25 August 2021: Clinical Research
A Comparative Study of Early Mucosal Healing Following Hot Polypectomy and Cold Polypectomy
Hirofumi Fukushima1ABCDEF, Naoto Sakamoto1ABF, Tomoyoshi Shibuya1BCDEF*, Keiichi Haga1DE, Osamu Nomura1DE, Takashi MurakamiDOI: 10.12659/MSM.933043
Med Sci Monit 2021; 27:e933043
Abstract
BACKGROUND: Cold polypectomy (CP) and hot polypectomy (HP) are both accepted methods for polypectomy. In recent years, the use of CP has increased for reasons of safety. However, there have been few investigations of conditions at follow-up early after resection. This prospective study from a single center aimed to compare colonic mucosal healing at 1 week following HP vs CP of benign colonic polyps <10 mm in diameter.
MATERIAL AND METHODS: Six patients with a total of 52 lesions under 10 mm in size were randomized to either the HP group (n=25) or CP group (n=27) using information in opaque envelopes. One week after endoscopic treatment, the site of treatment was evaluated using colonoscopy. We assessed the mean tumor size, ulcer diameter, exposed blood vessels, residual lesion, and complications.
RESULTS: Mean tumor size did not differ between the 2 groups (CP vs HP: 5.41 mm vs 5.68 mm). The CP group had a smaller ulcer base diameter (2.70 mm vs 4.84 mm; P<0.05) and fewer exposed blood vessels than the HP group (3.7% vs 36.0%; P<0.05). One residual lesion was found in the CP group. No patients experienced delayed perforation or post-polypectomy bleeding.
CONCLUSIONS: Our study findings showed that at 1-week follow-up, cold polypectomy resulted in improved colonic mucosal healing, with a smaller ulcer diameter and fewer blood vessels, when compared with hot polypectomy.
Keywords: Colorectal Neoplasms, Endoscopes, Gastrointestinal, Wound Healing, colonic polyps, Aged, 80 and over, Colonoscopy, endoscopic mucosal resection, Follow-Up Studies, Mucous Membrane, Postoperative Hemorrhage, Prospective Studies, young adult
Background
Colonoscopic resection of polyps is useful to prevent colorectal cancer and has been shown to reduce the colorectal cancer mortality rate [1,2]. Both snare polypectomy and forceps polypectomy were shown to be effective for removing diminutive colorectal polyps (DCPs) and are further classified into hot polypectomy (HP) and cold polypectomy (CP). The basic difference is the use of electrocautery by a high-frequency generator in HP, which can minimize immediate procedural bleeding by coagulation [3,4]. HP can damage deeper vessels, with increased risk of delayed post-polypectomy bleeding (PPB) or perforation [3,5–8].
Over recent years, there has been a tendency to transition from electrocautery to non-electrocautery for the complete removal of DCPs. As CP presents a low risk of PPB, it can be performed safely even in patients taking antithrombotic drugs [9–12]. CP is considered safer than HP, and its use has greatly increased in recent years due to the development of specific CP snares and forceps [13–17]. Cold forceps polypectomy (CFP) uses jumbo forceps, which are larger than conventional forceps. It was reported that jumbo forceps are more effective for the complete resection of DCPs than conventional forceps [13,18]. Jumbo forceps polypectomy (JFP) is especially effective for DCPs 4 to 5 mm in size [12]. Recent European Society for Gastrointestinal Endoscopy (ESGE) Clinical Guidelines recommend CFP as the preferred technique to remove polyps ≤5 mm [19]. However, local recurrence is a major problem after polypectomy [12,20]. To address that issue, indications for CP are limited to benign lesions <10 mm, such as adenomas and sessile-serrated polyps (SSP), because of the risk of cancerous and malignant lesions [9,10,21].
A recent retrospective case-control study also provided support that CP could effectively reduce the risk of PPB [5,6,20]. For this reason, CP has increasingly been used over HP. However, the early condition at follow-up after resection as been little investigated [22]. Therefore, this prospective study from a single center aimed to compare colonic mucosal healing following hot polypectomy and cold polypectomy in benign colonic polyps of less than 10 mm in diameter.
Material and Methods
STATEMENT OF ETHICS:
Prior to the initial diagnostic colonoscopy, each patient provided written informed consent about the possibility of subsequent enrollment in a research study. The study protocol was approved by the Institutional Review Board of the Ethics Committee of the Juntendo University School of Medicine before initiating this study (IRB number: 14-050). The ethics standards of the Helsinki Declaration of 2013 were followed.
PATIENTS AND METHODS:
We prospectively examined data on patients who underwent endoscopic polyp resection in our institution from October 2014 to December 2015. Lesions that were followed up by colonoscopy 1 week after treatment were examined. This analysis included men and women 20 to 80 years of age who provided informed consent for participation in the study. The target number of registered patients was set at 15 cases, with a recruitment period of about 1 year. Colonoscopy was indicated based on positive fecal immunohistochemical test results, surveillance, or resection of polyps. Exclusion criteria were: use of an anticoagulant or antiplatelet therapy according to Japanese guidelines or by patients with an existing hemorrhagic diathesis, pregnancy, history of previous abdominal surgical operation, and overweight (body weight over 100 kg). We tried to minimize the possibility of response bias much as possible. Indications for resection included suspected benign tumors such as adenoma and SSP of <10 mm diagnosed according to surface patterns by narrow band imaging (NBI) magnification [23–26]. Exclusion criteria of colorectal lesion were: (1) pedunculated polyps; (2) polyps >10 mm; (3) history of inflammatory bowel disease; (4) history of familial adenomatous polyposis; or (5) apparent colorectal cancer diagnosed by colonoscopic observation. Adenomas were diagnosed using magnifying endoscopy findings and vessel and surface patterns (JNET [Japan NBI Expert Team] classification Type 2A), and either an enlarged crypt opening or thick and branched vessels were used to identify SSP [23,24]. Within 1 week after polypectomy, all patients visited our hospital to be informed of the histology results and to undergo colonoscopy using NBI magnification to observe the site of the resection. Judgments of the diameter of the ulcer base, exposed blood vessels, and presence of a residual lesion were made by both an operator and an assistant doctor. A residual lesion was defined as a lesion on the scar after polypectomy and was diagnosed using NBI magnification in as much detail as possible. Thereafter, to confirm the accuracy of the data in the registry, we arranged for another endoscopist to verify the captured image.
STUDY ENDPOINTS:
The primary endpoint of interest in this study was the resection width achieved by HP or CP. The diameter of the ulcer base was evaluated by a measuring device (M1-1C, Olympus Optical Co, Tokyo, Japan).
PROCEDURES:
This was a prospective randomized comparison of HP and CP for treatment of colorectal polyps. Patients were randomized according to treatment by HP or CP when a polyp that met the inclusion criteria was found during colonoscopy. The whole colon was divided into left side and right side with the splenic flexure as the boundary; in each patient, one side was treated with CP and the other side with HP. For patients who met the inclusion criteria, the treatment procedure, either HP or CP, was randomly selected using the envelope method at the moment of treatment.
One week after treatment, the bilateral post-polypectomy state was confirmed by colonoscopic examination. Polypectomy had been performed using the same technique, either CP or HP, on either the right or left side, respectively, in each patient. The alternative technique was then used on the other side in each patient, with the exception of Case 6, who only had eligible polyps on the right side, which were treated with HP. With the exception of this 1 patient, we could follow up differences in healing according to differences in treatment in the same patient.
POLYPECTOMY PROTOCOL: Patients underwent standard bowel preparation. On the day before colonoscopy, patients took 5 mg of Mosapride citrate hydrate tablets (Gasmotin®, Sumitomo Dainippon Pharma Co, Ltd. Osaka, Japan) 30 min before each meal and 24 mg of Sennoside (Pursennid®, Mitsubishi Tanabe Pharma Corporation, Osaka, Japan) at 9: 00 PM. No dietary restrictions were imposed for breakfast and lunch the day before the test, but dinner was to be completed by 8: 00 PM. From 4 to 6 h prior to the colonoscopy, the patient drank olyethylene glycol (PEG: Niflec®, EA Pharma Co. Ltd, Tokyo, Japan) at a rate of about 1.5–2.0 L/h. Patients were then prohibited from eating or drinking until after the examination. While performing the endoscopy, patients were given an anticholinergic agent (butylscopolammonium bromide) or glucagon. Sedation was administered at the endoscopists’ discretion when patients reported feeling pain or discomfort. A high-definition colonoscope (PCF-Q260AZI; Olympus Optical Co, Tokyo, Japan) was used for polypectomy by operators who each had performed >3000 colonoscopies. The endoscope-dedicated equipment had a distal tip 11.7 mm in diameter and insertion tube 11.8 mm in diameter (working length, 1330 cm: accessory channel diameter, 3.2 mm). Cecal intubation was confirmed either by ileal intubation or visualization of the appendiceal orifice and the ileocecal valve. Polypectomy was selected for polyps diagnosed to be neoplastic by high-definition white-light endoscopy and NBI endoscopy. [23–26]. Polyp shape was classified according to the Paris classification as elevated (Is, Isp, Ip) or flat (IIa, IIb) [27]. Polyp size was estimated using the complete width of the opening of conventional biopsy forceps. If the size of the polyp was eligible for the study (≤10 mm), polypectomy was performed by 1 of 2 randomized methods. After cecal intubation, the operator searched for polyps while withdrawing the endoscope. The operator performed the polypectomies by the allocated method (CP or HP). Polyps <5 mm were assigned forceps polypectomy and those ≥5 mm were assigned snare polypectomy. The CFP was performed by a radial jaw 4 jumbo forceps (M00513360, jaw volume 12.44 mm, maximum jaw opening 8.8 mm; Boston Scientific, Marlborough, MA, USA). The cold snare polypectomy (CSP) was performed as follows: the polyp was excised to include approximately 1 to 2 mm of normal mucosa around the base of the polyp without using electrocautery and retrieved into the trap. In our study, a traditional oval snare (Captivator II Boston Scientific, Natick, MA, USA) with a 10-mm opening width was used. Regardless of allocation, the same forceps and snare wire were decided upon in advance. Retrieved polyps underwent histopathologic evaluations. An ERBE VIO300 (Amco, Tokyo, Japan) was used in the endo-cut mode with the effect 3 current set at output limit 120 W and forced coagulation current set at an output limit of 35 W for HP. Prophylactic clipping at the site of resection was not performed. We evaluated the colonic mucosa after resection using a high-definition colonoscope. The diameter of the ulcer base was evaluated by a measuring device (M1-1C, Olympus Optical Co.).
STATISTICAL ANALYSES:
Results were analyzed by the Mann-Whitney U test and chi-squared test. Continuous variables such as polyp size and ulcer size were analyzed using the Mann-Whitney U test. An SPSS software program was used for statistical analyses (version 22.0 for Windows; IBM Japan, Ltd., Tokyo, Japan).
Results
In our study, 6 patients (mean age 70.1 y (67–75 y; 3 males, 3 females) were registered. At the end of the trial, only 6 patients were enrolled because of difficulties in obtaining consent from a larger number of patients. We reviewed 52 neoplastic lesions resected by polypectomy in 6 patients who underwent follow-up colonoscopy 1 week after treatment (Figure 1). Study subjects’ demographic and clinical characteristics population are shown in Table 1. The mean tumor size did not differ between the 2 groups – CP vs HP: 5.41 (3–8) mm vs 5.68 (3–8) mm. The CP group consisted of 10 lesions in the CFP group and 17 lesions in the CSP group, and the HP group consisted of 7 lesions in the HFP group and 18 lesions in the HSP group. Figure 2 shows the distribution of lesions and details of treatment in each patient. In 3 cases CP was performed on the right side and in the other 3 cases HP was performed on the left side. The opposite treatment, either CP or HP, was administered to the alternate side except in 1 patient (Case 6 had lesions on only the right side). Regarding localization, in the CP group, 66.6% (17 lesions) were right-sided and 32.4% (10 lesions) were left-sided. In the HP group, 56.0% (13 lesions) were right-sided and 44.0% (12 lesions) were left-sided. Ulcer base diameter 1 week after treatment in the CP group was smaller than in the HP group (2.70 (1–5) mm vs 4.84 (2–8) mm;
Discussion
Both CP and HP are major methods of polypectomy for removing DCPs. In this study, findings showed that 1 week after treatment, the ulcer diameter was smaller and there were fewer exposed blood vessels with CP than with HP. However, 1 residual lesion case was found in the CP group.
There have been many reports about therapeutic results, usefulness of devices, and adverse events after endoscopic resection [10,12,14,28–30]; however, there have been few studies on early-phase follow-up after resection. In previous reports, Suzuki et al endoscopically confirmed the condition of the wound immediately after the procedure and 1 day later [30]. They found that the mucosal defect immediately after CSP was significantly larger than that after HSP, and that it decreased within 1 day after CSP but increased with HSP. In the present study, we compared HP and CP 1 week after resection.
In the CP group, the ulcer base diameter was smaller and there were fewer exposed blood vessels 1 week after treatment than in the HP group, suggesting that the cold technique did not damage ulcers and their edges because of the lack of a burn artifact. Non-energized devices perform very well in making incisions with a low level of iatrogenic damage compared to energized devices [31]. Electrocautery provides an efficient method of early hemostasis, but involves several drawbacks such as lateral tissue damage from the passage of electrical current. HP can damage deeper vessels and the mucous membrane, with an increased risk of delayed healing [32]. HP appears to delay mucosal regeneration compared to CP. In fact, edema and redness were prominent in the ulcer margins in the HP group.
In many studies, measurements of the depth of injury have shown histologic effects of various lasers (electrocautery) in the upper aerodigestive tract [33–37]. A greater depth of injury with electrocautery than with non-electrocautery was shown in most of these studies. In a prospective randomized study in a rat model, histologic depth of injury was measured in the upper aerodigestive tract of rats after creating incisions using a scalpel and electrocautery [29]. Wounds created by non-electrocautery resulted in the least tissue destruction, whereas wounds created by electrocautery had a significantly greater postoperative depth of damage [35,37].
Further, another animal study suggested that energized incisions resulted in more substantial effects at both the gene transcription level and the protein level than low-energized incisions [38]. Tissue incised with energized techniques exhibited higher levels of hemostasis remnants, more inflammation, more immune-related genes and proteins, and less wound healing [38].
In our study, non-electrical treatment caused less mucosal damage than energized treatment in removing DCPs. The risk of PPB could be theoretically reduced by avoiding electrocautery in CP. Also, with CP there were fewer exposed blood vessels compared to HP, which is important when considering PPB complications, although no delayed PPB complications occurred in the present study. There are several reasons for the failure to demonstrate a difference between the 2 groups. In general, the incidence of delayed PPB is 0.6–2.4% [7,39]. Because the number of lesions among study participants was small, this result was not unexpected, and this may also have been because no patients received antithrombotic treatment. This issue should be further evaluated in more patients to detect delayed PPB.
Our study revealed 1 residual lesion in the CP group. Din et al reported that after CFP for polyps ≤5 mm, the overall recurrence rate was 17% [29]. Several recent reports showed that CSP was associated with histopathologically incomplete resection rates of 1.8% and 3.9% [8,40], which are thought to be involved in recurrence after resection. As CP does not use an energized unit, specimens resected by CP are fragile and show no effects of burning. It is sometimes difficult to discriminate the resected margin of lesions without a burning effect or lesions that are partially collapsed. Also, polypectomy specimens were small and fragile and were not pinned, so a real horizonal vertical margin was not evaluated in some of the specimens. For these reasons, it is important to make an accurate endoscopic diagnosis before treatment and to exclude the presence of a residual tumor after treatment.
This study had some limitations. Firstly, the number of samples was small, and the study took place at a single center, making randomization inadequate. It was a heavy burden for the patient to undergo 2 colonoscopies during 1 week. Therefore, the target number of cases was not reached. Secondly, since the study patients participated on a voluntary basis, they may have been healthier than the general population, causing a selection bias. Thirdly, experienced operators are aware that HP is believed to cause more delayed PPB than CP. Thus, operators in this clinical study may have unconsciously performed HP more carefully to reduce damage to the deep submucosal layer in the HP group.
Despite the above limitations, the main strength of this study is that observations were made in the early phase after treatment by different methods in the same patient; therefore, patient-specific healing factors could be ruled out, lessening the chance for between-patient errors.
Conclusions
Our findings from this prospective study from a single center showed that at 1 week after treatment, CP resulted in superior colonic mucosal healing with a smaller ulcer diameter and fewer blood vessels when compared with HP.
In conclusion, CSP is a safe method for the removal of DCPs. These results support the safety of CP.
Figures
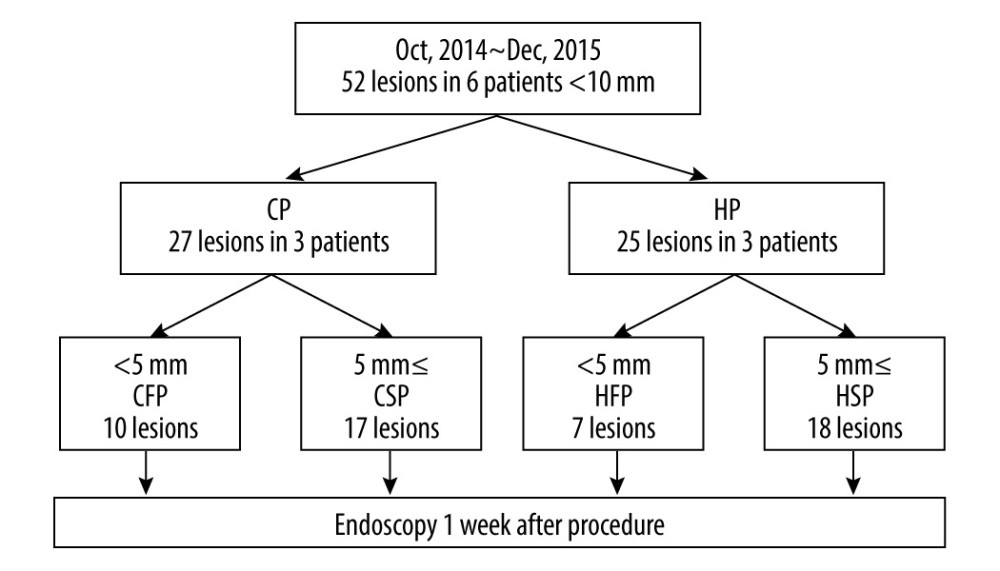 Figure 1. Study flow. HP – hot polypectomy; CP - cold polypectomy; CFP – cold forceps polypectomy; CSP – cold snare polypectomy; HFP – hot forceps polypectomy; HSP – hot snare polypectomy.
Figure 1. Study flow. HP – hot polypectomy; CP - cold polypectomy; CFP – cold forceps polypectomy; CSP – cold snare polypectomy; HFP – hot forceps polypectomy; HSP – hot snare polypectomy.  Figure 2. The distribution of lesions and details of treatment in each patient. In 3 cases, CP was performed on the right side and HP was performed on the left side. In the other 3 cases the opposite treatment was administered. There was 1 exception (Case 6), who only had lesions on the left side, for which CP was used. C – Cecum; A – Ascending colon; T – Transverse colon; D – Descending colon; S – Sigmoid colon; R – Rectum; CFP – cold forceps polypectomy; HFP – hot forceps polypectomy; CSP – cold snare polypectomy; HSP – hot snare polypectomy; L – residual lesion; V – exposed blood vessels.
Figure 2. The distribution of lesions and details of treatment in each patient. In 3 cases, CP was performed on the right side and HP was performed on the left side. In the other 3 cases the opposite treatment was administered. There was 1 exception (Case 6), who only had lesions on the left side, for which CP was used. C – Cecum; A – Ascending colon; T – Transverse colon; D – Descending colon; S – Sigmoid colon; R – Rectum; CFP – cold forceps polypectomy; HFP – hot forceps polypectomy; CSP – cold snare polypectomy; HSP – hot snare polypectomy; L – residual lesion; V – exposed blood vessels. 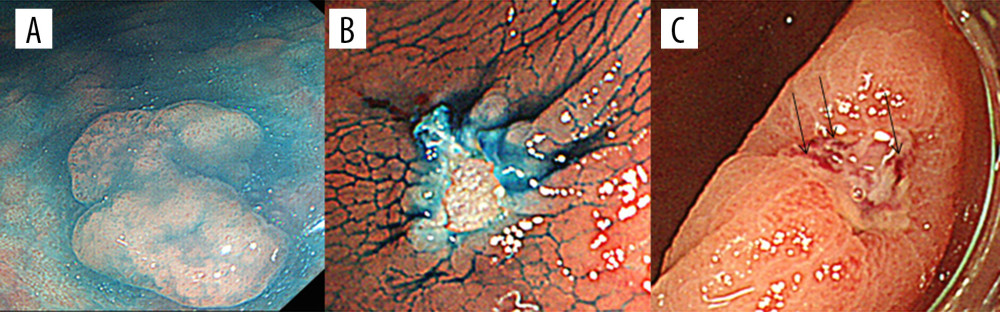 Figure 3. Representative case of HP. (A) A non-polypoid (IIa) lesion (7 mm) located in the descending colon. (B) The lesion was resected en bloc by HSP. (C) One week after HP, the ulcer was 7 mm and had an exposed blood vessel. HP – hot polypectomy; HSP – hot snare polypectomy
Figure 3. Representative case of HP. (A) A non-polypoid (IIa) lesion (7 mm) located in the descending colon. (B) The lesion was resected en bloc by HSP. (C) One week after HP, the ulcer was 7 mm and had an exposed blood vessel. HP – hot polypectomy; HSP – hot snare polypectomy 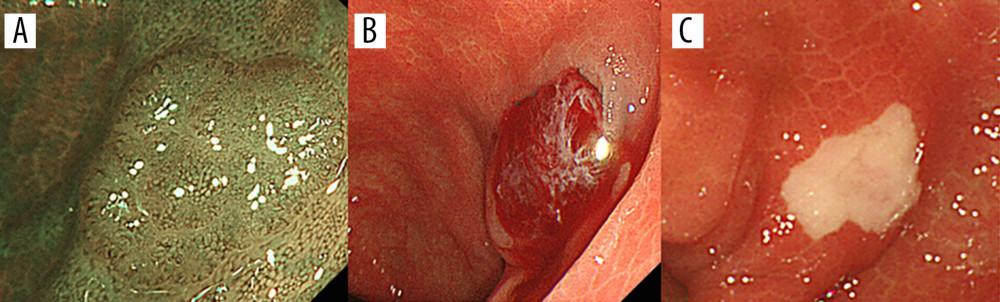 Figure 4. Representative case of CP. (A) A non-polypoid (IIa) lesion (6 mm) located in the descending colon. (B) The lesion was resected en bloc by CSP. (C) One week after CP, the ulcer was 3 mm and no exposed blood vessels were observed. CP – cold polypectomy; CSP – cold snare polypectomy.
Figure 4. Representative case of CP. (A) A non-polypoid (IIa) lesion (6 mm) located in the descending colon. (B) The lesion was resected en bloc by CSP. (C) One week after CP, the ulcer was 3 mm and no exposed blood vessels were observed. CP – cold polypectomy; CSP – cold snare polypectomy. References
1. Winawer SJ, Zauber AG, Ho MN, Prevention of colorectal cancer by colonoscopic polypectomy. The National Polyp Study Workgroup: N Engl J Med, 1993; 329; 1977-81
2. Zauber AG, Winawer SJ, O’Brien MJ, Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths: N Engl J Med, 2012; 366; 687-96
3. Metz A, Moss A, Mcleod D, A blinded comparison of the safety and efficacy of hot biopsy forceps electrocauterization and conventional snare polypectomy for diminutive colonic polypectomy in a porcine model: Gastrointest Endosc, 2013; 77; 484-90
4. Fujiya M, Sato H, Ueno N, Efficacy and adverse events of cold vs hot polypectomy: A meta-analysis: World J Gastroenterol, 2016; 22; 5436-44
5. Yamashina T, Fukuhara M, Maruo T, Cold snare polypectomy reduced delayed postpolypectomy bleeding compared with conventional hot polypectomy: A propensity score-matching analysis: Endosc Int Open, 2017; 05; E587-94
6. Boumitri C, Mir FA, Ashraf I, Prophylactic clipping and post-polypectomy bleeding: A meta-analysis and systemat c review: Ann Gastroenterol, 2016; 29; 502-8
7. Kwon MJ, Kim YS, Bae SI, Risk factors for delayed post-polypectomy bleeding: Intest Res, 2015; 13; 160-65
8. Iwashita H, Takedatsu H, Murao H, Safety and efficacy of cold polypectomy compared to endoscopic mucosal resection and hot biopsy polypectomy: Scand J Gastroenterol, 2019; 54; 678-83
9. Uraoka T, Oka S, Ichihara S, Endoscopic management of colorectal tumors less than 10 mm in size: Current status and future perspectives in Japan from a questionnaire survey: Dig Endosc, 2018; 30; 36-40
10. Horiuchi A, Nakayama Y, Kajiyama M, Removal of small colorectal polyps in anticoagulated patients: A prospective randomized comparison of cold snare and conventional polypectomy: Gastrointest Endosc, 2014; 79; 417-23
11. Hirose R, Yoshida N, Murakami T, Histological analysis of cold snare polypectomy and its indication for colorectal polyps 10–14 mm in diameter: Dig Endosc, 2017; 29; 594-601
12. Kawamura T, Takeuchi Y, Asai S, A comparison of the resection rate for cold and hot snare polypectomy for 4–9 mm colorectal polyps: A multicentre randomised controlled trial (CRESCENT study): Gut, 2018; 67; 1950-57
13. Aslan F, Cekic C, Camci , What is the most accurate method for the treatment of diminutive colonic polyps? Standard versus jumbo forceps polypectomy: Medicine (Baltimore), 2015; 94; e621
14. Piraka C, Saeed A, Waljee AK, Cold snare polypectomy for nonpedunculated colon polyps greater than 1 cm: Endosc Int Open, 2017; 05; E184-89
15. Repici A, Hassan C, Vitetta E, Safety of cold polypectomy for <10 mm polyps at colonoscopy: A prospective multicenter study: Endoscopy, 2011; 44; 27-31
16. Chukmaitov A, Bradley CJ, Dahman B, Polypectomy techniques, endoscopist characteristics, and serious gastrointestinal adverse events: J Surg Oncol, 2014; 110; 207-13
17. Aslan F, Camci M, Alper E, Cold snare polypectomy versus hot snare polypectomy in endoscopic treatment of small polyps: Turk J Gastroenterol, 2014; 25; 279-83
18. Ichise Y, Horiuchi A, Nakayama Y, Tanaka N, Prospective randomized comparison of cold snare polypectomy and conventional polypectomy for small colorectal polyps: Digestion, 2011; 84; 78-81
19. Ferlitsch M, Moss A, Hassan C, Colorectal polypectomy and endoscopic mucosal resection (EMR): European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline: Endoscopy, 2017; 49; 270-97
20. Murakami T, Yoshida N, Yasuda R, Local recurrence andits risk factors after cold snare polypectomy of colorectal polyps: Surg Endosc, 2020; 34; 2918-25
21. Gschwantler M, Kriwanek S, Langner E, High-grade dysplasia and invasive carcinoma in colorectal adenomas: A multivariate analysis of the impact of adenoma and patient characteristics: Eur J Gastroenterol Hepatol, 2002; 14; 183-88
22. Suzuki S, Gotoda T, Kusano C, Width and depth of resection for small colorectal polyps: Hot versus cold snare polypectomy: Gastrointest Endosc, 2018; 87; 1095-103
23. Tanaka S, Hayashi N, Oka S, Chayama K, Endoscopic assessment of colorectal cancer with superfcial or deep submucosal invasion using magnifying colonoscopy: Clin Endosc, 2013; 46; 138-46
24. Sano Y, Tanaka S, Kudo SE, Narrow-band imaging (NBI) magnifying endoscopic classifcation of colorectal tumors proposed by the Japan NBI Expert Team: Dig Endosc, 2016; 28; 526-33
25. Wada Y, Kashida H, Kudo SE, Diagnostic accuracy of pit pattern and vascular pattern analyses in colorectal lesions: Dig Endosc, 2010; 22; 192-99
26. Yoshida N, Yagi N, Inada Y, Ability of a novel blue laser imaging system for the diagnosis of colorectal polyps: Dig Endosc, 2014; 26; 250-58
27. , The Paris endoscopic classification of superficial neoplastic lesions: Esophagus, stomach, and colon: November 30 to December 1, 2002;: Gastrointest Endosc, 2003; 58; S3-43
28. Lee HS, Park HW, Lee JS, Treatment outcomes and recurrence following standard cold forceps polypectomy for diminutive polyps: Surg Endosc, 2017; 31; 159-69
29. Din S, Ball AJ, Riley SA, A randomized comparison of cold snare polypectomy versus a suction pseudopolyp technique: Endoscopy, 2015; 47; 1005-10
30. Lee CK, Shim JJ, Jang JY, Cold snare polypectomy vs. cold forceps polypectomy using double-biopsy technique for removal of diminutive colorectal polyps: A prospective randomized study: Am J Gastroenterol, 2013; 108; 1593-600
31. Carew JF, Ward RF, LaBruna A: Laryngoscope, 1998; 108; 373-80
32. Voorhees JR, Cohen-Gadol AA, Laws ER, Spencer DD, Battling blood loss in neurosurgery: Harvey Cushing’s embrace of electrosurgery: J Neurosurg, 2005; 102; 745-52
33. Norris CW, Mullarky MB, Experimental skin incision made with the carbon dioxide laser: Laryngoscope, 1982; 92; 416-19
34. Kyzer MD, Aly AS, Davidson JM, Sub ablation effects of the KTP laser on wound healing: Laser Surg Med, 1993; 13; 62-71
35. Cochrane JP, Beacon JP, Creasey GH, Russell RC, Wound healing after laser surgery: An experimental study: Br J Surg, 1980; 67; 7403
36. Hambley R, Hebda PA, Abell E, Russellet RC, Wound healing of skin incisions produced by ultrasonically vibrating knife, scalpel, electrosurgery, and carbon dioxide laser: J Dermatol Surg Oncol, 1988; 14; 1213-17
37. Pogrel MA, Yen CK, Hansen LS, A comparison of carbon dioxide laser, liquid nitrogen cryosurgery and scalpel wounds in healing: Oral Surg Oral Med Oral Pathol, 1990; 69; 269-73
38. Nanduri B, Pendarvis K, Shack LA, Ultrasonic incisions produce less inflammatory mediator response during early healing than electrosurgical incisions: PLoS One, 2013; 8; e73032
39. Zhang Q, An SL, Chen ZY, Assessment of risk factors for delayed colonic post-polypectomy hemorrhage: A study of 15553 polypectomies from 2005 to 2013: PLoS One, 2014; 9; e108290
40. Repici A, Hassan C, Vitetta E, Safety of cold polypectomy for <10 mm polyps at colonoscopy: A prospective multicenter study: Endoscopy, 2012; 44; 27-31
Figures
 Figure 1. Study flow. HP – hot polypectomy; CP - cold polypectomy; CFP – cold forceps polypectomy; CSP – cold snare polypectomy; HFP – hot forceps polypectomy; HSP – hot snare polypectomy.
Figure 1. Study flow. HP – hot polypectomy; CP - cold polypectomy; CFP – cold forceps polypectomy; CSP – cold snare polypectomy; HFP – hot forceps polypectomy; HSP – hot snare polypectomy. Figure 2. The distribution of lesions and details of treatment in each patient. In 3 cases, CP was performed on the right side and HP was performed on the left side. In the other 3 cases the opposite treatment was administered. There was 1 exception (Case 6), who only had lesions on the left side, for which CP was used. C – Cecum; A – Ascending colon; T – Transverse colon; D – Descending colon; S – Sigmoid colon; R – Rectum; CFP – cold forceps polypectomy; HFP – hot forceps polypectomy; CSP – cold snare polypectomy; HSP – hot snare polypectomy; L – residual lesion; V – exposed blood vessels.
Figure 2. The distribution of lesions and details of treatment in each patient. In 3 cases, CP was performed on the right side and HP was performed on the left side. In the other 3 cases the opposite treatment was administered. There was 1 exception (Case 6), who only had lesions on the left side, for which CP was used. C – Cecum; A – Ascending colon; T – Transverse colon; D – Descending colon; S – Sigmoid colon; R – Rectum; CFP – cold forceps polypectomy; HFP – hot forceps polypectomy; CSP – cold snare polypectomy; HSP – hot snare polypectomy; L – residual lesion; V – exposed blood vessels. Figure 3. Representative case of HP. (A) A non-polypoid (IIa) lesion (7 mm) located in the descending colon. (B) The lesion was resected en bloc by HSP. (C) One week after HP, the ulcer was 7 mm and had an exposed blood vessel. HP – hot polypectomy; HSP – hot snare polypectomy
Figure 3. Representative case of HP. (A) A non-polypoid (IIa) lesion (7 mm) located in the descending colon. (B) The lesion was resected en bloc by HSP. (C) One week after HP, the ulcer was 7 mm and had an exposed blood vessel. HP – hot polypectomy; HSP – hot snare polypectomy Figure 4. Representative case of CP. (A) A non-polypoid (IIa) lesion (6 mm) located in the descending colon. (B) The lesion was resected en bloc by CSP. (C) One week after CP, the ulcer was 3 mm and no exposed blood vessels were observed. CP – cold polypectomy; CSP – cold snare polypectomy.
Figure 4. Representative case of CP. (A) A non-polypoid (IIa) lesion (6 mm) located in the descending colon. (B) The lesion was resected en bloc by CSP. (C) One week after CP, the ulcer was 3 mm and no exposed blood vessels were observed. CP – cold polypectomy; CSP – cold snare polypectomy. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387