08 April 2022: Clinical Research
A Retrospective Study to Determine the Association Between Serum Albumin Levels and Atrial Fibrillation by Sex in 950 Patients from a Single Center in China
Xia Zhong1ACDE, Huachen Jiao2ADEG*, Dongsheng Zhao1BCF, Jing Teng1ADF, Mengqi Yang1CFDOI: 10.12659/MSM.935347
Med Sci Monit 2022; 28:e935347
Abstract
BACKGROUND: It remains unclear whether there is a sex difference in the association between serum albumin (ALB) and atrial fibrillation (AF). This retrospective case-control study from a single center in China aimed to determine the association between serum ALB levels and AF by sex in 950 patients.
MATERIAL AND METHODS: Data of 950 AF patients and 963 age- and sex-matched non-AF patients with sinus rhythm were collected and analyzed retrospectively. Clinical baseline data were analyzed using the t test or Mann-Whitney U test, analysis of variance (ANOVA), and chi-square test. The interrelationships were determined by Pearson correlation analysis, and multivariate logistic regression analysis was performed to adjust for covariables.
RESULTS: ALB levels of AF patients were significantly lower in both sexes (P<0.05), especially paroxysmal AF. ALB was positively correlated with total cholesterol (TC) (r=0.359, P<0.05), low-density lipoprotein cholesterol (LDL-C) (r=0.283, P<0.05), and serum apolipoprotein A1 (APOA1) (r=0.429, P<0.05) and was negatively correlated with serum creatinine (SCr) (r=0.129, P<0.05) in patients with AF. We found an independent negative association between ALB levels and AF in men after adjusting for confounding factors (OR=0.889, 95% CI: 0.845-0.934, P<0.05).
CONCLUSIONS: In patients at a single center in China, low serum ALB levels in male patients were significantly associated with AF. These findings support those from previous studies in other populations and highlight the importance of monitoring and treating the cause of hypoalbuminemia in cardiac patients.
Keywords: Atrial Fibrillation, Risk Factors, Serum Albumin, Case-Control Studies, China, Cholesterol, LDL, Female, Humans, Male
Background
Atrial fibrillation (AF), a worldwide epidemic and increasingly common arrhythmia, affects 33 million people worldwide [1,2]. In the United States and Europe, AF will affect 1 in 4 middle-aged adults during their lifetimes [3,4]. As a growing public health problem with anincreasing prevalence and significant substantial financial cost, AF is related to an increased risk of heart failure, stroke, cognitive impairment, and even mortality [5,6]. Unfortunately, there have been no major advances in equipment and novel anti-arrhythmic drugs to combatAF in the past decade [7]. Although catheter ablation is an effective therapy for AF, the invasive procedure can still have potential complications [8–10]. Furthermore, the success rate of a single operation is only 66.6% in paroxysmal AF [11]. Meanwhile, despite multifaceted efforts, prevention of AF remains challenging [12] and it is essential to investigate the correlation and mechanisms between potential modifiable risk factors and AF.
Although risk factors and mechanisms of AF are complex and far from clear, it has been confirmed that inflammation and oxidative stress play an important role in the occurrence and maintenance of AF [13,14]. Many scholars have demonstrated a relationship between low serum albumin (ALB) levels as an independent risk factor and cardiovascular diseases [15,16]. More recent studies additionally showed that ALB levels were associated with the risk of AF [17–21]. Clinical evidence suggests that ALB has several physiological properties, including antioxidant, anti-inflammatory, and anticoagulant, as well as colloid osmosis [22]. Furthermore, several studies demonstrated that ALB is a valuable biomarker of many diseases, mainly as a result of malnutrition and inflammation [23–25]. Nevertheless, from the clinical standpoint, the association between AF and ALB levels also can be influenced by multiple parameters, complications, and particularly by sex [26,27].
The relationship between ALB and AF among men and women has been less clearly elucidated, especially in the Chinese population. Therefore, this retrospective case-control study from a single center in China aimed to determine the association between serum ALB levels and AF by sex in 950 patients.
Material and Methods
DATA SOURCE AND STUDY DESIGN:
This investigation was conducted in accordance with the principles of the Helsinki Declaration and was approved by the Medical Research Ethics Committee of the Affiliated Hospital of Shandong University of Traditional Chinese Medicine before data collection, and the requirement of informed consent was waived. This case-control study retrospectively evaluated the clinical information of 1913 hospitalized patients (male/female: 949/964, mean age 68.26±11.02 years) in the Affiliated Hospital of Shandong University of Traditional Chinese Medicine from January 2019 to September 2021. All participants were from a short-term hospitalization population in the community who had lived with normal nutritional status. A total of 950 AF patients aged 28 to 85 years old and 963 age- and sex-matched non-AF patients with sinus rhythm served as controls. Additionally, considering that several confounding factors may interfere with the results, we excluded patients with cardiac surgery, structural heart disease, valvular disease, heart failure, hyperthyroidism, current liver or kidney dysfunction, malignancy, gout, use of uric acid-lowering drugs, and diuretics, as well as pregnant women. Baseline information of participants including age, sex, laboratory test results, AF type, and complications was documented from electronic medical record review.
DEFINITION OF AF:
According to guidelines [28], paroxysmal AF was considered to be AF terminated spontaneously or with intervention within 7 days of initiation. Permanent AF was defined as AF for which sinus rhythm could not be restored or maintained.
ALB MEASUREMENT AND DEFINITION OF HYPOPROTEINEMIA:
The bromocresol green (BCG) was used to measure ALB levels. The conversion standard of ALB levels is as follows: 1 g/L=0.1 g/dL. Hypoproteinemia was identified as ALB levels <3.5 g/dL [29].
SCREENED INDICATORS:
We screened baseline data of all participants, including age, sex, AF type, AF complication, and laboratory indicators, including ALB, triglyceride (TG), total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), alanine aminotransferase (ALT), serum creatinine, aspartate aminotransferase (AST), serum apolipoprotein A1(APOA1), serum apolipoprotein B (APOB), lipoprotein (a) (Lp (a)), serum creatinine (SCr), and serum uric acid (SUA).
STATISTICAL ANALYSIS:
All statistical analyses were performed using SPSS software (version 26.0; SPSS Inc., Chicago, IL, USA) and GraphPad Prism software (version 9.0.0). Specifically, continuous data are presented as mean±standard deviations (SD) or medians and interquartile ranges (IQR) [25th–75th percentile] and compared by
Results
BASELINE CHARACTERISTICS:
Table 1 shows the baseline characteristics of the AF group and controls. We enrolled 1913 patients and divided them into 2 groups: an AF group (n=950) and a control group (n=963). Overall, patients with AF were more likely to experience coronary heart disease (CHD), hypertension, diabetes, and hypoproteinemia (P<0.05), significantly higher levels of AST, SCr, and SUA (P<0.05), and significantly lower levels of TG, TC, LDL-C, HDL-C, APOA1, and APOB (P<0.05). Figure 1 shows the comparison of ALB levels between the AF group and controls by sex. Specifically, ALB levels of AF patients were significantly lower in both sexes (P<0.05). Furthermore, ALB levels of paroxysmal AF were significantly lower than permanent AF (P<0.05). In addition, Figure 2 shows the comparison of ALB levels between the AF group and controls by age. Compared with controls, ALB levels of AF patients with age ≤60 years were significantly lower (P<0.05) and ALB levels of AF patients age >60 years also were significantly lower (P<0.05).
THE RELATED FACTORS OF ALB LEVELS IN PATIENTS WITH AF:
Figure 3 shows the correlation between ALB levels and AF-related factors. Our results suggested that ALB was positively correlated with TC (r=0.359, P<0.05, Figure 3A), LDL-C (r=0.283, P<0.05, Figure 3B), and APOA1(r=0.429, P<0.05, Figure 3C) in patients with AF. Conversely, ALB was negatively correlated with SCr in AF patients (r=0.129, P<0.05, Figure 3D).
ASSOCIATION BETWEEN ALB AND AF:
Table 2 shows the association between ALB and AF by multivariate logistic regression analysis. Specifically, after adjusting for CHD, hypertension, and diabetes, ALB was considered to be a factor associated with AF (OR=0.931, 95% CI: 0.906–0.957, P<0.05). After adjusting for TG, TC, LDL-C, HDL-C, ALT, AST, APOA1, APOB, SCr, and SUA, ALB remained as a significant factor related to AF (OR=0.910, 95% CI: 0.884–0.936, P<0.05). After further adjustment for all confounding factors, ALB remained as an important relevant factor for AF (OR=0.930, 95% CI: 0.898–0.963, P<0.05). Moreover, this independent association was more significant in men with AF, regardless of age (P<0.05).
ASSOCIATION BETWEEN ALB LEVELS AND AF BY TYPE AND COMPLICATION:
As shown in Table 3, compared with the permanent AF group, the ALB levels of the paroxysmal AF group were significantly lower (37.49±4.96 vs 38.35±4.43 g/L, P<0.05). However, no significant differences in ALB levels between AF complications were observed (P>0.05).
ASSOCIATION BETWEEN ALB LEVELS AND METABOLIC INDICATORS IN PATIENTS WITH PAROXYSMAL AF:
As shown in Table 4, patients with paroxysmal AF of lower ALB had lower APOA1, TG, TC, LDL-C, and HDL-C in both sexes (P<0.05), lower SUA in men (P<0.05), and lower ALT and APOB in women (P<0.05).
ASSOCIATION BETWEEN ALB LEVELS AND METABOLIC INDICATORS IN PATIENTS WITH PERMANENT AF:
As shown in Table 5, patients with permanent AF and lower ALB had lower APOA1, TG, TC, LDL-C, and HDL-C in both sexes (P<0.05), and lower APOB in women (P<0.05).
Discussion
The present results show that in patients at a single center in China, low serum ALB levels in male patients were significantly associated with AF, regardless of age. Also, ALB was positively correlated with TC, LDL-C, and APOA1, and was negatively correlated with SCr. ALB levels of patients with paroxysmal AF were significantly lower than in patients with permanent AF, but no significant differences were observed in SUA levels among patients with different AF complications. We also found a consistent relationship between paroxysmal AF and permanent AF with lower ALB had lower blood lipid profiles and APOA1 in both sexes. These findings support those from previous studies in other populations and highlight the importance of monitoring and treating the cause of hypoalbuminemia in cardiac patients.
ALB is a primary protein with a variety of biochemical properties in human plasma [30]. Hypoalbuminemia, generally defined as ALB <35 g/L, is considered as a significant factor of inflammation, malnutrition and cachexia, as well as a useful marker of multiple diseases [31,32]. We found a close relationship between serum ALB levels and AF. The current finding are supported by those of previous studies. A prospective epidemiological and Mendelian randomization study based on a large-scale showed that ALB level is inversely related to the risk of AF in an approximately linear pattern [17]. A dose-response meta-analysis found a significant negative linear association between serum ALB and the risk of AF [18]. Another study showed that lower ALB was a risk factor for paroxysmal AF [19]. A prospective cohort study indicated that low ALB was associated with the risk and the number of attacks of NOAF for ICU patients [20]. In addition, several studies have shown that the relationship between albumin and AF may differ by sex. A study of 8870 individuals in the Copenhagen City Heart Study reported that lower ALB levels were independently associated with the occurrence of AF in women [21]. Nevertheless, in the current study, we found an independent negative correlation between ALB levels and AF in men, which was inconsistent with previous findings. Therefore, further studies are needed to confirm this association and elucidate the sex-specific mechanisms involved.
Several potential mechanisms have been identified. Inflammation has been proposed as a significant mechanism contributing to AF [33]. Evidence has shown that ALB exerts a powerful anti-inflammatory property under certain conditions [22]. Previous studies have shown that ALB is positively correlated with inflammatory factors such as C-reactive protein [22]. Meanwhile, ALB also has important antioxidant effects, which prevent the shortening of atrial action potential duration and delay of after-depolarizations caused by increasing sarcoplasmic reticulum Ca2+ [34–36]. ALB is also involved in left ventricular remodeling, a key link in AF [37]. Moreover, ALB is regarded as an important indicator of individual malnutrition and may interfere with the proportional loss of myocardial muscle and electrophysiological stability due to its deficiency [38–40]. Malnutrition-related AF warrants further investigation.
Additionally, we found that ALB was positively correlated with TC, LDL-C, and APOA1, and there was a consistent relationship between paroxysmal AF and permanent AF with lower ALB, lower blood lipid profiles, and APOA1 in both sexes, suggesting that ALB contributes to AF development by affecting lipid profiles and APOA1. A potential relationship between lipids and AF has been demonstrated, and a cohort study has shown TC and LDL-C levels were inversely related to AF [41]. Likewise, APOA1 exerts anti-inflammatory and antioxidant effects through related enzymes, which may mediate the occurrence of AF and ischemic outcomes [42–44]. Thus, it could be speculated that ALB combined with other clinical factors affects the occurrence and maintenance of AF. The limited sample size and confounding factors may also affect the relationship, which still needs to be confirmed by further studies.
In the present study, we performed a subgroup analysis stratified by sex and AF type to investigate the relationship between ALB and AF. On this basis, we also investigated the relationship between ALB and some metabolic factors of AF. To the best of our knowledge, few such studies have been conducted. Certainly, there might be several potential limitations. First, the outcome of the limited sample size is worth considering. Second, the association between ALB and AF was only analyzed based on single-center retrospective data, but not causality. Third, only paroxysmal AF and permanent AF were included, and the observation of patients with persistent AF was ignored. Fourth, the inflammatory and oxidative stress state of participants was not assessed. Additionally, several confounding factors may not have been adequately adjust for and high comorbidities risk requires further investigation. Finally, due to the limited sample size, we were unable to match hypertension, coronary heart disease, and diabetes in the AF patients and controls. We will address these deficiencies in future research.
Conclusions
In conclusion, the present results showed that in patients at a single center in China, low serum ALB levels in male patients were significantly associated with AF. These findings support those from previous studies in other populations and highlight the importance of monitoring and treating the causes of hypoalbuminemia in cardiac patients, including heart failure, nephrotic syndrome, liver cirrhosis, and nutritional deficiency.
Figures
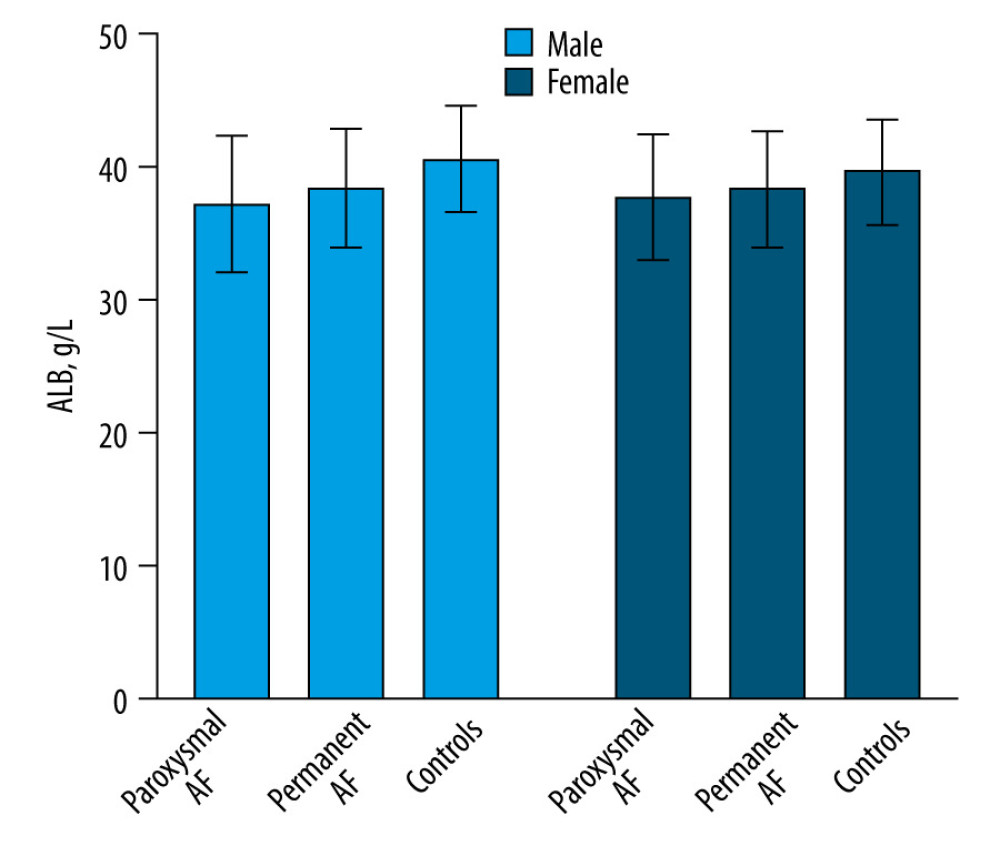 Figure 1. ALB levels in AF patients and controls by sex. Compared with controls, ALB levels of AF patients were significantly lower in men (paroxysmal AF vs permanent AF vs controls: 37.25±5.14 vs 38.39±4.48 vs 40.62± 4.04 g/L, P<0.05). Compared with controls, ALB levels of AF patients were significantly lower in women (paroxysmal AF vs permanent AF vs controls: 37.77±4.75 vs 38.31±4.39 vs 39.63±4.14 g/L, P<0.05). AF – atrial fibrillation; ALB – serum albumin. The figure was created using GraphPad Prism software (version 9.0.0).
Figure 1. ALB levels in AF patients and controls by sex. Compared with controls, ALB levels of AF patients were significantly lower in men (paroxysmal AF vs permanent AF vs controls: 37.25±5.14 vs 38.39±4.48 vs 40.62± 4.04 g/L, P<0.05). Compared with controls, ALB levels of AF patients were significantly lower in women (paroxysmal AF vs permanent AF vs controls: 37.77±4.75 vs 38.31±4.39 vs 39.63±4.14 g/L, P<0.05). AF – atrial fibrillation; ALB – serum albumin. The figure was created using GraphPad Prism software (version 9.0.0). 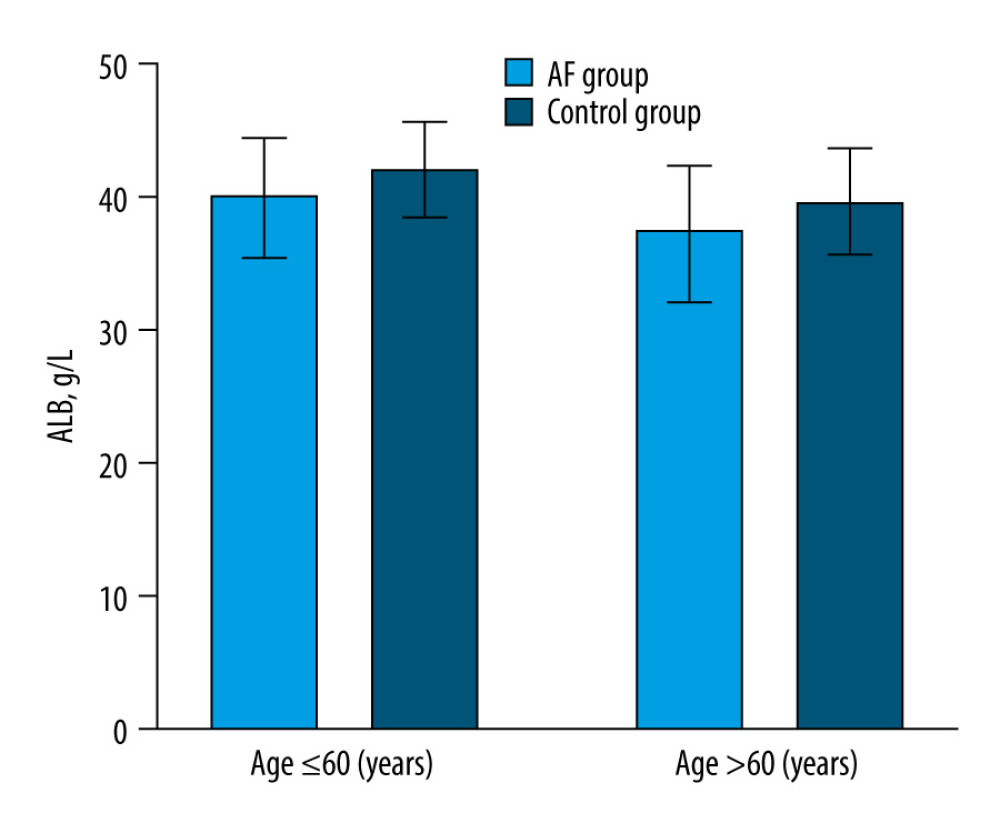 Figure 2. ALB levels in AF patients and controls by age. Compared with controls, ALB levels of AF patients with age ≤60 years were significantly lower (AF group vs control group: 40.04±4.35 vs 41.96±3.54 g/L, P<0.05), ALB levels of AF patients with age >60 years also were significantly lower (AF group vs control group: 37.53±4.57 vs 39.59±4.12 g/L, P<0.05). AF – atrial fibrillation; ALB – serum albumin. The figure was created using GraphPad Prism software (version 9.0.0).
Figure 2. ALB levels in AF patients and controls by age. Compared with controls, ALB levels of AF patients with age ≤60 years were significantly lower (AF group vs control group: 40.04±4.35 vs 41.96±3.54 g/L, P<0.05), ALB levels of AF patients with age >60 years also were significantly lower (AF group vs control group: 37.53±4.57 vs 39.59±4.12 g/L, P<0.05). AF – atrial fibrillation; ALB – serum albumin. The figure was created using GraphPad Prism software (version 9.0.0). 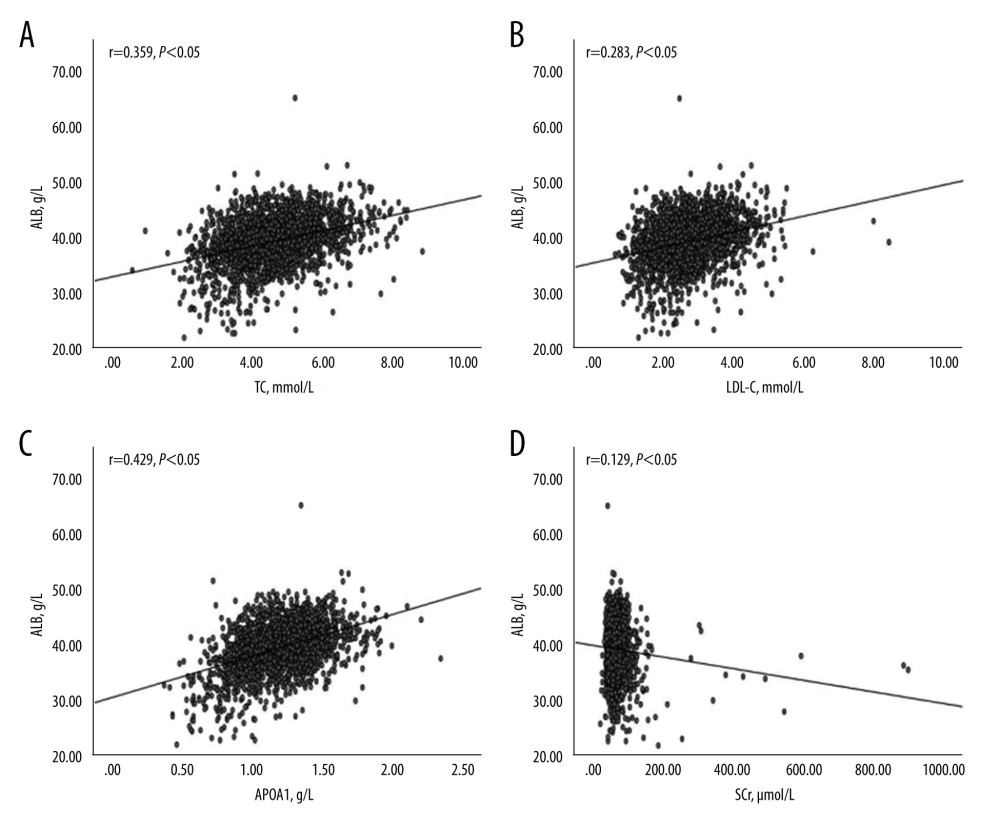 Figure 3. Factors associated with ALB levels in AF patients. (A) Correlation between ALB and TC in AF patients (r=0.359, P<0.05). (B) Correlation between ALB and LDL-C in AF patients (r=0.283, P<0.05). (C) Correlation between ALB and APOA1 in AF patients (r=0.429, P<0.05). (D) Correlation between ALB and SCr in AF patients (r=0.129, P<0.05). ALB – serum albumin; TC – total cholesterol; LDL-C – low-density lipoprotein cholesterol; APOA1 – serum apolipoprotein A1(APOA1); SCr – serum creatinine. The figure was created using SPSS software (version 26.0, SPSS Inc., Chicago, IL, USA).
Figure 3. Factors associated with ALB levels in AF patients. (A) Correlation between ALB and TC in AF patients (r=0.359, P<0.05). (B) Correlation between ALB and LDL-C in AF patients (r=0.283, P<0.05). (C) Correlation between ALB and APOA1 in AF patients (r=0.429, P<0.05). (D) Correlation between ALB and SCr in AF patients (r=0.129, P<0.05). ALB – serum albumin; TC – total cholesterol; LDL-C – low-density lipoprotein cholesterol; APOA1 – serum apolipoprotein A1(APOA1); SCr – serum creatinine. The figure was created using SPSS software (version 26.0, SPSS Inc., Chicago, IL, USA). Tables
Table 1. Baseline characteristics of AF group and controls.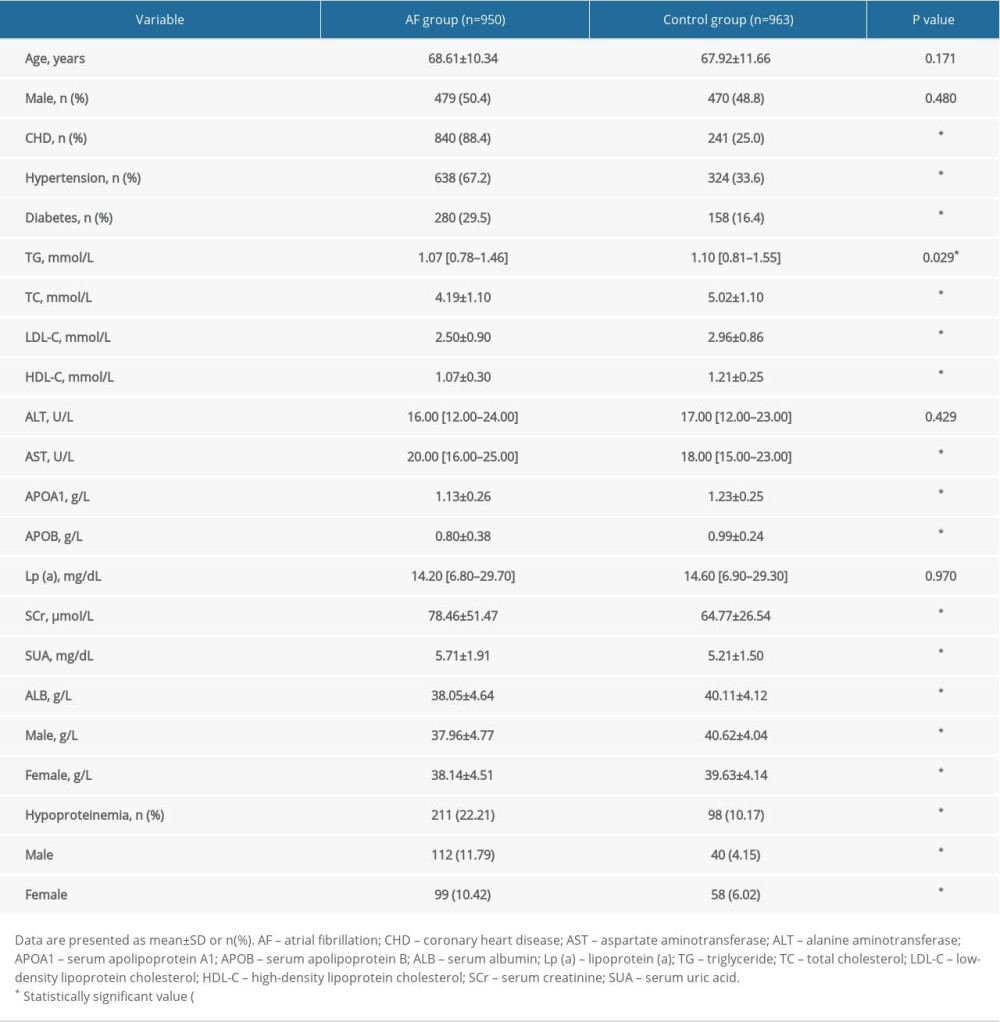 Table 2. Association between ALB and AF.
Table 2. Association between ALB and AF.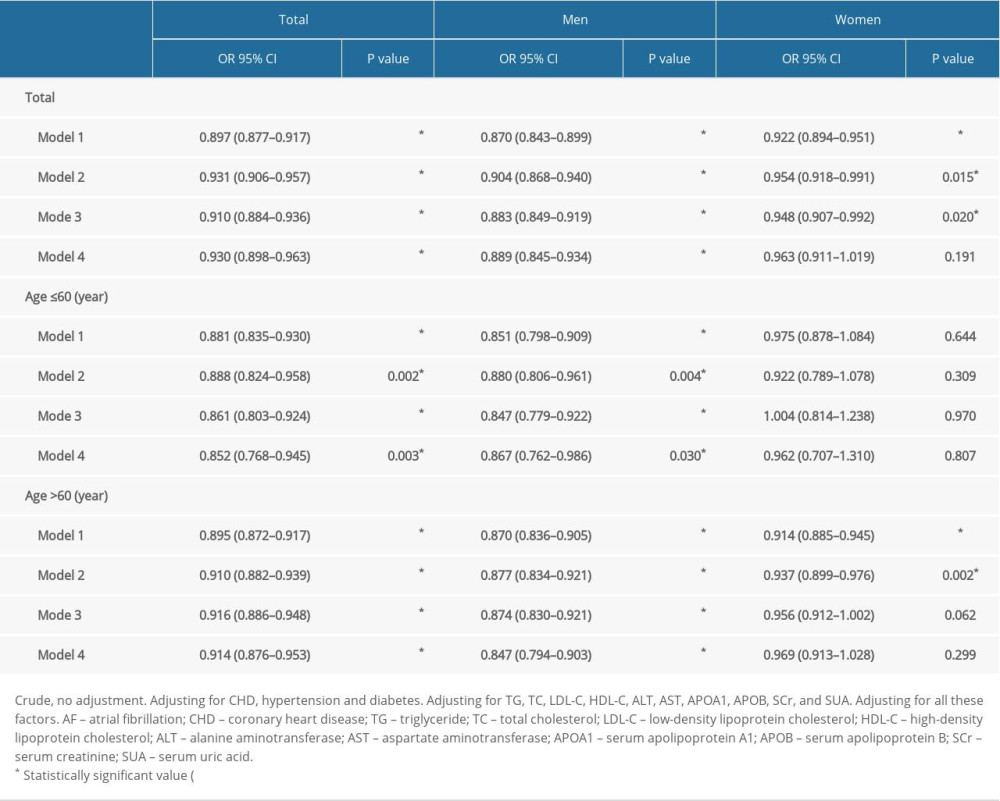 Table 3. Association between ALB levels and AF by type and complication.
Table 3. Association between ALB levels and AF by type and complication.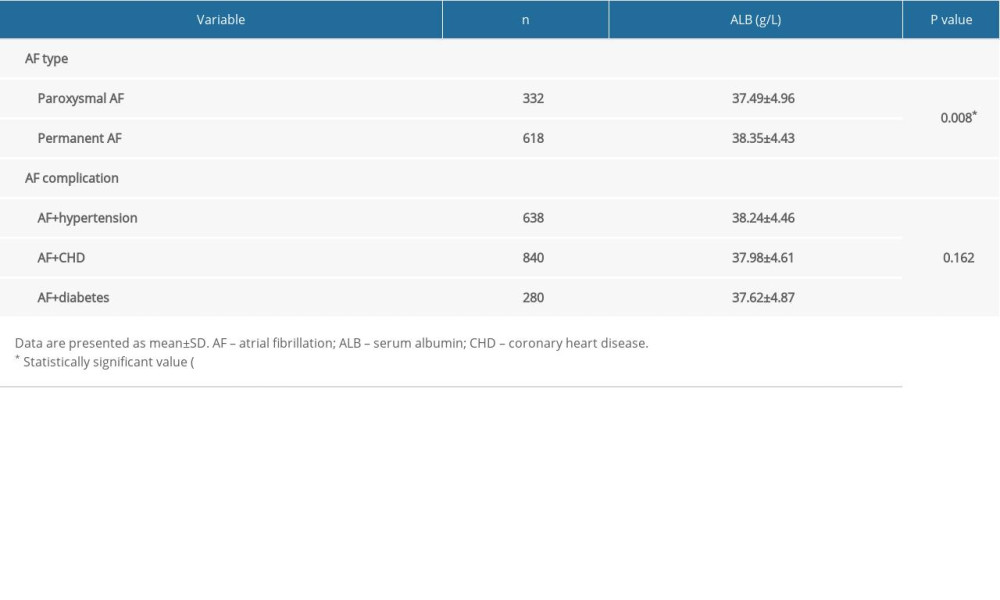 Table 4. Association between levels of SUA and metabolic indicators in patients with paroxysmal AF by sex.
Table 4. Association between levels of SUA and metabolic indicators in patients with paroxysmal AF by sex.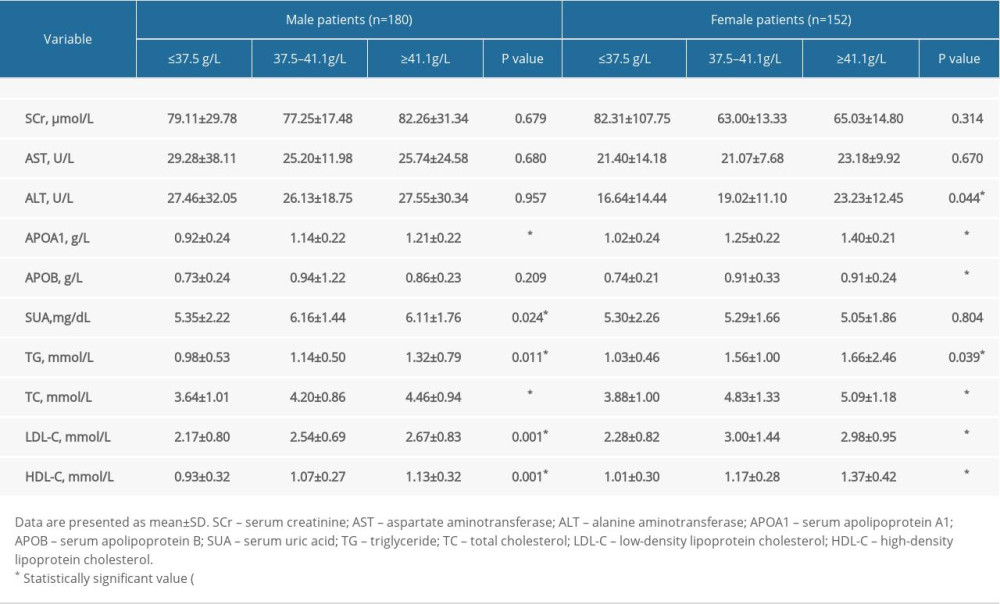 Table 5. Association between ALB levels and metabolic indicators in patients with permanent AF by sex.
Table 5. Association between ALB levels and metabolic indicators in patients with permanent AF by sex.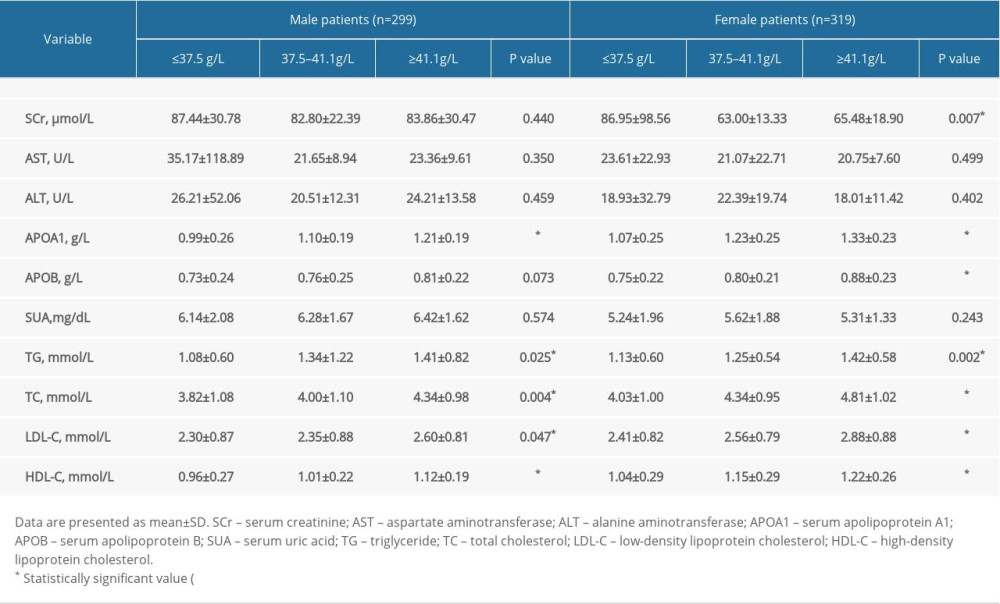
References
1. Waldmann V, Jouven X, Narayanan K, Association between atrial fibrillation and sudden cardiac death: pathophysiological and epidemiological insights: Circ Res, 2020; 127(2); 301-9
2. Chugh SS, Havmoeller R, Narayanan K, Worldwide epidemiology of atrial fibrillation: A Global Burden of Disease 2010 Study: Circulation, 2014; 129(8); 837-47
3. Heeringa J, van der Kuip DA, Hofman A, Prevalence, incidence and lifetime risk of atrial fibrillation: the Rotterdam study: Eur Heart J, 2006; 27(8); 949-53
4. Andrade J, Khairy P, Dobrev D, Nattel S, The clinical profile and pathophysiology of atrial fibrillation: Relationships among clinical features, epidemiology, and mechanisms: Circ Res, 2014; 114(9); 1453-68
5. Ren J, Yang Y, Zhu J, The characteristics, long-term outcomes, risk factors, and antithrombotic therapy in chinese patients with atrial fibrillation and bioprosthetic valves: Front Cardiovasc Med, 2021; 8; 665124
6. Staerk L, Sherer JA, Ko D, Atrial fibrillation: Epidemiology, pathophysiology, and clinical outcomes: Circ Res, 2017; 120(9); 1501-17
7. Lau DH, Nattel S, Kalman JM, Sanders P, Modifiable risk factors and atrial fibrillation: Circulation, 2017; 136(6); 583-96
8. Gupta A, Perera T, Ganesan A, Complications of catheter ablation of atrial fibrillation: A systematic review: Circ Arrhythm Electrophysiol, 2013; 6(6); 1082-88
9. January CT, Wann LS, Alpert JS, 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society [published correction appears in Circulation. 2014;130(23):e270–71]: Circulation, 2014; 130(23); 2071-104
10. Link MS, Haïssaguerre M, Natale A, Ablation of atrial fibrillation: Patient selection, periprocedural anticoagulation, techniques, and preventive measures after ablation: Circulation, 2016; 134(4); 339-52
11. Ganesan AN, Shipp NJ, Brooks AG, Long-term outcomes of catheter ablation of atrial fibrillation: A systematic review and meta-analysis: J Am Heart Assoc, 2013; 2(2); e004549
12. Benjamin EJ, Chen PS, Bild DE, Prevention of atrial fibrillation: Report from a national heart, lung, and blood institute workshop: Circulation, 2009; 119(4); 606-18
13. Kanellis J, Kang DH, Uric acid as a mediator of endothelial dysfunction, inflammation, and vascular disease: Semin Nephrol, 2005; 25(1); 39-42
14. Strazzullo P, Puig JG, Uric acid and oxidative stress: Relative impact on cardiovascular risk?: Nutr Metab Cardiovasc Dis, 2007; 17(6); 409-14
15. Shaper AG, Wannamethee SG, Whincup PH, Serum albumin and risk of stroke, coronary heart disease, and mortality: The role of cigarette smoking: J Clin Epidemiol, 2004; 57(2); 195-202
16. Nelson JJ, Liao D, Sharrett AR, Serum albumin level as a predictor of incident coronary heart disease: The Atherosclerosis Risk in Communities (ARIC) study: Am J Epidemiol, 2000; 151(5); 468-77
17. Liao LZ, Zhang SZ, Li WD, Serum albumin and atrial fibrillation: Insights from epidemiological and mendelian randomization studies: Eur J Epidemiol, 2020; 35(2); 113-22
18. Wang Y, Du P, Xiao Q, Relationship between serum albumin and risk of atrial fibrillation: A dose-response meta-analysis: Front Nutr, 2021; 8; 728353
19. Corti MC, Salive ME, Guralnik JM, Serum albumin and physical function as predictors of coronary heart disease mortality and incidence in older persons: J Clin Epidemiol, 1996; 49(5); 519-26
20. van Beek DEC, Kuijpers YAM, Königs MHH, Low serum albumin levels and new-onset atrial fibrillation in the ICU: A prospective cohort study: J Crit Care, 2020; 56; 26-30
21. Mukamal KJ, Tolstrup JS, Friberg J, Fibrinogen and albumin levels and risk of atrial fibrillation in men and women (the Copenhagen City Heart Study): Am J Cardiol, 2006; 98(1); 75-81
22. Arques S, Human serum albumin in cardiovascular diseases: Eur J Intern Med, 2018; 52; 8-12
23. Fanali G, di Masi A, Trezza V, Human serum albumin: From bench to bedside: Mol Aspects Med, 2012; 33(3); 209-90
24. Kuzuya M, Izawa S, Enoki H, Is serum albumin a good marker for malnutrition in the physically impaired elderly?: Clin Nutr, 2007; 26(1); 84-90
25. Don BR, Kaysen G, Serum albumin: Relationship to inflammation and nutrition: Semin Dial, 2004; 17(6); 432-37
26. Tamariz L, Hernandez F, Bush A, Palacio A, Hare JM, Association between serum uric acid and atrial fibrillation: A systematic review and meta-analysis: Heart Rhythm, 2014; 11(7); 1102-8
27. Kuwabara M, Niwa K, Nishihara S, Hyperuricemia is an independent competing risk factor for atrial fibrillation: Int J Cardiol, 2017; 231; 137-42
28. Calkins H, Hindricks G, Cappato R, 2017 HRS/EHRA/ECAS/APHRS/SOLAECE expert consensus statement on catheter and surgical ablation of atrial fibrillation: Europace, 2018; 2(1); e1-e160
29. Sullivan SA, Van Le L, Association between hypoalbuminemia and surgical site infection in vulvar cancers: Gynecol Oncol, 2016; 142(3); 435-39
30. Soeters PB, Wolfe RR, Shenkin A, Hypoalbuminemia: Pathogenesis and clinical significance: J Parenter Enteral Nutr, 2019; 43(2); 181-93
31. Ronit A, Kirkegaard-Klitbo DM, Dohlmann TL, Plasma albumin and incident cardiovascular disease: Results from the CGPS and an updated meta-analysis: Arterioscler Thromb Vasc Biol, 2020; 40(2); 473-82
32. Peacock F, Morris DL, Anwaruddin S, Meta-analysis of ischemia-modified albumin to rule out acute coronary syndromes in the Emergency Department: Am Heart J, 2006; 152(2); 253-62
33. Hu YF, Chen YJ, Lin YJ, Chen SA, Inflammation and the pathogenesis of atrial fibrillation: Nat Rev Cardiol, 2015; 12(4); 230-43
34. Roche M, Rondeau P, Singh NR, The antioxidant properties of serum albumin: FEBS Lett, 2008; 582(13); 1783-87
35. Sovari AA, Dudley SC, Reactive oxygen species-targeted therapeutic interventions for atrial fibrillation: Front Physiol, 2012; 3; 311
36. Ziolo MT, Mohler PJ, Defining the role of oxidative stress in atrial fibrillation and diabetes: J Cardiovasc Electrophysiol, 2015; 26(2); 223-25
37. Enia G, Mallamaci F, Benedetto FA, Long-term CAPD patients are volume expanded and display more severe left ventricular hypertrophy than haemodialysis patients: Nephrol Dial Transplant, 2001; 16(7); 1459-64
38. Annoura M, Ogawa M, Kumagai K, Cholesterol paradox in patients with paroxysmal atrial fibrillation: Cardiology, 1999; 92(1); 21-27
39. Webb JG, Kiess MC, Chan-Yan CC, Malnutrition and the heart: CMAJ, 1986; 135(7); 753-58
40. He YM, Yang XJ, Hui J, Low serum albumin levels in patients with paroxysmal atrial fibrillation: What does it mean?: Acta Cardiol, 2006; 61(3); 333-37
41. Li X, Gao L, Wang Z, Lipid profile and incidence of atrial fibrillation: A prospective cohort study in China: Clin Cardiol, 2018; 41(3); 314-20
42. Du R, Winarsih I, Ho B, Ding JL, Lipid-free apolipoprotein A-I exerts an antioxidative role against cell-free hemoglobin: Am J Clin Exp Immunol, 2012; 1(1); 33-48
43. Barter PJ, Puranik R, Rye KA, New insights into the role of HDL as an anti-inflammatory agent in the prevention of cardiovascular disease: Curr Cardiol Rep, 2007; 9(6); 493-98
44. Pol T, Held C, Westerbergh J, Dyslipidemia and risk of cardiovascular events in patients with atrial fibrillation treated with oral anticoagulation therapy: Insights from the ARISTOTLE (Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation) Trial: J Am Heart Assoc, 2018; 7(3); e007444
Figures
 Figure 1. ALB levels in AF patients and controls by sex. Compared with controls, ALB levels of AF patients were significantly lower in men (paroxysmal AF vs permanent AF vs controls: 37.25±5.14 vs 38.39±4.48 vs 40.62± 4.04 g/L, P<0.05). Compared with controls, ALB levels of AF patients were significantly lower in women (paroxysmal AF vs permanent AF vs controls: 37.77±4.75 vs 38.31±4.39 vs 39.63±4.14 g/L, P<0.05). AF – atrial fibrillation; ALB – serum albumin. The figure was created using GraphPad Prism software (version 9.0.0).
Figure 1. ALB levels in AF patients and controls by sex. Compared with controls, ALB levels of AF patients were significantly lower in men (paroxysmal AF vs permanent AF vs controls: 37.25±5.14 vs 38.39±4.48 vs 40.62± 4.04 g/L, P<0.05). Compared with controls, ALB levels of AF patients were significantly lower in women (paroxysmal AF vs permanent AF vs controls: 37.77±4.75 vs 38.31±4.39 vs 39.63±4.14 g/L, P<0.05). AF – atrial fibrillation; ALB – serum albumin. The figure was created using GraphPad Prism software (version 9.0.0). Figure 2. ALB levels in AF patients and controls by age. Compared with controls, ALB levels of AF patients with age ≤60 years were significantly lower (AF group vs control group: 40.04±4.35 vs 41.96±3.54 g/L, P<0.05), ALB levels of AF patients with age >60 years also were significantly lower (AF group vs control group: 37.53±4.57 vs 39.59±4.12 g/L, P<0.05). AF – atrial fibrillation; ALB – serum albumin. The figure was created using GraphPad Prism software (version 9.0.0).
Figure 2. ALB levels in AF patients and controls by age. Compared with controls, ALB levels of AF patients with age ≤60 years were significantly lower (AF group vs control group: 40.04±4.35 vs 41.96±3.54 g/L, P<0.05), ALB levels of AF patients with age >60 years also were significantly lower (AF group vs control group: 37.53±4.57 vs 39.59±4.12 g/L, P<0.05). AF – atrial fibrillation; ALB – serum albumin. The figure was created using GraphPad Prism software (version 9.0.0). Figure 3. Factors associated with ALB levels in AF patients. (A) Correlation between ALB and TC in AF patients (r=0.359, P<0.05). (B) Correlation between ALB and LDL-C in AF patients (r=0.283, P<0.05). (C) Correlation between ALB and APOA1 in AF patients (r=0.429, P<0.05). (D) Correlation between ALB and SCr in AF patients (r=0.129, P<0.05). ALB – serum albumin; TC – total cholesterol; LDL-C – low-density lipoprotein cholesterol; APOA1 – serum apolipoprotein A1(APOA1); SCr – serum creatinine. The figure was created using SPSS software (version 26.0, SPSS Inc., Chicago, IL, USA).
Figure 3. Factors associated with ALB levels in AF patients. (A) Correlation between ALB and TC in AF patients (r=0.359, P<0.05). (B) Correlation between ALB and LDL-C in AF patients (r=0.283, P<0.05). (C) Correlation between ALB and APOA1 in AF patients (r=0.429, P<0.05). (D) Correlation between ALB and SCr in AF patients (r=0.129, P<0.05). ALB – serum albumin; TC – total cholesterol; LDL-C – low-density lipoprotein cholesterol; APOA1 – serum apolipoprotein A1(APOA1); SCr – serum creatinine. The figure was created using SPSS software (version 26.0, SPSS Inc., Chicago, IL, USA). Tables
 Table 1. Baseline characteristics of AF group and controls.
Table 1. Baseline characteristics of AF group and controls. Table 2. Association between ALB and AF.
Table 2. Association between ALB and AF. Table 3. Association between ALB levels and AF by type and complication.
Table 3. Association between ALB levels and AF by type and complication. Table 4. Association between levels of SUA and metabolic indicators in patients with paroxysmal AF by sex.
Table 4. Association between levels of SUA and metabolic indicators in patients with paroxysmal AF by sex. Table 5. Association between ALB levels and metabolic indicators in patients with permanent AF by sex.
Table 5. Association between ALB levels and metabolic indicators in patients with permanent AF by sex. Table 1. Baseline characteristics of AF group and controls.
Table 1. Baseline characteristics of AF group and controls. Table 2. Association between ALB and AF.
Table 2. Association between ALB and AF. Table 3. Association between ALB levels and AF by type and complication.
Table 3. Association between ALB levels and AF by type and complication. Table 4. Association between levels of SUA and metabolic indicators in patients with paroxysmal AF by sex.
Table 4. Association between levels of SUA and metabolic indicators in patients with paroxysmal AF by sex. Table 5. Association between ALB levels and metabolic indicators in patients with permanent AF by sex.
Table 5. Association between ALB levels and metabolic indicators in patients with permanent AF by sex. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








