16 August 2021: Clinical Research
Association Between Serum Albumin Levels and Obesity and Risk of Developing Chronic Kidney Disease Using Data from the Korean Multi-Rural Communities Cohort (MRCohort) Population Database
Kwang Ho Mun1ABCDEFG*DOI: 10.12659/MSM.933840
Med Sci Monit 2021; 27:e933840
Abstract
BACKGROUND: The Multi-Rural Communities Cohort (MRCohort) was established as part of the Korean Genome and Epidemiology Study, which identified risk factors for cardiovascular diseases in the population between January 2005 and December 2011. This was a prospective study, aimed to investigate the association between serum albumin levels and the risk of developing chronic kidney disease (CKD) in obese patients using data from the MRCohort population database.
MATERIAL AND METHODS: For analysis, we selected a total of 9695 people from the MRCohort from 2004 to 2013: 3105 people from Goryeong, 3183 from Yangpyeong, and 3407 from Namwon, without previous history of myocardial infarction, cerebrovascular disease, cancer, and CKD, or missing values on confounders. As a result, 2300 metabolically healthy participants were studied as a whole, and then by body mass index of >23 kg/m². The groups were divided into 4 subgroups by albumin quartiles, and the effects of albumin levels compared to the lowest quartile were studied by Cox proportional hazard analysis, after adjusting for potential confounders.
RESULTS: The results showed that the highest quartile of albumin, compared to the lowest quartile, was a protective factor of CKD development only in the overweight and obese group with adjusted hazard ratio (HR)=0.38 (95% confidence interval (CI) 0.16-0.95, p<0.05). In the normal-weight group, results were insignificant, with adjusted HR=0.54 (95% CI 0.24-1.25, not significant).
CONCLUSIONS: Data from the Korean MRCohort population database showed that obesity and increased serum albumin levels were protective factors for the development of CKD.
Keywords: Cohort Studies, Kidney Failure, Chronic, Serum Albumin, Body Mass Index, Obesity, Asians, Cardiovascular Diseases, Glomerular Filtration Rate, overweight, Proportional Hazards Models, Prospective Studies, Renal Insufficiency, Chronic, Republic of Korea, Risk Assessment, Risk Factors, Rural Population
Background
The Multi-Rural Communities Cohort (MRCohort) was established as part of the Korean Genome and Epidemiology Study (KoGES), which identified risk factors for cardiovascular diseases in the population between January 2005 and December 2011 [1]. The cohort consists of 3 regions, Goryeong, Yangpyeong, and Namwon, each located in southeastern, northwestern, and southwestern areas of Korea, respectively. Most of the subjects were farmers and housewives, and all were age 40 years or older [1]. Therefore, this population was suitable for studying long-term effects of various factors on chronic diseases, including chronic kidney disease (CKD).
CKD is a major global health problem, with 2 million or more patients in renal replacement therapy, and more than 10 times this number in need [2]. The most common cause of CKD was diabetes (48%), and hypertension (21.2%), with other causes including old age, obesity, inflammation, abnormal levels of uric acid, and rural residential area [3,4]. Therefore, preventing and predicting CKD development is a major public health problem.
Albumin levels tend to be low in CKD patients and are a predictor of poor clinical outcomes [5,6]. Although the mechanisms by which hypoalbuminemia is associated with mortality and other clinical outcomes are unclear, albumin is an acute-phase reactant, and this could be associated with clinical outcomes [5,6]. Albumin levels of <3.8 g/dL, if corrected, could prevent 19% of patient deaths in maintenance hemodialysis patients [5]. Furthermore, hypoalbuminemia was associated with inflammation and mortality in peritoneal dialysis patients [5,6]. Low serum albumin levels have been known to increase poor outcomes in CKD patients, as seen in various studies, but few studies have reported an association with CKD development, and this needs further research [5,6].
Several studies have shown a positive relationship between obesity and CKD [7]. A study has shown a protective effect of high body mass index (BMI) on short-term and long-term mortality [8]. Also, another study has shown lower mortality risk in old people, compared to normal-weight [9]. However, the effect of obesity on CKD development has been challenged by various studies [10–12]. The obesity paradox, a phenomenon observed in CKD patients, reports better outcomes in obese patients [10]. The diabetic CKD group had a lower prevalence of obesity compared to the non-CKD group in study in Thailand [11]. A meta-analysis suggested that in CKD patients, in both predialysis and dialysis patients, BMIs equivalent to overweight and obese were associated with longer survival [12]. In conclusion, obesity has been associated with better clinical outcomes of CKD, regardless of treatment or patient status.
Recent studies have classified obesity with low levels of metabolic abnormalities as metabolically obese, thereby classifying patients into metabolically healthy non-obese, metabolically healthy obese, metabolically unhealthy non-obese, and metabolically unhealthy obese [13]. Metabolically healthy patients are free of metabolic abnormalities, including hypertension, insulin resistance, and dyslipidemia [13]. However, whether a metabolically healthy obese state is benign or has an effect on mortality and clinical outcomes such as CKD are debatable [14]. In metabolically healthy patients, the risk of CKD development is controversial, with both reports on lower and higher risk of CKD development [15,16]. In a Japanese study, metabolically healthy subjects were not associated with CKD development [14]. On the other hand, studies have shown that metabolically healthy status increased CKD development, even in subjects with low-grade inflammation [15,16].
Several studies have reported an association between obesity and serum albumin levels. Obesity could reflect malnutrition and deficiency in various nutritional factors, including antioxidants [17,18]. These factors of obesity might result in hypoalbuminemia, as albumin decreases in chronic inflammation and malnutrition [19]. As obesity affects serum albumin levels through various pathways, the presence of obesity should be considered when studying the effect of serum albumin on CKD development.
A study regarding albumin as an indicator of nutritional status showed that the combined effect of low BMI and low albumin is a risk factor of CKD development [20] and that BMI or albumin alone was insufficient factor of CKD development. However, the participants were predialysis patients, with baseline characteristics of the study including mean blood pressure of 140.5/78.2 mmHg and hemoglobin of 11.9 g/dL [20]. Another study has shown that albumin and BMI to be independent risk factors of mortality in hemodialysis patients [21]. There are no published studies on albumin, BMI, and CKD in the general population. Therefore, further study is needed to understand these relationships in the general population.
To sum up, a few studies have shown an association between serum albumin and clinical outcomes in CKD patients, regardless of treatment or severity [5,6]. However, the effect of albumin on CKD development itself is yet to be clarified. Furthermore, the effect of obesity is also unclear, and diseases affecting metabolic health such as diabetes and hypertension are major contributors to CKD development. Thus, the present study hypothesized that high levels of serum albumin protect against CKD development and kidney function decline, and that the effect of albumin differs by obesity status, after excluding metabolically unhealthy individuals. Therefore, this study aimed to investigate the association between serum albumin levels and the risk of developing CKD in obese patients using data from the MRCohort population database.
Material and Methods
STUDY POPULATION:
The study participants were recruited from the MRCohort, a cohort started as a part of the Korean Genomic and Epidemiology Study, which began in 2004 [1]. The study was conducted in 3 communities: Goryeong, Yangpyeong, and Namwon. Candidate villages were chosen by multistage cluster sampling, and participants age 40 years and older were recruited. The baseline study was conducted until 2009, with 9695 participants recruited. Participants were followed up every 2–4 years. As of 2013, 7020 participants have returned for follow-up.
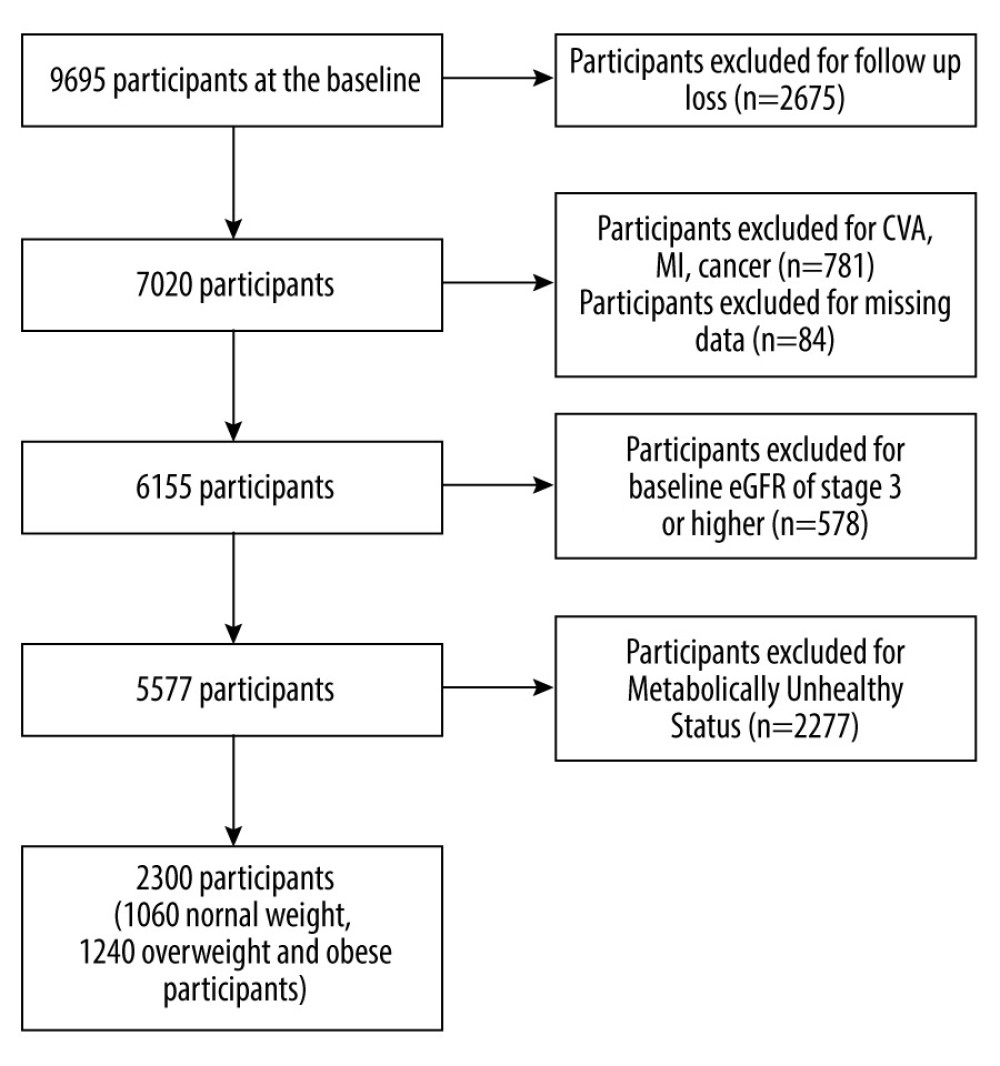
The patient selection is shown in Figure 1. From 9695 participants, 2675 participants without follow-up data were excluded. Furthermore, 781 participants were excluded due to presence of myocardial infarction, cerebrovascular disease, or cancer at the baseline examination. We also excluded participants without laboratory data, including albumin, creatinine, total cholesterol, triglyceride, high-density lipoprotein (HDL), C-reactive protein (CRP), aspartate transaminase (AST), alanine transferase (ALT), and gamma- glutamyltransferase (γ-GTP), fasting glucose levels were excluded from the study. Participants with void information on lifestyle factors (smoking, drinking, exercise), and other confounding variables such as BMI, systolic blood pressure (SBP), and diastolic blood pressure (DBP). Participants with baseline estimated glomerular filtration rate (eGFR) of 60 mL/min/1.73 m2 or lower were excluded, as it is equivalent to CKD stage of 3 or higher. Lastly, participants defined as metabolically unhealthy were excluded because metabolic abnormalities such as diabetes and hypertension are major causes of CKD development. As a result, 2300 participants were used for the study, of which consisted 1060 were normal-weight (BMI <23 kg/m2) and 1240 were overweight and obese (BMI ≥23 kg/m2).
DATA COLLECTION:
Standardized protocols were used for questionnaires and examinations, including questionnaires on general characteristics, blood pressure, blood sampling, and measuring of anthropometric measures [1]. All of the questionnaires and examinations were performed by trained interviewers and examiners who were trained by the same coordinating center staff.
Structured questionnaire research was performed, asking questions on demographic information including identification number, age, and gender. Lifestyle patterns, including smoking, drinking and exercise status, classified by non, ex, and current, were assessed. The information on previous history of disease and medicine, including hypertension (HTN), diabetes (DM), myocardial infarction, cerebrovascular disease, and cancer, were also collected by questionnaire. Patients with current or former history of disease and medicine were asked to answer by “non-“, “ex-“, and “current” status of medical history.
The food frequency questionnaire (FFQ) was used to collect dietary data. For each 108 food items, frequency (never to 3 times/day) and normal intake amount was collected. Duration of intake (3, 6, 9, 12 months) was also collected for seasonal items. Nutrient intake, such as calorie and protein intake, was calculated from the total of all food and dietary supplement consumption.
Anthropometric measurements were obtained by examiners who were trained in a single center, using the standard protocol. Height and weight were obtained by standard height and weight scales, with zero-balancing before each measurement. Height was measured to the nearest 0.1 cm, and weight was measured to the nearest 0.01 kg. BMI was calculated as weight (kg) divided by height squared (m2). Participants were categorized by BMI: BMI of <23 kg/m2 as the normal group and BMI of ≥23 kg/m2 as the overweight and obese group.
Blood pressure was measured in seated position, from the right arm at the heart level using a standard sphygmomanometer. Participants were measured after resting of 10 min, followed by 2 consecutive measures with 5-min intervals. SBP and DBP were obtained by the average of the 2 measurements to the nearest 2 mmHg. If the difference of the 2 measures was greater than 5 mmHg, an additional measurement was performed. HTN was defined as blood pressure of >140/90 mmHg, or the use of antihypertensives, or prior diagnosis of HTN in other clinics.
Laboratory data were collected from blood samples, which were collected after 8 h of overnight fasting. All of the samples were analyzed on the same day. Total cholesterol, HDL, triglyceride (TG), fasting glucose, WBC (white blood cell), CRP, AST, ALT, γ-GTP, albumin, and creatinine were measured using an ADVIA 1650 Automated Analyzer (Siemens, New York, NY, USA) [1].
DEFINITION OF RENAL FUNCTIONS:
eGFR was obtained using the CKD-EPI equation, which has been validated elsewhere [22]. eGFR of 60 mL/min/1.73 m2 or lower at the time of follow-up was defined as CKD development. All of the participants were followed up until CKD development or until the final follow-up examination.
DEFINITION OF METABOLIC HEALTH:
The standard ATP-III protocol was used to define metabolically healthy status [23]. As BMI has strong collinearity with waist circumference, the waist was excluded from the definition criteria. Participant with fewer than 2 of the following criteria were defined as metabolically healthy: 1) blood pressure of 130/85 mmHg or higher, or diagnosed of HTN, or taking HTN medication; 2) fasting glucose of 100 mg/dL or higher, or diagnosed of DM, or taking DM medication; 3) TG of 150 mg/dL or higher; 4) HDL of 40 mg/dL or lower in men, or 50 mg/dL or lower in women.
STATISTICAL ANALYSIS:
Participants were studied according to Q1–4 by albumin levels of quartile cut-off levels (<4.3 mg/L, 4.3–4.4 mg/L, 4.5–4.6 mg/L, >4.6 mg/L). Furthermore, participants were divided into 4 groups according to high albumin (Q4), and BMI of 23 kg/m2; G1, high albumin, low BMI group; G2, high albumin, high BMI group; G3, low albumin, low BMI group; G4, low albumin, and high BMI group. Lastly, participants were divided into Q1–4 in each overweight and obese group and normal-weight group, with identical cut-off points. Lifestyle factors, laboratory data, and other confounders were compared between each group using one-way analysis of variance for continuous variables and chi-square test for categorical variables. Results are shown as mean±standard deviation (SD) for continuous variables, while the outcome was CKD development.
Cox proportional hazard analysis was used to obtain hazard ratios (HR) and 95% confidence interval (CI) to assess the effect of albumin and obesity on CKD development. Firstly, the effects of albumin levels compared to the lowest quartile (Q1) in the whole participants were studied. Secondly, the combined effect of albumin and obesity was studied in all participants, with the high albumin and low BMI group as a reference. Thirdly, in both obese and normal-weight groups, the effect of albumin was studied by albumin quartiles, with lowest albumin (Q1) as a reference in each group. The analyses were adjusted for confounders significantly associated in the analysis, or were strong confounders in other studies. In Model 1, no confounders were adjusted. In Model 2, age (by every 10 years), gender, smoking, drinking, exercise, total cholesterol, triglyceride, HDL, CRP, AST, ALT, γ-GTP, WBC, and CRP were further adjusted. In Model 3, the effect of albumin was further adjusted by calorie and protein, by recommended amount, to adjust for dietary components. A
Results
The baseline characteristics of the participants by obesity are shown in Table 1. During the average follow-up period of 47.2±19.0 months, 156 people (6.8%) developed CKD. The average age of the participants was 59.3±9.6 years, and 38.9% were men. Among the participants, the blood pressure was 117.3±15.1/75.7±9.0 mmHg, with fasting glucose of 92.7±17.7 mg/dL. Furthermore, 14.3% were current smokers, 29.2% were current drinkers, and 29.2% were actively exercising among the participants. The average level of serum albumin was 4.4±0.2 mg/L. The prevalence of CKD in normal-weight participants was 5.7%, while the prevalence was 7.7% in overweight and obese participants. However, the difference in eGFR decline was not statistically significant, as 12.1% in normal-weight and 11.4% in overweight and obese participants were reported with eGFR decline.
In normal-weight participants, age, HDL, AST, baseline eGFR, number of men, and smokers were higher compared to obese participants. Baseline eGFR was 78.9±9.4 mL/min/1.73 m2 in the normal group, compared to 78.1±9.5 mL/min/1.73 m2 in the obese group. In the obese group, follow-up time, total cholesterol, TG, ALT, number of drinkers, and number of exercisers were higher compared to the normal group. Calorie and protein intake were also higher in the obese group, and albumin levels were also higher in the obese group (
The participants were divided into 4 groups according to the albumin and BMI levels, as shown in Table 2. A lower CKD development of 3.8% was observed in the high albumin low BMI group (G1), compared to the low albumin high BMI group (G4) of 9.0% (p<0.01). Compared to G1, higher rates of drinking, ALT, and calorie and protein intake were observed in G4. G1 had longer follow-up time, number of men, smokers, and exercisers. SBP, DBP levels were higher in G1 compared to G4. Furthermore, total cholesterol, TG, and WBC levels were also higher in G1 compared to G4.
Table 3 shows the effect of albumin on CKD development. Compared to lowest albumin level (Q1), CKD development decreased in highest albumin levels (Q4) with adjusted HR=0.49 95% CI 0.25–0.93 (p<0.05). The joint effect of albumin on CKD development by albumin and obesity is shown in Table 4. Compared to the high albumin low BMI group (G1), higher CKD development was observed in the low albumin high BMI group (G4), with adjusted HR=2.66, 95% CI 1.35–5.24 (
Table 5 shows the effect of albumin on CKD development in the overweight and obese groups and in the normal-weight group. Compared to lower quartile of albumin (Q1), lower CKD development was observed in the highest albumin level (Q4) with adjusted HR= 0.38, 95% CI 0.16–0.95 (
Discussion
Data from the Korean MRCohort population database showed that obesity and increased serum albumin levels were protective factors for CKD development. Compared to the low BMI high albumin group, only the high BMI low albumin group showed higher CKD development. Moreover, the effect of albumin was consistent only in the overweight obese group. Finally, the effect of albumin levels was compared in overweight and obese and normal-weight patients, with albumin levels having an effect on CKD development only in overweight and obese groups.
CKD development, defined by the CKD-EPI equation [22], was 10.4% in the whole group, and 6.8% in the metabolically healthy population, during 4-year follow-up period. In Korea, the prevalence of CKD in adults age ≥30 is 4.1% [4], but the prevalence of CKD in those ages 60–69 and ≥70 is 7.9% and 20.4%, respectively. Thus, the prevalence of CKD increases dramatically as age increases, especially in old age. Moreover, other studies show similar results compared to our study. During an average follow-up of 4 years, 7.6% of the population developed CKD, with an average age of 45.4 years [24]. Another study with an average follow-up of 6 years, have shown 17.9% of CKD development across the population [25]. Therefore, the CKD development of the study is plausible, with similarity and explicability by statistics on the Korean general population as well as studies of foreign populations.
Albumin function is an indicator of nutritional status and actions of antioxidants. The serum levels of albumin are changed by several factors, including reduction of albumin synthesis in the liver, due to cirrhosis, liver failure, or cancer [17,26]. In the study, albumin was shown to reduce WBC levels in the overweight and obese group, compared to the normal-weight group, even though the baseline WBC and CRP levels were higher in the overweight and obese group (data not shown). These results suggest the role of inflammation in CKD development, as well as serum albumin levels.
However, in metabolically healthy participants, CRP levels showed an insignificant association with albumin level or CKD development. There are other explanations of CKD development, suggesting diverse roles of albumin independent of inflammation [3]. Firstly, serum albumin might represent true GFR levels, due to increased tubular creatinine secretion [27]. Secondly, low serum albumin might represent impairment of protein reabsorption, which has been filtrated in the renal tubules, therefore representing tubular damage prior to the decline of kidney function [28]. Thus, serum albumin levels might reflect various conditions of the human body resulting in CKD prior to its development.
Serum albumin of 3.4–3.5 g/dL was a cut-off point of outcomes in clinical practice, such as heart failure and mortality [5,29]. However, various studies have shown that albumin levels higher than 3.3 have clinical significance, such as 3.9 g/dL of end-stage renal disease development in patients with immunoglobulin A nephropathy, or 3.8 g/dL of increased kidney function decline in elderly patients [30,31]. Moreover, serum albumin less than 4.0 g/dL showed consistently high incidences of heart failure compared those with higher albumin levels [29]. In the REGARDS study, serum albumin levels of 4.0 g/dL or lower were associated with higher risk of CKD compared to high serum albumin levels [32]. These findings suggest that low serum albumin, even normal levels, should be considered as a risk factor in clinical practice.
Only metabolically healthy participants were enrolled in the present study. “Metabolically healthy” subjects are free of insulin resistance, hypertension, dyslipidemia, and inflammation, and have been reported to have lower mortality [13]. The criteria of metabolically healthy varies from study to study, with diabetes, hypertension, and dyslipidemia being common criteria used [33]. In the current study, metabolically healthy was defined based on results of previous studies with similar populations and outcomes [15,16]. The effect of metabolically healthy obesity on CKD development is controversial [14–16]. The present study shows that metabolically healthy obese patients have increased risk of developing CKD. The specific mechanisms linking obesity and CKD regardless of metabolic abnormalities need to be identified, although some studies have suggested hemodynamic factors and burden on nephrons as potential factors [7,15].
The reason why BMI affects CKD development only in people with low albumin levels might involve various causes. As obesity has ambivalent effects on CKD development, other indicators, such as albumin, should be considered. Albumin, which is an indicator of various conditions such as nutritional status and inflammation, should be considered in obese patients to understand the true conditions when predicting clinical outcomes.
There are several strengths to this study. Firstly, this is the first study to explore the effect of albumin on the development of CKD, especially between obese and non-obese patients in Korea. Secondly, the study included a large number of participants from a rural cohort study, unlike other Korean studies which had recruited participants from health examinations in medical centers [15,16]. The current cohort consists of 3 different rural areas, located in northern, western, and eastern parts of Korea, allowing more generalization of study results to rural communities. Lastly, as this was a large, prospective study, a strong causal relationship between serum albumin and CKD development is suggested.
However, there are some limitations of the study that should be considered. First, the current study has defined CKD by eGFR, without considering albuminuria or other factors that define CKD, due to lack of data in the cohort database. However, eGFR of <60 mL/min/1.73 m2 has shown to be an acceptable definition of CKD in population-based studies [34]. Secondly, the current study followed the participants every 2–4 years. However, the exact time of CKD development within the follow-up period of the participants is unknown, which might alter the time of CKD development. Lastly, due to the relatively short follow-up period, generalization of the study results should be confirmed in a long-term follow-up study, although various studies have used similar follow-up periods of 4–6 years [24,25].
Conclusions
Data from the Korean MRCohort population database showed that obesity and increased serum albumin levels were protective factors for the development of CKD.
Tables
Table 1. Baseline characteristics of the participants by obesity.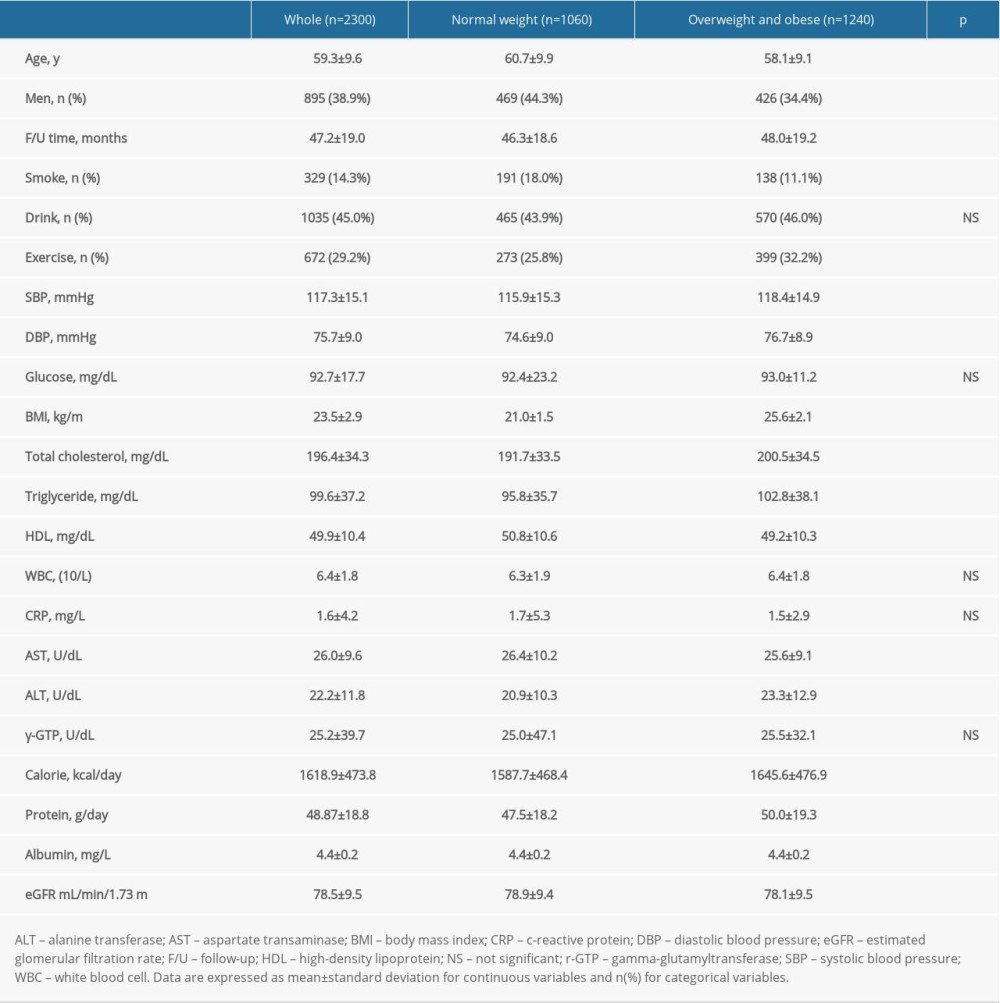 Table 2. Baseline characteristics of the participants by albumin and obesity.
Table 2. Baseline characteristics of the participants by albumin and obesity.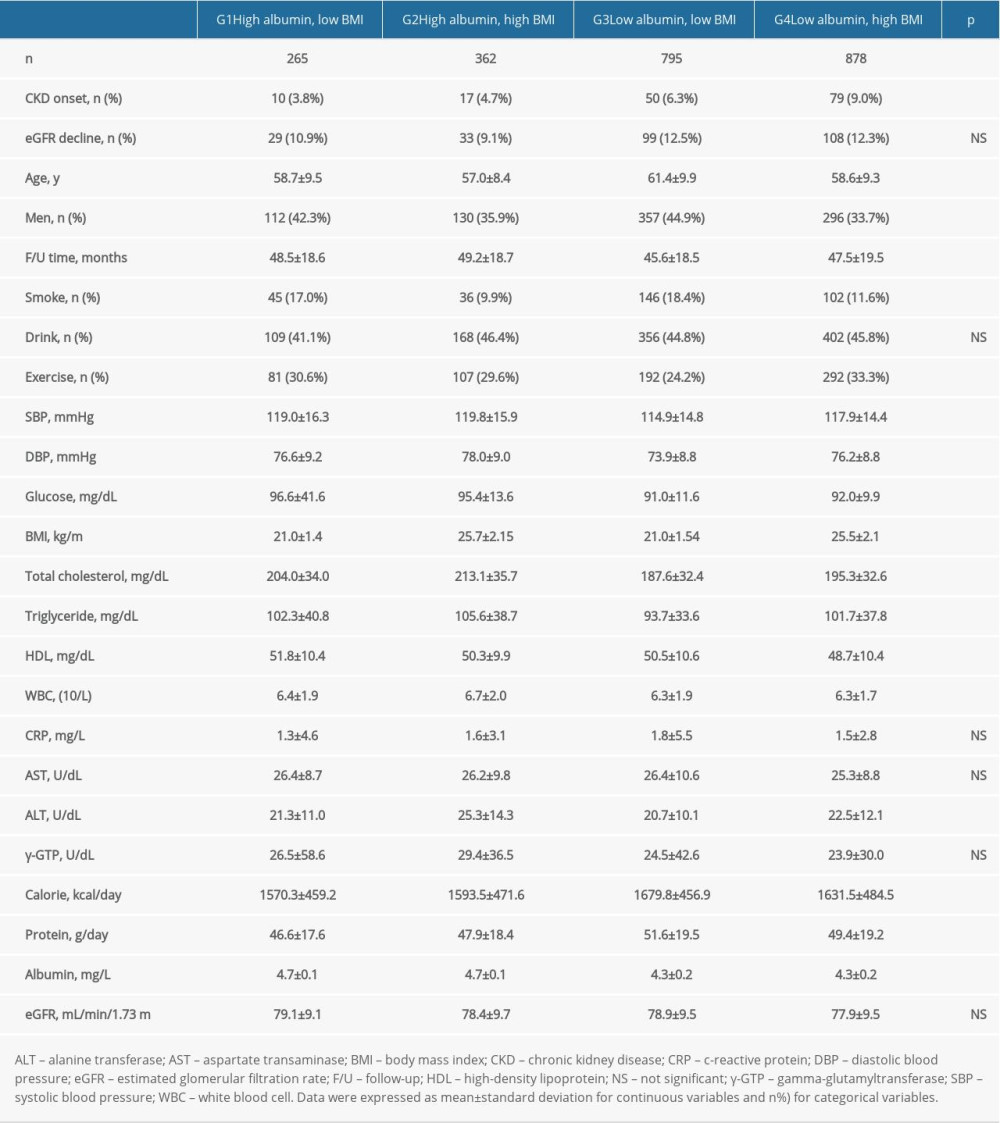 Table 3. Hazard ratios of chronic kidney disease development according to albumin quartiles in all participants.
Table 3. Hazard ratios of chronic kidney disease development according to albumin quartiles in all participants.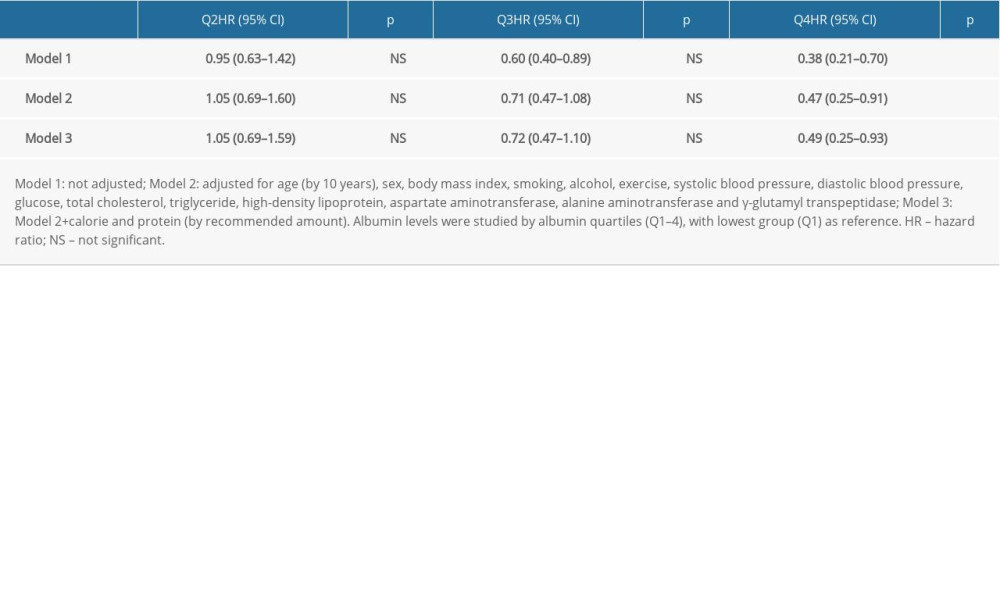 Table 4. Hazard ratios of chronic kidney disease development according to albumin and obesity.
Table 4. Hazard ratios of chronic kidney disease development according to albumin and obesity.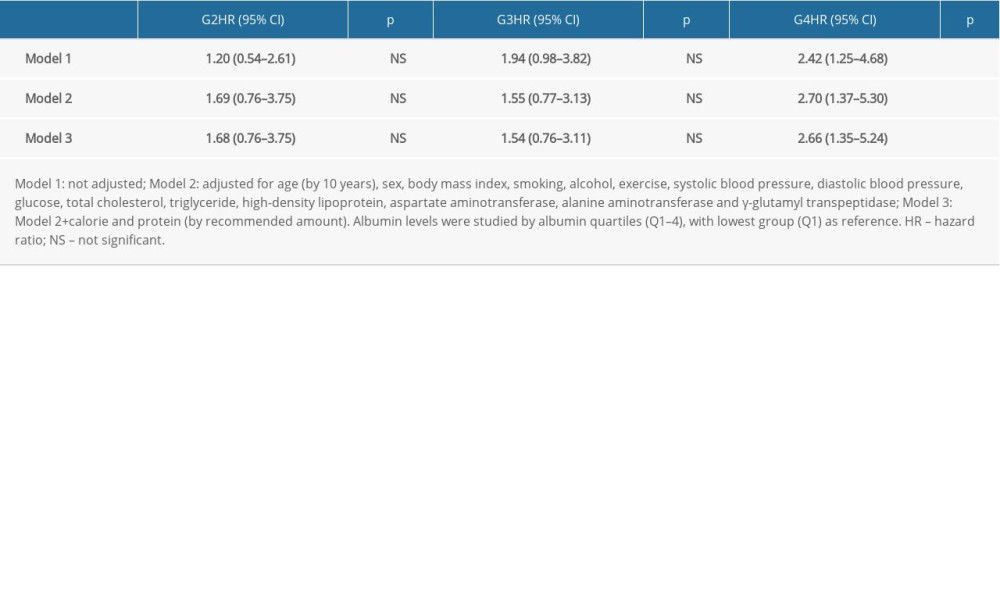 Table 5. Hazard ratios for risk of CKD development according to quartiles of albumin.
Table 5. Hazard ratios for risk of CKD development according to quartiles of albumin.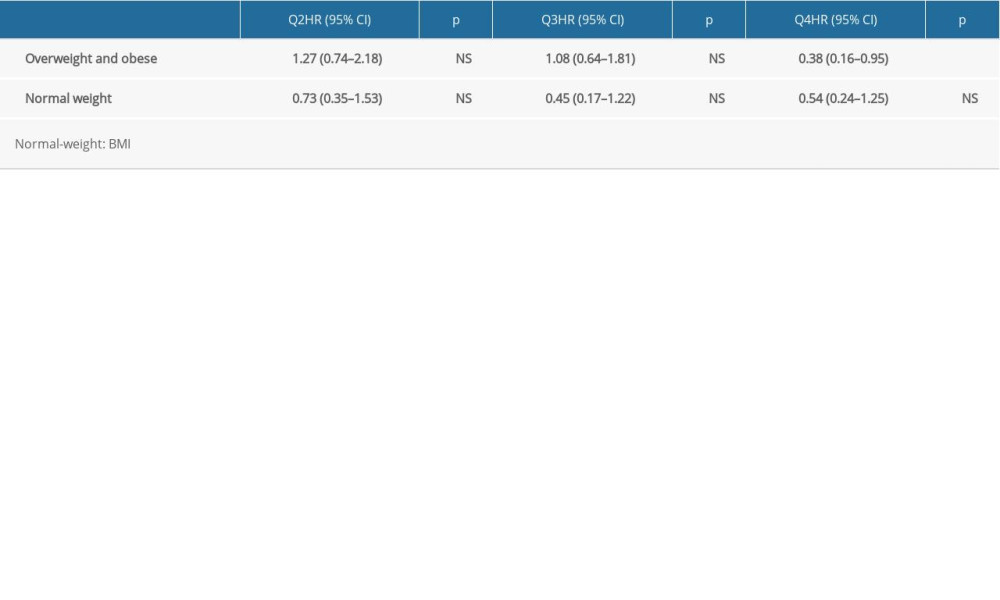
References
1. Kim Y, Han BGKoGES Group, Cohort profile: The Korean Genome and Epidemiology Study (KoGES) Consortium: Int J Epidemiol, 2017; 46; e20 [Erratum in: Kim Y, Han BG, KoGES group. Cohort profile: The Korean Genome and Epidemiology Study (KoGES) Consortium. Int J Epidemiol. 2017;46(4):1350]
2. Couser WG, Remuzzi G, Mendis S, Tonelli M, The contribution of chronic kidney disease to the global burden of major noncommunicable diseases: Kidney Int, 2011; 80; 1258-70
3. Amdur RL, Feldman HI, Gupta J, Inflammation and progression of CKD: The CRIC study: Clin J Am Soc Nephrol, 2016; 11; 1546-56
4. Park JI, Baek H, Jung HH, Prevalence of chronic kidney disease in Korea: The Korean National Health and Nutritional Examination Survey 2011–2013: J Korean Med Sci, 2016; 31; 915-23
5. Kalantar-Zadeh K, Kilpatrick RD, Kuwae N, Revisiting mortality predictability of serum albumin in the dialysis population: Time dependency, longitudinal changes and population-attributable fraction: Nephrol Dial Transplant, 2005; 20; 1880-88
6. Perez-Fontan M, Rodriguez-Carmona A, Garcia-Naveiro R, Peritonitis-related mortality in patients undergoing chronic peritoneal dialysis: Perit Dial Int, 2005; 25; 274-84
7. Ejerblad E, Fored CM, Lindblad P, Obesity and risk for chronic renal failure: J Am Soc Nephrol, 2006; 17; 1695-702
8. Ertilav M, Levin WN, Celtik A, Impact of body mass index on short-term and long-term survival in prevalent hemodialysis patients: Hemodial Int, 2019; 23; 375-83
9. Dahl AK, Fauth EB, Ernsth-Bravell M, Body mass index, change in body mass index, and survival in old and very old persons: J Am Geriatr Soc, 2013; 61; 512-18
10. Fung F, Sherrard DJ, Gillen DL, Increased risk for cardiovascular mortality among malnourished end-stage renal disease patients: Am J Kidney Dis, 2002; 40; 307-14
11. Kittiskulnam P, Thokanit NS, Katavetin P, The magnitude of obesity and metabolic syndrome among diabetic chronic kidney disease population: A nationwide study: PLoS One, 2018; 13; e0196332
12. Ahmadi SF, Zahmatkesh G, Ahmadi E, Association of body mass index with clinical outcomes in non-dialysis-dependent chronic kidney disease: A systematic review and meta-analysis: Cardiorenal Med, 2015; 6; 37-49
13. Phillips CM, Metabolically healthy obesity: Definitions, determinants and clinical implications: Rev Endocr Metab Disord, 2013; 14; 219-27
14. Hashimoto Y, Tanaka M, Okada H, Metabolically healthy obesity and risk of incident CKD: Clin J Am Soc Nephrol, 2015; 10; 578-83
15. Jung CH, Lee MJ, Kang YM, The risk of chronic kidney disease in a metabolically healthy obese population: Kidney Int, 2015; 88; 843-50
16. Chang Y, Ryu S, Choi Y, Metabolically healthy obesity and development of chronic kidney disease: A cohort study: Ann Intern Med, 2016; 164; 305-12
17. Nelson CL, Elkassabany NM, Kamath AF, Liu J, Low albumin levels, more than morbid obesity, are associated with complications after TKA: Clin Orthop Relat Res, 2015; 473; 3163-72
18. Strauss RS, Comparison of serum concentrations of alpha-tocopherol and beta-carotene in a cross-sectional sample of obese and nonobese children (NHANES III). National Health and Nutrition Examination Survey: J Pediatr, 1999; 134; 160-65
19. Navaneethan SD, Kirwan JP, Arrigain S, Obesity, anthropometric measures and chronic kidney disease complications: Am J Nephrol, 2012; 36; 219-27
20. Kikuchi H, Kanda E, Mandai S, Combination of low body mass index and serum albumin level is associated with chronic kidney disease progression: The chronic kidney disease-research of outcomes in treatment and epidemiology (CKD-ROUTE) study: Clin Exp Nephrol, 2017; 21; 55-62
21. Takahashi R, Ito Y, Takahashi H, Combined values of serum albumin, C-reactive protein and body mass index at dialysis initiation accurately predicts long-term mortality: Am J Nephrol, 2012; 36; 136-43
22. Levey AS, Stevens LA, Schmid CH, A new equation to estimate glomerular filtration rate: Ann Intern Med, 2009; 150; 604-12
23. Hinnouho GM, Czernichow S, Dugravot A, Metabolically healthy obesity and risk of mortality: Does the definition of metabolic health matter?: Diabetes Care, 2013; 36; 2294-300
24. Toyama T, Furuichi K, Shimizu M, Relationship between serum uric acid levels and chronic kidney disease in a Japanese cohort with normal or mildly reduced kidney function: PLoS One, 2015; 10; e0137449
25. Mirmiran P, Nazeri P, Bahadoran Z, Dietary sodium to potassium ratio and the incidence of chronic kidney disease in adults: A longitudinal follow-up study: Prev Nutr Food Sci, 2018; 23; 87-93
26. Valerio C, Theocharidou E, Davenport A, Agarwal B, Human albumin solution for patients with cirrhosis and acute on chronic liver failure: Beyond simple volume expansion: World J Hepatol, 2016; 8; 345-54
27. Horio M, Imai E, Yasuda Y, Lower serum albumin level is associated with higher fractional excretion of creatinine: Clin Exp Nephrol, 2014; 18; 469-74
28. Otaki Y, Watanabe T, Takahashi H, Comorbid renal tubular damage and hypoalbuminemia exacerbate cardiac prognosis in patients with chronic heart failure: Clin Res Cardiol, 2016; 105; 162-71
29. Gopal DM, Kalogeropoulos AP, Georgiopoulou VV, Serum albumin concentration and heart failure risk: The health, aging, and body composition study: Am Heart J, 2010; 160; 279-85
30. Kawai Y, Masutani K, Torisu K, Association between serum albumin level and incidence of end-stage renal disease in patients with immunoglobulin A nephropathy: A possible role of albumin as an antioxidant agent: PLoS One, 2018; 13; e0196655
31. Lang J, Katz R, Ix JH, Association of serum albumin levels with kidney function decline and incident chronic kidney disease in elders: Nephrol Dial Transplant, 2018; 33; 986-92
32. Walther CP, Gutiérrez OM, Cushman M, Serum albumin concentration and risk of end-stage renal disease: The REGARDS study: Nephrol Dial Transplant, 2018; 33; 1770-77
33. Jung CH, Lee WJ, Song KH, Metabolically healthy obesity: A friend or foe?: Korean J Intern Med, 2017; 32; 611-21
34. Bash LD, Coresh J, Köttgen A, Defining incident chronic kidney disease in the research setting: The ARIC Study: Am J Epidemiol, 2009; 170; 414-24
Tables
 Table 1. Baseline characteristics of the participants by obesity.
Table 1. Baseline characteristics of the participants by obesity. Table 2. Baseline characteristics of the participants by albumin and obesity.
Table 2. Baseline characteristics of the participants by albumin and obesity. Table 3. Hazard ratios of chronic kidney disease development according to albumin quartiles in all participants.
Table 3. Hazard ratios of chronic kidney disease development according to albumin quartiles in all participants. Table 4. Hazard ratios of chronic kidney disease development according to albumin and obesity.
Table 4. Hazard ratios of chronic kidney disease development according to albumin and obesity. Table 5. Hazard ratios for risk of CKD development according to quartiles of albumin.
Table 5. Hazard ratios for risk of CKD development according to quartiles of albumin. Table 1. Baseline characteristics of the participants by obesity.
Table 1. Baseline characteristics of the participants by obesity. Table 2. Baseline characteristics of the participants by albumin and obesity.
Table 2. Baseline characteristics of the participants by albumin and obesity. Table 3. Hazard ratios of chronic kidney disease development according to albumin quartiles in all participants.
Table 3. Hazard ratios of chronic kidney disease development according to albumin quartiles in all participants. Table 4. Hazard ratios of chronic kidney disease development according to albumin and obesity.
Table 4. Hazard ratios of chronic kidney disease development according to albumin and obesity. Table 5. Hazard ratios for risk of CKD development according to quartiles of albumin.
Table 5. Hazard ratios for risk of CKD development according to quartiles of albumin. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387