22 June 2022: Review Articles
A Review of the Role of Transthoracic and Transesophageal Echocardiography, Computed Tomography, and Magnetic Resonance Imaging in Cardioembolic Stroke
Sergiu Florin ArnautuDOI: 10.12659/MSM.936365
Med Sci Monit 2022; 28:e936365
Abstract
ABSTRACT: Stroke is a major source of morbidity and mortality worldwide, accounting for the second largest cause of mortality and the third greatest cause of disability. Stroke is frequently preceded by a transient ischemic attack (TIA). The etiologies of 20-30% of ischemic strokes are unknown, and thus are termed “cryptogenic strokes”. About 25% of ischemic strokes are cardioembolic. Strokes occur at a rate of around 2% per year in individuals with heart failure with reduced ejection fraction (HFrEF), with a strong correlation between stroke risk and the degree of ventricular impairment. Furthermore, stroke risk is augmented in the absence of anticoagulation therapy. Cardioembolic strokes, when treated inadequately, have a greater predilection for recurrences than atherothrombotic strokes, both early and late in life. The role of a patent foramen ovale in strokes, specifically in “cryptogenic strokes”, is a matter of concern that deserves due attention. The use of tissue-engineered heart valves and aspirin for minimizing the risk of stroke is recommended. Transthoracic echocardiography (TTE) is advantageous for assessing heart function in the acute phase of ischemic stroke. Transesophageal echocardiography (TEE) is considered the criterion standard procedure for detecting LAA thrombi. Computed tomography (CT) scans are good imaging modalities for identifying and excluding bleeding. Magnetic resonance imaging (MRI) images are by far the most effective imaging technique available for assessing the brain parenchymal state. We conducted a thorough review of the literature on the use of imaging modalities, highlighting the important contribution of TTE, TEE, CT, and MRI in the evaluation of cardioembolic stroke.
Keywords: Atrial Fibrillation, embolic stroke, Echocardiography, Transesophageal, Heart Failure, Humans, Magnetic Resonance Imaging, Stroke, Stroke Volume, Tomography, X-Ray Computed
Background
Stroke is a major source of morbidity and mortality worldwide, accounting for the second largest cause of death and the third greatest cause of disability-adjusted life years (DALYs), as shown in the World Health Organization (WHO) Report on Mortality and Global Health estimates [1]. Frequently, it is preceded by transient ischemic attacks (TIAs). Since the etiologies of 20–30% of ischemic strokes are unknown, they are classified as cryptogenic [2]. However, using novel diagnostic procedures, the cause of 30% of the cryptogenic strokes can be attributed to the cardioembolic events of silent atrial fibrillation (AF) [3]. The most common sustained cardiac arrhythmia is AF, with a prevalence of 2–4% in adults and 5–15% in the elderly [3]. The yearly absolute risk of occurrence of stroke in AF is 3–4% [4–7]. This risk is amplified in patients with asymptomatic and paroxysmal atrial fibrillation (PAF), as these patients are vulnerable due to inadequate use of anticoagulants. Around 15% of TIAs can be attributed to silent PAF [2]. Although Holter monitoring is commonly used for diagnosing PAF, its efficiency is limited by the sporadic nature of PAF and the inability of patients to identify it. Long-term monitoring is only feasible by the use of insertable loop recorders, but their invasive implantation is expensive and thus cannot be achieved in most stroke patients [8]. Situations become even more complex when cardioembolic stroke is distinguished from extracranial and intracranial atherosclerotic strokes, which can be caused by a variety of diverse processes, including in situ thromboembolic obstruction, artery-to-artery embolism, branch occlusion, and circulatory insufficiency [9].
About 25% of ischemic strokes are cardioembolic. Strokes caused by cardioembolic thrombosis are typically more severe than those caused by atherothrombotic thrombosis. Furthermore, they have a greater proclivity for relapses, both earlier and late in life. Therefore, we conducted this review to elucidate the role of transthoracic echocardiography (TTE), transesophageal echocardiography (TEE), magnetic resonance imaging (MRI), and computed tomography (CT) in diagnosing cardioembolic stroke. The following cardioembolic stroke risk factors are among the most common ones.
Cardioembolic Stroke Risk Factors
Atrial Factors
Clinical Characteristics of Cardioembolic Stroke
Cardioembolic stroke is characterized by the following characteristics: sudden deterioration of mental state, presence of neurological impairments, and modification of level of consciousness. Cardiac abnormalities can manifest themselves in the following ways: In the recent past, the patient may have had an arrhythmia (atrial fibrillation), a cardiac murmur, signs of congestive heart failure, acute myocardial infarction, or acute infective endocarditis.
Cardiovascular Assessment Methods
Electrophysiologic Evaluation
TWELVE-LEAD ELECTROCARDIOGRAM:
It is only effective for detecting continuing arrhythmias. Additionally, it can offer important information on the myocardial state of the ventricles (evidence of ventricular hypertrophy) and previous cardiac ischemia events. However, transitory arrhythmias can be overlooked, most notably paroxysmal atrial fibrillation.
HOLTER MONITORING:
It is now routinely used on all suspected cardioembolic stroke patients. While it is fundamentally identical to an electrocardiogram, it has the limitation of examining the conduction system for just 24 h.
IMPLANTABLE LOOP RECORDERS (ILRS):
They can maintain a 3-year record of activities. As a result, their clinical value continues to grow, and their use has aided identification of numerous cases of “missing” atrial fibrillation.
Imaging Methods for Assessment of Structural and Functional Cardiac Abnormalities
Risk Evaluation and Treatment
THROMBUS FORMATION:
LA enlargement is a strong predictor of newly diagnosed atrial fibrillation and embolic events in individuals with atrial fibrillation. Echocardiographic abnormalities, such as LA indexed volume, are related to the diagnosis of AF in patients with cardiac embolic stroke of unknown cause [22,45,46].
The pulsed-wave Doppler transmitral inflow (TMF) pattern is another previously described predictor of AF. TMF is composed of 2 components: early diastole filling induced by the left atrioventricular pressure gradient (E wave) and late diastole filling induced by atrial contraction (A wave). Around the age of 60 years, a decline in E velocity occurs, followed by an increase in compensatory age-related A velocity due to left ventricular diastolic failure, and reversal of the E/A ratio. This change happened regardless of age in response to an increase in blood pressure, even during the acute phase of stroke. Individuals with AF did not exhibit an increase in the velocity of their A-waves [47,48]. Meanwhile, LA stunning is another factor that contributes to patients with atrial fibrillation having a low A wave velocity [49]. Anticoagulation for subclinical device-detected AF is debatable, as short-term AF has not been demonstrated to warrant it [4].
Neurological Imaging Methods
Evaluation of the Cerebral Parenchyma
COMPUTED TOMOGRAPHY (CT) SCANS: This is a good imaging modality for identifying and excluding bleeding. Rapid imaging and simplicity of reporting, especially in environments with minimal information, continue to be its main benefits. Nonetheless, it demonstrates low sensitivity for detecting infarcts early in their evolution. Subtle symptoms such as sulcal effacement or “the hyperdense middle cerebral artery (MCA) sign” can be present in large infarcts, although they are inconsistent and often ignored (Figure 3).
MAGNETIC RESONANCE IMAGING (MRI) SCANS: This is by far the best imaging modality available for assessing the brain parenchymal state. Numerous imaging sequences aid in not only delineating the infarcted region but also in providing a chronological context in cases when the history is not well known. By selecting the right b-value, diffusion-weighted imaging (DWI) (Figure 4), MRI can assist in identifying even hyper-acute infarcts. Cortical stroke in various vascular areas raises the possibility of a cardioembolic origin. The presence of an infarct in the fluid-attenuated inversion recovery (FLAIR) sequence implies that the infarct formed partially over 6 h ago. Using susceptibility-weighted imaging (SWI), MRI scans can also correctly detect hemorrhagic infarcts.
MAGNETIC RESONANCE ANGIOGRAPHY:
This is an advantageous imaging modality, especially in patients with renal impairment, because it does not involve administration of intravenous contrast medium. A disadvantage is that it is susceptible to a variety of errors, including overestimation of stenotic lesions.
CT ANGIOGRAPHY:
This is the criterion standard for examining the cerebral vasculature, but has the disadvantage of not being suitable in individuals with renal impairment.
Complications of Cardioembolic Strokes
Cardioembolic strokes, when not treated adequately, have a greater proclivity for early and late recurrences than athero-thrombotic strokes. Hemorrhagic events, both spontaneous and as a result of anticoagulant treatment, are potentially fatal complications of this illness. Long-term impairment and consequences such as internal cranial hypertension can emerge, although their degree and extent of deterioration corresponds to the severity and depth of the neuro-deficiency [51].
Conclusions
Due to the high likelihood of recurrence and death associated with cardioembolic stroke, cardioembolic embolism should be suspected and evaluated in all patients presenting with stroke. Owing mostly to the increasing prevalence of AF with age, the incidence of cardioembolic stroke is predicted to rise in the future. Both 2-D TTE and 2-D TEE are critical in detecting cardioembolic causes of stroke. These imaging modalities give critical and additional data that can be utilized for secondary prevention and determining the therapeutic plan for stroke patients. However, the novel technological innovation, known as the RT3DE, provides better insight into the pathogenesis of cardioembolic stroke.
Figures
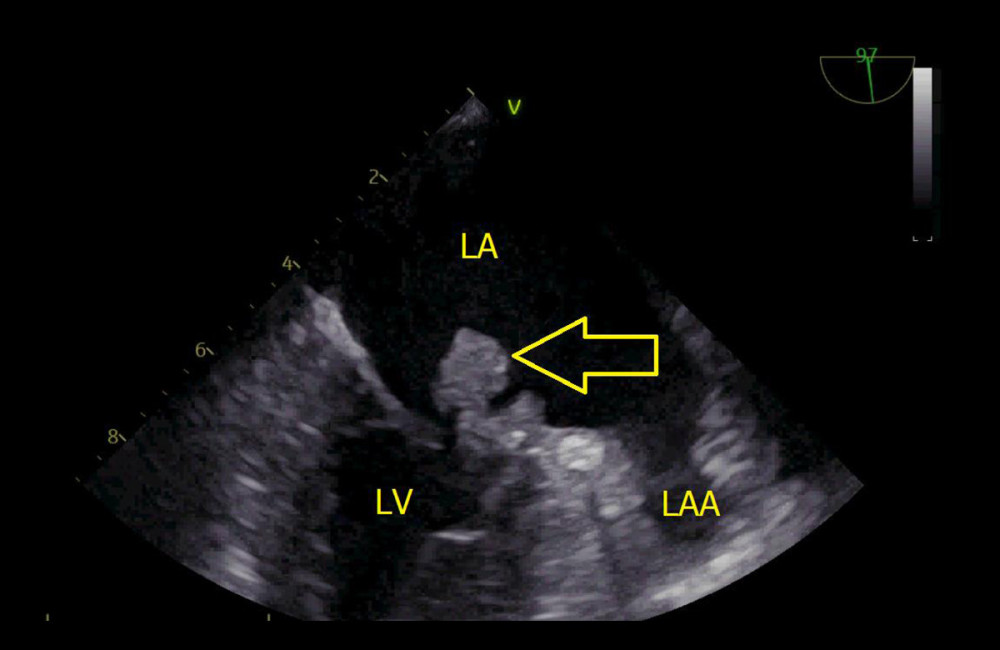 Figure 1. Transesophageal echocardiography. Vegetations of the mitral valve (indicated by yellow arrow). The image was acquired using a VIVID5S, General Electrics phased array ultrasonoscope (Tirat Carmel, Israel). LV – left ventricle; LA – left atrium.
Figure 1. Transesophageal echocardiography. Vegetations of the mitral valve (indicated by yellow arrow). The image was acquired using a VIVID5S, General Electrics phased array ultrasonoscope (Tirat Carmel, Israel). LV – left ventricle; LA – left atrium. 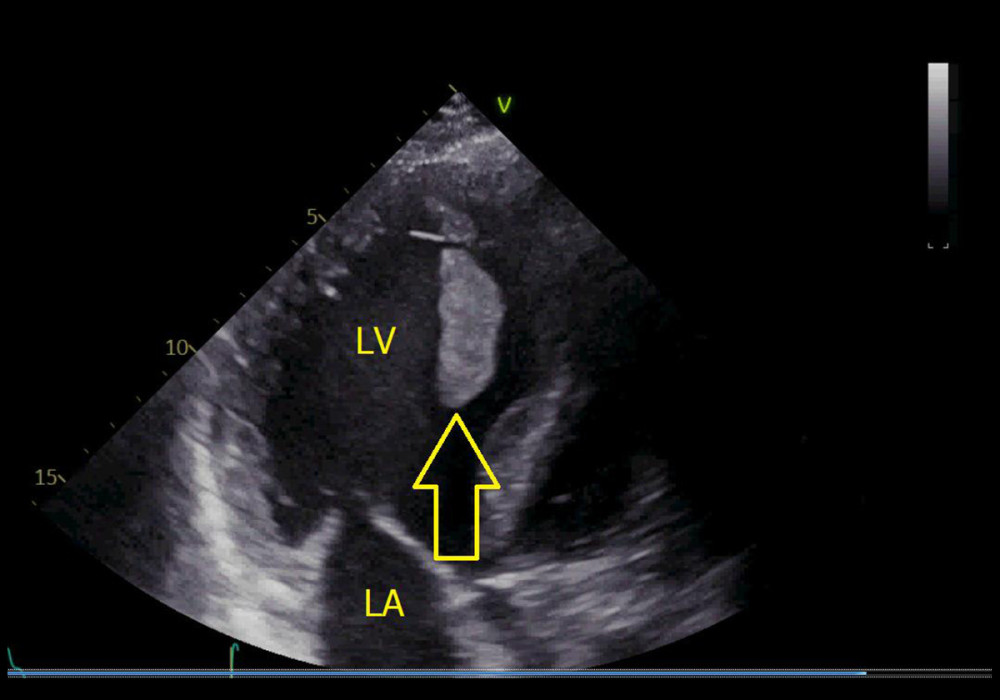 Figure 2. Transthoracic echocardiography. Thrombus in the left ventricle (indicated by yellow arrow). The image was acquired using a VIVID5S, General Electrics phased array ultrasonoscope (Tirat Carmel, Israel). LV – left ventricle.
Figure 2. Transthoracic echocardiography. Thrombus in the left ventricle (indicated by yellow arrow). The image was acquired using a VIVID5S, General Electrics phased array ultrasonoscope (Tirat Carmel, Israel). LV – left ventricle. 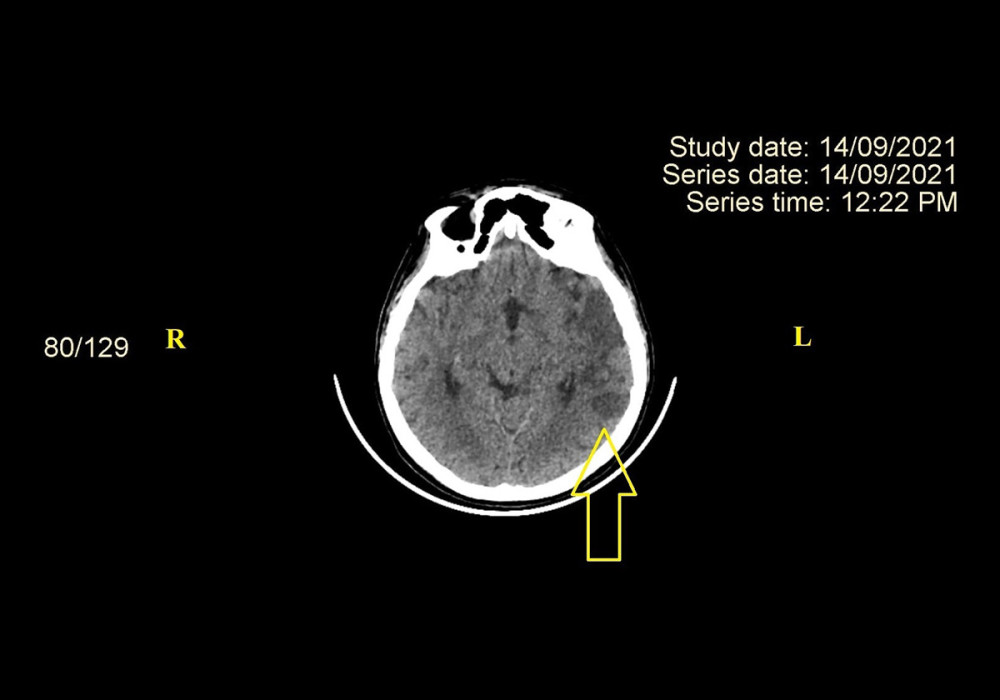 Figure 3. CT scan. Ischemic stroke in left superficial MCA territory (indicated by yellow arrow). The image was acquired using GE REVOLUTION EVO 128 SLICE computed tomograph produced by General Electric Healthcare Japan Corporation – Japan. CT – computed tomography; MCA – middle cerebral artery, R – right; L – left.
Figure 3. CT scan. Ischemic stroke in left superficial MCA territory (indicated by yellow arrow). The image was acquired using GE REVOLUTION EVO 128 SLICE computed tomograph produced by General Electric Healthcare Japan Corporation – Japan. CT – computed tomography; MCA – middle cerebral artery, R – right; L – left. 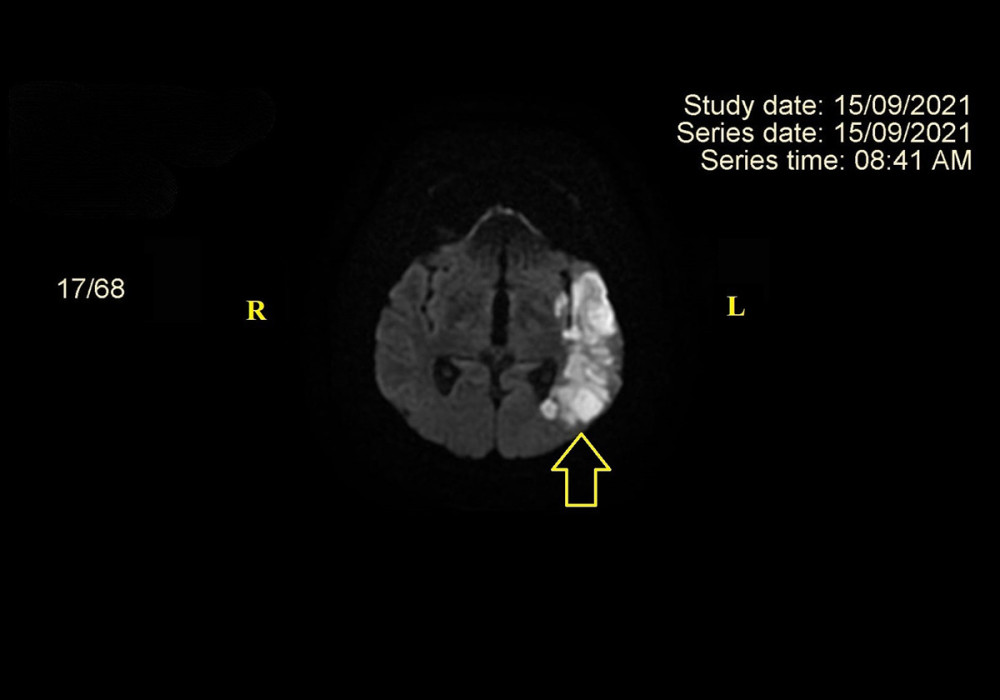 Figure 4. MRI in DWI. Recent ischemic stroke in superficial left MCA (indicated by yellow arrow). The image was acquired using GE SIGNA EXPLORER 1.5T magnetic resonance imaging device produced by General Electric Healthcare Japan Corporation – Japan. MRI – magnetic resonance imaging; DWI – diffusion-weighted imaging; R – right; L – left.
Figure 4. MRI in DWI. Recent ischemic stroke in superficial left MCA (indicated by yellow arrow). The image was acquired using GE SIGNA EXPLORER 1.5T magnetic resonance imaging device produced by General Electric Healthcare Japan Corporation – Japan. MRI – magnetic resonance imaging; DWI – diffusion-weighted imaging; R – right; L – left. References
1. World Health Organization: Report on Mortality and Global Health Estimates Mortality and global health estimates (who.int) 12 27, 2021 https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates
2. Saver JL, Clinical practice. Cryptogenic stroke: N Engl J Med, 2016; 374(21); 2065-74
3. Gladstone DJ, Spring M, Dorian P, Atrial fibrillation in patients with cryptogenic stroke: N Engl J Med, 2014; 370(26); 2467-77
4. Hindricks G, Potpara T, Dagres N, 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): The Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC: Eur Heart J, 2021; 42(5); 373-498
5. Wolf PA, Abbott RD, Kannel WB, Atrial fibrillation as an independent risk factor for stroke: The Framingham Study: Stroke, 1991; 22(8); 983-88
6. Sanna T, Diener HC, Passman RS, Cryptogenic stroke and underlying atrial fibrillation: N Engl J Med, 2014; 370(26); 2478-86
7. Kundnani NR, Rosca CI, Sharma A, Selecting the right anticoagulant for stroke prevention in atrial fibrillation: Eur Rev Med Pharmacol Sci, 2021; 25(13); 4499-505
8. Liao J, Khalid Z, Scallan C, Noninvasive cardiac monitoring for detecting paroxysmal atrial fibrillation or flutter after acute ischemic stroke: A systematic review: Stroke, 2007; 38(11); 2935-40
9. Jianu DCJS, Munteanu G, Dan TF, Birsan C: Diagnosis of symptomatic intracranial atherosclerotic disease, in new insight into cerebrovascular diseases – an updated comprehensive review, 2019, IntechOpen, London
10. Rosca CI, Kundnani NR, Tudor A, Benefits of prescribing low-dose digoxin in atrial fibrillation: Int J Immunopathol Pharmacol, 2021; 35 20587384211051955
11. Sharma A, Christodorescu R, Agbariah A, Cardiovascular risk prediction parameters for better management in rheumatic diseases: Healthcare, 2022; 10(2); 312
12. Yuan K, Kasner SE, Patent foramen ovale and cryptogenic stroke: Diagnosis and updates in secondary stroke prevention: Stroke Vasc Neurol, 2018; 3(2); 84-91
13. Habib G, Lancellotti P, Antunes MJ, 2015 ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM): Eur Heart J, 2015; 36(44); 3075-128
14. Vahanian A, Beyersdorf F, Praz F, 2021 ESC/EACTS Guidelines for the management of valvular heart disease: Eur Heart J, 2022; 43(7); 561-632
15. Otto CM, Nishimura RA, Bonow RO, 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines: Circulation, 2021; 143(5); e35-e71
16. García-Cabrera E, Fernández-Hidalgo N, Almirante B, Neurological complications of infective endocarditis: Risk factors, outcome, and impact of cardiac surgery: A multicenter observational study: Circulation, 2013; 127(23); 2272-84
17. Pathickal SM, Park TE, Sharma R, Clinical outcomes associated with the use of anticoagulant and antiplatelet agents in patients undergoing treatment for infective endocarditis: A pilot study: Clin Ther, 2020; 42(9); 1828-38
18. Alec V, Fabien P, Milan M, 2021 ESC/EACTS Guidelines for the management of valvular heart disease: EuroIntervention, 2022; 17(14); e1126-e95
19. McDonagh TA, Metra M, Adamo M, 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: Eur Heart J, 2021; 42(36); 3599-726
20. Ibanez B, James S, Agewall S, 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC): Eur Heart J, 2018; 39(2); 119-77
21. Nakanishi K, Homma S, Role of echocardiography in patients with stroke: J Cardiol, 2016; 68(2); 91-99
22. Toh N, Kanzaki H, Nakatani S, Left atrial volume combined with atrial pump function identifies hypertensive patients with a history of paroxysmal atrial fibrillation: Hypertension, 2010; 55(5); 1150-56
23. Vasan RS, Larson MG, Levy D, Doppler transmitral flow indexes and risk of atrial fibrillation (the Framingham Heart Study): Am J Cardiol, 2003; 91(9); 1079-83
24. Lang RM, Bierig M, Devereux RB, Recommendations for chamber quantification: a report from the American Society of Echocardiography’s Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of Cardiology: J Am Soc Echocardiogr, 2005; 18(12); 1440-63
25. Hagen PT, Scholz DG, Edwards WD, Incidence and size of patent foramen ovale during the first 10 decades of life: An autopsy study of 965 normal hearts: Mayo Clin Proc, 1984; 59(1); 17-20
26. Lechat P, Mas JL, Lascault G, Prevalence of patent foramen ovale in patients with stroke: N Engl J Med, 1988; 318(18); 1148-52
27. Webster MW, Chancellor AM, Smith HJ, Patent foramen ovale in young stroke patients: Lancet, 1988; 2(8601); 11-12
28. Homma S, Messé SR, Rundek T, Patent foramen ovale: Nat Rev Dis Primers, 2016; 2; 15086
29. Pearson AC, Nagelhout D, Castello R, Atrial septal aneurysm and stroke: A transesophageal echocardiographic study: J Am Coll Cardiol, 1991; 18(5); 1223-29
30. Overell JR, Bone I, Lees KR, Interatrial septal abnormalities and stroke: A meta-analysis of case-control studies: Neurology, 2000; 55(8); 1172-79
31. Porter TR, Abdelmoneim S, Belcik JT, Guidelines for the cardiac sonographer in the performance of contrast echocardiography: A focused update from the American Society of Echocardiography: J Am Soc Echocardiogr, 2014; 27(8); 797-810
32. Lip GY, Nieuwlaat R, Pisters R, Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: The euro heart survey on atrial fibrillation: Chest, 2010; 137(2); 263-72
33. Saw J, Lempereur M, Percutaneous left atrial appendage closure: Procedural techniques and outcomes: JACC Cardiovasc Interv, 2014; 7(11); 1205-20
34. Tsang TS, Abhayaratna WP, Barnes ME, Prediction of cardiovascular outcomes with left atrial size: Is volume superior to area or diameter?: J Am Coll Cardiol, 2006; 47(5); 1018-23
35. Iwataki M, Takeuchi M, Otani K, Measurement of left atrial volume from transthoracic three-dimensional echocardiographic datasets using the biplane Simpson’s technique: J Am Soc Echocardiogr, 2012; 25(12); 1319-26
36. Senior R, Becher H, Monaghan M, Contrast echocardiography: Evidence-based recommendations by European Association of Echocardiography: Eur J Echocardiogr, 2009; 10(2); 194-212
37. Amarenco P, Cohen A, Hommel M, Atherosclerotic disease of the aortic arch as a risk factor for recurrent ischemic stroke: N Engl J Med, 1996; 334(19); 1216-21
38. Di Tullio MR, Sacco RL, Savoia MT, Aortic atheroma morphology and the risk of ischemic stroke in a multiethnic population: Am Heart J, 2000; 139(2 Pt 1); 329-36
39. Di Tullio MR, Sacco RL, Savoia MT, Gender differences in the risk of ischemic stroke associated with aortic atheromas: Stroke, 2000; 31(11); 2623-27
40. Amarenco P, Davis S, Jones EF, Clopidogrel plus aspirin versus warfarin in patients with stroke and aortic arch plaques: Stroke, 2014; 45(5); 1248-57
41. Amarenco P, Davis S, Jones EF, Clopidogrel plus aspirin versus warfarin in patients with stroke and aortic arch plaques: Stroke, 2014; 45(5); 1248-57
42. Kernan WN, Ovbiagele B, Black HR, Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: A guideline for healthcare professionals from the American Heart Association/American Stroke Association: Stroke, 2014; 45(7); 2160-236
43. Kronzon I, Tunick PA, Aortic atherosclerotic disease and stroke: Circulation, 2006; 114(1); 63-75
44. Piazzese C, Tsang W, Sotaquira M, Semiautomated detection and quantification of aortic plaques from three-dimensional transesophageal echocardiography: J Am Soc Echocardiogr, 2014; 27(7); 758-66
45. Rosenberg MA, Gottdiener JS, Heckbert SR, Echocardiographic diastolic parameters and risk of atrial fibrillation: The Cardiovascular Health Study: Eur Heart J, 2012; 33(7); 904-12
46. Jordan K, Yaghi S, Poppas A, Left atrial volume index is associated with cardioembolic stroke and atrial fibrillation detection after embolic stroke of undetermined source: Stroke, 2019; 50(8); 1997-2001
47. Nakagawa T, Hara H, Yamamoto M, Transmitral inflow wave and progression from paroxysmal to permanent atrial fibrillation in Asian people: Heart Asia, 2019; 11(2); e011166
48. Henein MY, Lindqvist P, Assessment of left ventricular diastolic function by Doppler echocardiography: Card Fail Rev, 2015; 1(2); 87-89
49. Perino AC, Fan J, Askari M, Practice variation in anticoagulation prescription and outcomes after device-detected atrial fibrillation: Circulation, 2019; 139(22); 2502-12
50. Lin MP, Liebeskind DS, Imaging of ischemic stroke: Continuum (Minneap Minn), 2016; 22(5, Neuroimaging); 1399-423
51. Griñán K, Arboix A, Massons J, Cardioembolic stroke: Risk factors, clinical features, and early outcome in 956 consecutive patients: Rev Invest Clin, 2020; 73(1); 23-30
Figures
 Figure 1. Transesophageal echocardiography. Vegetations of the mitral valve (indicated by yellow arrow). The image was acquired using a VIVID5S, General Electrics phased array ultrasonoscope (Tirat Carmel, Israel). LV – left ventricle; LA – left atrium.
Figure 1. Transesophageal echocardiography. Vegetations of the mitral valve (indicated by yellow arrow). The image was acquired using a VIVID5S, General Electrics phased array ultrasonoscope (Tirat Carmel, Israel). LV – left ventricle; LA – left atrium. Figure 2. Transthoracic echocardiography. Thrombus in the left ventricle (indicated by yellow arrow). The image was acquired using a VIVID5S, General Electrics phased array ultrasonoscope (Tirat Carmel, Israel). LV – left ventricle.
Figure 2. Transthoracic echocardiography. Thrombus in the left ventricle (indicated by yellow arrow). The image was acquired using a VIVID5S, General Electrics phased array ultrasonoscope (Tirat Carmel, Israel). LV – left ventricle. Figure 3. CT scan. Ischemic stroke in left superficial MCA territory (indicated by yellow arrow). The image was acquired using GE REVOLUTION EVO 128 SLICE computed tomograph produced by General Electric Healthcare Japan Corporation – Japan. CT – computed tomography; MCA – middle cerebral artery, R – right; L – left.
Figure 3. CT scan. Ischemic stroke in left superficial MCA territory (indicated by yellow arrow). The image was acquired using GE REVOLUTION EVO 128 SLICE computed tomograph produced by General Electric Healthcare Japan Corporation – Japan. CT – computed tomography; MCA – middle cerebral artery, R – right; L – left. Figure 4. MRI in DWI. Recent ischemic stroke in superficial left MCA (indicated by yellow arrow). The image was acquired using GE SIGNA EXPLORER 1.5T magnetic resonance imaging device produced by General Electric Healthcare Japan Corporation – Japan. MRI – magnetic resonance imaging; DWI – diffusion-weighted imaging; R – right; L – left.
Figure 4. MRI in DWI. Recent ischemic stroke in superficial left MCA (indicated by yellow arrow). The image was acquired using GE SIGNA EXPLORER 1.5T magnetic resonance imaging device produced by General Electric Healthcare Japan Corporation – Japan. MRI – magnetic resonance imaging; DWI – diffusion-weighted imaging; R – right; L – left. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








