31 December 2021: Clinical Research
A Prospective Observational Study of 42 Patients with COVID-19 infection and a History of Hepatitis C Virus Infection and Thyroid Disease with Follow-Up Thyroid Function and Autoantibody Testing
Dumitru Cristinel Badiu1B, Gabriel Cristian Popescu1F, Anca Zgura2A*, Adriana Mercan Stanciu3F, Mihai Daniel Dodot3F, Claudia Mehedintu4C, Letitia Toma3E, Simu Razvan3B, Xenia Bacinschi2D, Bogdan Haineala5ADOI: 10.12659/MSM.935075
Med Sci Monit 2021; 27:e935075
Abstract
BACKGROUND: Thyroiditis is an important extrahepatic association in chronic hepatitis C virus (HCV) infection. There have been reports of an association between SARS-CoV-2 infection and the onset or re-activation of autoimmune hypothyroidism. Therefore, we performed this prospective observational study of 42 patients with COVID-19 infection and a history of hepatitis C virus infection and thyroid disease with follow-up thyroid function and autoantibody testing.
MATERIAL AND METHODS: From April 2020 to October 2020, we performed a prospective observational study of patients with cured hepatitis C virus (HCV) infection and documented thyroid disease who became infected with SARS-CoV-2 (confirmed by SARS-CoV-2 RNA detection via reverse-transcription polymerase chain reaction [RT-PCT] from the upper respiratory tract, both nasal and pharyngeal swabs). Evaluation at 1 and 3 months after SARS-CoV-2 infection included serum determination of antithyroid antibodies (anti-thyroglobulin [anti-Tg] and antithyroid peroxidase [ATPO]), thyroid-stimulating hormone (TSH), free thyroxine (fT4), free triiodothyronine (fT3), and evaluation of thyroid medication, with dose adjustment if required.
RESULTS: One-month follow-up showed that both patients with autoimmune thyroiditis as well as patients without antibodies had increased ATPO levels. Also, levels of TSH, fT3, and fT4 were significantly decreased. At 3-month follow-up, levels of ATPO were decreased in all patient groups and the levels of thyroid hormones increased to normal values.
CONCLUSIONS: This study supports previous reports of an association between SARS-CoV-2 infection and thyroid dysfunction associated with thyroid autoantibodies. Thyroid function tests may be considered as part of the laboratory work-up in patients with COVID-19.
Keywords: ACE2 Protein, Human, Hepatitis C, Chronic, Thyroid Diseases, Adult, COVID-19, Female, Follow-Up Studies, Hepacivirus, Hepatitis C, Humans, Hypothyroidism, Male, Prospective Studies, RNA, Viral, Romania, SARS-CoV-2, Thyroid Function Tests, Thyroid Gland, Thyroiditis, Autoimmune, thyrotropin, thyroxine, Triiodothyronine
Background
The COVID-19 pandemic has proven an important test of the ability of the scientific medical world to effectively understand, describe, and manage pathophysiology processes when dealing with epidemics of newly-emerged pathogens. As time progresses, more and more studies are published reflecting the systemic impact of what was originally thought to be a respiratory infection [1]. It appears that COVID-19 can directly affect the cardiovascular, gastrointestinal (including the liver and the pancreas), renal, nervous, and musculoskeletal systems [2]. Furthermore, due to the abundance of ACE2 and TMPRSS2 receptors in the thyroid, required for internalization of the virus, it seems likely that SARS-CoV-2 can affect the thyroid directly, as well as in the context of an increased systemic inflammatory response [3]. The presence of SARS-CoV-2 virus in the thyroid tissue obtained by autopsy after death due to COVID-19 suggests a possible direct infection of the thyroid and may provide an explanation for the thyroid dysfunction in these patients [4].
A recent meta-analysis has found an association between SARS-CoV-2 infection and the triggering of Graves’ disease, as well as subacute thyroiditis [5]. Cases of autoimmune hypothyroidism have been described, but there were no indicators that the condition developed before or during SARS-CoV-2 infection [5]. One interesting article reported a temporal correlation between COVID-19 and thyroid damage: the first to develop is subacute thyroiditis, followed by Hashimoto’s disease (in patients with pre-existing autoimmunity), Graves’ disease (peak incidence at 4 weeks after infection), and painless thyroiditis in patients with pre-existing autoimmunity [6]. In this regard, a study published in 2021 including 122 patients without previous thyroid conditions found an increase in antithyroid peroxidase antibodies during COVID-19 and convalescence [7].
In the general population, subclinical hypothyroidism has a prevalence of 4.3–8.5%, while overt disease is found in 0.3–0.4%; it is commonly associated with environmental iodine deficiency and autoimmune thyroid diseases. Substitution therapy is recommended in overt disease [8].
Thyroid damage has been associated with acute conditions and several viral infections, among which hepatitis C virus (HCV) occupies an important place [9]. HCV chronic infection is associated with a multitude of extrahepatic manifestations, either by inflammatory or autoimmune mechanisms or even by direct infection of other organs, such as the thyroid [10–13]. In fact, thyroid involvement is considered the most frequent endocrine disorder associated with HCV chronic infection; mechanisms responsible for thyroid damage are thyroid destruction induced by an increased inflammatory response (resulting in autoimmune thyroiditis) or by direct HCV infection [14,15]. Current HCV guidelines recommend all oral antiviral therapies for chronic HCV infection, with benefits of liver function improvement, decline of progression toward fibrosis and cirrhosis, and improvement of HCV extrahepatic manifestations [16].
Therefore, this prospective observational study of 42 patients with SARS-CoV-2 infection and a history of hepatitis C virus infection and thyroid disease evaluated thyroid dysfunction with follow-up thyroid function and autoantibody testing.
Material and Methods
The study was performed following STROBE guidelines and was approved by the local Ethics Committee (Fundeni Clinical Institute Ethics Committee approval no 3513/2020). On hospital admission, patients signed inform consent forms for blood analysis and the use of their data in scientific research, under the condition of anonymity.
This study aimed to determine the effects of COVID-19 infection on thyroid diseases associated with HCV chronic hepatitis. From April 2020 to October 2020, we performed a prospective observational study on patients with cured HCV infection and documented thyroid disease who became infected with SARS-CoV-2. We routinely monitor patients with chronic HCV infection every 6 months, even after obtaining sustained virologic response after direct-acting antiviral therapy, in accordance with current guidelines [16,17].
Inclusion criteria for the current study were: history of HCV chronic hepatitis, with undetectable HCV-RNA at 3 months after direct-acting antiviral therapy (either ombitasvir/paritaprevir/ritonavir and dasabuvir or ledipasvir/sofosbuvir); documented thyroid disease (autoimmune thyroiditis, hyperthyroidism or hypothyroidism) with antibody and hormonal determination within 6 months before SARS-CoV-2 infection; and documented SARS-CoV-2 infection cured within 1 month before the first visit.
We excluded patients who had a history of thyroid cancer associated with HCV chronic infection; hepatitis B virus or HIV co-infection; changes in thyroid substitution therapy or antithyroid drugs within 6 months of the first visit; decompensated cirrhosis, solid or hematological neoplasia.
Demographic and medical data were retrieved from electronic source documents. Patients came from the south-eastern part of Romania, which is well known for a high prevalence of iodine deficiency-induced goiter [18]. The diagnosis of HCV infection was established by the presence of anti-HCV antibodies and a history of negative viremia after direct antiviral treatment. SARS-CoV-2 RNA detection was performed on nasal and pharyngeal swab samples by reverse-transcription-polymerase chain reaction (RT-PCR) using the Cobas® SARS-COV-2 RT-PCR test (Roche Diagnostics, Basel, Switzerland). Evaluation at 1 and 3 months after SARS-CoV-2 infection included serum determination of antithyroid antibodies (anti-thyroglobulin [anti-Tg] and antithyroid peroxidase [ATPO]) thyroid-stimulating hormone (TSH), free thyroxine (fT4), free triiodothyronine (fT3), and evaluation of thyroid medication, with dose adjustment if required. TSH, fT3, fT4, and anti-Tg antibodies were determined using electrochemiluminescence assays (normal values 0.27–4.2 μUI/mL for TSH, 2.2–4.4 pg/ml for fT3, 12–22 pmol/l for fT4, and less than 115 IU/ml for anti-Tg), while ATPO was determined using chemiluminescence with microparticles (normal values less than 34 IU/ml).
Data were evaluated using SPSS 18.0 (SPSS, Inc., Chicago, IL, USA) statistical software. Numerical values are expressed as mean±standard deviation. ANOVA was performed to compare values at the 3 evaluation visits, with statistical significance set at a
Within our study group, patients with evidence of thyroid hormone impairment were defined as either having hypothyroidism or hyperthyroidism (regardless of the underlying mechanism and regardless of the presence/absence of autoimmune antibodies). We only used the term autoimmune thyroiditis if the patient had euthyroidism and positive autoimmune antibodies.
Results
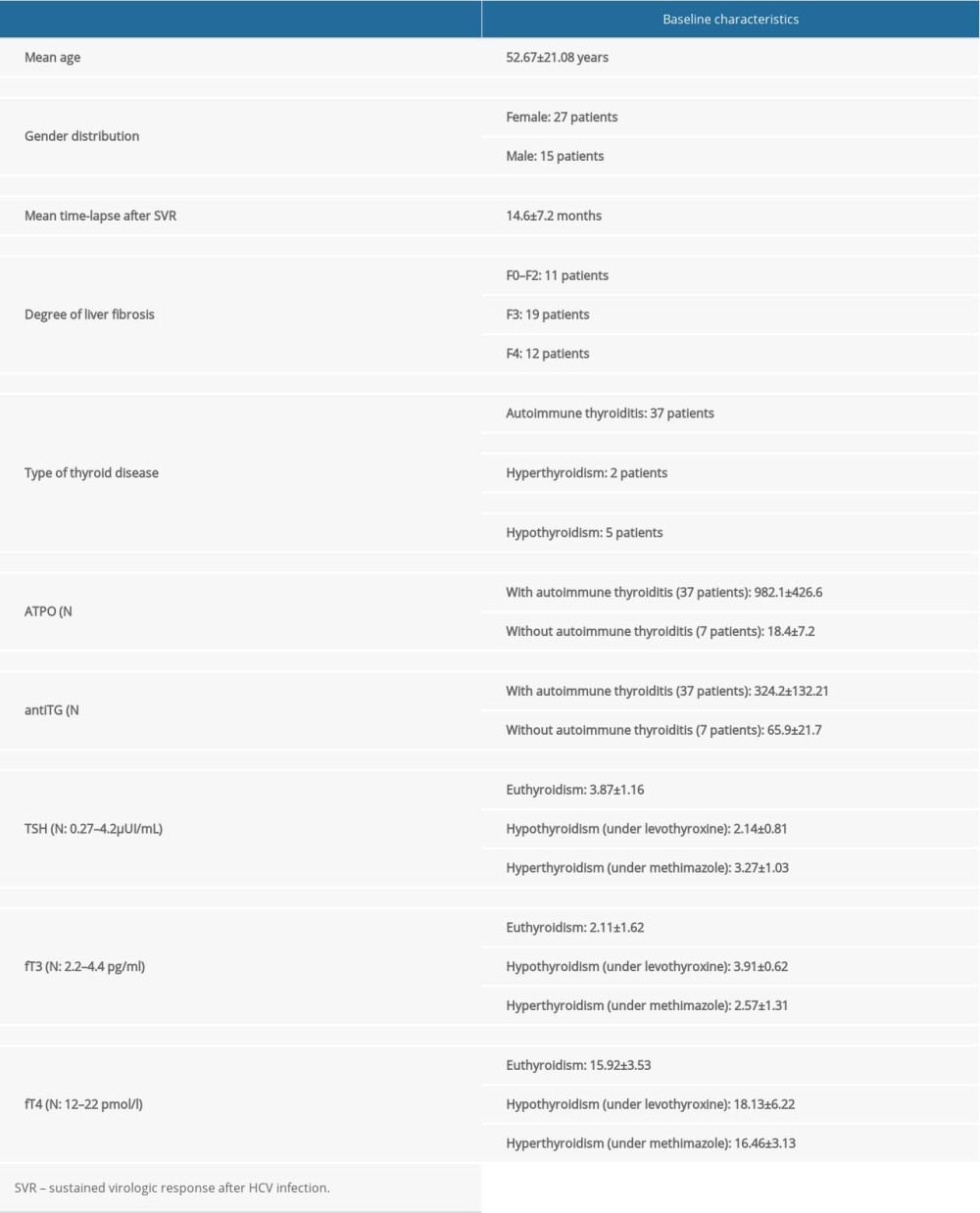
A total of 42 patients were included in the study, with a mean age of 52.67 years, and female patients representing 64.28%. Baseline characteristics of the study group are presented in Table 1. Out of the 42 patients, 7 patients were receiving medication for thyroid disease: 5 patients with hypothyroidism were receiving levothyroxine (mean dose of 55.4 mcg daily) and 2 patients with hyperthyroidism were receiving methimazole (mean dose 7.5 mg daily).
There were 36 patients with mild COVID-19 infection, while 6 patients had a moderate form of infection. The most frequent symptoms were myalgia (80.95%), fatigue (73.80%), fever (59.52%), headaches (50%), and digestive symptoms (54.76%).
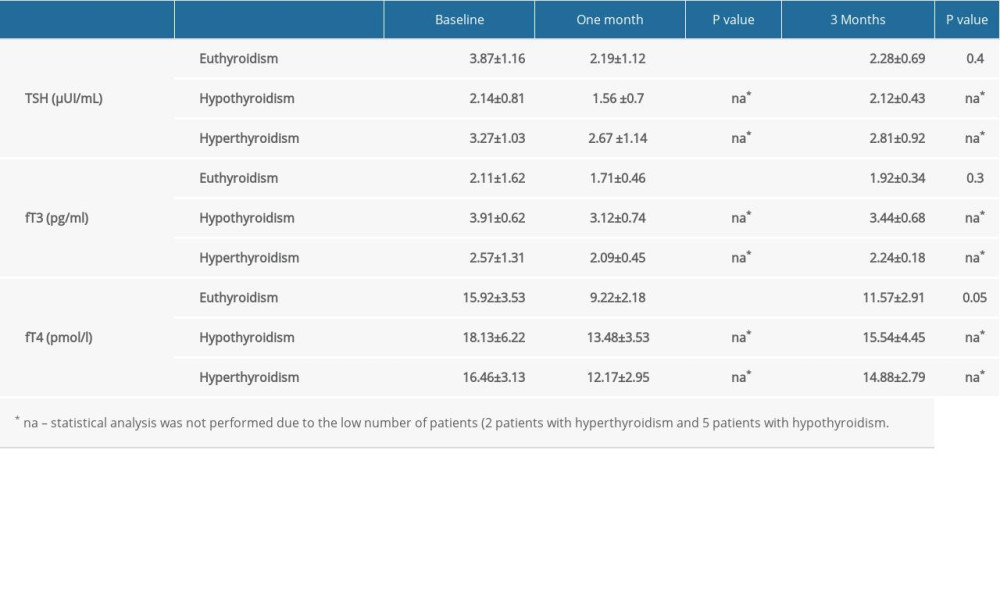
At 1-month follow-up, we found an increase in antithyroid antibodies, in patients with autoimmune thyroiditis as well as patients with baseline normal values of antibodies. In patients with autoimmune thyroiditis, mean values of ATPO were 1187.7±285IU/ml (versus 982.1±426.6 IU/ml at baseline,
Moreover, out of the 5 patients receiving levothyroxine, 2 patients required discontinuation of substitution therapy. Both of the patients had been on a stable dose of 25 mcg/day for more than 6 months and had a decrease of TSH levels from 2.1 μUI/mL to 0.5 μUI/mL and from 1.92 μUI/mL to 0.7 μUI/mL, respectively. One patient who had been on a stable dose of methimazole of 2.5 mg daily for 4 months discontinued the therapy due to a TSH increase from 3.2 μUI/mL to 4.6 μUI/mL.
At 3-month follow-up, we noted a significant decrease in ATPO levels (84.13±25.12 IU/ml versus 123.7±32.8 IU/ml,
Discussion
The present study shows that COVID-19 infection influences the thyroid function and the presence and levels of antithyroid antibodies in patients with pre-existing conditions associated with chronic HCV infection. We found that at 1-month follow-up, levels of ATPO and anti-Tg antibodies were increased in patients with and without known autoimmune thyroiditis; the levels declined during the 3-month follow-up until close to baseline. Furthermore, during the acute phase, patients presented lower levels of TSH, fT3, and fT4; these also gradually increased to baseline levels at 3-month follow-up.
It is a well-known fact that there is a thyroid response associated with systemic inflammation; a “low triiodothyronine” syndrome has been described in association with sepsis, potentially accompanied by low levels of T4 [19]. Moreover, septic shock can induce hypophyseal hypoperfusion, causing central hypothyroidism [20]. It has also been shown that decreased baseline thyroid function is associated with a poor prognosis in patients with sepsis or septic shock, independent of other prognostic factors [21]. Sepsis-induced thyroid dysfunction appears to be a transient condition and may represent an adaptive mechanism to protect the thyroid from cellular death caused by systemic inflammatory response syndrome. Furthermore, there are several pro-inflammatory cytokines (IL1β, IL6, and TNF-α) that have inhibitory effects on thyroid function [22]. These mechanisms partially explain the results of our study, particularly the decrease in TSH, fT3, and fT4 levels shortly after COVID-19 infection.
Recent articles have investigated the varieties of thyroid disease associated with COVID-19 infection, regardless of a pre-existing thyroid dysfunction or HCV infection. In a case series, it has been suggested that COVID-19 can induce subacute thyroiditis, developing as late as 36 days after typical COVID-19 symptoms appear [23]. The 4 patients reported presented with thyrotoxicosis and received low-dose steroid therapy. The incidence of thyroiditis in intensive care unit patients has also been evaluated in a prospective study comparing COVID-19 infected to non-infected patients [24]. This study also presents a high incidence of thyrotoxicosis in the COVID-19 subgroup without underlying thyroid disease. Another large single-center retrospective study found a high incidence of thyroid dysfunction in 287 patients admitted to non-intensive care units for COVID-19 infection: thyrotoxicosis in 20.2% and hypothyroidism in 5.2% [25]. On the other hand, a retrospective study on 50 patients with COVID-19 infection without previous thyroid disease found low levels of TSH and total triiodothyronine, which normalized after the infection resolution [26]. Furthermore, a large multicenter trial including 621 patients reported lower values of TSH and fT4 in COVID-19 infected patients than in non-infected patients. Interestingly, none of the patients in this study presented overt thyrotoxicosis [27]. A recent systematic review found a prevalence of thyroid dysfunction ranging from 13% to 64% in a total of 1237 patients [28].
More relevant studies have evaluated the impact of severe COVID-19 infection on thyroid function. In a retrospective study of 100 COVID-19 patients, low levels of fT3, TSH, and fT3/fT4 were correlated with severity of disease and with patient prognosis [29]. Another retrospective study of 46 COVID-19 patients without a history of thyroid disease also found that these patients had lower values of fT3 and TSH compared to a control group. Values were significantly lower in patients requiring ICU admission [30]. These studies support our findings that SARS-CoV-2 has a major impact on thyroid function. However, these studies excluded patients with a previous thyroid disease, while our study group consists of patients with a known history of thyroid dysfunction as well as systemic inflammation induced by HCV infection. Therefore, the results cannot be generalized.
Autopsy studies in patients who died from COVID-19 revealed that the presence of the virus in the thyroid tissue was associated with a significant inflammatory infiltration and upregulation of immune genes involved in the interferon pathways, while tissues negative for SARS-CoV-2 did not present these inflammatory changes [31]. In our study, we evaluated patients with previously diagnosed thyroid conditions (associated with the chronic HCV infection); this may explain the increased thyroid inflammation we found in our patients, expressed by the high levels of autoantibodies,
Important similarities between HCV and SARS-CoV-2 have been studied. Both viruses, which were originally thought to have specific target organs, have proven to be systemic infections. The resemblance between the 2 viruses originates at a genomic lever, both being single-strained RNA viruses. Furthermore, exacerbation of the immune response, particularly from T helper2 lymphocytes has been associated with HCV infection and the previous SARS infections, inducing immune-mediated tissue damage, which is also suspected in COVID-19 [32]. On a molecular level, both HCV and SARS-CoV-2 use ion channels (viroporins) as entry pathways into the cells; these viruses have structurally similar proteins, p7 and E, that bond to viroporins [33]. One of the most important ion channel impairments in both these infections is the damage to chloride channels, which are essential for physiologic processes such as neuronal excitation, muscle contraction, and transepithelial fluid transportation [34]. In the ongoing SARS-CoV-2 pandemic, hypochloremia has been associated with the diagnosis of COVID-19 and with increased illness severity [35].
Inconclusive data also emerge when studying the impact of COVID-19 on patients with a history of thyroid illness. A report of 2 cases presented the recurrence of Graves’ disease during infection, both after more than 2 years of stable thyroid function without medication [36]. An article regarding the relationship between thyroid cancer and SARS-CoV-2 showed an increased prevalence of the infection in this subgroup of patients [37]. The presence of thyroid cancer appears not to be an additional risk factor for mortality in COVID-19 [38].
To the best of our knowledge, none of the articles published analyzed the impact of COVID-19 on patients with HCV- induced thyroid disease, although this is a frequently-encountered condition. A systematic review published in 2016 showed an increased prevalence of high anti-Tg and ATPO antibodies and a 3-fold risk of hypothyroidism in patients with chronic HCV infection [39]. Modern interferon-free therapies in HCV infection, which are associated with high cure rates and few adverse reactions, are expected to reduce the rate and severity of HCV extrahepatic manifestations [40–43]. It appears that HCV-associated thyroid disorders can persist after all oral antiviral therapy but are less frequent and severe than after the classical interferon-based regimens [44,45]. Our findings regarding COVID-19 consequences among HCV-infected patients with preexistent thyroid dysfunction are comparable with those described by other recent studies (that included patients without known HCV-induced thyroidal dysfunction) [46].
From a cost-effectiveness point of view, we consider that thyroid hormone screening should be done at least in patients with risk factors for thyroid disease (other autoimmune conditions, patients coming from regions with endemic goiter, HCV-infected patients, or patients with abnormal clinical thyroid examination results).
Our study reveals that COVID-19 infection has a significant impact on the values of thyroid hormones and antibodies and may lead to medication adjustments in patients with known thyroid illness. However, the study has several limitations: absence of patients with severe COVID-19 infection, insufficient data regarding corticoid therapy received during infection, and a small number of patients, not permitting sufficient correlations with the stage of liver disease or in patients receiving antithyroid drugs or substitution therapy. Another important factor that should be taken into account is the time passed since HCV cure was obtained in these patients, as HCV chronic infection is associated with persistent systemic inflammation, which could have altered the results. Also, evaluations during the acute COVID-19 infection were performed in other medical facilities and could not be obtained for a better description of the changes in time. Prospective studies monitoring these patients should estimate the impact of SARS-CoV-2 infection on the metabolism and dose adjustment of these drugs.
Conclusions
This study supports previous reports of an association between SARS-CoV-2 infection and thyroid dysfunction associated with thyroid autoantibodies. Thyroid function tests may be considered as part of the laboratory work-up in patients with COVID-19 infection.
References
1. Ramos-Casals M, Brito-Zerón P, Mariette X, Systemic and organ-specific immune-related manifestations of COVID-19: Nat Rev Rheumatol, 2021; 17; 315-32
2. Temgoua MN, Endomba FT, Nkeck JR, Coronavirus disease 2019 (COVID-19) as a multi-systemic disease and its impact in low- and middle-income countries (LMICs): SN Compr Clin Med, 2020 [Online ahead of print]
3. Scappaticcio L, Pitoia F, Esposito K, Impact of COVID-19 on the thyroid gland: An update: Rev Endocr Metab Disord, 2020 [Online ahead of print]
4. Poma AM, Bonuccelli D, Giannini R, COVID-19 autopsy cases: Detection of virus in endocrine tissues: J Endocrinol Invest, 2021 [Online ahead of print]
5. Duntas LH, Jonklaas J, COVID-19 and thyroid diseases: A bidirectional impact: J Endocr Soc, 2021; 5(8) bvab076
6. Inaba H, Aizawa T, Coronavirus disease 2019 and the thyroid – progress and perspectives: Front Endocrinol (Lausanne), 2021; 12; 708333
7. Lui DTW, Lee CH, Chow WS, Insights from a prospective follow-up of thyroid function and autoimmunity among COVID-19 survivors: Endocrinol Metab (Seoul), 2021; 36(3); 582-89
8. Garber JR, Cobin RH, Gharib H, American Association of Clinical Endocrinologists and American Thyroid Association Taskforce on Hypothyroidism in Adults. Clinical practice guidelines for hypothyroidism in adults: Cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association: Thyroid, 2012; 22(12); 1200-35
9. Cacoub P, Comarmond C, Domont F, Extrahepatic manifestations of chronic hepatitis C virus infection: Ther Adv Infect Dis, 2016; 3(1); 3-14
10. Obrişcă B, Jurubi ă R, Sorohan B, Clinical outcome of HCV-associated cryoglobulinemic glomerulonephritis following treatment with direct acting antiviral agents: A case-based review: Clin Rheumatol, 2019; 38(12); 3677-87
11. Iliescu L, Herlea V, Toma L, Orban C, Association between chronic HCV hepatitis, membranoproliferative glomerulopathy and cutaneous sarcoidosis: J Gastrointestin Liver Dis, 2015; 24(1); 8
12. Iliescu L, Mercan-Stanciu A, Ioanitescu ES, Toma L, Hepatitis C-associated B-cell non-hodgkin lymphoma: A pictorial review: Ultrasound Q, 2018; 34(3); 156-66
13. Iliescu L, Mercan-Stanciu A, Toma L, Ioanitescu ES, A severe case of hyperglycemia in a kidney transplant recipient undergoing interferon-free therapy for chronic hepatitis C: Acta Endocrinol (Buchar), 2018; 14(4); 533-38
14. Ferri C, Colaci M, Fallahi P, Thyroid involvement in hepatitis C virus-infected patients with/without mixed cryoglobulinemia: Front Endocrinol (Lausanne), 2017; 8; 159
15. Pastore F, Martocchia A, Stefanelli M, Hepatitis C virus infection and thyroid autoimmune disorders: A model of interactions between the host and the environment: World J Hepatol, 2016; 8(2); 83-91
16. ASLD/IDSA HCV Guidance Panel, Hepatitis C guidance: AASLD-IDSA recommendations for testing, managing, and treating adults infected with hepatitis C virus: Hepatology, 2015; 62(3); 932-54
17. Suceveanu A-I, Mazilu L, Katsiki N, NLRP3 inflammasome biomarker – could be the new tool for cu cardiometabolic syndrome outcome: Metabolites, 2020; 10(11); 448
18. : WHO Global Database on Iodine Deficiency Last Updated: 2007-07-24, available at https://www.who.int/vmnis/iodine/data/database/countries/rou_idd.pdf
19. Luo B, Yu Z, Li Y, Thyroid hormone disorders and sepsis: Biomed Mater Eng, 2017; 28(s1); S237-41
20. Benea SN, Lazar M, Hristea A, Central hypothyroidism in severe sepsis: Acta Endocrinol (Buchar), 2019; 15(3); 372-77
21. Angelousi AG, Karageorgopoulos DE, Kapaskelis AM, Falagas ME, Association between thyroid function tests at baseline and the outcome of patients with sepsis or septic shock: A systematic review: Eur J Endocrinol, 2011; 164(2); 147-55
22. McIver B, Gorman CA, Euthyroid sick syndrome: An overview: Thyroid, 1997; 7(1); 125-32
23. Brancatella A, Ricci D, Cappellani D, Is subacute thyroiditis an underestimated manifestation of SARS-CoV-2 infection? Insights from a case series: J Clin Endocrinol Metab, 2020; 105(10) dgaa537
24. Muller I, Cannavaro D, Dazzi D, SARS-CoV-2-related atypical thyroiditis: Lancet Diabetes Endocrinol, 2020; 8(9); 739-41
25. Lania A, Sandri MT, Cellini M, Thyrotoxicosis in patients with COVID-19: The THYRCOV study: Eur J Endocrinol, 2020; 183(4); 381-87
26. Dascalu AM, Tudosie MS, Smarandache GC, Serban D, Impact of the COVID-19 pandemic upon the ophthalmological clinical practice: Rom J Leg Med, 2020; 28; 96-100
27. Khoo B, Tan T, Clarke SA, Thyroid function before, during, and after COVID-19: J Clin Endocrinol Metab, 2021; 106(2); e803-11
28. Giovanella L, Ruggeri RM, Ovčariček PP, Prevalence of thyroid dysfunction in patients with COVID-19: A systematic review: Clin Transl Imaging, 2021 [Online ahead of print]
29. Gao W, Guo W, Guo Y, Thyroid hormone concentrations in severely or critically ill patients with COVID-19: J Endocrinol Invest, 2021; 44(5); 1031-40
30. Baldelli R, Nicastri E, Petrosillo N, Thyroid dysfunction in COVID-19 patients: J Endocrinol Invest, 2021; 44(12); 2735-39
31. Poma AM, Basolo A, Bonuccelli D, Activation of type I and type II interferon signaling in SARS-CoV-2-positive thyroid tissue of patients dying from COVID-19: Thyroid, 2021; 31(12); 1766-75
32. Tay MZ, Poh CM, Rénia L, The trinity of COVID-19: Immunity, inflammation and intervention: Nat Rev Immunol, 2020; 20(6); 363-74
33. Surya W, Li Y, Torres J, Pentameric viral ion channels: From structure to function: J Receptor Ligand Channel Res, 2015; 8; 9-18
34. Liu B, Billington CK, Henry AP, Chloride intracellular channel 1 (CLIC1) contributes to modulation of cyclic AMP-activated whole-cell chloride currents in human bronchial epithelial cells: Physiol Rep, 2018; 6(2); e13508
35. Alothaid H, Aldughaim MSK, El Bakkouri K, Similarities between the effect of SARS-CoV-2 and HCV on the cellular level, and the possible role of ion channels in COVID19 progression: A review of potential targets for diagnosis and treatment: Channels (Austin), 2020; 14(1); 403-12
36. Jiménez-Blanco S, Pla-Peris B, Marazuela M, COVID-19: A cause of recurrent Graves’ hyperthyroidism?: J Endocrinol Invest, 2021; 44(2); 387-88
37. Prete A, Falcone M, Bottici V, Thyroid cancer and COVID-19: Experience at one single thyroid disease referral center: Endocrine, 2021; 27; 332-39
38. Kathuria-Prakash N, Mosaferi T, Xie M, COVID-19 outcomes of patients with differentiated thyroid cancer: A multicenter Los Angeles cohort study: Endocr Pract, 2021; 27(2); 90-94
39. Shen Y, Wang XL, Xie JP, Thyroid disturbance in patients with chronic hepatitis C infection: A systematic review and meta-analysis: J Gastrointestin Liver Dis, 2016; 25(2); 227-34
40. Iliescu EL, Mercan-Stanciu A, Toma L, Safety and efficacy of direct-acting antivirals for chronic hepatitis C in patients with chronic kidney disease: BMC Nephrol, 2020; 21(1); 21
41. Iliescu L, Stanciu MA, Toma L, All oral antiviral treatment with paritaprevir/ombitasvir/ritonavir and dasabuvir in chronic HCV infection real life experience, 2018; 138-43
42. Zignego AL, Ramos-Casals M, Ferri C, ISG-EHCV. International therapeutic guidelines for patients with HCV-related extrahepatic disorders. A multidisciplinary expert statement: Autoimmun Rev, 2017; 16(5); 523-41
43. Wahid B, Waqar M, Rasool N, Prevalence of thyroid stimulating hormone dysfunction among sofosbuvir-treated HCV-infected patients: A real-world clinical experience: J Med Virol, 2019; 91(3); 514-17
44. Zgura A, Gales L, Bratila E, Variation of the T lymphocytes according to treatment in breast cancer: Rev Chim, 2019; 70(5); 1649-54
45. Serban D, Socea B, Balasescu SA, Safety of laparoscopic cholecystectomy for acute cholecystitis in the elderly: A multivariate analysis of risk factors for intra and postoperative complications: Medicina, 2021; 57(3); 230
46. Şerban D, Brănescu CM, Smarandache GC, Safe surgery in day care centers: Focus on preventing medical legal issues: Rom J Leg Med, 2021; 29(1); 60-64
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387