03 December 2022: Clinical Research
Comparison of the Effectiveness of Transnasal Humidified Rapid Insufflation Ventilator Exchange (THRIVE) with Facemask Pre-Oxygenation in 40 Patients ≥65 Years of Age Undergoing General Anaesthesia During Gastrointestinal Surgery for Intestinal Obstruction
Wanling Wang1AE, Wenwen Zhang1ABCE, Yu Lu1C, Yajie Xu1BEF, Yong Zhang1CF, Hongwei Shi1BF, Xiaoliang Wang1AE*DOI: 10.12659/MSM.938168
Med Sci Monit 2022; 28:e938168
Abstract
BACKGROUND: In this study, we aimed to compare the effectiveness of transnasal humidified rapid insufflation ventilator exchange (THRIVE) with facemask pre-oxygenation in 40 patients ≥65 years of age undergoing general anesthesia during gastrointestinal surgery for intestinal obstruction.
MATERIAL AND METHODS: Patients with gastrointestinal obstruction were randomized to either a facemask group (group M, n=20) or THRIVE group (group T, n=20). During pre-oxygenation, the 2 groups used a facemask (100% oxygen, 6 L/min) and THRIVE (100% oxygen, 40 L/min) to supply oxygen, respectively. Induction of anesthesia was performed in both groups using facemasks and without mechanical or assisted ventilation. The intubation occurred after myorelaxant action began. When the peripheral oxygen saturation (SpO₂) dropped below 95%, or 480 s after administration of muscle relaxants, mechanical ventilation was initiated immediately. The primary outcome was arterial partial pressure of oxygen (PaO₂) at 5 min after pre-oxygenation. A secondary outcome was time to SpO₂ of 95% during apnea, with a cut-off time of 480 s.
RESULTS: PaO₂ at 5 min after pre-oxygenation was (261.5±30.9) mmHg for group M and (446.1±84.4) mmHg for group T (P<0.001). Based on survival analysis, the median time-to-event in group T was 480 s (95% CI 415.7 s – upper limit unknown) and 240 s (95% CI 225.9-254.1 s) in group M (P<0.001).
CONCLUSIONS: In elderly patients undergoing rapid sequence induction, pre-oxygenation with THRIVE could improve oxygenation and extend safe apnea time, compared with facemask pre-oxygenation.
Keywords: Hyperbaric Oxygenation, Rapid Sequence Induction and Intubation, Aged, Humans, Masks, Insufflation, Digestive System Surgical Procedures, Apnea, Ventilators, Mechanical, Intestinal Obstruction, Anesthesia, General, Oxygen
Background
Hypoxia during the rapid sequence induction (RSI) of anesthesia is still an important issue that can considerably impact patient outcomes [1]. Normally, pre-oxygenation with 100% oxygen before intubation is used to denitrogenate, increase the oxygen reserve, and extend safe apnea time [2]. However, it is common for patients with gastrointestinal obstruction to have increased intra-abdominal pressure, diaphragmatic muscle activity, and decreased abdominal breathing [3]. Most of these patients are in an emergency situation and usually have a full stomach before surgery. The nasogastric tube and gastrointestinal tube are typically placed before surgery for gastrointestinal decompression or enteral nutrition, which prevents the facemask from fitting tightly in the pre-oxygenation stage. In addition, elderly patients with insufficient oxygen reserves are more likely to have hypoxia [4].
Transnasal humidified rapid-insufflation ventilatory exchange (THRIVE) is a new high-flow ventilation method that delivers warmed and humidified high-flow oxygen through a nasal cannula and is increasingly used in anesthetic practice and complex airway management [5]. Oxygen flow rates of up to 70 L/min can reduce anatomical dead space in the upper airway and improve end-expiratory compliance for ventilation and oxygenation [6]. Many studies have shown that THRIVE provides better oxygenation during induction of general anesthesia and can prolong safe apnea time by up to 17 min [7].
In 2020, Hua et al found that THRIVE extended the apnea time and provided better pre-oxygenation in elderly patients under general anesthesia [6]. However, they focused on using THRIVE during the peri-intubation period, including the phase of pre-oxygenation and apneic oxygenation. Because oxygen is provided continuously until intubation, this can raise intragastric pressure, possibly causing reflux or aspiration in patients with gastrointestinal obstruction. No studies have looked at the use of THRIVE alone during pre-oxygenation with RSI. Therefore, in this study, we aimed to compare the effectiveness of THRIVE with that of facemask pre-oxygenation in 40 patients ≥65 years of age undergoing general anaesthesia during gastrointestinal surgery for intestinal obstruction.
Material and Methods
STUDY DESIGN:
This single-blind and prospective trial was approved by the Ethics Committee of Nanjing First Hospital (KY20201102-04) and registered with the Chinese Clinical Trial Center (ChiCTR2100050184) before patient enrollment. All patients signed an informed consent form.
PATIENTS:
Patients with gastrointestinal obstruction requiring RSI of anesthesia, ≥65 year of age, with American Society of Anesthesiologists classification I–III, Mallampati score I–II, and correctly cooperating with the use of THRIVE after explanation, were selected. The gastrointestinal obstructions were all diagnosed by surgeons, based on computed tomography. Exclusion criteria included the following: anticipating difficult airways, body mass index (BMI) >30 kg/m2, severe systemic disease (neurological, cardiovascular, pulmonary, hepatic, or renal disorders), epistaxis and nasopharyngeal abnormalities, and moderate or higher anemia.
RANDOMIZATION AND BLINDING:
The research equipment included THRIVE (Optiflow THRIVE, Fisher & Paykel Healthcare, Auckland, New Zealand) and facemasks (Zhejiang SuJia Medical Device Co., Ltd., Zhejiang, China).
Using computer randomization software (SPSS 24.0), each patient was randomly categorized into the facemask group (group M) or THRIVE group (group T) in a 1: 1 ratio. The grouping results were secured in a sealed opaque envelope that was opened only by the researchers before anesthesia induction. A blinded research team member conducted postoperative interviews with the patients. All patients, preoperative and postoperative follow-up assessors, and statisticians were blinded to group allocation.
INTERVENTION:
Patients were decompressed through a nasal gastric tube before surgery. Negative pressure suction devices were prepared, as well as routine monitoring. The patients were placed supine with the head elevated approximately 25° to 30° [12]. In addition to routine monitoring, each patient underwent invasive blood pressure monitoring. Group M underwent pre-oxygenation via a facemask with a fraction of inspiration oxygen (FiO2) of 100% at 6 L/min and opened adjustable pressure limiting valve. The THRIVE device was heated and humidified 5 min before use, and patients in group T were instructed to use THRIVE oxygen with a tightly closed mouth, with a FiO2 of 100% at 40 L/min. Induction of anesthesia after 5 min of pre-oxygenation consisted of intravenous midazolam 0.02 to 0.05 mg/kg, propofol 1 to 2 mg/kg, rocuronium bromide 0.6 mg/kg, and remifentanil 1 to 2 ug/kg. When induction started in group T, THRIVE was removed, and the facemask was put with oxygenation as in group M. During the induction period, the upper airways of both groups were kept open with 2-handed airway maneuvers and breathing without using any pressure-assisted techniques. A video laryngoscope was inserted within the 60 s after rocuronium bromide was administered. The tracheal cuff was inflated immediately after successful intubation in both groups, and no mechanical ventilation was performed in either group. When the peripheral oxygen saturation (SpO2) dropped to 95% or the safe apnea time (the time between the start of rocuronium bromide injection and the drop of SpO2 to 95%) reached 480 s, mechanical ventilation was started.
In cases of severe reflux, the patients were placed in the Trendelenburg position with the head tilted to the side, the oropharynx was suctioned and cleared, and an emergency endotracheal intubation was performed. Intubation was stopped when SpO2 was greather than 90% during intubation, and oxygenation was maintained by manually ventilating (tidal volumes of 4–6 mL/kg, pressure-limiting valve set at 20 cmH2O) until SpO2 reached 98%. The 2015 Difficult Airway Society guidelines for unanticipated difficult endotracheal intubation in adults were used when the patient proved difficult for endotracheal intubation [13]. Patients with the above 3 situations were excluded from the study.
OUTCOMES AND DATA COLLECTION:
During this study, we defined different time points as follows: upon entering the operating room while breathing room air (T0), after 5 min of pre-oxygenation (T1), after successful endotracheal intubation (T2), and safe apnea time up to 480 s or SpO2 down to 95% (T3).
The primary outcome was PaO2 at T1. Secondary outcomes were the number of patients with safe apnea time up to 480 s, safe apnea time, re-oxygenation time, PaO2, arterial partial pressure of carbon dioxide (PaCO2), heart rate, and mean arterial pressure at each time point, intubation time, adverse events after induction of anesthesia, and complications related to THRIVE ventilation.
Apnea time was defined as the time from the start of rocuronium bromide injection until SpO2 dropped to 95%. For patient safety, the maximum duration of the apneic period has been set at 480 s [6]. When the patient’s SpO2 level did not drop to 95% within 480 s, it was finally recorded as 480 s.
Re-oxygenation time was defined as the time from the beginning of mechanical ventilation until SpO2 reached 98%. When SpO2 ≥98% was obtained at the end of the apnea, the re-oxygenation time was recorded as 0.
The intubation time was defined as the time from video laryngoscope placement into the mouth to passing a tracheal tube through the vocal cords.
SAMPLE SIZE ESTIMATION:
A pilot study with 4 patients undergoing facemask pre-oxygenation was conducted to estimate the sample size for the study. With facemask pre-oxygenation, these patients’ mean PaO2 was 305 mmHg, with an average standard deviation of 80 mmHg. The calculation of the sample size used a mean PaO2 of 400 mmHg to achieve an adequate elongation of safe apnea margin with the 2 devices [14]. Based on mean PaO2 values of 300 and 400 mmHg with a standard deviation of 80 mmHg, the significance criterion was set at α=0.05 (2-sided), power=90%, and in PASS11.0 software, a sample size of 16 was calculated for each group. Considering the data loss, the final sample size was 20 patients per group.
STATISTICAL ANALYSIS:
SPSS software (version 24.0, IBM Corp, Armonk, NY, USA) was used for the statistical analysis. The Shapiro-Wilk test was used to determine the normality of the data. The variance was tested for homogeneity with Levene’s test. Means ± standard deviations were used to represent normally distributed data. The
A secondary result was time to SpO2 of 95% during apnea, with a cut-off time of 480 s. A maximum apneic period of 480 s was set to ensure patient safety. SpO2 levels were initially documented as 480 s if they did not drop to 95% before 480 s. Since there were many patients whose SpO2 fell below 95% within 480 s in the THRIVE group, the outcome was analyzed as a time-to-event analysis (survival framework) that censored a patient if their SpO2 levels did not fall below 95% by 480 s. For comparison between group T and group M, a Kaplan-Meier survival analysis was performed, but not the Mann-Whitney U test [11]. Statistical significance was set at P<0.05.
Results
PRIMARY OUTCOME:
The primary outcome, PaO2 at T1, was 261.5±30.9 mmHg for group M and 446.1±84.4 mmHg for group T (Table 2; P<0.001). A higher PaO2 was observed in group T than in group M at T1–T3. PaO2 at T1–T2 in both groups was higher than that at T0. As for the intubation time, group M measured 42.7±8.1 s, and group T measured 41.6±6.5 s (P=0.640).
SAFE APNEA TIME:
Figure 2 shows the Kaplan-Meier survival curves for both groups. The safe apnea time of group T was 377.5 (360, 480) s and for group M was 239 (217, 273.8) s. The safe apnea time for group T was significantly longer than that of group M (P=0.01). Group T had a higher percentage of safe apnea time up to 480 s and a shorter time to re-oxygenation than group M (Figure 3, P<0.05).
:
Both groups had higher PaCO2 levels at T2–T3 than at T0–T1. At T2–T3, group T had a higher PaCO2 than group M. At the beginning of mechanical ventilation, group T had a higher partial pressure of end-tidal carbon dioxide (PETCO2) (Table 3).
HEMODYNAMIC CHANGES AND ADVERSE EVENTS:
There was no statistically significant difference in hemodynamic changes or adverse events after induction of anesthesia between the 2 groups (Figure 4). No THRIVE ventilation-related complications were observed in the patients in group T.
Discussion
To study only the pre-oxygenation period and avoid gastric content regurgitation of THRIVE continuous oxygenation, we replaced THRIVE with a facemask during the apneic period in group T. Compared with PaO2 with facemask pre-oxygenation, the study results showed that PaO2 was significantly higher after pre-oxygenation with THRIVE in elderly patients undergoing RSI. In addition, the safe apnea time was extended, and the time to re-oxygenation was shortened in the THRIVE group.
In 2017, Mir et al used either THRIVE or facemask for oxygen supply during pre-oxygenation for RSI anesthesia [8]. Although they found no statistically significant difference in PaO2, the THRIVE group’s arterial blood gases might have been more favorable if the intubation time was the same in both groups. In 2018, Lodenius et al randomly allocated patients having RSI anesthesia for emergency surgery to pre-oxygenation with 100% oxygen with a facemask or THRIVE [9]. There were no differences in apnea time or the lowest SpO2 (1 min after intubation) between the groups. However, there were 5 patients (12.5%) whose oxygen saturation level fell below 93% on pre-oxygenation with the facemask, whereas there were none in the THRIVE group. Similar results have also been reported in patients with acute hypoxic respiratory failure [10]. Both of these reports and ours suggest that THRIVE may provide a pre-oxygenation benefit.
Due to the relative elevation of the diaphragm, increased intra-abdominal pressure, and lower effective lung volume, combined with the elderly patients’ diminished lung function, elderly patients with gastrointestinal obstruction are more at risk of hypoxia. However, THRIVE could provide continuous positive airway pressure by delivering heated and humidified high-flow pure oxygen, more than the patient needs, opening the upper airway and even the distal alveoli, thereby reducing dead space and decreasing the incidence of atelectasis and improving oxygenation [15]. Furthermore, THRIVE increased end-expiratory volume by increasing end-expiratory lung impedance, thus increasing functional residual air volume and preventing the collapse of alveoli and airways, which was beneficial for airway opening and improving pulmonary shunts in elderly patients [16]. Finally, the thin cheeks and lost teeth of elderly patients with gastrointestinal obstruction made facemask ventilation inefficient and less airtight, while THRIVE ventilation was not affected. Clinically, safe apnea time was usually defined as when SpO2 dropped to 90% after stopping ventilation after induction of general anesthesia [17]. However, considering combined cardiovascular and cerebrovascular illnesses in elderly individuals and impaired respiratory capacity, we defined the safe apnea time as when SpO2 dropped to 95% following breathing stoppage, and the end of the test was set to 480 s. In our study, PaCO2 was elevated in both groups during the apneic oxygenation period compared with preoperative PaCO2. Nevertheless, it was significantly higher in the THRIVE group than in the facemask pre-oxygenation group because the safe apnea time was significantly longer with THRIVE pre-oxygenation, which increased carbon dioxide accumulation.
In 2019, Wong et al found that the safe apnea time was significantly longer in the THRIVE group than in the facemask pre-oxygenation group [18]. Similar conclusions were obtained in a study by Guy et al on the duration of asphyxia in patients with obesity [11]. Hua et al discovered that THRIVE pre-oxygenation resulted in higher PaO2 and longer safe apnea time than facemask pre-oxygenation, consistent with our findings [6]. In contrast, Pillai et al found no difference between THRIVE pre-oxygenation and facemask pre-oxygenation [19]. The difference between the 2 studies might be explained by pre-oxygenation time (3 min vs 5 min), and the index monitored (transcutaneous partial pressure of oxygen vs PaO2). Compared with our study, Mir et al found that PaO2 after endotracheal intubation was comparable with THRIVE or facemask pre-oxygenation in patients with RSI undergoing emergency general anesthesia, which differed from our findings [8]. In their study, the endotracheal intubation time in the THRIVE group was longer than in the facemask group. With the exact duration of endotracheal intubation, PaO2 should be higher in the THRIVE group than in the facemask group. According to Tan et al, THRIVE pre-oxygenation in full-term pregnant women did not achieve similar (EtO2 ≥90%) results as facemask pre-oxygenation [20]. We speculate that this was associated with flow rate, pre-oxygenation time, and respiratory status. Their study had a pre-oxygenation time of 3 min and a flow rate of 30 L/min, and the patients were not instructed to close their mouths before pre-oxygenation. On the other hand, in our study, the patients in the THRIVE group breathed nasally with their mouths shut, the pre-oxygenation time was 5 min, and the flow rate was 40 L/min. Open-mouth and closed-mouth use of THRIVE produces EtO2 of 86 kPa and 49 kPa, respectively. Higher EtO2 partial pressure promotes oxygen diffusion and ensures oxygen supply for the patient.
Additionally, THRIVE’s PEEP is flow-dependent, and higher flow rates create higher PEEP values, which are more beneficial for maintaining airway opening [19]. Depending on the patient’s physiological state, a relatively longer pre-oxygenation time may be necessary to maximize the pre-oxygenation effect. THRIVE was used at a flow rate of 50 L/min, pre-oxygenation was for 3 min, and pre-oxygenation resulted in better results than facemask for pregnant women with RSI [21]. Thus, the application of THRIVE for pre-oxygenation may have specificity in some patients.
This study had the following limitations. First, the optimal flow rate regarding the application of THRIVE was not explored, and further studies will be conducted subsequently. Second, whether the 2 pre-oxygenation methods will affect gastric volume was not evaluated. Third, we have not yet determined the best pre-oxygenation time.
Conclusions
Pre-oxygenation using THRIVE during RSI improved oxygenation and safe apnea time. Once the patient’s airway is handled for a long period, THRIVE may be proven beneficial to maintain oxygenation and ensure patient safety.
Figures
 Figure 1. Flow diagram of patient enrollment (Visio 2016, Microsoft Inc., USA).
Figure 1. Flow diagram of patient enrollment (Visio 2016, Microsoft Inc., USA). ![Kaplan-Meier curve of time to SpO2 ≤95% for the 2 study groups: group M (facemask [M]) and group T (Optiflow THRIVE™ [T]) (GraphPad Prism 9, GraphPad Software Inc., USA).](https://jours.isi-science.com/imageXml.php?i=medscimonit-28-e938168-g002.jpg&idArt=938168&w=1000) Figure 2. Kaplan-Meier curve of time to SpO2 ≤95% for the 2 study groups: group M (facemask [M]) and group T (Optiflow THRIVE™ [T]) (GraphPad Prism 9, GraphPad Software Inc., USA).
Figure 2. Kaplan-Meier curve of time to SpO2 ≤95% for the 2 study groups: group M (facemask [M]) and group T (Optiflow THRIVE™ [T]) (GraphPad Prism 9, GraphPad Software Inc., USA). 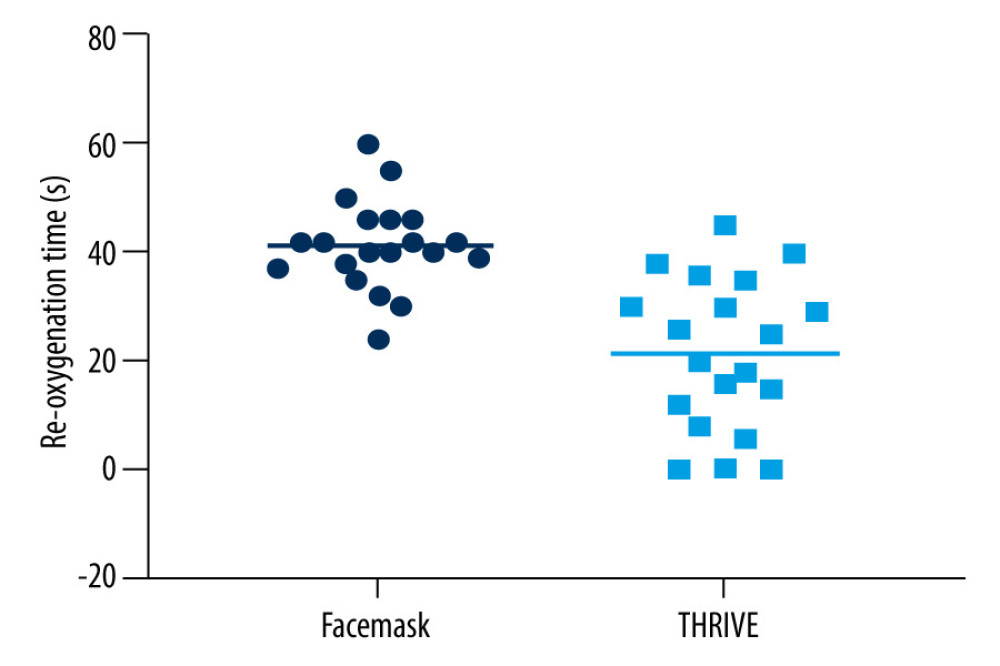 Figure 3. Re-oxygenation time in the facemask and THRIVE groups (GraphPad Prism 9, GraphPad Software Inc., USA).
Figure 3. Re-oxygenation time in the facemask and THRIVE groups (GraphPad Prism 9, GraphPad Software Inc., USA). 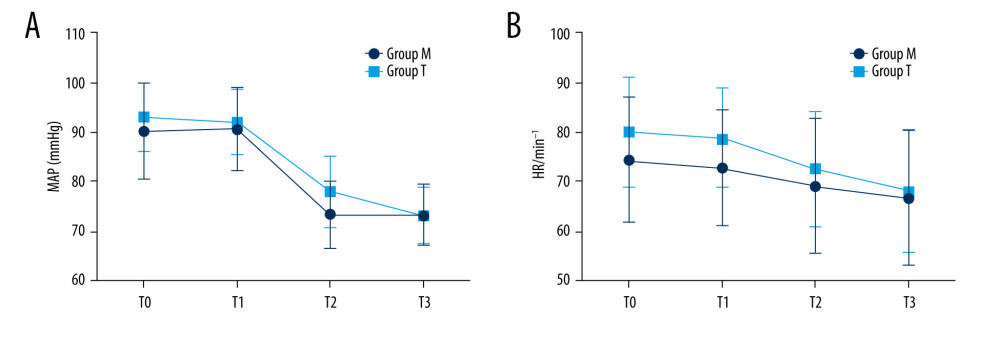 Figure 4. Mean arterial pressure and heart rate in the facemask and THRIVE groups (GraphPad Prism 9, GraphPad Software Inc., USA).
Figure 4. Mean arterial pressure and heart rate in the facemask and THRIVE groups (GraphPad Prism 9, GraphPad Software Inc., USA). References
1. Sun J, Li XH, Zuo YX, Comparison of Incidence of hypoxia during modified rapid sequence induction and an alternative technique: A prospective randomized controlled trial: Int J Clin Exp Med, 2015; 8; 16231-37
2. Eichelsbacher C, Ilper H, Noppens RRapid sequence induction and intubation in patients with risk of aspiration: Recommendations for action for practical management of anesthesia: Anaesthesist, 2018; 67; 568-83 [in German]
3. Jackson PG, Raiji MT, Evaluation and management of intestinal obstruction: Am Fam Physician, 2011; 83; 159-65
4. Sprung J, Gajic O, Warner DO, Review article: Age related alterations in respiratory function – anesthetic considerations: Can J Anaesth, 2006; 53; 1244-57
5. Badiger S, John M, Fearnley RA, Ahmad I, Optimizing oxygenation and intubation conditions during awake fibre-optic intubation using a high-flow nasal oxygen-delivery system: Br J Anaesth, 2015; 115; 629-32
6. Hua Z, Liu Z, Li Y, Transnasal humidified rapid insufflation ventilatory exchange vs. facemask oxygenation in elderly patients undergoing general anaesthesia: A randomized controlled trial: Sci Rep, 2020; 10; 5745
7. Patel A, Nouraei SA, Transnasal humidified rapid-insufflation ventilatory exchange (THRIVE): A physiological method of increasing apnoea time in patients with difficult airways: Anaesthesia, 2015; 70; 323-29
8. Mir F, Patel A, Iqbal R, A randomised controlled trial comparing transnasal humidified rapid insufflation ventilatory exchange (THRIVE) pre-oxygenation with facemask pre-oxygenation in patients undergoing rapid sequence induction of anaesthesia: Anaesthesia, 2017; 72; 439-43
9. Lodenius A, Piehl J, Ostlund A, Transnasal humidified rapid-insufflation ventilatory exchange (THRIVE) vs. facemask breathing pre-oxygenation for rapid sequence induction in adults: A prospective randomised non-blinded clinical trial: Anaesthesia, 2018; 73; 564-71
10. Frat JP, Thille AW, Mercat A, High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure: N Engl J Med, 2015; 372; 2185-96
11. Guy L, Christensen R, Dodd B, The effect of transnasal humidified rapid-insufflation ventilator exchange (THRIVE) versus nasal prongs on safe apnoea time in paralysed obese patients: A randomised controlled trial: Br J Anaesth, 2022; 128; 375-81
12. Hinkelbein J, Kranke PRapid sequence induction: Anasthesiol Intensivmed Notfallmed Schmerzther, 2018; 53; 631-34 [in German]
13. Frerk C, Mitchell VS, McNarry AF, Difficult Airway Society 2015 guidelines for management of unanticipated difficult intubation in adults: Br J Anaesth, 2015; 115; 827-48
14. Heinrich S, Horbach T, Stubner H, Benefits of heated and humidified high flow nasal oxygen for preoxygenation in morbidly obese patients undergoing bariatric surgery: A randomized controlled study: Journal of Obesity and Bariatrics, 2014; 1; 2377-9284.1000003
15. Parke R, McGuinness S, Eccleston M, Nasal high-flow therapy delivers low level positive airway pressure: Br J Anaesth, 2009; 103; 886-90
16. Corley A, Caruana LR, Barnett AG, Oxygen delivery through high-flow nasal cannulae increase end-expiratory lung volume and reduce respiratory rate in post-cardiac surgical patients: Br J Anaesth, 2011; 107; 998-1004
17. Rajan S, Joseph N, Tosh P, Effectiveness of transnasal humidified rapid-insufflation ventilatory exchange versus traditional preoxygenation followed by apnoeic oxygenation in delaying desaturation during apnoea: A preliminary study: Indian J Anaesth, 2018; 62; 202-7
18. Wong DT, Dallaire A, Singh KP, High-flow nasal oxygen improves safe apnea time in morbidly obese patients undergoing general anesthesia: A randomized controlled trial: Anesth Analg, 2019; 129; 1130-36
19. Pillai A, Daga V, Lewis J, High-flow humidified nasal oxygenation vs. standard face mask oxygenation: Anaesthesia, 2016; 71; 1280-83
20. Tan PCF, Millay OJ, Leeton L, Dennis AT, High-flow humidified nasal preoxygenation in pregnant women: A prospective observational study: Br J Anaesth, 2019; 122; 86-91
21. Zhou S, Zhou Y, Cao X, The efficacy of high flow nasal oxygenation for maintaining maternal oxygenation during rapid sequence induction in pregnancy: A prospective randomised clinical trial: Eur J Anaesthesiol, 2021; 38; 1052-58
Figures
 Figure 1. Flow diagram of patient enrollment (Visio 2016, Microsoft Inc., USA).
Figure 1. Flow diagram of patient enrollment (Visio 2016, Microsoft Inc., USA).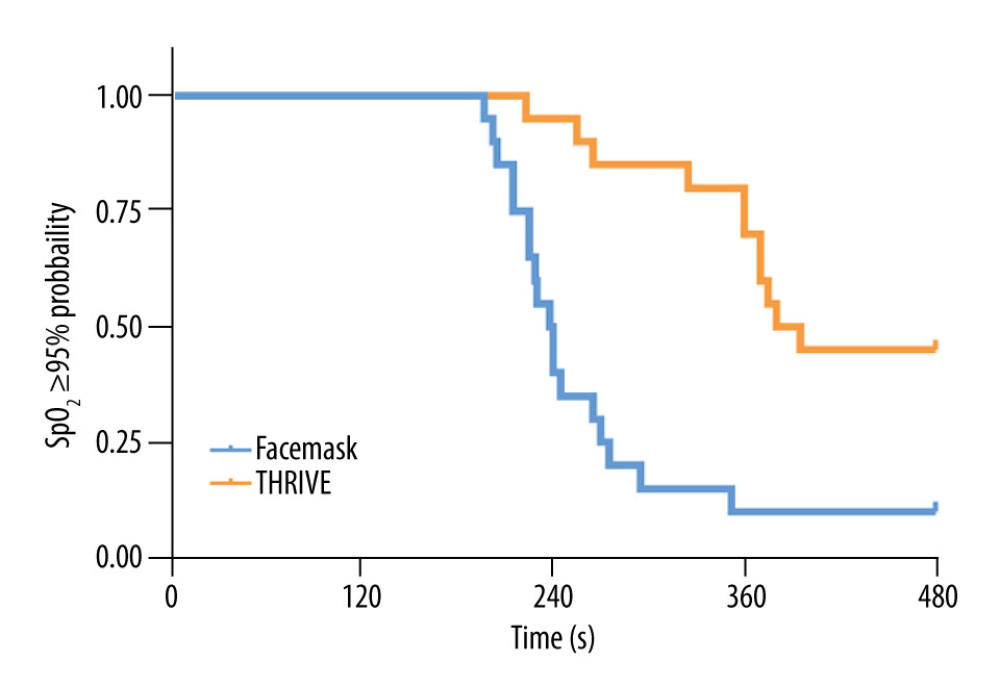 Figure 2. Kaplan-Meier curve of time to SpO2 ≤95% for the 2 study groups: group M (facemask [M]) and group T (Optiflow THRIVE™ [T]) (GraphPad Prism 9, GraphPad Software Inc., USA).
Figure 2. Kaplan-Meier curve of time to SpO2 ≤95% for the 2 study groups: group M (facemask [M]) and group T (Optiflow THRIVE™ [T]) (GraphPad Prism 9, GraphPad Software Inc., USA). Figure 3. Re-oxygenation time in the facemask and THRIVE groups (GraphPad Prism 9, GraphPad Software Inc., USA).
Figure 3. Re-oxygenation time in the facemask and THRIVE groups (GraphPad Prism 9, GraphPad Software Inc., USA). Figure 4. Mean arterial pressure and heart rate in the facemask and THRIVE groups (GraphPad Prism 9, GraphPad Software Inc., USA).
Figure 4. Mean arterial pressure and heart rate in the facemask and THRIVE groups (GraphPad Prism 9, GraphPad Software Inc., USA). Tables
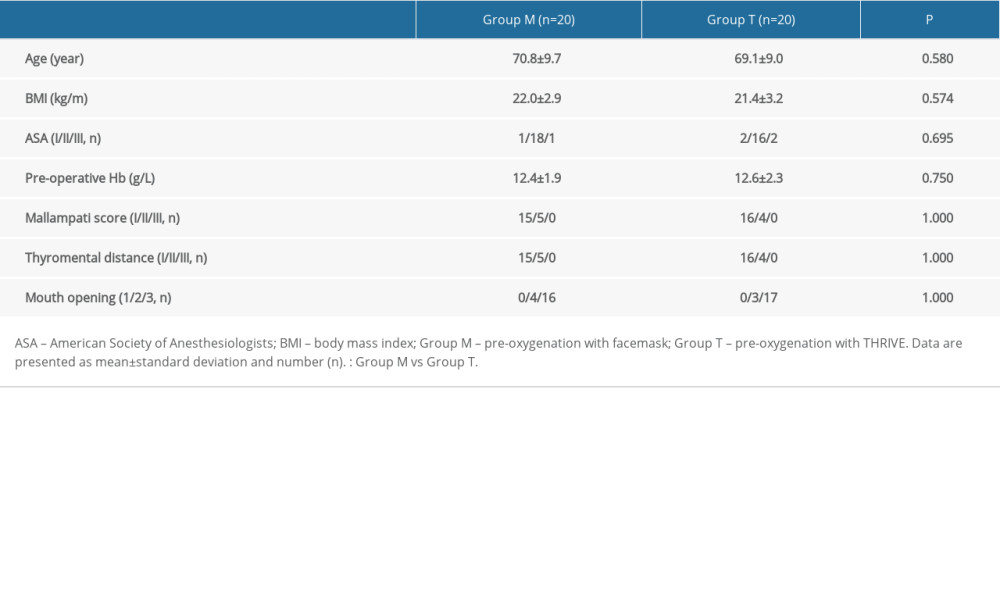 Table 1. Patient characteristics.
Table 1. Patient characteristics.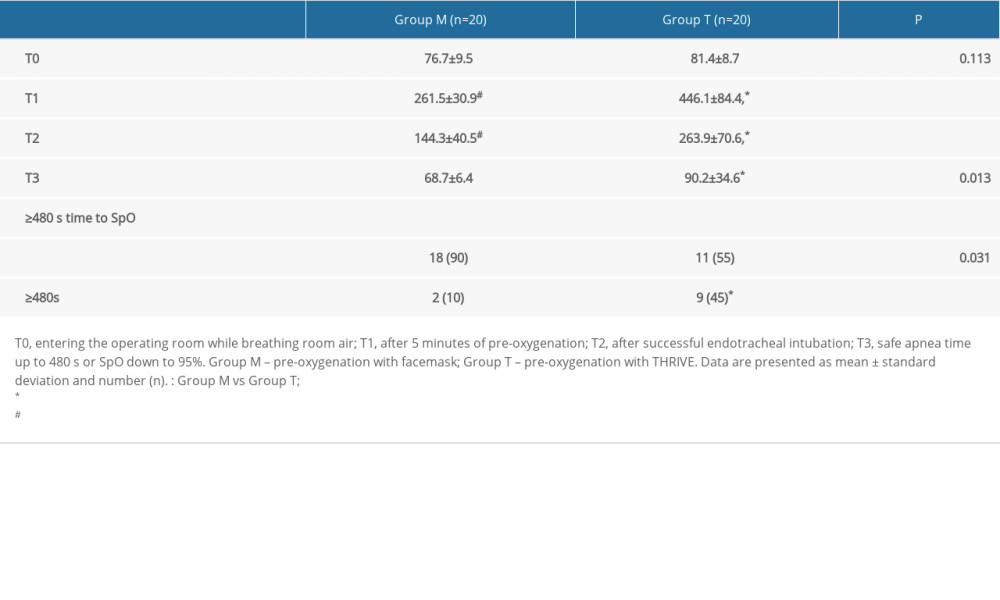 Table 2. Comparison of PaO2 at different times and safe apnea time of 480 s between the 2 groups.
Table 2. Comparison of PaO2 at different times and safe apnea time of 480 s between the 2 groups. Table 3. Comparison of PaCO2 and PETCO2 after intubation between the facemask and THRIVE groups.
Table 3. Comparison of PaCO2 and PETCO2 after intubation between the facemask and THRIVE groups. Table 1. Patient characteristics.
Table 1. Patient characteristics. Table 2. Comparison of PaO2 at different times and safe apnea time of 480 s between the 2 groups.
Table 2. Comparison of PaO2 at different times and safe apnea time of 480 s between the 2 groups. Table 3. Comparison of PaCO2 and PETCO2 after intubation between the facemask and THRIVE groups.
Table 3. Comparison of PaCO2 and PETCO2 after intubation between the facemask and THRIVE groups. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








