22 November 2021: Clinical Research
Serum Beta-2 Microglobulin: A Possible Biomarker for Atrial Fibrillation
Bangying Zhang1ABCDEF, Xinpei Chen1BCDE, Xiaofeng Mu1BDE, Enzhao Liu1AB, Tong Liu1BE, Gang Xu1BC, Qiankun Bao1CDE, Guangping Li1AFG*DOI: 10.12659/MSM.932813
Med Sci Monit 2021; 27:e932813
Abstract
BACKGROUND: Atrial fibrillation (AF) is the most common persistent arrhythmia that can cause complications (including stroke). Therefore, its diagnosis and treatment require increased attention. Although beta-2 microglobulin (b2-MG) is a novel marker of cardiovascular disease, its role in AF has not been evaluated.
MATERIAL AND METHODS: We conducted a case-control study with 61 patients who had normal heart rhythm (control group) and 60 patients with AF (research group). We analyzed the serum b2-MG levels in both groups and performed multivariate analysis to assess the correlation between b2-MG and left atrial remodeling. In addition, b2-MG levels were compared between the left atrial blood and peripheral venous blood of another set of 57 patients with AF, who underwent cryoballoon ablation.
RESULTS: There were no statistically significant differences in the baseline characteristics (age, sex, history of hypertension, diabetes mellitus, previous stroke, coronary heart disease, and estimated glomerular filtration rate) of the control and research groups. The left atrial anteroposterior diameters (LAD) and left ventricular end-systolic diameters in the AF group were significantly larger compared to the control group (P<0.01). Serum β2-MG levels in patients with AF were significantly higher (P<0.01) and positively correlated with the LAD (B-coefficient 25.482, 95% CI 14.410~36.554, P<0.01), serum β2-MG levels in the left atrial blood were significantly higher than those in peripheral venous blood (P<0.01), and serum β2-MG levels were an independent predictor of AF.
CONCLUSIONS: With the development of atrial fibrillation, the serum β2-MG levels increase and are closely related to the left atrial remodeling due to AF. Therefore, β2-MG can be an effective biomarker for predicting AF.
Keywords: Arrhythmias, Cardiac, Atrial Fibrillation, Cardiology, Case-Control Studies, Female, Heart Atria, Humans, Male, Sensitivity and Specificity, beta 2-Microglobulin
Background
Atrial fibrillation (AF) is one of the most common arrhythmias. It refers to the loss of normal and ordered atrial electrical activity, which is replaced by disorderly fibrillation waves [1]. The incidence and prevalence of AF are increasing globally. The Framingham Heart Study demonstrated that the prevalence of AF has tripled in the last 50 years [2,3]. In addition, future projections indicate that the absolute burden of AF could increase by >60% by 2050 [4]. AF can cause serious complications (including heart failure and arterial embolism) and the most common complication is cardiogenic stroke [5–7]. However, patients with AF lack its typical clinical symptoms and about 15% to 30% of AF patients are asymptomatic [8]. Many patients fail to receive effective treatment and have complications as an AF diagnosis can be easily missed or misdiagnosed [9]. Therefore, the early and timely diagnosis of AF is particularly important for an improved prognosis.
Previous studies have shown that the pathogenesis of AF is related to myocardial strain, myocardial fibrosis, and inflammation [10,11]. Over the past decade, there is growing evidence of the use of the natriuretic peptide system in AF, which includes brain natriuretic peptide (BNP) and N-terminal pro-B-type natriuretic peptide (NT-proBNP). A cohort study showed that patients with elevated BNP levels are at greater risk of progression to persistent or permanent forms of AF. In addition, BNP and NT-proBNP are associated with AF incidence, post-operative AF incidence, and prognosis in AF [12,13]. Biomarkers that suggest myocardial fibrosis (including soluble suppressor of tumorigenicity 2 and galectin-3 [gal-3]) and inflammation indicators (C-reactive protein and interleukin 6) increased in patients with AF [14–18]. However, there is a lack of serum markers for the early diagnosis of AF [19].
Beta-2 microglobulin (β2-MG) is an endogenous low-molecular-weight serum globulin produced by the lymphocytes, platelets, and leukocytes [20]. It is widely distributed in the human body, exists in free form in serum, and its concentration remains relatively stable in the serum of healthy individuals [21]. The serum level of β2-MG is commonly used to assess early renal impairment and as a marker for multiple myeloma [22–24]. In recent years, increasing evidence has shown that β2-MG is associated with the occurrence of coronary heart disease, heart failure, and hypertension and β2-MG levels are positively related to the incidence and mortality of cardiovascular diseases [25–27]. The pathogenesis of AF has not been fully elucidated. Most pathophysiological mechanisms of AF are mainly ectopic excitation and reentry mechanisms, and these changes include ion-channel dysfunction, Ca2+-handling abnormalities, structural remodeling, and autonomic neural dysregulation [1]. Atrial fibrosis can promote atrial arrhythmogenesis, leading to AF with local conduction slowdown and a unidirectional blockade [28–30]. The expression of β2-MG is associated with fibrosis of the kidneys, liver, lungs, and heart. Previous studies have shown that β2-MG is an indicator of the severity of pulmonary fibrosis [31]. In addition, exogenous β2-MG has a profibrotic effect on cardiac fibroblasts [32]. Therefore, we proposed the hypothesis that serum β2-MG is associated with AF. In this study, we determined serum β2-MG levels in patients with AF and analyzed the relationship between serum β2-MG levels and AF.
Material and Methods
STUDY POPULATION:
We conducted a case-control study on patients with or without AF who were hospitalized in the Second Hospital of Tianjin Medical University between December 2018 and December 2019. The study was conducted while the patients were in hospital and patient information was obtained from the hospital’s electronic medical record system (including sex, age, comorbidities, and laboratory test results). Transthoracic echocardiography was performed on the enrolled patients for related parameters, including left atrial anteroposterior diameter, left ventricular end-diastolic diameter, left ventricular end-systolic diameter, interventricular septum thickness, left ventricular posterior wall thickness, right ventricular anterior wall thickness, right ventricular end-diastolic diameter, and left ventricular ejection fraction. These parameters were measured in the parasternal long-axis view using 2-dimensional methods. The inclusion criteria were patients with AF or non-AF who were hospitalized in the Department of Cardiology of the Second Hospital of Tianjin Medical University and patients diagnosed with AF who met the diagnostic criteria of the 2016 Atrial Fibrillation Management Guidelines developed by the European Society of Cardiology and the European Heart Rhythm Association [33]. The exclusion criteria were patients with severe cardiac insufficiency (ejection fraction <40% or New York Heart Failure Classification IV), severe hypertension (3 measurements of systolic blood pressure ≥180 mmHg and/or diastolic blood pressure ≥110 mmHg on different days), acute or chronic autoimmunity or infectious diseases, liver, kidney, gallbladder diseases or abnormal liver and kidney function, other heart diseases (dilated cardiomyopathy, hypertrophic cardiomyopathy, rheumatic heart disease, congenital heart disease, malignant arrhythmia), and systemic blood diseases.
Subsequently, to explore the possible source of serum β2-MG in AF patients, another set of 57 patients with AF who underwent cryoballoon ablation were chosen as the research subjects to determine the β2-MG levels in left atrial blood and peripheral venous blood. The selection criteria for these patients were the same as described above for patients in the control and research groups. In addition, these patients underwent cryoballoon ablation (a treatment for atrial fibrillation). We collected atrial blood samples before the cryoballoon ablation (between September 2019 and January 2021).
All the procedures performed in studies involving human participants were in accordance with the Ethical Standards of the Institutional Research Committee (Medical Ethics Committee of the Second Hospital of Tianjin Medical University, approval number: KY2019K145) and the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from each patient.
BLOOD SAMPLES AND LABORATORY ASSAYS:
A fasting venous blood sample (5 mL) was taken from the patients within 24 h after hospital admission. Blood samples from the left atrial (5 mL) were collected from patients undergoing cryoballoon ablation. The supernatant serum from the blood samples was centrifuged at 3000 g for 5 min to 10 min and frozen at −80°C. The serum β2-MG level was determined by an enzyme-linked immunosorbent assay (ELISA) (R&D Systems, Minneapolis, MN, USA).
STATISTICAL ANALYSIS:
Continuous variables are expressed as means±standard deviations, and independent sample
Results
RELATIONSHIP BETWEEN SERUM β-2 MICROGLOBULIN AND PREVALENCE OF ATRIAL FIBRILLATION:
The baseline characteristics of the control and research groups are shown in Table 1. There were no statistically significant differences in the baseline clinical characteristics (age, sex, history of hypertension, diabetes, previous stroke, coronary heart disease, and eGFR) between the 2 groups. The anteroposterior of the left atrial diameters (LAD) (36.75±4.78 mm and 44.03±5.15 mm) and left ventricular end-systolic diameters (26.03±3.30 mm and 29.35±4.71 mm) in patients with AF were significantly larger compared to the control group (P<0.01) (Table 2). However, there were no significant differences between the 2 groups for the left ventricular end-diastolic diameter, left ventricular ejection fraction, and other parameters from the transthoracic echocardiography (Table 2).
We analyzed the serum β2-MG levels between the 2 groups. These levels in the AF group (1999.68±498.26 ng/mL) were significantly higher than seen in the control group (1567.20±288.21 ng/mL) (P<0.01) (Figure 1). Multivariate analysis was performed to evaluate the correlation between β2-MG levels and left atrial remodeling. The results showed that the increase in the β2-MG levels was significantly correlated with the size of the LAD (P<0.01) and negatively correlated to the eGFR (Table 3). β2-MG showed a moderate predictive ability to diagnose AF according to the area under the ROC curve (AUC=0.80, 95% CI 0.720–0.876, P<0.01) (Figure 2).
DISTRIBUTION OF β2-MICROGLOBULIN IN PATIENTS WITH ATRIAL FIBRILLATION:
In the light of the above results, we promote the hypothesis that the increase of serum β2-MG contributes to the structural remodeling of the left atrial. Therefore, another set of 57 AF patients who underwent cryoballoon ablation were selected as the research subjects and we determined the concentration of β2-MG in their left atrial blood and peripheral venous blood. The clinical characteristics of these patients are shown in Table 4. The mean age of these patients was 64.12±8.97 years, 57.9% were men and 42.1% were women. The CHA2DS2VASc score [34] was 2.21±1.29 points and the LAD was 41.16±4.77 mm. We determined the concentration of β2-MG in the left atrial blood and peripheral venous blood of patients with AF using ELISA. The level of β2-MG in the left atrial blood (2395.96±604.47 ng/mL) was significantly higher than that in the peripheral venous blood (2036.33±614.317 ng/mL) of patients with AF (P<0.01) (Figure 3).
Discussion
LIMITATIONS:
There were several limitations in our research. Firstly, as this is a single-center study, the sample size is small compared to a multicenter study. Secondly, the level of serum β2-MG is affected by many factors, and our research lacks further information on the specific mechanism of β2-MG on AF. Therefore, the use of plasma β2-MG levels to predict the occurrence of AF in the population could be biased. We hope to study a larger population in the future to verify the predictive value of β2-MG in AF.
Conclusions
We demonstrated that serum β2-MG levels increased with the development of atrial fibrillation. The levels of β2-MG are closely related to the left atrial remodeling due to AF. β2-MG could be an effective biomarker for AF prediction. Therefore, in the future, we could diagnose early AF by measuring serum β2-MG levels.
Figures
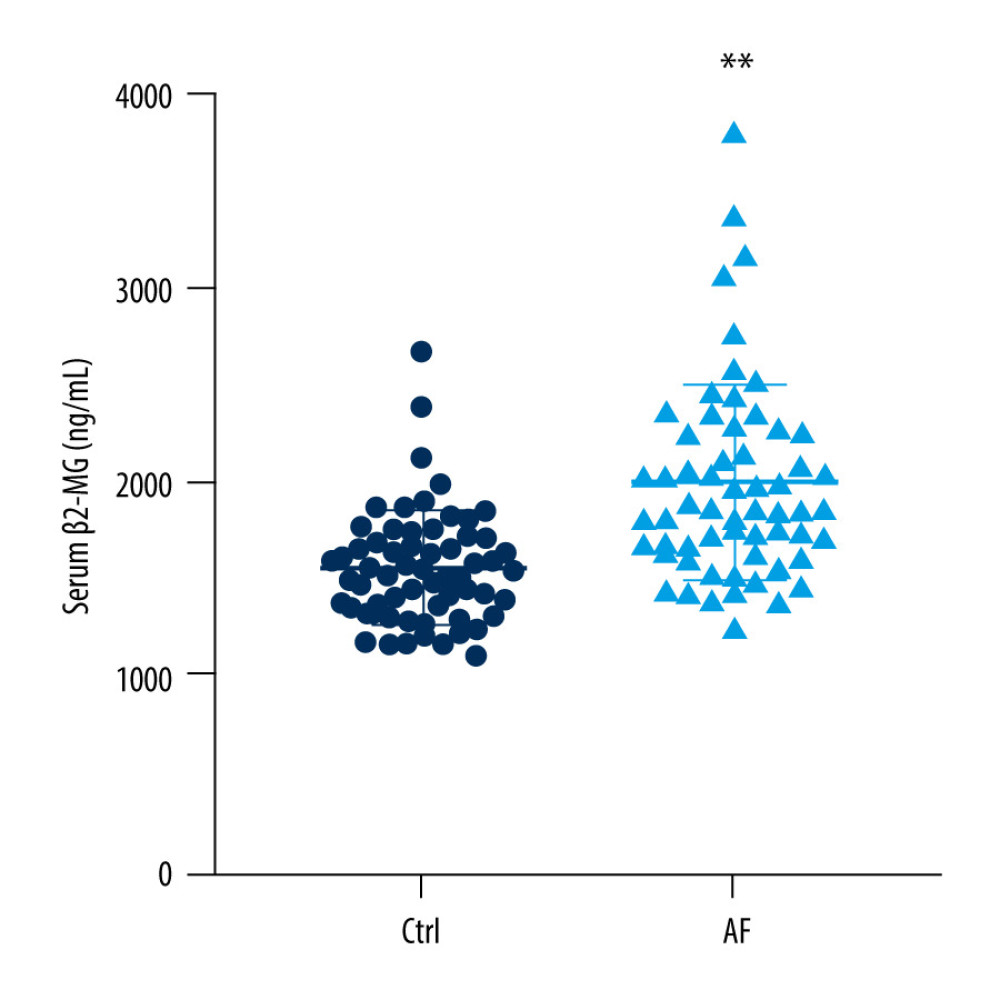 Figure 1. Serum levels of beta-2 microglobulin in the control group and research group. Data are mean±standard deviation and an unpaired 2-tailed t test. ** P<0.01 (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA).
Figure 1. Serum levels of beta-2 microglobulin in the control group and research group. Data are mean±standard deviation and an unpaired 2-tailed t test. ** P<0.01 (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA). 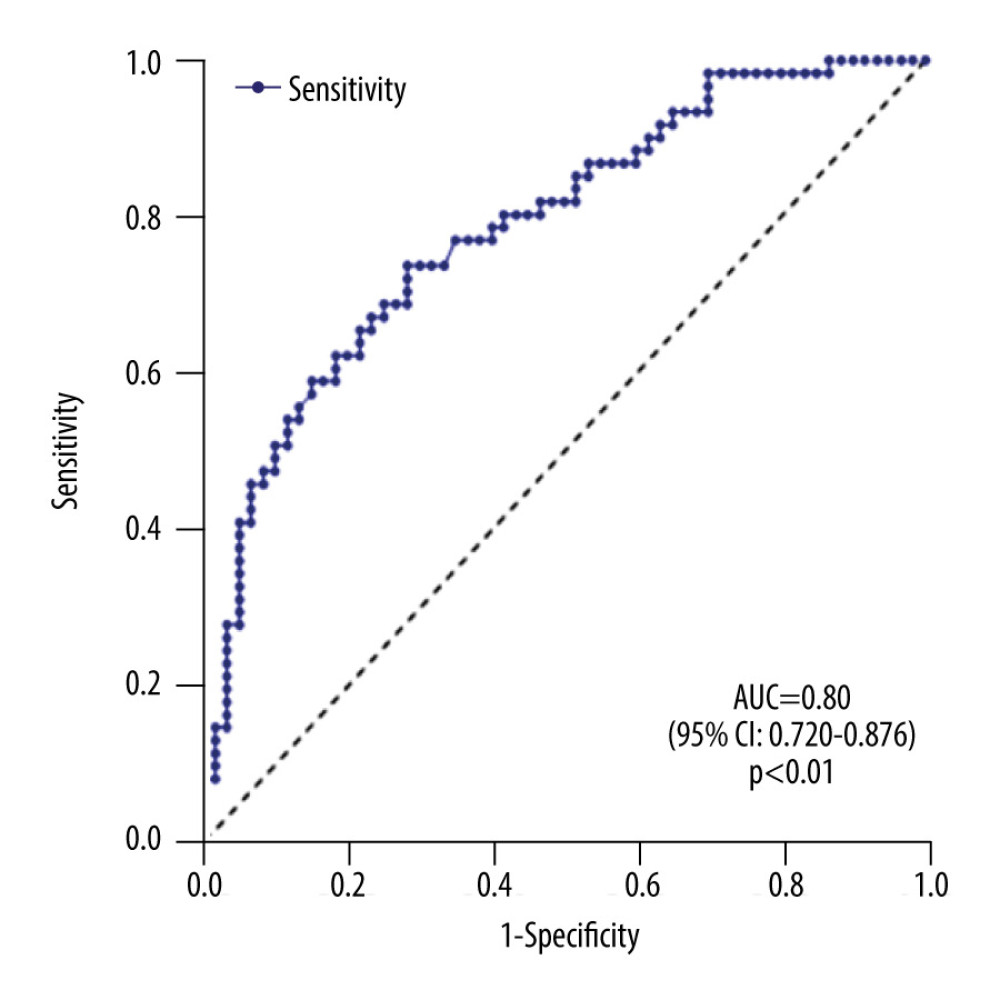 Figure 2. Receiver operating characteristic curve analysis for predictive values of beta-2 microglobulin in diagnosing atrial fibrillation. (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA).
Figure 2. Receiver operating characteristic curve analysis for predictive values of beta-2 microglobulin in diagnosing atrial fibrillation. (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA). 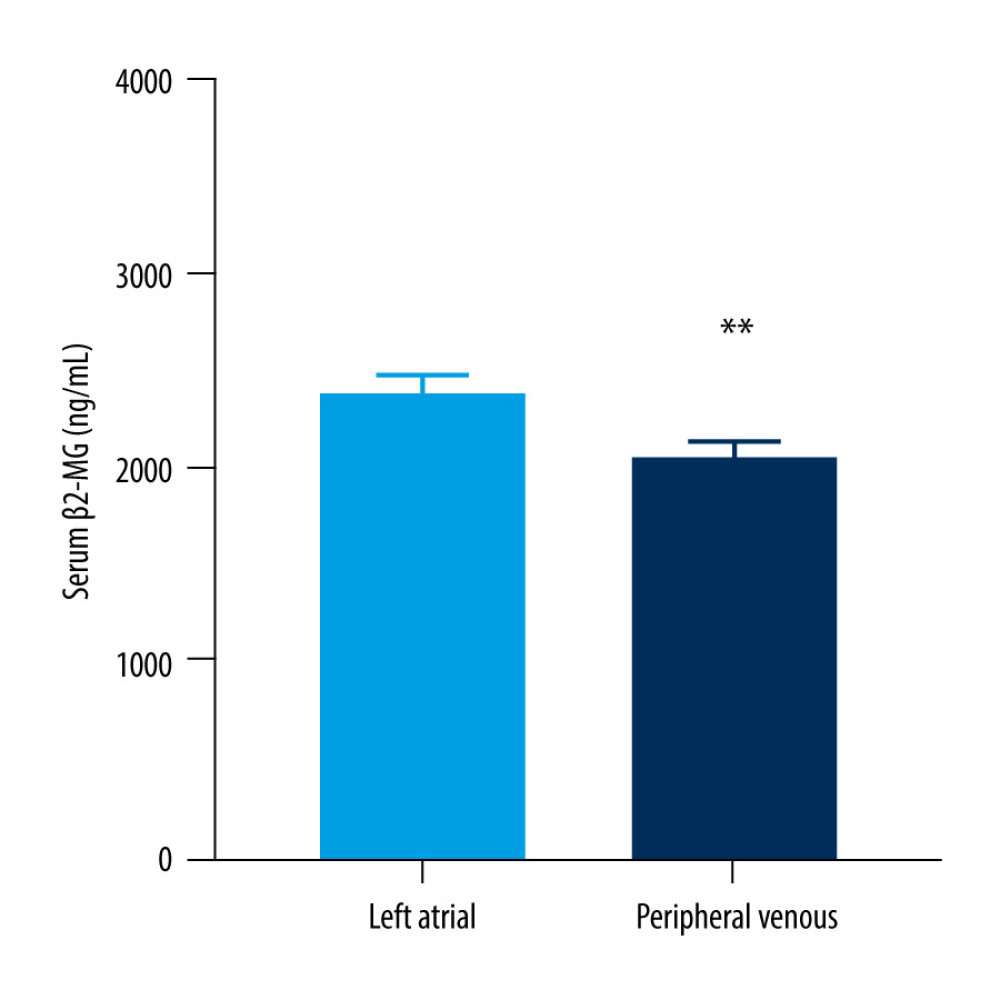 Figure 3. Left atrial and peripheral venous serum levels of beta-2 microglobulin in patients with atrial fibrillation. Data are mean±standard deviation and an unpaired 2-tailed t test. ** P<0.01 (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA).
Figure 3. Left atrial and peripheral venous serum levels of beta-2 microglobulin in patients with atrial fibrillation. Data are mean±standard deviation and an unpaired 2-tailed t test. ** P<0.01 (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA). Tables
Table 1. Baseline characteristics of the study participants.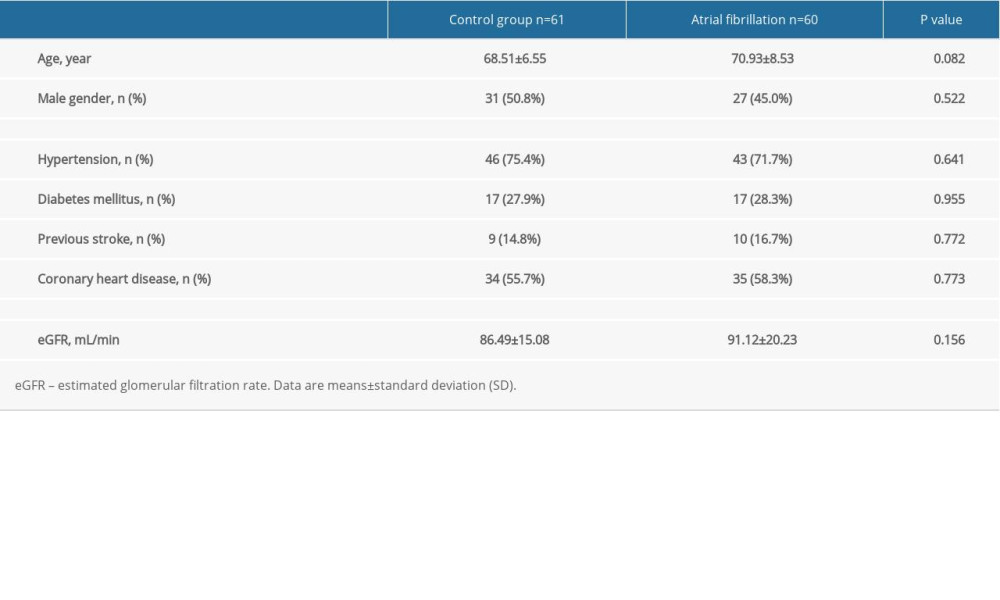 Table 2. Echocardiographic parameters of the study participants.
Table 2. Echocardiographic parameters of the study participants.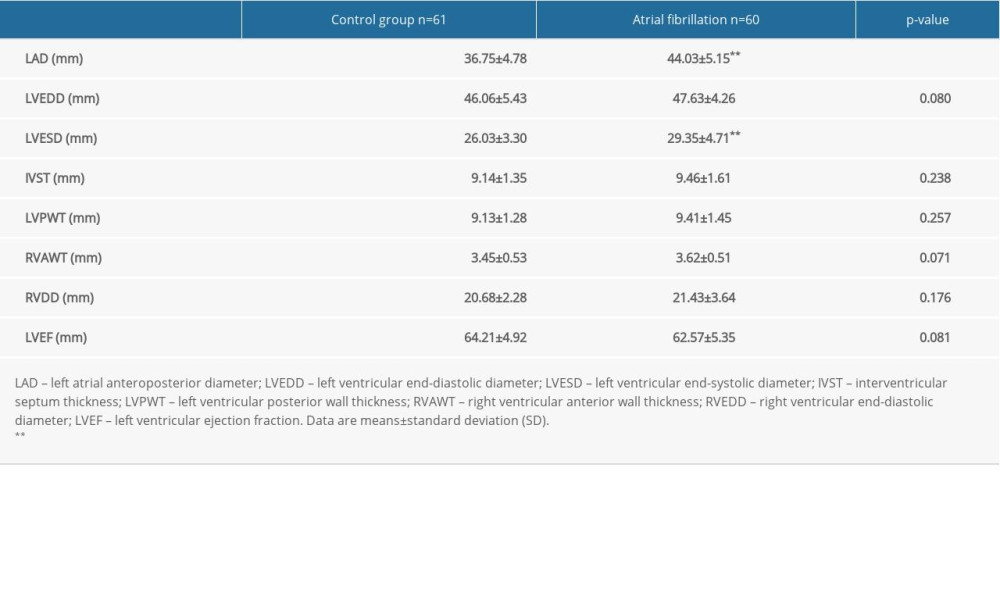 Table 3. Multivariate linear regression analysis for the association between serum beta-2 microglobulin levels and the left atrial diameter in the study participants.
Table 3. Multivariate linear regression analysis for the association between serum beta-2 microglobulin levels and the left atrial diameter in the study participants.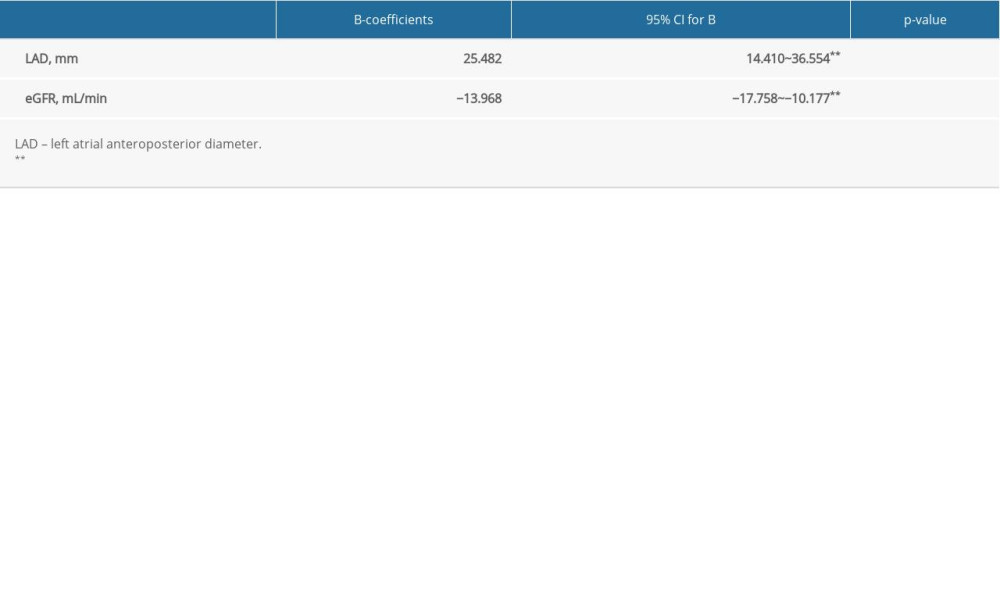 Table 4. Clinical and laboratory characteristics of patients in the research.
Table 4. Clinical and laboratory characteristics of patients in the research.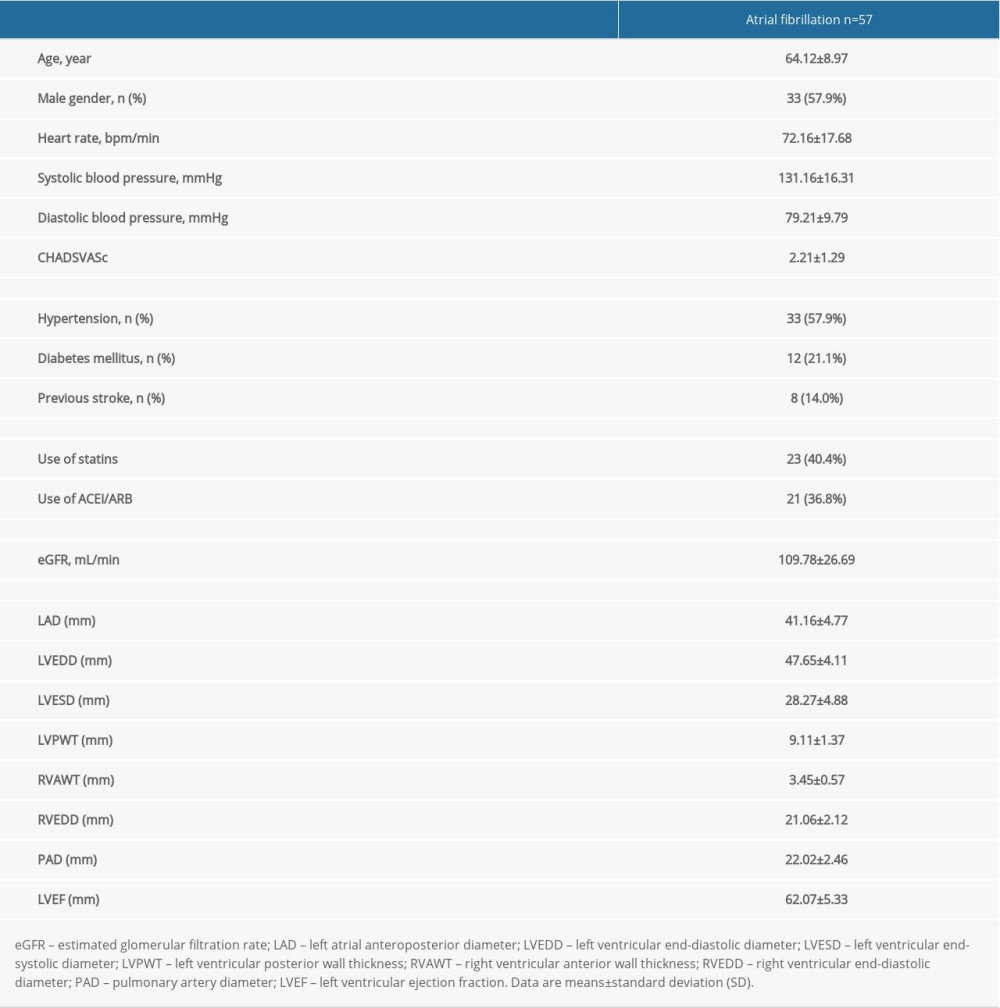
References
1. Andrade J, Khairy P, Dobrev D, The clinical profile and pathophysiology of atrial fibrillation: Relationships among clinical features, epidemiology, and mechanisms: Circ Res, 2014; 114(9); 1453-68
2. Kornej J, Borschel CS, Benjamin EJ: Circ Res, 2020; 127(1); 4-20
3. Staerk L, Sherer JA, Ko D, Atrial fibrillation: Epidemiology, pathophysiology, and clinical outcomes: Circ Res, 2017; 120(9); 1501-17
4. Lippi G, Sanchis-Gomar F, Cervellin G, Global epidemiology of atrial fibrillation: An increasing epidemic and public health challenge: Int J Stroke, 2021; 16(2); 217-21
5. Chen LY, Chung MK, Allen LA, Atrial fibrillation burden: Moving beyond atrial fibrillation as a binary entity: A Scientific Statement from the American Heart Association: Circulation, 2018; 137(20); e623-44
6. Lin HJ, Wolf PA, Benjamin EJ, Newly diagnosed atrial fibrillation and acute stroke. The Framingham Study: Stroke, 1995; 26(9); 1527-30
7. Pistoia F, Sacco S, Tiseo C, The epidemiology of atrial fibrillation and stroke: Cardiol Clin, 2016; 34(2); 255-68
8. Rienstra M, Lubitz SA, Mahida S, Symptoms and functional status of patients with atrial fibrillation: State of the art and future research opportunities: Circulation, 2012; 125(23); 2933-43
9. Rogers PA, Bernard ML, Madias C, Current evidence-based understanding of the epidemiology, prevention, and treatment of atrial fibrillation: Curr Probl Cardiol, 2018; 43(6); 241-83
10. Wijesurendra RS, Casadei B, Mechanisms of atrial fibrillation: Heart, 2019; 105(24); 1860-67
11. Nattel S, Molecular and cellular mechanisms of atrial fibrosis in atrial fibrillation: JACC Clin Electrophysiol, 2017; 3(5); 425-35
12. Chang KW, Hsu JC, Toomu A, Clinical applications of biomarkers in atrial fibrillation: Am J Med, 2017; 130(12); 1351-57
13. Kumarathurai P, Mouridsen MR, Mattsson N, Atrial ectopy and N-terminal pro-B-type natriuretic peptide as predictors of atrial fibrillation: A population-based cohort study: Europace, 2017; 19(3); 364-70
14. Wang Z, Cheng L, Zhang J, Serum-soluble ST2 is a novel biomarker for evaluating left atrial low-voltage zone in paroxysmal atrial fibrillation: Med Sci Monit, 2020; 26; e926221
15. Xiao M, Zhang M, Bie M, Galectin-3 induces atrial fibrosis by activating the TGF-beta1/Smad pathway in patients with atrial fibrillation: Cardiology, 2020; 145(7); 446-55
16. Clementy N, Piver E, Bisson A, Galectin-3 in atrial fibrillation: Mechanisms and therapeutic implications: Int J Mol Sci, 2018; 19(4); 976
17. Lewicka E, Dudzinska-Gehrmann J, Dabrowska-Kugacka A, Neopterin and interleukin-6 as predictors of recurrent atrial fibrillation: Anatol J Cardiol, 2016; 16(8); 563-71
18. Aulin J, Hijazi Z, Siegbahn A, Serial measurement of interleukin-6 and risk of mortality in anticoagulated patients with atrial fibrillation: Insights from ARISTOTLE and RE-LY trials: J Thromb Haemost, 2020; 18(9); 2287-95
19. Oikonomou E, Zografos T, Papamikroulis GA, Biomarkers in atrial fibrillation and heart failure: Curr Med Chem, 2019; 26(5); 873-87
20. Vincent C, Revillard JP, Beta-2-microglobulin and HLA-related glycoproteins in human urine and serum: Contrib Nephrol, 1981; 26; 66-88
21. Drueke TB, Massy ZA, Beta2-microglobulin: Semin Dial, 2009; 22(4); 378-80
22. Maaroufi A, Khadem-Ansari MH, Khalkhali HR, Serum levels of bone sialoprotein, osteopontin, and beta2-microglobulin in stage I of multiple myeloma: J Cancer Res Ther, 2020; 16(1); 98-101
23. Michels TC, Petersen KE, Multiple myeloma: Diagnosis and treatment: Am Fam Physician, 2017; 95(6); 373-83
24. Stakhova T, Shcherbak AV, Kozlovskaia LV, Clinical value of the determination of markers for endothelial dysfunction (endothelin-1, microalbuminuria) and tubulointerstitial tissue lesion (beta2-microglobulin, monocyte chemotactic protein-1) in hypertensive patients with uric acid metabolic disorders: Ter Arkh, 2014; 86(6); 45-51
25. You L, Xie R, Hu H, High levels of serum beta2-microglobulin predict severity of coronary artery disease: BMC Cardiovasc Disord, 2017; 17(1); 71
26. Brioschi M, Gianazza E, Agostoni P, Multiplexed MRM-based proteomics identified multiple biomarkers of disease severity in human heart failure: Int J Mol Sci, 2021; 22(2); 838
27. Shi F, Sun L, Kaptoge S, Association of beta-2-microglobulin and cardiovascular events and mortality: A systematic review and meta-analysis: Atherosclerosis, 2021; 320; 70-78
28. Li D, Fareh S, Leung TK, Promotion of atrial fibrillation by heart failure in dogs: Atrial remodeling of a different sort: Circulation, 1999; 100(1); 87-95
29. Nattel S, Dobrev D, The multidimensional role of calcium in atrial fibrillation pathophysiology: Mechanistic insights and therapeutic opportunities: Eur Heart J, 2012; 33(15); 1870-77
30. Yue L, Xie J, Nattel S, Molecular determinants of cardiac fibroblast electrical function and therapeutic implications for atrial fibrillation: Cardiovasc Res, 2011; 89(4); 744-53
31. Wu Z, Yan M, Zhang M, Beta2-microglobulin as a biomarker of pulmonary fibrosis development in COPD patients: Aging (Albany NY), 2020; 13(1); 1251-63
32. Li Y, Zhang X, Li L, Mechanical stresses induce paracrine beta-2 microglobulin from cardiomyocytes to activate cardiac fibroblasts through epidermal growth factor receptor: Clin Sci (Lond), 2018; 132(16); 1855-74
33. Kirchhof P, Benussi S, Kotecha D, 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS: Eur Heart J, 2016; 37(38); 2893-962
34. Camm AJ, Kirchhof P, Lip GY, Guidelines for the management of atrial fibrillation: The Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC): Europace, 2010; 12(10); 1360-420
35. Huang J, Xiang Y, Zhang H, Plasma level of interferon-gamma predicts the prognosis in patients with new-onset atrial fibrillation: Heart Lung Circ, 2020; 29(7); e168-76
36. Hussein AA, Saliba WI, Martin DO, Plasma B-type natriuretic peptide levels and recurrent arrhythmia after successful ablation of lone atrial fibrillation: Circulation, 2011; 123(19); 2077-82
37. Tang Z, Zeng L, Lin Y, Circulating Galectin-3 is associated with left atrial appendage remodelling and thrombus formation in patients with atrial fibrillation: Heart Lung Circ, 2019; 28(6); 923-31
38. Boyer MJ, Eguchi S, Novel role for cardiac myocyte-derived beta-2 microglobulin in mediating cardiac fibrosis: Clin Sci (Lond), 2018; 132(19); 2117-20
Figures
 Figure 1. Serum levels of beta-2 microglobulin in the control group and research group. Data are mean±standard deviation and an unpaired 2-tailed t test. ** P<0.01 (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA).
Figure 1. Serum levels of beta-2 microglobulin in the control group and research group. Data are mean±standard deviation and an unpaired 2-tailed t test. ** P<0.01 (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA). Figure 2. Receiver operating characteristic curve analysis for predictive values of beta-2 microglobulin in diagnosing atrial fibrillation. (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA).
Figure 2. Receiver operating characteristic curve analysis for predictive values of beta-2 microglobulin in diagnosing atrial fibrillation. (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA). Figure 3. Left atrial and peripheral venous serum levels of beta-2 microglobulin in patients with atrial fibrillation. Data are mean±standard deviation and an unpaired 2-tailed t test. ** P<0.01 (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA).
Figure 3. Left atrial and peripheral venous serum levels of beta-2 microglobulin in patients with atrial fibrillation. Data are mean±standard deviation and an unpaired 2-tailed t test. ** P<0.01 (GraphPad Prism 8.0.1, GraphPad Software, Inc., San Diego, CA, USA). Tables
 Table 1. Baseline characteristics of the study participants.
Table 1. Baseline characteristics of the study participants. Table 2. Echocardiographic parameters of the study participants.
Table 2. Echocardiographic parameters of the study participants. Table 3. Multivariate linear regression analysis for the association between serum beta-2 microglobulin levels and the left atrial diameter in the study participants.
Table 3. Multivariate linear regression analysis for the association between serum beta-2 microglobulin levels and the left atrial diameter in the study participants. Table 4. Clinical and laboratory characteristics of patients in the research.
Table 4. Clinical and laboratory characteristics of patients in the research. Table 1. Baseline characteristics of the study participants.
Table 1. Baseline characteristics of the study participants. Table 2. Echocardiographic parameters of the study participants.
Table 2. Echocardiographic parameters of the study participants. Table 3. Multivariate linear regression analysis for the association between serum beta-2 microglobulin levels and the left atrial diameter in the study participants.
Table 3. Multivariate linear regression analysis for the association between serum beta-2 microglobulin levels and the left atrial diameter in the study participants. Table 4. Clinical and laboratory characteristics of patients in the research.
Table 4. Clinical and laboratory characteristics of patients in the research. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








