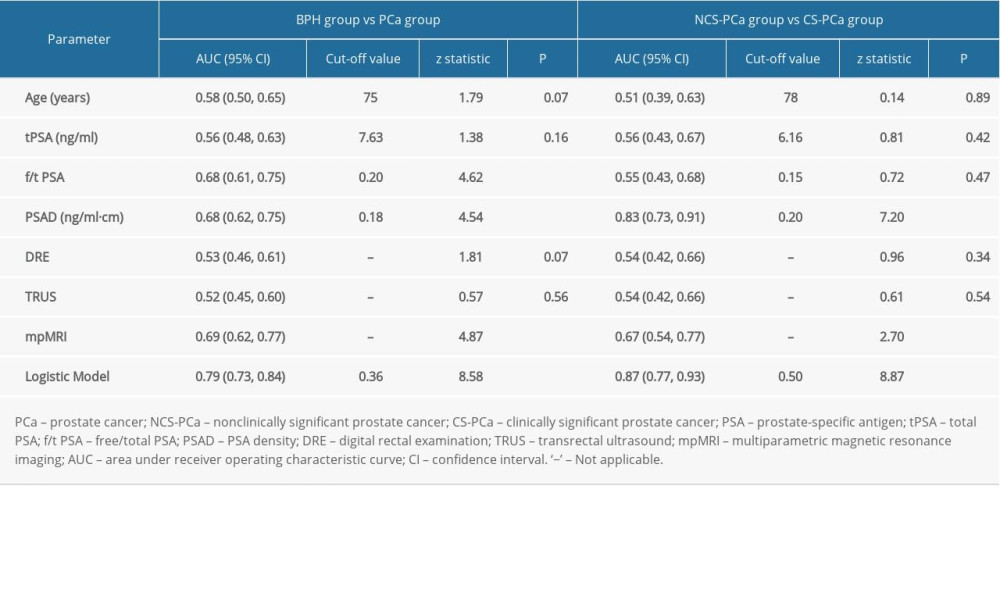 Table 4. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting NCS-PCa and CS-PCa.
Table 4. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting NCS-PCa and CS-PCa.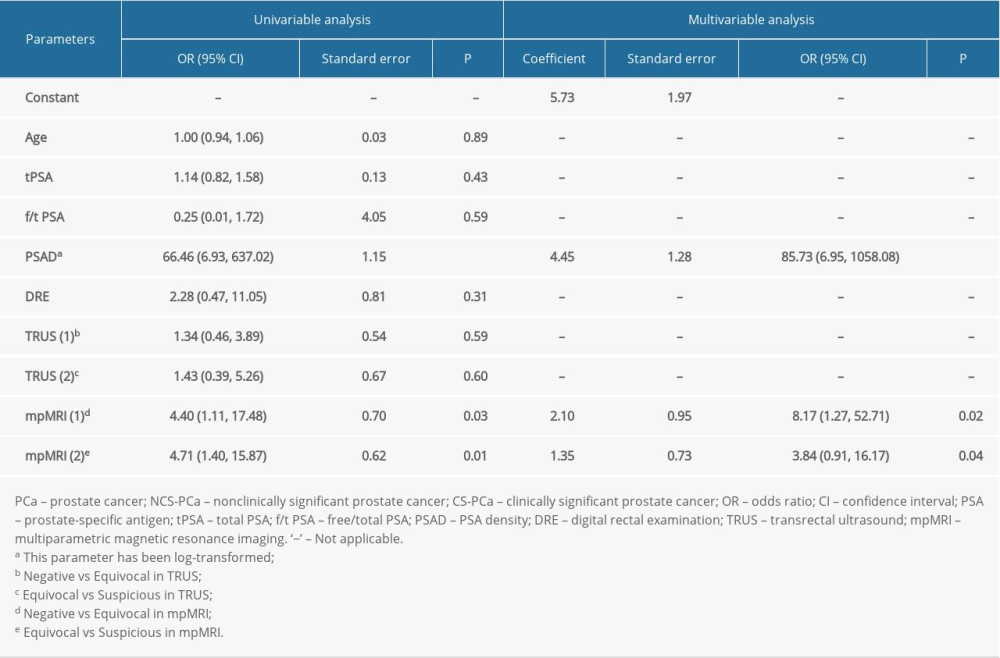
References
1. Cai Z, Liu Q, Understanding the Global Cancer Statistics 2018: Implications for cancer control: Science China Life Sciences, 2019 [Online ahead of print]
2. Ye D, Zhu YEpidemiology of prostate cancer in China: An overview and clinical implication: Zhonghua Wai Ke Za Zhi, 2015; 53(4); 249-52 [in Chinese]
3. Chen WQ, Zeng HM, Zheng RS, Cancer incidence and mortality in China, 2007: Chin J Cancer Res, 2012; 24(1); 1-8
4. Chen W, Sun K, Zheng R, Cancer incidence and mortality in China, 2014: Chin J Cancer Res, 2018; 30(1); 1-12
5. Chen M, Cao CL, Cui XWShear-wave elastosonography for diagnosis of prostatic cancer: A meta-analysis: International Journal of Clinical and Experimental Medicine, 2019; 12(8); 9533-47 [in Chinese]
6. Van Den Bergh RCN, Mottet N, Briers E: European Association of Urology Guidelines on Prostate Cancer, 2019 https://uroweb.org/guideline/prostate-cancer/
7. Xia J, Gulati R, Au M, Effects of screening on radical prostatectomy efficacy: The prostate cancer intervention versus observation trial: J Natl Cancer Inst, 2013; 105(8); 546-50
8. Chen R, Xie LP, Zhou L-QCurrent status of prostate biopsy in Chinese Prostate Cancer Consortium member hospitals: Chinese Journal of Urology, 2015; 36(5); 342-45 [in Chinese]
9. Huang Y, Li Z-Z, Huang Y-L, Value of free/total prostate-specific antigen (f/t PSA) ratios for prostate cancer detection in patients with total serum prostate-specific antigen between 4 and 10 ng/mL: A meta-analysis: Medicine (Baltimore), 2018; 97(13); e0249
10. Ha YS, Yu J, Salmasi AH, Prostate-specific antigen density toward a better cutoff to identify better candidates for active surveillance: Urology, 2014; 84(2); 365-71
11. Ceylan C, Gazel E, Keles I, Can the free/total PSA ratio predict the Gleason score before prostate biopsy?: Curr Urol, 2016; 9(1); 24-27
12. Sang L, Wang X-M, Xu D-Y, Cai Y-F, Accuracy of shear wave elastography for the diagnosis of prostate cancer: A meta-analysis: Sci Rep, 2017; 7(1); 1949
13. Harmon SA, Tuncer S, Sanford T, Artificial intelligence at the intersection of pathology and radiology in prostate cancer: Diagn Interv Radiol, 2019; 25(3); 183-88
14. Beyersdorff D, Taymoorian K, Knösel T, MRI of prostate cancer at 1.5 and 3.0 T: Comparison of image quality in tumor detection and staging: Am J Roentgenol, 2005; 185; 1214-20
15. Epstein JI, Allsbrook WC, Amin MB, Egevad LLand the Isup Grading Committee, The 2005 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma: Am J Surg Pathol, 2005; 29(9); 1228-42
16. Christie D, How accurately can prostate gland imaging measure the prostate gland volume? Results of a systematic review: Prostate Cancer, 2019; 2019; 1-12
17. Alonzo D, Mure A, Soloway M, Prostate cancer and the increasing role of active surveillance: Postgrad Med, 2013; 125; 109-16
18. Liu J, Wang ZQ, Li M, Establishment of two new predictive models for prostate cancer to determine whether to require prostate biopsy when the PSA level is in the diagnostic gray zone (4–10 ng ml(−1)): Asian J Androl, 2020; 22(2); 213-16
19. Junxiao L, Biao D, Wugong Q, Using clinical parameters to predict prostate cancer and reduce the unnecessary biopsy among patients with PSA in the gray zone: Sci Rep, 2020; 10(1); 5157
20. Okotie O, Roehl K, Han M, Characteristics of prostate cancer detected by digital rectal examination only: Urology, 2008; 70; 1117-20
21. Gosselaar C, Roobol M, Roemeling S, Schröder F, The role of the digital rectal examination in subsequent screening visits in the European Randomized Study of Screening for Prostate Cancer (ERSPC), Rotterdam: Eur Urol, 2008; 54; 581-88
22. Sun Y-HConsensus of Chinese experts on prostate puncture: Chinese Journal of Urology, 2016; 37(4); 241-44 [in Chinese]
23. Bachour DM, Chahin E, Al-Fahoum S, Human kallikrein-2, prostate specific antigen and free-prostate specific antigen in combination to discriminate prostate cancer from benign diseases in Syrian patients: Asian Pac J Cancer Prev, 2015; 16(16); 7085-88
24. Thakur V, Singh P, Talwar M, Mukherjee U, Utility of free/total prostate specific antigen (f/t PSA) ratio in diagnosis of prostate carcinoma: Dis Markers, 2004; 19; 287-92
25. Stephan C, Lein M, Jung K, The influence of prostate volume on the ratio of free to total prostate specific antigen in serum of patients with prostate carcinoma and benign prostate hyperplasia: Cancer, 1997; 79(1); 104-9
26. Schoots IG, Osses DF, Drost F-JH, Reduction of MRI-targeted biopsies in men with low-risk prostate cancer on active surveillance by stratifying to PI-RADS and PSA-density, with different thresholds for significant disease: Transl Androl Urol, 2018; 7(1); 132-44
27. Ujike T, Uemura M, Kawashima A, A novel model to predict positive prostate biopsy based on serum androgen level: Endocr Relat Cancer, 2018; 25(1); 59-67
28. Udeh EI, Nnabugwu II, Ozoemena FO, Prostate-specific antigen density values among patients with symptomatic prostatic enlargement in Nigeria: World J Surg Oncol, 2016; 41(1); 174
29. Smeenge M, Barentsz J, Cosgrove D, Role of transrectal ultrasonography (TRUS) in focal therapy of prostate cancer: Report from a Consensus Panel: BJU Int, 2012; 110(7); 942-48
30. Porcaro AB, Novella G, Molinari A, Prostate volume index and chronic inflammation of the prostate type IV with respect to the risk of prostate cancer: Urol Int, 2015; 94(3); 270-85
31. Zhu Y, Han CT, Zhang GM, Effect of body mass index on the performance characteristics of PSA-related markers to detect prostate cancer: Sci Rep, 2016; 6; 19034
32. Catalona WJ, Richie JP, Ahmann FR, Comparison of digital rectal examination and serum prostate specific antigen in the early detection of prostate cancer: Results of a multicenter clinical trial of 6,630 men: J Urol, 2017; 197(2 Suppl); S200-7
33. Weinreb JC, Barentsz JO, Choyke PL, PI-RADS Prostate Imaging – Reporting And Data System: 2015, Version 2: Eur Urol, 2016; 69(1); 16-40
34. Zhang L, Tang M, Chen S, A meta-analysis of use of Prostate Imaging Reporting and Data System Version 2 (PI-RADS V2) with multiparametric MR imaging for the detection of prostate cancer: Eur Radiol, 2017; 27(12); 5204-14
35. Zhu LY, Ding XF, Huang TBCorrelation analysis between prostate imaging report and data system score and pathological results of prostate cancer: Zhonghua Yi Xue Za Zhi, 2020; 100(34); 2663-68 [in Chinese]
36. Gündoğdu E, Emekli E, Kebapçı M, Evaluation of relationships between the final Gleason score, PI-RADS v2 score, ADC value, PSA level, and tumor diameter in patients that underwent radical prostatectomy due to prostate cancer: Radiol Med, 2020; 125(9); 827-37
37. Wang S-C, Chen C-C, Yang C-K, Pathological outcomes in men with prostate cancer who are eligible for active surveillance: J Chin Med Assoc, 2017; 81(4); 348-51
38. Sebesta EM, Anderson CB, The surgical management of prostate cancer: Semin Oncol, 2017; 44(5); 347-57
39. Kaufmann S, Bedke J, Gatidis S, Prostate cancer gene 3 (PCA3) is of additional predictive value in patients with PI-RADS grade III (intermediate) lesions in the MR-guided re-biopsy setting for prostate cancer: World J Urol, 2016; 34(4); 509-15
40. Selvi I, Basar H, Baydilli N, The importance of plasma arginine level and its downstream metabolites in diagnosing prostate cancer: Int Urol Nephrol, 2019; 51(11); 1975-83
41. Sklinda K, Mruk B, Walecki J, Active surveillance of prostate cancer using multiparametric magnetic resonance imaging: A review of the current role and future perspectives: Med Sci Monit, 2020; 26; e920252
42. Schoots IG, Nieboer D, Giganti F, Is magnetic resonance imaging-targeted biopsy a useful addition to systematic confirmatory biopsy in men on active surveillance for low-risk prostate cancer? A systematic review and meta-analysis: BJU Int, 2018; 122(6); 946-58
Figures
 Figure 1. The ROC curve of univariate and multivariate analysis for predicting PCa. PSAD – prostate-specific antigen density; f/t PSA – free/total prostate-specific antigen; mpMRI – multiparametric magnetic resonance imaging.
Figure 1. The ROC curve of univariate and multivariate analysis for predicting PCa. PSAD – prostate-specific antigen density; f/t PSA – free/total prostate-specific antigen; mpMRI – multiparametric magnetic resonance imaging. Figure 2. The ROC curve of univariate and multivariate analysis for predicting CS-PCa. PSAD – prostate-specific antigen density; mpMRI – multiparametric magnetic resonance imaging.
Figure 2. The ROC curve of univariate and multivariate analysis for predicting CS-PCa. PSAD – prostate-specific antigen density; mpMRI – multiparametric magnetic resonance imaging. Tables
 Table 1. Characteristics of clinical information for enrolled patient.
Table 1. Characteristics of clinical information for enrolled patient. Table 2. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting BPH vs PCa.
Table 2. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting BPH vs PCa. Table 3. Diagnostic value of clinical parameters and logistic regression models in predicting PCa and CS-PCa.
Table 3. Diagnostic value of clinical parameters and logistic regression models in predicting PCa and CS-PCa. Table 4. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting NCS-PCa and CS-PCa.
Table 4. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting NCS-PCa and CS-PCa. Table 1. Characteristics of clinical information for enrolled patient.
Table 1. Characteristics of clinical information for enrolled patient. Table 2. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting BPH vs PCa.
Table 2. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting BPH vs PCa. Table 3. Diagnostic value of clinical parameters and logistic regression models in predicting PCa and CS-PCa.
Table 3. Diagnostic value of clinical parameters and logistic regression models in predicting PCa and CS-PCa. Table 4. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting NCS-PCa and CS-PCa.
Table 4. Verifying the diagnostic value of clinical parameters by using univariate and multivariate logistic regression analyses in predicting NCS-PCa and CS-PCa. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








