20 December 2021: Animal Study
Electroacupuncture Ameliorates Depressive-Like State and Synaptic Deficits Induced by Hyper-Cholinergic Tone During Chronic Stress in Rats
Jing Gao12BCDE, Mingyin Lai3BCF, Wen Fu2B, Mengyu Wang2B, Thi Thanh Tam Mai2B, Baile Ning42AF*, Wenbin Fu245AGDOI: 10.12659/MSM.933833
Med Sci Monit 2021; 27:e933833
Abstract
BACKGROUND: Major depressive disorder (MDD) is the leading cause of disability around the world. It is generally agreed that the central cholinergic system plays an important role in emotional regulation. Acetylcholine (ACh) is now a new target for antidepressants. Therefore, the aim of this study was to evaluate the effect of acupuncture on depressive behaviors, cholinergic tones, and synaptic plasticity in the prefrontal cortex (PFC) in chronic unpredictable mild stress (CUMS).
MATERIAL AND METHODS: We randomly divided 36 male Sprague-Dawley (SD) rats into the Normal group, Stress group, Physostigmine+stress (Phys+stress) group, and Electroacupuncture+physostigmine+stress (EA+Phys+stress) group. Rats underwent CUMS exposure for 42 days. After 28 days of CUMS, rats received physostigmine or EA treatment for 2 weeks. Rats in the Phys+stress and EA+Phys+stress group received an intraperitoneal injection of physostigmine (TOCRIS, UK, 5 mg/kg) daily. Rats in the EA+Phys+stress group also received EA stimulation at GV 20 (Baihui), GV 29 (Yintang), LI 4 (Hegu), and LR 3 (Taichong) daily for 2 weeks.
RESULTS: We found that EA ameliorated weight loss and the depressive-like behaviors in the sucrose preference test, novelty-suppressed feeding test, and open-field test. There was significantly decreased expression of ACh and increased expression of acetylcholinesterase (AChE) after EA treatment. Consistent with the behavior tests and cholinergic tones, there were increased spine density and expressions of synaptic proteins, including brain-derived neurotrophic factor (BDNF), glutamate receptor 1 (GluR1), glutamate receptor 2 (GluR2), postsynaptic density protein 95 (PSD95), and synapsin I in the PFC.
CONCLUSIONS: The results suggest that EA can reverse the depressive-like behaviors and synaptic deficits induced by hyper-cholinergic tone during chronic stress via the modulation of hyper-cholinergic tone.
Keywords: Depression, Electroacupuncture, Synapses, Acetylcholine, Acetylcholinesterase, Animals, Blotting, Western, Cholinesterase Inhibitors, Male, Neuronal Plasticity, open field test, Physostigmine, Prefrontal Cortex, Rats, Rats, Sprague-Dawley, Stress, Psychological
Background
Major depressive disorder (MDD) is the leading cause of disability worldwide and the proportion of the global population living with depression is estimated to be approximately 350 million people [1]. However, the etiology of MDD is not yet fully understood. So far, it has been hypothesized that dysfunction within the neurotransmitter systems occurs in people with MDD, including the serotonergic, noradrenergic, dopaminergic, γ aminobutyric acidergic, and glutamatergic systems [2]. Currently, pharmacotherapies, including monoaminergic-acting, tricyclic-acting, and even compound type antidepressants, are the main treatments that target these systems [2]. However, such antidepressants have their drawbacks. First, it takes anywhere from 2 weeks to several months to achieve clinical improvement [3]. Additionally, antidepressants are ineffective in approximately two-thirds of patients [4] and, as suicide risk is elevated in depressed individuals, more efficacious antidepressant therapies with less significant adverse effects are needed. Acupuncture, one of the most widely used therapies in traditional Chinese medicine, is effective in the treatment of many diseases, including depression [5–7]. As such, depression is one of the most commonly treated acupuncture indications [8]. However, the mechanism of action responsible for acupuncture’s antidepressant effects remains to be elucidated.
The cholinergic hypothesis of depression posited that the mechanism underlying depression is a central predominance of an acetyl-cholinergic to catecholaminergic tone [9,10]. Higher levels of acetylcholine (ACh) were found in patients with depression or people with a history of depression [11,12]. Meanwhile, many studies have demonstrated that hyper-cholinergic tone results in depressive symptoms in human and rodent models [13,14]. Acetylcholinesterase (AChE) is the key enzyme in ACh breakdown and is extremely efficient at modulating extracellular ACh levels [15]. Physostigmine, an inhibitor of acetylcholinesterase, can block the breakdown of ACh and induce the excessive accumulation of ACh in the synaptic cleft [15]. Studies have shown that increased sensitivity promotes depressive behaviors induced by mild stressors [16,17], and that enhanced cholinergic tone induced by physostigmine during stress can induce a depressive-like state in rodents [17]. These novel findings suggest that anticholinergic drugs (eg, scopolamine) can induce antidepressant effects in depressed patients and rodents with hyper-cholinergic tone [18–20]. The mechanism might be activation of brain-derived neurotrophic factor (BDNF) release, mammalian target of rapamycin complex 1 (mTORC 1) signaling, and synthesis of synaptic proteins in the prefrontal cortex (PFC) [19,21,22]. It has been demonstrated that regulation of the cholinergic system is a potential target for antidepression therapy.
Our previous clinical studies have identified the therapeutic effects of acupuncture. Specifically, we demonstrated that acupuncture can decrease HAMD and PSQI scores and ameliorate depressive symptoms, as well as alleviate insomnia by improving sleep quality, and enhance the overall quality of life in patients with depression [23–27]. The clinical efficacy of acupuncture in treating depression patients was maintained after 4 weeks and even at 3 months of follow-up. Furthermore, we found the underlying mechanism responsible for the antidepressant effects of acupuncture may be the increase of synaptic proteins in the PFC and hippocampi [28,29]. However, the underlying effect on the regulation of cholinergic tone in antidepression therapy using acupuncture has not been elucidated.
In the present study, we detected the expression of cholinergic tone and the structure and number of synaptic spines, as well as the related proteins, in the PFC of rats. We sought to determine whether modulating hyper-cholinergic tone and alleviating synaptic deficits contributes to the antidepressant effect of acupuncture.
Material and Methods
ANIMALS:
We obtained 36 adult male Sprague-Dawley rats (180±20 g) from Guangdong Medical Laboratory Animal Center and maintained them in standard conditions with a 12-h light–dark cycle and
CHRONIC STRESS PROCEDURE:
Chronic unpredictable mild stress (CUMS) procedures were conducted for 42 days. The animals were exposed to a variable sequence of mild unpredictable stressors, consisting of 10 stressors in total: 24-h water deprivation, 24-h food deprivation, 5-min cold swimming, reversed light–dark-cycle, 24-h wet bedding, 24-h 45° cage tilt, 2-min tail clip, 3-h intermittent white noise, stroboscope overnight, and 4-h activity limitation. Two of these stressors were performed daily in random order throughout the experiment.
GROUPS AND TREATMENT:
Rats were randomly divided into 4 groups (9 rats in each group): the Normal group, Stress group, physostigmine+stress (Phys+stress) group, and EA+physostigmine+stress (EA+Phys+stress) group. All rats except for the Normal group were exposed to chronic unpredictable stressors. Rats in the Phys+stress group also received an intraperitoneal injection of physostigmine (TOCRIS, UK, 5mg/kg) after 28 days of CUMS, once a day for 2 weeks. In the EA+Phys+stress group, rats received the same electroacupuncture stimulation and physostigmine administration as the Phys+stress group. All electroacupuncture was carried out 30 min after administration in the EA+Phys+stress group.
EA TREATMENT:
We placed soft and loose loops under the armpits of rats before treatment. The rats were then placed on a custom-made acupuncture experimental apparatus (National Utility Model patent of China, NO. ZL 2019 2 1340810.6), which is less confining, allowing some freedom of movement. Sterilized, disposable stainless steel needles, 0.16 mm in diameter and 7 mm in length (Hwato Medical Instruments Co, Ltd., Suzhou, China), were inserted 2–3 mm at the GV 20 (Baihui), GV 29 (Yintang), LI 4 (Hegu), and LR 3 (Taichong) acupoints, as reported in previous studies [29,30]. GV 20 is located above the apex auriculate, on the midline of the head; GV 29 is located at the midpoint between the eyes; LI 4 is located on the depression between the first and second metacarpal of the forelimbs; and LR 3 is located on the depression of the first and second metacarpal of hind limbs, as shown in Figure 1A. Then, the EA apparatus (Hans-LH202H, Nanjing Jisheng Medicine Science Co., Ltd., Nanjing, China) was energized for EA stimulation (2/100 Hz, 0.2 mA, for 20 min). EA treatment was started on Day 29 of the CUMS procedure and performed for 2 weeks.
BODY WEIGHT MEASUREMENT (BWM):
All rats were weighed before and after the intervention.
SUCROSE PREFERENCE TEST (SPT):
A SPT was conducted before and after the intervention. In this test, rats were exposed to 2 identical bottles of 1% sucrose solution for the first 24 h, then exposed to a bottle of water and a bottle of 1% sucrose solution for the second 24 h. After 24 h of water deprivation, rats were exposed to 2 identical bottles, one filled with 1% sucrose solution and one with water. Sucrose and water consumption over the following 12 h were measured. Sucrose preference rate was calculated by the equation as follows:
NOVELTY-SUPPRESSED FEEDING TEST (NSFT):
A NSFT was conducted after intervention, in a manner identical to that previously described. Before the test, rats were food-deprived for 24 h. Rats were placed in an open field (80×80×40 cm) with a small amount of food in the center. The test was conducted in a dim environment. During a testing period of 6 min, the latency to seek food, specifically the time it took for the rat to approach and take the first bite of the food, was recorded with a stopwatch. Home cage food intake was measured immediately following the test as a control. Excretions were cleaned up before putting in the next rat. A 75% alcohol solution was sprayed on the boxes to remove odors from the previous rats.
OPEN-FIELD TEST (OFT):
An OFT was conducted after NSFT. The open-field apparatus was made of wood, with an 80×80 cm field and walls 40 cm high. On the floor of the apparatus were 25 identical squares, each 16×16 cm, printed in black. Rats were placed at one corner of the open field and allowed to roam freely for 3 min. Similar to the NSFT, the test was conducted in a dim environment. Throughout the testing period, the following behaviors were recorded: horizontal activity frequency (ie, the total number of squares entered by rats with 4 paws), vertical activity frequency (ie, the number of times that the rat rose onto its hand paws), and grooming frequency (ie, the number of times that the rat licked itself or adjusted its hair with its paws). Excretions were cleaned up before putting in the next rat. The behavioral test timeframe is shown in Figure 1B.
ENZYME-LINKED IMMUNOSORBENT ASSAY (ELISA):
After the behavioral tests, rats were anesthetized and killed. Brains were quickly removed and blood was collected from the abdominal aorta. The serum was then separated by centrifugation (3000 rpm for 5 min) and stored at −20°C for further analysis. The prefrontal cortex tissue samples were quickly put on ice and stored at −80°C for later analysis. The concentrations of acetylcholine (Shanghai Enzyme-linked Biotechnology Co., Ltd., China, ACh) and acetylcholinesterase (US Biological, USA, AChE) in the prefrontal cortex were tested by ELISA kit strictly according to the protocol provided by the manufacturer.
GOLGI-COX STAINING AND SPINE DENSITY ANALYSIS:
A FD Golgi Stain kit (FD Neuro-Technologies, USA) was used to conduct Golgi staining, with strict adherence to manufacturer instructions. Animals were anesthetized and killed after 2 weeks of intervention. Brains were quickly removed and immediately immersed in the impregnation solution (made by equal volumes of solution A and solution B) for 2 weeks, then solution C for 1 week. Brain slices at a thickness of 150 μm were obtained via a freezing microtome (HM525, Thermo Science, USA) and stained following the staining procedure supplied by the manufacturer. The brain slices were washed twice with ddH2O for 4 min each, stained with the solution (contains solution D, solution E and ddH2O, and the volume ratio is 1: 1: 2) for 10 min, and washed twice with ddH2O again. Then, the slices were dehydrated by 50%, 75%, 95% ethanol in sequence for 4 min each, and by 100% ethanol 4 times for 4 min each. Finally, slices were cleared 3 times with xylene for 4 min each and sealed with Balsam Mounting Medium. Three independent coronal sections containing prelimbic cortex (PrL), a part of PFC, were imaged per rat (Bregma 2.52 mm to 4.68 mm). Spines were counted on the tertiary branches of apical dendrites in the layer V pyramidal neurons of the PFC. A 10-μm segment of tertiary dendrite from the layer V pyramidal neurons of the PFC was randomly selected in each section. All segments were imaged using NIS-Element Viewer software under an inverted microscope (DM100, Nikon, Japan) with a 100× oil-immersion lens (NF2, Nikon, Japan) by investigators blind to the experimental scheme. Spine density and mature or immature spine density were determined [31]. The number of spines along a 10-μm segment was counted to determine the number of spines/μm.
WESTERN BLOT ANALYSIS:
PFC tissue samples were homogenized and western blot analysis was conducted. A BCA protein assay kit (Beyotime, Shanghai, China) was used to measure protein concentrations. Protein in each lane (50 μm) was separated by SDA-PAGE (Beyotime, Shanghai, China) and transferred to polyvinylidene fluoride (PVDF, Bio-Rad, USA). After blocking with 5% milk/TBST for 2 h, membranes were incubated for 2 h with primary antibodies. The primary antibodies were BDNF, glutamate receptor 1 (GluR1), glutamate receptor 2 (GluR2), postsynaptic density protein 95 (PSD95), synapsin I, and β-actin (all from ABclonal, Wuhan, China, 1: 1000 dilution). Then, samples were incubated with HRP anti-mouse secondary antibody (Yijia, Shanghai, China, 1: 2500 dilution) for 1 h. Bio-Rad Image Lab software was used to scan, and the gray value of bands was analyzed. Data were normalized to β-actin.
STATISTICAL ANALYSIS:
Data are presented as mean±standard error of the mean (SEM). Differences among groups were determined using a one-way analysis of variance (ANOVA) followed by calculation of least significant difference (LSD) when equal variances were assumed. Otherwise, a Dunnett’s T3 test was conducted.
Results
EFFECT OF EA ON BEHAVIORS INDUCED BY CUMS AND PHYSOSTIGMINE:
As shown in Figure 2A, 2B, prior to the start of the intervention (Day 0), compared with the Normal group, the sucrose preference rate was decreased significantly in the Stress group (P<0.05), which reflects that anhedonia is the core symptom of depression, as well as the Phys+stress and EA+Phys+stress group. Due to the accumulation of physostigmine administration or CUMS at Day 14, as shown in Figure 3A–3D, there were significant differences in BWM, SPT, NSFT, and OFT among 4 groups (all P<0.001). Rats in the Phys+stress group manifested depressive symptoms, including decreased sucrose preference rate, prolonged latency feeding time, as well as the reduction of horizontal activity and grooming frequency (all P<0.05), except for vertical activity (P>0.05), when compared with the Stress group. Compared with the Phys+stress group, the sucrose preference rate, latency feeding time, as well as the horizontal activity, vertical activity, and grooming frequency in OFT were ameliorated by EA in the EA+Phys+stress group (all P<0.05). The data above demonstrate that physostigmine administration during chronic stress can aggravate depressive-like behaviors, but EA can reverse the symptoms, creating an antidepressant action.
EFFECT OF EA ON THE CONCENTRATION OF CHOLINERGIC TONE ACH AND ACHE IN THE PFC AND SERUM:
As shown in Figure 4A–4D, chronic stress significantly increased the concentration of ACh in the PFC and serum, while the concentration of AChE decreased in the PFC and serum compared with the Normal group (P<0.001, P<0.05). In the Phys+stress group, the concentration of ACh increased in the PFC and serum (P<0.001, P<0.05). There was a significant decrease in the concentration of AChE in the serum of the Phys+stress group (P<0.05) when compared to the Stress group. However, there was no difference in the concentration of AChE in the PFC in the Phys+stress group (P>0.05). Interestingly, compared with the Phys+stress group, rats in the EA+Phys+stress group manifested a decreased concentration of ACh and increased concentration of AChE in the PFC and serum (all P<0.05).
EFFECT OF EA ON SPINE DENSITY IN THE SPECIAL DENDRITES OF LAYER V PYRAMIDAL NEURONS IN THE PFC:
As illustrated in Figure 5, the dendrite spines in the Stress group were less luxuriant compared with the Normal group. The quantified data demonstrate that the total spine density of the rats in the Stress group significantly decreased (P<0.001), especially in the mature spines (P<0.001). The dendrite spines manifested as bare branches in the Phys+stress group. Meanwhile, there were significant decreases in total spine density in both mature and immature spines (all P<0.001). With the intervention of EA, total spine density was increased significantly in the EA+Phys+stress group (P<0.001). Particularly, EA increased the spine density of mature and immature spines when compared with the Phys+stress group (both P<0.001).
EFFECT OF EA ON EXPRESSIONS OF SYNAPSE-RELATED PROTEINS IN PFC:
Previous studies suggested that the induction of synapse formation requires synthesis of new synaptic proteins, including glutamate receptors and synaptic structural proteins [32]. Synapse-related proteins in the PFC were detected. As shown in Figure 6A, 6B, there were significant differences in the expressions of BDNF, GluR1, GluR2, PSD95, and synapsin I in the PFC in all 4 groups (all P<0.000). Compared with the Normal group, chronic stress caused a significant reduction in the expression of synapse-related proteins in the Stress group (all P<0.000). In addition to the deficits in synaptic morphology observed, there were significantly decreased expressions of BDNF, GluR1, GluR2, PSD95, and synapsin I in the PFC in the Phys+stress group compared with the Stress group (all P<0.000). EA resulted in a significant increase in the expression of those synapse-related proteins compared with the Phys+stress group (all P<0.05).
Discussion
The present study demonstrated that both chronic stress and physostigmine during chronic stress induced hyper-cholinergic tone in the PFC, including a decrease in AChE and increase in ACh. We also found that the enhanced ACh induced by physostigmine during chronic stress could also cause or aggravate depressive-like behaviors with synaptic deficits and decreased synthesis of synapse-related proteins in the PFC. Further, the antidepressant action of EA was associated with behavioral improvement, as well as an increase in the spine density and synthesis of BDNF and synapse-related proteins in the PFC. The underlying mechanism responsible for the antidepressant action of EA may be its involvement in the regulation of AChE, consequently achieving a balance of ACh in the PFC.
It is generally agreed that the central cholinergic system plays an important role in learning and memory processes [15]. Furthermore, the cholinergic system also participates in emotional regulation [10]. Cholinergic system alterations involving cholinesterase, acetyltransferase, and cholinergic receptors have been found in depressive patients and animal models in previous studies [33,34]. In the present study, depressive-like behaviors were observed in rats exposed to chronic stress. Enhanced cholinergic tone was also found in the Stress group, manifesting as a decrease in AChE and resulting in an increase of ACh. These results confirm that chronic stress can induce hyper-cholinergic tone, which may be involved in development of depression.
As early as the 1970s, it was found that increases in central ACh could lead to depressed moods, and the sources include physostigmine administration in healthy people [35] and exposure of rural people to pesticides (most are irreversible cholinesterase inhibitors) [14]. Similarly, observations in rodent studies showed similar results. Addy et al found that physostigmine administration, either systemically or directly into the ventral tegmental area, significantly increased the immobility time in the forced swim test [16]. Fernandes et al found that animals that received physostigmine and were exposed to stress showed less social interaction and more learned helplessness, anhedonia, and anxious-like behaviors [17]. AChE knockdown in the PFC in mice also caused depressive-like behaviors, which provides strong evidence for hyper-acetylcholinergic tone playing an important role in development of depression [16]. Stress is one of the main triggers of depression. Studies have observed that rats with hyper-cholinergic sensitivity are more likely to develop depressive-like behaviors induced by stress [17]. In the present research, depressive-like behaviors induced by physostigmine administration during chronic stress in rats were worse than in rats exposed to stress only. Cholinergic alteration indicated an increase in ACh and a decrease in AChE. It was demonstrated in previous studies that the different results from the intervention of acetylcholinesterase inhibitor during acute stress induced an increase in AChE and Ach [34]. Recent studies also verified that acute stress induced an increase in choline uptake and newly synthesized ACh release, while chronic stress induced adaptive changes in the cholinergic terminal, with a reduction in choline uptake and an elevation in muscarinic binding-site number [36]. Therefore, acute stress may lead to a compensatory increase in AChE activity, reducing extracellular ACh levels, while the cumulative and adaptive change in the cholinergic system during chronic stress induces a decrease in AChE and elevation in ACh. We speculate that aggravated depressive symptoms induced by physostigmine might be relevant to the increasing ACh on the postsynaptic membrane, and hyper-cholinergic tone increased the susceptibility to depression.
Studies demonstrate a role for neuronal atrophy, synaptic remodeling, and loss of neurotrophic factor support [37]. Decreases in the volume and number of synapses in the PFC and hippocampus were observed in MDD patients. In preclinical studies, decreases in the synaptic connections and the number of synapses were also found in the PFC and hippocampus of rodents exposed to chronic stress. Similar to previous studies [38], the present study found a decreased spine density in the PFC caused by chronic stress, especially in mature spines. Consistent with the synaptic morphological alteration, reduced synthesis of synaptic proteins (including the glutamate receptors GluR1 and GLuR2, and the synaptic structural proteins PSD95 and synapsin I) and BDNF, which are necessary for the induction of synapse formation [21], were also observed. Enhanced cholinergic tone induced by physostigmine during chronic stress induced synaptic deficits with a decreased density of mature and immature spines. Reductions in the synthesis of synapse-related proteins were detected as well. We speculated that physostigmine might induce synaptic deficits, but little evidence supports this argument. Previous studies demonstrated that depressive-like behaviors induced by physostigmine were related to higher vulnerability and resilience to stress [17]. We speculated that it might be the vulnerability to stress that contributed to the behavioral and synaptic deficits of the rats the acetyl-cholinergic Phys+stress group.
In the present study, the action of EA on symptoms induced by physostigmine was also detected. EA reversed the depressive-like behaviors and synaptic deficits induced by hyper-cholinergic tone during stress, as well as the synthesis of synaptic proteins. Our previous studies demonstrated that EA could ameliorate behavioral alterations caused by CUMS, and the underlying mechanism attributed to the antidepressant action was relevant to the regulation of glutamate metabolism and activation of the BDNF postsynaptic signaling pathway, consequently playing a protective role with respect to neurons [29,30,39,40]. In the present study, EA reversed the hyper-cholinergic tone caused by physostigmine by decreasing the concentration of ACh and increasing the concentration of AChE. Based on the knowledge that hyper-cholinergic tone plays a role in susceptibility to stress, the effect of EA on the cholinergic system may help recover the balance of cholinergic tone, decreasing vulnerability to stress. However, how EA regulates the cholinergic system needs further study.
Admittedly, there are some limitations to the present study. First, we only detected the alteration of behaviors and synaptic plasticity under the conditions of hyper-cholinergic tone and chronic stress, and the connection between the cholinergic system and synaptic plasticity responsible for antidepression should be further explored. Second, we only detected the regulation in hyper-cholinergic tone induced by physostigmine of EA. More methods (eg, gene knockout) are needed to examine regulation of the cholinergic system of EA.
Conclusions
In conclusion, the present results suggest that physostigmine can induce elevated levels of ACh, which results in decreases in sucrose preference rate, horizontal activity, vertical activity, and grooming frequency, prolonged latency feeding time, and decreased spine density and synaptic proteins. However, EA can reverse the depressive-like behaviors and synaptic deficits induced by hyper-cholinergic tone during chronic stress via the modulation of hyper-cholinergic tone.
Figures
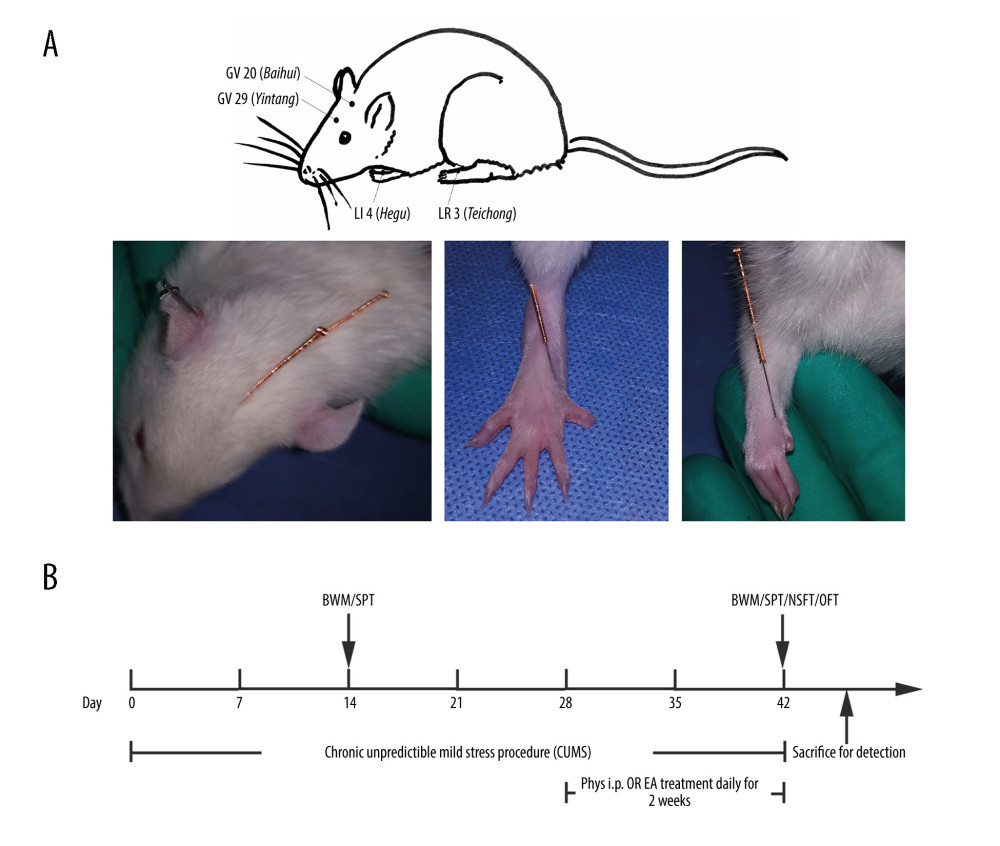 Figure 1. (A) Acupoints utilized. (B) Schematic diagram of the experimental design and behavioral test time frame. The figure was created using Adobe Photoshop V2019 software.
Figure 1. (A) Acupoints utilized. (B) Schematic diagram of the experimental design and behavioral test time frame. The figure was created using Adobe Photoshop V2019 software. 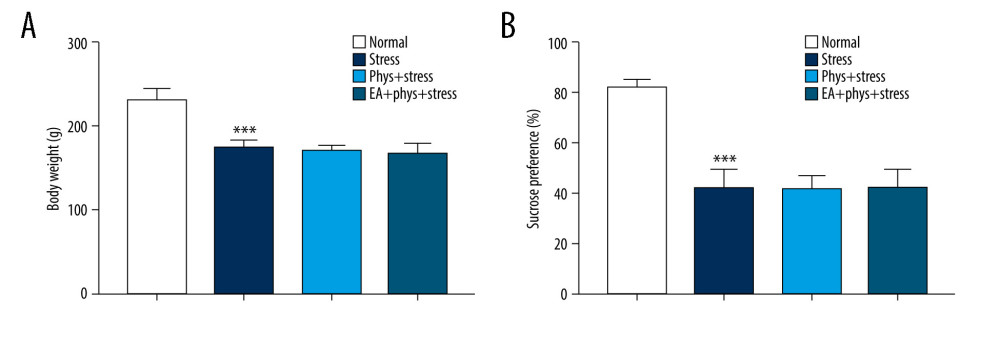 Figure 2. Body weight measurement and sucrose preference test after chronic stress. (A, B) Body weight and sucrose preference rate in Normal, Stress, Phys+Stress, and EA+Phys+stress group after 28 days. Results are the mean±SEM, n=9 per group. *** p<0.001 compared with the Normal group. The Figure was created by the GraphPad Prism version 7.0 software.
Figure 2. Body weight measurement and sucrose preference test after chronic stress. (A, B) Body weight and sucrose preference rate in Normal, Stress, Phys+Stress, and EA+Phys+stress group after 28 days. Results are the mean±SEM, n=9 per group. *** p<0.001 compared with the Normal group. The Figure was created by the GraphPad Prism version 7.0 software. 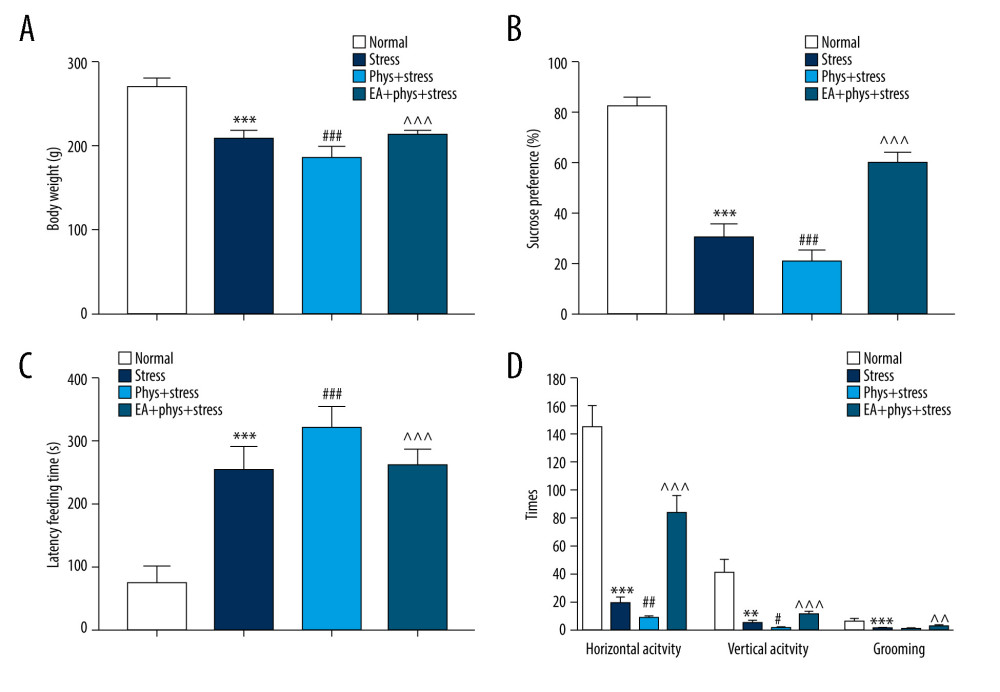 Figure 3. Body weight and behavioral response within the Normal, Stress, Phys+Stress, and EA+Phys+stress group. (A) Body weight measurement. (B) Sucrose preference test. (C) Latency time to feeding in novelty-suppressed feeding test (NSFT), and (D) The open-field test (OFT). Results are the mean±SEM, n=9 per group. ** p<0.01, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using GraphPad Prism version 7.0 software.
Figure 3. Body weight and behavioral response within the Normal, Stress, Phys+Stress, and EA+Phys+stress group. (A) Body weight measurement. (B) Sucrose preference test. (C) Latency time to feeding in novelty-suppressed feeding test (NSFT), and (D) The open-field test (OFT). Results are the mean±SEM, n=9 per group. ** p<0.01, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using GraphPad Prism version 7.0 software. 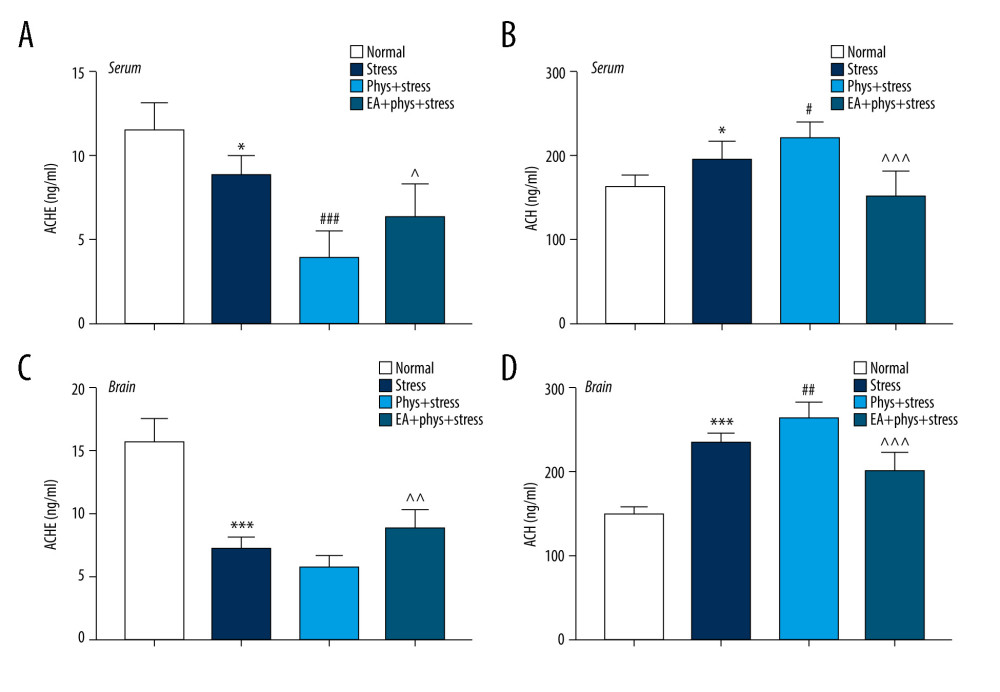 Figure 4. Cholinergic tones, acetylcholinesterase (AChE), and acetyl choline (ACh) in the serum and prefrontal cortex (PFC). (A, B) AChE and ACh in the serum within every group. (C, D) AChE and ACh in the PFC within every group. Results are the mean±SEM, n=9 per group detected in the serum, n=6 per group detected in the PFC. * p<0.05, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using GraphPad Prism version 7.0 software.
Figure 4. Cholinergic tones, acetylcholinesterase (AChE), and acetyl choline (ACh) in the serum and prefrontal cortex (PFC). (A, B) AChE and ACh in the serum within every group. (C, D) AChE and ACh in the PFC within every group. Results are the mean±SEM, n=9 per group detected in the serum, n=6 per group detected in the PFC. * p<0.05, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using GraphPad Prism version 7.0 software. 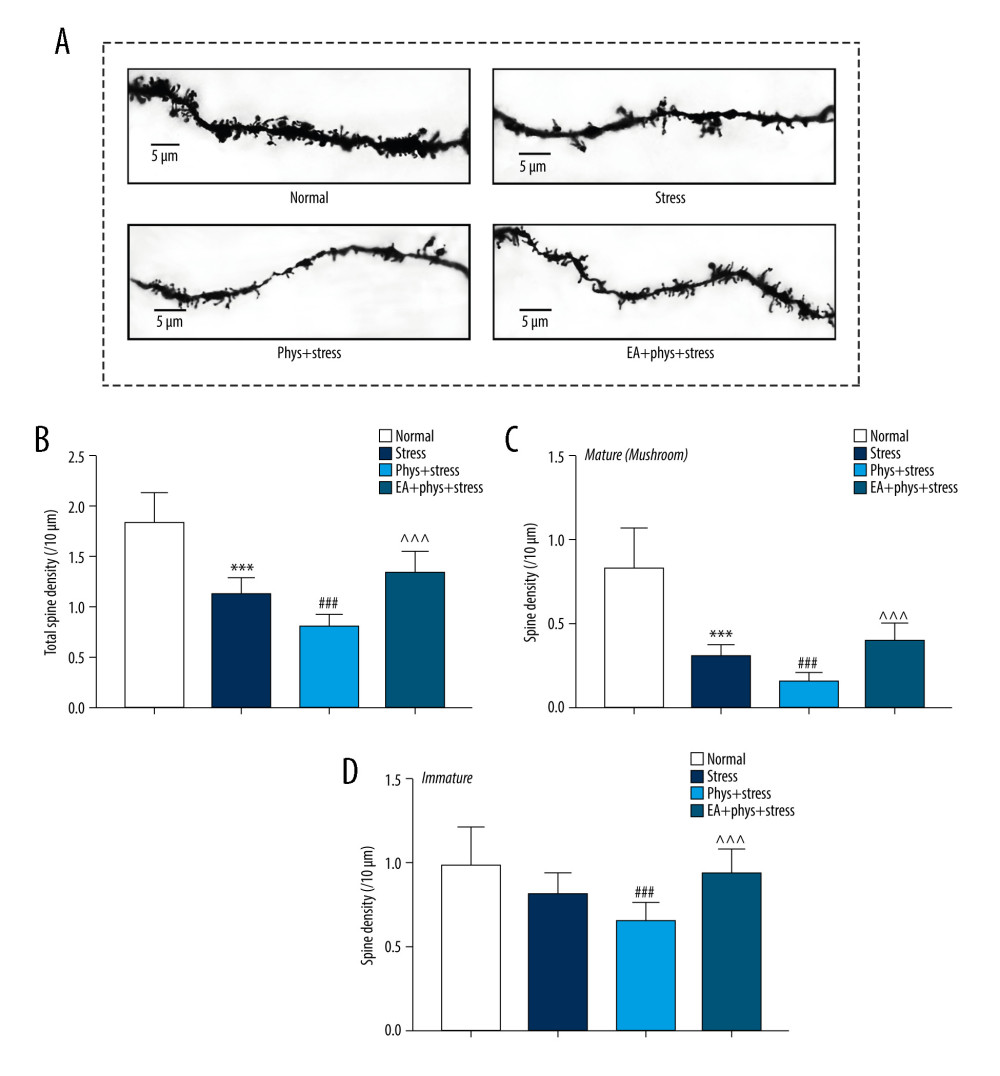 Figure 5. Analysis of spine density in PFC layer V pyramidal neurons (Golgi-Cox staining, Scale bar: 5 μm). (A) Apical dendrites in layer V pyramidal neurons of the PFC, the prelimbic subregions (PrL) were observed. Representative images are shown of high-magnification projections of layer V pyramidal neuron apical tuft dendrites (scale bar: 5 μm). (B–D) The total spine density, mature, immature spine density. Results are presented as mean±SEM (5 dendritic segments from 1 animal. Three rats for each group). *** p<0.001 compared with the Normal group; ## p<0.01, ### p<0.001 compared with the Stress group, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using Adobe Photoshop V2019 and GraphPad Prism version 7.0 software.
Figure 5. Analysis of spine density in PFC layer V pyramidal neurons (Golgi-Cox staining, Scale bar: 5 μm). (A) Apical dendrites in layer V pyramidal neurons of the PFC, the prelimbic subregions (PrL) were observed. Representative images are shown of high-magnification projections of layer V pyramidal neuron apical tuft dendrites (scale bar: 5 μm). (B–D) The total spine density, mature, immature spine density. Results are presented as mean±SEM (5 dendritic segments from 1 animal. Three rats for each group). *** p<0.001 compared with the Normal group; ## p<0.01, ### p<0.001 compared with the Stress group, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using Adobe Photoshop V2019 and GraphPad Prism version 7.0 software. 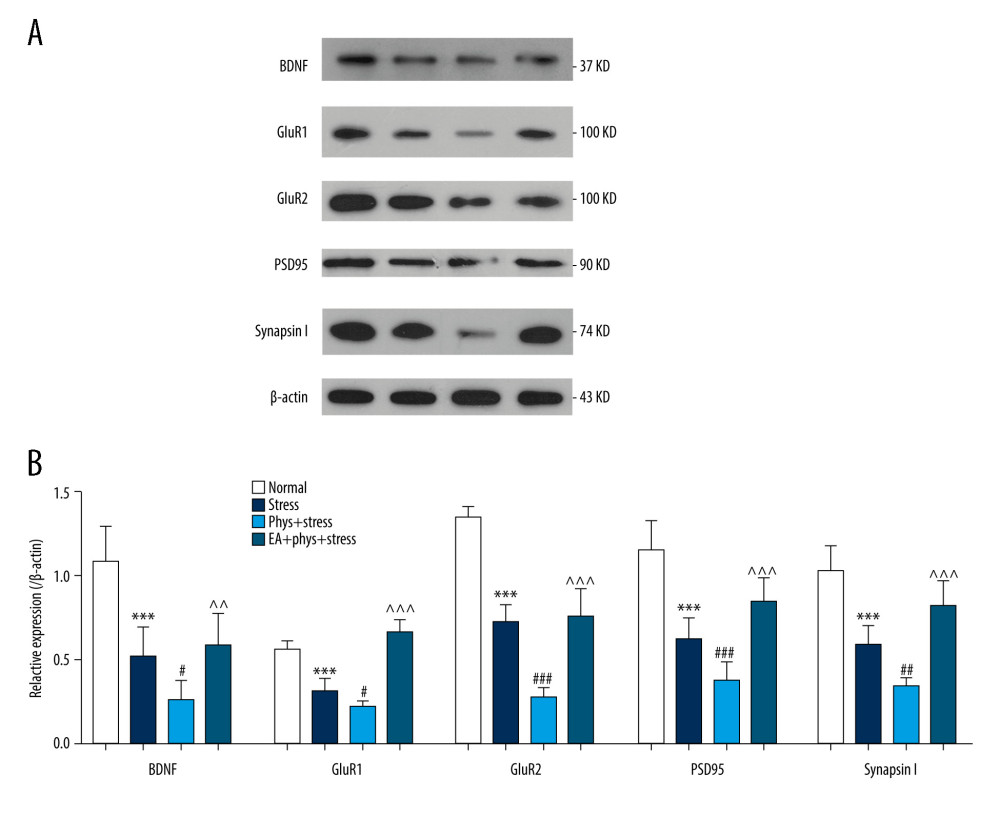 Figure 6. The expression of BDNF and synaptic proteins in the PFC of each group. (A) Respective immune exposures to BDNF, GluR1, GluR2, PSD95, Synapsin I, and β-actin. (B) The levels of BDNF, GluR1, GluR2, PSD95, and synapsin I in the PFC. Results are the mean±SEM (n=6 rats per group, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The Figure was created using Adobe Photoshop V2019 and GraphPad Prism version 7.0 software.
Figure 6. The expression of BDNF and synaptic proteins in the PFC of each group. (A) Respective immune exposures to BDNF, GluR1, GluR2, PSD95, Synapsin I, and β-actin. (B) The levels of BDNF, GluR1, GluR2, PSD95, and synapsin I in the PFC. Results are the mean±SEM (n=6 rats per group, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The Figure was created using Adobe Photoshop V2019 and GraphPad Prism version 7.0 software. References
1. Malhi GS, Mann JJ, Depression: Lancet, 2018; 392(10161); 2299-312
2. Papakostas GI, Ionescu DF, Towards new mechanisms: An update on therapeutics for treatment-resistant major depressive disorder: Mol Psychiatry, 2015; 20(10); 1142-50
3. Sramek JJ, Murphy MF, Cutler NR, Sex differences in the psychopharmacological treatment of depression: Dial Clin Neurosci, 2016; 18(4); 447-57
4. Duman RS, Sanacora G, Krystal JH, Altered connectivity in depression: GABA and glutamate neurotransmitter deficits and reversal by novel treatments: Neuron, 2019; 102(1); 75-90
5. Smith CA, Armour M, Lee MS, Wang LQ, Hay PJ, Acupuncture for depression: Cochrane Database Syst Rev, 2018; 3(3); CD004046
6. Armour M, Smith CA, Wang LQ, Acupuncture for depression: A systematic review and meta-analysis: J Clin Med, 2019; 8(8); 1140
7. Qaseem A, Barry MJ, Kansagara D, Nonpharmacologic versus pharmacologic treatment of adult patients with major depressive disorder: A clinical practice guideline from the American College of Physicians: Ann Intern Med, 2016; 164(5); 350-59
8. Wang H, Yang G, Wang S, The most commonly treated acupuncture indications in the United States: A cross-sectional study: Am J Chin Med, 2018; 46(7); 1-33
9. Janowsky DS, Davis JM, El-Yousef MK, Sekerke HJ, A cholinergic-adernergic hyothesis of mania and depression: Lancet, 1972; 09(23); 632-35
10. Dulawa SC, Janowsky DS, Cholinergic regulation of mood: From basic and clinical studies to emerging therapeutics: Mol Psychiatry, 2019; 24(5); 694-709
11. Davis MT, Holmes SE, Pietrzak RH, Esterlis I, Neurobiology of chronic stress-related psychiatric disorders: Evidence from molecular imaging studies: Chronic stress (Thousand Oaks, Calif), 2017; 1
12. Saricicek A, Esterlis I, Maloney KH, Persistent beta2*-nicotinic acetylcholinergic receptor dysfunction in major depressive disorder: Am J Psychiatry, 2012; 169(8); 851-59
13. Mineur YS, Obayemi A, Wigestrand MB, Cholinergic signaling in the hippocampus regulates social stress resilience and anxiety- and depression-like behavior: Proc Natl Acad Sci USA, 2013; 110(9); 3573
14. Suarez-Lopez JR, Hood N, Suarez-Torres J, Associations of acetylcholinesterase activity with depression and anxiety symptoms among adolescents growing up near pesticide spray sites: Int J Hyg Environ Health, 2019; 222(7); 981-90
15. Picciotto MR, Higley MJ, Mineur YS, Acetylcholine as a neuromodulator: Cholinergic signaling shapes nervous system function and behavior: Neuron, 2012; 76(1); 116-29
16. Addy NA, Nunes EJ, Wickham RJ, Ventral tegmental area cholinergic mechanisms mediate behavioral responses in the forced swim test: Behav Brain Res, 2015; 288; 54-62
17. Fernandes SS, Koth AP, Parfitt GM, Enhanced cholinergic-tone during the stress induce a depressive-like state in mice: Behav Brain Res, 2018; 347(1); 17-25
18. Drevets WC, Furey ML, Replication of scopolamine’s antidepressant efficacy in major depressive disorder: A randomized, placebo-controlled clinical trial: Biol Psychiatry, 2010; 67(5); 432-38
19. Navarria A, Wohleb ES, Voleti B, Rapid antidepressant actions of scopolamine: Role of medial prefrontal cortex and M1-subtype muscarinic acetylcholine receptors: Neurobiol Dis, 2015; 82; 254-61
20. Howland RH, The antidepressant effects of anticholinergic drugs: J Psychosoc Nurs Ment Health Serv, 2009; 7(6); 17-20
21. Ghosal S, Bang E, Yue W, Activity-dependent brain-derived neurotrophic factor release is required for the rapid antidepressant actions of scopolamine: Biol Psychiatry, 2018; 83(1); 29-37
22. Levy MJF, Boulle F, Steinbusch HW, Neurotrophic factors and neuroplasticity pathways in the pathophysiology and treatment of depression: Psychopharmacology, 2018; 235(8); 2195-20
23. Fu WB, Fan L, Zhu XP, Depressive neurosis treated by acupuncture for regulating the liver – a report of 176 cases: J Tradit Chin Med, 2009; 29(2); 83-86
24. Fan L, Fu W, Zhao C, Curative effect of acupuncture on quality of life in patient with depression: A clinical randomized single-blind placebo-controlled study: J Tradit Chin Med, 2016; 36(2); 151
25. Xiuyun W, Qian W, Jianhua L, Randomized single-blind multicenter trial comparing the effects of standard and augmented acupuncture protocols on sleep quality and depressive symptoms in patients with depression: Psychol Health Med, 2017; 23(4); 375-90
26. Chen YF, Liu JH, Xu NG, Effects of acupuncture treatment on depression insomnia: a study protocol of a multicenter randomized controlled trial: Trials, 2013; 14; 2
27. Li S, Li ZF, Wu Q, A multicenter, randomized, controlled trial of electroacupuncture for perimenopause women with mild-moderate depression: Biomed Res Int, 2018; 2018; 5351210
28. Fan L, Zhao Q, Lv AEffect of acupuncture on the release of presynaptic glutamate mediated by SNARE protein in hippocampus of rats with depression: Chinese Journal of Gerontology, 2016; 36(13); 3123-26 [in Chinese]
29. Luo D, Ma R, Wu Y, Mechanism underlying acupuncture-ameliorated depressive behaviors by enhancing glial glutamate transporter in chronic unpredictable mild stress (CUMS) Rats: Med Sci Monit, 2017; 23; 3080-87
30. Fan L, Chen Z, Fu W, Soluble N-ethylmaleimide-sensitive factor attachment receptor (SNARE) protein involved in the remission of depression by acupuncture in rats: J Acupunct Meridian Stud, 2016; 9(5); 242-49
31. Papadakakis A, Sidiropoulou K, Panagis G, Music exposure attenuates anxiety- and depression-like behaviors and increases hippocampal spine density in male rats: Behav Brain Res, 2019; 372; 112023
32. Duman RS, Aghajanian GK, Sanacora G, Krystal JH, Synaptic plasticity and depression: New insights from stress and rapid-acting antidepressants: Nat Med, 2016; 22(3); 238-49
33. Alzarea S, Rahman S, Alpha-7 nicotinic receptor allosteric modulator PNU120596 prevents lipopolysaccharide-induced anxiety, cognitive deficit and depression-like behaviors in mice: Behav Brain Res, 2019; 366; 19-28
34. Kaufer D, Friedman A, Seidman S, Soreq H, Acute stress facilitates long-lasting changes in cholinergic gene expression: Nature, 1998; 393(6683); 373-77
35. Risch SC, Cohen RM, Janowsky DS, Physostigmine induction of depressive symptomatology in normal human subjects: Psychiatry Res, 1981; 4(1); 89-94
36. Finkelstein Y, Koffler B, Rabey JM, Gilad GM, Dynamics of cholinergic synaptic mechanisms in rat hippocampus after stress: Brain Res, 1985; 343(2); 314-19
37. Duman RS, Pathophysiology of depression and innovative treatments: Remodeling glutamatergic synaptic connections: Dialogues Clin Neurosci, 2014; 16(1); 11-27
38. Xu C, Ma XM, Chen HB, Orbitofrontal cortex 5-HT2A receptor mediates chronic stress-induced depressive-like behaviors and alterations of spine density and Kalirin7: Neuropharmacology, 2016; 109; 7-17
39. Fu W, Liu J, Bai YEffect of electroacupuncture on CREB-BDNF postreceptor signal transduction pathway in hippocampus of depression rats: Chinese Journal of Gerontology, 2009; 29(23); 3038-42 [in Chinese]
40. Liu JH, Wu ZF, Sun J, Role of AC-cAMP-PKA cascade in antidepressant action of electroacupuncture treatment in rats: Evid Based Complement Alternat Med, 2012; 2012; 932414
Figures
 Figure 1. (A) Acupoints utilized. (B) Schematic diagram of the experimental design and behavioral test time frame. The figure was created using Adobe Photoshop V2019 software.
Figure 1. (A) Acupoints utilized. (B) Schematic diagram of the experimental design and behavioral test time frame. The figure was created using Adobe Photoshop V2019 software. Figure 2. Body weight measurement and sucrose preference test after chronic stress. (A, B) Body weight and sucrose preference rate in Normal, Stress, Phys+Stress, and EA+Phys+stress group after 28 days. Results are the mean±SEM, n=9 per group. *** p<0.001 compared with the Normal group. The Figure was created by the GraphPad Prism version 7.0 software.
Figure 2. Body weight measurement and sucrose preference test after chronic stress. (A, B) Body weight and sucrose preference rate in Normal, Stress, Phys+Stress, and EA+Phys+stress group after 28 days. Results are the mean±SEM, n=9 per group. *** p<0.001 compared with the Normal group. The Figure was created by the GraphPad Prism version 7.0 software. Figure 3. Body weight and behavioral response within the Normal, Stress, Phys+Stress, and EA+Phys+stress group. (A) Body weight measurement. (B) Sucrose preference test. (C) Latency time to feeding in novelty-suppressed feeding test (NSFT), and (D) The open-field test (OFT). Results are the mean±SEM, n=9 per group. ** p<0.01, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using GraphPad Prism version 7.0 software.
Figure 3. Body weight and behavioral response within the Normal, Stress, Phys+Stress, and EA+Phys+stress group. (A) Body weight measurement. (B) Sucrose preference test. (C) Latency time to feeding in novelty-suppressed feeding test (NSFT), and (D) The open-field test (OFT). Results are the mean±SEM, n=9 per group. ** p<0.01, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using GraphPad Prism version 7.0 software. Figure 4. Cholinergic tones, acetylcholinesterase (AChE), and acetyl choline (ACh) in the serum and prefrontal cortex (PFC). (A, B) AChE and ACh in the serum within every group. (C, D) AChE and ACh in the PFC within every group. Results are the mean±SEM, n=9 per group detected in the serum, n=6 per group detected in the PFC. * p<0.05, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using GraphPad Prism version 7.0 software.
Figure 4. Cholinergic tones, acetylcholinesterase (AChE), and acetyl choline (ACh) in the serum and prefrontal cortex (PFC). (A, B) AChE and ACh in the serum within every group. (C, D) AChE and ACh in the PFC within every group. Results are the mean±SEM, n=9 per group detected in the serum, n=6 per group detected in the PFC. * p<0.05, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using GraphPad Prism version 7.0 software. Figure 5. Analysis of spine density in PFC layer V pyramidal neurons (Golgi-Cox staining, Scale bar: 5 μm). (A) Apical dendrites in layer V pyramidal neurons of the PFC, the prelimbic subregions (PrL) were observed. Representative images are shown of high-magnification projections of layer V pyramidal neuron apical tuft dendrites (scale bar: 5 μm). (B–D) The total spine density, mature, immature spine density. Results are presented as mean±SEM (5 dendritic segments from 1 animal. Three rats for each group). *** p<0.001 compared with the Normal group; ## p<0.01, ### p<0.001 compared with the Stress group, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using Adobe Photoshop V2019 and GraphPad Prism version 7.0 software.
Figure 5. Analysis of spine density in PFC layer V pyramidal neurons (Golgi-Cox staining, Scale bar: 5 μm). (A) Apical dendrites in layer V pyramidal neurons of the PFC, the prelimbic subregions (PrL) were observed. Representative images are shown of high-magnification projections of layer V pyramidal neuron apical tuft dendrites (scale bar: 5 μm). (B–D) The total spine density, mature, immature spine density. Results are presented as mean±SEM (5 dendritic segments from 1 animal. Three rats for each group). *** p<0.001 compared with the Normal group; ## p<0.01, ### p<0.001 compared with the Stress group, ^^^ p<0.001 compared with the Phys+stress group. The figure was created using Adobe Photoshop V2019 and GraphPad Prism version 7.0 software. Figure 6. The expression of BDNF and synaptic proteins in the PFC of each group. (A) Respective immune exposures to BDNF, GluR1, GluR2, PSD95, Synapsin I, and β-actin. (B) The levels of BDNF, GluR1, GluR2, PSD95, and synapsin I in the PFC. Results are the mean±SEM (n=6 rats per group, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The Figure was created using Adobe Photoshop V2019 and GraphPad Prism version 7.0 software.
Figure 6. The expression of BDNF and synaptic proteins in the PFC of each group. (A) Respective immune exposures to BDNF, GluR1, GluR2, PSD95, Synapsin I, and β-actin. (B) The levels of BDNF, GluR1, GluR2, PSD95, and synapsin I in the PFC. Results are the mean±SEM (n=6 rats per group, *** p<0.001 compared with the Normal group; # p<0.05, ## p<0.01, ### p<0.001 compared with the Stress group, ^^ p<0.01, ^^^ p<0.001 compared with the Phys+stress group. The Figure was created using Adobe Photoshop V2019 and GraphPad Prism version 7.0 software. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








