22 September 2022: Clinical Research
Impact of Adherence with Disease-Modifying Therapies on All-Cause Mortality Rates Among Veterans with Multiple Sclerosis
Meheroz H. Rabadi12ABCDEF*, Kimberly Just1B, Chao XuDOI: 10.12659/MSM.938116
Med Sci Monit 2022; 28:e938116
Abstract
BACKGROUND: Adherence to disease-modifying therapies (DMTs) is essential for reducing multiple sclerosis (MS)-related relapses and disability. However, no known data exist regarding rates of adherence to DMTs and their impact on mortality. The present study aimed to determine the effect of adherence to DMTs on all-cause mortality in patients with MS in a real-world setting.
MATERIAL AND METHODS: We reviewed electronic records of 279 patients with MS and followed them longitudinally in our MS clinic between Januray1, 2000 and December 31, 2019. The inclusion criteria were complete electronic records along with documentation of initial and final functional outcome measures, including mortality. The exclusion criteria were incomplete electronic records and lack of documentation of initial and final functional outcome measures.
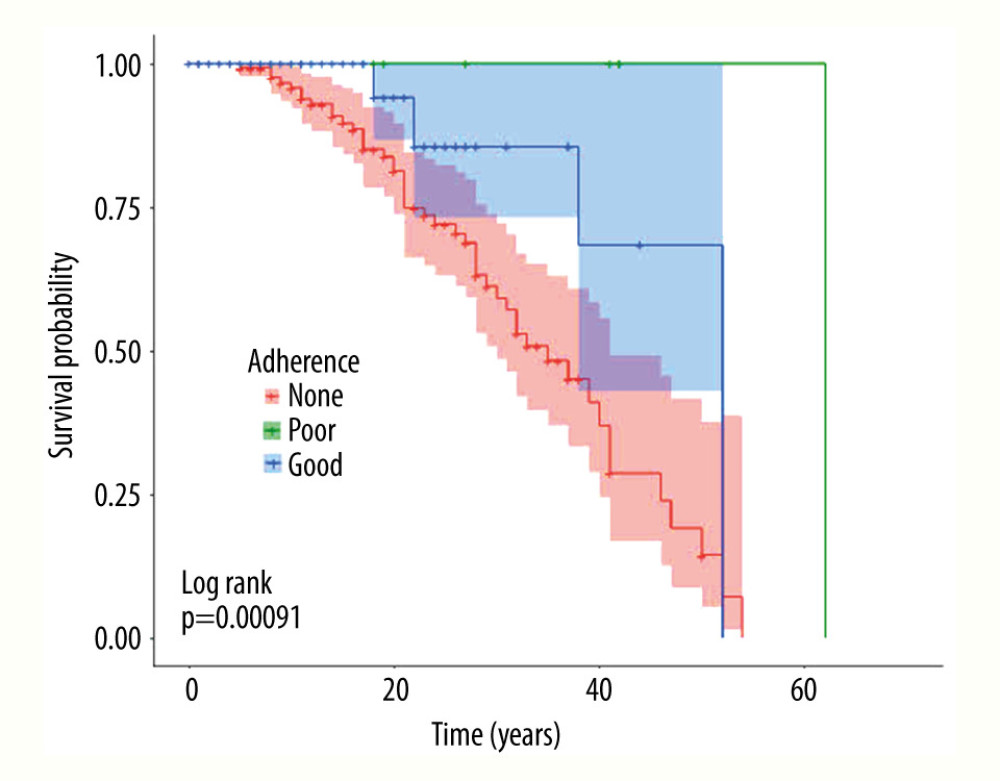
RESULTS: Of 279 patients with MS, 148 (53.0%) were non-adherent to any DMT medication(s). Of the 131 (47.0%) MS patients who were adherent, 13 (4.7%) had poor adherence and 118 (42.3%) had good adherence. More patients in the good-adherence group survived (94.9%) compared to the non-adherence group (66.9%, P<0.001). The odds of being alive were 12 times higher among those who adhered to their DMT compared to those who did not.
CONCLUSIONS: This study indicates that veterans who adhere to their DMTs are 12 times more likely to be alive than those who are non-adherent, even after adjusting for variables known to affect S-related mortality such as age at entry, MS type, MS duration, body mass index, and diabetes.
Keywords: Multiple Sclerosis, Veterans, medication adherence, Mortality, Humans, Recurrence
Background
Multiple sclerosis (MS) is a chronic inflammatory disease of the central nervous system that mainly affects young adults, with the highest incidence at ages 20–40 years, and it follows a variable course. Its etiology is multifactorial, with both genetic susceptibility and environmental exposure contributing to its pathogenesis [1]. MS remains an incurable disease, hence treatments designed to modify disease progression or alleviate symptoms using disease-modifying therapies (DMTs) are essential. Maintaining adherence to DMTs to help derive the maximum possible clinical benefit and reduce substantial disability is challenging [2].
Adherence describes the extent to which a patient acts in accordance with the timing, dosing, and frequency of medication prescribed by a physician who has prescribed a DMT. This study uses the term “adherence” rather than “compliance” because adherence better reflects the action required of the patient and avoids judgmental connotations associated with the term “noncompliance” [3]. Patients have found it challenging to maintain long-term adherence to therapies for chronic medical conditions [4]. Published estimates of adherence among patients with relapsing-remitting MS from clinical trials vary between 41% and 88% depending on the study’s definition of adherence [5–8]. Adherence rates reported in clinical trials for different injectable DMTs vary from 79% to 85% for once-weekly interferon beta-1a (IM IFNb-1a) to 49–78% for other injectable DMTs [4]. Poor adherence has been associated with worsening morbidity, increased health care cost, and increased mortality [9].
MS carries a high morbidity and shortens lifespan by 6–10 years despite advances in MS-specific treatment and in treatments for its co-morbidities [10,11]. Amezcua et al studied patients with MS in the United States from 1999 to 2015 and found that mortality increased with age in both sexes and in both non-Hispanic Whites and Blacks, peaking at 55–64 years for Blacks and 65–74 years for Whites, with White females being the most affected [12].
Our previous study of 226 veterans with MS reported a mortality of 14% at the end of the 15-year period. Patients with MS died prematurely, with a standardized mortality ratio (SMR) of 1.35 relative to the general (Oklahoma) population [13]. The main causes of death documented were MS disease (57% of cases), infection (43%), and cancer and respiratory failure (18% each). Similarly, in their study of patients with MS in British Columbia, Canada, Kingwell et al [14] found a high SMR of 2.71, with MS being the underlying cause in 50.4% cases, followed by infection (genitourinary, respiratory, and septicemia), respiratory diseases, and suicide. Thorman et al [15], in their study of patients with MS in Denmark between 1980 and 2005, found that MS-related mortality was higher in MS patients with stroke, Parkinson’s disease, cardiovascular disease, lung cancer, cancer, and diabetic co-morbidities.
Previous studies of adherence in patients with MS have covered relatively brief time periods [16], have been retrospective [17,18], or have focused on patients in clinical trials [19] whose experience differs from patients in many real-world clinical practices. Since our previous study in military veterans with MS followed in the MS clinic at the Oklahoma City Veterans Affairs Medical Center (VAMC) showed effective management of co-morbidities to reduce all-cause mortality over time [13], the aim of the present study was to compare the effect of adherence vs non-adherence to DMTs on all-cause mortality in veterans with MS. The results of the present study will help guide neurologists taking care of patients with MS to actively encourage adherence to DMTs to reduce MS-related mortality.
Material and Methods
PARTICIPANTS:
This study was approved by our local Institution Review Board, and they determined that the study was exempt from patient informed consent. In this observational study we reviewed the electronic records of 279 veterans who were diagnosed with MS (using the McDonald criteria) [20] and followed longitudinally in our MS clinic between Januray1, 2000 and December 31, 2019. The MS clinic has a structured approach in which all patients with MS on initial and yearly clinical evaluation undergo neuroimaging and blood tests for complete blood count for lymphocyte count, blood chemistry for renal and liver functions, vitamin D level [25-Hydroxy], and JVC antibody titers.
ELIGIBILITY CRITERIA:
The inclusion criteria were complete electronic records of veterans with MS who were regularly followed-up in our MS clinic with documentation of both initial and final functional outcome measures including mortality. Exclusion criteria were incomplete electronic records and lack of documentation of initial and final functional outcome measures plus mortality.
STUDY DESIGN: SINGLE-CENTER:
Data collected included demographic and clinical measures (age, gender, race, height, weight), MS status, age at MS onset, clinical MS subtype (relapsing-remitting [RR], secondary-progressive [SP], and primary-progressive [PP] MS) [21], duration of the disease, initial documented cognition (Mini-Mental Status Examination [MMSE]), presence of co-morbidities (hypertension, hyperlipidemia, diabetes mellitus, hypothyroidism, current smoking habit, alcohol use), and presence of MS-related complications (fatigue, depression). These comorbidities are the most common causes of disability and death in the general US population [22]. MS-related severity and disability was measured using the Expanded Disability Severity (EDSS) [23] and Total Function Independence Measures (TFIM) scales [24]. This study conforms to all STROBE guidelines and reports the required information accordingly (see Supplementary Checklist).
INTERVENTION(S) OR TREATMENT:
We classified adherence to DMT use as (1) non-adherence, (2) poor adherence, and (3) good adherence. Investigators made the classification after verbally inquiring about each individual’s DMT use during face-to-face interactions and subjectively examining injection sites as indicated during follow-up visits. Information gathered from interviews and injection site examination was cross-checked with data on prescription refills in electronic medical records from our facility’s pharmacy, which is the sole dispenser of DMT medications for our veterans with MS. Switching between different DMTs (oral, injections, and infusions) was allowed.
We judged veterans to be non-adherent if they initially took but then stopped taking the prescribed DMTs or if they refused to take the prescribed DMTs to begin with due to adverse effects, because they perceived lack of effect on disease progression, or because they believed their disease was stable or improving. Veterans were categorized as demonstrating poor adherence if they took their prescribed DMT infrequently either because they were forgetful (cognitive impairment) or depressed. We considered veterans to show good adherence if they took their DMT regularly as prescribed.
We validated their categorization of adherence by calculating the medication possession ratio (MPR) using pharmacy refill data. The MPR was calculated as the sum of the day’s supply obtained between the first and last pharmacy refill divided by the total number of days over a 1-year period. This clinical definition was supported by an MPR cut score of <0.8 equals non-adherence, 0.8–0.9 equals poor adherence, and greater than 0.9–1.0 equals good adherence [25].
ASSESSMENT OF MORTALITY:
Deaths were ascertained for all patients with MS who were followed-up in our clinic at any time over the 20-year study period. Causes, dates, and locations of death (home, long-term care facility, or hospital) were obtained from information provided to our clinic coordinator or social worker by the patient’s care provider or next of kin, and from death certificates filed with the Oklahoma State Department of Health.
STATISTICAL ANALYSIS:
Statistical significance was defined as a
Results
Of the 279 patients with MS, 148 (53.0%) were non-adherent to any DMT medications, 131 (47.0%) were adherent, 13 (4.7%) had poor adherence, and 118 (42.3%) had good adherence. Of the 279 patients with MS who were followed over the 20-year study period, 223 were alive (79.9%) and 56 were dead (20.1%). Those who had died and those who remained alive differed at the time of initial presentation in terms of age of entry in years, duration of MS in years, MS type, MS-related impairment, DMT use, EDSS and TFIM scores, hypertension, DM, and BMI (Table 1).
Table 2 compares the primary outcome – mortality – among the 3 groups defined by adherence to DMTs. Of the 148 non-adherent patients, 99 (66.9%) were alive and 49 (33.1%) were dead compared to 112 of the 118 (94.9%) adherent patients who were alive and 6 (5.1%) who were dead (
A Cox proportional hazards model was fit for the groups with good adherence and non-adherence. The 13 patients with poor adherence were excluded from the analysis. The model included covariates known to affect MS-related survival, which included age at entry, MS type and duration, BMI, and DM. After adjustment, the risk of death in the non-adherence group was 7.5 times (95% CI [2.71, 20.5],
When we performed a subset analysis of primary outcome measure between the groups only for patients with RRMS, there were 44 non-adherent patients; 37 (84.1%) were alive and 7 (15.9%) were dead compared to 60 of the 61 (98.4%) adherent patients who were alive and 1 (1.6%) who died. All 10 patients who demonstrated poor adherence survived. The Cochran-Armitage test indicated a significant association between the trend of non-/poor/good adherence and mortality (
Discussion
The main findings of this study in veterans with MS who were regularly followed (longitudinally) over our clinic’s 20-year history were as follows: (1) 53.0% of veterans with MS were non-adherent to their DMT medications; (2) mortality among the veterans was 20.1%; (3) veterans who were adherent to their DMT medications were 12 times more likely to be alive than veterans who were non-adherent to their DMT medications; (4) veterans with MS were more likely to be alive if they began attending the clinic when younger (<30 years), were men, whose duration of MS was less than 10 years, whose diagnosis was RRMS, who were not cognitively impaired, whose MS was less severe (initial EDSS score 0–3), and who initially had less functional disability (higher TFIM score).
The reasons why 53.0% of veterans were non-adherent to DMT medication included: perceived lack of beneficial effect on the disease; adverse effects of medication, especially injection site reactions and depression; and complaints of having to take the DMT over a prolonged period. Of the veterans who took their DMT as prescribed, 9.9% (13/131) had poor adherence due to forgetfulness or depression. Other investigators have found similar rates of non-adherence (30–50%) among adults with chronic illnesses such as diabetes or hypertension [26]. Over the last 15 years, although several studies have been conducted to improve rates of medication adherence, the rate of medication non-adherence has not appreciably changed [26].
Although there is no information in the literature regarding adherence to DMTs and mortality in a clinical setting, Goodin et al [27] in their cohort of 372 patients who participated in the pivotal randomized clinical trial (RCT) of interferon beta (IFNβ)-1b on all-cause mortality over a 21-year period showed patients assigned to IFNβ-1b 250 μg had a 46% reduction in all-cause mortality compared with placebo (
The present study had several limitations. First, because 80% of the participants were veterans who were men and non-Hispanic White, its generalizability to the general population of patients with MS is limited. Second, the small sample size limited the statistical power. Third, this was an observational study in a real-world clinic setting and assignment to the groups was not by deliberate randomization.
Despite these limitations, this study has certain strengths. All the patients in the electronic database of the MS registry were included. Thus, the data were standardized and complete, and reflected regular follow-up at 4, 8, and 12 months. The data collected over a 20-year period included hospital pharmacy prescription and dispensing data, which permitted validation of adherence to DMTs. These factors limit the potential for selection and recall biases [29] and reflect real-world clinical practice.
Based on the study results, we hope to add a clinical pharmacist to our MS team to co-manage (including education, increased frequency of disease monitoring via telephone or in-person follow-up visits, and refill reminders) in addition to our present ongoing effort of patient and family (caregiver) education (eg, recurrent and personalized telephone counseling sessions) and medication regimen management (to reduce the frequency of taking the medication on a daily to weekly basis) and further help improve the adherence rate.
Conclusions
This study indicates that veterans who adhere to their DMTs are 12 times more likely to be alive than non-adherent groups, even after adjusting for age at entry, MS type, MS duration, BMI, and DM, which are variables known to affect MS-related mortality.
Tables
Table 1. Demographics based on multiple sclerosis survival (dichotomized).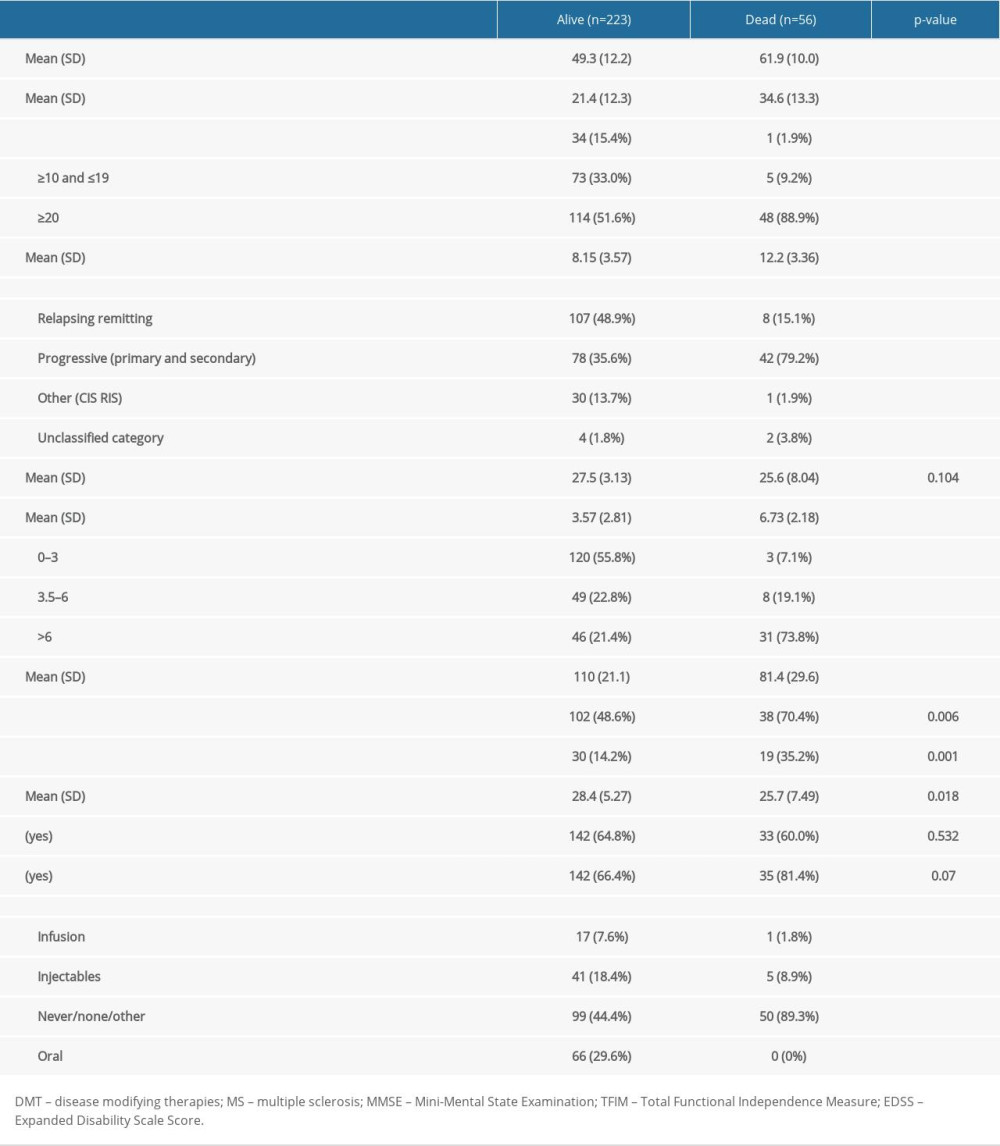 Table 2. Comparison of mortality in groups defined by adherence to adherence to disease-modifying therapies.
Table 2. Comparison of mortality in groups defined by adherence to adherence to disease-modifying therapies.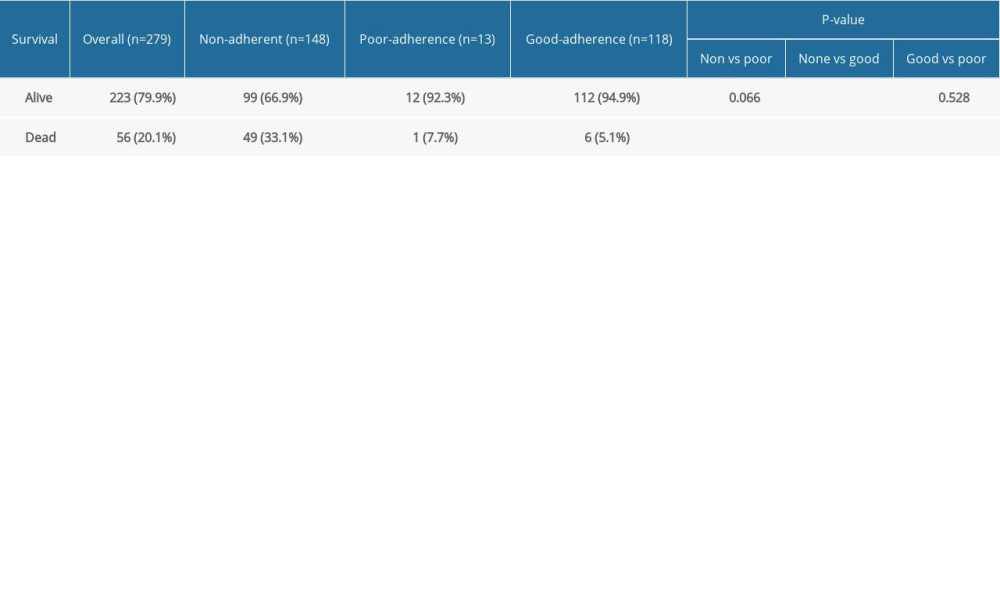 Table 3. Comparison of mortality in groups defined by adherence to disease-modifying therapies only for patients with relapsing-remitting multiple sclerosis.
Table 3. Comparison of mortality in groups defined by adherence to disease-modifying therapies only for patients with relapsing-remitting multiple sclerosis.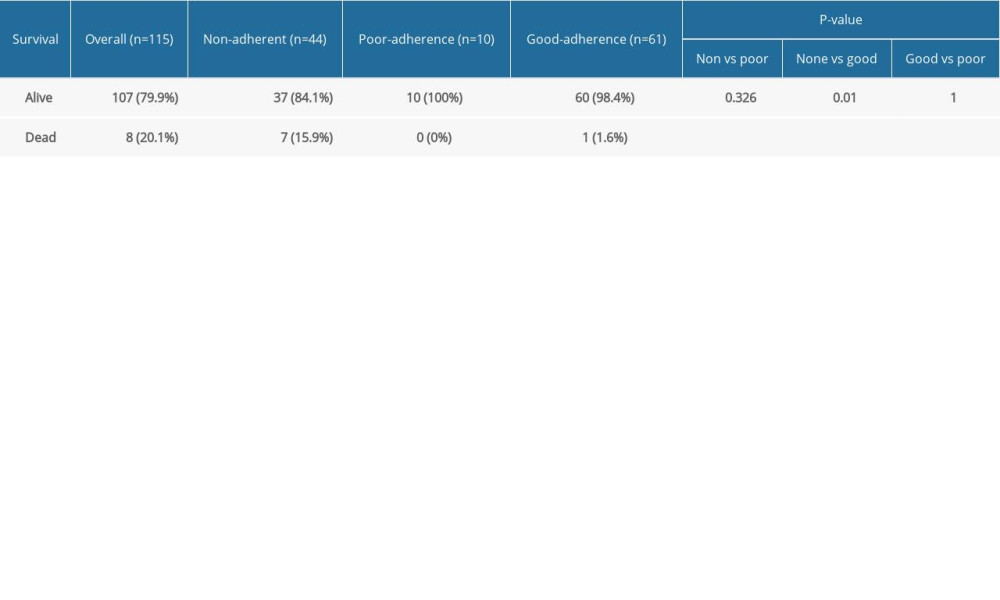
References
1. Dobson R, Giovannoni G, Multiple sclerosis – a review: Eur J Neurol, 2019; 26(1); 27-40
2. Thompson AJ, Baranzini SE, Geurts J, Multiple sclerosis: Lancet, 2018; 391(10130); 1622-36
3. Aronson JK, Compliance, concordance, adherence: Br J Clin Pharmacol, 2007; 63(4); 383-84
4. World Health Organization: Adherence to long-term therapies: evidence for action, 2003 Available from: http://www.who.int/chp/knowledge/publications/adherence_report_fin.pdf?ua=1
5. Treadaway K, Cutter G, Salter A, Factors that influence adherence with disease-modifying therapy in MS: J Neurol, 2009; 256(4); 568-76
6. Devonshire V, Lapierre Y, Macdonell R, The Global Adherence Project (GAP): A multicenter observational study on adherence to disease-modifying therapies in patients with relapsing-remitting multiple sclerosis: Eur J Neurol, 2011; 18(1); 69-77
7. Lafata JE, Cerghet M, Dobie E, Measuring adherence and persistence to disease-modifying agents among patients with relapsing remitting multiple sclerosis: J Am Pharm Assoc, 2008; 48(6); 752-57
8. Menzin J, Caon C, Nichols C, Narrative review of the literature on adherence to disease-modifying therapies among patients with multiple sclerosis: J Manag Care Pharm, 2013; 19(1 Suppl A); S24-40
9. Osterberg L, Blaschke T, Adherence to medication: N Engl J Med, 2005; 353; 487-97
10. Koch-Henriksen N, Laursen B, Stenager E, Magyari M, Excess mortality among patients with multiple sclerosis in Denmark has dropped significantly over the past six decades: A population based study: J Neurol Neurosurg Psychiatry, 2017; 88(8); 626-31
11. Lunde HMB, Assmus J, Myhr KM, Bø L, Grytten N, Survival and cause of death in multiple sclerosis: A 60-year longitudinal population study: J Neurol Neurosurg Psychiatry, 2017; 88(8); 621-25
12. Amezcua L, Rivas E, Joseph S, Multiple sclerosis mortality by race/ethnicity, age, sex, and time period in the United States, 1999–2015: Neuroepidemiology, 2018; 50(1–2); 35-40
13. Rabadi MH, Aston CE, Predictors of mortality in veterans with multiple sclerosis in an outpatient clinic setting: Int J MS Care, 2017; 19(5); 265-73
14. Kingwell E, Zhu F, Evans C, Causes that contribute to the excess mortality risk in multiple sclerosis: A population-based study: Neuroepidemiology, 2019; 18; 1-9
15. Thormann A, Sørensen PS, Koch-Henriksen N, Comorbidity in multiple sclerosis is associated with diagnostic delays and increased mortality: Neurology, 2017; 89(16); 1668-75
16. Katsarava Z, Ehlken B, Limmroth VC.A.R.E. Study Group, Adherence and cost in multiple sclerosis patients treated with IM IFN beta-1a: Impact of the CARE patient management program: BMC Neurol, 2015; 15; 170
17. Zhang T, Kingwell E, Zhu F, Effect of adherence to the first-generation injectable immunomodulatory drugs on disability accumulation in multiple sclerosis: A longitudinal cohort study: BMJ Open, 2017; 7(9); e018612
18. Jernas Ł, Wencel J, Wiak A: PLoS One, 2016; 11(10); e0157950
19. Rudge P, Are clinical trials of therapeutic agents for MS long enough?: Lancet, 1999; 353; 1033-34
20. McDonald WI, Compston A, Edan G, Recommended diagnostic criteria for multiple sclerosis: Guidelines from the International Panel on the diagnosis of multiple sclerosis: Ann Neurol, 2001; 50; 121-27
21. Lublin FD, Reingold SC, Defining the clinical course of multiple sclerosis: Results of an international survey. National Multiple Sclerosis Society (USA) Advisory Committee on Clinical Trials of New Agents in Multiple Sclerosis: Neurology, 1996; 46; 907-11
22. Murray CJ, Lopez AD, Evidence-based health policy: Lessons from the Global Burden of Disease Study: Science, 1996; 274; 740-43
23. Kurtzke JF, Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS): Neurology, 1983; 33; 1444-52
24. Granger CV, The emerging science of functional assessment: Our tool for outcomes analysis: Arch Phys Med Rehabil, 1998; 79; 235-40
25. Sikka R, Xia F, Aubert RE, Estimating medication persistency using administrative claims data: Am J Manag Care, 2005; 11(7); 449-57
26. Briesacher BA, Andrade SE, Fouayzi H, Chan KA, Comparison of drug adherence rates among patients with seven different medical conditions: Pharmacotherapy, 2008; 28(4); 437-43
27. Goodin DS, Reder AT, Ebers GC, Survival in MS: A randomized cohort study 21 years after the start of the pivotal IFNβ-1b trial: Neurology, 2012; 78(17); 1315-22
28. Kingwell E, Leray E, Zhu F, Multiple sclerosis: Effect of beta interferon treatment on survival: Brain, 2019; 142(5); 1324-33
29. Bruce JM, Hancock LM, Lynch SG, Objective adherence monitoring in multiple sclerosis: Initial validation and association with self-report: Mult Scler, 2010; 16(1); 112-20
Tables
 Table 1. Demographics based on multiple sclerosis survival (dichotomized).
Table 1. Demographics based on multiple sclerosis survival (dichotomized). Table 2. Comparison of mortality in groups defined by adherence to adherence to disease-modifying therapies.
Table 2. Comparison of mortality in groups defined by adherence to adherence to disease-modifying therapies. Table 3. Comparison of mortality in groups defined by adherence to disease-modifying therapies only for patients with relapsing-remitting multiple sclerosis.
Table 3. Comparison of mortality in groups defined by adherence to disease-modifying therapies only for patients with relapsing-remitting multiple sclerosis. Table 1. Demographics based on multiple sclerosis survival (dichotomized).
Table 1. Demographics based on multiple sclerosis survival (dichotomized). Table 2. Comparison of mortality in groups defined by adherence to adherence to disease-modifying therapies.
Table 2. Comparison of mortality in groups defined by adherence to adherence to disease-modifying therapies. Table 3. Comparison of mortality in groups defined by adherence to disease-modifying therapies only for patients with relapsing-remitting multiple sclerosis.
Table 3. Comparison of mortality in groups defined by adherence to disease-modifying therapies only for patients with relapsing-remitting multiple sclerosis. In Press
07 Mar 2024 : Clinical Research
Knowledge of and Attitudes Toward Clinical Trials: A Questionnaire-Based Study of 179 Male Third- and Fourt...Med Sci Monit In Press; DOI: 10.12659/MSM.943468
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952