18 March 2023: Clinical Research
Effect of Polylactic Acid Membrane on Guided Bone Regeneration in Anterior Maxillary Implantation
Xin Li1ABCDEFG, Qiao Jin2ADFG, Hao Xu1ABCF, Shuang Zhang2AFG, Wenxue Wang1ACEF, Baodong Zhao1ABCDEFG*DOI: 10.12659/MSM.938566
Med Sci Monit 2023; 29:e938566
Abstract
BACKGROUND: This study evaluated the clinical efficacy of polylactic acid (PLA) membranes in guiding bone regeneration in anterior maxillary implants.
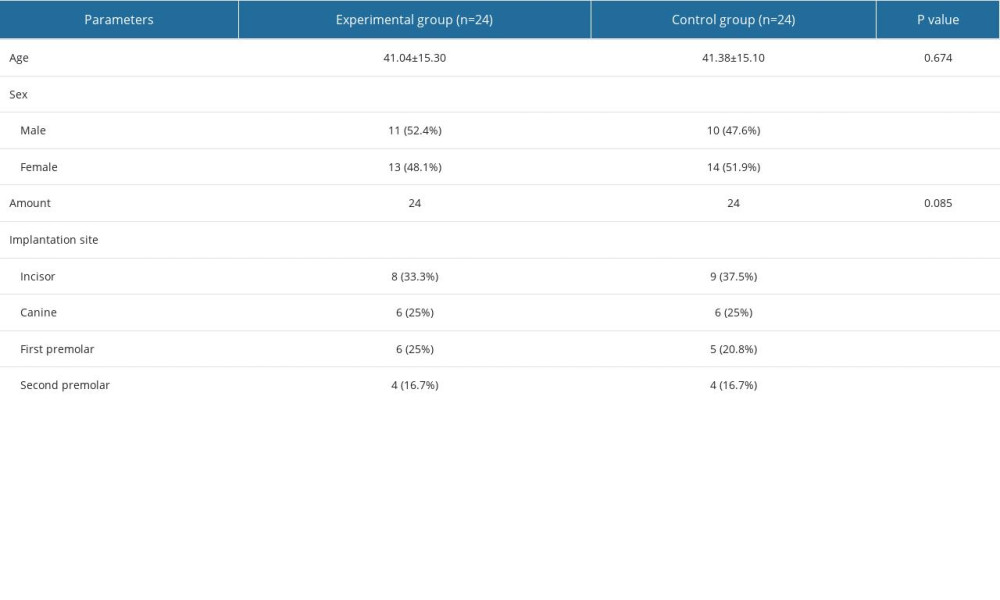
MATERIAL AND METHODS: Forty-eight participants with maxillary anterior tooth loss requiring implantation with guided bone regeneration were recruited and randomly divided into 2 groups (n=24): the PLA membrane (experimental) and the Bio-Gide membrane (control) group. Wound healing was observed at 1 week and 1 month postoperatively. Cone beam CT was performed immediately and at 6 and 36 months postoperatively. Soft-tissue parameters were measured at 18 and 36 months postoperatively. Implant stability quotient (ISQ) and patient satisfaction were separately evaluated at 6 and 18 months after the operation. The independent sample t test and chi-square test were preformed to analyze quantitative and descriptive statistics, respectively.
RESULTS: No implants were lost and there were no statistically significant differences in ISQ between the 2 groups. The labial bone plates in the experimental group exhibited a non-significantly greater extent of absorption than did the control group at 6 and 18 months postoperatively. Soft-tissue parameters did not reveal inferior results for the experimental group. Patients in both groups were satisfied.
CONCLUSIONS: PLA membranes are comparable to Bio-Gide in terms of the effectiveness and safety and can be used as a barrier membrane to guide bone regeneration in clinical practice.
Keywords: Dental Implants, Polylactic Acid-Polylipoic Acid, Humans, Polyesters, Treatment Outcome, Wound Healing
Background
Implant dentures are the most used aesthetic method for repairing tooth loss [1]. To obtain a good long-term prognosis, there should be enough bone at the implant site to ensure osseointegration of the implant during functional loading [2–6]. However, the anatomical characteristics in the maxillary aesthetic area are as follows: 1) a thin labial bone plate occurs in most cases; 2) the long axis of the teeth is in an angular relationship with the alveolar process, which is prone to centripetal absorption; and 3) a lack of bone width in the aesthetic area is more common than that in the mandible [7,8]. If the tooth is extracted because of periodontal disease, trauma, tumor, or other reasons, the bone defect will be more serious. If the implant is placed deeper to obtain sufficient alveolar ridge width, the aesthetic effect will be adversely affected owing to an imbalance in the proportion and contour of the prosthesis. Different strategies such as onlay bone grafting, distraction osteogenesis, and guided bone regeneration (GBR) have been applied to repair the missing bone tissue.
In 1989, Dahlin et al introduced guided tissue regeneration for oral implantation and began research on the application of GBR technology [9]. GBR refers to the use of a barrier membrane to guide bone regeneration and prevent the surrounding fibrous connective tissue from prematurely growing into the bone defect area, changing the healing environment and thus promoting bone tissue regeneration and repair [10–12]. GBR can achieve high predictability of bone augmentation for horizontal defects. The application of GBR technology promotes the development and progress of modern oral implantation, and the increasing requirements for barrier membranes are constantly improving [13]. The biocompatibility, space maintenance ability, operation convenience, ability to induce bone regeneration, and anti-infection properties of barrier membranes have emerged as new research directions [14].
There are many types of barrier membranes used to guide bone regeneration; these membranes are mainly divided into non-absorbable and absorbable membranes. The representative non-absorbable membranes mainly include polytetrafluoroethylene and titanium membranes and exhibit advantages of good histocompatibility, strong mechanical performance, and space maintenance ability; moreover, the retention time in the body can be arbitrarily adjusted. There are also shortcomings, including requirement of a second operation, hard texture, time-consuming operation, higher infection rate, and high cost [15,16]. The absorbable membrane can be degraded and absorbed in vivo without a second operation to remove it. Moreover, the decomposition products have no toxic adverse effects on patients and are increasingly used clinically [17–19]. Bio-Gide membranes are relatively mature for clinical applications and exhibit good histocompatibility, low antigenicity, strong anti-infection ability, good wound healing ability, strong toughness, easy operation, degradation cycle of 4–6 months, and strong tissue regeneration ability; however, the price is relatively high. Moreover, because the material properties of collagen membranes are slightly poor, their texture is relatively soft and their space maintenance capacity is low [20,21].
Polylactic acid (PLA) has the advantages of good mechanical properties, elastic modulus, thermoplastic properties, biocompatibility, degradability, and easy moulding. It has been widely used in medical materials, such as fracture fixation materials and bone scaffold materials in the medical field [14,22,23]. Some studies have found that the combination of a PLA membrane and composite artificial bone material has obvious clinical effects in the treatment of maxillofacial bone defects [24,25]. Owing to their advantages, such as simplicity, low cost, good biocompatibility, strong mechanical performance, controllable degradation time, and strong ability to guide tissue regeneration, PLA membranes have attracted much attention in the field of oral implantation. Previous animal experiments also confirmed that the degradation rate of the PLA membrane in vivo was slower than that of the Bio-Gide membrane, which can better match the repair rate of bone tissue [26–28].
In this study, PLA membrane was compared with Bio-Gide membrane and to evaluate its clinical effect, effectiveness, and safety for guiding bone regeneration around implants in anterior aesthetic areas.
Material and Methods
MATERIAL:
We used PLA oral isolation membrane (Qingdao Jieshengbo Biotechnology Co., Ltd., Qingdao, China), length×width: 15×30 mm, thickness 0.2 mm. PLA membrane is an absorbable biofilm with a specific microporous structure, which is prepared by a freeze-drying process using polyd, l-lactic acid as the raw material. The surface of this product has a two-sided structure: one side is smooth and the other side is rough. We also assessed the effect of Bio-Gide (Geistlich Biomaterials, Wolhusen, LU, Switzerland), an absorbable collagen membrane, length×width: 13×25 mm. Bio-Oss (Geistlich Biomaterials, Wolhusen, LU, Switzerland). Internal connection implants (Straumann BL, Switzerland; Nobel Biocare, Sweden; WeiGao, China), are 3.3–3.8 mm in diameter and 10–14 mm in length.
PATIENT RECRUITMENT:
This study was approved by the Ethics Committee of the Affiliated Hospital of Qingdao University (NO. QYFYEC 2018-007-01) and conformed to STROBE guidelines. To evaluate the sample size, 10 patients with mild bone defects in the maxillary anterior area were randomly divided into a PLA membrane group and a Geistlich Bio-Gide group (n=5). Changes in labial bone dimensions immediately and 6 months postoperatively were preliminarily analyzed. The standard deviation s was calculated as 0.61. According to the formula n=tα2s2/d2, (n is the sample size; α is the significance level; when 0.05 is determined, t≈2; s is 0.61; d is the allowable error, and is determined as 0.1mm). After calculation, the sample size was determined as about 50.
The study included patients treated consecutively at the Department of Oral Implantology, Affiliated Hospital of Qingdao University, from October 2018 to October 2019.
THE INCLUSION CRITERIA WERE AS FOLLOWS:
1) age 18–70 years of either sex; 2) missing maxillary anterior teeth (from central incisors to premolars) requiring implant restoration, with only 1 missing tooth included for each patient; 3) insufficient alveolar bone mass that required GBR immediately after implant placement; and 4) clear mind and understanding ability.
EXCLUSION CRITERIA WERE AS FOLLOWS:
1) specific allergic constitution, especially those sensitive to polylactic acid and collagen; 2) pregnant and lactating women and patients planning to conceive during the study period; 3) patients with general contraindications to implant surgery; 4) those who smoked more than 10 cigarettes a day; and 5) those who were addicted to alcohol or drugs or those who had an addiction tendency.
EXPERIMENTAL DESIGN:
This clinical trial was a randomized, blind, positive control, non-inferiority, prospective study. A diagram of the experimental protocol is shown in Figure 1.
RANDOMIZATION:
Random envelopes were used for grouping. All patients who provided informed consent were provided a unique participant screening number and randomly assigned 1: 1 to either the PLA membrane (experimental) or Geistlich Bio-Gide (control) group.
BLINDING METHOD:
The observer and participant were both blinded; however, the surgeon could not be blinded. That is, the doctor who followed up to observe and evaluate the efficacy did not participate in the early treatment and operation, and the non-blind surgical operator was not allowed to participate in the follow-up evaluation, which was performed by 3 dentists who were not involved in implant placement and GBR.
SURGICAL PROCEDURE:
Cefdinir (Hai Tian Ri Zhongtian Pharmaceutical Co., Ltd., Hainan, China) 100-mg dispersible tablets and paracetamol dihydrocodeine (Weihai Lutan Pharmaceutical Co., Ltd., Weihai, China) 0.51 g (LUGEC) were used 30 minutes before surgery, and 0.12% chlorhexidine (Jiangsu Enweizhong Biological Medicine Co., Ltd., Jiangsu, China) was used to rinse 3 times for 30 seconds each time before surgery [29]. All surgeries were performed by an experienced surgeon. Under local infiltration of anaesthesia with primacaine, a lateral palatal incision of 2–3 mm was made along the alveolar ridge, followed by a distal vertical relaxation incision. A full-thickness mucoperiosteal flap was raised, and the residual granulation tissue on the bone surface was scraped to fully expose the surgical area. Holes were prepared step-by-step following the principles of surgery, and the implants were inserted; each implant had a good initial stability (30–45 N·cm) and was covered with screws. A small ball drill (1.4 mm) (Straumann, Switzerland) was used to drill holes, resulting in spotty bleeding on the facial bone surface. An appropriate amount of Bio-Oss material was placed in the bone defect area, and membranes were trimmed to roughly the same shape as the bone graft area, exceeding more than 3 mm beyond the edge of the bone defect, and 1–2 mm away from the incision. The smooth upper surface was applied outwards to the wound defect and the rough surface was placed adversely. The periosteal suture technique was used to fix the barrier membrane. Sutures were placed without tension. Antibiotics (cefdinir dispersible tablets, 100 mg 3 times daily) and ornidazole tablets (0.5 g twice daily) were administered postoperatively to every patient for 3 days [29]. The surgical procedure is illustrated in Figure 2.
RADIOGRAPHIC EVALUATION (PRIMARY OUTCOME):
Cone beam CT (CBCT) (Ruike Medical Instruments Co., Ltd., Shanghai, China) was performed immediately (T1) and at 6 months (T2) and 36 months (T3) postoperatively. CBCT images were processed using Dental Imaging Software. As shown in Figure 3, on the sagittal plane along the central axis of the implant, the long axis of the implant (from the shoulder to the root) was divided into 5 sites (D1, D2, D3, D4, and D5) at 1/4 intervals, and a straight line perpendicular to the long axis of the implant was created at each point. The horizontal facial bone thickness is the distance from the lip edge of the implant to the outer edge of the labial bone plate on a straight line, and the average value of different sites from D1–D5 at the same time was taken as the average labial bone thickness at that time [30].
IMPLANT MOBILITY (ISQ) TEST:
At 6 months after the operation, the ISQ values of implants were measured and recorded using a resonance frequency analyzer (Osstell ISQ™, Sweden), which is a simple, practical, reproducible, clinically feasible, and non-invasive apparatus for assessment of implant mobility. The resonant frequency sensor rod was connected to the implant and measured 3 times in different directions with Osstell ISQ™. The mean value was recorded.
EVALUATION OF THE SOFT TISSUE CONDITION:
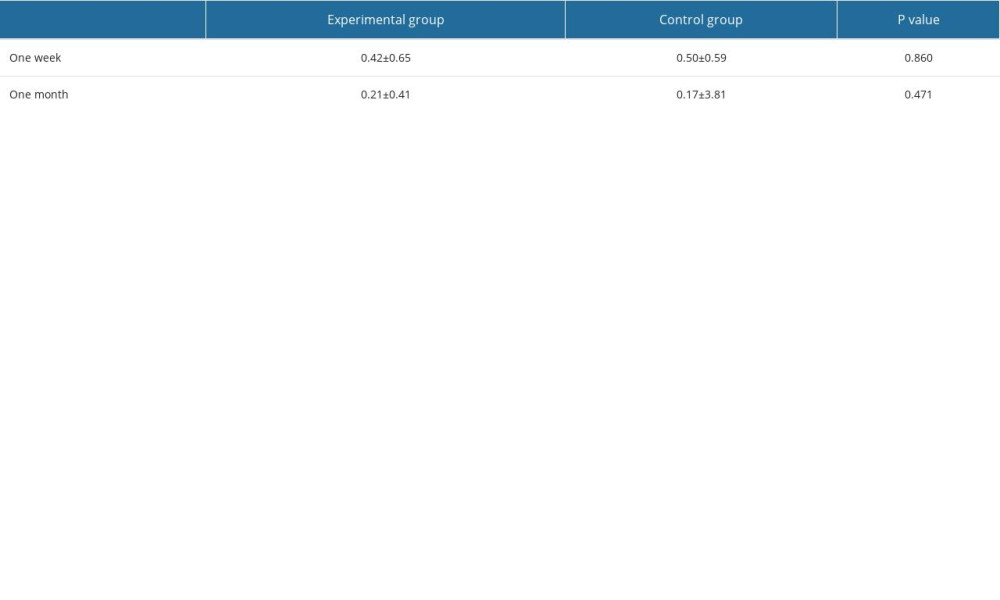
Wound healing of the 2 groups was examined at 1 week and 1 month postoperatively (0=No inflammation; 1=Mild inflammation; 2=Mild inflammation with texture changes; 3=Moderate inflammation; 4=Severe inflammation) [31]. At 18 and 36 months postoperatively, soft-tissue conditions around the implants were examined, including 1) probing depth around the implant: the mesial, central, and distal sites of the labial and lingual sides of the implant were measured, and the average values were taken; 2) plaque index around the implant was measured (0=No plaque; 1=Thin plaque; 2=Moderate amount; 3=A large amount) [32]; and 3) modified bleeding index: the gingival sulcus was gently probed with a periodontal probe to observe and record bleeding (0=No bleeding; 1=Isolated punctate bleeding; 2=Linear bleeding; 3=Severe bleeding) [33].
PATIENT SATISFACTION EVALUATION:
Patient satisfaction was evaluated using the “oral health impact profile for partially edentate with implant supported prostheses” scale (OHIP-I) [34]. The scale consists of 14 items in 3 domains: functional limitation, physical pain and discomfort, and psychological and social impact. Each question includes a score range of 0–4: very healthy=0, healthy=1, moderately healthy=2, unhealthy=3, and very unhealthy=4. The total score is 0–56, with lower scores indicating higher patient satisfaction.
STATISTICAL ANALYSIS:
SPSS22.0 statistical software (SPSS Inc, Chicago, IL, USA) was used to process the data. Quantitative measurements were described as mean±standard deviation (SD). The age data of the 2 groups were tested for normality, and an independent sample
Results
RADIOGRAPHIC EVALUATIONS:
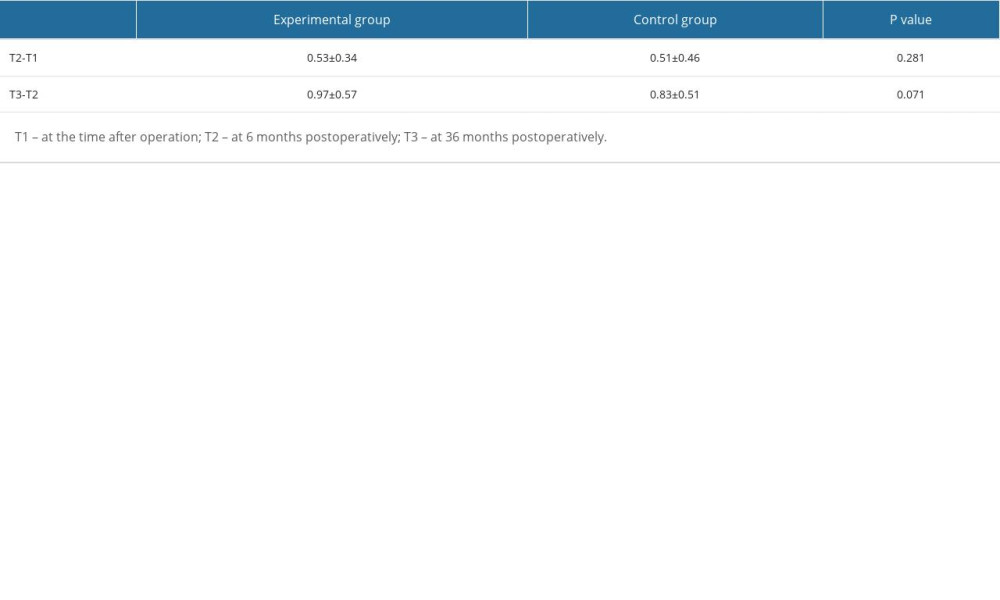
The radiographic images and histograms of the facial bone dimensions of the 2 groups at different times are presented in Figure 4. The means and standard deviations of the changes in the facial bone dimensions are presented in Table 2. Immediately postoperatively (T1), the labial plate thickness (LPH) of the experimental and control group averaged 2.99±0.76 mm and 2.83±0.81 mm, respectively. At 6 months postoperatively (T2), the labial bone of the 2 groups had different degrees of resorption, and the average volume of resorption was 0.53±0.34 and 0.51±0.46, respectively, but there was no significant difference in the changes between the groups (P=0.281). At 36 months postoperatively (T3), the thickness of the labial bony plate changed more obviously, and the absorption volume was close to 1 mm, which was statistically significant compared with that of the same group at T1 and T2; however, there was no statistically significant difference between the 2 groups (P=0.71).
ISQ VALUES:
No implants loosened or fell off, and the implant retention rate was 100%. ISQ values were 71.67±4.30 and 72.83±5.63 in the experimental and control group, respectively, indicating that all implants formed good osseointegration. Although the ISQ values in the control group were slightly higher than that in the experimental group, there was no significant difference (
SOFT TISSUE EVALUATIONS:
The wound healing scores are shown in Table 3. One week and 1 month postoperatively, 2 cases (8.33%) in the experimental group and 1 case (4.17%) in the control group had wound dehiscence and membrane exposure. However, with oral health care, the gingiva was completely covered and healed smoothly, and no infection symptoms occurred in the surrounding tissues, with no statistically significant difference. At 18 and 36 months postoperatively, the soft tissues around the implants were healthy in both groups, and there were no significant differences in the probing depth, plaque index, and modified bleeding index between the 2 groups (Figure 5). The periodontal index of the same group at 36 months was slightly higher than that at 18 months, which may be related to the decrease of patients’ awareness of oral hygiene maintenance.
PATIENT SATISFACTION:
One year after loading, there was no significant gingival recession, porcelain collapse, pain in occlusion, or serious biological or prosthetic complications in either group. Forty-eight participants completed the satisfaction survey according to the OHIP-I score, and all participants were satisfied with the results of the implant dentures, including aesthetic effect, comfort, and function. OHIP-I scores were 2.17±0.96 and 2.08±1.31 in the experimental and control group, respectively. There was no significant difference in satisfaction between the groups (
Discussion
GBR has become the most used bone augmentation technique to restore insufficient bone width in the aesthetic area. In clinical practice, implantation combined with guided bone regeneration usually requires 6 months to heal before starting the prosthetic procedure, and an annual follow-up is required after wearing the crown. Therefore, CBCT is performed immediately and at 6 months and 36 months (about 2.5 years after restoration) postoperatively to assess the changes in labial plate thickness, and soft tissue is evaluated at 1 year (18 months postoperatively) and 2.5 years (36 months postoperatively) after restoration. In the present study, CBCT showed that labial bone thickness in both groups recovered to an average of approximately 3 mm immediately postoperatively. Six months postoperatively, both groups showed different degrees of bone resorption, which was more obvious in the experimental group. However, the between-group difference was less than 0.5 mm and was not statistically significant. Moreover, the average labial bone thickness was still greater than 2 mm after absorption. These results confirmed the effectiveness of the PLA membrane in guiding bone regeneration.
The PLA membrane surface is microporous and has good tissue permeability, which allows tissue fluid to pass through [35] but prevents connective tissue and epithelial cells from passing through, thus providing good space protection for new bone formation. It has a two-sided structure: one side is smooth and the other is rough. The smooth side faces the soft tissue, promoting good healing of the soft tissue. The rough surface is directed towards the bone tissue to induce optimal bone healing. Early animal experiments have demonstrated that PLA membranes have good biocompatibility and shielding effects, which are conducive to the formation of bone at the defect and improve the quality of new bone.
The PLA component of the experimental membrane is obtained from corn, sugarcane, and sugar beets through fermentation and distillation. It is a fully degradable synthetic polymer material without toxic, thermogenic, or immunogenic properties; nonetheless, it has good stability and is not prone to tissue reaction. The final degradation products, carbon dioxide and water, do not cause immune and inflammatory reactions, change the pH value of surrounding tissues, or have any adverse effect on osteogenesis. In this study, the soft-tissue evaluation results revealed that PLA has good biocompatibility and reliable long-term effects.
Labial bone thickness in the aesthetic area greatly impacts appearance and function, and the data are more intuitive; therefore, it is often used to compare the effect of GBR [36]. Considering the different implant lengths, the long axis of the implant on CBCT was divided into 4 equal sections from the shoulder to the root [37]; thus, the average value of labial bone plate thickness at the 4 horizontal lines can better reflect the real situation of labial bone changes. To avoid more bone resorption, labial bone thickness ≥2 mm should be used as the standard in GBR [38]. Bone resorption after GBR is related to many factors, such as the tension of the suture wound, displacement of the bone-filling material, absorption and reconstruction mode of the labial bone, and collapse and degradation time of the barrier membrane [39].
Bio-Gide collagen membranes have great advantages in promoting cell migration and angiogenesis, but their hardness and degradation rate are insufficient [19,40]. However, the PLA membrane in the experimental group has good mechanical strength and supporting performance and does not easily collapse, enabling it to maintain a better osteogenic space. The effective action time of the PLA membrane can reach 4–6 months, and the complete degradation time is 9–12 months. The degradation rate is consistent with the osteogenesis process. Histological observation of previous animal experiments showed that at 12 weeks postoperatively, the collagen membrane was completely degraded, while the PLA membrane maintained a relatively complete structure [28,35]; therefore, it can better protect new bone formation after bone grafting. Its good degradation rate prevents the invasion of soft tissue from interfering with bone healing, and its good supporting performance provides space for the growth of new bone units to obtain a good osteogenic thickness. After the formation and remodelling of new bone, the PLA membrane is broken and gradually degrades into carbon dioxide and water, which are excreted metabolically.
PLA membranes also have shortcomings in terms of their clinical use. The hardness and toughness make the operation of covering the barrier membrane more difficult. In addition, the degree of fit of the bone-filling material is slightly lower than that of the Bio-Gide membrane, which may be related to their chemical structure. The lack of reactive functional groups results in insufficient hydrophilicity and poor cell adhesion [41]. Arunjaroensuk et al recommended preparation of the membrane before covering because of its relative hardness [39]. In the present study, to improve its fitting performance and reduce the risk of cracking during the operation because of its brittleness, the PLA membranes were first soaked in physiological saline to increase their softness. By contrast, the Bio-Gide membrane in the control group was softer and easier to operate. In the future, we will focus on improving the bonding performance and brittleness of PLA membranes to increase the convenience of operation and improve the bone augmentation effect. Additionally, some unexplored variables can have a significant influence on oral environment. The use of paraprobiotics [42], lysates [43], postbiotics [44], and other natural compounds [45] can modify clinical and microbiological parameters in periodontal patients, and they can also affect wound healing after guided bone regeneration. All these variables should be considered in future clinical trials.
The limitation of this study is that the sample size was small. Prospective case-control studies with longer observation periods and larger sample sizes are needed.
Conclusions
The PLA membrane is comparable to Bio-Gide in terms of the effectiveness and safety of implantation in aesthetic areas and can be clinically used as a barrier membrane to guide bone regeneration.
Figures
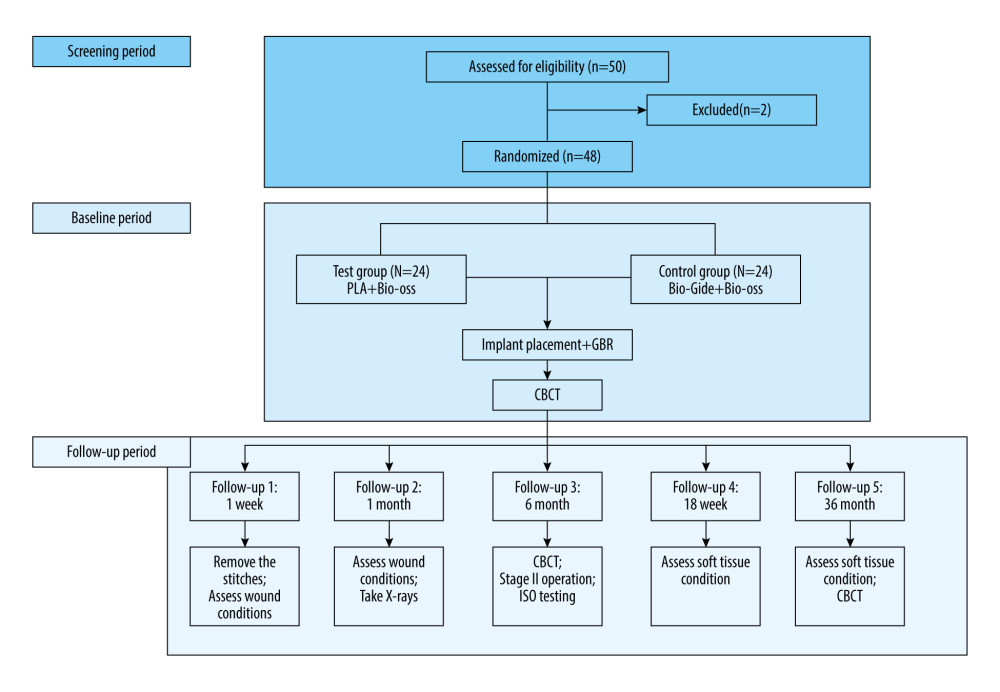 Figure 1. Diagram of the experimental protocol. In the screening period, participants were screened according to the inclusion and exclusion criteria. In the baseline period, the participants included were grouped, underwent surgery, received CBCT (cone beam CT). In follow-up period, soft-tissue and CBCT evaluations were performed. (Microsoft PowerPoint 2003).
Figure 1. Diagram of the experimental protocol. In the screening period, participants were screened according to the inclusion and exclusion criteria. In the baseline period, the participants included were grouped, underwent surgery, received CBCT (cone beam CT). In follow-up period, soft-tissue and CBCT evaluations were performed. (Microsoft PowerPoint 2003). 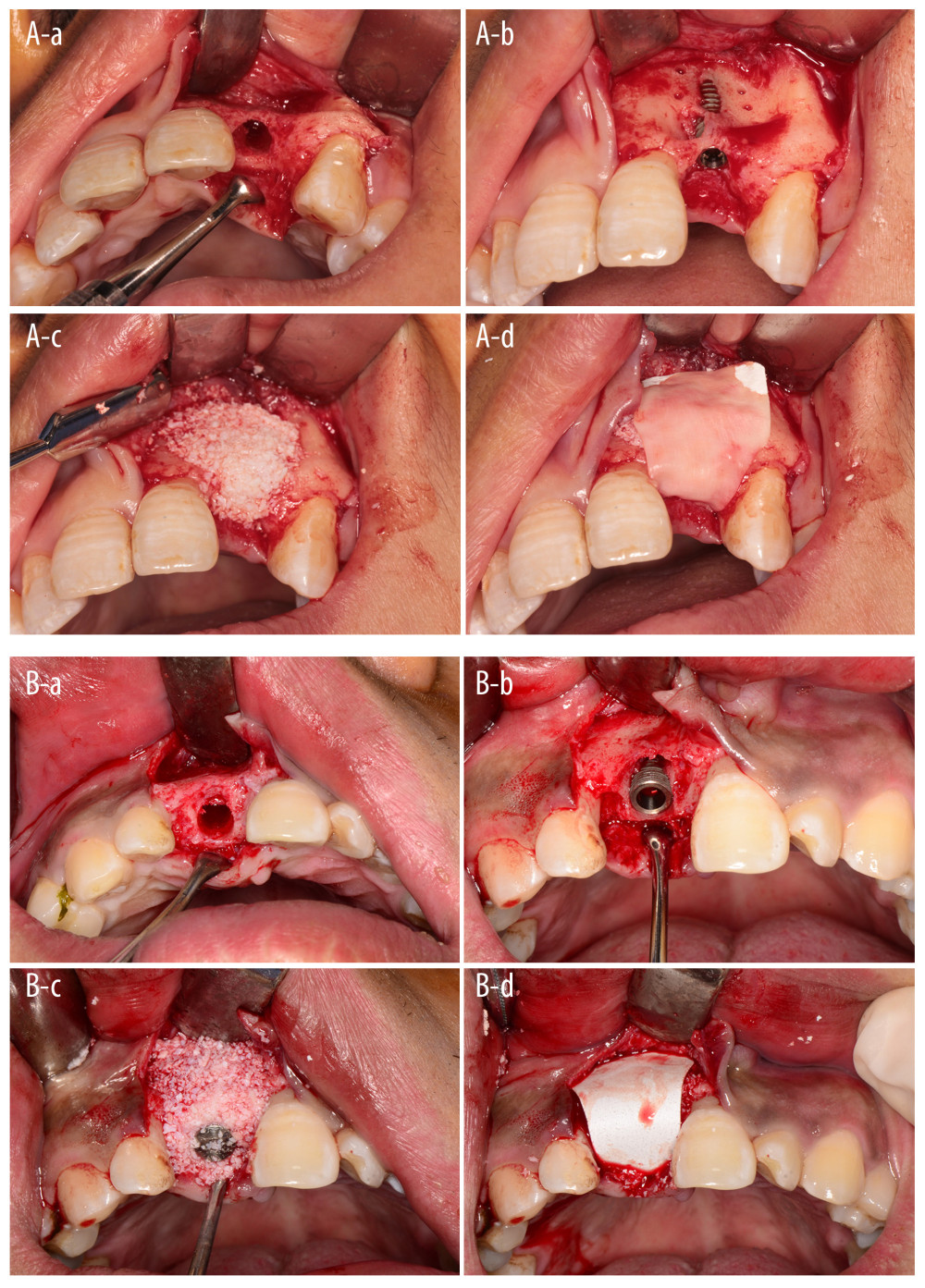 Figure 2. Surgery procedures. A control group, B experimental group. a. Hole preparation; b. Implant placement; c. Bio-Oss material placement on the labial plate; d. Membrane coverage. (Canon EOS 60D).
Figure 2. Surgery procedures. A control group, B experimental group. a. Hole preparation; b. Implant placement; c. Bio-Oss material placement on the labial plate; d. Membrane coverage. (Canon EOS 60D). 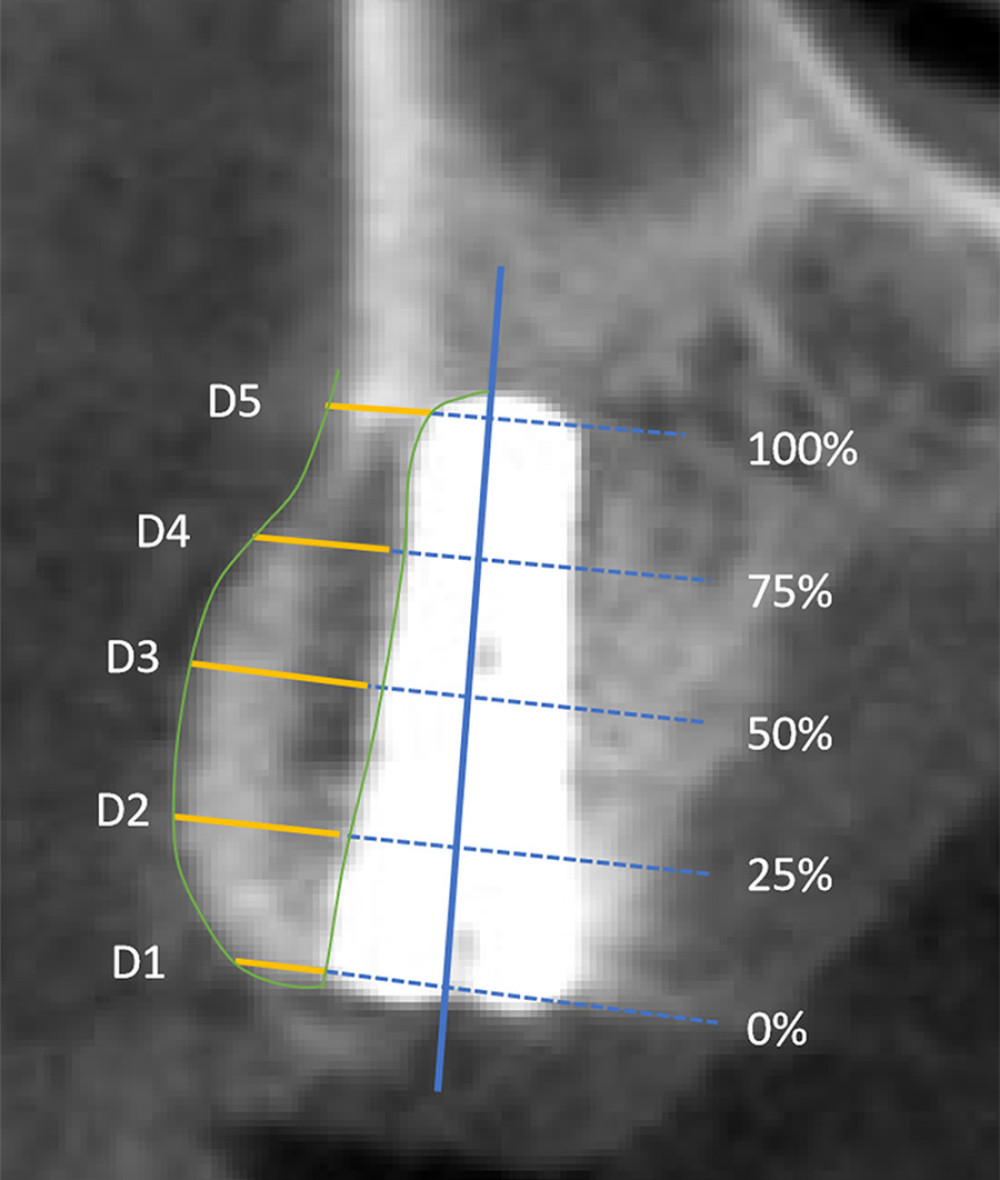 Figure 3. CBCT illustrating of labial alveolar bone thickness. (Microsoft PowerPoint 2003).
Figure 3. CBCT illustrating of labial alveolar bone thickness. (Microsoft PowerPoint 2003). 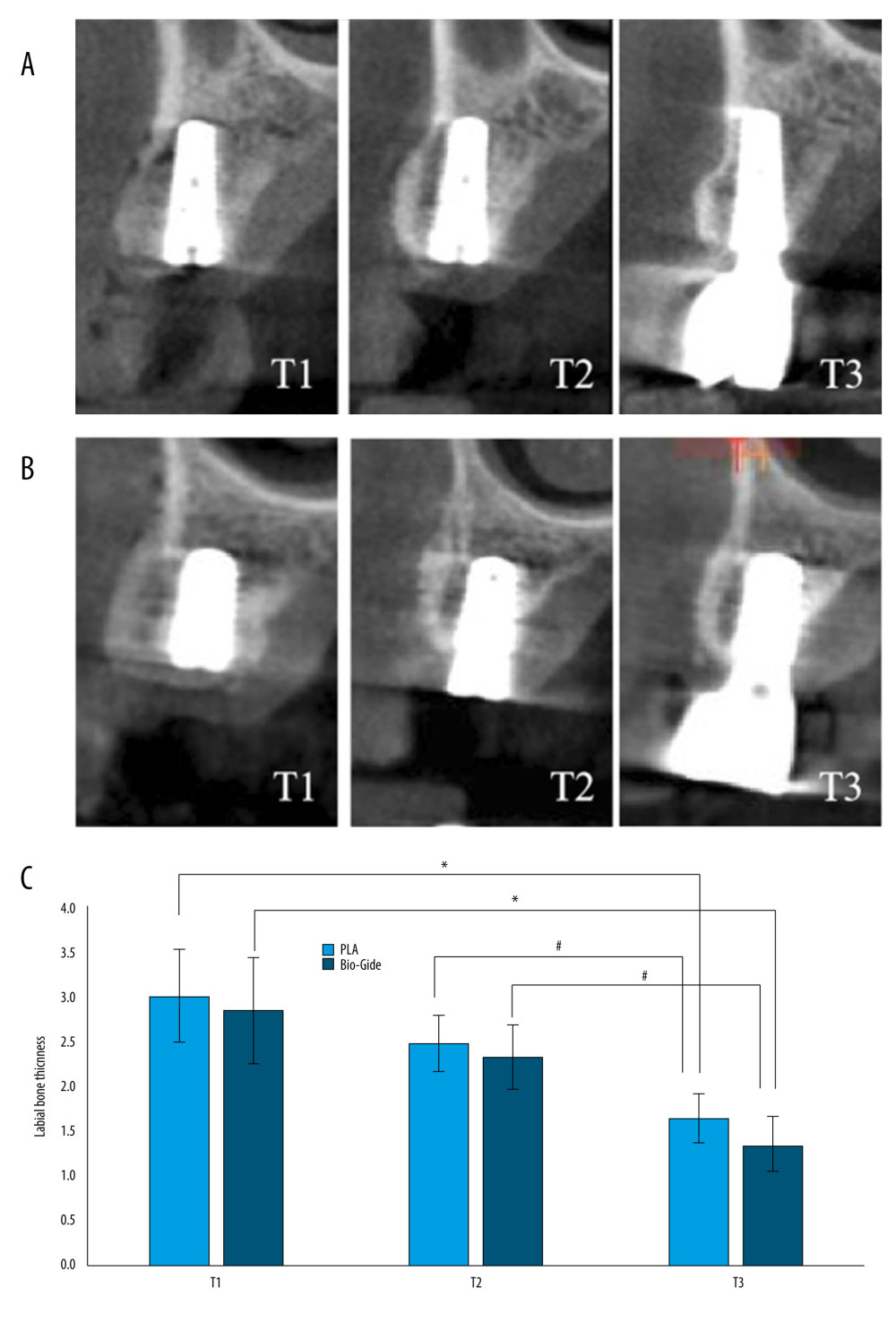 Figure 4. The radiographic images (Ruike Medical Instruments Co., Ltd.) and histograms of the facial bone dimensions of the 2 groups at different times (Microsoft PowerPoint 2003). A experimental group, B control group, C histograms of the facial bone dimensions. * P<0.05 compared with T1, # P<0.05 compared with T2. T1 – at the time after operation; T2 – at 6 months postoperatively; T3 – at 36 months postoperatively. LPH – labial plate thickness.
Figure 4. The radiographic images (Ruike Medical Instruments Co., Ltd.) and histograms of the facial bone dimensions of the 2 groups at different times (Microsoft PowerPoint 2003). A experimental group, B control group, C histograms of the facial bone dimensions. * P<0.05 compared with T1, # P<0.05 compared with T2. T1 – at the time after operation; T2 – at 6 months postoperatively; T3 – at 36 months postoperatively. LPH – labial plate thickness. 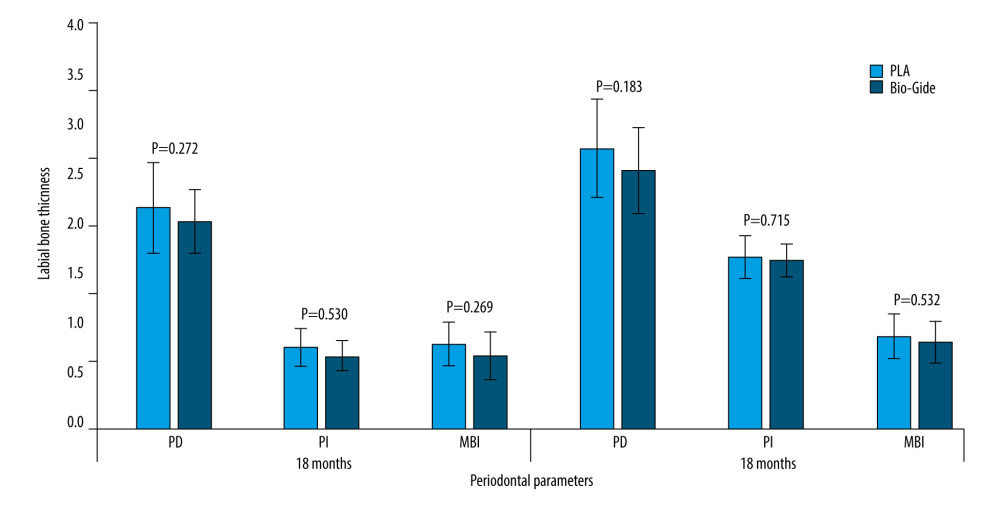 Figure 5. Histograms of peri-implant soft-tissue scores at 18 and 36 months postoperatively. PD – probing depth; PI – plaque index; MBI – modified bleeding index. (Microsoft PowerPoint 2003).
Figure 5. Histograms of peri-implant soft-tissue scores at 18 and 36 months postoperatively. PD – probing depth; PI – plaque index; MBI – modified bleeding index. (Microsoft PowerPoint 2003). References
1. Buser D, Chappuis V, Kuchler U, Long-term stability of early implant placement with contour augmentation. J Dent Res: Dec, 2013; 92(12 Suppl); 176S-82S
2. Chiapasco M, Zaniboni M, Boisco M, Augmentation procedures for the rehabilitation of deficient edentulous ridges with oral implants: Clin Oral Implants Res, 2006; 17(Suppl 2); 136-59
3. Bernstein S, Cooke J, Fotek P, Wang HL, Vertical bone augmentation: Where are we now?: Implant Dentistry, 2006; 15(3); 219
4. Donos N, Mardas N, Chadha V, Clinical outcomes of implants following lateral bone augmentation: Systematic assessment of available options (barrier membranes, bone grafts, split osteotomy): J Clin Periodontol, 2008; 35(8 Suppl); 173-202
5. Rocchietta I, Fontana F, Simion M, Clinical outcomes of vertical bone augmentation to enable dental implant placement: A systematic review: J Clin Periodontol, 2008; 35(8 Suppl); 203-15
6. Elgali I, Omar O, Dahlin C, Thomsen P, Guided bone regeneration: Materials and biological mechanisms revisited: Wiley-Blackwell Online Open, 2017; 125(5); 315-37
7. Arunyanak SP, Pollini A, Ntounis A, Morton D, Clinician assessments and patient perspectives of single-tooth implant restorations in the esthetic zone of the maxilla: A systematic review: J Prosthet Dent, 2017; 118(1); 10-17
8. Gultekin BA, Bedeloglu E, Kose TE, Mijiritsky E, Comparison of bone resorption rates after intraoral block bone and guided bone regeneration augmentation for the reconstruction of horizontally deficient maxillary alveolar ridges: BioMed Res Int, 2016; 2016; 4987437
9. Dahlin C, Sennerby L, Lekholm U, Generation of new bone around titanium implants using a membrane technique: an experimental study in rabbits: Int J Oral Maxillofac Implants, 1989; 4(1); 19-25
10. Rakhmatia YD, Ayukawa Y, Furuhashi A, Koyano K, Current barrier membranes: Titanium mesh and other membranes for guided bone regeneration in dental applications: J Prosthodont Res, 2013; 57(1); 3-14
11. Urban IA, Monje A, Guided bone regeneration in alveolar bone reconstruction: Oral Maxillofac Surg Clin North Am, 2019; 31(2); 331-38
12. Johnson TB, Siderits B, Nye S, Effect of guided bone regeneration on bone quality surrounding dental implants: J Biomech, 2018; 80; 166-70
13. Aghaloo TL, Moy PK, Which hard tissue augmentation techniques are the most successful in furnishing bony support for implant placement?: Int J Oral Maxillofac Implants, 2007; 22(Suppl); 49-70
14. Caballé-Serrano J, Munar-Frau A, Ortiz-Puigpelat O, On the search of the ideal barrier membrane for guided bone regeneration: Journal of clinical and experimental dentistry, 2018; 10(5); e477-83
15. Guo Y, Su L, Chen C, Clinical efficacy and safety of different dental prosthetic membranes in guided bone regeneration during dental implants: A meta-analysis: Comput Intell Neurosci, 2022; 31; 3245014
16. Wolff LF, Mullally B, New clinical materials and techniques in guided tissue regeneration: Int Dent J, 2000; 50(5); 235-44
17. Allan B, Ruan R, Landao-Bassonga E, Collagen membrane for guided bone regeneration in dental and orthopedic applications: Tissue Eng Part A, 2021; 27(5–6); 372-81
18. You P, Liu Y, Wang X, Acellular pericardium: A naturally hierarchical, osteoconductive, and osteoinductive biomaterial for guided bone regeneration: J Biomed Mater Res A, 2021; 109(2); 132-45
19. Di Raimondo R, Sanz-Esporrín J, Sanz-Martin I, Hard and soft tissue changes after guided bone regeneration using two different barrier membranes: An experimental in vivo investigation: Clin Oral Investig, 2021; 25(4); 2213-27
20. Benic GI, Hämmerle CH, Horizontal bone augmentation by means of guided bone regeneration: Periodontology 2000, 2014; 66(1); 13-40
21. Fiorellini JP, Kim DM, Nakajima Y, Weber HP, Osseointegration of titanium implants following guided bone regeneration using expanded polytetrafluoroethylene membrane and various bone fillers: Int J Periodontics Restorative Dent, 2007; 27(3); 287-94
22. Annunziata M, Nastri L, Cecoro G, Guida L, The use of poly-d,l-lactic acid (PDLLA) devices for bone augmentation techniques: A systematic review: Molecules, 2017; 22(12); 2214
23. He H, Lu Y, Qi J, Adapting liposomes for oral drug delivery: Acta Pharm Sin B, 2019; 9(1); 36-48
24. Huang J, Xia X, Zou Q, The long-term behaviors and differences in bone reconstruction of three polymer-based scaffolds with different degradability: J Mater Chem B, 2019; 7(48); 7690-703
25. Tyler B, Gullotti D, Mangraviti A, Polylactic acid (PLA) controlled delivery carriers for biomedical applications: Adv Drug Deliv Rev, 2016; 107; 163-75
26. Ni P, Fu S, Fan M, Preparation of poly(ethylene glycol)/polylactide hybrid fibrous scaffolds for bone tissue engineering: Int J Nanomedicine, 2011; 6; 3065-75
27. Zhang HY, Jiang HB, Ryu JH, Comparing properties of variable pore-sized 3D-printed PLA membrane with conventional PLA membrane for guided bone/tissue regeneration: Materials, 2019; 12(10); 1718
28. Ma K, Mei D, Lin X, A synthetic biodegradable polymer membrane for guided bone regeneration in bone defect: J Biomed Nanotechnol, 2021; 17(3); 456-65
29. Lambert PM, Morris HF, Ochi S, The influence of 0.12% chlorhexidine digluconate rinses on the incidence of infectious complications and implant success: J Oral Maxillofac Surg, 1997; 55(12 Suppl 5); 25-30
30. Roe P, Kan JY, Rungcharassaeng K, Horizontal and vertical dimensional changes of peri-implant facial bone following immediate placement and provisionalization of maxillary anterior single implants: A 1-year cone beam computed tomography study: Int J Oral Maxillofac Implants, 2012; 27(2); 393-400
31. Byun HY, Oh TJ, Abuhussein HM, Significance of the epithelial collar on the subepithelial connective tissue graft: J Periodontol, 2009; 80(6); 924-32
32. Löe H, The gingival index, the plaque index and the retention index systems: J Periodontol, 1967; 38(6 Suppl); 610-16
33. Lin GH, Chan HL, Wang HL, The significance of keratinized mucosa on implant health: A systematic review: J Periodontol, 2013; 84(12); 1755-67
34. Wong MC, Lo EC, McMillan AS, Validation of a Chinese version of the oral health impact profile (OHIP): Community Dent Oral Epidemiol, 2002; 30(6); 423-30
35. Elshahat A, Inoue N, Marti G, Role of guided bone regeneration principle in preventing fibrous healing in distraction osteogenesis at high speed: Experimental study in rabbit mandibles: J Craniofac Surg, 2004; 15(6); 916-21
36. Buser D, Chappuis V, Bornstein MM, Long-term stability of contour augmentation with early implant placement following single tooth extraction in the esthetic zone: A prospective, cross-sectional study in 41 patients with a 5- to 9-year follow-up: J Periodontol, 2013; 84(11); 1517-27
37. Wang D, Künzel A, Golubovic V, More about accuracy of peri-implant bone thickness and validity of assessing bone augmentation material using cone beam computed tomography: Clin Oral Investig, 2013; 17(7); 1787-88
38. Miyamoto Y, Obama T, Dental cone beam computed tomography analyses of postoperative labial bone thickness in maxillary anterior implants: Comparing immediate and delayed implant placement: Int J Periodontics Restorative Dent, 2011; 31(3); 215-25
39. Arunjaroensuk S, Panmekiate S, Pimkhaokham A, The stability of augmented bone between two different membranes used for guided bone regeneration simultaneous with dental implant placement in the esthetic zone: Int J Oral Maxillofac Implants, 2018; 33(1); 206-16
40. Sanz M, Dahlin C, Apatzidou D: J Clin Periodontol, 2019; 46(Suppl 21); 82-91
41. Floren M, Spilimbergo S, Motta A, Migliaresi C, Porous poly(D,L-lactic acid) foams with tunable structure and mechanical anisotropy prepared by supercritical carbon dioxide: J Biomed Mater Res B Appl Biomater, 2011; 99(2); 338-49
42. Butera A, Gallo S, Maiorani C, Management of gingival bleeding in periodontal patients with domiciliary use of toothpastes containing hyaluronic acid, lactoferrin, or paraprobiotics: A randomized controlled clinical trial: Applied Sciences, 2021; 11(18); 8586
43. Vale GC, Mayer MPA: Arch Oral Biol, 2021; 128; 105174
44. Butera A, Gallo S, Pascadopoli M, Home oral care of periodontal patients using antimicrobial gel with postbiotics, lactoferrin, and aloe barbadensis leaf juice powder vs. conventional chlorhexidine gel: A split-mouth randomized clinical trial: Antibiotics (Basel), 2022; 11(1); 118
45. Ghasemi N, Behnezhad M, Asgharzadeh M: Int J Dent, 2020; 2020; 8855277
Figures
 Figure 1. Diagram of the experimental protocol. In the screening period, participants were screened according to the inclusion and exclusion criteria. In the baseline period, the participants included were grouped, underwent surgery, received CBCT (cone beam CT). In follow-up period, soft-tissue and CBCT evaluations were performed. (Microsoft PowerPoint 2003).
Figure 1. Diagram of the experimental protocol. In the screening period, participants were screened according to the inclusion and exclusion criteria. In the baseline period, the participants included were grouped, underwent surgery, received CBCT (cone beam CT). In follow-up period, soft-tissue and CBCT evaluations were performed. (Microsoft PowerPoint 2003). Figure 2. Surgery procedures. A control group, B experimental group. a. Hole preparation; b. Implant placement; c. Bio-Oss material placement on the labial plate; d. Membrane coverage. (Canon EOS 60D).
Figure 2. Surgery procedures. A control group, B experimental group. a. Hole preparation; b. Implant placement; c. Bio-Oss material placement on the labial plate; d. Membrane coverage. (Canon EOS 60D). Figure 3. CBCT illustrating of labial alveolar bone thickness. (Microsoft PowerPoint 2003).
Figure 3. CBCT illustrating of labial alveolar bone thickness. (Microsoft PowerPoint 2003). Figure 4. The radiographic images (Ruike Medical Instruments Co., Ltd.) and histograms of the facial bone dimensions of the 2 groups at different times (Microsoft PowerPoint 2003). A experimental group, B control group, C histograms of the facial bone dimensions. * P<0.05 compared with T1, # P<0.05 compared with T2. T1 – at the time after operation; T2 – at 6 months postoperatively; T3 – at 36 months postoperatively. LPH – labial plate thickness.
Figure 4. The radiographic images (Ruike Medical Instruments Co., Ltd.) and histograms of the facial bone dimensions of the 2 groups at different times (Microsoft PowerPoint 2003). A experimental group, B control group, C histograms of the facial bone dimensions. * P<0.05 compared with T1, # P<0.05 compared with T2. T1 – at the time after operation; T2 – at 6 months postoperatively; T3 – at 36 months postoperatively. LPH – labial plate thickness. Figure 5. Histograms of peri-implant soft-tissue scores at 18 and 36 months postoperatively. PD – probing depth; PI – plaque index; MBI – modified bleeding index. (Microsoft PowerPoint 2003).
Figure 5. Histograms of peri-implant soft-tissue scores at 18 and 36 months postoperatively. PD – probing depth; PI – plaque index; MBI – modified bleeding index. (Microsoft PowerPoint 2003). In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387