06 April 2022: Review Articles
Signal Crosstalk and the Role of Estrogen Receptor beta (ERβ) in Prostate Cancer
Jialin Li1ACEF, Quanliang Liu2AE*, Chengming Jiang1BFDOI: 10.12659/MSM.935599
Med Sci Monit 2022; 28:e935599
Abstract
ABSTRACT: Prostate cancer remains the most prevalent cancer among men worldwide; however, as a sex hormone-dependent cancer, sex hormones and their receptor signaling play an important role in the development and progression of cancer. Most current treatment options for prostate cancer thus revolve around the inhibition of androgen signaling (eg, ADT), which, although effective in the early stages, eventually progresses to treatment-resistant prostate cancer with no effective follow-up options. Recent studies have shown that among the nuclear receptor family members, in addition to androgen receptors, estrogen receptor (ER) plays an important biological function as a transcription factor and regulatory protein in various cancers, acting either directly or indirectly by forming homodimers or heterodimers with ligands. In this paper, we review the application of ERβ in animal models and in vitro experiments in the last 5 years, as well as the presence and role of some of its splice variants. We summarize the overview and update of ERβ in prostate cancer, and provide a corresponding analysis of some current research disagreements. Its crosstalk action on some important cancer growth-related signaling pathways (eg, TGF-β and ERK), regulation of downstream target proteins (eg, nuclear translocation of EGFR and expression of oncogenic -related protein MMP-2), and interactions with related ERβ co-regulators (eg, ZFHX3), agonists, and antagonists in prostate cancer are highlighted, and the resulting effects on tumor progression are described. In addition, the paper describes its current potential clinical application as a novel therapeutic strategy and some of the challenges it faces.
Keywords: Estrogen Receptor beta, Phyllodes Tumor of the Prostate, Prostatic Neoplasms, Receptors, Androgen, Animals, Cell Line, Tumor, Humans, Male, Prostate, Receptors, Estrogen
Background
Prostate cancer is the most commonly diagnosed cancer worldwide, and is the most common cancer in men. According to World Health Organization statistics, there were about 1 276 000 new cases and 359 000 related deaths in 2018 [1]. The androgen receptor (AR) pathway is considered to be the main factor in the pathogenesis of prostate cancer [2], and most patients benefit from androgen deprivation therapy (ADT). Unfortunately, although there is some curative effect in the early stage, the inevitable progression is to androgen castration-resistant prostate cancer (CRPC) [3,4]. Multiple life-prolonging treatments for CRPC have emerged, including systemic taxane chemotherapy, androgen receptor pathway inhibitors, radioisotope therapy, and cell immunotherapy [5,6]. However, such therapeutic strategies are limited by the nature of metastatic tissues or are complicated by new somatic cell mutations at the metastatic sites, resulting in a weakening correlation with primary cancer.
In the past, the treatment of prostate cancer has focused on inhibiting the androgen signaling pathway, while discounting the important regulatory role of estrogen receptor beta [7,8].
Prostate cancer development is closely related to estrogen receptor action, which includes 2 main subtypes: estrogen receptor α (ERα) and estrogen receptor β (ERβ) [9]. Estrogen receptor signaling regulates a variety of physiological processes, such as cell growth, apoptosis, and differentiation, and abnormally altered signaling often causes a series of malignant changes in prostate cancer [10,11]. The present study reviews the role of ERβ and provides an updated perspective, with a focus on its crosstalk with other signals and role in cancer. In addition, potential clinical applications and some of the current issues faced are discussed.
The Structure of Estrogen Receptorsβ (ERβ)
ERβ is a member of the nuclear hormone receptor superfamily, with regulatory functions as a transcription factor. In the human genome, on the one hand, ERα has a full length of 595 amino acids and 67 kDa and is encoded by the ESR1 gene located on chromosome 6 at locus 6q25.1; on the other hand, ERβ has a full length of 530 amino acids and 59 kDa and is encoded by the ESR2 gene located on chromosome 14 at locus 14q23.2 [12,13]. Structurally, ERα and ERβ are composed of various functional domains and regions: the N-terminal transactivation domain (NTD), DNA-binding domain (DBD), hinge region, C-terminal co-activator/cofactor interaction domain, and ligand-binding domain (LBD) (Figure 1) [13]. The NTD contains zinc-finger structures capable of binding and transcriptionally activating target genes, and it is only 16% similar to another estrogen receptor subtype, ERα, and this is one of the most significant structural differences between the them [14]. DBD mediates ERβ with estrogen response elements (EREs) in target genes, which promote ERβ dimerization and binding to chromosome-specific sequences (Figure 2). The hinge region is capable of binding to molecular chaperones and involves nuclear localization signals. Finally, the carboxyl terminus, which is known as the ligand-binding domain (LBD), contains the binding sites for co-activators, co-repressors, and the estrogen binding region [15]. LBD also includes a ligand-dependent activation domain (AF2) [16].
Alternative splicing of ER-mRNAs leads to the existence of several splice variants of ERβ, which have different roles but are structurally similar (Figure 1) [17]. For example, it has been suggested that ERβ splice variants are associated with biological behaviors such as drug resistance and apoptosis in prostate cancer [18,19]. Faria et al found that, in prostate cancer, ERβ2 and ERβ can induce stem cell properties of tumors as well as the development of chemoresistance. In contrast, in the same study, ERβ5 was shown to enhance the sensitivity of cancer cells to chemotherapeutic agents [20]. In most cases, the various splice variants are co-expressed in cells; therefore, the expression ratio between splice variants is likely to be a key determinant of clinical therapeutic efficacy.
Estrogen Receptors (ERs) in Prostate Cancer
Prostate tissue mainly expresses 2 types of estrogen receptors, ERα and Erβ. Although they are structurally similar, their areas of expression and effects are different. ERα is mainly expressed in prostate stromal cells [21], while ERβ is mainly expressed in prostate epithelial cells and stromal cells, mediating the differentiation of prostate epithelium and the maintenance of cell morphology [22].
Studies on ERα in prostate cancer have consistently suggested that this receptor protein is highly expressed in prostate cancer cells as a pro-oncogene, facilitating the processes of cancer cell proliferation, invasion, and migration, and gradually increasing the expression level with the grading and staging of the tumor [23,24]. In the VP (ventral prostate) ERαKO and ERα overexpression mouse models established by Omoto et al, ERα showed extremely high proliferative effects on normal prostate tissue and induced intraepithelial neoplasia in prostate tissue [25], suggesting that ERα plays a positive activating role in the growth of prostate cells. In addition, it was noted in several studies that the site of ERα expression changes after cancer genesis, mainly in the form of high levels of ERα expression detected in epithelial cells, which were predominantly expressed in prostate stromal cells [22,26,27]. In contrast, there are some controversial studies on ERβ, most of which suggest that ERβ is low or undetectable in prostate cancer tissues and plays a cancer-suppressive role, further confirming the suppressive effect of ERβ in prostate cancer [28,29]. However, a few recent studies have suggested that ERβ may also have a carcinogenic role. Migliaccio et al found that in LNCaP cells (an estrogen-versus androgen-dependent prostate cancer cell line), ERβ was able to drive the cells into S-phase and promote cell proliferation and epidermal growth factor secretion [30]. In a clinical study, an assay of CPEC (cells expressing prostate-specific antigens) in prostate cancer patient specimens found that ERβ, but not ERα, enhanced the survival and migration of CPEC [31]. Both of these lines of evidence suggest a possible dual role for ERβ in prostate cancer.
The discrepancies in studies on the role of ERβ in the prostate may be due to a variety of factors. First, there are multiple splice variants of ERβ and the differences in their effects, and the specificity of each splice variant of ERβ currently on the market is not high. Second, all of the immunohistochemical analyses of steroid receptors rely on cell permeabilization, which leads to the possibility of nuclear or cytoplasmic staining of ERβ. Third, expression of antibodies against the amino-terminal structural domain of ERβ in
The Regulatory Effect of ERβ on AR Signal in Prostate Cancer
Androgen signaling is critical in prostate cancer, but the mechanism is poorly understood, and ERβ may play an important function in the AR signaling pathway. Recent investigations have shown that androgen-activated androgen receptors can interact not only directly through binding to estrogen response elements, but also indirectly with SP-1 (specificity protein-1), AF-1 (activation protein-1), and nuclear factor κB proteins by binding to the corresponding ligands and regulating AR signaling at the transcriptional or post-transcriptional level, which in turn affects prostate cancer progression [32–35]. Researchers found elevated epithelial hyperplasia and AR downstream genes level in a ERβ knockout mouse model, most of which were also upregulated in prostate cancer, prompting consideration of whether crosstalk exists between these 2 signaling pathways [36]. Subsequently, Chaurasiva et al used ERβ overexpression and LY3201 (ERβ-specific binding ligand) to enhance ERβ activity in LNCaP cell lines (AR-positive) to investigate alteration of the AR signaling pathway and related genes, which resulted in not only the downregulation of AR expression at transcriptional and protein levels, but also the decrease of related genes such as FKBP5 and TBC1D4. ERβ has also been shown to affect the expression level of FKBP5 by binding to its intron region [37], but the mechanism of action on TBC1D4 needs further investigation. In addition, ligand-activated ERβ was also found to inhibit the phosphorylation of its target through the regulation of CAMKK2, which inhibits the AMPK signaling pathway, which in turn affects the metabolism of tumor cells. Wu et al reached similar conclusions in establishing an ERβ-deficient mouse VP model, whereby ERβ indirectly affects the activation of AR signaling by inhibiting the AR co-activator RORC, or by enhancing the action of the AR co-repressor DACH1 [38]. Whether it affects the expression level of AR or the regulation of its control genes by AR, these indicate a close regulatory linkage of ERβ in the AR signaling pathway.
The expression level of ERβ in prostate cancer gradually decreased with increasing Gleason’s score grade, during which epidermal growth factor receptor (EGFR) shifted from the cell surface to the nucleus and there was enhanced expression of proliferating cell nuclear antigen (PCNA) on epithelial cells. In a variety of types of tumors, the nuclear localization of EGFR is closely related to the progression of cancer and patient survival. For instance, nuclear localization of EGFR enhances the resistance of cancer cells to radiotherapy and to EGFR drugs [39,40], while AR plays an indispensable function in this process and is regulated by ERβ. In the clinical use of combined ADT+enzalutamide, Wu et al found increased the nuclear translocation of EGFR in prostate cells, implying that this blockade of AR can affect the nuclear translocation of EGFR. After short-term treatment of such patients with an AR signaling pathway inhibitor (abiraterone), it was found that early specific blockade of AR signaling was followed by an increase in ERβ expression and inhibition of nuclear translocation of EGFR was inhibited, while nuclear translocation of the oncogene PTEN was stimulated, leading to inhibition of prostate cell growth. However, with the prolongation of ADT treatment, ERβ expression in prostate cancer gradually disappears. At this time, when the same treatment is given to patients, the initial treatment effect is often not attained or is of little benefit, and the tumor also further deteriorates to a higher grade [41]. As important co-regulatory receptor proteins in prostate cancer, AR and ERβ interact with each other and crosstalk in signaling, which has a significant impact on the pathogenesis of prostate cancer. Therefore, maintenance of the balance of action between AR and ERβ is crucial for the treatment of prostate cancer.
The Role of ERβ in Prostate Cancer in Relation to Other Signaling Molecules
The ERβ acts as an essential tumor regulator, regulating processes such as cell proliferation and metabolism by direct or indirect means. In vivo, ERβ normally binds to specific ligands, forming homo- or heterodimers that modify target genes at the transcriptional and post-transcriptional levels. Zinc-finger homeobox 3 (ZFHX3) is a class of transcription factors that package multiple zinc-finger structural domains and other motifs, which are often mutated in metastatic prostate cancer or high-grade prostate cancer [42]. Several studies have shown that deletion of ZFHX3 not only leads to prostate intraepithelial neoplasia in mice, but also affects the expression and function of PTEN and Myc, inducing prostate cancer development [43,44]. Hu et al found that ZFHX3 is an indispensable factor for ERβ tumor-suppressive activity and by binding regulates the activation state of ERβ. In further experiments, the oncogenic effects of ERβ and its agonist DPN (including inhibition of clone formation and Myc expression) were dramatically attenuated after inhibition of ZFHX3 expression on C4-2B and LNCaP cells [45], which suggests that ZFHX3 expression and activation need to be considered in clinical treatment of prostate cancer targeting agonists to restore or enhance ERβ activity.
The ERβ in prostate cancer is triggered by ligand activation, which further translocates to the nucleus and binds to specific sequences on nuclear DNA. This suggests that plasmonuclear translocation of ERβ may play an important role in prostate cancer. CRM1 (chromosome region maintenance 1) is a class of nuclear transport proteins, and a study of an androgen-independent prostate cancer PC-3 cell line showed an essential contribution of CRM1, which is involved in intracellular ERβ translocation between nucleoplasms [46]. Rice et al found that ER signaling could promote phosphorylation of ERK1/2 in PC-3 cells by forming homo- or heterodimers interacting with Src after rapid activation in the extranuclear area, but ERβ was mainly distributed outside the nucleus in PC-3 cells and the use of estrogen did not affect the localization of ERs after 24 h [47], suggesting that another protein or proteins are involved in ERβ transport in this process. Further tests revealed that CRM1 may be involved in this process and that it indirectly regulates the activation level of ERs by affecting the subcellular localization of ERs. After specifically reducing CRM1 expression in cells, reactivation of ERβ revealed a significant decrease in ERK1/2 phosphorylation levels in PC-3 cells at this time compared to the previous level, and a decrease in ERβ levels in the nucleus [48]. Rapid activation signaling of ERs in the extranuclear region is extremely important, affecting the activation of multiple signaling pathways, including MAPK/ERK, whereby endocrine therapy by blocking pathways associated with the nuclear export of ERβ can be an important strategy for the treatment of prostate cancer in clinical practice. However, in several studies on androgen non-dependent prostate cancer cells, it was noted that silencing ERβ was able to upregulate the expression level of p-ERK1/2 in prostate cancer cells, activate the ERK1/2 signaling pathway, and promote cell proliferation by decreasing the ratio of G0/G1 phase cells, increasing cyclinD1 and other effects on the cell cycle, along with anti-apoptosis [49,50].
In addition, upon the decrease of ERβ expression, the expression of metalloproteinase-2 (MMP-2), vascular endothelial growth factor (VEGF), and other key proteins for invasion and migration also appeared to be elevated, accompanied by the enhanced invasion and migration ability of prostate cancer cells. This result was further validated in a nude mouse model, in which the transplanted tumor volume and metastatic ability were significantly greater in the ERβ-silenced nude mouse model than in the control group [18,49,51]. Xiao et al showed, by regulating ERβ expression in androgen non-dependent prostate cancer cells, that transforming growth factor-1 (TGFβ-1) and insulin-like growth factor-1 (IGF-1) expression was significantly reduced when ERβ expression was upregulated, thereby inhibiting the expression of TGFβ-1 class IGF-1 downstream anti-apoptotic proteins Bcl-2 and Survivin and inducing apoptosis. After the combined use of ER inhibitor PHTPP and TGFβ inhibitor LY2109761, the expression of anti-apoptotic proteins Survivin and Bcl-2 with TGFβ-1 and IGF-1 were all downregulated compared with the control and PHTPP groups [52], indicating that in androgen non-dependent prostate cancer cells, both of these anti-apoptotic proteins were regulated by the combination of ERβ and TGFβ/IGF-1. Each of these studies suggests that ERβ may provide a new therapeutic strategy for clinical use in the treatment of anti-androgen non-hormone-dependent prostate cancer.
Conclusions
Prostate cancer is the second most common hormone-dependent tumor after breast cancer, with changes in the levels of sex hormones and their receptors having a significant impact on cancer pathogenesis and etiology. Studies on the actions of ERβ have demonstrated its suppressive effects in cancers, but some splice variants present in ERβ also exhibit opposing cancer growth-promoting effects. In conclusion, the study of the effects of ERβ in cell biology should combine a variety of factors, including the cell type, cancer stage, the ligands and other regulatory factors bound, and the proportion of each splice variant expressed in the cancer. More studies are needed to elaborate on ERβ in normal prostate and prostate cancer.
Our investigation of ERβ revealed that among multiple tumor types, ERβ crosstalk with other ligands and signaling pathways exists and promotes the amplification of cancer cell growth signals or activation of cascade responses, accelerating further differentiation of cancer cells to higher malignancy. In particular, in prostate cancer, ERβ can interact with AR to modify the upstream regulators and downstream target genes of AR at the transcriptional or post-transcriptional levels; or it can promote the phosphorylation levels of key proteins in the ERK1/2 MAPK and AMPK pathways to influence the activation status of the signaling pathway, thus regulating the expression levels and effects of various target proteins such as VEGF, Bcl-2, cyclinD1, and MMP-2 in prostate cancer cells, affecting the processes of prostate proliferation, invasion, migration, and apoptosis. Multiple regulators (ZFHX3, CRM1, and others) are involved in this process and mediate the level of ERβ action.
Despite what is known about ERβ, there are still many questions about the molecular events of estrogen and its receptors during prostate cancer progression, in which new findings will always further complicate the current conclusions. For example, the splice variant isoforms identified in the study of ERβ produce completely different cell biological effects when forming homo- and heterodimers, a result that always seems to be contradictory for reasons related to the failure to distinguish the isoforms for systematic evaluation. In addition, ERβ is now known to act by affecting the nuclear translocation of some proteins, like EGFR, along with the translocation activation of some oncogenes, besides regulating the expression of some target proteins.
Targeted therapy for tumors is currently attracting much attention as a focus of clinical treatment. Despite a series of achievements in targeted therapy for prostate cancer, including novel anti-androgens, polyene paclitaxel, and radium-223, prostate cancer will eventually develop into a drug-resistant type, so new effective targets need to be found. Further understanding of the role of ERβ in prostate cancer will enhance our understanding of the molecular mechanisms of prostate cancer development and provide assistance in the future treatment of this disease.
Figures
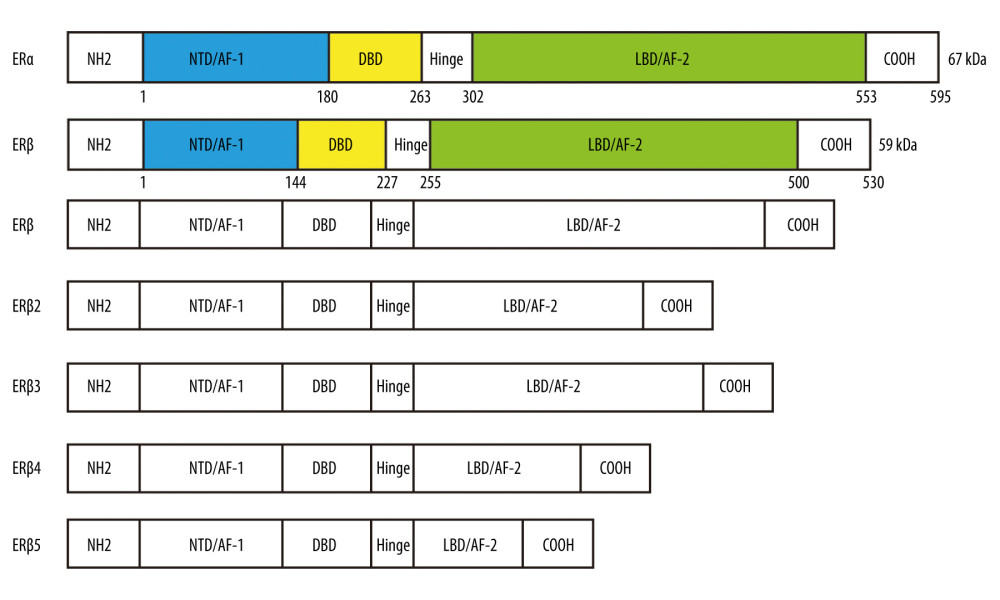 Figure 1. Structures of ERα, ERβ, and several of their major splice variant (ERβ2, ERβ3, ERβ4, ERβ5) proteins: N-terminal transactivation domain (NTD), DNA-binding domain (DBD), hinge region, C-terminal co-activator/cofactor interaction domain and ligand-binding domain (LBD), amino-terminal, and carboxy-terminal. (Created using Adobe Illustrator CS6, Version 16.0.0.682, Adobe Systems, Incorporated).
Figure 1. Structures of ERα, ERβ, and several of their major splice variant (ERβ2, ERβ3, ERβ4, ERβ5) proteins: N-terminal transactivation domain (NTD), DNA-binding domain (DBD), hinge region, C-terminal co-activator/cofactor interaction domain and ligand-binding domain (LBD), amino-terminal, and carboxy-terminal. (Created using Adobe Illustrator CS6, Version 16.0.0.682, Adobe Systems, Incorporated). 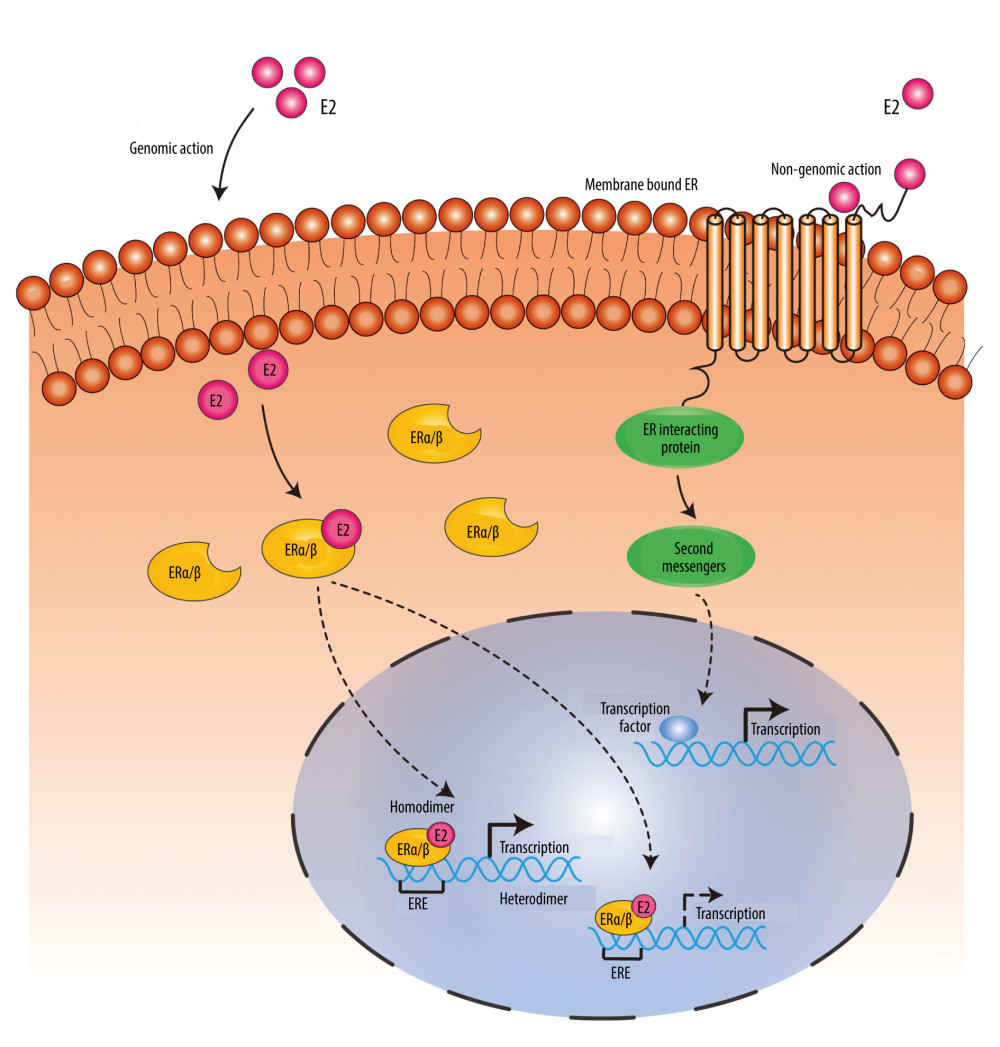 Figure 2. In genomic action, estrogen regulates the transcription of target genes by binding to and activating inactive ER receptors in the cytoplasm or nucleus, and the activated estrogen receptor monomer forms a dimer with another activated estrogen receptor monomer, which in combination with other co-regulatory proteins (not labeled) regulates the transcription of target genes after binding to the estrogen response element (ERE) within the DNA. Non-genomic action: Estrogen induces the initiation of estrogen action by binding to receptors on the cell membrane, which in turn regulates downstream protein expression by acting as second messengers and transcription factors. (Created using Adobe Illustrator CS6, Version 16.0.0.682, Adobe Systems, Incorporated).
Figure 2. In genomic action, estrogen regulates the transcription of target genes by binding to and activating inactive ER receptors in the cytoplasm or nucleus, and the activated estrogen receptor monomer forms a dimer with another activated estrogen receptor monomer, which in combination with other co-regulatory proteins (not labeled) regulates the transcription of target genes after binding to the estrogen response element (ERE) within the DNA. Non-genomic action: Estrogen induces the initiation of estrogen action by binding to receptors on the cell membrane, which in turn regulates downstream protein expression by acting as second messengers and transcription factors. (Created using Adobe Illustrator CS6, Version 16.0.0.682, Adobe Systems, Incorporated). References
1. Culp MB, Soerjomataram I, Efstathiou JA, Recent global patterns in prostate cancer incidence and mortality rates: Eur Urol, 2020; 77(1); 38-52
2. Howard N, Clementino M, Kim D, New developments in mechanisms of prostate cancer progression: Semin Cancer Biol, 2019; 57; 111-16
3. Chen X, Lu J, Xia L, Li G, Drug resistance of enzalutamide in CRPC: Curr Drug Targets, 2018; 19(6); 613-20
4. Moussa M, Papatsoris A, Abou Chakra M, Pharmacotherapeutic strategies for castrate-resistant prostate cancer: Expert Opin Pharmacother, 2020; 21(12); 1431-48
5. Hussain M, Fizazi K, Saad F, Enzalutamide in men with nonmetastatic, castration-resistant prostate cancer: N Engl J Med, 2018; 378(26); 2465-74
6. Teo MY, Rathkopf DE, Kantoff P, Treatment of advanced prostate cancer: Annu Rev Med, 2019; 70; 479-99
7. Bonkhoff H, Estrogen receptor signaling in prostate cancer: Implications for carcinogenesis and tumor progression: Prostate, 2018; 78(1); 2-10
8. Miao L, Yun X, Tao R, Bakuchiol exhibits anti-metastasis activity through NF-κB cross-talk signaling with AR and ERβ in androgen-independent prostate cancer cells PC-3: J Pharmacol Sci, 2018; 138(1); 1-8
9. Hua H, Zhang H, Kong Q, Jiang Y, Mechanisms for estrogen receptor expression in human cancer: Exp Hematol Oncol, 2018; 7; 24
10. Tang L, Platek ME, Yao S, Associations between polymorphisms in genes related to estrogen metabolism and function and prostate cancer risk: Results from the Prostate Cancer Prevention Trial: Carcinogenesis, 2018; 39(2); 125-33
11. Di Zazzo E, Galasso G, Giovannelli P, Estrogens and their receptors in prostate cancer: Therapeutic implications: Front Oncol, 2018; 8; 2
12. Herynk MH, Fuqua SA, Estrogen receptor mutations in human disease: Endocr Rev, 2004; 25(6); 869-98
13. Fuentes N, Silveyra P, Estrogen receptor signaling mechanisms: Adv Protein Chem Struct Biol, 2019; 116; 135-70
14. Kumar R, Zakharov MN, Khan SH, The dynamic structure of the estrogen receptor: J Amino Acids, 2011; 2011; 812540
15. Ascenzi P, Bocedi A, Marino M, Structure-function relationship of estrogen receptor alpha and beta: impact on human health: Mol Aspects Med, 2006; 27(4); 299-402
16. Altwegg KA, Vadlamudi RK, Role of estrogen receptor coregulators in endocrine resistant breast cancer: Explor Target Antitumor Ther, 2021; 2; 385-400
17. Moore JT, McKee DD, Slentz-Kesler K, Cloning and characterization of human estrogen receptor beta isoforms: Biochem Biophys Res Commun, 1998; 247(1); 75-78
18. Mak P, Leav I, Pursell B, ERbeta impedes prostate cancer EMT by destabilizing HIF-1alpha and inhibiting VEGF-mediated snail nuclear localization: Implications for Gleason grading: Cancer Cell, 2010; 17(4); 319-32
19. Qu LG, Wardan H, Davis ID, Circulating oestrogen receptor mutations and splice variants in advanced prostate cancer: BJU Int, 2019; 124(Suppl 1); 50-56
20. Faria M, Shepherd P, Pan Y, The estrogen receptor variants β2 and β5 induce stem cell characteristics and chemotherapy resistance in prostate cancer through activation of hypoxic signaling: Oncotarget, 2018; 9(91); 36273-88
21. Talesa VN, Ferri I, Bellezza G, Glyoxalase 2 is involved in human prostate cancer progression as part of a mechanism driven by PTEN/PI3K/AKT/mTOR signaling with involvement of PKM2 and ERα: Prostate, 2017; 77(2); 196-210
22. Shen Y, Cao J, Liang Z, Estrogen receptor α-NOTCH1 axis enhances basal stem-like cells and epithelial-mesenchymal transition phenotypes in prostate cancer: Cell Commun Signal, 2019; 17(1); 50
23. Semenas J, Wang T, Sajid Syed Khaja A, Targeted inhibition of ERα signaling and PIP5K1α/Akt pathways in castration-resistant prostate cancer: Mol Oncol, 2021; 15(4); 968-86
24. Kowalska K, Habrowska-Górczyńska DE, Urbanek KA, Estrogen receptor α is crucial in zearalenone-induced invasion and migration of prostate cancer cells: Toxins (Basel), 2018; 10(3); 98
25. Ratliff TL, Estrogen receptor alpha and imprinting of the neonatal mouse ventral prostate by estrogen: J Urol, 2005; 174(3); 1149
26. Liang Z, Cao J, Tian L, Aromatase-induced endogenous estrogen promotes tumour metastasis through estrogen receptor-α/matrix metalloproteinase 12 axis activation in castration-resistant prostate cancer: Cancer Lett, 2019; 467; 72-84
27. Fan Y, Song TR, Wei Q, Modulatory effect of aquaporin 5 on estrogen-induced epithelial-mesenchymal transition in prostate epithelial cells: Chin Med J, 2020; 134(4); 448-55
28. Xiao L, Luo Y, Tai R, Zhang N, Estrogen receptor β suppresses inflammation and the progression of prostate cancer: Mol Med Rep, 2019; 19(5); 3555-63
29. Grindstad T, Skjefstad K, Andersen S, Estrogen receptors α and β and aromatase as independent predictors for prostate cancer outcome: Sci Rep, 2016; 6; 33114
30. Migliaccio A, Castoria G, Di Domenico M, Steroid-induced androgen receptor-oestradiol receptor beta-Src complex triggers prostate cancer cell proliferation: EMBO J, 2000; 19(20); 5406-17
31. Rossi V, Di Zazzo E, Galasso G, Estrogens modulate somatostatin receptors expression and synergize with the somatostatin analog pasireotide in prostate cells: Front Pharmacol, 2019; 10; 28
32. Xiao L, Xiao M, Gao L, Xu W, Involvement of estrogen receptor β in androgen receptor-induced growth inhibition in prostate cancer PC-3 cells: Oncol Lett, 2017; 14(3); 2796-802
33. Shorning BY, Dass MS, Smalley MJ, Pearson HB, The PI3K-AKT-mTOR pathway and prostate cancer: At the crossroads of AR, MAPK, and WNT signaling: Int J Mol Sci, 2020; 21(12); 4507
34. Gao L, Zhang W, Zhang J, KIF15-mediated stabilization of AR and AR-V7 contributes to enzalutamide resistance in prostate cancer: Cancer Res, 2021; 81(4); 1026-39
35. Vanderschueren D, Vandenput L, Boonen S, Androgens and bone: Endocr Rev, 2004; 25(3); 389-425
36. Chaurasiya S, Widmann S, Botero C, Estrogen receptor β exerts tumor suppressive effects in prostate cancer through repression of androgen receptor activity: PLoS One, 2020; 15(5); e0226057
37. Kitraki E, Nalvarte I, Alavian-Ghavanini A, Rüegg J, Developmental exposure to bisphenol A alters expression and DNA methylation of Fkbp5, an important regulator of the stress response: Mol Cell Endocrinol, 2015; 417; 191-99
38. Wu WF, Maneix L, Insunza J, Estrogen receptor β, a regulator of androgen receptor signaling in the mouse ventral prostate: Proc Natl Acad Sci USA, 2017; 114(19); e3816-22
39. Yang CC, Lin LC, Lin YW, Higher nuclear EGFR expression is a better predictor of survival in rectal cancer patients following neoadjuvant chemoradiotherapy than cytoplasmic EGFR expression: Oncol Lett, 2019; 17(2); 1551-58
40. Alsahafi E, Begg K, Amelio I, Clinical update on head and neck cancer: molecular biology and ongoing challenges: Cell Death Dis, 2019; 10(8); 540
41. Wu WF, Wang L, Spetsieris N, Estrogen receptor β and treatment with a phytoestrogen are associated with inhibition of nuclear translocation of EGFR in the prostate: Proc Natl Acad Sci USA, 2021; 118(13); e2011269118
42. Miura Y, Tam T, Ido A, Cloning and characterization of an ATBF1 isoform that expresses in a neuronal differentiation-dependent manner: J Biol Chem, 1995; 270(45); 26840-48
43. Sun X, Xing C, Fu X, Additive effect of Zfhx3/Atbf1 and Pten deletion on mouse prostatic tumorigenesis: J Genet Genomics, 2015; 42(7); 373-82
44. Sun X, Fu X, Li J, Deletion of atbf1/zfhx3 in mouse prostate causes neoplastic lesions, likely by attenuation of membrane and secretory proteins and multiple signaling pathways: Neoplasia (New York, NY), 2014; 16(5); 377-89
45. Hu Q, Zhang B, Chen R, ZFHX3 is indispensable for ERβ to inhibit cell proliferation via MYC downregulation in prostate cancer cells: Oncogenesis, 2019; 8(4); 28
46. Dickmanns A, Monecke T, Ficner R, Structural basis of targeting the exportin CRM1 in cancer: Cells, 2015; 4(3); 538-68
47. Rice L, Lepler S, Pampo C, Siemann DW, Impact of the SRC inhibitor dasatinib on the metastatic phenotype of human prostate cancer cells: Clin Exp Metastasis, 2012; 29(2); 133-42
48. Souza DS, Lombardi APG, Vicente CM, Estrogen receptors localization and signaling pathways in DU-145 human prostate cancer cells: Mol Cell Endocrinol, 2019; 483; 11-23
49. Zhao Z, Yu H, Kong Q, Effect of ERβ-regulated ERK1/2 signaling on biological behaviors of prostate cancer cells: Am J Transl Res, 2017; 9(6); 2775-87
50. Silva RS, Lombardi APG, de Souza DS, Activation of estrogen receptor beta (ERβ) regulates the expression of N-cadherin, E-cadherin and β-catenin in androgen-independent prostate cancer cells: Int J Biochem Cell Biol, 2018; 96; 40-50
51. Lombardi APG, Vicente CM, Porto CS, Estrogen receptors promote migration, invasion and colony formation of the androgen-independent prostate cancer cells PC-3 through β-Catenin pathway: Front Endocrinol, 2020; 11; 184
52. Xiao L, Xiao M, Zou M, Xu W, Estrogen receptor β inhibits prostate cancer cell proliferation through downregulating TGF-β1/IGF-1 signaling: Int J Clin Exp Pathol, 2017; 10(8); 8569-76
Figures
 Figure 1. Structures of ERα, ERβ, and several of their major splice variant (ERβ2, ERβ3, ERβ4, ERβ5) proteins: N-terminal transactivation domain (NTD), DNA-binding domain (DBD), hinge region, C-terminal co-activator/cofactor interaction domain and ligand-binding domain (LBD), amino-terminal, and carboxy-terminal. (Created using Adobe Illustrator CS6, Version 16.0.0.682, Adobe Systems, Incorporated).
Figure 1. Structures of ERα, ERβ, and several of their major splice variant (ERβ2, ERβ3, ERβ4, ERβ5) proteins: N-terminal transactivation domain (NTD), DNA-binding domain (DBD), hinge region, C-terminal co-activator/cofactor interaction domain and ligand-binding domain (LBD), amino-terminal, and carboxy-terminal. (Created using Adobe Illustrator CS6, Version 16.0.0.682, Adobe Systems, Incorporated). Figure 2. In genomic action, estrogen regulates the transcription of target genes by binding to and activating inactive ER receptors in the cytoplasm or nucleus, and the activated estrogen receptor monomer forms a dimer with another activated estrogen receptor monomer, which in combination with other co-regulatory proteins (not labeled) regulates the transcription of target genes after binding to the estrogen response element (ERE) within the DNA. Non-genomic action: Estrogen induces the initiation of estrogen action by binding to receptors on the cell membrane, which in turn regulates downstream protein expression by acting as second messengers and transcription factors. (Created using Adobe Illustrator CS6, Version 16.0.0.682, Adobe Systems, Incorporated).
Figure 2. In genomic action, estrogen regulates the transcription of target genes by binding to and activating inactive ER receptors in the cytoplasm or nucleus, and the activated estrogen receptor monomer forms a dimer with another activated estrogen receptor monomer, which in combination with other co-regulatory proteins (not labeled) regulates the transcription of target genes after binding to the estrogen response element (ERE) within the DNA. Non-genomic action: Estrogen induces the initiation of estrogen action by binding to receptors on the cell membrane, which in turn regulates downstream protein expression by acting as second messengers and transcription factors. (Created using Adobe Illustrator CS6, Version 16.0.0.682, Adobe Systems, Incorporated). In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








