24 October 2022: Animal Study
Effect of Antilogous Platelet-Rich Plasma on the Revascularization of Rabbit Prefabricated Flap
Ke Xie1ABCDEF, Meixia Huang1BC, Yan Zheng1B, Daiqing Chen1BC, Junling Hu1ABC, Jiansheng Zheng1ADFG*DOI: 10.12659/MSM.937718
Med Sci Monit 2022; 28:e937718
Abstract
BACKGROUND: We aimed to investigate the effect of the local application of autologous platelet-rich plasma (PRP) on the angiogenesis of the rabbit prefabricated flap.
MATERIAL AND METHODS: Femoral arteriovenous bundle prefabricated flaps were constructed on both sides of the abdomen of 20 New Zealand white rabbits and were randomly divided into experimental sides and control sides, with 40 sides in total. Autologous PRP was injected around the prefabricated femoral vascular bundle on the experimental side, and the same amount of saline was injected on the control side. After 14 days, stage II surgery was performed to lift the island flap with the implanted femoral vascular bundle at the tip of the preconfigured flap marker line and suture it in situ. Twenty rabbits were divided equally into 4 groups: group A, 7 days after stage I surgery; group B, 14 days after stage I surgery; group C, 7 days after stage II surgery; and group D, 14 days after stage II surgery. The survival of neovascularization and the flaps in the 4 groups were compared by microscopic observation, histology, and immunohistochemistry.
RESULTS: The density of neovascularization was much higher on the experimental side than on the control side in the 4 groups, and statistically significantly increased with time (P<0.05). Seven days after stage II surgery, flap survival was better on the experimental side than on the control side.
CONCLUSIONS: PRP effectively promoted the revascularization of a prefabricated flap, and vascular density increased with time.
Keywords: Endothelial Growth Factors, platelet-rich plasma, Surgical Flaps, Animals, Vascular Surgical Procedures
Background
The prefabricated flap is a method to transform any flap into an axial flap by transplanting a known vascular nerve bundle, thereby enhancing the function and application range of the flap. Since it is not necessary to consider the original blood supply of the flap, the selection range of the flap donor site is expanded, which is an important method for tissue repair in the clinical setting [1–4]. However, the survival core of the prefabricated flap is the degree of revascularization of transplanted blood vessels. Therefore, determining how to effectively promote the revascularization of the prefabricated flap is a key problem to be solved [5,6]. Studies have shown that platelet-rich plasma (PRP) is rich in a variety of growth factors that can promote random flap vascularization; however, the role of PRP in the prefabricated flap is rarely reported [7–9]. PRP has a high level of growth factor content, which is clinically used to promote bone and wound healing [10,11]. In this study, New Zealand white rabbits were used as research subjects. Through this experimental study of PRP on the vascularization of the prefabricated flap, we aimed to clarify the mechanism of PRP vascularization and lay a theoretical foundation for future clinical treatment.
Material and Methods
ANIMAL GROUPING:
This study was approved by the Experimental Animals Ethics Committee of Southeast Hospital Affiliated to Xiamen University (approval no. 2019-002-01) and was conducted in the Laboratory Animals and Experimental Research Center of Xiamen University. The project was part of the Fujian Natural Science Fund (no. 2019J01143).
Twenty male New Zealand white rabbits, weighing 2.0 to 2.5 kg, were provided by the Songlian Experimental Animal Farm, Songjiang District, Shanghai (license no. SCXK [Shanghai] 2019-0011). They were acclimated to the conditions of the animal laboratory for 1 week, including feeding. The room temperature was kept at 18°C to 22°C, and the rabbits were regularly ventilated and disinfected. The rabbits were randomly divided into 4 groups, with 5 rabbits in each group, as follows: group A, 7 days after stage I surgery; group B, 14 days after stage I surgery; group C, 7 days after stage II surgery; and group D, 14 days after stage II surgery.
MAIN REAGENTS AND INSTRUMENTS:
The following reagents and instruments were used: hematoxylin staining solution (Fuzhou Maixin Biotechnology Co., Ltd.); 0.5% eosin ethanol solution (Fuzhou Maixin Biotechnology); sodium citrate buffer (Fuzhou Maixin Biotechnology); 0.22 μm filter (Millipore, USA); PV-9004 immunohistochemical detection kit (Beijing Zhongshan Jinqiao Biotechnology Co., Ltd.) namely, mouse anti-human CD34 monoclonal antibody; goat anti-mouse IgG polyclonal antibody labeled with horseradish peroxidase (Fuzhou Maixin Biotech); HEAEUS CRYOFUHE 6000i low-temperature large capacity centrifuge (Beijing Dongxun Tiandi Medical Instrument Co., Ltd.) IXUS210 digital camera (Canon, Japan); WI93961 medical image analysis system (Beijing Beiruida Pharmaceutical Technology Co., Ltd.); and DM4B biological microscope (Leica, Germany).
PRP PREPARATION:
An amount of 12 mL of whole blood was collected from the central artery of the rabbit ear in a sterile centrifuge tube, and PRP was prepared by the Landbesberg method [12]. The PRP was mixed with 10% sterile CaCl2 solution 2: 1 and prepared as needed. The platelet concentration was measured by taking the activated PRP.
EXPERIMENTAL METHOD:
In the first stage of surgery (stage I), 0.2 mL/kg Lumianning and ketamine hydrochloride 1: 1 were mixed intramuscularly for anesthesia. Pre-purchased abdominal flaps were constructed on both sides of the abdomen of 20 rabbits, with one side being the experimental side and the other side being the control side. A 4×2-cm rectangular pre-purchased area was marked on each side of the abdomen of the rabbits in the experimental group; the short side was parallel to the inguinal ligament bundle (Figure 1), and the distal end was ligated and disconnected. No special treatment was required for the blood vessels in the flap area. A subcutaneous tunnel was made under the dermis of the central axis of the prefabricated skin flap area on both sides, and 0.2 mL of PRP was injected evenly at 4 points equally spaced around the tunnel in the experimental group. In the same way, the control group was injected with the same volume of normal saline. The stripped femoral vascular bundle was inverted and placed in the corresponding subcutaneous tunnel for fixation. Three days later, 0.2 mL of PRP was injected into 4 points of each tunnel wall in the experimental group.
In the second stage of surgery (stage II), 14 days after the stage I, the island flaps were lifted along the original prefabricated flap marking line with the implanted femoral vascular bundle as the pedicle in group C and group D and then sutured in situ.
DETECTION INDICATORS:
The platelet concentration of whole blood was detected by the automatic blood cell analyzer, and the PRP platelet concentration was detected by the manual sampling mode.
The flap survival rate was the ratio of the flap survival area to the total flap area. Seven days after stage II surgery (group C), the flap area was photographed, and the IPP6.0 image was used for image analysis to calculate the flap survival rate.
For histological detection, the implanted femoral vascular bundle was used as the axis, and a tissue size of 2×1 cm was taken from the center of the flap, fixed in 4% formaldehyde, and stained with conventional hematoxylin and eosin (H&E) and immunohistochemical staining (MaxVision method). Ten visual fields were randomly selected under a light microscope with 200× magnification to count the blood vessel sections and calculate the microvessel density.
STATISTICAL ANALYSIS:
SPSS16.0 statistical software was used for statistical analysis of test results. All index data were expressed as mean±standard deviation. The flap survival rate and neovascular density in each group were tested by the paired
Results
PLATELET CONCENTRATION:
The average concentration of platelets in whole blood was 316×109/L, the average concentration of PRP platelets was 1370×109/L, and the concentration ratio was about 4.3 times.
FLAP SURVIVAL RATE:
Seven days after stage II surgery (group C), the flap survival rates of the experimental side and the control side were 76.60±8.91 and 64.75±6.49%, respectively (t=6.579, P<0.05; Figure 2).
H&E STAINING:
In group A, there were new small blood vessels in the dermis of the experimental side, with thin blood vessel walls and endothelial cell proliferation, which were distributed in clusters, and a small amount of inflammatory cells were scattered around; there were a few new small blood vessels in the dermis of the control side, and new small blood vessels were formed. The cavity was irregular in shape, with a small amount of red blood cells in the cavity, accompanied by a small amount of endothelial cell proliferation and inflammatory cell infiltration around the cavity, which was more obvious than on the experimental side. In group B, small blood vessels, fibroblasts, and endothelial cells under the epidermis on the experimental side could be seen with hyperplasia, interstitial edema, and inflammatory cell infiltration; a few small blood vessels, fibroblasts, and endothelial cells proliferated under the epidermis on the control side, and interstitial hemorrhage could be seen, accompanied by a mild inflammatory reaction. In group C, the proliferation of small blood vessels on the experimental side was very active, especially in the dermis layer. The small blood vessels were densely distributed linearly and in clusters, and actively proliferating fibroblasts were seen. The interstitium showed edema, with a small amount of inflammatory cell infiltration. On the control side, small blood vessels and fibroblast proliferation could be seen in a scattered distribution, and interstitial hemorrhage could be seen with moderate infiltration of inflammatory cells. In group D, the proliferation of small blood vessels and fibroblasts on the experimental side was very active, and the small blood vessels tended to mature, with a small amount of scattered inflammatory cells seen around them. Although the proliferation of small blood vessels and fibroblasts was seen on the control side, the interstitial inflammatory reaction was more severe than that on the experimental side (Figure 3).
IMMUNOHISTOCHEMICAL STAINING:
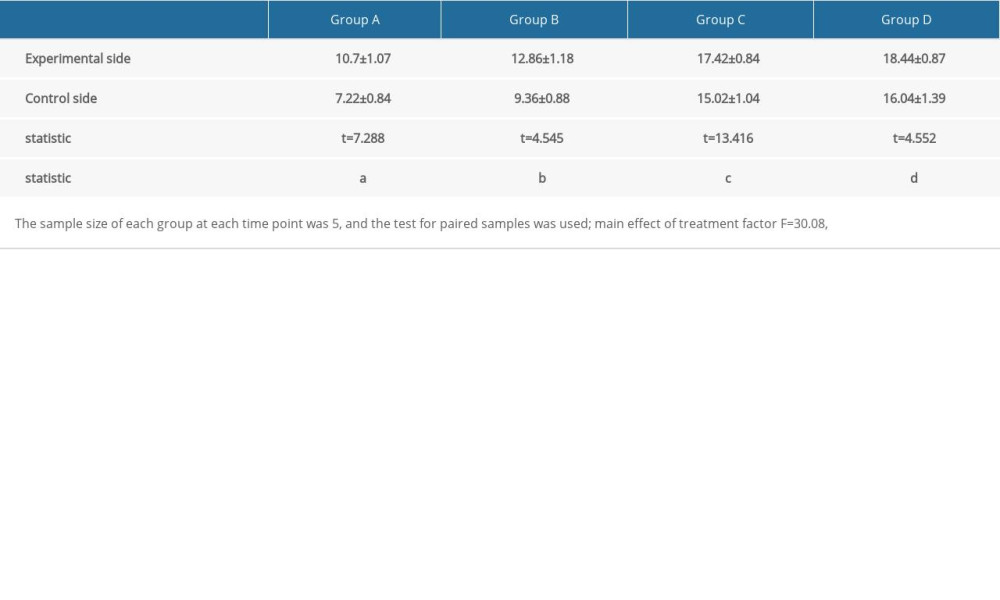
Vascular endothelial growth factor (VEGF) was mainly expressed in fibroblasts and neovascular endothelial cells. A large number of brown-yellow particles were deposited in the PRP side of flap tissue in each group; that is, a large amount of VEGF was expressed. Also, the positive rate of the experimental side in each group was higher than that of the control side (Figure 4). The expression levels of VEGF and CD34 in each group reflected the density of new blood vessels. In the 4 groups, the positive expression rates of VEGF and CD34 in the experimental side were higher than those in the control side, and the difference was significant (Table 1, Figure 5).
Discussion
The concept of the prefabricated flap was proposed by Yao in 1982 [13]. The prefabricated flap allows the existing blood vessels to be transplanted under the random flap, or the free skin graft is transplanted to the fascia, omentum, and other tissues with vascular bundles and rich blood supply. This creates an axial flap, and actively increases the blood flow of the flap to supply the flap with a new blood supply system [14]. It is not necessary to consider the original blood supply of the flap. Therefore, it breaks through the limitation of the distribution of blood vessels on the body surface, thus expanding the selection range of the donor site of the flap and prefabricating axial flap with a thin cortex and large flat area. This has gradually become an effective means for the treatment of complex trauma and high repair requirements of deformity or defect. The maturation process of the prefabricated flap is the process of flap vascular regeneration and blood supply reconstruction. The core problem is revascularization, and insufficient revascularization is the main reason for distal necrosis of the prefabricated flap. Therefore, determining how to make the prefabricated flap survive is one of the hotspots in plastic surgery research.
PRP is a first-generation platelet concentrate obtained by centrifugation of whole blood and contains a high-volume fraction of platelets. The platelets PRP contains are activated to release a variety of growth factors, including VEGF, which have multiple effects on wound repair and can promote wound healing and improve healing quality.
PRP activation releases a large number of growth factors, such as platelet-derived growth factor, VEGF, and fibroblast growth factor (FGF). Among them, VEGF has a strong pro-angiogenic effect and acts on endothelial cells by means of extravascular mesenchymal cells in a paracrine manner to generate blood vessels [15]. Fibrin gels provide a fibrous network for the migration of endothelial cells and fibroblasts, which facilitates angiogenesis [16,17]. Platelet-derived growth factor, epidermal growth factor, transforming growth factor, and FGF in PRP can upregulate VEGF expression by 3- to 20-fold [18]. Among them, basic FGF has the most synergistic effect with VEGF. Basic FGF is able to prevent premature cell death by upregulating telomere terminal transferase activity while promoting small artery formation. PRP participates in the process of hemostasis through fibrinogen, chemotactic inflammatory cells, mediating the trauma inflammatory response, and reducing infection to some extent. It provides a stable environment for postoperative preconstructed flap viability [19,20].
In the present experiment, we used our own left and right controls to construct a 2-sided abdominal femoral arteriovenous vascular bundle transfer flap model and observed the neovascular growth of the preconstructed flap on the experimental side and the control side at different periods. The results showed that PRP could accelerate the formation of the neovascular network and thus improve the survival rate of the preconstructed flap.
This study had certain limitations. We used a small sample size and a short postoperative follow-up period, and therefore, we could not well assess the variability of long-term sensory recovery. We will increase the sample size in subsequent research to lay a reliable experimental foundation for clinical treatment.
Conclusions
In summary, we successfully constructed the rabbit abdominal prefabricated flap model and showed that PRP could effectively promote the vascularization of the prefabricated flap, thereby promoting the flap survival. Moreover, with time, neovascularization increased, which provides the theoretical guidance for obtaining larger prefabricated axial flaps in clinical practice. Thus, we can greatly expand the use of the prefabricated flap and provide an effective choice for wound repair.
Figures
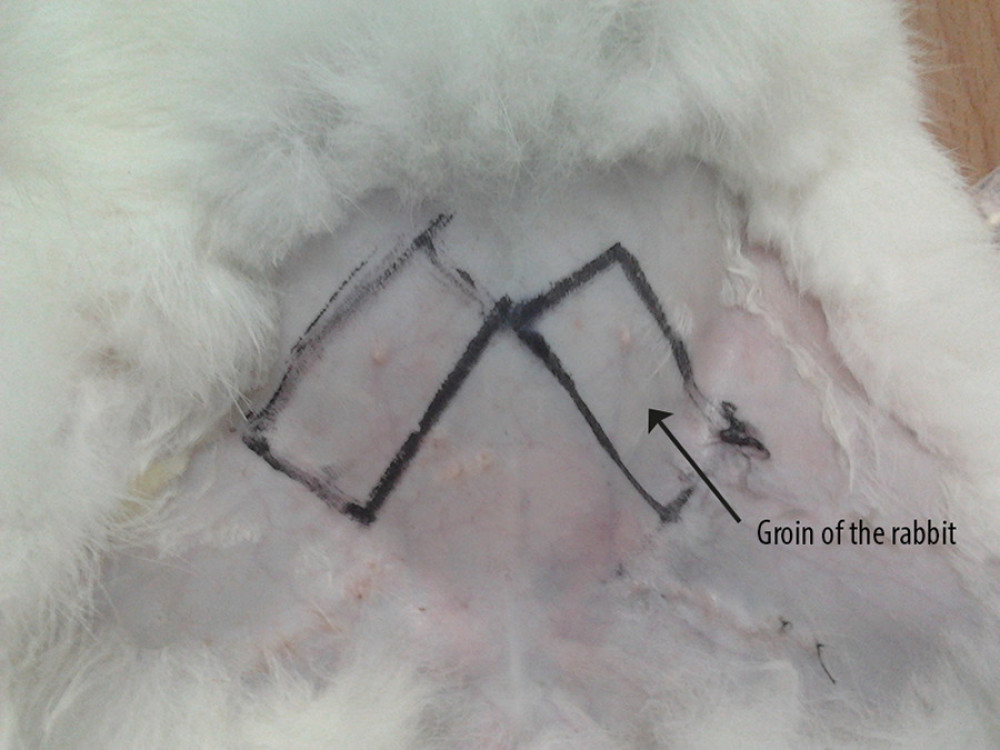 Figure 1. Preformed skin flap design.
Figure 1. Preformed skin flap design. 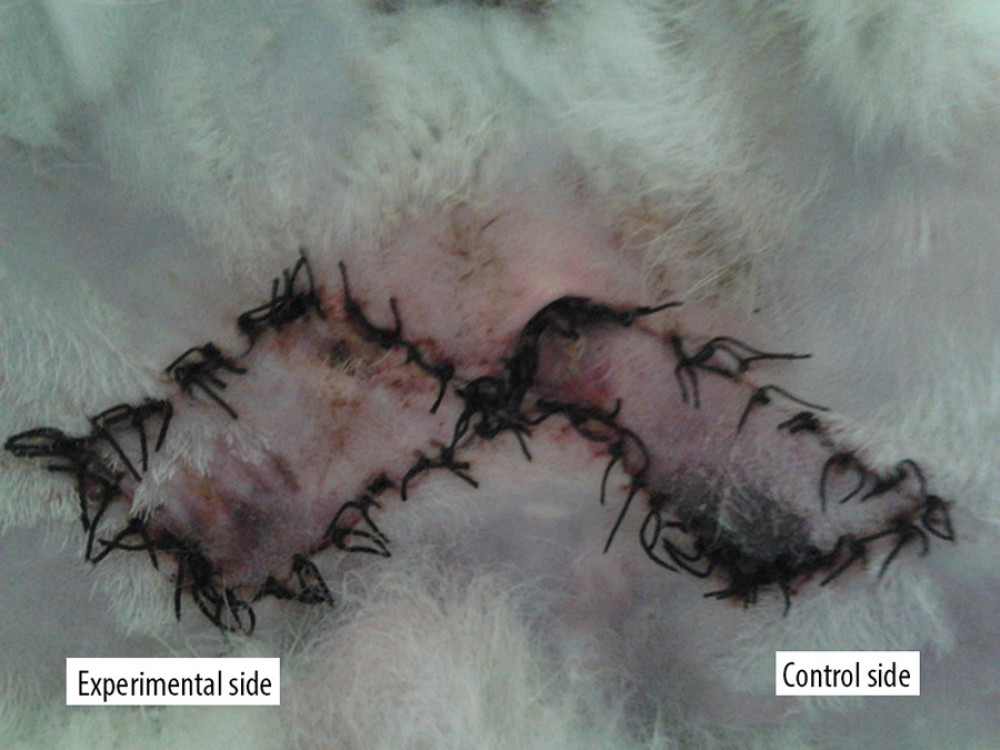 Figure 2. Survival of the 2 flaps at 7 days after surgical stage.
Figure 2. Survival of the 2 flaps at 7 days after surgical stage. 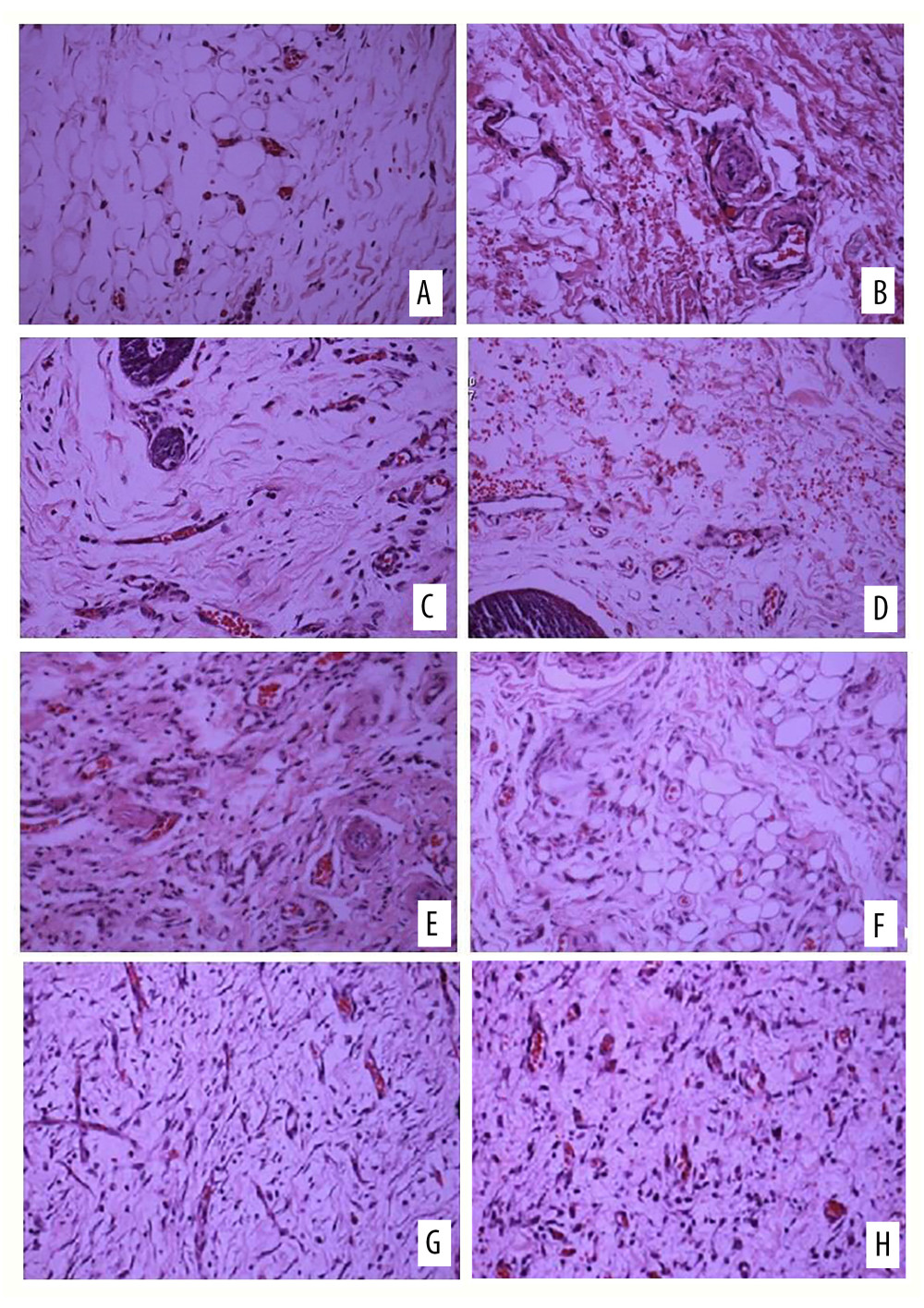 Figure 3. Hematoxylin and eosin (H&E) staining of (A) the experimental side and (B) the control side 7 days after surgery (group A). H&E staining of (C) the experimental side and (D) the control side 14 days after surgery (group B). H&E staining of (E) the experimental side and (F) the control side 7 days after surgery (group C). H&E staining of (G) the experimental side and (H) the control side 14 days after surgery (group D). All magnification is ×200.
Figure 3. Hematoxylin and eosin (H&E) staining of (A) the experimental side and (B) the control side 7 days after surgery (group A). H&E staining of (C) the experimental side and (D) the control side 14 days after surgery (group B). H&E staining of (E) the experimental side and (F) the control side 7 days after surgery (group C). H&E staining of (G) the experimental side and (H) the control side 14 days after surgery (group D). All magnification is ×200. 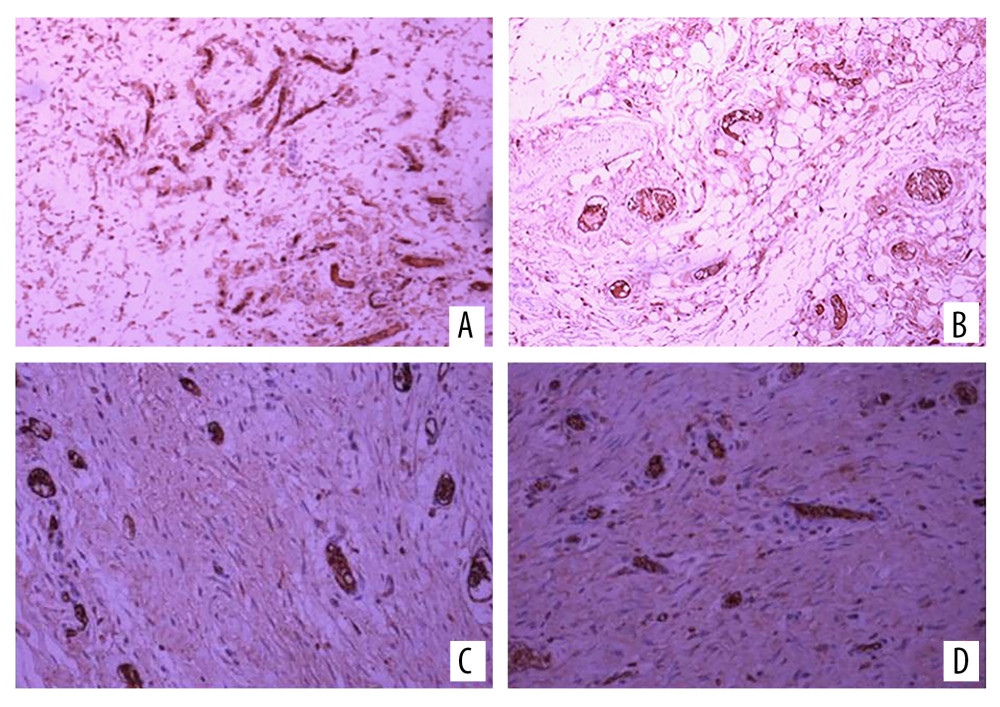 Figure 4. Vascular endothelial growth factor (VEGF) immunohistochemical staining on (A) the experimental side and (B) the control side 7 days after the second stage surgery (group C); VEGF immunohistochemical staining 14 days after the second stage surgery (group D) on (C) the experimental side and (D) the control side. All magnification is ×200.
Figure 4. Vascular endothelial growth factor (VEGF) immunohistochemical staining on (A) the experimental side and (B) the control side 7 days after the second stage surgery (group C); VEGF immunohistochemical staining 14 days after the second stage surgery (group D) on (C) the experimental side and (D) the control side. All magnification is ×200. 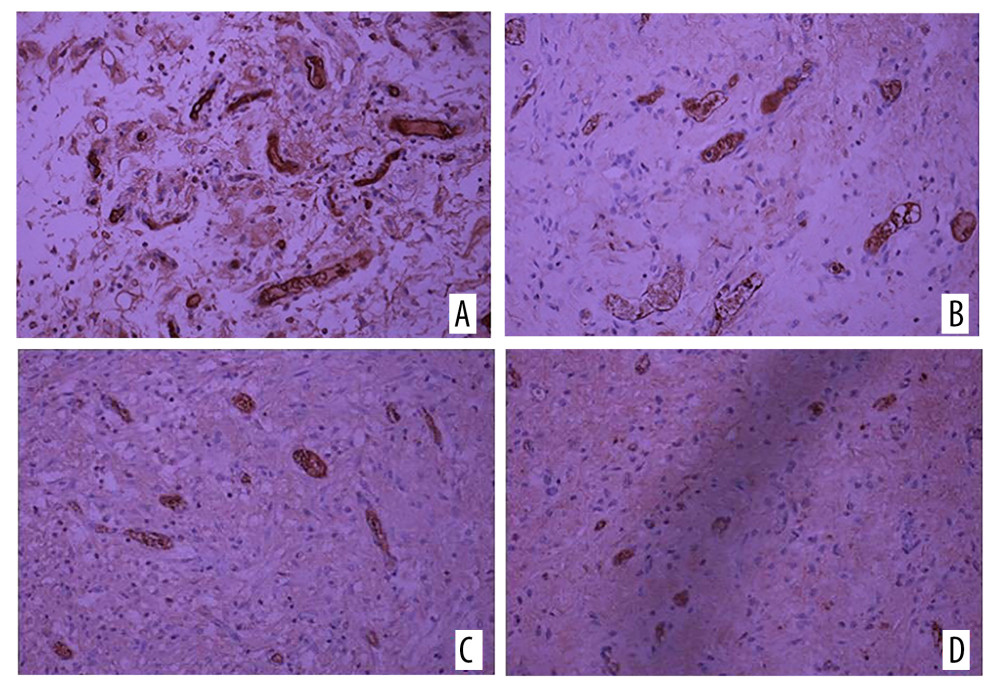 Figure 5. Immunohistochemical staining of CD34 on (A) the experimental side and (B) the control side 7 days after the second surgery stage (group C). CD34 immunohistochemical staining on (C) the experimental side and (D) the control side 14 days after the second surgery stage (group D). All magnification is ×200.
Figure 5. Immunohistochemical staining of CD34 on (A) the experimental side and (B) the control side 7 days after the second surgery stage (group C). CD34 immunohistochemical staining on (C) the experimental side and (D) the control side 14 days after the second surgery stage (group D). All magnification is ×200. References
1. Orgill DP, Ogawa R, Current methods of burn reconstruction: Plast Reconstr Surg, 2013; 131; e827-36
2. Daugherty THF, Pribaz JJ, Neumeister MW, The use of prefabricated flaps in burn reconstruction: Clin Plast Surg, 2017; 44(4); 813-21
3. Li K, Min P, Sadigh P, Prefabricated cervical skin flaps for hemi-facial resurfacing: elucidating the natural history of postoperative edema using indocyanine green: Lymphat Res Biol, 2018; 16(1); 100-8
4. Diab J, Leinkram D, Wykes J, Maxillofacial reconstruction with prefabricated prelaminated osseous free flaps: ANZ J Surg, 2021; 91(3); 430-38
5. Li B, Li H, Jin R, Desferrioxamine: A practical method for improving neovascularization of prefabricated flaps: Ann Plast Surg, 2015; 74(2); 252-55
6. Li W, Enomoto M, Ukegawa M, Subcutaneous injections of platelet-rich plasma into skin flaps modulate proangiogenic gene expression and improve survival rates: Plast Reconstr Surg, 2012; 129; 858-66
7. Wang B, Geng QH, Hu JL, Platelet-rich plasma reduces skin flap inflammatory cells infiltration and improves survival rates through induction of angiogenesis: An experiment in rabbits: J Plast Surg Hand Surg, 2016; 50; 239-45
8. Li W, Enomoto M, Ukegawa M, Subcutaneous injections of platelet-rich plasma into skin flaps modulate proangiogenic gene expression and improve survival rates: Plast Reconstr Surg, 2012; 129; 858-66
9. Sönmez TT, Vinogradov A, Zor F, The effect of platelet rich plasma on angio-genesis in ischemic flaps in VEGFR2-luc mice: Biomaterials, 2013; 34; 2674-82
10. Lee JW, Kwon OH, Kim TK, Platelet-rich plasma: Quantitative assessment of growth factor levels and comparative analysis of activated and inactivated groups: Arch Plast Surg, 2013; 40(5); 530
11. Akita S, Yoshimoto H, Akino K, Early experiences with stemcells in treating chronic wounds: Clin Plast Surg, 2012; 39(3); 281-92
12. Carter MJ, Fyling CP, Parnell LK, Use of platelet rich plasma gel on wound healing: A systematic review and meta-analysis: Eplasty, 2011; 11; e38
13. Yao ST, Microvascular transplantation of prefabricated free thigh flap: Plast Reconstr Surg, 1982; 69(3); 568
14. Orgill DP, Ogawa R, Current methods of burn reconstruction J1: Plast Reconstr Surg, 2013; 131(5); e827-36
15. Tille JC, Pepper MS, Mesenchymal cells potentiate vascular endothelial growth factor-induced angiogenesis in vitro: Exp Cell Res, 2002; 280; 179-91
16. Hom DB, Linzie BM, Huang TC, The healing effects of autologous platelet gel on acute human skin wounds: Arch Facial Plast Surg, 2007; 9(3); 174-83
17. Schade VL, Roukis TS, Use of platelet-rich plasma with split-thickness skin grafts in the high-risk patient: Foot Ankle Spec, 2008; 1(3); 155-59
18. Chakroborty D, Sarkar C, Kai L, Activation of Dopamine D 1, receptors in dermal fibroblasts restores vascular endothelial growth factor – a production by these cells and subsequent angiogenesis in diabetic cutaneous wound tissues: Am J Pathol, 2016; 186(9); 2262-70
19. Andia I, Maffulli N, Platelet-rich plasma for managing pain and inflammation in osteoarthritis: Nat Rev Rheumatol, 2013; 9(12); 721-30
20. Kim H-J, Yeom JS, Koh YG, Anti-inflammatory effect of platelet-rich plasma on nucleus pulposus cells with response of TNF-α and IL-1: J Orthop Res, 2014; 32(4); 551-56
Figures
 Figure 1. Preformed skin flap design.
Figure 1. Preformed skin flap design. Figure 2. Survival of the 2 flaps at 7 days after surgical stage.
Figure 2. Survival of the 2 flaps at 7 days after surgical stage. Figure 3. Hematoxylin and eosin (H&E) staining of (A) the experimental side and (B) the control side 7 days after surgery (group A). H&E staining of (C) the experimental side and (D) the control side 14 days after surgery (group B). H&E staining of (E) the experimental side and (F) the control side 7 days after surgery (group C). H&E staining of (G) the experimental side and (H) the control side 14 days after surgery (group D). All magnification is ×200.
Figure 3. Hematoxylin and eosin (H&E) staining of (A) the experimental side and (B) the control side 7 days after surgery (group A). H&E staining of (C) the experimental side and (D) the control side 14 days after surgery (group B). H&E staining of (E) the experimental side and (F) the control side 7 days after surgery (group C). H&E staining of (G) the experimental side and (H) the control side 14 days after surgery (group D). All magnification is ×200. Figure 4. Vascular endothelial growth factor (VEGF) immunohistochemical staining on (A) the experimental side and (B) the control side 7 days after the second stage surgery (group C); VEGF immunohistochemical staining 14 days after the second stage surgery (group D) on (C) the experimental side and (D) the control side. All magnification is ×200.
Figure 4. Vascular endothelial growth factor (VEGF) immunohistochemical staining on (A) the experimental side and (B) the control side 7 days after the second stage surgery (group C); VEGF immunohistochemical staining 14 days after the second stage surgery (group D) on (C) the experimental side and (D) the control side. All magnification is ×200. Figure 5. Immunohistochemical staining of CD34 on (A) the experimental side and (B) the control side 7 days after the second surgery stage (group C). CD34 immunohistochemical staining on (C) the experimental side and (D) the control side 14 days after the second surgery stage (group D). All magnification is ×200.
Figure 5. Immunohistochemical staining of CD34 on (A) the experimental side and (B) the control side 7 days after the second surgery stage (group C). CD34 immunohistochemical staining on (C) the experimental side and (D) the control side 14 days after the second surgery stage (group D). All magnification is ×200. In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952