25 April 2022: Clinical Research
A Retrospective Study of 320 Patients Treated with Carotid Artery Stenting at 4 General Hospitals in Zhejiang Province to Establish a Risk Score for Cerebral Hypoperfusion Syndrome
Yuan Liu1B, Changyang Zhong1ABCEF*, Peng Wang2BC, Jianhong Yang3DF, Chenghua Xu4BF, Zuyong Zhang5AFGDOI: 10.12659/MSM.935697
Med Sci Monit 2022; 28:e935697
Abstract
BACKGROUND: Cerebral hypoperfusion syndrome (CHS) includes a spectrum of clinical symptoms, ranging from focal neurologic deficit to intracerebral hemorrhage. CHS was initially described as a complication of carotid endarterectomy but also occurs following carotid artery stenting. This retrospective study included 320 patients treated with carotid artery stenting at 4 general hospitals in Zhejiang Province between June 2019 and June 2021 and aimed to establish a risk score for CHS.
MATERIAL AND METHODS: Through retrospective case analysis, a risk model and scoring model for CHS were established and evaluated.
RESULTS: Poor integrity of the circle of Willis, preoperative cerebrovascular resistance, mean transit time, peak time at CTP, and preoperative cerebral circulation time were significant in the univariate analysis and were entered into the regression equation to establish the logistic and additive scoring model for predicting the risk of CHS after carotid stenting. The area under the receiver operating characteristic (ROC) curve of the logistic scoring system for the early warning risk of CHS after carotid stenting was 0.964 (95% confidence interval [CI] [0.931-0.996]), and the area under the ROC curve of the CHS early risk additive scoring model after carotid stenting was 0.968 (95% CI [0.936-1.000]), The Hosmer-Lemeshow test chi-square values were 0.037 (P=0.848) and 2.671 (P=0.102).
CONCLUSIONS: Two methods of scoring risk for CHS were developed from a retrospective analysis of 320 patients treated with carotid artery stenting at 4 general hospitals in Zhejian Province between June 2019 and June 2021.
Keywords: carotid stenosis, Early Warning Score, Risk Factors, Carotid Arteries, Cerebrovascular Circulation, Cerebrovascular Disorders, Endarterectomy, Carotid, Hospitals, General, Humans, Stents, Syndrome
Background
Carotid stenosis has become one of the most common causes of death and endangers the health of patients. Carotid stenting (CAS) is currently one of the main treatment methods for carotid artery stenosis owing to its minimal trauma and rapid recovery of neurological function [1]. However, after the diseased vascular stenosis is relieved, blood flow increases asymptotically (20–40% higher than the baseline) for several hours; however, some patients experience a prolonged state of high hyperemia (100–200% higher cerebral blood flow than at baseline), which peaks 3–4 days after the operation. Generally, patients return to a steady state after 6–7 days, which lasts for 1–2 weeks, although this can last for as long as 4 weeks [2]. When the brain tissue cannot tolerate the series of clinical symptoms and signs caused by the sudden increase in cerebral blood flow in the blood supply area of the blood vessels, this phenomenon is called cerebral hyperperfusion syndrome (CHS) [3]. Several studies have reported that the incidence of CHS after carotid artery stenting (CAS) fluctuates between 2.2% and 2.9% [4,5] and is a serious complication following carotid artery stenosis treatment. Once CHS occurs, the prognosis is often poor and results in serious disability or death. Therefore, early prevention of CHS is crucial. At present, research on the prevention of CHS has mainly focused on treatment management, cause analysis, and influencing factor analysis [6,7], and few reports have comprehensively explored the risk factors that affect the occurrence of CHS, internal relationships among the influencing factors, and quantification of the risk factor level, targeted early warning model, and scoring model. The establishment of a CHS early risk model after carotid stenting will allow early clinical identification of groups at high risk of CHS, timely detection, and early intervention, which is of clinical significance for reducing medical risk and the rational use of medical resources. Therefore, this retrospective study included 320 patients treated with carotid artery stenting at 4 general hospitals in Zhejian Province between June 2019 and June 2021 and aimed to establish a risk score for CHS.
Material and Methods
DEMOGRAPHIC INFORMATION:
The following demographic characteristics and history information were obtained: sex, age, smoking, drinking, history of hypertension, history of diabetes, and history of stroke.
DISEASE AND OPERATION:
Anesthesia mode, degree of preoperative carotid stenosis, occlusion of the carotid artery on the opposite side of the stenosis, integrity of the circle of Willis, collateral circulation, and whether carotid stenting was performed within 2 weeks were recorded.
OTHER RELEVANT VARIABLES:
We also gathered information on the following: 1) cerebrovascular resistance; 2) preoperative asymmetry index; 3) pulsatility index (PI); 4) mean transit time difference (dMTT); 5) blood flow difference between the ipsilateral and contralateral middle cerebral artery (MCA); 6) postoperative and preoperative MCA; 7) postoperative-preoperative blood pressure ratio; 8) time to peak (TTP) during CTP (computed tomography perfusion imaging) inspection of the rTTP index (affected side TTP-contralateral TTP/contralateral TTP); 9) rCBV (central blood volume) index; 10) resting mean transit time (MTT); and 11) cerebral circulation time (CCT).
IMAGING EXAMINATION METHODS 1.2.1 MSCT (PERFUSION SCAN):
All patients underwent whole-brain perfusion CT examination within 1 week before the treatment of cerebral artery stenosis. A Siemens 64-slice spiral CT was used for spiral scan, and the whole-brain CT scan was performed from the base of the skull to the top of the skull (thickness: 5 mm, 120 kV, 36 mAs). Then, 50 mL of non-ionic iodine contrast agent (iodophenol, 350 mL/dL) was injected through the cubital vein with an 18G injection needle, and 40 mL of 0.9 mL% normal saline was added. The injection rate was 6.0 mL/s, and scanning was performed 5 s later (110 kV, 80 mAs). Data were collected in 33.99 s and 846 imaging images with a thickness of 5.0 mm were generated.
IMAGE AND DATA PROCESSING:
Imaging images were observed and analyzed by 2 experienced imaging physicians, and a unified opinion was reached on the abnormal perfusion area. Siemens MMWP workstation was used for calculation. The middle cerebral artery was the input artery and the internal jugular vein was the output vein. The location and size of possible lesions were determined manually, and vascular calcification and old infarcted tissue were avoided as far as possible. Quantitative function graphs and perfusion parameters such as cerebral blood flow (CBF), cerebral blood volume (CBV), mean peak time (MTT) and peak time (TTP) were automatically generated by computer. Then, the mirror image method was used to place the region of interest on the opposite side, and the circular region of interest on the corresponding part of the opposite side was placed, and the average value was taken. The relative parameter values of lesions (rCBF, rCBV, rMTT, rTTP) were obtained by comparing the parameters of lesions with those of normal lesions.
STATISTICAL ANALYSIS:
Continuous variables with a normal distribution, continuous variables with a non-normal distribution, and classified variables are expressed as means±standard deviations (x±s), medians and interquartile spacing, and percentages, respectively. The normal distribution of variables was examined using Kolmogorov-Smirnov tests. The chi-square and Fisher’s tests were used to compare categoric variables. The
Results
BASIC PATIENT INFORMATION:
A total of 320 patients who underwent carotid stenting were included in this study, which included 68 patients with CHS (21.25%) and 252 patients without CHS (78.75%), which was in agreement with the sample inclusion method (1: 4). There were 279 men (87.19%) and 41 women (12.81%). The average age was 68.92±7.97 years. There were 272 cases (85%) with a history of hypertension, 131 cases (40.94%) with a history of stroke, and 122 cases (38.13%) with a history of diabetes. There were 225 cases in the test set and 95 cases in the validation set. The incidence and clinical outcomes of patients with CHS after carotid stenting are shown in Table 1.
COMPARISON OF DEMOGRAPHIC CHARACTERISTICS AND HISTORY BETWEEN THE 2 GROUPS: There were significant differences in age, smoking, history of diabetes, and history of stroke both before and after CAS between the CHS and non-CHS groups (P<0.05; Table 2).
COMPARISON OF DISEASE AND OPERATIONS BETWEEN THE 2 GROUPS: There were significant differences in the degree of carotid stenosis, integrity of the circle of Willis, and collateral circulation between patients with and without CHS after carotid stenting (P<0.05; Table 3).
The circle of Willis can be divided into 4 types: complete (type I, 24.5%), anterior complete (type II, 57.2%), posterior complete (type III, 3.2%), and anterior and posterior incomplete (type IV, 15.2%). According to the literature, type I was defined as better integrity of the circle of Willis, type II and III were defined good integrity of the circle of Willis, and type IV was defined poor integrity of the circle of Willis [9].
Collateral circulation is the commonly used ASITN/SIR in the grading system of collateral circulation evaluation standard: Level 0 is without collateral blood supply to the ischemia area, Level 1 is slow collateral blood supply to the periphery of ischemic area, Level 2 is fast collateral blood supply to the periphery of ischemic area, Level 3 is slow collateral blood supply to the whole ischemic area, and Level 4 is fast collateral blood supply to the whole ischemic area [10].
COMPARISON OF RELEVANT INDICES BETWEEN THE 2 GROUPS: Preoperative CVR (cerebral vascular resistance), preoperative asymmetry index, PI, dMTT, ipsilateral and contralateral MCA blood flow difference, V (velocity) postoperative/V (velocity) preoperative (TCD [transcranial Doppler]) detects the blood flow velocity ratio of the MCA 30 min before and after the operation), preoperative and postoperative blood pressure ratio, rTTP index, rCBV index, mean transit time, and resting MTT, and preoperative CCT between CCT and ΔCCT were significant differences (P<0.05; Table 4).
LOGISTIC REGRESSION MODEL OF RISK FACTORS FOR CHS AFTER CAROTID STENTING: Using the occurrence of CHS after carotid stenting as the dependent variable and the factors that were significantly different in the univariate analysis as independent variables for the logistic regression analysis, results showed that poor integrity of the circle of Willis, preoperative CVR, MTT, peak time at CTP (rTTP), and preoperative CCT were included in the regression equation (P<0.05; Table 5).
According to the results of multiple stepwise logistic regression analysis, the regression equation was constructed as follows:
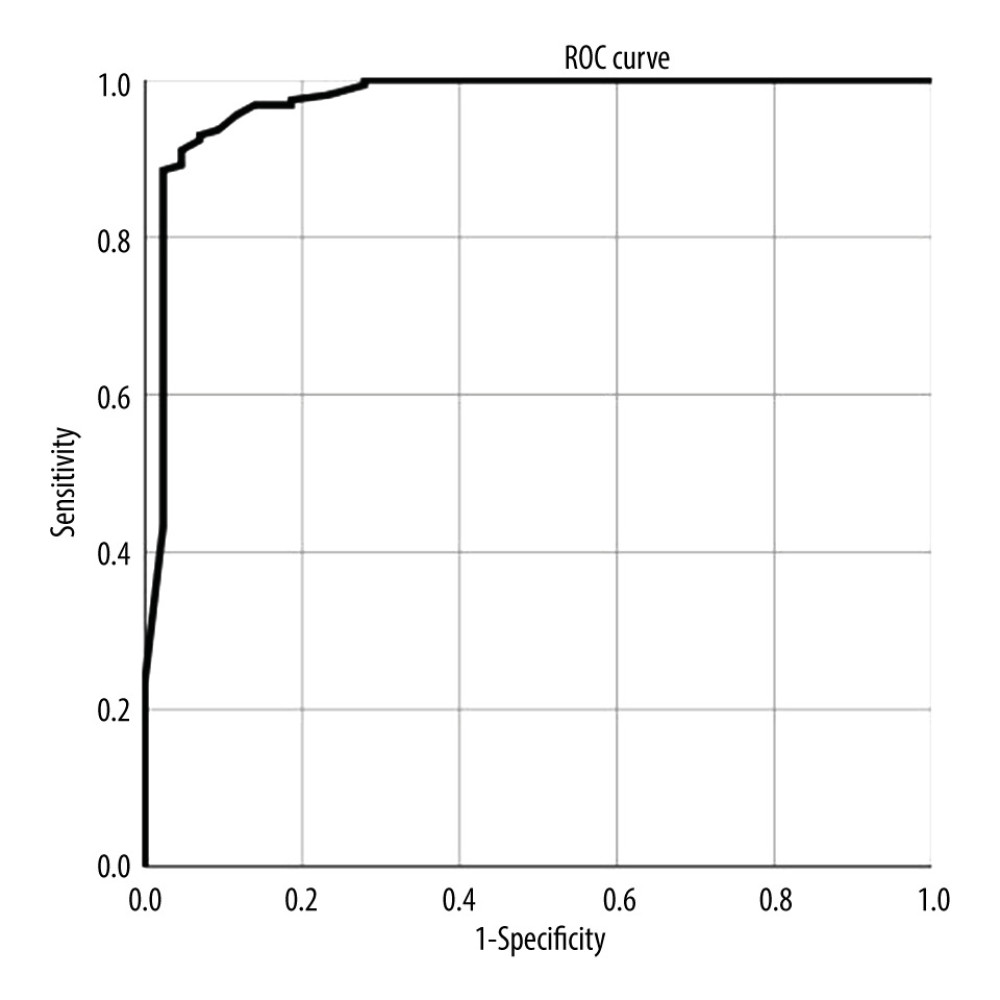
When different risk factors exist in each case, this equation allows the calculation of the probability of the occurrence of CHS after carotid stenting. Taking the probability of occurrence as the test variable and the occurrence of CHS as the state variable, we drew an ROC curve, as shown in Figure 1.
The area under the ROC curve was 0.973 (95% confidence interval [CI] [0.942–1.000]), which indicated good discrimination ability. The chi-square value of the Hosmer-Lemeshow test was 6.210 (
ESTABLISHMENT OF A RISK EARLY WARNING SCORING SYSTEM FOR CHS AFTER CAROTID STENTING:
According to the regression coefficient B value (integer) and/or value (integer) of each variable that entered the regression equation, the logistic and additional scoring systems for predicting the risk of CHS after carotid stenting were established, respectively, and the total scores were 0–11 and 5–61, respectively (Table 6).
EVALUATION OF CHS RISK EARLY WARNING SCORING SYSTEM AFTER CAROTID STENTING:
The risk of CHS after carotid stenting in all patients was divided into 3 levels: low risk (logistic score ≤4 or additive score ≤25); medium risk (logistic score 5–6 or additive score 26–50); high risk (logistic score ≥7 or additive score ≥51). The low-, medium-, and high-risk probabilities of the occurrence of CHS were 2.7%, 44.4%, and 96.9%, respectively. The low-, medium-, and high-risk rates of CHS were 2.6%, 69.6%, and 95.8%, respectively (Tables 7, 8).
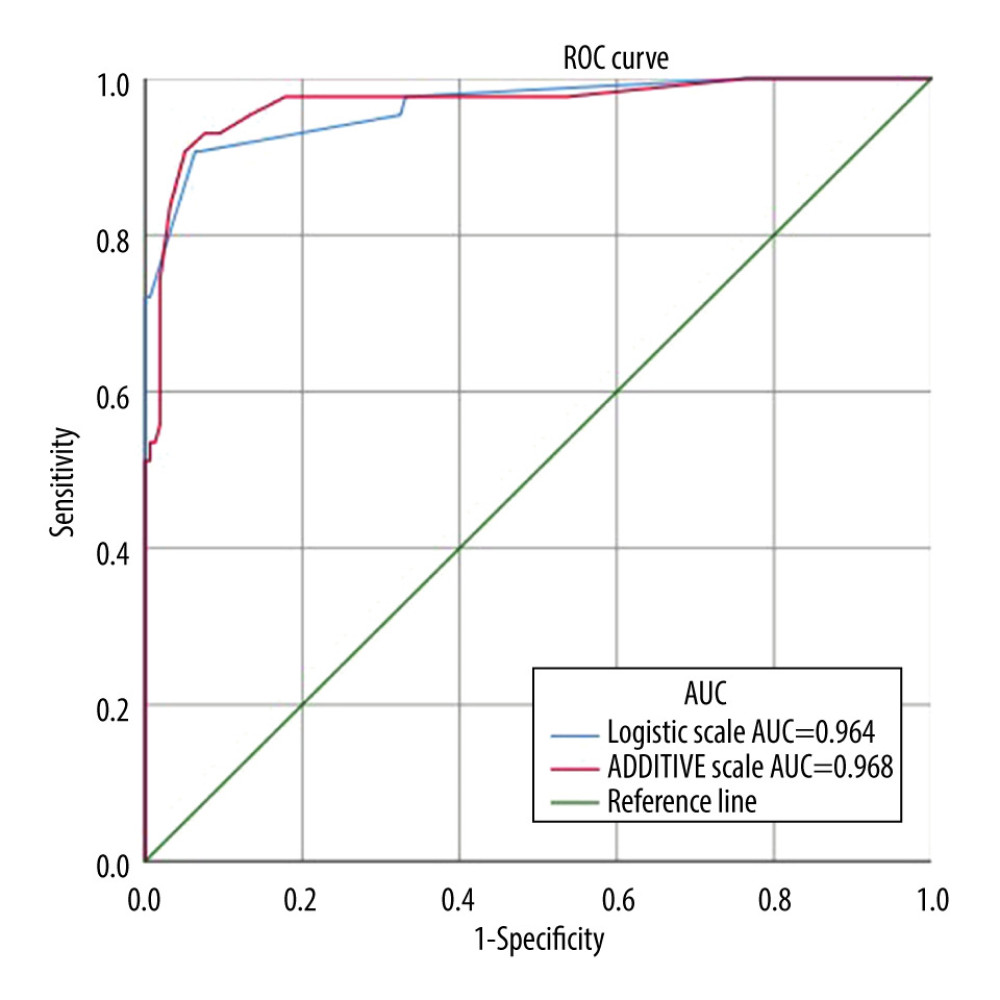
The validation set results are provided in Figure 2. The area under the ROC curve of the CHS early warning risk logistic scoring system after carotid stenting was 0.964 (95% CI [0.931–0.996]), and the area under the ROC curve of the CHS early warning risk additive scoring system after carotid stenting was 0.968 (95% CI [0.936–1.000]). The chi-square values of the Hosmer-Lemeshow test were 0.037 (P=0.848) and 2.671 (P=0.102), respectively, which indicated that the actual probability was in good agreement with the predicted probability of the simulation fit and had good correction ability.
Discussion
In this study, the mortality rate of CHS was 8.82%, and the disability rate was 38.24%. Therefore, there is an urgent need to develop methods to reduce the incidence rate and mortality of CHS after CAS. Our results showed that poor integrity of the circle of Willis, preoperative CVR, MTT, peak time at CTP (rTTP), and preoperative CCT were 5 important variables of the early warning scoring system and were included in the regression equation.
Poor integrity of the circle of Willis was related to the occurrence of CHS. We found that the risk of developing CHS in patients with poor circle of Willis integrity was 5 times higher than that in patients with a complete circle of Willis. Moreover, poor long-term collateral circulation compensation led to long-term hypoperfusion of the cerebral hemisphere on the diseased side. Once vascular stenosis is relieved, cerebral blood flow cannot be diverted through the compensated vessels, which results in poor cerebrovascular reactivity and a greater likelihood of the occurrence of CHS.
Preoperative CVR was also related to the occurrence of CHS. Our study showed that the risk of developing CHS in patients with abnormal preoperative CVR was 6 times higher than that in patients with normal preoperative CVR. CVR reflects the ability to maintain stable cerebral blood flow. Cerebral blood flow is regulated via the compensatory expansion or contraction of arterioles and capillaries and the opening of the cerebrovascular collateral circulation. Abnormal preoperative CVR indicates a decrease in the patient’s self-regulation ability, which leads to difficulties in adapting to the sudden recovery of cerebral blood flow over a short period and, ultimately, CHS.
MTT was related to the occurrence of CHS. We revealed that the risk of developing CHS in patients with an MTT >4.85 s was 8 times higher than that in patients with an MTT ≤4.85 s. This finding is consistent with that of Yoshie et al [11]. They found that MTT is an independent risk factor for CHS and recommended that MTT should be monitored in the clinic; however, further reasons for this recommendation were not offered. We found that the time required for the contrast agent to pass through the capillaries (ie, MTT) was prolonged, which indicated that the time required for the blood flow of the original blood supply artery to reach the tissue was longer. This may be because the degree of carotid artery stenosis was aggravated, which increased the medial cerebral branches, and the often-small collateral vessels gradually formed a capillary bed, resulting in poor reactivity of the cerebral vessels. In addition, the automatic regulation ability may have been insufficient. When the capillary bed perfusion pressure increases suddenly, the blood-brain barrier is destroyed, and CHS occurs.
The time to peak (rTTP) of CTP was related to the occurrence of CHS. The risk of developing CHS in patients with a CTP peak time (rTTP) >0.22 s after carotid stenting was 31 times higher than that in patients with an rTTP ≤0.22s. This is consistent with the findings of Chang et al [12], who found that the rTTP index ([affected rTTP-contralateral rTTP]/contralateral rTTP) >0.22 independently predicted CHS in 54 patients with severe internal carotid artery stenosis who were treated by CAS. This study only found that rTTP is related to CHS through case studies. Therefore, we sought to analyze the reasons. TTP refers to the time from the beginning of the contrast agent injection to the peak concentration of the contrast agent in a specific region of the brain tissue; thus, it reflects the blood vessels to the study area and the perfusion characteristics of the brain tissue itself, and the rTTP index reflects the perfusion difference index of the capillaries and brain tissue on both sides. The peak time of the cerebral blood flow on the affected side was greater than that on the opposite side, which indicated that the reserve capacity of cerebral blood flow decreased, and the decline in the reserve capacity of cerebral blood flow was important for the pathogenesis of CHS. Therefore, we revealed that the peak time of CTP (rTTP) was the most sensitive risk factor for predicting the occurrence of CHS.
Preoperative CCT was also related to the occurrence of CHS. The risk of CHS after carotid stent implantation in patients with a preoperative CCT >7.1 s was 11 times higher than that in patients with a preoperative CCT ≤7.1 s. Lin et al [13] studied 49 patients with severe unilateral carotid stenosis and found that a prolonged preoperative CCT led to postoperative CHS. The optimal time for prolonged preoperative CCT to predict CHS was 7.1 s. Yamauchi et al [14] measured the CCT of 33 patients with CAS or angioplasty using color-coded DSA (digital subtraction angiography) and similarly found that prolonged CCT before treatment increased the likelihood of CHS. However, the above studies found that CCT was related to CHS through case studies only, and no additional research has been conducted. Our finding that CCT is an important risk factor for the occurrence of CHS may be because preoperative CCT is measured from the time from the arterial phase to the venous phase. The prolonged CCT time indicates that the higher the degree of stenosis, the greater the increase in collateral vessels and extension of the blood flow path, which reflects the decline in brain structure reserve and cerebral blood flow reserve capacity.
Based on the retrospective collection of clinical data and the single-factor and logistic regression analyses of the data, we established and verified a logistic risk model (total score 0–11) and an additional risk scoring model (total score 5–61) for the occurrence of CHS after carotid stenting. Both early risk models had good prediction efficiency and discrimination ability. According to the different assignments of the 2 early risk models, we determined 3 risk levels: low, medium, and high risk. These early risk models and gradings would be valuable for clinical practitioners to predict and implement targeted measures in advance. Firstly, targeted preventive interventions can be administered to patients according to the risk level obtained from the score. Secondly, the application of the risk scoring model of CHS after carotid stenting to patient evaluations will help patients and their families to correctly understand the risk of the operation, improve their preparation, reduce psychological expectations, and reduce the contradiction between doctors and patients.
Compared with previous studies, our study has 2 improvements. The first is result innovation.
In this study, the risk warning model and risk warning scoring system for CHS after carotid stenting were established, which further supplemented and improved the previous research results on risk factors of CHS after carotid stenting. The second is methodological innovation. On the basis of univariate analysis and multiple regression analysis, this study further assigned scores to each factor according to the odds ratio (OR) value of each independent risk factor and combined with the specific clinical situation, and obtained the area under receiver operating characteristic (ROC) curve of the scoring warning system to evaluate its discriminant ability. The Hosmer-Lemeshow goodness-of-fit test was used to evaluate its correction ability, but previously, domestic and foreign scholars only carried out single-factor analysis and multi-factor analysis.
Finally, there are 2 limitations/deficiencies in this study. First of all, due to limited time, we performed a retrospective study, which entails research difficulties with data integrity and information accessibility. Although this difficulty was finally overcome and enough effective samples were collected in 4 tertiary general hospitals, it also suggested that prospective studies could be carried out in the future to further explore the independent risk factors of hyperperfusion syndrome after carotid stenting. Secondly, this study developed a high-perfusion syndrome after 2 carotid stent early warning assessments. Although it was verified in 95 patients, and has high discriminant ability and correction ability, larger studies are needed to further explore the utility of the early warning and assessment system.
Conclusions
Two methods of scoring risk for CHS were developed from a retrospective analysis of 320 patients treated with carotid artery stenting at 4 general hospitals in Zhejian Province between June 2019 and June 2021.
Tables
Table 1. Incidence and clinical outcomes of CHS after carotid stenting.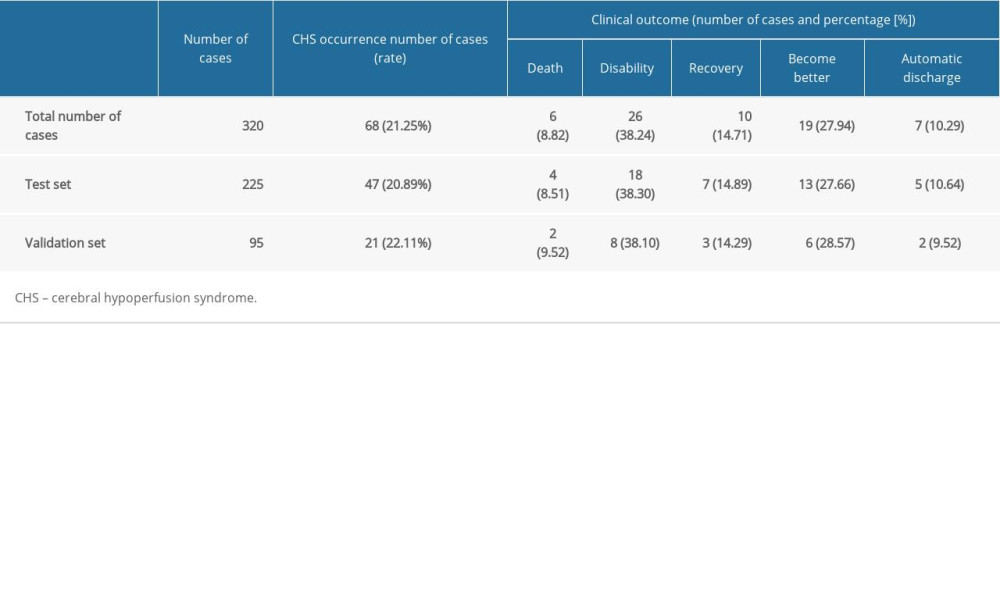 Table 2. Comparison of demographic characteristics and history between the 2 groups.
Table 2. Comparison of demographic characteristics and history between the 2 groups.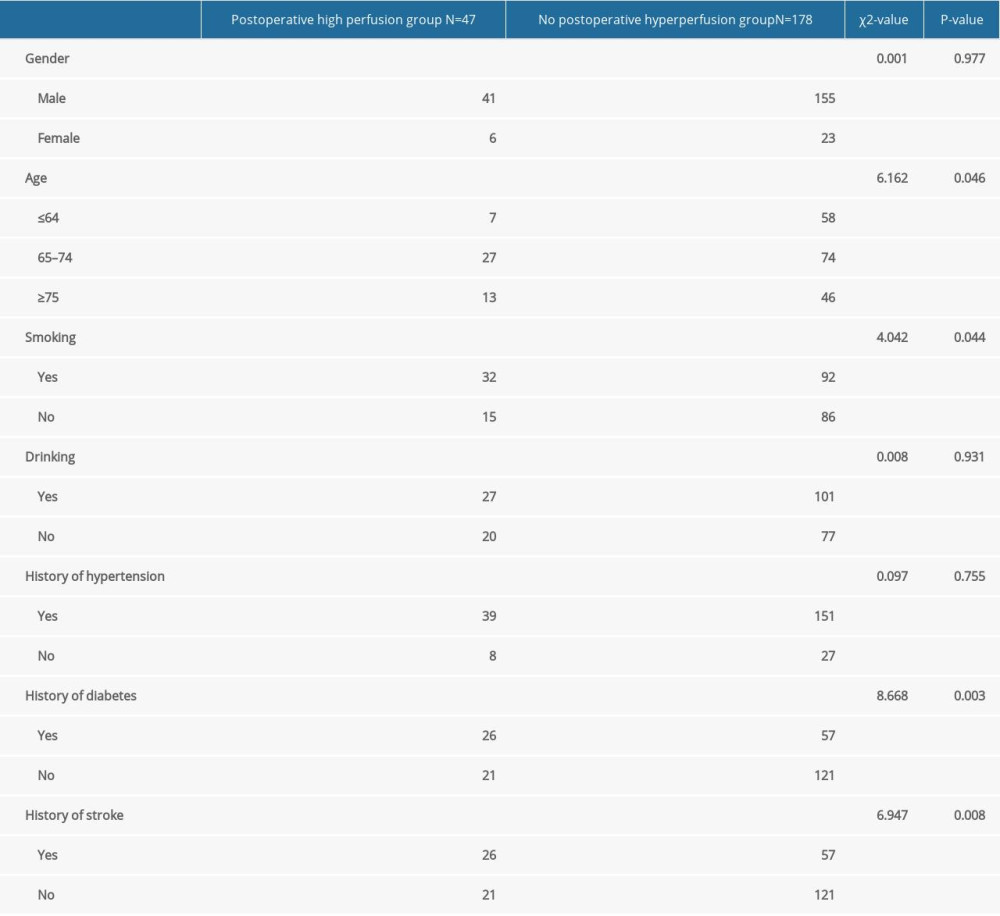 Table 3. Comparison of disease and operations between the 2 groups.
Table 3. Comparison of disease and operations between the 2 groups.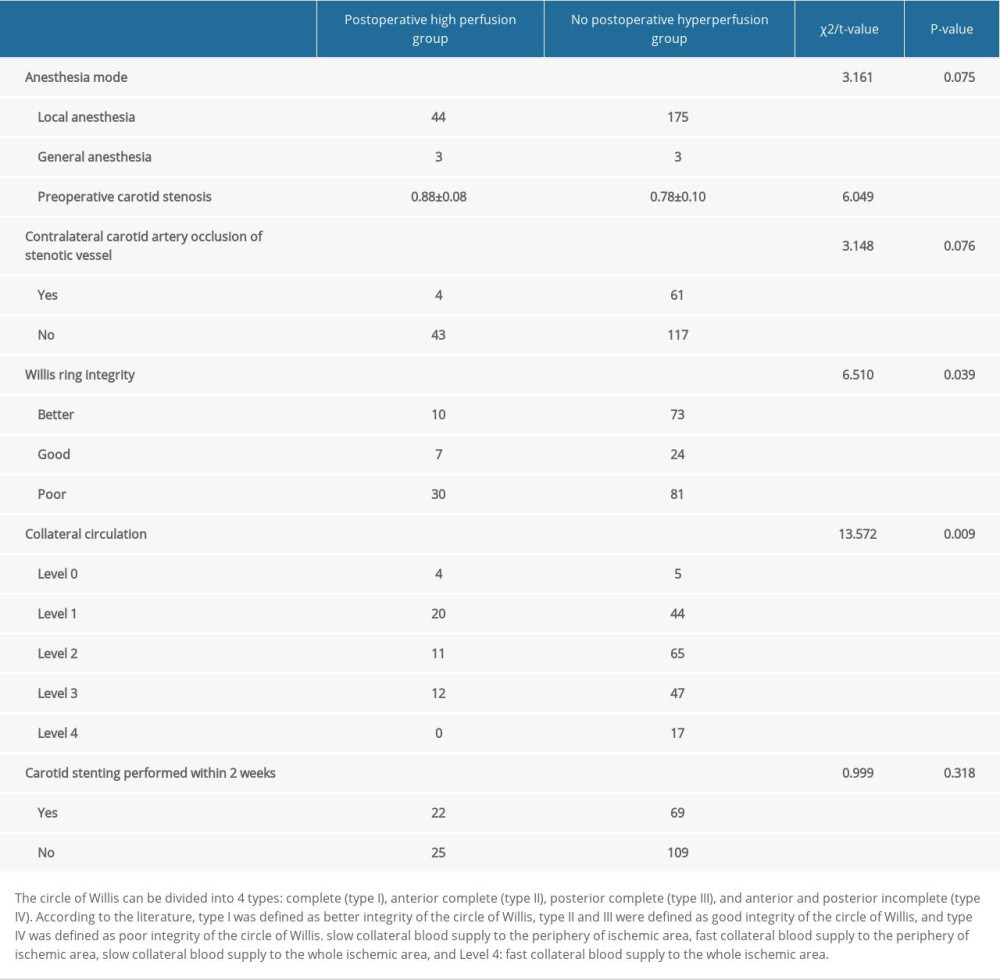 Table 4. Comparison of relevant indices between the 2 groups.
Table 4. Comparison of relevant indices between the 2 groups.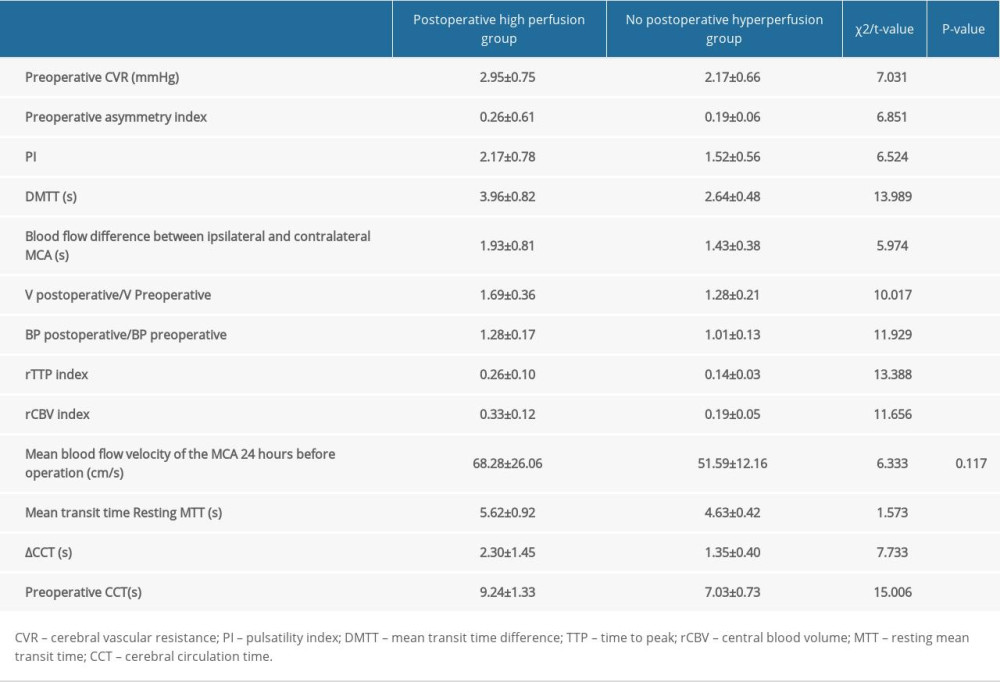 Table 5. Logistic regression analysis results for the influencing factors of CHS after carotid stenting.
Table 5. Logistic regression analysis results for the influencing factors of CHS after carotid stenting.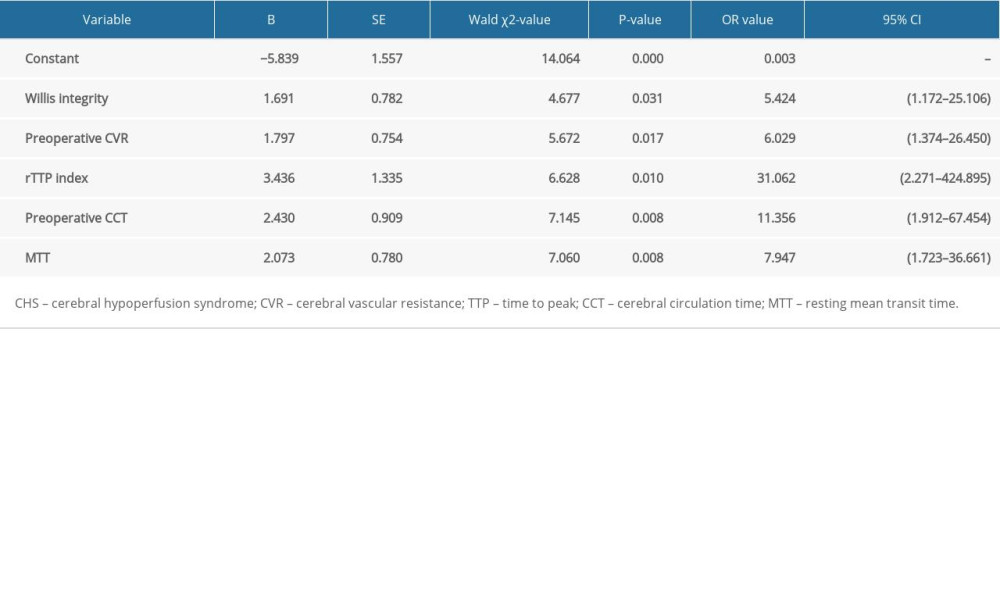 Table 6. Scoring system for the risk of CHS after carotid stenting.
Table 6. Scoring system for the risk of CHS after carotid stenting.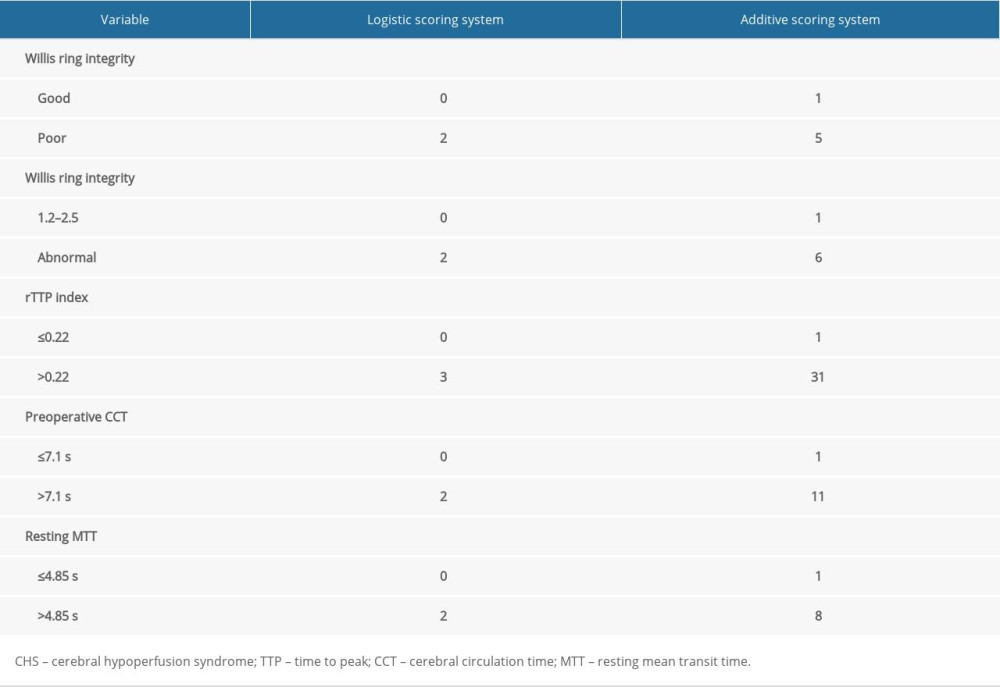 Table 7. Logistic score and incidence of CHS after carotid stenting.
Table 7. Logistic score and incidence of CHS after carotid stenting.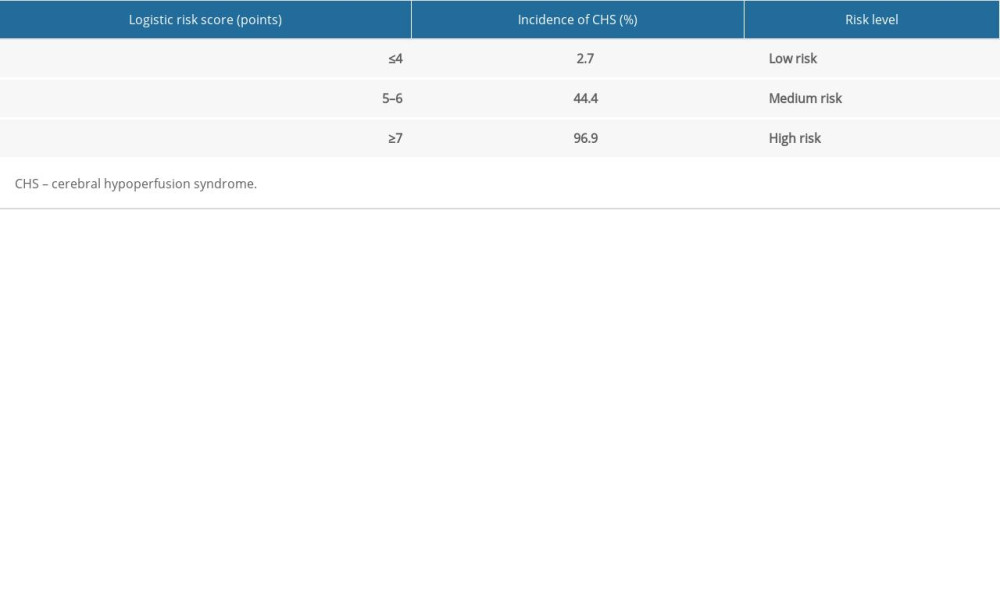 Table 8. CHS early warning risk additional score and incidence of CHS after carotid stenting.
Table 8. CHS early warning risk additional score and incidence of CHS after carotid stenting.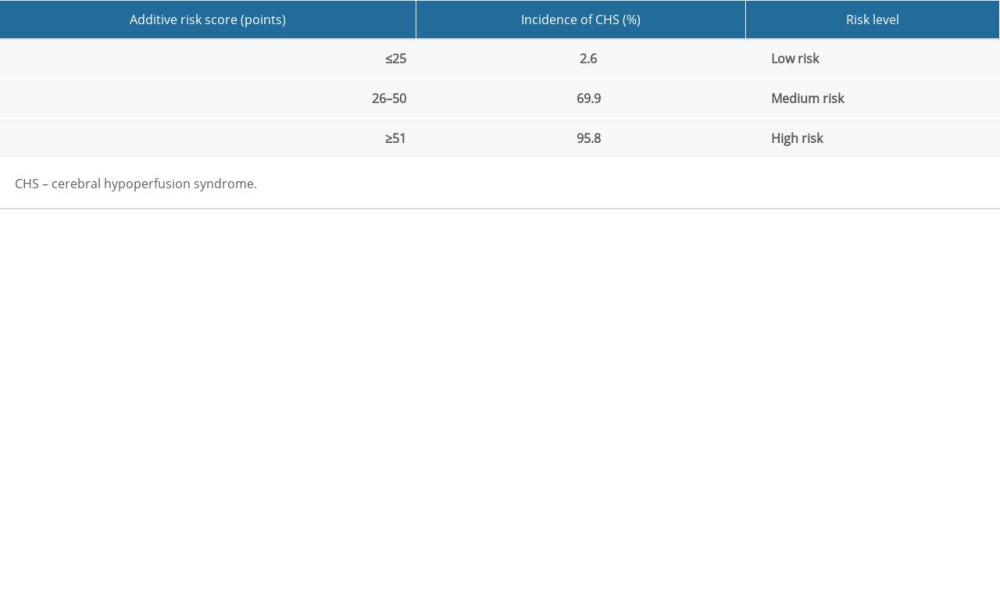
References
1. Bae C, Szuchmacher M, Chang JB, Comparative review of the treatment methodologies of carotid stenosis: Int J Angiol, 2015; 24(3); 215-22
2. Anand S, Sahoo AK, Cerebral hyperperfusion syndrome after carotid revascularization: A brief review: J Neuroanaesthesiol Crit Care, 2019; 6(3); 292-98
3. Okamura A, Nakaoka M, Ohbayashi N, Intraoperative idiopathic subarachnoid hemorrhage during carotid artery stenting: A case report and literature review: Interv Neuroradiol, 2015; 21(5); 592-97
4. Galyfos G, Sianou A, Filis K, Cerebral hyperperfusion syndrome and intracranial hemorrhage after carotid endarterectomy or carotid stenting: A meta-analysis: J Neurol Sci, 2017; 381; 74-82
5. González García A, Moniche F, Escudero-Martínez I, Clinical predictors of hyperperfusion syndrome following carotid stenting: Results from a national prospective multicenter study: JACC Cardiovasc Interv, 2019; 12(9); 873-82
6. Murai S, Sugiu K, Hishikawa T, Safety and efficacy of staged angioplasty for patients at risk of hyperperfusion syndrome: A single-center retrospective study: Neuroradiology, 2020; 62(4); 503-10
7. Dongpo Y, Zhongrong M, Clinical research status and progress of cerebral hyperperfusion syndrome after carotid stent implantation: Chinese J Cerebrovasc Dis, 2020; 14(03); 129-32
8. Ates ER, Babb JD, Casey DE, Clinical expert consensus document on carotid stenting: Vasc Med, 2007; 12(1); 35-83
9. Liebeskind DS, Collateral circulation: Stroke, 2003; 34(9); 2279-84
10. Hofrneijer J, Klijn CJM, Kappelle LJ, Collateral ckculation via the ophthalmic artery or leptomeningeal vessels is associated with impaired cerebral vasore. Activity in patients with symptomatic carotid artery occlusion: Cerebrovasc Dis, 2002; 14(1); 22-26
11. Yoshie T, Ueda T, Takada T, Prediction of cerebral hyperperfusion syndrome after carotid artery stenting by CT perfusion imaging with acetazolamide challenge: Neuroradiology, 2016; 58(3); 253-59
12. Chang CH, Chang TY, Chang YJ, The role of perfusion computed tomography in the prediction of cerebral hyperperfusion syndrome: PLoS One, 2011; 6(5); e19886
13. Lin CJ, Chang FC, Tsai FY, Stenotic transverse sinus predisposes to poststenting hyperperfusion syndrome as evidenced by quantitative analysis of peritherapeutic cerebral circulation time: Am J Neuroradiol, 2014; 35(6); 1132-36
14. Yamauchi K, Enomoto Y, Otani K, Prediction of hyperperfusion phenomenon after carotid artery stenting and carotid angioplasty using quantitative DSA with cerebral circulation time imaging: J Neurointerv Surg, 2018; 10(6); 576-79
Figures
Tables
 Table 1. Incidence and clinical outcomes of CHS after carotid stenting.
Table 1. Incidence and clinical outcomes of CHS after carotid stenting. Table 2. Comparison of demographic characteristics and history between the 2 groups.
Table 2. Comparison of demographic characteristics and history between the 2 groups. Table 3. Comparison of disease and operations between the 2 groups.
Table 3. Comparison of disease and operations between the 2 groups. Table 4. Comparison of relevant indices between the 2 groups.
Table 4. Comparison of relevant indices between the 2 groups. Table 5. Logistic regression analysis results for the influencing factors of CHS after carotid stenting.
Table 5. Logistic regression analysis results for the influencing factors of CHS after carotid stenting. Table 6. Scoring system for the risk of CHS after carotid stenting.
Table 6. Scoring system for the risk of CHS after carotid stenting. Table 7. Logistic score and incidence of CHS after carotid stenting.
Table 7. Logistic score and incidence of CHS after carotid stenting. Table 8. CHS early warning risk additional score and incidence of CHS after carotid stenting.
Table 8. CHS early warning risk additional score and incidence of CHS after carotid stenting. Table 1. Incidence and clinical outcomes of CHS after carotid stenting.
Table 1. Incidence and clinical outcomes of CHS after carotid stenting. Table 2. Comparison of demographic characteristics and history between the 2 groups.
Table 2. Comparison of demographic characteristics and history between the 2 groups. Table 3. Comparison of disease and operations between the 2 groups.
Table 3. Comparison of disease and operations between the 2 groups. Table 4. Comparison of relevant indices between the 2 groups.
Table 4. Comparison of relevant indices between the 2 groups. Table 5. Logistic regression analysis results for the influencing factors of CHS after carotid stenting.
Table 5. Logistic regression analysis results for the influencing factors of CHS after carotid stenting. Table 6. Scoring system for the risk of CHS after carotid stenting.
Table 6. Scoring system for the risk of CHS after carotid stenting. Table 7. Logistic score and incidence of CHS after carotid stenting.
Table 7. Logistic score and incidence of CHS after carotid stenting. Table 8. CHS early warning risk additional score and incidence of CHS after carotid stenting.
Table 8. CHS early warning risk additional score and incidence of CHS after carotid stenting. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387