23 May 2021: Clinical Research
Risk Factors Associated with All-Cause Death Among Dialysis Patients with Diabetes
Anna Grzywacz1ABCDEFG*, Arkadiusz Lubas1ACDE, Jerzy Smoszna1AD, Stanisław Niemczyk1ADGDOI: 10.12659/MSM.930152
Med Sci Monit 2021; 27:e930152
Abstract
BACKGROUND: Patients receiving dialysis who also have diabetes mellitus have high mortality. This study aimed to investigate the risk factors associated with all-cause death among Polish patients with diabetes receiving dialysis.
MATERIAL AND METHODS: This prospective observational study included 100 patients with type 1 or type 2 diabetes who were treated with peritoneal dialysis or hemodialysis. Blood laboratory tests, the occurrence of diabetes complications, and comorbidity, using the Charlson Comorbidity Index, were estimated. Survival analysis was performed using the multivariate Cox proportional hazard model, and Kaplan-Meyer survival analyses with log-rank tests were performed to show differences between groups.
RESULTS: During 16.0±5.0 months, 23 patients died. The deceased group had significantly higher levels of HbA1c (P=0.046) and fructosamine (P=0.011) than the surviving group. The deceased patients also had higher comorbidity scores (P=0.013). In the stepwise multivariate Cox proportional hazard regression model, history of stroke or transient ischemic attack was an independent risk factor of all-cause death (hazard ratio [HR] 3.15, 95% CI 1.34-7.39; P=0.009), while regular physical activity significantly reduced the risk of all-cause death (HR 0.26, 95% CI 0.08-0.87; P=0.029).
CONCLUSIONS: Deceased patients had higher HbA1c and fructosamine levels and higher comorbidity. However, history of stroke or transient ischemic attack was an independent risk factor of all-cause death, while regular physical activity was associated with the reduction of the risk of all-cause death in patients with type 1 and type 2 diabetes treated with peritoneal dialysis or hemodialysis. Regular physical activity should be recommended to improve survival in this population.
Keywords: Diabetes Mellitus, Mortality, Motor Activity, Stroke, Renal Dialysis, Aged, 80 and over, Cause of Death, Comorbidity, Diabetes Complications, Life Style, Poland, Prospective Studies
Background
Diabetes mellitus is a primary cause of end-stage renal disease (ESRD) [1,2]. According to the European Renal Association-European Dialysis and Transplant Association (ERA-EDTA) Registry from 2016, which included 36 countries, the incidence of renal replacement therapy (RRT) was 121 per million people. Almost two-thirds of patients were men, over one-half were ≥65 years old, and in nearly one-fourth of cases, diabetes was the main cause of kidney failure. At the end of 2016, the RRT prevalence in these countries was 823 per million people. For all patients initiating RRT from 2007 to 2011, the 5-year survival probability was 50.5% [3]. Among the dialysis population, patients with diabetes represent the group where the highest mortality rates are observed [4], with the 5-year mortality rate in this population exceeding 70% [5]. Cardiovascular diseases are the main cause of death in patients receiving dialysis, and they occur twice as often in patients with diabetes [4]. The second most frequent cause of death are infectious complications, which are more than twice as common in this same population [5]. A recent systematic review and meta-analysis assessing risk factors of mortality among patients undergoing hemodialysis (HD) revealed that increased age, previous cardiovascular disease, and higher C-reactive protein (CRP), HbA1c, adiponectin, ferritin, troponin T, and brain natriuretic peptide levels were associated with increased risk of all-cause mortality, while a higher body mass index (BMI) and higher hemoglobin, albumin, total iron-binding capacity, serum iron, and apolipoprotein A2 and A3 levels were associated with a reduced risk of all-cause mortality [6]. Independent predictors of mortality among patients receiving peritoneal dialysis (PD) with diabetes were older age, female sex, previous cardiovascular disease, protein-energy wasting, and low renal residual function [7]. In a small Polish study, independent risk factors of all-cause mortality among patients with diabetes on PD were hypoalbuminemia and older age, while low serum albumin level and low cholesterol concentration were independent risk factors of all-cause mortality in patients with diabetes on HD [8]. According to the meta-analysis of 17 cohort studies, including 441 842 patients on HD and 62 462 patients on PD, patients with diabetes and ESRD in the HD group had a significantly lower mortality risk than did patients with diabetes and ESRD in the PD group [9]. Transient ischemic attack (TIA) and stroke are serious vascular events. Chinese patients with diabetes and a history of stroke who were treated with PD had shorter survival than did those treated with HD, while the mortality rate was similar between patients receiving PD and HD treatment with a history of stroke but not diabetes [10].
In the general population, regular physical activity is an important element of a healthy lifestyle, reducing the risk of cardiovascular disease and thus contributing to improved survival. However, there is little data on the effect of physical activity on mortality in the dialysis population. One study demonstrated that low physical activity was associated with higher mortality in patients on dialysis [11]. In another study that included patients on HD, very active aerobic exercise significantly reduced the risk of death [12]. There are no studies including the impact of lifestyle on mortality among patients who are on dialysis treatment and have diabetes.
The study aimed to investigate the risk factors, including lifestyle, associated with all-cause death in patients with type 1 and type 2 diabetes and ESRD who were on HD or PD.
Material and Methods
ETHICS APPROVAL AND CONSENT TO PARTICIPATE:
This work was conducted according to the principles of the Declaration of Helsinki. The study is a part of the project that was approved by the Bioethics Committee at the Military Institute of Medicine (resolution No. 29/WIM/2017). All patients gave their written informed consent to participate in the study.
ELIGIBILITY CRITERIA:
Inclusion criteria were age ≥18 years, ESRD during HD or PD, duration of RRT ≥3 months, and type 1 or type 2 diabetes for ≥3 months. Three months of RRT or diabetes before inclusion to the study was used arbitrarily to eliminate bias from increased mortality in patients initiating HD and to allow for the stabilization of laboratory test results after initiation of RRT or diabetes treatment. This time also allowed patients to adjust their lifestyle to the limitations related to the diseases and their treatment.
Exclusion criteria were a patient’s lack of consent to participate in the study, other types of diabetes, severe verbal or cognitive disorders that did not allow for the collection of a medical interview.
STUDY POPULATION:
We initially intended to assess mortality in a group consisting of 4 equally sized subgroups of patients (all n ≥20) with type 1 or type 2 diabetes and different types of RRT (PD or HD). Thus, we qualified all patients with diabetes treated in the 2 largest dialysis centers in central Poland who met the inclusion and exclusion criteria. Because of a significant disproportion of patients in the groups, we searched for patients with type 1 diabetes in other minor dialysis centers. However, up until the recruitment deadline, only 7 patients with type 1 diabetes and HD were eligible for inclusion in the study (Figure 1).
Patients with diabetes were identified based on the analysis of the medical records from the dialysis centers. The presence of diabetes micro- and macroangiopathic complications and all comorbidities were assessed by medical record analysis and an interview. Patients were asked about their duration of diabetes, residual diuresis, and lifestyle factors including regular physical activity and cardiovascular risk factors like smoking.
STUDY MEASUREMENTS:
Comorbidity was assessed using the Charlson Comorbidity Index (CCI) [13], and cognitive condition was tested with Mini-Mental State Examination [14].
Patients’ weight and height were obtained on scales with a height measuring device. Waist and hip circumference were measured according to the World Health Organization recommendations, using a stretch-resistant tape. The waist was measured at the midpoint between the lower margin of the last rib and the top of the iliac crest in the mid axillary line. Hip circumference was measured around the widest part of the buttocks [15]. These measurements were obtained before HD or at the control visit for patients receiving PD. The waist-to-hip ratio (WHR) was calculated according to the following formula: WHR=waist circumference/hip circumference. The BMI was calculated according to the following formula: BMI=body weight (kg)/height2 (m).
Systolic and diastolic blood pressure, as well as pulse rate, were measured with an oscillometric blood pressure device on the arm at the beginning of HD or at the control visit for patients receiving PD. Heart rate was estimated from the measured pulse rate.
Laboratory tests were performed in the Department of Laboratory Diagnostics at the Military Institute of Medicine. Blood samples were drawn at the beginning of HD or at the control visit for patients receiving PD. Blood HbA1c levels were obtained by the turbidimetric inhibition immunoassay; serum fructosamine, total protein, and albumin were assessed by the colorimetric method; total cholesterol, high-density lipoprotein, low-density lipoprotein, and triglycerides were obtained with enzymatic colorimetric reactions; and serum CRP was determined using the immunoturbidimetric method; all tests were performed using a Cobas c 501 analyzer (Roche Diagnostics). Blood hemoglobin concentration was assessed by an automated hematology analyzer SYSMEX XN-1000.
The duration of the study was limited by the framework of the project. Patients were recruited from June 1, 2018 to April 30, 2019. From the first evaluation, patients were followed for hospitalizations and mortality to the end of May 2020. The causes of death were collected from hospital discharge summary reports along with diagnoses and data from dialysis center archive records.
STATISTICAL ANALYSIS:
Statistical tests were performed using Statistica 12 software (StatSoft, Cracow, Poland). The normal di
Results
One hundred patients were included in this prospective observational study. The homogeneity of the study participants resulted from the homogeneity of the local Polish population. Patients were recruited from June 1, 2018 to April 30, 2019, and observation for mortality continued until May 31, 2020, for a total of 16.0±5.0 months.
Study participants’ mean age was 64.0±13.3 years, the mean diabetes duration was 22.4±11.3 years, and patients were undergoing RRT for median (IQR) 24.5 (36.0) months. During the observation period, 23 patients died (23%) in the following groups: diabetes type 1 5/23 (21.74%), diabetes type 2 18/77 (23.38%), HD 17/59 (22.37%), and PD 6/24 (25%), with no statistically significant differences between groups. After dividing into subgroups and taking into account the modality of RRT and type of diabetes, the mortality also did not differ significantly: diabetes type 1 and HD in 3/15 (20%) patients, diabetes type 2 and HD in 14/61 (23%) patients, diabetes type 1 and PD in 2/8 (25%) patients, and diabetes type 2 and PD in 4/16 (25%) patients.
In the studied cohort, the leading causes of death in 9/23 patients (39.13%) were concomitant cardiovascular and infectious complications. In 7/23 patients (30.43%), cardiovascular diseases were the only cause of death, and in 6/23 patients (26.09%), infectious complications were the main cause of death. One person (4.35%) died due to respiratory failure. The characteristics of the study participants divided into deceased and surviving groups are shown in Table 1.
The deceased group had significantly higher HbA1c and fructosamine levels than did the surviving group. The deceased patients also had significantly higher comorbidity, as determined by the CCI score, with a substantially lower estimated 10-year survival rate and more hospitalizations during observation. Further, significantly more patients from the deceased group had a history of stroke or TIA.
In the stepwise multivariate Cox proportional hazard regression model, which included duration of chronic kidney disease, duration of tobacco smoking, number of hospitalizations, HbA1c, fructosamine, stroke or TIA, and physical activity, only stroke or TIA and regular physical activity were independent factors significantly affecting survival. Although CCI scores differed significantly between groups, they were excluded from the Cox regression model because of significant collinearity with other included variables. History of stroke or TIA was a statistically significant predictor of all-cause death (hazard ratio [HR] 3.15, 95% confidence interval [CI] 1.34–7.39;
Discussion
In our study, the history of stroke or TIA was an independent risk factor of all-cause death (HR 3.15, 95% CI 1.34–7.39;
Regular physical activity as a part of a healthy lifestyle contributes to improving prognosis by reducing cardiovascular risk [16]. Lack of physical activity leads to sarcopenia, which increases the risk of death [17]. In our present study, undertaking regular physical activity significantly reduced the risk of death (HR 0.26, 95% CI 0.08–0.87;
In a US study including 1554 patients beginning HD or PD, of which 56% had diabetes, the association between physical activity scores and mortality was linear. Patients with the lowest physical activity level had a higher risk of death than those with average physical activity or physical activity above an average HR of 3.5 (95% CI 2.54–4.89) [11].
HbA1c is the recommended marker of long-term glycemic control. Although a higher level of HbA1c is associated with poor outcome, its optimal level in diabetic patients with ESRD is unknown [18]. Serum fructosamine is also a good indicator of glycemic control in patients on HD and PD [19,20], as well as a significant predictor of hospitalization and infection in patients with diabetes on HD [19]. Fructosamine is also connected with an increased risk of cardiovascular and all-cause mortality, first cardiovascular disease event, and first sepsis hospitalization in HD [5].
According to the results of our study, worse diabetes control, expressed by higher levels of HbA1c (7.38±1.53% vs 6.72±1.47%;
Patients from our study with diabetes and ESRD had a very high cardiovascular risk due only to having these diseases, based on the SCORE scale, which was developed for the Polish population [24]. In the present study, deceased patients had a trend of longer tobacco smoking history (26 (33) vs 10 (30) years;
Patients with cardiovascular diseases, ESRD, and diabetes have higher mortality rates. However, the coexistence of diabetes and ESRD increases the risk of cardiovascular disease. Chang et al investigated the influence of ESRD and diabetes on the risk of cardiovascular events, including congestive heart failure, acute MI, and stroke. Their study included 648 851 patients without ESRD and 71 397 patients with ESRD, of which 53 342 and 34 754, respectively, had diabetes. Participants were followed up from 1998 to 2009, and the study demonstrated that in comparison with people without ESRD or diabetes, concomitant diabetes and ESRD had a synergistic effect for acute MI and stroke (HR 5.24, 95% CI 4.83–5.68;
In the present study, mortality in subgroups regarding the type of diabetes and dialysis modality was similar: type 1 diabetes 21.74%, type 2 diabetes 23.38%, HD 22.37%, and PD 25% (P>0.05). In the literature, data on the influence of type of dialysis modality on mortality differ. There are studies reporting higher overall mortality in patients on HD [26,27], in patients on PD [28,29], or showing no difference between them [8,30–33]. In some works, during the first months of RRT, survival is better in patients on PD [30,34–36]. In many studies, diabetes [25,33,35,36] and, as expected, older age [32,33,35,36] are associated with the higher mortality of patients on dialysis.
Patients with type 1 diabetes or type 2 diabetes on prevalent HD or PD treatment have complex health problems. This is also reflected by the fact that in our research, concomitant cardiovascular and infectious complications were the cause of death in the majority of analyzed cases (39.13%). Combining the above number with cases of only cardiovascular deaths (30.43%) or with only infectious deaths (26.09%), cardiovascular complications were responsible for 69.56% of deaths and infection led to 65.22% of deaths.
In patients with ESRD and diabetes, other chronic concomitant diseases also affect prognosis. The CCI was created to investigate the influence of comorbid conditions on the risk of mortality in prospective studies and estimates a 10-year survival due to this additional burden [13]. The CCI is widely used as a valid and reliable method [37]. It was also validated in assessing comorbidity and predicting survival in patients with ESRD [38]. In our study, comorbidity assessed by the CCI in both groups was high, but it was statistically significantly higher in the deceased patients (9.0±1.8 vs 8.0 (3.0) points;
In spite of the limitations of the study, which included a rather small number of participants and a relatively short observation time, we hope that our work demonstrated a broader view of Polish patients undergoing dialysis who also have diabetes. We believe that the presented work will contribute to a better understanding of the complex health problems of these patients. More research is needed in the ESRD population with diabetes owing to the increased mortality rates in these patients. We plan further observation of this study group.
Conclusions
Based on the conducted research, deceased diabetic patients with ESRD on HD or PD had worse control of diabetes, expressed as higher HbA1c and fructosamine levels. Higher comorbidity was found among patients with worse outcomes. A history of stroke or TIA was an independent risk factor of all-cause death, while regular physical activity was associated with the reduced risk of all-cause death in patients with type 1 or 2 diabetes mellitus treated with PD or HD. Undertaking regular physical activity should be recommended to improve survival in the evaluated population as a modifiable factor. New solutions in education and medical care should be introduced to improve outcomes in this population.
Figures
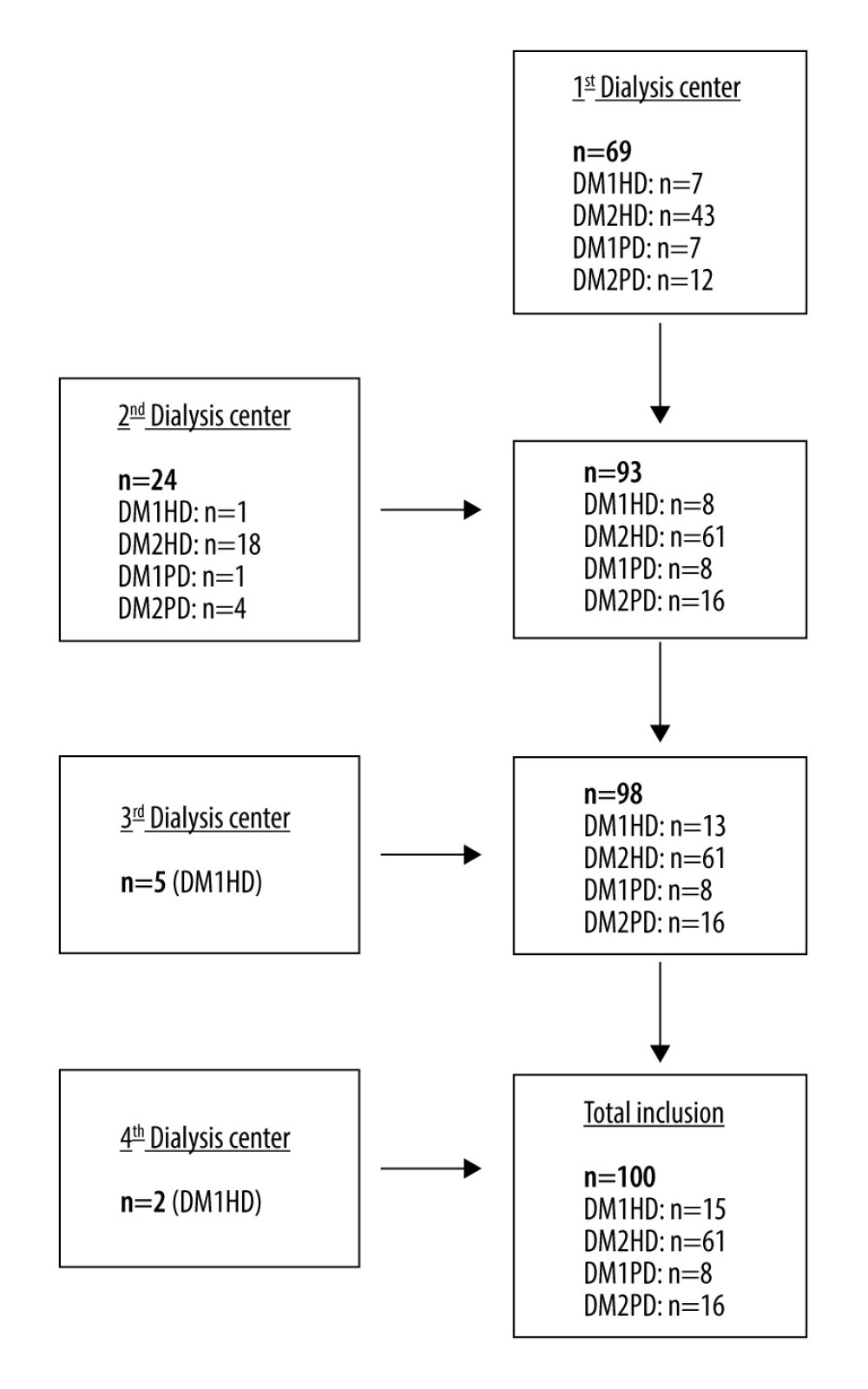 Figure 1. Patients’ inclusion data. DM1 – type 1 diabetes mellitus; DM2 – type 2 diabetes mellitus; HD – hemodialysis; PD – peritoneal dialysis.
Figure 1. Patients’ inclusion data. DM1 – type 1 diabetes mellitus; DM2 – type 2 diabetes mellitus; HD – hemodialysis; PD – peritoneal dialysis. 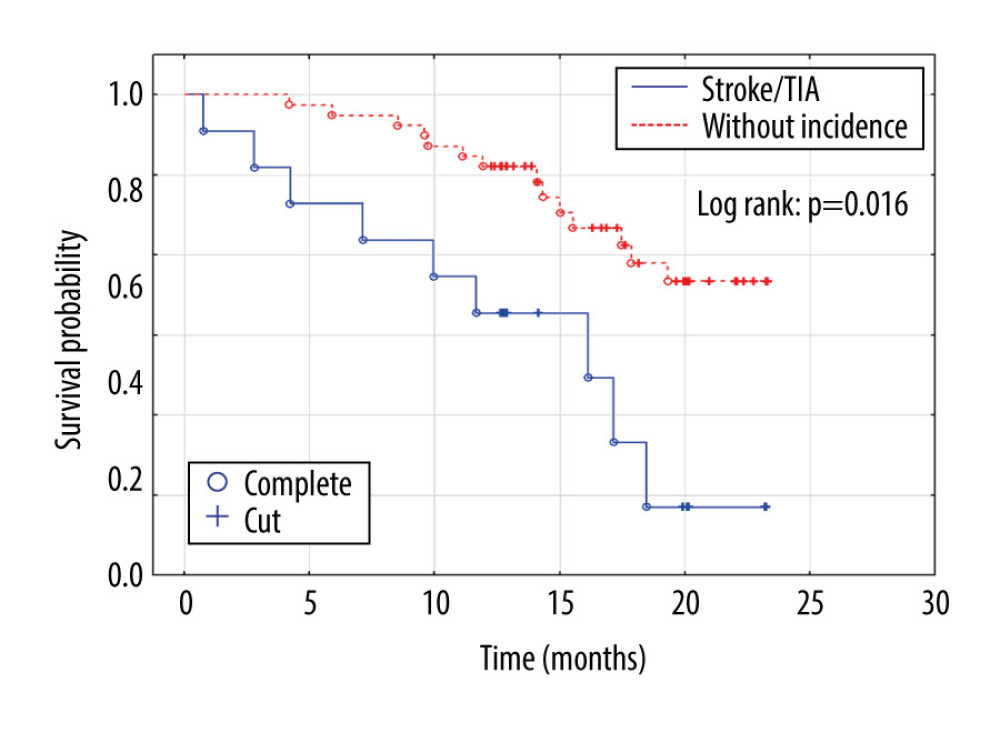 Figure 2. Kaplan-Meier survival function. The difference in mortality between patients with diabetes and end-stage renal disease according to history of stroke or transient ischemic attack.
Figure 2. Kaplan-Meier survival function. The difference in mortality between patients with diabetes and end-stage renal disease according to history of stroke or transient ischemic attack. 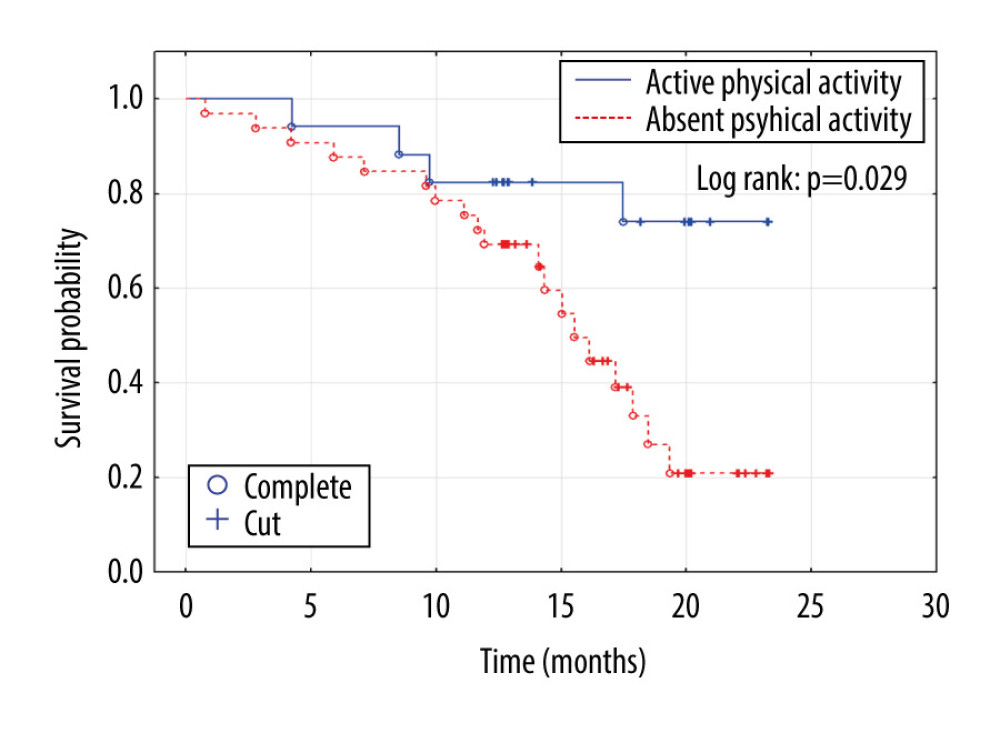 Figure 3. Kaplan-Meier survival function. The difference in mortality between patients with diabetes and end-stage renal disease according to regular physical activity.
Figure 3. Kaplan-Meier survival function. The difference in mortality between patients with diabetes and end-stage renal disease according to regular physical activity. References
1. Saran R, Robinson B, Abbott KC, US Renal Data System 2019 Annual Data Report: Epidemiology of kidney disease in the United States: Am J Kidney Dis, 2020; 75; A6-7
2. Collins AJ, Foley RN, Chavers B, US Renal Data System 2013 Annual Data Report: Preface: Am J Kidney Dis, 2014; 63; A7
3. Kramer A, Pippias M, Noordzij M, The European Renal Association – European Dialysis and Transplant Association (ERA-EDTA) Registry Annual Report 2016: A summary: Clin Kidney J, 2019; 12; 702-20
4. Williams ME, Lacson E, Wang W, Glycemic control and extended hemodialysis survival in patients with diabetes mellitus: Comparative results of traditional and time-dependent Cox model analyses: Clin J Am Soc Nephrol, 2010; 5; 1595-601
5. Shafi T, Sozio SM, Plantinga LC, Serum fructosamine and glycated albumin and risk of mortality and clinical outcomes in hemodialysis patients: Diabetes Care, 2013; 36; 1522-33
6. Ma L, Zhao S, Risk factors for mortality in patients undergoing hemodialysis: A systematic review and meta-analysis: Int J Cardiol, 2017; 238; 151-58
7. Chung SH, Han DC, Noh H, Risk factors for mortality in diabetic peritoneal dialysis patients: Nephrol Dial Transpl, 2010; 25; 3742-48
8. Madziarska K, Weyde W, Penar J, Different mortality predictor pattern in hemodialysis and peritoneal dialysis diabetic patients in 4-year prospective observation: Postepy Hig Med Dosw, 2013; 67; 1076-82
9. Xue J, Li H, Zhou Q, Comparison of peritoneal dialysis with hemodialysis on survival of diabetic patients with end-stage kidney disease: A meta-analysis of cohort studies: Ren Fail, 2019; 41; 521-31
10. Wang IK, Liang WM, Lin CL, Impact of dialysis modality on the survival of patients with end-stage renal disease and prior stroke: Int Urol Nephrol, 2016; 48; 139-47
11. Johansen KL, Kaysen GA, Dalrymple LS, Association of physical activity with survival among ambulatory patients on dialysis: The comprehensive dialysis study: Clin J Am Soc Nephrol, 2013; 8; 248-53
12. Lopes AA, Lantz B, Morgenstern H, Associations of self-reported physical activity types and levels with quality of life, depression symptoms, and mortality in hemodialysis patients: The DOPPS: Clin J Am Soc Nephrol, 2014; 9; 1702-12
13. Charlson ME, Pompei P, Ales KL, A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation: J Chronic Dis, 1987; 40; 373-83
14. Folstein MF, Folstein SE, McHugh PR, “Mini-mental state” A practical method for grading the cognitive state of patients for the clinician: J Psychiatr Res”, 1975; 12; 189-98
15. World Health Organization: Waist circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation December, 2008, Geneva, Switzerland, WHO [Cited 9 Feb 2021], www.cdc.gov/nchs/data/nhanes/nhanes_07_08/manual_an.pdf
16. Hamdy O, Mottalib A, Morsi A, Long-term effect of intensive lifestyle intervention on cardiovascular risk factors in patients with diabetes in real-world clinical practice: A 5-year longitudinal study: BMJ Open Diabetes Res Care, 2017; 5; 259
17. Mori K, Nishide K, Okuno S, Impact of diabetes on sarcopenia and mortality in patients undergoing hemodialysis: BMC Nephrol, 2019; 20; 105
18. , KDIGO 2020 Clinical Practice Guideline for Diabetes Management in Chronic Kidney Disease: Kidney Int, 2020; 98; S1-115
19. Mittman N, Desiraju B, Fazil I, Serum fructosamine versus glycosylated hemoglobin as an index of glycemic control, hospitalization, and infection in diabetic hemodialysis patients: Kidney Int, 2010; 78; S41-45
20. Lee S-Y, Chen Y-C, Tsai I-C, Glycosylated hemoglobin and albumin-corrected fructosamine are good indicators for glycemic control in peritoneal dialysis patients: PLoS One, 2013; 8; e57762
21. Rhee JJ, Zheng Y, Montez-Rath ME, Associations of glycemic control with cardiovascular outcomes among us hemodialysis patients with diabetes mellitus: J Am Heart Assoc, 2017; 6; e005581
22. Ricks J, Molnar MZ, Kovesdy CP, Glycemic control and cardiovascular mortality in hemodialysis patients with diabetes: A 6-year cohort study: Diabetes, 2012; 61; 708-15
23. Oomichi T, Emoto M, Tabata T, Impact of glycemic control on survival of diabetic patients on chronic regular hemodialysis: A 7-year observational study: Diabetes Care, 2006; 29; 1496-500
24. Zdrojewski T, Jankowski P, Bandosz PA new version of cardiovascular risk assessment system and risk charts calibrated for Polish population: Kardiol Pol, 2015; 73; 958-61 [in Polish]
25. Chang YT, Wu JL, Hsu CC, Diabetes and end-stage renal disease synergistically contribute to increased incidence of cardiovascular events: A nationwide follow-up study during 1998–2009: Diabetes Care, 2014; 37; 277-85
26. Mehrotra R, Chiu YW, Kalantar-Zadeh K, Similar outcomes with hemodialysis and peritoneal dialysis in patients with end-stage renal disease: Arch Intern Med, 2011; 171; 110-18
27. Schaubel DE, Morrison HI, Fenton SSA, Comparing mortality rates on CAPD/CCPD and hemodialysis. The Canadian experience: Fact or fiction?: Perit Dial Int, 1998; 18; 478-84
28. Kim HJ, Park JT, Han SH, The pattern of choosing dialysis modality and related mortality outcomes in Korea: A national population-based study: Korean J Intern Med, 2017; 32; 699-710
29. Bloembergen WE, Port FK, Mauger EA, A comparison of mortality between patients treated with hemodialysis and peritoneal dialysis1: J Am Soc Nephrol, 1995; 6(2); 177-83
30. Lukowsky LR, Mehrotra R, Kheifets L, Article comparing mortality of peritoneal and hemodialysis patients in the first 2 years of dialysis therapy: A marginal structural model analysis: Clin J Am Soc Nephrol, 2013; 8; 619-28
31. Lee CC, Sun CY, Wu MS, Long-term modality-related mortally analysis in incident dialysis Patients: Perit Dial Int, 2009; 29; 182-90
32. Waldum-Grevbo B, Leivestad T, Reisaeter AV, Impact of initial dialysis modality on mortality: A propensity-matched study, 2015; 16; 179
33. Yeates K, Zhu N, Vonesh E, Hemodialysis and peritoneal dialysis are associated with similar outcomes for end-stage renal disease treatment in Canada 2012: Nephrol Dial Transplant, 2012; 27(9); 3568-75
34. Wilkinson TJ, Clarke AL, Nixon DGD, Prevalence and correlates of physical activity across kidney disease stages: An observational multicentre study: Nephrol Dial Transplant, 2019 [Epub ahead of print]
35. Liem YS, Wong JB, Hunink MGM, Comparison of hemodialysis and peritoneal dialysis survival in The Netherlands: Kidney Int, 2007; 71; 153-58
36. Mircescu G, Ştefan G, Gârneaţă L, Outcomes of dialytic modalities in a large incident registry cohort from Eastern Europe: The Romanian Renal Registry: Int Urol Nephrol, 2014; 46; 443-51
37. De Groot V, Beckerman H, Lankhorst GJ, How to measure comorbidity: A critical review of available methods: J Clin Epidemiol, 2003; 56; 221-29
38. Hemmelgarn BR, Manns BJ, Quan H, Adapting the Charlson comorbidity index for use in patients with ESRD: Am J Kidney Dis, 2003; 42; 125-32
Figures
 Figure 1. Patients’ inclusion data. DM1 – type 1 diabetes mellitus; DM2 – type 2 diabetes mellitus; HD – hemodialysis; PD – peritoneal dialysis.
Figure 1. Patients’ inclusion data. DM1 – type 1 diabetes mellitus; DM2 – type 2 diabetes mellitus; HD – hemodialysis; PD – peritoneal dialysis. Figure 2. Kaplan-Meier survival function. The difference in mortality between patients with diabetes and end-stage renal disease according to history of stroke or transient ischemic attack.
Figure 2. Kaplan-Meier survival function. The difference in mortality between patients with diabetes and end-stage renal disease according to history of stroke or transient ischemic attack. Figure 3. Kaplan-Meier survival function. The difference in mortality between patients with diabetes and end-stage renal disease according to regular physical activity.
Figure 3. Kaplan-Meier survival function. The difference in mortality between patients with diabetes and end-stage renal disease according to regular physical activity. Tables
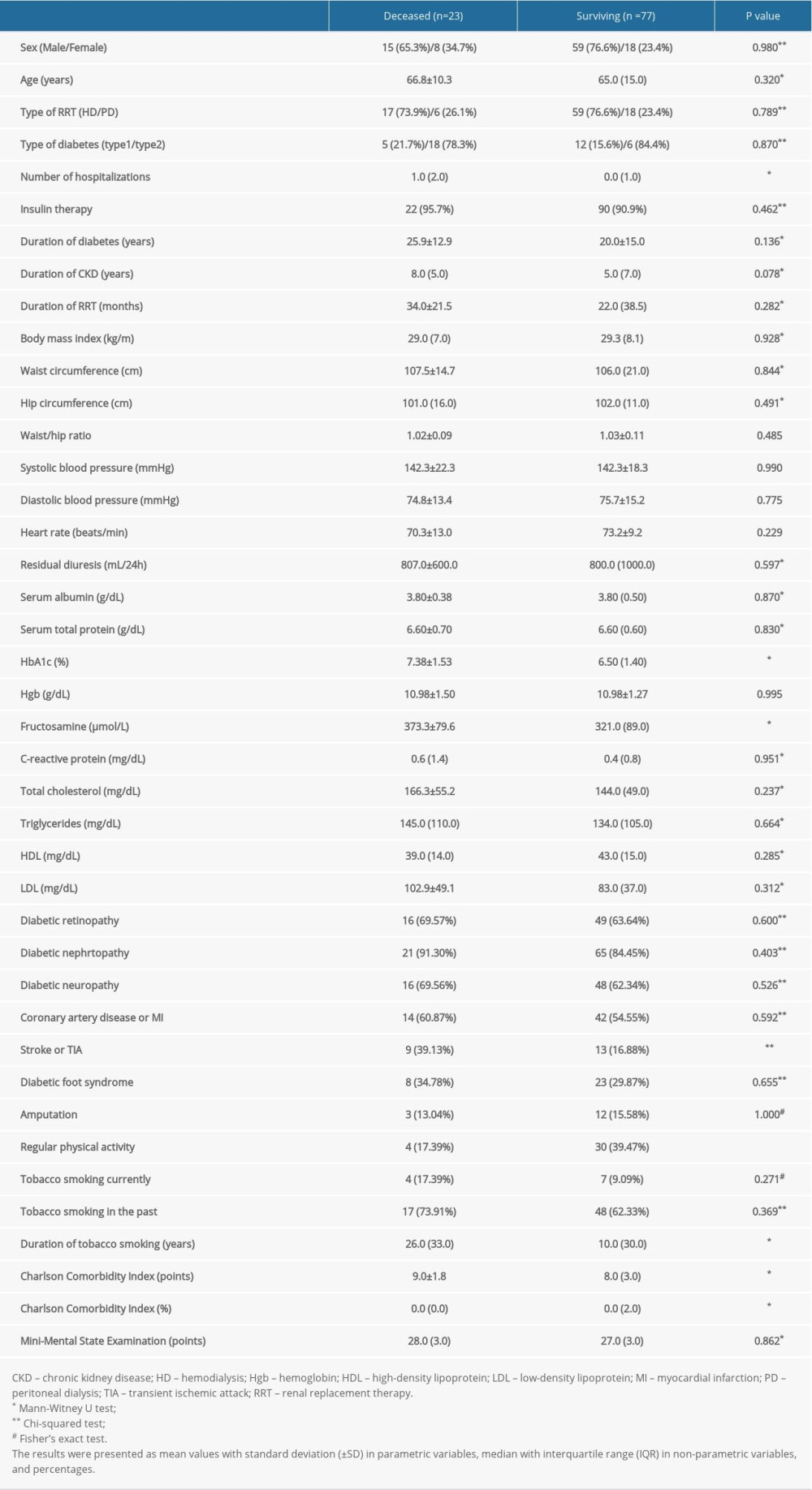 Table 1. Characteristics of study participants.
Table 1. Characteristics of study participants.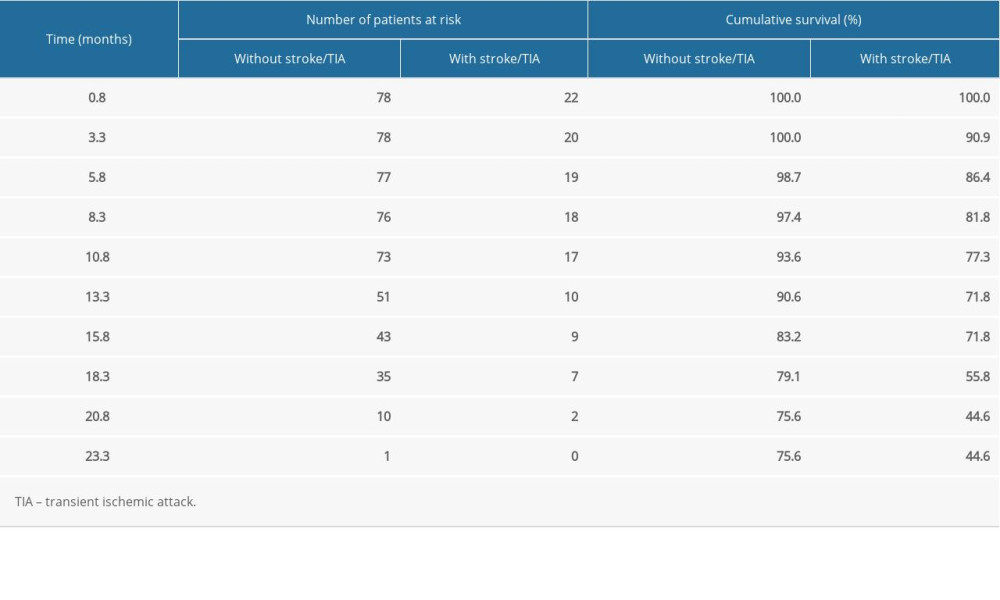 Table 2. Cumulative survival of patients without and with stroke/TIA.
Table 2. Cumulative survival of patients without and with stroke/TIA.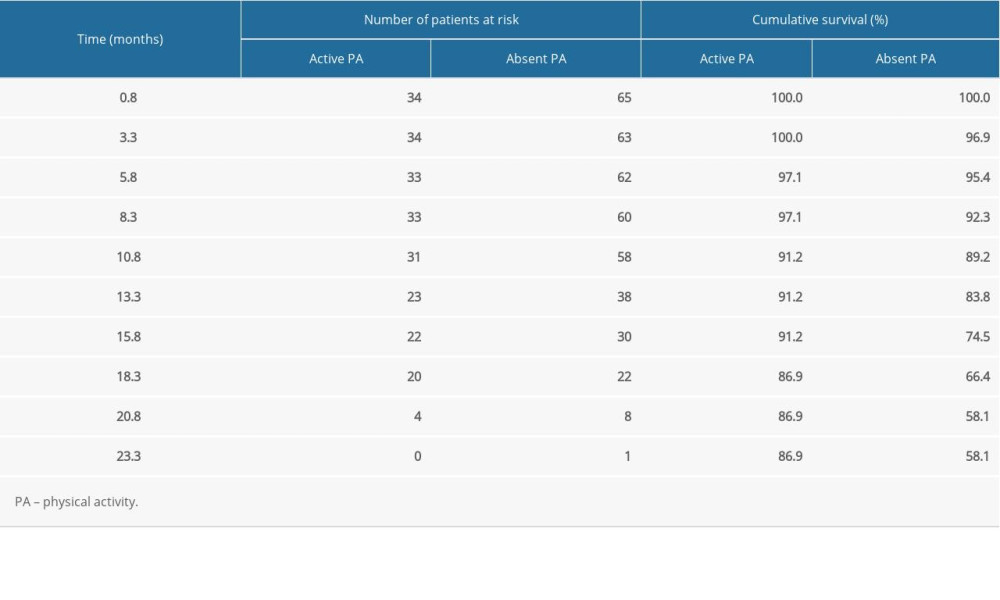 Table 3. Cumulative survival of patients with and without physical activity.
Table 3. Cumulative survival of patients with and without physical activity. Table 1. Characteristics of study participants.
Table 1. Characteristics of study participants. Table 2. Cumulative survival of patients without and with stroke/TIA.
Table 2. Cumulative survival of patients without and with stroke/TIA. Table 3. Cumulative survival of patients with and without physical activity.
Table 3. Cumulative survival of patients with and without physical activity. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








