08 October 2021: Clinical Research
Left Main Bronchus Stenosis Lesion, Neutrophil Count, and Platelet Count Are Predictors of Post-Tuberculosis Bronchomalacia
Yongchang Wu12BE*, Yishi Li2DF, Yang Bai2E, Jinyue Jiang2E, Xiaohui Wang2E, Shuliang Guo2AGDOI: 10.12659/MSM.931779
Med Sci Monit 2021; 27:e931779
Abstract
BACKGROUND: Post-tuberculosis bronchomalacia (PTBM) is one of the main conditions occurring in patients after tracheobronchial tuberculosis (TBTB), and is also associated with the recurrence of symptoms. The present study aimed to investigate the predictors of PTBM in patients who had been undergoing appropriate TB treatment.
MATERIAL AND METHODS: Clinical data of 104 patients with symptomatic airway stenosis after TBTB between January 01, 2019 and June 31, 2020 were recorded and analyzed. The association between baseline clinical characteristics, laboratory results, and PTBM was calculated with logistical regression. The time from onset of bronchoscopic intervention was examined by Kaplan-Meier estimates; differences between the 2 groups were tested by the log-rank test.
RESULTS: Fifty-seven patients (54.81%) had PTBM. In the multivariate logistical analysis, the left main bronchus stenosis lesion (odds ratio [OR]=3.763), neutrophil (NEUT) count (OR=1.527), and platelet (PLT) (OR=1.010) count were predictors of PTBM. During follow-up, patients with BM had a significantly longer duration from onset of bronchoscopic intervention than patients without BM (hazard ratio=2.412, P<0.0001). Further, all patients needing long-term bronchoscopic intervention therapy were subsequently identified as having PTBM. Additionally, blood PLT counts were significantly decreased to normal levels in the non-BM group (P<0.05), but not in the BM group (P>0.05).
CONCLUSIONS: PTBM is most likely to occur in the left main bronchus. The inflammatory and immune responses associated with NEUT and PLT may represent therapeutic targets of PTBM. Our study is the first to report that decreased blood PLT count has the potential to monitor the treatment response.
Keywords: Blood Platelets, Neutrophils, Tracheobronchomalacia, Bronchi, Bronchial Diseases, bronchomalacia, Bronchoscopy, Constriction, Pathologic, Female, Humans, Mycobacterium tuberculosis, Platelet Count, Risk Assessment, Risk Factors, Tomography, X-Ray Computed, Tuberculosis, Pulmonary, young adult
Background
Tracheomalacia is characterized by weakening of the airway wall and dynamic collapse of the airway lumen during respiration [1–4]; the condition is called tracheobronchomalacia if the main bronchi are also affected [5]. At present, there is no generally accepted adult classification of the causes of tracheomalacia. Tracheomalacia may result from tumor invasion [6], cicatricial tracheal stenosis associated with a tracheal tube [7], trauma and surgery [8], compression of an abnormal artery [9,10], or goiter [11], and can be associated with esophageal achalasia [12], cystic fibrosis [13], and chronic obstructive pulmonary disease [14].
Tracheobronchial tuberculosis (TBTB) is a tuberculosis (TB) infection of the trachea and bronchus, and has been reported in 10–50% of patients with pulmonary TB (PTB) [15,16]. Due to the residual and long-term damage after TBTB, 90% of patients with TBTB develop some degree of airway stenosis in the long term, despite adequate antituberculous therapy [17,18]. Post-tuberculosis bronchomalacia (PTBM) is one of the main conditions affecting patients after TBTB [15,19,20]. Consistent with our clinical observations, Lee et al [15] reported that PTBM is associated with the recurrence of symptoms after TBTB. Therefore, in addition to antituberculous treatment, monitoring and determining PTBM is important for early diagnosis and treatment. However, no studies to date have investigated the predictors associated with PTBM. Therefore, we performed this retrospective study to investigate the predictors of PTBM in patients who had been undergoing appropriate TB treatment.
Material and Methods
2 STUDY DESIGN AND POPULATION:
We analyzed the medical records of 104 consecutive hospitalized patients with symptomatic airway stenosis after TBTB in the Department of Respiratory and Critical Care Medicine at the First Affiliated Hospital of Chongqing Medical University between January 01, 2019 and June 31, 2020. All these patients had been undergoing appropriate TB treatment for at least 6 months. The exclusion criteria were active TB, malignant and other infectious diseases, and evidence of other systemic diseases. Data regarding clinical characteristics, laboratory examination, chest computed tomography (CT), bronchoscopic intervention treatment, and outcome were obtained from the patients’ medical records. The final date of follow-up was December 31, 2020. The study was conducted in accordance with the Declaration of Helsinki and Uniform Requirements for Manuscripts Submitted to Biomedical Journals, and the protocol was approved by the Ethics Committee of the First Affiliated Hospital of Chongqing Medical University (Approval ID: No 2020-147).
LABORATORY EXAMINATION VARIABLES:
The laboratory examination included complete blood counts and liver-kidney function. A complete blood count included white blood cell (WBC), red blood cell (RBC), neutrophil (NEUT), lymphocyte (LYM), and platelet (PLT) counts, and hemoglobin (Hb) levels. Liver-kidney function tests included total protein (TP), albumin (Alb), total bilirubin (Tbil), alanine transaminase (ALT), aspartate aminotransferase (AST), glutamyl transpeptidase (GGT), and creatinine (Crea) levels.
DIAGNOSTIC CRITERIA FOR PTBM AND BRONCHOSCOPIC INTERVENTION:
PTBM was diagnosed according to Chung’s classification [20] using a bronchoscope (CV-290; Olympus, Tokyo, Japan). All symptomatic patients with airway stenosis after TBTB were treated using bronchoscopic intervention according to treatment guidelines [21] using an electrocautery needle knife (VIO 300S, ERBE, Tubingen, Germany), a dilatation balloon (ENDO-FLEX, Voerde, Germany), and a multi-use cryosurgery system (ERBOKRYO CA, ERBE, Tuebingen, Germany). Patients were followed up for at least 6 months after the last intervention. As the high incidence of recurrence is the most common problem among patients with PTBM, we therefore investigated the duration of bronchoscopic intervention after the onset of intervention in this population.
STATISTICAL ANALYSIS:
Normality was tested using the Shapiro-Wilk test to separate parametric from non-parametric variables. The continuous variables of normally distributed data are expressed as means±standard deviation and differences between any 2 groups were evaluated using the unpaired
Results
DIAGNOSIS AND INCIDENCE OF PTBM:
There were 104 consecutive hospitalized patients with airway stenosis after TBTB who underwent bronchoscopy; all the patients had cicatrices strictures, and 57 (54.81%) had PTBM, but other types of stenosis were not found. The chest HRCT and bronchoscopy features of patients with PTBM are shown in Figure 1. All patients underwent laboratory examination, chest CT, and bronchoscopic intervention on primary admission.
CLINICAL CHARACTERISTICS OF THE STUDY PARTICIPANTS:
The relevant clinical characteristics of patients with BM and non-BM are summarized in Table 1. The significant clinical characteristics included age, sex, residence, smoking, bronchoscopic intervention history, and symptoms at presentation. Compared to patients without BM, those with BM were more likely to live in a rural area, and had a shorter duration from symptom onset to intervention, more acute intervention therapy, more stent placement, and longer average intervention interval history (all, P<0.05; Table 1).
There were no significant differences in age, sex, smoking, duration of PTB to TBTB diagnosis, and cough, dyspnea, or, more wheeze symptoms at presentation between patients with BM and those without BM (
CHEST CT AND BRONCHOSCOPY FEATURES OF STUDY PARTICIPANTS:
The chest CT and bronchoscopy features are summarized in Table 2. The chest CT features included atelectasis, cavitary, fibrous lesion, mucus plugging, and bronchiectasis. There were no significant differences in any features between patients with BM and those without BM (P>0.05).
The bronchoscopy features primarily included stenosis lesion sites and stenosis degree. Compared to patients without BM, patients with BM had more trachea or left main bronchus stenosis lesions, and had ≥2 bronchi stenosis lesions more frequently (
LABORATORY EXAMINATION OF STUDY PARTICIPANTS:
The results of the laboratory examinations are summarized in Table 3. Compared to patients without BM, those with BM had higher blood WBC, NEUT, and PLT counts; as well as a higher NEUT/LYM ratio (all, P<0.05). NEUT and PLT counts remained elevated in the initial stage. After bronchoscopic intervention, symptoms improved immediately, and about 2 weeks later NEUT and PLT counts began to fall in all the enrolled patients. Then, NEUT and PLT counts continued to increase as airway stenosis and bronchomalacia (BM) worsened. No significant differences were found in blood RBC counts, Hb levels, and LYM counts between the 2 groups (P>0.05). Regarding liver-kidney function, the blood TP, Alb, Tbil, ALT, AST, GGT, and Crea levels were not significantly different between BM and non-BM patients (P>0.05).
PREDICTORS ASSOCIATED WITH PTBM:
Among the variables that showed significant differences in the univariate analysis (Tables 1–3), 4 were selected using the forward stepwise method for the multivariate logistic regression model for PTBM. In the univariate analysis, living in a rural area, left main bronchus stenosis lesion, NEUT count, and PLT count were all associated with the development of PTBM (Table 4). In the multivariate logistic analysis, left main bronchus stenosis lesion, NEUT count, and PLT counts were independently associated with PTBM. Living in a rural area was excluded, which suggested that living in a rural area was not independently related to PTBM (Table 4).
OUTCOMES OF THE STUDY PARTICIPANTS:
Symptoms improved immediately after bronchoscopic intervention in all the enrolled patients. As shown in Figure 2, the time from onset of bronchoscopic intervention was significantly longer in patients with BM than that in patients without BM (hazard ratio [HR]=2.412, P<0.0001). During follow-up, there were 6 (10.53%) patients in the BM group and 2 (4.26%) patients in the non-BM group who required long-term bronchoscopic intervention therapy to restore airway patency. Further, the patients needing long-term bronchoscopic intervention therapy in the BM and non-BM groups were all subsequently identified as having BM.
The complete blood count results from 84 (53 with BM, 31 with non-BM) patients were used to study the long-term effect of bronchoscopic intervention therapy. The changes are summarized in Table 5. PLT counts were significantly decreased to normal levels in the non-BM group (P<0.05), but not in the BM group (P>0.05). The PLT counts in both groups showed a fluctuating trend in the long-term follow-up. The PLT counts in the non-BM group gradually decreased to normal levels, while the PLT counts in the BM group were always higher than normal levels. Regarding WBC, RBC, Hb, NEUT, and LYM levels, the changes were not significant in both groups (P>0.05).
Discussion
LIMITATIONS OF THE STUDY:
The main limitation of our study is that it was a single-center retrospective study with a relatively small sample size; therefore, the possibility of bias exists. In addition, longer follow-up periods after bronchoscopic intervention are required to evaluate the recurrence of airway stenosis after TBTB. Prospective, multicenter, large-sample studies are needed to confirm our findings.
Conclusions
PTBM is most likely to occur in the left main bronchus. The inflammatory and immune responses associated with NEUT and PLT counts may represent therapeutic targets of PTBM in the future. Non-invasive strategies for monitoring PTBM are clinically important but lacking. Our study is the first to report that decreased blood PLT count has the potential to monitor the treatment response. In future studies, we will enroll more patients in a multicenter study to provide enough data to confirm the findings of the present study.
Figures
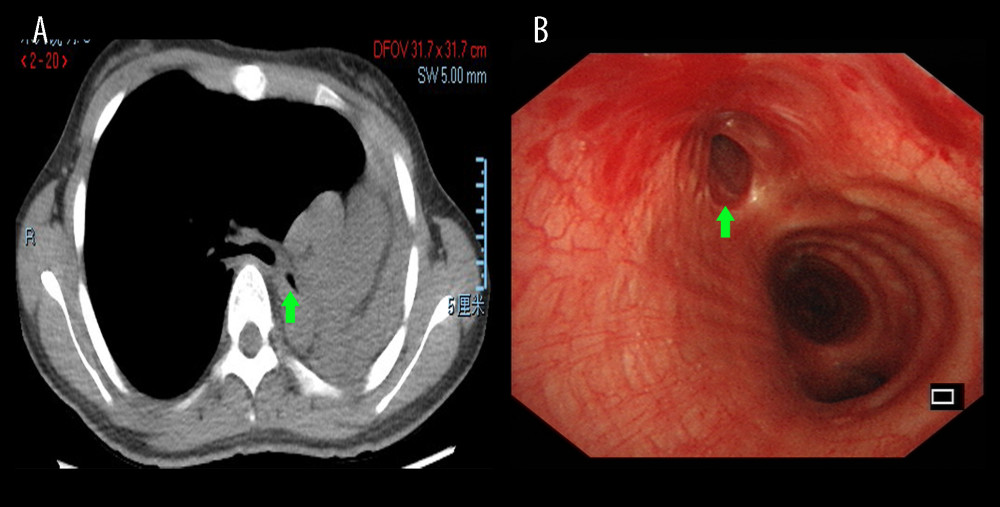 Figure 1. Chest HRCT and bronchoscopy features of patients with PTBMChest HRCT showing left main bronchus stenosis lesion (A, green arrow) due to TBTB. Diagnostic bronchoscopic appearance of PTBM (B, green arrow) can be observed in the left main bronchus. HRCT – high-resolution computed tomography; PTBM – post-tuberculosis bronchomalacia; TBTB – tracheobronchial tuberculosis.
Figure 1. Chest HRCT and bronchoscopy features of patients with PTBMChest HRCT showing left main bronchus stenosis lesion (A, green arrow) due to TBTB. Diagnostic bronchoscopic appearance of PTBM (B, green arrow) can be observed in the left main bronchus. HRCT – high-resolution computed tomography; PTBM – post-tuberculosis bronchomalacia; TBTB – tracheobronchial tuberculosis. 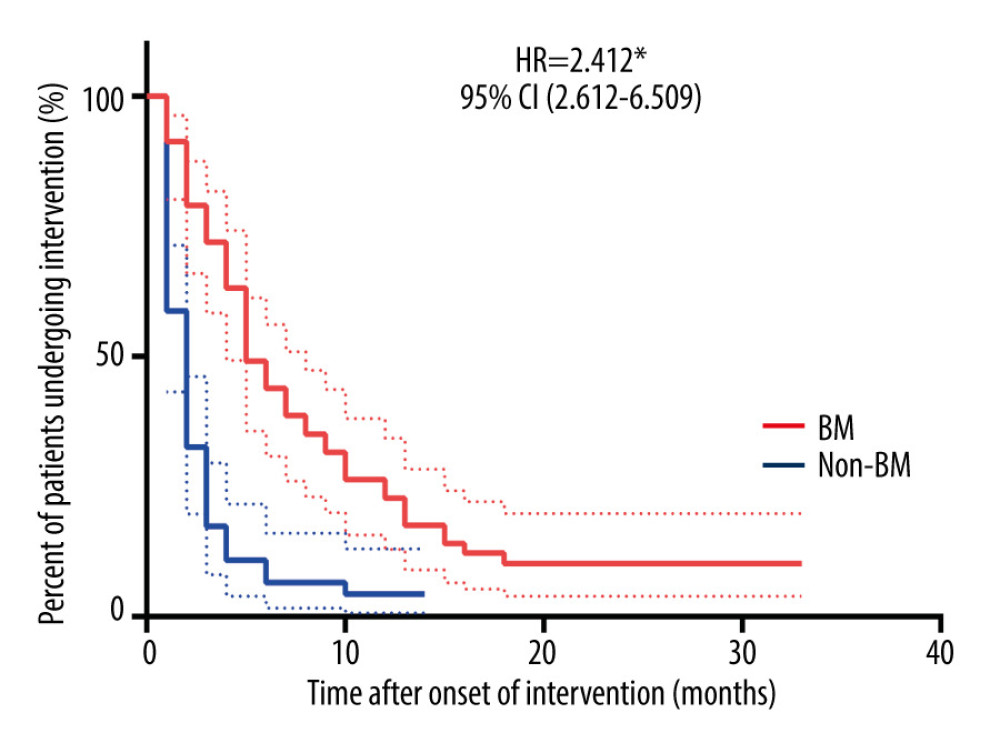 Figure 2. Duration of bronchoscopic intervention therapy in BM and non-BM groupsThe time from onset of bronchoscopic intervention was examined by Kaplan-Meier estimates. Differences between the 2 groups were tested by the log-rank test. * P<0.0001. BM – bronchomalacia; HR – hazard ratio; CI – confidential interval.
Figure 2. Duration of bronchoscopic intervention therapy in BM and non-BM groupsThe time from onset of bronchoscopic intervention was examined by Kaplan-Meier estimates. Differences between the 2 groups were tested by the log-rank test. * P<0.0001. BM – bronchomalacia; HR – hazard ratio; CI – confidential interval. Tables
Table 1. Clinical characteristics of study participants.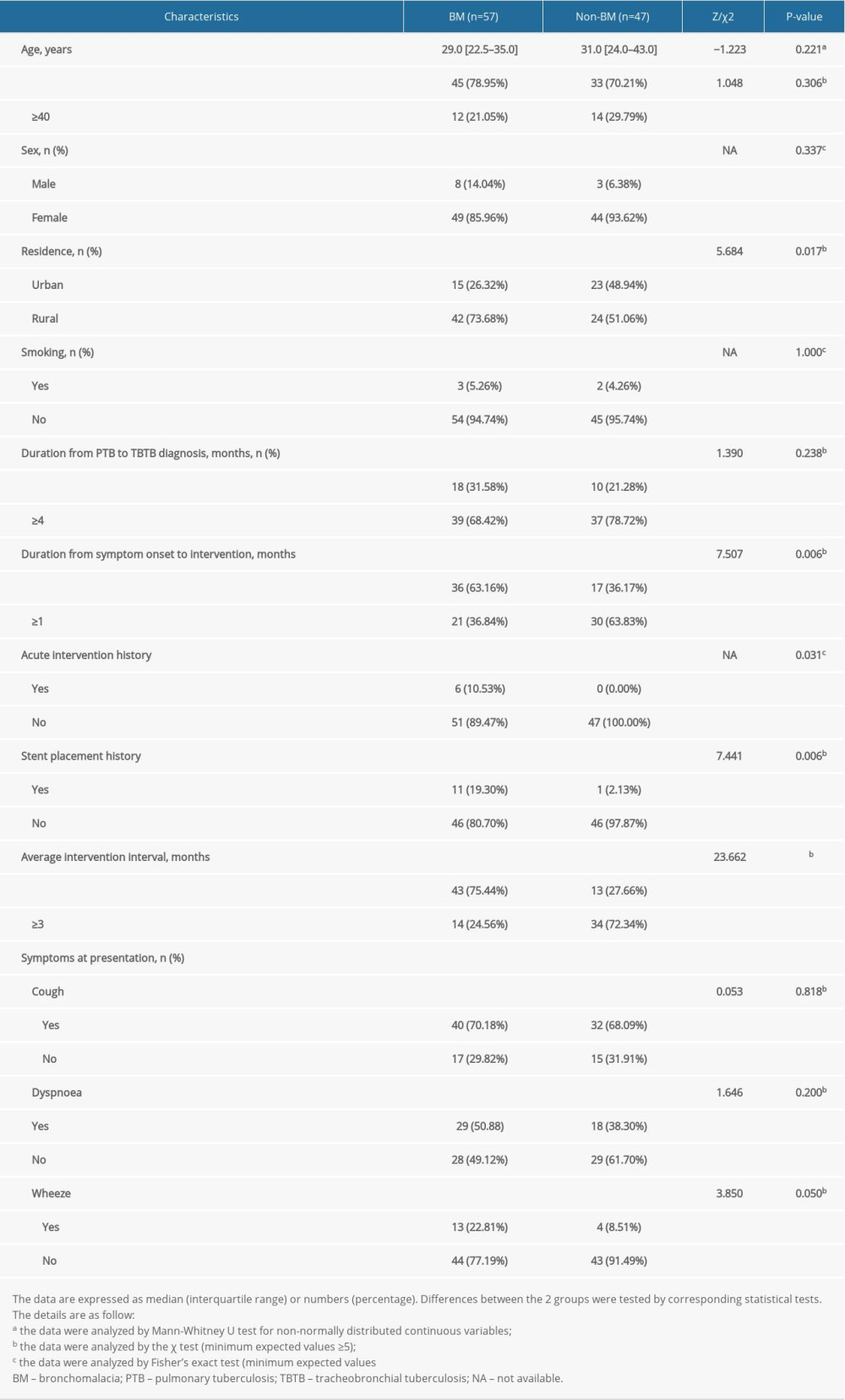 Table 2. Chest CT and bronchoscopy features of study participants.
Table 2. Chest CT and bronchoscopy features of study participants.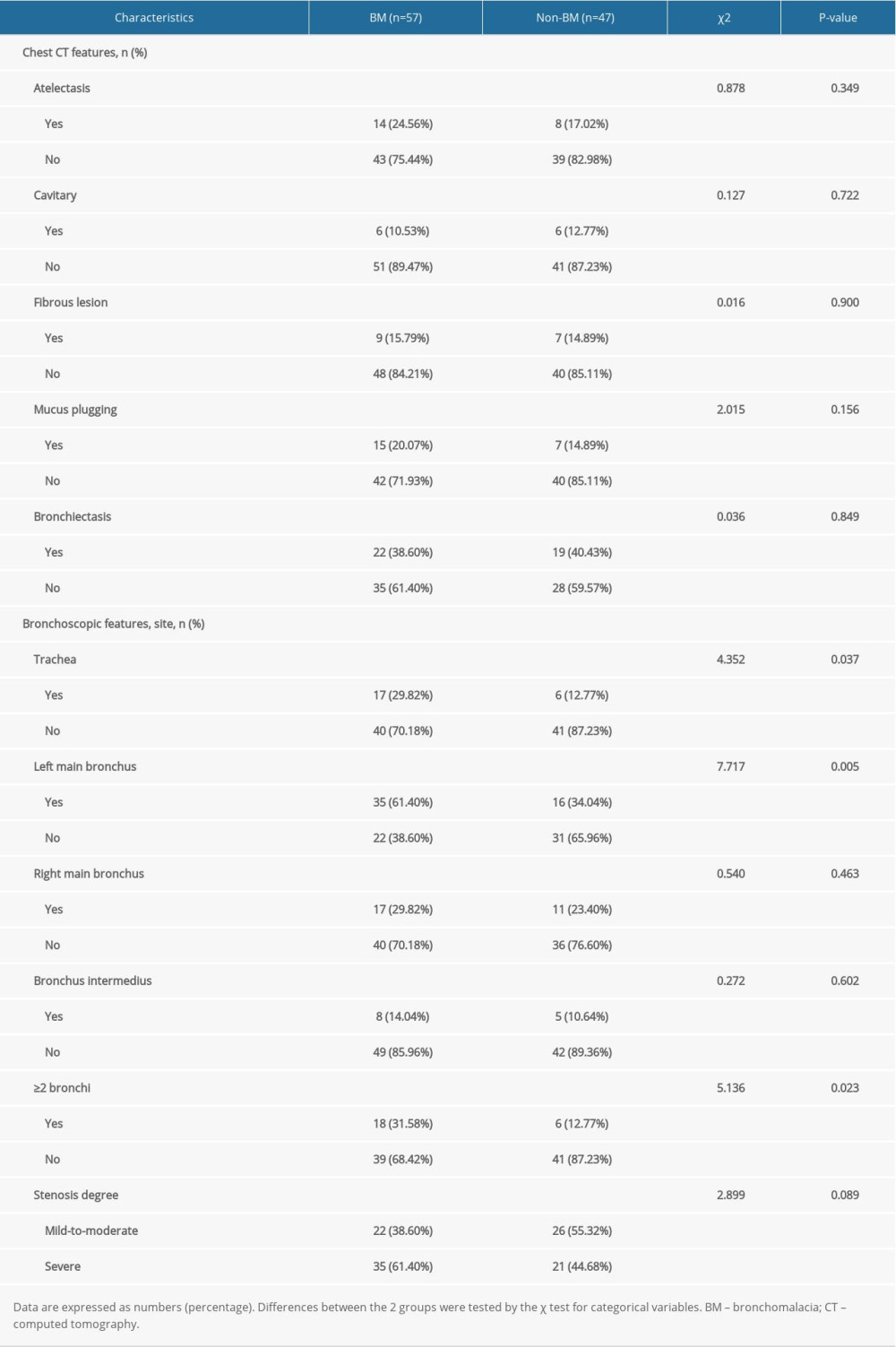 Table 3. Laboratory examination of the study participants.
Table 3. Laboratory examination of the study participants.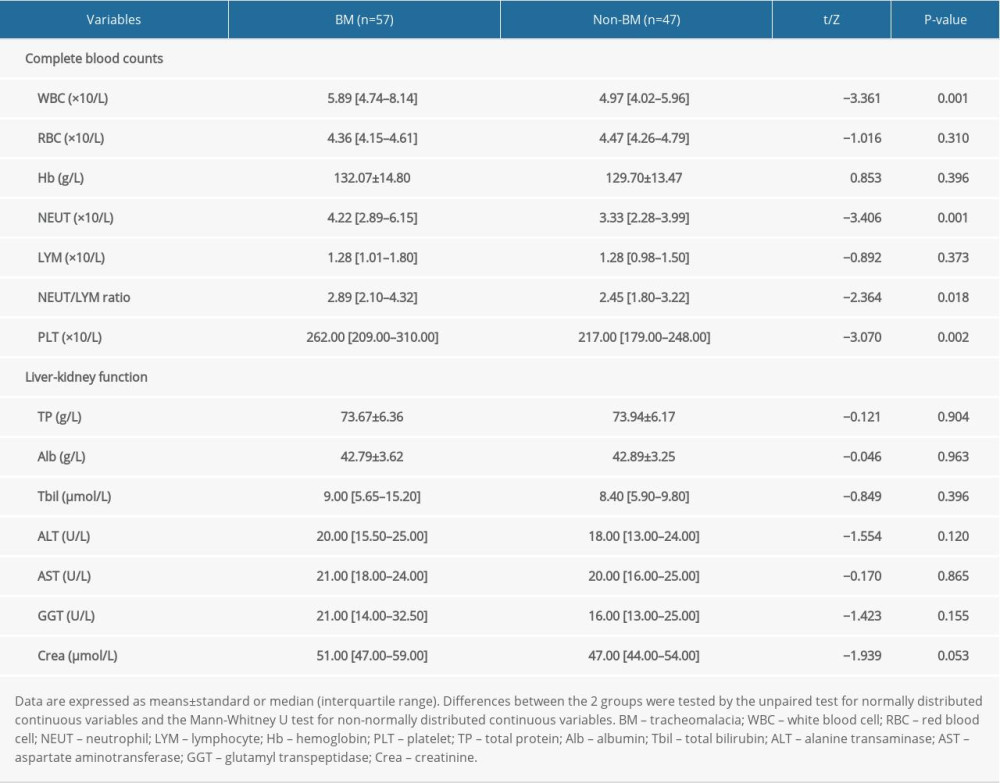 Table 4. Predictors of PTBM.
Table 4. Predictors of PTBM.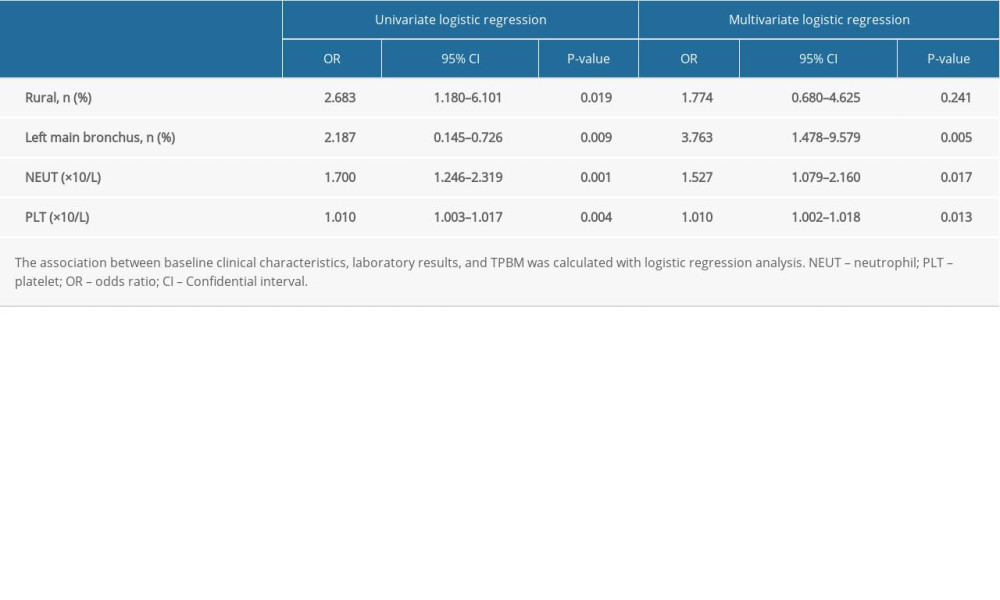 Table 5. Comparison of long-term changes of complete blood counts between the 2 groups.
Table 5. Comparison of long-term changes of complete blood counts between the 2 groups.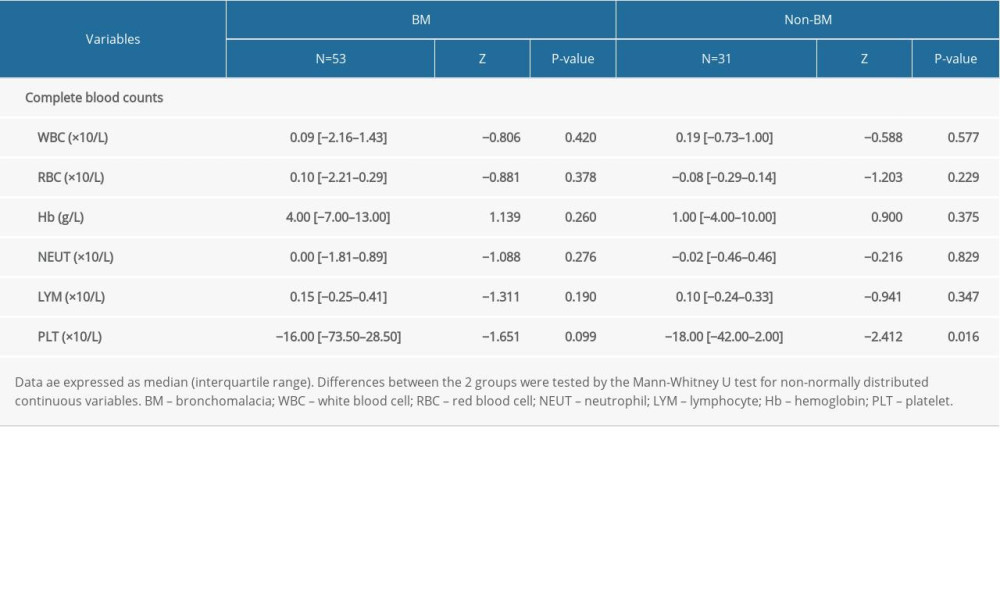
References
1. Hysinger EB, Bates AJ, Higano NS, Ultrashort echo-time MRI for the assessment of tracheomalacia in neonates: Chest, 2020; 157; 595-602
2. Gunatilaka CC, Higano NS, Hysinger EB, Increased work of breathing due to tracheomalacia in neonates: Ann Am Thorac Soc, 2020; 17; 1247-56
3. Hysinger EB, Hart CK, Burg G, Differences in flexible and rigid bronchoscopy for assessment of tracheomalacia: Laryngoscope, 2020; 131; 201-4
4. Dewberry L, Wine T, Prager J, Thoracoscopic posterior tracheopexy is a feasible and effective treatment for tracheomalacia: J Laparoendosc Adv Surg Tech A, 2019; 29; 1228-31
5. Wallis C, Alexopoulou E, Antón-Pacheco JL, ERS statement on tracheomalacia and bronchomalacia in children: Eur Respir J, 2019; 54; 1900382
6. Xu S, Zhu J, Zhao G, Li S, Tracheal suspension with autogenous rib cartilage in a patient with severe tracheomalacia: J Cardiothorac Surg, 2019; 14; 21
7. Brewis C, Pracy JP, Albert DM, Localized tracheomalacia as a complication of the Cole tracheal tube: Paediatr Anaesth, 1999; 9; 531-33
8. Parshin VD, Koroleva IM, Mishchenko MADiagnosis and treatment of acquired tracheomalacia in patients with cicatricial tracheal stenosis: Khirurgiia (Mosk), 2016(8); 73-82 [in Russian]
9. Keng LT, Chang CJ, All that wheezes is not asthma: Adult tracheomalacia resulting from innominate artery compression: Postgrad Med J, 2017; 93; 54-55
10. Kamran A, Friedman KG, Jennings RW, Aortic uncrossing and tracheobronchopexy corrects tracheal compression and tracheobronchomalacia associated with circumflex aortic arch: J Thorac Cardiovasc Surg, 2020; 160; 796-804
11. Paul M, Kannaujia A, Chatterjee A, Serial fiber optic bronchoscopy (FOB) to predict the need of tracheostomy in tracheomalacia after thyroidectomy in long standing goiter: J Clin Anesth, 2018; 47; 9-10
12. De Pieri C, Cogo P, Barbato A, Tracheomalacia due to esophageal achalasia: Arch Bronconeumol, 2017; 53; 78-79
13. McDermott S, Barry SC, Judge EP, Tracheomalacia in adults with cystic fibrosis: Determination of prevalence and severity with dynamic cine CT: Radiology, 2009; 252; 577-86
14. Kandaswamy C, Balasubramanian V, Review of adult tracheomalacia and its relationship with chronic obstructive pulmonary disease: Curr Opin Pulm Med, 2009; 15; 113-19
15. Lee KCH, Tan S, Goh JK, Long-term outcomes of tracheobronchial stenosis due to tuberculosis (TSTB) in symptomatic patients: Airway intervention vs. conservative management: J Thorac Dis, 2020; 12; 3640-50
16. Pathak V, Shepherd RW, Shojaee S, Tracheobronchial tuberculosis: J Thorac Dis, 2016; 8; 3818-25
17. Meghji J, Lesosky M, Joekes E, Patient outcomes associated with post-tuberculosis lung damage in Malawi: A prospective cohort study: Thorax, 2020; 75; 269-78
18. Allwood B, van der Zalm M, Makanda G, The long shadow post-tuberculosis: Lancet Infect Dis, 2019; 19; 1170-71
19. Heimendinger E, Klotz G, Mounier-Kuhn PTracheal stenosis and tracheomalacia in a fibrous tuberculosis patient: Ann Otolaryngol, 1956; 73; 709-13 [in French]
20. Chung HS, Lee JH, Bronchoscopic assessment of the evolution of endobronchial tuberculosis: Chest, 2000; 117; 385-92
21. Low SY, Hsu A, Eng P, Interventional bronchoscopy for tuberculous tracheobronchial stenosis: Eur Respir J, 2004; 24; 345-47
22. Su Z, Cheng Y, Wu Z, Incidence and predictors of tracheobronchial tuberculosis in pulmonary tuberculosis: A multicentre, large-scale and prospective study in southern China: Respiration, 2019; 97; 153-59
23. Wang L, Liu J, Chin DP, Progress in tuberculosis control and the evolving public-health system in China: Lancet, 2007; 369; 691-96
24. Wang L, Zhang H, Ruan Y, Tuberculosis prevalence in China, 1990–2010; A longitudinal analysis of national survey data: Lancet, 2014; 383; 2057-64
25. Xin H, Zhang H, Liu J, Mycobacterium tuberculosis infection among the elderly in 20 486 rural residents aged 50–70 years in Zhongmu county, China: Clin Microbiol Infect, 2019; 25; 1120-26
26. Pellegrini JM, Sabbione F, Morelli MP, Neutrophil autophagy during human active tuberculosis is modulated by SLAMF1: Autophagy, 2020 [Online ahead of print]
27. Soehnlein O, Steffens S, Hidalgo A, Neutrophils as protagonists and targets in chronic inflammation: Nat Rev Immunol, 2017; 17; 248-61
28. Muefong CN, Sutherland JS, Neutrophils in tuberculosis-associated inflammation and lung pathology: Front Immunol, 2020; 11; 962
29. Bickett TE, McLean J, Creissen E, Characterizing the BCG induced macrophage and neutrophil mechanisms for defense against mycobacterium tuberculosis: Front Immunol, 2020; 11; 1202
30. Thieblemont N, Wright HL, Edwards SW, Human neutrophils in auto-immunity: Semin Immunol, 2016; 28; 159-73
31. Xu Y, Zhang Q, Zhao Y, The functional diversity of neutrophils and clustered polarization of immunity: Cell Mol Immunol, 2020; 17; 1212-14
32. Demirin H, Ozhan H, Ucgun T, Normal range of mean platelet volume in healthy subjects: Insight from a large epidemiologic study: Thromb Res, 2011; 128; 358-60
33. Chen Y, Zhong H, Zhao Y, Role of platelet biomarkers in inflammatory response: Biomark Res, 2020; 8; 28
34. Cognasse F, Laradi S, Berthelot P, Platelet inflammatory response to stress: Front Immunol, 2019; 10; 1478
35. Adatia K, Farag MF, Gue YX, Relationship of platelet reactivity and inflammatory markers to recurrent adverse events in patients with ST-elevation myocardial infarction: Thromb Haemost, 2019; 119; 1785-94
36. Middleton EA, Weyrich AS, Zimmerman GA, Platelets in pulmonary immune responses and inflammatory lung diseases: Physiol Rev, 2016; 96; 1211-59
37. Morrell CN, Pariser DN, Hilt ZT, The platelet napoleon complex-small cells, but big immune regulatory functions: Annu Rev Immunol, 2019; 37; 125-44
38. Büyükaşik Y, Soylu B, Soylu AR, In vivo platelet and T-lymphocyte activities during pulmonary tuberculosis: Eur Respir J, 1998; 12; 1375-79
39. Meyer A, Wang W, Qu J, Platelet TGF-β1 contributions to plasma TGF-β1, cardiac fibrosis, and systolic dysfunction in a mouse model of pressure overload: Blood, 2012; 119; 1064-74
40. Li H, Hicks JJ, Wang , Customized platelet-rich plasma with transforming growth factor β1 neutralization antibody to reduce fibrosis in skeletal muscle: Biomaterials, 2016; 87; 147-56
41. Forcina L, Miano C, Scicchitano BM, Signals from the Niche: Insights into the role of IGF-1 and IL-6 in modulating skeletal muscle fibrosis: Cells, 2019; 8(3); 232
42. Andrade D, Oliveira G, Menezes L, Insulin-like growth factor-1 short-period therapy improves cardiomyopathy stimulating cardiac progenitor cells survival in obese mice: Nutr Metab Cardiovasc Dis, 2020; 30; 151-61
43. Squecco R, Chellini F, Idrizaj E, Platelet-rich plasma modulates gap junction functionality and connexin 43 and 26 expression during TGF-β1-induced fibroblast to myofibroblast transition: clues for counteracting fibrosis: Cells, 2020; 9; 1199
44. Wang T, Zhang J, Qiu XJ, Scarring airway stenosis in Chinese adults: Characteristics and interventional bronchoscopy treatment: Chin Med J (Engl), 2018; 131; 276-81
45. Khvilivitzky K, Trivedi PN, McFadden PM, Tuberculous tracheobronchial stenosis: Avoiding resection-when less is more: J Thorac Dis, 2017; 9; E779-82
Figures
 Figure 1. Chest HRCT and bronchoscopy features of patients with PTBMChest HRCT showing left main bronchus stenosis lesion (A, green arrow) due to TBTB. Diagnostic bronchoscopic appearance of PTBM (B, green arrow) can be observed in the left main bronchus. HRCT – high-resolution computed tomography; PTBM – post-tuberculosis bronchomalacia; TBTB – tracheobronchial tuberculosis.
Figure 1. Chest HRCT and bronchoscopy features of patients with PTBMChest HRCT showing left main bronchus stenosis lesion (A, green arrow) due to TBTB. Diagnostic bronchoscopic appearance of PTBM (B, green arrow) can be observed in the left main bronchus. HRCT – high-resolution computed tomography; PTBM – post-tuberculosis bronchomalacia; TBTB – tracheobronchial tuberculosis. Figure 2. Duration of bronchoscopic intervention therapy in BM and non-BM groupsThe time from onset of bronchoscopic intervention was examined by Kaplan-Meier estimates. Differences between the 2 groups were tested by the log-rank test. * P<0.0001. BM – bronchomalacia; HR – hazard ratio; CI – confidential interval.
Figure 2. Duration of bronchoscopic intervention therapy in BM and non-BM groupsThe time from onset of bronchoscopic intervention was examined by Kaplan-Meier estimates. Differences between the 2 groups were tested by the log-rank test. * P<0.0001. BM – bronchomalacia; HR – hazard ratio; CI – confidential interval. Tables
 Table 1. Clinical characteristics of study participants.
Table 1. Clinical characteristics of study participants. Table 2. Chest CT and bronchoscopy features of study participants.
Table 2. Chest CT and bronchoscopy features of study participants. Table 3. Laboratory examination of the study participants.
Table 3. Laboratory examination of the study participants. Table 4. Predictors of PTBM.
Table 4. Predictors of PTBM. Table 5. Comparison of long-term changes of complete blood counts between the 2 groups.
Table 5. Comparison of long-term changes of complete blood counts between the 2 groups. Table 1. Clinical characteristics of study participants.
Table 1. Clinical characteristics of study participants. Table 2. Chest CT and bronchoscopy features of study participants.
Table 2. Chest CT and bronchoscopy features of study participants. Table 3. Laboratory examination of the study participants.
Table 3. Laboratory examination of the study participants. Table 4. Predictors of PTBM.
Table 4. Predictors of PTBM. Table 5. Comparison of long-term changes of complete blood counts between the 2 groups.
Table 5. Comparison of long-term changes of complete blood counts between the 2 groups. In Press
15 Apr 2024 : Laboratory Research
The Role of Copper-Induced M2 Macrophage Polarization in Protecting Cartilage Matrix in OsteoarthritisMed Sci Monit In Press; DOI: 10.12659/MSM.943738
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








