12 September 2022: Editorial
Editorial: Treatment with Dual Incretin Receptor Agonists to Maintain Normal Glucose Levels May Also Maintain Normal Weight and Control Metabolic Dysfunction-Associated Fatty Liver Disease (MAFLD)
Ana Luisa Ordóñez-Vázquez1CDEF, Sofía Murúa Beltrán-Gall1CDEF, Shreya C. Pal12DEF, Nahum Méndez-Sánchez34DF*DOI: 10.12659/MSM.938365
Med Sci Monit 2022; 28:e938365
Abstract
ABSTRACT: Worldwide, metabolic dysfunction-associated fatty liver disease (MAFLD) is the most common chronic liver disease. MAFLD is associated with insulin resistance, type 2 diabetes mellitus (T2DM), obesity, hypertension, and dyslipidemia. Early diagnosis and management are vital to improving hepatic and cardiometabolic outcomes. Dietary change, weight loss, and structured exercise are the main treatment approaches for fatty liver disease. Since 2010, several investigational drug treatments failed to achieve regulatory approval due to mixed and unsatisfactory results. Although glucagon-like peptide 1 receptor agonists (GLP1-RAs) showed initial promise as therapeutic agents, metabolic liver damage can recur after monotherapy cessation. Dual incretin receptor agonists target the receptors for glucagon-like peptide 1 (GLP-1) and gastric inhibitory peptide (GIP). Importantly, on May 13, 2022, the US Food and Drug Administration (FDA) approved tirzepatide as the first dual GLP-1 and GIP receptor agonist for the treatment of T2DM. Dual incretin receptor agonists induce weight loss and enhance hepatic lipid metabolism and systemic insulin sensitivity. Insulin resistance and hepatic steatosis are the main contributors to the development of MAFLD. Treatment with dual incretin analogs reduces hepatic steatosis, lobular inflammation, liver cell damage, fibrosis, and total liver triglyceride levels. The availability of dual incretin receptor agonists for patients with MAFLD may result in weight control, normalizing insulin sensitivity, and reducing or even reversing metabolic dysfunction and liver damage. This Editorial aims to provide an update and discuss how treatment with dual incretin receptor agonists may maintain normal glucose levels and weight and control MAFLD.
Keywords: Editorial, Incretins, Fatty Liver, Non-alcoholic fatty liver disease, Insulin Resistance, Diabetes Mellitus, Type 2, Glucagon-Like Peptide 1, Glucose, Humans, United States, Weight Loss
Worldwide, metabolic dysfunction–associated fatty liver disease (MAFLD) is the most common chronic liver disease and a leading reason for liver transplantation [1]. The global prevalence of MAFLD is 30.4% in the general population and up to 80% in patients with type 2 diabetes mellitus (T2DM) or obesity [1]. MAFLD is associated with insulin resistance, T2DM, obesity, hypertension, and atherogenic dyslipidemia [2,3]. Insulin resistance is the key risk factor for the development and progression of MAFLD [2,3]. Since 2020 an international consensus has proposed the term, MAFLD, to describe the presence of hepatic steatosis with one or more of the following: overweight/obesity, T2DM, or evidence of two or more features of metabolic dysfunction in patients of normal weight [4,5]. Recognizing this close association with metabolic risk factors identifies patients with a worse prognosis when compared with patients diagnosed using the past criteria for non-alcoholic fatty liver disease (NAFLD) [1]. Multiple consensus panels have accepted the definition and use of the term MAFLD [4–6]. New criteria for the diagnosis of MAFLD have resulted in the inclusion of a significant proportion of the population who are now recognized to be at increased cardiometabolic risk, resulting in improved diagnosis and management [4–6]. Even though MAFLD is associated with a range of metabolic and hepatic changes, the reduced life expectancy is mainly related to cardiovascular disease, which is the leading cause of death in people with MAFLD [4–6]. Early diagnosis and intervention are vital to improving liver and cardiometabolic outcomes and reducing the health care burden of MAFLD [7]. Currently, the latest international guidelines recommend dietary change, weight loss, and structured exercise intervention as the cornerstone of therapy in the absence of regulatory-approved drug treatments [7].
Since 2010, several investigational drug treatments have failed to achieve regulatory approval due to mixed and unsatisfactory results. For example, the results from the Pioglitazone vs Vitamin E vs Placebo for Treatment of Non-Diabetic Patients With Nonalcoholic Steatohepatitis (PIVENS) trial (NCT00063622), resulted in pioglitazone and vitamin E being considered as possible adjuvant therapies to lifestyle, dietary changes, and weight loss [8]. However, later studies, including a clinical trial (NCT02970942), found this therapy to have unsatisfactory results [9]. The search for treatments to reduce hepatic steatosis, liver injury, and metabolic and cardiovascular risk has resulted in several clinical trials of new drugs [10]. Notably, elafibranor, cenicriviroc, simtuzumab, selonsertib, volixibat, aldafermin, obeticholic acid, lanifibranor, resmetirom, liraglutide and semaglutide were all studied, as well as developments in bariatric surgery to treat obesity, T2DM, and MAFLD [11].
Currently, weight loss and reduced hepatic insulin resistance are the most useful and promising approaches to managing MAFLD [11]. A recent systematic review and network meta-analysis included randomized controlled trials involving patients with non-alcoholic fatty liver disease (NAFLD) [11]. Meta-analysis data showed that for every 1% reduction in body mass index (BMI), the NAFLD activity score (NAS) was reduced by 1.3% (β=1.28%; P=0.01) and a 1% reduction in the homeostatic model assessment of insulin resistance (HOMA) index reduced the NAS by 0.3% (β=0.31%; P<0.001) [11].
In 2016, the findings from the liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN) phase 2 trial (NCT01237119) were published [12]. The basis for the LEAN trial was that glucagon-like peptide 1 receptor agonist drugs (GLP-1RAs) could be expected to reduce hepatic steatosis since they reduce body weight and reduced insulin resistance. The 72-week phase 2 LEAN trial included 52 patients with biopsy-proven steatohepatitis who were treated with liraglutide at doses of 1.8 mg daily for 48 weeks and weekly semaglutide with subcutaneous doses of 0.1, 0.2, or 0.4 mg [12]. The findings showed efficacy in MAFLD, improved resolution of steatohepatitis, and reduced progression to fibrosis compared with placebo [9,12].
In 2021, Dai and colleagues published the findings from a systematic review and meta-analysis to compare the efficacy of GLP1-RAs in patients with MAFLD [13]. This study identified eight randomized controlled trials that included 396 patients with MAFLD [13]. There were 265 patients with MAFLD who had T2DM [13]. GLP1-RA treatment resulted in a significant reduction in hepatic fat, patient waist circumference, alanine aminotransferase (ALT) levels, γ-glutamyl transferase (γGT) levels, fasting blood glucose levels, and hemoglobin A1c (HbA1C) [13]. The studies reviewed showed no serious adverse events, while most gastrointestinal symptoms resolved within two weeks after dose titration [13]. In a further systematic review and meta-analysis of controlled trials conducted by Mantovani and colleagues in 2021, placebo or standard therapy was compared with GLP-1 RAs for a median of 26 weeks [14]. Treatment was associated with significant reductions in the absolute percentage of liver fat content assessed using magnetic resonance imaging (MRI)-based techniques [14]. Treatment with liraglutide and semaglutide reduced serum liver enzyme levels [14]. Also, liver biopsies showed a histologically-confirmed reduction in steatohepatitis without an increase in liver fibrosis [14]. However, despite these promising results, cessation of treatment with GLP1-RAs resulted in the return of hepatic steatosis and abnormal liver function tests [14].
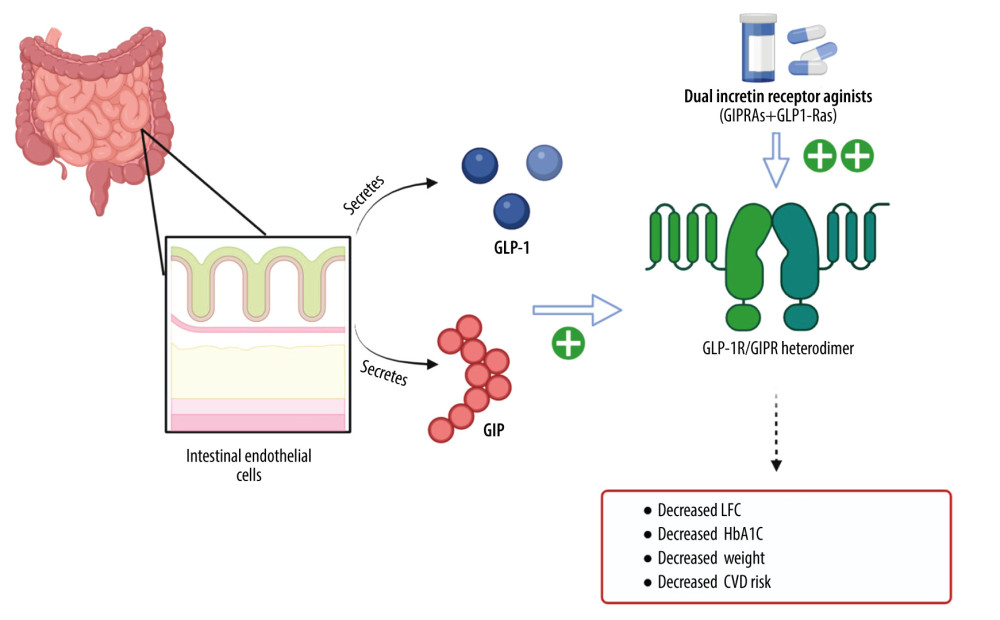
Dual incretin receptor agonists are novel pharmacologic agents that target the receptors for both glucagon-like peptide 1 (GLP-1) and the glucose-dependent insulinotropic polypeptide (GIP) [15]. The gut-derived incretin hormones, GLP-1 and GIP, modulate glucagon secretion, amino acid metabolism, energy expenditure, body weight, and lipid metabolism [15]. GLP-1 inhibits glucagon secretion in hyperglycemic states but not during hypoglycemia or euglycemia [15]. As a result, GLP-1RAs reduce hyperglycemia with little risk of hypoglycemia [16]. In non-diabetics, GIP stimulates glucagon secretion during hypoglycemia and potentiates GLP-1-induced insulin secretion [16]. Also, the action of GIP on glucose metabolism is reduced during hyperglycemic states, which occurs most commonly in patients with T2DM [16]. The enteroinsular axis originates with the activation of enteroendocrine cells in the intestine in response to food intake and ultimately regulates gastric motility, nutrient absorption, blood flow, and food intake [17]. The GIP constituent of dual GIP/GLP-1 receptor agonism is believed to have a central action in promoting GLP-1-induced weight loss and enhanced white adipose tissue function, leading to improved lipid metabolism and systemic insulin sensitivity (Figure 1) [18].
Furthermore, a recently described axis involving the G-coupled receptor GPR119 (GPR119/incretin axis) has been described as a potentially important pathway [19]. The GPR119/incretin axis may have a role in reducing insulin resistance, fat production, and total body mass, appetite reduction, and inhibiting effect on inflammation, potentially reducing the development and outcomes of MAFLD [19]. A further important consideration is the relationship between the incretins and the mechanisms of fibrosis [19]. GIP exerts biological functions through binding to its receptor and influences macrophage activation [19]. GLP-1 binds to its receptor to downregulate collagen formation by downregulating the expression of transforming growth factor-beta 1 (TGF-β1) and fibroblast growth factor-21 (FGF-21) in mouse models of steatohepatitis and AMP-activated protein kinase (AMPK) in diabetic pulmonary fibrosis [19]. Therefore, combined treatment with dual incretins in animal models reduced steatohepatitis, lobular inflammation, hepatocyte damage, and liver fibrosis [19].
Several clinical studies now support the clinical evidence for the efficacy and safety of dual GLP-1/GIP receptor agonists. In May 2022, the dual receptor agonist tirzepatide was approved by the US Food and Drug Administration (FDA) for glycemic control in patients with T2DM [20]. Tirzepatide is administered subcutaneously and showed a greater reduction in levels of HbA1C when compared with semaglutide, insulin degludec, and insulin glargine [20]. Also, in a 2020 study reported by Hartman and colleagues, tirzepatide was shown to reduce the levels of steatohepatitis-related biomarkers, including pro-C3, K-18, AST, and ALT, and to increase adiponectin levels in a dose-dependent manner [21]. Another study by Pirroin and colleagues in 2021 showed that tirzepatide improved glycemic control, reduced triacylglycerides, reduced markers of insulin resistance, and improved beta cell function by direct pharmacologic effects, rather than by the effects of weight loss alone [22]. In a phase 2b, 26-week trial in T2DM patients, tirzepatide (dual GIP/GLP1 RA) demonstrated superior glucose control and reduction in body weight compared with monotherapy with the GLP-1RA, dulaglutide (NCT03131687)4[23]. The findings from this trial showed that tirzepatide reduced serum glycosylated HbA1C by 1.6%, 2.0%, and 2.4% in the 5 mg, 10 mg, and 15 mg dose groups, respectively, compared with 1.1% for dulaglutide at a dose of 1.5 mg [23]. Also, 48% of patients achieved normoglycemia (HbA1C 5.7%) compared with 2% of subjects treated with dulaglutide [23]. More patients treated with 5 mg, 10 mg, and 15 mg of tirzepatide reached normal body weight targets (<5%, <10%, and <15% weight loss from baseline, respectively) when compared with dulaglutide [23]. Reduced appetite was more common with tirzepatide, which was consistent with the putative role of GIP to complement the anorectic action of GLP-1. The incidence of nausea, diarrhea, and vomiting was similar for tirzepatide at 5 mg and 10 mg to dulaglutide, but higher at the 15 mg dose for tirzepatide [18,23]. However, GIP appears to have a dose-related effect on adipose tissue inflammation and may have anti-atherogenic effects [23,24]. Nitric oxide production prevents vessel obstruction by inhibiting vascular smooth muscle cell proliferation [23,24]. Also, there may be effects that suppress the inflammatory responses in specific inflammatory cells and adipocytes [24]. Therefore, the effects of dual incretin receptor agonists may have beneficial effects on managing comorbidities associated with MAFLD, particularly cardiometabolic outcomes.
This overview of the dual function of incretins and the synergic effects on insulin regulation, hepatic fat content, and fibrosis has highlighted the importance of conducting further longitudinal studies. Future studies should primarily evaluate weight loss, hepatic steatosis, and fibrosis. Recent studies have shown the early impact of dual incretin receptor agonists on insulin resistance, obesity, and the metabolic profile, which could be a solution to prevent or treat MAFLD.
Conclusions
The use and effect of dual incretin receptor agonists for patients with MAFLD are promising and increase the probability of inducing and maintaining normal weight and insulin sensitivity and reducing or reversing metabolic dysfunction. Achieving metabolic homeostasis and maintaining a healthy lifestyle could reduce the likelihood of recurring obesity, steatosis, steatohepatitis, and the hepatic and cardiometabolic consequences of MAFLD. Ideally, treatment for each patient should be individualized according to their metabolic state to identify which therapeutic strategy is most effective. However, there is a long way to go before establishing precision medicine in the field of metabolic disease. Clinical trials are awaited to provide evidence for managing specific patient groups according to age, gender, comorbidities such as T2DM, cardiovascular disease, and renal disease, and genetic susceptibility of patient groups according to factors such as ethnicity.
References
1. Spiers J, Brindley JH, Li W, Alazawi W, What’s new in non-alcoholic fatty liver disease?: Frontline Gastroenterol, 2022; 13(e1); e102-8
2. Pal SC, Eslam M, Mendez-Sanchez N, Detangling the interrelations between MAFLD, insulin resistance, and key hormones: Hormones (Athens), 2022 [Epub ahead of print] Erratum in: Hormones (Athens). 2022 [Epub ahead of print]
3. Cusi K, Isaacs S, Barb D, American Association of Clinical Endocrinology Clinical Practice Guideline for the diagnosis and management of nonalcoholic fatty liver disease in primary care and endocrinology clinical settings: Co-sponsored by the American Association for the Study of Liver Diseases (AASLD): Endocr Pract, 2022; 28(5); 528-62
4. Méndez-Sánchez N, Bugianesi E, Gish RG, Global multi-stakeholder endorsement of the MAFLD definition: Lancet Gastroenterol Hepatol, 2022; 7(5); 388-90
5. Mendez-Sanchez N, Arrese M, Gadano A, The Latin American Association for the Study of the Liver (ALEH) position statement on the redefinition of fatty liver disease: Lancet Gastroenterol Hepatol, 2021; 6(1); 65-72
6. Ramírez-Mejía MM, Díaz-Orozco LE, Barranco-Fragoso B, Méndez-Sánchez N, A review of the increasing prevalence of metabolic-associated fatty liver disease (MAFLD) in children and adolescents worldwide and in Mexico and the implications for public health: Med Sci Monit, 2021; 27; 1-9
7. Eslam M, Sarin SK, Wong VWS, The Asian Pacific Association for the Study of the Liver clinical practice guidelines for the diagnosis and management of metabolic associated fatty liver disease: Hepatol Int, 2020; 14; 889-919
8. Sanyal AJ, Chalasani N, Kowdley KV, Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis: N Engl J Med, 2010; 362(18); 1675-85
9. Newsome PN, Buchholtz K, Cusi K, A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis: N Engl J Med, 2021; 384(12); 1113-24
10. Ando Y, Jou JH, Nonalcoholic fatty liver disease and recent guideline updates: Clin Liver Dis (Hoboken), 2021; 17(1); 23-28
11. Sumida Y, Yoneda M, Ogawa Y, Current and new pharmacotherapy options for non-alcoholic steatohepatitis: Expert Opin Pharmacother, 2020; 21(8); 953-67
12. Armstrong MJ, Gaunt P, Aithal GP, Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): A multicentre, double-blind, randomised, placebo-controlled phase 2 study: Lancet, 2016; 387(10019); 679-90
13. Dai Y, He H, Li S, Comparison of the efficacy of glucagon-like peptide-1 receptor agonists in patients with metabolic associated fatty liver disease: Updated systematic review and meta-analysis: Front Endocrinol (Lausanne), 2021; 11; 622589
14. Mantovani A, Petracca G, Beatrice G, Glucagon-like peptide-1 receptor agonists for treatment of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis: An updated meta-analysis of randomized controlled trials: Metabolites, 2021; 11(2); 73
15. Thomas MK, Nikooienejad A, Bray R, Dual GIP and GLP-1 receptor agonist tirzepatide improves beta-cell function and insulin sensitivity in type 2 diabetes: J Clin Endocrinol Metab, 2021; 106(2); 388-96
16. Mathiesen DS, Bagger JI, Bergmann NC, The effects of dual GLP-1/GIP receptor agonism on glucagon secretion – a review: Int J Mol Sci, 2019; 20(17); 1-18
17. Campbell JE, Targeting the GIPR for obesity: To agonize or antagonize? Potential mechanisms: Mol Metab, 2021; 46; 101139
18. Samms RJ, Coghlan MP, Sloop KW, How May GIP enhance the therapeutic efficacy of GLP-1? Trends Endocrinol Metab, 2020; 31(6); 410-21
19. Zhao J, Zhao Y, Hu Y, Peng J, Targeting the GPR119/incretin axis: A promising new therapy for metabolic-associated fatty liver disease: Cell Mol Biol Lett, 2021; 26(1); 32
20. Food and Drug Administration (FDA) News Release: FDA approves novel, dual-targeted treatment for type 2 diabetes in clinical trials, treatment proved more effective than other therapies evaluated May 13, 2022 Available at: https://www.fda.gov/news-events/press-announcements/fda-approves-novel-dual-targeted-treatment-type-2-diabetes
21. Hartman ML, Sanyal AJ, Loomba R, Effects of novel dual GIP and GLP-1 receptor agonist tirzepatide on biomarkers of nonalcoholic steatohepatitis in patients with type 2 diabetes: Diabetes Care, 2020; 43(6); 1352-55
22. Pirro V, Roth KD, Lin Y, Effects of tirzepatide, a dual GIP and GLP-1 RA, on lipid and metabolite profiles in subjects with type 2 diabetes: J Clin Endocrinol Metab, 2022; 107(2); 363-78
23. Frias JP, Nauck MA, Van J, Efficacy and safety of LY3298176, a novel dual GIP and GLP-1 receptor agonist, in patients with type 2 diabetes: A randomised, placebo-controlled and active comparator-controlled phase 2 trial: Lancet, 2018; 392(10160); 2180-93
24. Rizvi AA, Rizzo M, The emerging role of dual GLP-1 and GIP receptor agonists in glycemic management and cardiovascular risk reduction: Diabetes Metab Syndr Obes, 2022; 15; 1023-30
In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387