22 February 2022: Clinical Research
Comparison of the Color Doppler Vascularity Index in Patients with Pancreatic Adenocarcinoma and Chronic Pancreatitis from a Single Center in Poland
Przemysław Dyrla1AE*, Arkadiusz LubasDOI: 10.12659/MSM.935249
Med Sci Monit 2022; 28:e935249
Abstract
BACKGROUND: Pancreatic cancer is one of the most common cancers in the world and a major cause of cancer mortality. Therefore, it is extremely important to distinguish between malignant and benign changes quickly and accurately. This single-center study aimed to assess the discriminatory properties of the color Doppler vascularity index (CDVI) in the diagnosis of focal chronic pancreatitis and malignant pancreatic tumors.
MATERIAL AND METHODS: Seventy-nine patients (42 men, 37 women; age 62.0±13.5 years; 46 adenocarcinomas; 33 pancreatitis) qualified for this study. During endosonographic examination, pancreatic tumors were assessed in the color Doppler option. The dynamic tissue perfusion measurement was used to calculate tissue flow velocity (TFV), tissue perfusion intensity (TPI), and vascularization as the CDVI.
RESULTS: TFV, TPI, and CDVI were significantly lower in the group with malignant tumors than in the group with pancreatitis (P<0.001). In the receiver operating characteristic analysis, results of TFV=2.181 cm/s, TPI=0.009 cm/s, and CDVI=0.268 allowed for significant prediction of malignant tumors (P<0.001), with sensitivity of 75.8%, 69.7%, and 72.7% and specificity of 91.3%, 93.5%, and 80.4%, respectively, without significant differences between perfusion parameters and CDVI (P=0.07).
CONCLUSIONS: The findings from this study showed that color Doppler imaging and the use of the CDVI could provide an adjunctive diagnostic approach to distinguish between pancreatic adenocarcinoma and focal chronic pancreatitis. Owing to the possibility of calculating vascularization by non-Doppler methods, the method may be an easier and more accessible diagnostic option for malignant pancreatic tumors than perfusion assessed in external software.
Keywords: Endosonography, Pancreatic Diseases, Pancreatic Neoplasms, Diagnosis, Differential, Female, Humans, Male, Morbidity, Poland, ROC Curve, Ultrasonography, Doppler, Color
Background
In European countries, pancreatic cancer is the fourth most fatal cancer in men and women [1]. Pancreatic cancer most often occurs after the age of 70 years and is usually diagnosed in the advanced stage, which prevents effective treatment. Surgical treatment is possible in only 15% to 20% of patients, and the 5-year survival rate reaches only about 20% in patients treated in this way [2]. The evaluation of tissue perfusion parameters has been used as a diagnostic method that allows for the differentiation of the nature of focal pancreatic lesions [3,4]. The parameters enabling an adequate distinction of neoplastic lesions between inflammatory and cystic pancreatic tumors were found to be the average flow velocity and the lowest and highest percentile of the perfusion intensity assessed by dynamic tissue perfusion measurement (DTPM). To date, it is not possible to perform DTPM during endosonographic examination (EUS). To the best of our knowledge, no company has implemented this method in the internal software of an ultrasound machine. This, in turn, leads to the need to purchase additional software and involves the time-consuming transfer of recorded movie sequences with the color Doppler test to an external computer. On the other hand, an increasing number of new ultrasound devices have the ability to assess the number of color pixels shown in Doppler (color Doppler, power Doppler, microvascular flow [Samsung], superb microvascular imaging [Toshiba], micro flow imaging [Philips]), and non-Doppler, such as B-Flow [General Electric] and Fine Flow [Hitachi]) options compared to the total number of pixels in the area being evaluated and to calculate the vascularity index. We have not found any studies comparing these 2 methods in the diagnosis of focal chronic pancreatitis and malignant pancreatic tumors. Therefore, this single-center study aimed to assess the discriminatory properties of the color Doppler vascularity index (CDVI) in the diagnosis of focal chronic pancreatitis and malignant pancreatic tumors.
Material and Methods
TISSUE PERFUSION AND VASCULARIZATION:
During EUS diagnostics, the vascularization of the lesion was assessed in the color Doppler option of the Hitachi HI VISION Preirus (Fujifilm Healthcare, Japan), and the results were recorded in film sequences, which were 3 to 5 s in length. The color Doppler examination was performed at constant and unchanged frequency settings (5 MHz) and signal gain. To better visualize the vessels, the flow velocity scale was determined individually. To perform DTPM, recorded film sequences with flow testing in the color Doppler option were evaluated in an external program (PixelFlux Medical Device, Chameleon-Software, Leipzig, Germany). The region of interest (ROI) covered the entire color Doppler-gated tumor area, bypassing peripheral vessels and large vessels piercing the lesion. Mean tissue flow velocity (TFV [cm/s]) and mean tissue perfusion intensity (TPI [cm/s]=(average velocity×vascular area)/ROI area) were used to distinguish between malignant tumors and focal chronic pancreatitis in DTPM [5]. To assess the vascularization of the examined lesions based on the number of colored pixels reflecting the flow field in the vessels, the color Doppler vascular index (CDVI [%]) was calculated by referencing the calculated flow area to the ROI surface.
STATISTICAL ANALYSIS:
For statistical analysis, Statistica version 12 software (StatSoft Inc, Cracow, Poland) was used. The results are presented as means with standard deviation and median with interquartile range. The differences between the groups were tested using the
Based on the data of the first 20 patients, the power analysis for the necessary sample size to achieve the significant difference in CDVI between focal chronic pancreatitis and malignant lesions was 39 patients, for a statistical power of 90%.
Results
Of the 79 lesions assessed, only 1 (adenocarcinoma) failed to visualize any flow (TFV, TPI, and CDVI=0). The average values of the flow parameters are presented in Table 1. All assessed parameters differed significantly between the examined groups, and there were significantly higher values of TFV, TPI, and CDVI in the group with focal chronic pancreatitis (Table 1). In the ROC analysis, TFV, TPI, and CDVI enabled the differentiation between focal chronic pancreatitis and malignant pancreatic lesions (Table 2, Figure 3). Although sensitivity and specificity for CDVI ≤0.25 in the recognition of lesions was about 75% (Figure 4), in comparative analysis, TPI was slightly better than CDVI in differentiating the lesions studied (
Discussion
In the present study, we examined the usefulness of the CDVI in differentiating between focal chronic pancreatitis and malignant pancreatic lesions. The discriminatory properties of CDVI were not significantly different from those parameters assessing tissue perfusion, such as flow velocity and intensity, which were reported earlier [3,4].
Detection of a solid mass in the pancreas causes several diagnostic problems, the most important being whether the mass is malignant or benign. This problem should be noted as one of the most difficult diagnostic tasks, which is the differentiation of focal chronic pancreatitis from pancreatic cancer. Imaging methods play the most important roles, ranging from detection to staging assessment to planning the method of treatment. The current standard is diagnostic methods using computed tomography (CT), magnetic resonance imaging, and EUS [6]. Considering that in most cases the treatment of pancreatic cancer is ineffective and acute inflammation is often a sign of pancreatic cancer, methods enabling the proper detection and differentiation of solid pancreatic changes at an early stage are necessary [7,8]. Newer algorithms using external software and visualization options are emerging. Lastly, the possibility of using additional software enabling an adequate differentiation of malignant tumors and focal chronic pancreatitis, based on Doppler perfusion parameters, for example, has been introduced [3]. In the present study, we compared the discriminative properties of 2 color Doppler methods, DTPM and CDVI, in the diagnosis of focal chronic pancreatitis and malignant pancreatic tumors. Numerous publications are available on the assessment of perfusion parameters in pancreatic lesions using contrast-enhanced ultrasound. In previous years, owing to the imperfections of Doppler techniques, enhancing the obtained image by the contrast method has significantly expanded diagnostic options. In the EUS diagnostic results of 93 solid pancreatic focal lesions, Dietrich et al could not visualize any flow in focal pancreatic lesions using the Doppler option, while the use of contrast allowed them to differentiate all lesions into hypovascular, isovascular, and hypervascular types [9]. In their study, reduced vascularization of the lesion was associated with malignancy with 92% sensitivity and 100% specificity. Kitano et al, using contrast-enhanced harmonic EUS, assessed the vascularization of solid pancreatic lesions previously diagnosed in conventional EUS [10]. Contrast-enhanced harmonic identifies ductal carcinomas with 95.1% sensitivity and 89.0% specificity. Subsequent studies using contrast-enhanced ultrasound methods have shown that reduced vascularization of the pancreatic lesion helps diagnose and differentiate malignant pancreatic tumors from focal chronic pancreatitis [11–13]. Using an image analysis application specifically written in Java, Săftoiu et al used the VI calculated as a percentage of the number of pixels in the contrasting power Doppler option divided by the number of all pixels in the ROI [14]. The combination of VI with endoscopic elastographic examination allowed for the differential diagnosis of focal pancreatic masses with 75.8% sensitivity and 92.5% specificity. Despite the promising results of contrast-enhanced Doppler EUS in the differentiation of solid pancreatic lesions, it should be noted that it is characterized by high variability of results and poor reproducibility, and the results of the method do not have absolute values, such as cm/s or mL/min [15]. Certain factors, such as the complications after intravenous administration of ultrasound contrast, the necessity of having efficient resuscitation equipment during the examination, and contraindications for this examination in the case of known allergies, pulmonary hypertension, and uncontrolled hypertension, are also not negligible and may limit the availability of the method [15]. Dynamic 64-slice helical CT with perfusion imaging can be used to identify malignant lesions with 80% sensitivity and 75% specificity [16].
Unlike contrast-enhanced ultrasound, the assessment of perfusion in solid pancreatic lesions using the color Doppler option during EUS does not require additional precautions, and the ongoing improving methods of Doppler imaging allow for an adequate assessment of vascularization of lesions [3,4]. The purpose of the present study was to compare the discriminatory properties of organ perfusion parameters, such as the velocity and flow intensity, assessed in the previously tested DTPM method to the CDVI in the diagnosis of focal chronic pancreatitis and malignant pancreatic tumors. The selected CDVI can be calculated using Doppler and non-Doppler flow imaging methods and even in contrast-enhanced EUS, which is undoubtedly an advantage of this study. No contrast agents were used during the EUS examination in the present study, in contrast to the studies mentioned earlier, but only the color Doppler option was used. In our study, a total of 79 lesions were analyzed, and only 1 failed to register any flow. In the malignant lesion group, significantly lower values of both perfusion parameters (TFV and TPI) and CDVI (Table 1) were obtained. Perfusion and vascularization parameters assessed in the present study significantly differentiated between the group of malignant lesions and focal chronic pancreatitis (Table 2, Figure 3). Sensitivity and specificity for CDVI in the recognition of a lesion were about 75% (Figure 4), and in comparative analysis, TPI was only slightly better than CDVI in differentiating the lesions studied.
There were several limitations to our study. First, it had a relatively small sample size. Next, we used a conventional color Doppler method to calculate perfusion parameters and the CDVI. Thus, our findings can be repeated with most ultrasound endoscopy devices. However, this can be considered as a significant limitation of this study since the use of a microvascular imaging option and a calculation of microvessel density as CDVI could significantly improve the discriminatory properties of this index. Moreover, in the present study, pancreatic lesions were diagnosed with EUS-FNA, without the histological assessment. Thus, we could not evaluate the grade of tumor or the chronicity of pancreatitis. Moreover, the fact that FNA cytology has a rather high false-negative rate could mean that some cases of pancreatic adenocarcinoma were missed. Thus, these results relating to the discriminatory properties of CDVI should be verified in a group of pancreatic solid lesions with histopathological diagnoses. On the other hand, the 6-month follow-up period was designed to confirm the benign nature of lesions.
Nevertheless, in the scope of the differentiation of pancreatic lesions, our results were satisfactory, and this method can be an alternative to CT or EUS using contrast media. At present, EUS is recognized as the best method for detecting solid tumors in the pancreas. However, the differentiation between focal chronic pancreatitis and malignant lesions is still very difficult to obtain. The results of our study provide evidence of the usefulness of assessing perfusion parameters and the CDVI in differentiating the lesions mentioned above. This method can be successfully used in centers with EUS. Despite promising research results, one cannot forget about the absolute necessity to confirm the diagnosis with histological examination before qualifying a patient for further treatment.
Conclusions
The findings from this study showed that color Doppler imaging and the use of the CDVI could provide an adjunctive diagnostic approach to distinguish between pancreatic adenocarcinoma and focal chronic pancreatitis. Owing to the possibility of calculating the vascularization by non-Doppler methods, it may be an easier and more accessible diagnostic option for malignant pancreatic tumors than perfusion assessed in external software.
Figures
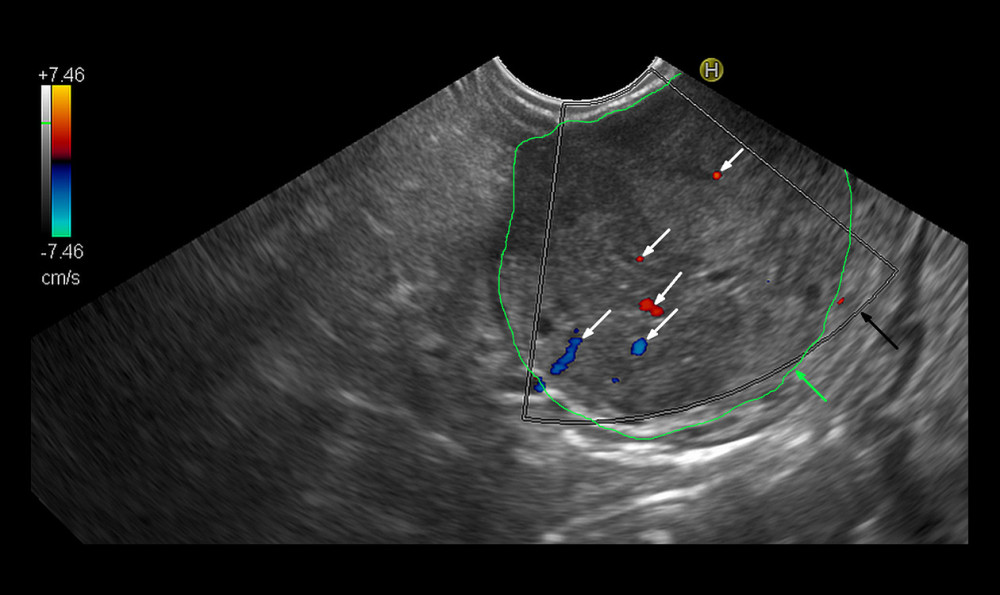 Figure 1. Endosonographic examination with color Doppler option of the malignant pancreatic lesion. Malignant tumor outlined with the green line (green arrow). The black arrow indicates the color Doppler frame. White arrows show sparse vessels detected in the color Doppler option. Calculated color Doppler vascularity index, 0.42%; tissue flow velocity, 1.63 cm/s; tissue perfusion intensity, 0.007 cm/s.
Figure 1. Endosonographic examination with color Doppler option of the malignant pancreatic lesion. Malignant tumor outlined with the green line (green arrow). The black arrow indicates the color Doppler frame. White arrows show sparse vessels detected in the color Doppler option. Calculated color Doppler vascularity index, 0.42%; tissue flow velocity, 1.63 cm/s; tissue perfusion intensity, 0.007 cm/s. 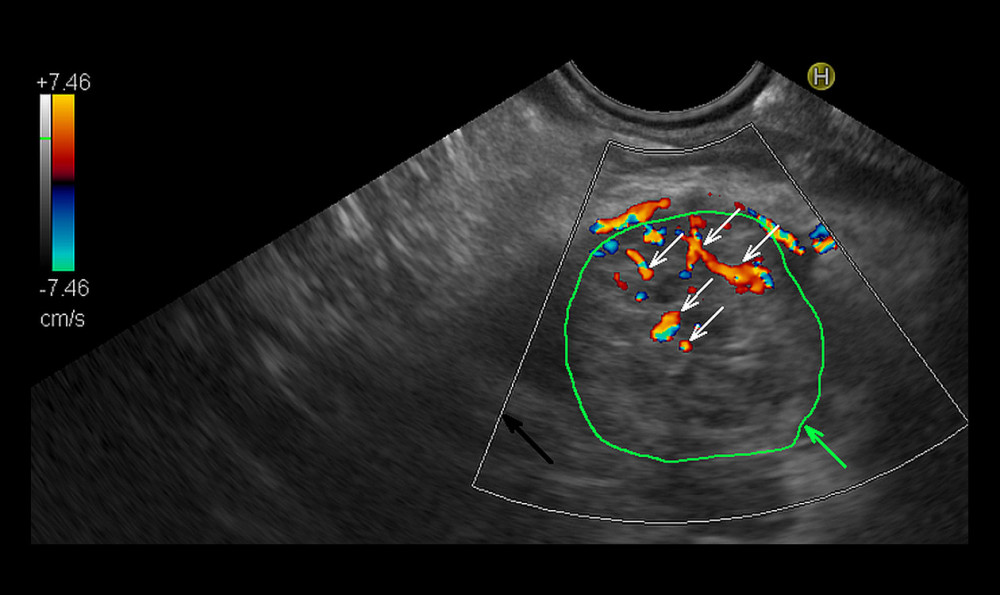 Figure 2. Endosonographic examination with color Doppler option of pancreatitis. Pancreatitis area outlined with the green line (green arrow). The black arrow indicates the color Doppler frame. White arrows show multiple vessels detected in the color Doppler option. Calculated color Doppler vascularity index, 5.97%; tissue flow velocity, 3.74 cm/s; tissue perfusion intensity, 0.220 cm/s.
Figure 2. Endosonographic examination with color Doppler option of pancreatitis. Pancreatitis area outlined with the green line (green arrow). The black arrow indicates the color Doppler frame. White arrows show multiple vessels detected in the color Doppler option. Calculated color Doppler vascularity index, 5.97%; tissue flow velocity, 3.74 cm/s; tissue perfusion intensity, 0.220 cm/s. 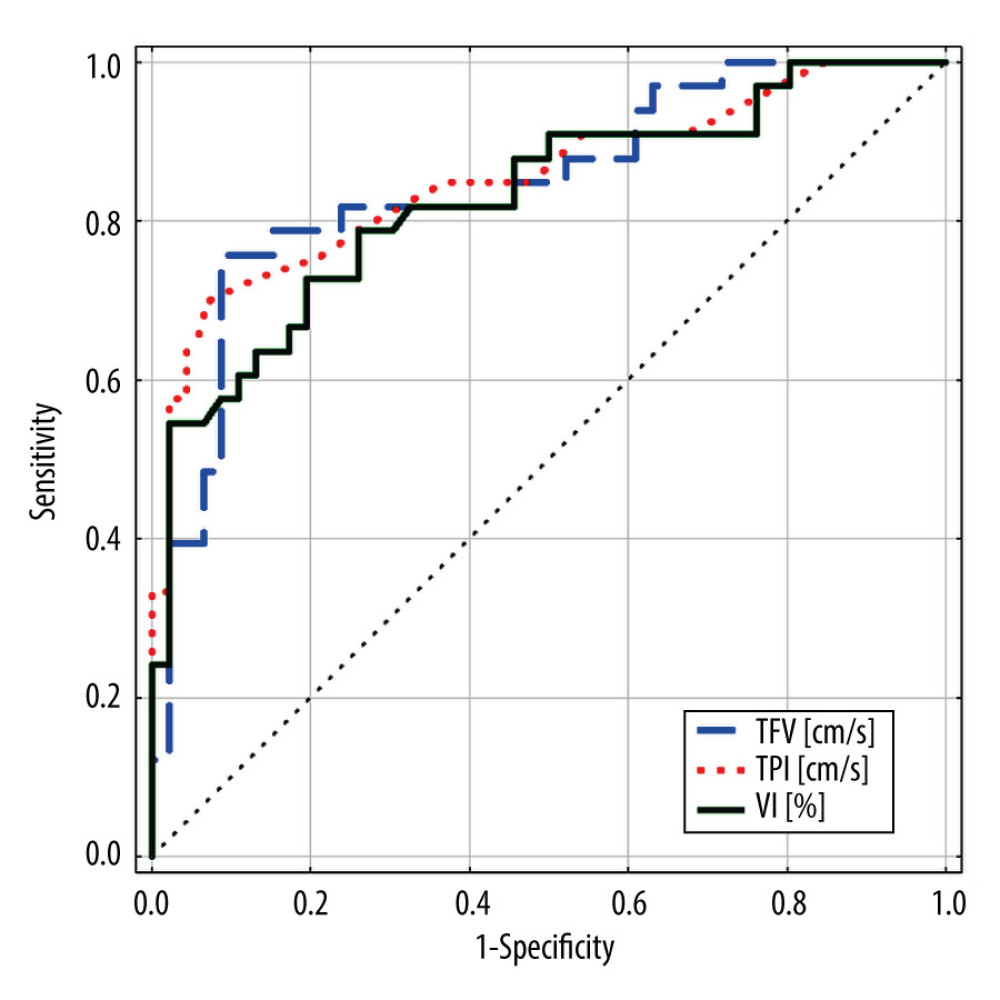 Figure 3. Receiver operating characteristic curve chart. Comparison of discriminatory properties of ultrasound assessment parameters in the diagnosis of pancreatic adenocarcinomas.
Figure 3. Receiver operating characteristic curve chart. Comparison of discriminatory properties of ultrasound assessment parameters in the diagnosis of pancreatic adenocarcinomas. 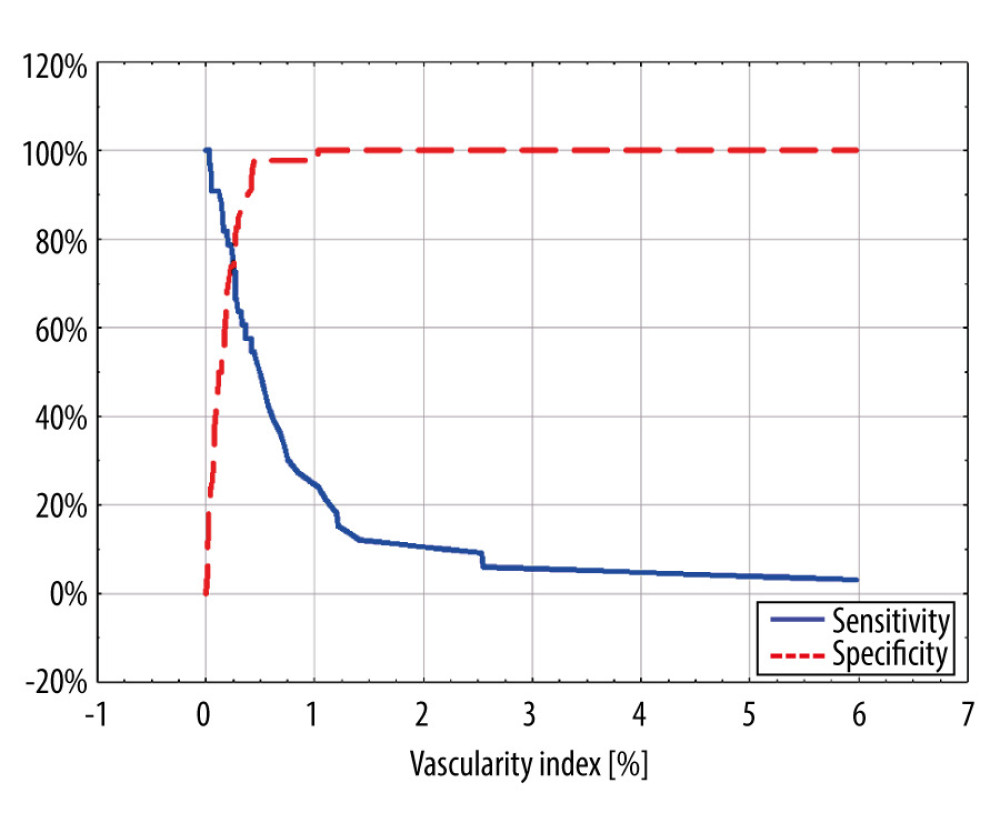 Figure 4. Sensitivity and specificity of the color Doppler vascularity index in the diagnosis of pancreatic adenocarcinomas.
Figure 4. Sensitivity and specificity of the color Doppler vascularity index in the diagnosis of pancreatic adenocarcinomas. References
1. Malvezzi M, Bertuccio P, Levi F, European cancer mortality predictions for the year 2014: Ann Oncol, 2014; 25; 1650-56
2. Ducreux M, Cuhna AS, Caramella CESMO Guidelines Committee, Cancer of the pancreas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up: Ann Oncol, 2015; 26(Suppl 5); v56-68
3. Dyrla P, Lubas A, Gil J, Niemczyk S, Doppler tissue perfusion parameters in recognizing pancreatic malignant tumors: J Gastroenterol Hepatol, 2016; 31(3); 691-95
4. Dyrla P, Gil J, Kosik K, Doppler tissue perfusion measurement is a sensitive and specific tool for a differentiation between malignant and inflammatory pancreatic tumors: PLoS One, 2019; 14(4); e0215944
5. Scholbach T, Scholbach J, Dynamic sonographic tissue perfusion measurement: J Med Ultrasound, 2009; 17(2); 71-85
6. Sharma C, Eltawil KM, Renfrew PD, Advances in diagnosis, treatment and palliation of pancreatic carcinoma: 1990–2010: World J Gastroenterol, 2011; 17; 867-97
7. Ryan DP, Hong TS, Bardeesy N, Pancreatic adenocarcinoma: N Engl J Med, 2014; 371; 1039-49
8. Munigala S, Kanwal F, Xian H, Increased risk of pancreatic adenocarcinoma after acute pancreatitis: Clin Gastroenterol Hepatol, 2014; 12; 1143-50
9. Dietrich CF, Ignee A, Braden B, Improved differentiation of pancreatic tumors using contrast-enhanced endoscopic ultrasound: Clin Gastroenterol Hepatol, 2008; 6(5); 590-97
10. Kitano M, Kudo M, Yamao K, Characterization of small solid tumors in the pancreas: The value of contrast-enhanced harmonic endoscopic ultrasonography: Am J Gastroenterol, 2012; 107(2); 303-10
11. Wang Y, Yan K, Fan Z, Contrast-enhanced ultrasonography of pancreatic carcinoma: Correlation with pathologic findings: Ultrasound Med Biol, 2016; 42(4); 891-98
12. Vitali F, Pfeifer L, Janson C, Quantitative perfusion analysis in pancreatic contrast enhanced ultrasound (DCE-US): A promising tool for the differentiation between autoimmune pancreatitis and pancreatic cancer: Z Gastroenterol, 2015; 53(10); 1175-81
13. Gheonea DI, Streba CT, Ciurea T, Săftoiu A, Quantitative low mechanical index contrast-enhanced endoscopic ultrasound for the differential diagnosis of chronic pseudotumoral pancreatitis and pancreatic cancer: BMC Gastroenterol, 2013; 13; 2
14. Săftoiu A, Iordache SA, Gheonea DI, Combined contrast-enhanced power Doppler and real-time sonoelastography performed during EUS, used in the differential diagnosis of focal pancreatic masses (with videos): Gastrointest Endosc, 2010; 72(4); 739-47
15. Emanuel AL, Meijer RI, van Poelgeest E, Contrast-enhanced ultrasound for quantification of tissue perfusion in humans: Microcirculation, 2020; 27(1); e12588
16. D’Onofrio M, Gallotti A, Mantovani W, Perfusion CT can predict tumoral grading of pancreatic adenocarcinoma: Eur J Radiol, 2013; 82(2); 227-33
Figures
 Figure 1. Endosonographic examination with color Doppler option of the malignant pancreatic lesion. Malignant tumor outlined with the green line (green arrow). The black arrow indicates the color Doppler frame. White arrows show sparse vessels detected in the color Doppler option. Calculated color Doppler vascularity index, 0.42%; tissue flow velocity, 1.63 cm/s; tissue perfusion intensity, 0.007 cm/s.
Figure 1. Endosonographic examination with color Doppler option of the malignant pancreatic lesion. Malignant tumor outlined with the green line (green arrow). The black arrow indicates the color Doppler frame. White arrows show sparse vessels detected in the color Doppler option. Calculated color Doppler vascularity index, 0.42%; tissue flow velocity, 1.63 cm/s; tissue perfusion intensity, 0.007 cm/s. Figure 2. Endosonographic examination with color Doppler option of pancreatitis. Pancreatitis area outlined with the green line (green arrow). The black arrow indicates the color Doppler frame. White arrows show multiple vessels detected in the color Doppler option. Calculated color Doppler vascularity index, 5.97%; tissue flow velocity, 3.74 cm/s; tissue perfusion intensity, 0.220 cm/s.
Figure 2. Endosonographic examination with color Doppler option of pancreatitis. Pancreatitis area outlined with the green line (green arrow). The black arrow indicates the color Doppler frame. White arrows show multiple vessels detected in the color Doppler option. Calculated color Doppler vascularity index, 5.97%; tissue flow velocity, 3.74 cm/s; tissue perfusion intensity, 0.220 cm/s. Figure 3. Receiver operating characteristic curve chart. Comparison of discriminatory properties of ultrasound assessment parameters in the diagnosis of pancreatic adenocarcinomas.
Figure 3. Receiver operating characteristic curve chart. Comparison of discriminatory properties of ultrasound assessment parameters in the diagnosis of pancreatic adenocarcinomas. Figure 4. Sensitivity and specificity of the color Doppler vascularity index in the diagnosis of pancreatic adenocarcinomas.
Figure 4. Sensitivity and specificity of the color Doppler vascularity index in the diagnosis of pancreatic adenocarcinomas. Tables
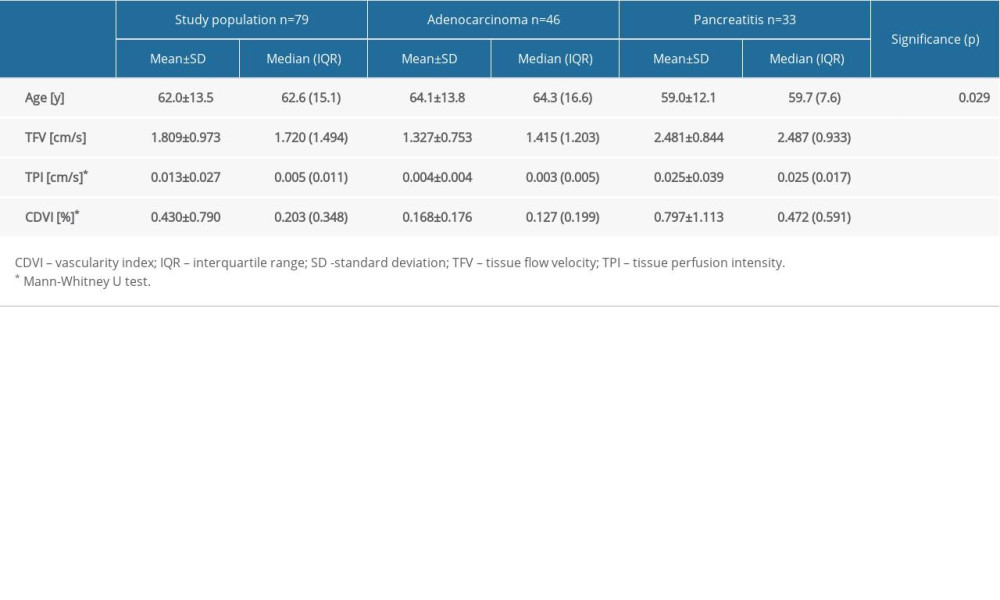 Table 1. Comparison of the results of perfusion and vascularization assessment of focal chronic pancreatitis and malignant pancreatic tumors.
Table 1. Comparison of the results of perfusion and vascularization assessment of focal chronic pancreatitis and malignant pancreatic tumors.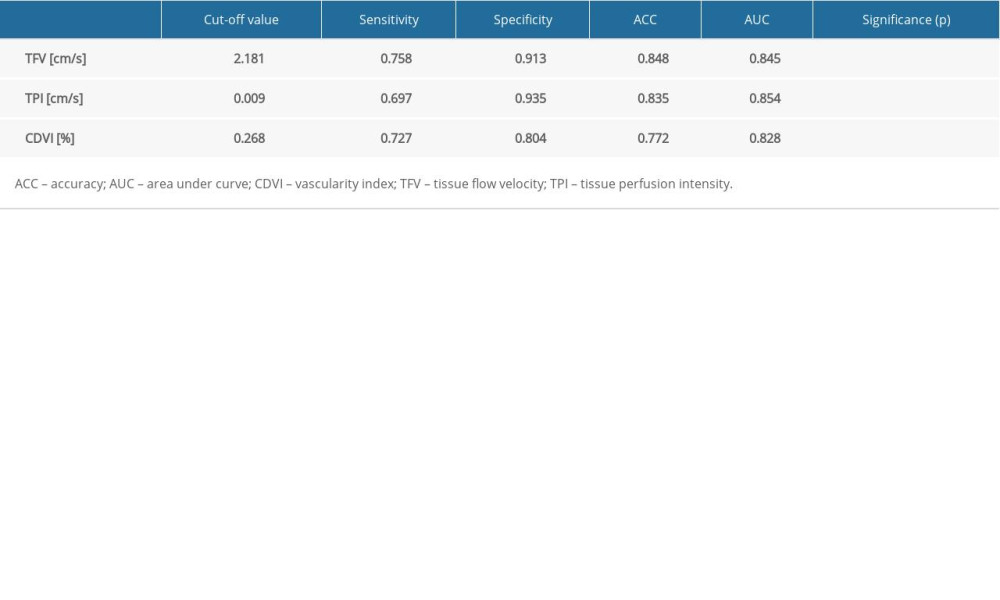 Table 2. Discriminatory properties of ultrasound parameters in the differentiation of pancreatic tumors.
Table 2. Discriminatory properties of ultrasound parameters in the differentiation of pancreatic tumors. Table 1. Comparison of the results of perfusion and vascularization assessment of focal chronic pancreatitis and malignant pancreatic tumors.
Table 1. Comparison of the results of perfusion and vascularization assessment of focal chronic pancreatitis and malignant pancreatic tumors. Table 2. Discriminatory properties of ultrasound parameters in the differentiation of pancreatic tumors.
Table 2. Discriminatory properties of ultrasound parameters in the differentiation of pancreatic tumors. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








