24 June 2022: Clinical Research
Medroxyprogesterone Acetate for Abnormal Uterine Bleeding Due to Ovulatory Dysfunction: The Effect of 2 Different-Duration Regimens
Rukiye Ada BenderDOI: 10.12659/MSM.936727
Med Sci Monit 2022; 28:e936727
Abstract
BACKGROUND: Abnormal uterine bleeding (AUB) lowers the quality of life of women. This study attempted to determine which treatment protocol of medroxyprogesterone acetate (MPA), 15 vs 10 day-administration in a luteal phase, provides better outcomes in women with ovulatory dysfunction-related AUB (AUB-O).
MATERIAL AND METHODS: The study included a total of 52 patients with AUB-O: Women in Group A were given MPA between days 11 and 25 of the menstrual cycle (15-day protocol), whereas women in Group B were given MPA between days 16 and 25 (10-day protocol). Outcomes were compared between the 2 groups.
RESULTS: Women in group B, compared with those in group A, more often showed regular menstrual cycles and decrement of AUB. In group B, 3 cycles of treatment were sufficient to achieve AUB-stop (p<0.05). Post-treatment hemogram parameters and surgical treatment requirements were not different between the 2 groups.
CONCLUSIONS: In the second half of the cycle/predicted luteal phase, 10-day cyclic use of MPA (the group B) better regulated the menstrual cycle and more frequently stopped AUB-O.
Keywords: Medroxyprogesterone Acetate, Menorrhagia, Metrorrhagia, Clinical Protocols, Female, Humans, Menstrual Cycle, Quality of Life, Uterine Hemorrhage
Background
AUB is experienced by approximately one-third of women of reproductive age, mostly in the premenopausal period, which impairs their quality of life [1]. As a medical condition, it manifests as increased blood loss and pain, decreased sexual desire and fertility, and increased cost due to non-routine follow-up and treatment [2].
The classification of AUB in non-pregnant women was updated by the International Federation of Gynecology and Obstetrics (FIGO) in 2011. According to the new classification, the causes of AUB are divided into 2 groups: Structural and non-structural causes. The group containing structural causes is divided further into PALM (Polyp, Adenomyosis, Leiomyoma, Malignancy-hyperplasia) subgroups, whereas the group containing non-structural causes is divided into COEIN (Coagulopathy, Ovulatory dysfunction, Endometrial disorders, Iatrogenic causes, Not yet classified) subgroups [1].
AUB-O describes a patient with anovulatory, oligo- or irregular ovulation, or experiencing luteal out-of-phase (LOOP) events, particularly toward the end of the reproductive period [3]. AUB-O occurs in women as a result of absence of cyclic progesterone due to absence of the corpus luteum and the stimulation of the endometrium by unopposed estrogen [4]. For the correct definition of women with AUB-O, excessive bleeding lasting more than 8 days in the last 6 to 12 months should not be due to pregnancy, diagnosed pelvic and systemic pathologies, or hormone-containing drug use [1]. It seems to emerge as a result of a luteal phase defect due to ovulatory dysfunction in intermenstrual hemorrhages in the cyclic and premenstrual periods.
Hormone-based therapies have an important place as medical therapy in the treatment of AUB-O [4]. Hormone-based treatment consists of synthetic progestins, combined oral contraceptives, GnRH analogs, and intrauterine device insertion with levonorgestrel [5]. Synthetic progestins are predominantly used to mimic the effects of endogenous progesterone produced by the ovary to treat ovulatory dysfunction. Of these progestins, medroxyprogesterone acetate (MPA), a viable 17α-hydroxyprogesterone derivative having a highly similar selectivity to progesterone, has a higher bioavailability and a longer half-life than progesterone and can be safely administered to women requiring hormone therapy without any change in endogenous progestin production [6,7]. The transformation dose of MPA that transforms the proliferative endometrium into the secretory endometrium is 5–10 mg daily, and 80 mg per cycle. This drug is considered to lack sufficient estrogenic activity, although androgenic and anabolic effects have been demonstrated [8]. MPA can be utilized in the medical treatment of AUB-O owing to these pharmacodynamic properties.
MPA therapy is used in the second half of the cycle, like other progestins, but the duration of use varies among clinicians. Although studies have compared the efficacy of MPA with different progestins, there are no data comparing MPA treatments used for different durations [9,10].
In this study, we aimed to evaluate the effect of different durations of MPA administration on the treatment of AUB-O in women diagnosed with AUB-O and treated cyclically with MPA.
Material and Methods
ETHICS STATEMENT:
The study was approved by the Biruni University Ethics Committee (Decision no: 2021/49-13). All methods were performed following the relevant guidelines and regulations by including a statement. Informed consent for the use of their medical records in scientific studies was obtained from all patients.
STUDY DESIGN:
The data of 52 women were included in the study by retrospectively scanning the data of women in the reproductive period who were diagnosed with AUB-O and underwent MPA treatment between April 2017 and March 2021 at Medicana International Istanbul Hospital Gynecology and Obstetrics Clinic. All of the patients were treated and followed up by the same gynecologist and obstetrician, and the data were recorded by the same specialist. AUB evaluation of all patients was performed with an appropriate algorithm according to FIGO’s recommendation.
An endometrial biopsy of all included AUB-O cases was required due to the endometrial thickness observations on ultrasonography, age status (>45 years), and/or an increased risk of neoplasia or hyperplasia.
For the study, causes other than ovulatory dysfunction in the PALM-COEIN classification were excluded in retrospectively scanned patient files. Anamnesis, physical examination, routine laboratory tests, and transvaginal ultrasonography (Voluson S6 [GE Healthcare, Milwaukee, WI]) of all women who applied to our clinic with AUB were evaluated. However, the inclusion criteria were excessive bleeding that lasted longer than 8 days and intermenstrual bleeding that was frequently in the premenstrual period.
Hemoglobin, hematocrit, and prolactin levels, leukocyte and platelet counts, aPTT, prothrombin time, the 2nd–5th day of the cycle FSH, LH, and estradiol levels, and the 19th–22nd day of the cycle progesterone levels were determined. FSH, LH, and estradiol levels of all women included were compatible with the reproductive period. Women with coagulopathy, systemic diseases (eg, thyroid diseases, diabetes) and hyperprolactinemia, or presenting bleeding in their postmenopausal or adolescent periods were not included in the study. According to the literature, if there is still uncertainty regarding ovulation after ruling out alternative causes of AUB-O, blood progesterone levels obtained at the predicted mid-luteal phase are used to confirm the diagnosis of AUB-O [1]. The progesterone level measured between the 19th and 22nd days of the cycle, which corresponds to the mid-luteal phase, was required to be <3 ng/mL.
All of these women had undergone endometrial biopsy with appropriate symptoms. Women reported to have endometrial polyps or secretory endometrium according to endometrial cytology analysis results, used hormone treatments, or having adenomyosis or leiomyoma detected by transvaginal ultrasonography and whose AUB-O was related to these pathologies were excluded from the study.
In this clinic, women with AUB-O are offered cyclic MPA as first-line therapy. The indications and contraindications for this treatment are evaluated, the patients are informed, and the patient’s regular use of the treatment is followed up for 3 cycles. These patients are re-evaluated after treatment and 6 months after the end of treatment. If MPA treatment fails, different surgical and alternative medical treatments are applied to women who continue to bleed, according to appropriate symptoms, patient request, and fertility desire.
In our clinic, 2 different MPA treatment protocols are applied with minimum effective dose and duration, which have no adverse effects, reduce bleeding, and increase the quality of life. The daily dose of 10 mg was applied between the 11th and 25th day of the cycle for patients who bleed for 10 days or longer. Although the dose is the same (10 mg), MPA treatment was applied between the 16th and 25th day of the cycle in patients with bleeding for less than 10 days. In this study, we aimed to compare the efficacy of these 2 treatment protocols on AUB-O in women.
To determine the efficacy of the treatment, hemoglobin and hematocrit levels, menstrual cycle patterns, and the need for additional surgical or medical treatment at 6 months post-treatment were evaluated.
STATISTICAL ANALYSIS:
The data obtained in the study were analyzed with SPSS (Version 25). Results were evaluated as mean ± standard deviation or frequencies (percentages). In group comparisons, normally distributed parameters were evaluated with the paired-sample
Results
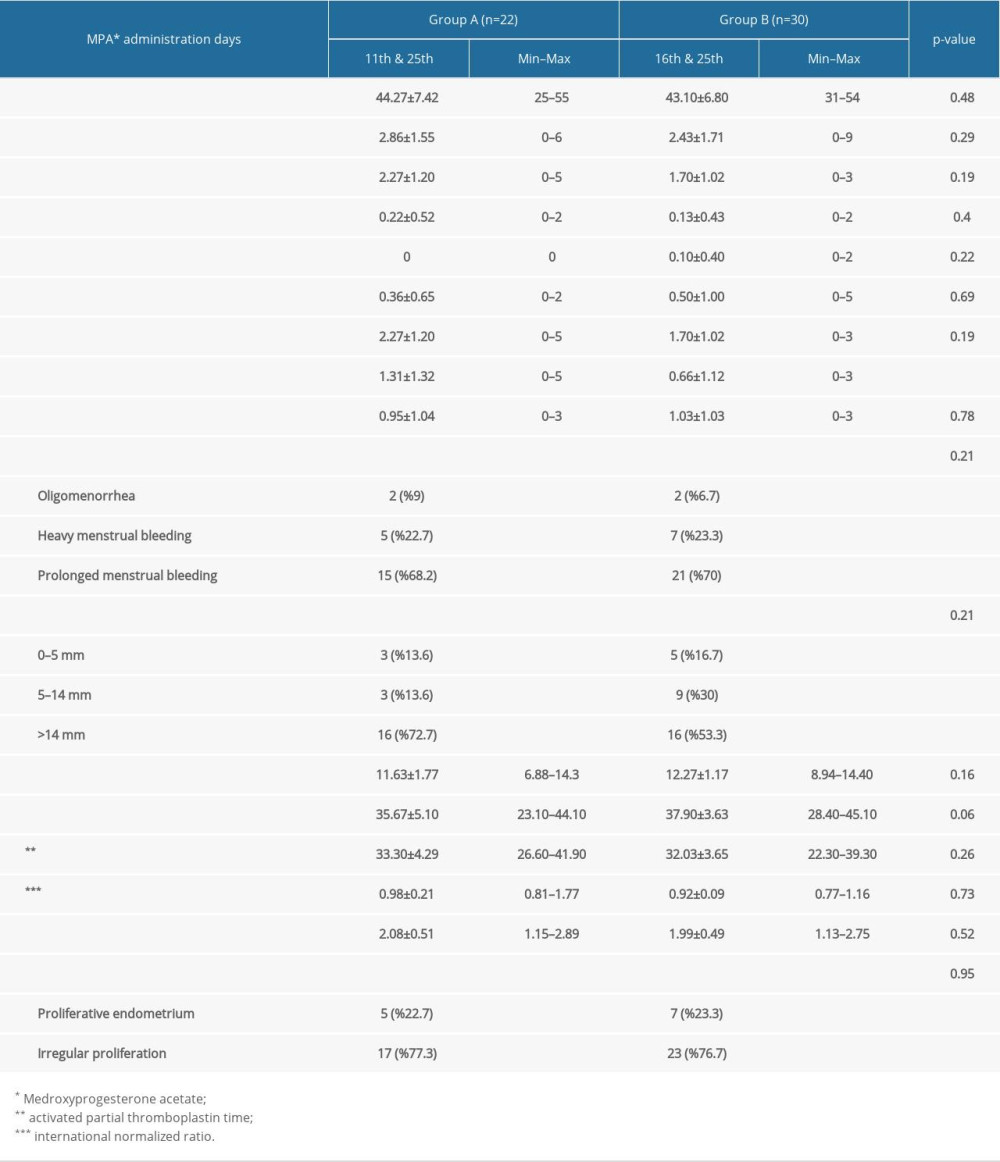
The women included in the study were divided into 2 groups according to the time they received MPA. The first group (group A) was treated with MPA between the 11th and 25th days of the menstrual cycle, while the second group (group B) was treated between the 16th and 25th days of the menstrual cycle. For both groups of patients, we recorded age, number of deliveries, number of cesarean-normal births, number of abortions if any, menstrual pattern, whether medical treatment was applied previously, endometrial thickness determined by ultrasonography at the diagnosis stage, pretreatment hemogram, and bleeding parameters, the progesterone level measured between the 19th and 22nd days of the cycle, and endometrial cytology results (Table 1). Statistically, a significant difference was only observed in the number of vaginal deliveries among the whole demographic data (
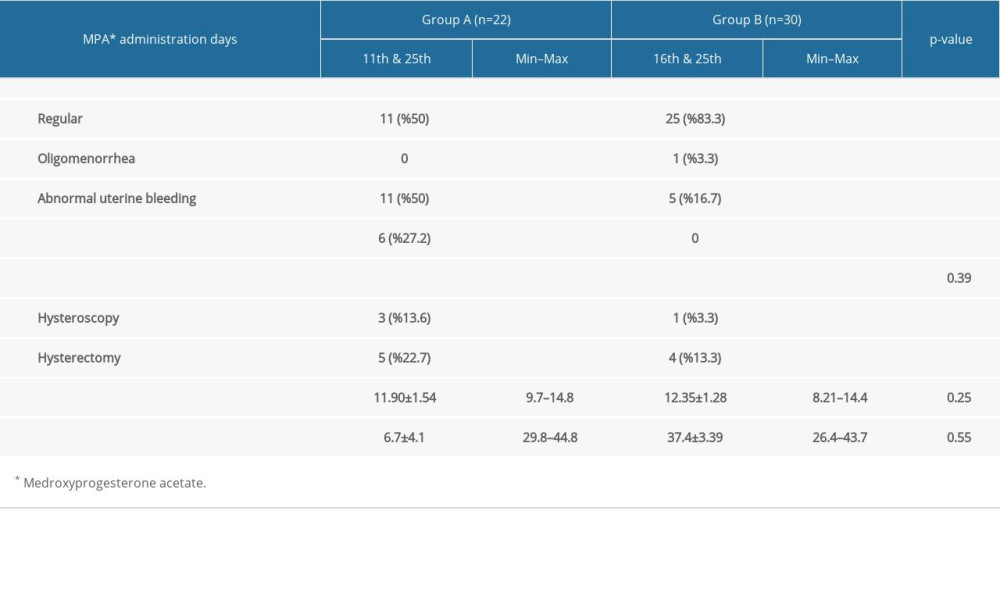
In comparison between groups, we observed that the regularity of the menstrual cycle after treatment was significantly higher in group B compared to group A, and, similarly, abnormal uterine bleeding was higher in group A compared to group B (
Discussion
AUB is a symptom used to describe bleeding outside the 5th to 95th percentiles of the population for menstrual cycle regularity, frequency, duration, and volume, which is considered acute in the presence of an immediate condition that requires immediate assistance and chronic when recurring frequently in the previous 6 months [11,12].
Bleeding times vary in AUB due to ovulatory dysfunction [13]. In AUB-O, prolonged estrogen stimulation continues without progesterone withdrawal. Therefore, progesterone, which is lacking in the second half of the cycle, can be used in the treatment [14]. Long-term use of progesterone causes endometrial atrophy by preventing estrogen-induced endometrial proliferation [15]. In addition, it is known that the use of MPA can significantly reduce or even stop bleeding after an average of 3 days [16].
There are insufficient data comparing durations of MPA treatment for AUB-O. A study in a mixed population of late reproductive women with AUB-O or severe AUB reported that MPA 5 mg tablets administered continuously for 2 months reduced menstrual blood loss by 33% [17]. As a common medical treatment agent, it is very important to determine the effective use of MPA. Increasingly serious adverse effects with continuous use will decrease significantly by determining the minimum but effective time and effective treatment will be provided. Although the sample range of this study is not very large, it provides important data to the literature with this comparison.
In a small study with only 6 patients, Fraser et al used 5 mg of norethisterone acetate or 10 mg of MPA to treat AUB-O from days 12 to 25 of the cycle and concluded that after the 1st and 2nd months of treatment, the menstrual blood losses of women decreased by 39% and 51%, respectively [14]. In another study, Dunphy et al evaluated the efficacy of 10 mg of MPA daily in the luteal phase for a short time (from days 16 to 25 of the cycle; n=11), and reported that bleeding after 1 and 2 months of administration, was reduced by 25% and 41%, respectively. Interestingly, a 12% increase in bleeding was observed after 3 months of treatment [18]. However, the results of these studies do not provide a comparison of the minimum effective doses of MPA.
In our study, in which the contribution of the difference in MPA administration times to the treatment was examined retrospectively, we observed that on the 16th and 25th days, compared to the 11th and 25th days, there was improved menstrual cycle, decreased abnormal uterine bleeding, and decreased prolonged MPA requirement after treatment. In our routine practice, we use MPA for a period of 15 days between the 11th and 25th days of the cycle, with earlier treatment in women whose bleeding lasts longer than 10 days, to reduce blood loss and increase the quality of life. However, our study showed that even if MPA is used for as long as 15 days for bleeding lasting longer than 10 days, it is not effective in regulating the menstrual cycle compared to women who bleed for less than 10 days and use MPA for 10 days. Post-treatment surgery requirements and total hemoglobin and hematocrit levels were similar between the 2 groups, suggesting that the use of MPA in bleeding lasting longer than 10 days may not be effective. In these women, it may be beneficial to continue treatment with other medical and surgical treatment methods.
The transformation dose of MPA, which transforms the proliferative endometrium into secretory endometrium, is 5–10 mg per day and 80 mg per cycle [8]. MPA can be used in the long phase (≥21 days of use per cycle) and short phase (≤14 days of use per cycle). However, in long-phase use, the patients tend to discontinue the medication due to the disturbing adverse effects.
The underlying reason for the similar treatment success in the 2 groups may be the insufficient dose of MPA. Like other clinicians, we treated our patients with a daily dose of 10 mg. BMI, age, or plasma estrogen levels of the patients were not taken into account when determining this dose for treatment. A dose adjustment could not be performed considering the pharmacokinetic and pharmacodynamic variables, which have become very important for most treatments today. For example, recent studies show that progesterone receptor levels and different microsomal enzyme polymorphisms (such as CYP2C19 and CYP2C9) significantly alter plasma progesterone levels after MPA use [19]. Considering all these, more studies are needed to adjust the daily dose.
Conclusions
The various medical treatment options available for the treatment of AUB-O should be used rationally to consider surgical procedures for specific situations and to avoid unnecessary surgical procedures. Although our study gave concrete results on how the use of MPA for different durations affects the success of treatment, more comprehensive and comparative studies are needed on the use of MPA at different doses.
According to the results of our study, prolonged use of MPA in women with bleeding for more than 10 days in the treatment of AUB-O is not sufficient to regulate the cycles. However, in women who bleed for less than 10 days, 10-day use is effective and has an advantage in terms of dose and time-related adverse effects.
References
1. Munro MG, Critchley HOD, Fraser ISFIGO Menstrual Disorders Committee, The two FIGO systems for normal and abnormal uterine bleeding symptoms and classification of causes of abnormal uterine bleeding in the reproductive years: 2018 revisions: Int J Gynaecol Obstet, 2018; 143(3); 393-408 Erratum in: Int J Gynaecol Obstet. 2019;144(2):237
2. Rahn DD, Abed H, Sung VWSociety of Gynecologic Surgeons-Systematic Review Group, Systematic review highlights difficulty interpreting diverse clinical outcomes in abnormal uterine bleeding trials: J Clin Epidemiol, 2011; 64(3); 293-300
3. Hale GE, Hughes CL, Burger HG, Atypical estradiol secretion and ovulation patterns caused by luteal out-of-phase (LOOP) events underlying irregular ovulatory menstrual cycles in the menopausal transition: Menopause, 2009; 16(1); 50-59
4. Wouk N, Helton M, Abnormal uterine bleeding in premenopausal women: Am Fam Physician, 2019; 99(7); 435-43
5. Sweet MG, Schmidt-Dalton TA, Weiss PM, Madsen KP, Evaluation and management of abnormal uterine bleeding in premenopausal women: Am Fam Physician, 2012; 85(1); 35-43
6. Piccinni MP, Lombardelli L, Logiodice F, Medroxyprogesterone acetate decreases Th1, Th17, and increases Th22 responses via AHR signaling which could affect susceptibility to infections and inflammatory disease: Front Immunol, 2019; 10; 642
7. Sitruk-Ware R, Nath A, Mishell DR, Contraception technology: Past, present and future: Contraception, 2013; 87(3); 319-30
8. Schindler AE, Campagnoli C, Druckmann R, Classification and pharmacology of progestins: Maturitas, 2003; 46(Suppl 1); S7-16
9. Bofill Rodriguez M, Lethaby A, Jordan V, Progestogen-releasing intrauterine systems for heavy menstrual bleeding: Cochrane Database Syst Rev, 2020; 6(6); CD002126
10. Lethaby AE, Cooke I, Rees M, Progesterone or progestogen-releasing intrauterine systems for heavy menstrual bleeding: Cochrane Database Syst Rev, 2005; 19(4) Update in: Cochrane Database Syst Rev. 2015;4:CD002126
11. Fraser IS, Critchley HO, Broder M, Munro MG, The FIGO recommendations on terminologies and definitions for normal and abnormal uterine bleeding: Semin Reprod Med, 2011; 29(5); 383-90
12. Munro MG, Critchley HO, Broder MS, Fraser ISFIGO Working Group on Menstrual Disorders, FIGO classification system (PALM-COEIN) for causes of abnormal uterine bleeding in nongravid women of reproductive age: Int J Gynaecol Obstet, 2011; 113(1); 3-13
13. Hickey M, Higham JM, Fraser I, Progestogens with or without oestrogen for irregular uterine bleeding associated with anovulation: Cochrane Database Syst Rev, 2012; 2012(9); CD001895
14. Fraser IS, Treatment of ovulatory and anovulatory dysfunctional uterine bleeding with oral progestogens: Aust N Z J Obstet Gynaecol, 1990; 30(4); 353-56
15. Dinh A, Sriprasert I, Williams AR, Archer DF, A review of the endometrial histologic effects of progestins and progesterone receptor modulators in reproductive age women: Contraception, 2015; 91(5); 360-67
16. Munro MG, Mainor N, Basu R, Oral medroxyprogesterone acetate and combination oral contraceptives for acute uterine bleeding: A randomized controlled trial: Obstet Gynecol, 2006; 108(4); 924-29
17. Küçük T, Ertan K, Continuous oral or intramuscular medroxyprogesterone acetate versus the levonorgestrel releasing intrauterine system in the treatment of perimenopausal menorrhagia: A randomized, prospective, controlled clinical trial in female smokers: Clin Exp Obstet Gynecol, 2008; 35(1); 57-60
18. Dunphy BC, Goerzen J, Greene CA, A double-blind randomised study comparing danazol and medroxyprogesterone acetate in the management of menorrhagia: J Obstet Gynaecol, 1998; 18(6); 553-55
19. Zubiaur P, Ochoa D, Gálvez MÁ, Effect of polymorphisms in CYP2C9 and CYP2C19 on the disposition, safety and metabolism of progesterone administrated orally or vaginally: Adv Ther, 2019; 36(10); 2744-55
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387