18 February 2023: Clinical Research
Comparison of 3 Enzyme-Linked Immunoassay Methods to Evaluate Serum Concentrations of Infliximab and Antibodies to Infliximab in 32 Patients with Moderate to Severe Inflammatory Bowel Disease
Tomislav Jukic1CDEF, David Drobne2ABCDE, Sasa Pusavec3BF, Alojz Ihan3AE, David Stubljar4ACDE*, Andrej Starc5DEDOI: 10.12659/MSM.939084
Med Sci Monit 2023; 29:e939084
Abstract
BACKGROUND: Monitoring of trough levels and anti-drug antibodies is important when patients with inflammatory bowel disease (IBD) are treated with anti-TNF biologics due to guided therapeutic decisions. The comparability of 3 ELISA tests for detection of the lowest serum concentration of infliximab (IFX) or antibodies to IFX (ATIs) was evaluated.
MATERIAL AND METHODS: Two commercial assays for measuring IFX levels were compared with the in-house (UHL) test. ATIs were measured with 1 commercial test and compared to the in-house test. According to the guidelines, IFX levels were within the range of 3 to 7 µg/mL.
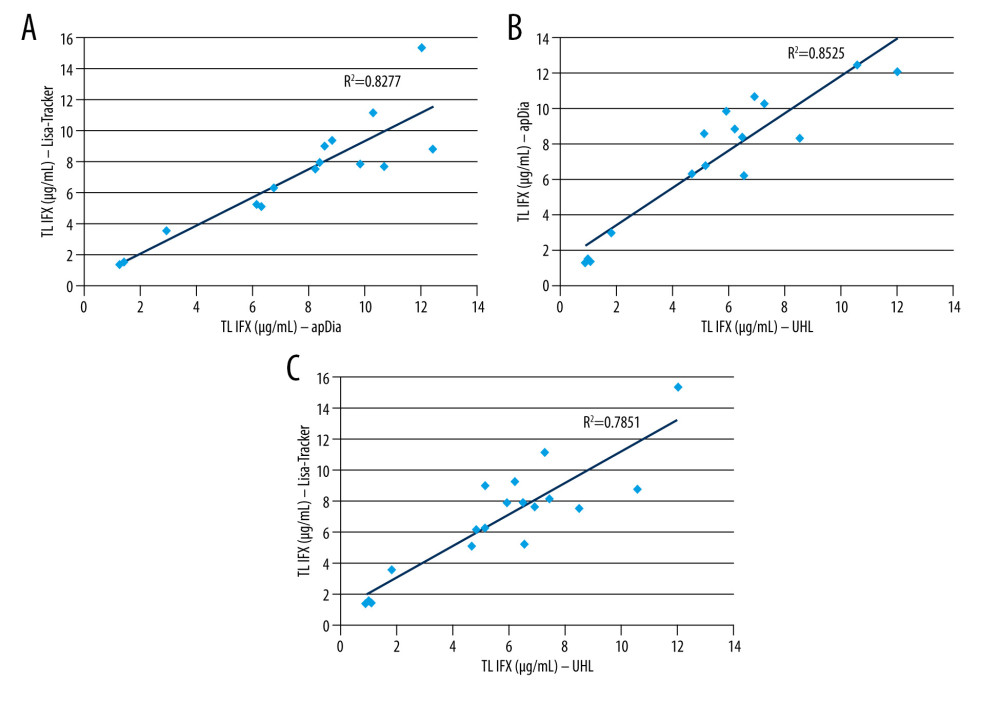
RESULTS: The decision to continue therapy would be the same for 11 out of 16 patients when comparing the apDia Infliximab ELISA and UHL test, and for 12 out of 18 patients when comparing the Lisa-Tracker and in-house UHL test. Linear correlations between the tests were R=0.92 (UHL and apDia), R=0.91 (apDia and Lisa-Tracker), and R=0.89 (UHL and Lisa-Tracker) with P<0.001, respectively.
CONCLUSIONS: As the IFX levels are important for decisions on further therapy, detectable IFX levels realistically reflect the presence of the drug in the patients’ blood and thus control inflammatory activity. The tests were found as comparable and performed well in this aspect and might be used in everyday clinical practice.
Keywords: Antibodies, Enzyme-Linked Immunosorbent Assay, infliximab, Materials Testing, Humans, Tumor Necrosis Factor Inhibitors, Drug Monitoring, Inflammatory Bowel Diseases
Background
Inflammatory bowel disease (IBD), which encompasses Crohn disease (CD) and ulcerative colitis (UC), is a lifelong gastrointestinal disease that takes place as alternating periods of disease flares and intermediate periods of disease remission. CD can affect the entire intestine from the mouth to the anus, while UC is restricted to the colon only [1].
The approach to treating IBD has changed significantly over the last 20 years [2]. In the past, the goal of treatment was only to alleviate symptoms, but today we want to achieve deep remission, that is, complete elimination of disease symptoms, normalization of biochemical and fecal indicators of inflammation, and healing of the intestinal mucosa (clinical, laboratory, and endoscopic remission of the disease) [3,4]. Several treatment options such as 5-aminosalicylic acid, corticosteroids, immunosuppressants, and biologics have been introduced [5]. With the introduction of biologics, the treatment modality has changed dramatically, as they specifically target molecules associated with the course of the disease. The first approved biological drug for the treatment of CD and UC was infliximab (IFX) [6], a chimeric antibody that belongs to the group of tumor necrosis factor (TNF)-α inhibitors [7].
Biologics have brought new opportunities for safe and effective treatment. For instance, regular IFX therapy leads to endoscopic mucosal healing, improves quality of life, and reduces hospitalization and surgery [8]. Although biologics usually provide strong control over the disease, some patients do not respond to the treatment (primary non-responsive patients), or the drug eventually loses its effect (secondary non-responders). One of the main reasons for this is the emergence of anti-drug antibodies (ADAs), which occur due to the immunogenicity of the biological drug. ADAs lower the serum level of a drug (the lowest serum concentration of a drug during treatment, trough level [TL]) by inactivating or accelerating its excretion from the body by urine [2]. A major limitation in IFX treatment is the development of antibodies to IFX (ATI) in some patients, which is due to the immunogenic potential of the murine variable region in the IFX molecule [9].
Therapeutic drug monitoring (TDM) and monitoring of ATI enable us to optimize patient treatment. With this approach, we can identify patients who have very low or undetectable serum IFX concentrations, due to ATI activity. These may develop at any time during treatment [10]. However, there are many different methods for determining TL IFX and ATI. The most used are enzyme-linked immunosorbent assay (ELISA), radioimmunoassay, and homogenous mobility shift assay [11]. As the monitoring of TL IFX and ATI is becoming a part of the clinical practice, competitive and precision tests are crucial for the diagnosis and decision-making of the patient’s subsequent therapy [12–15]. However, the discrepant accuracy rates of different assays for TDM might represent a challenge in improving the management of patients in clinical practice. ELISA assays vary for the quantitative determination of ATI in serum samples with different limits of detection.
Previously, assays have been tested and some showed acceptable accuracy within the quantification range. Although all tested assays were assessed as suitable for TDM, significant differences were observed. One with the best accuracy has been identified as the apDia assay, when compared to in-house Sanquin Diagnostics, Promonitor® IFX (Progenika Biopharma SA®), and the Lisa-Tracker® Duo infliximab (Theradiag®) assay. For instance, Promonitor measured the lowest IFX concentrations, and the apDia the highest [14]. Accordingly, the Lisa Tracker assay has already been evaluated in monitoring the concentration of IFX biosimilars and it showed valuable results [16]. Meanwhile, there are reports that showed a perfect comparisons alignment between 3 different assays (Ridascreen®, Promonitor, and Lisa-Tracker). The main drivers of different assessments are differences in (i) TNF coating, (ii) immune complexes, and (iii) interference with other anti-TNFα agents. Evaluation results vary significantly and are quantitatively interchangeable. This might lead to divergent clinical decisions [17]. Thus, comparisons of different assays are never obsolete.
The purpose of this study was thus to validate and determine the comparability of 3 ELISA tests (2 commercially available and 1 non-commercially available) to detect the lowest serum IFX concentration in IBD patients and to establish the mutual comparability of 2 ELISA tests, commercially available and non-commercially available, for the determination of ATI concentrations.
Material and Methods
PATIENTS:
Overall, 18 patients with moderate to severe IBD who were on IFX maintenance therapy at the time of sampling were tested and enrolled in the study, and 32 of their samples were tested. Inclusion criteria were age >18 years and confirmed diagnosis of UC or CD based on clinical, endoscopic, and histological examinations using European Crohn’s and Colitis Organization (ECCO) criteria. Patients were bio-naïve prior to the IFX therapy and settled for maintained disease remission. Prior to biological therapy, they were treated with conventional options, such as azathioprines, steroids, or oraminosalicylates, but the efficacy was lacking; therefore, these patients were eligible for biological therapy. Electronic clinical records of patients were reviewed and data on sex, age at diagnosis, age at first IFX infusion, disease phenotype (CD, UC), C-reactive protein (CRP) value, fecal calprotein value, endoscopic disease activity, IFX doses, patient body weight, and final patient status (resumed IFX therapy/discontinued IFX therapy) were obtained (Table 1).
TREATMENT OF PATIENTS WITH IFX:
Patients who were treated with IFX were treated according to the drug label, with 5 mg/kg of their body mass. IFX was given as an intravenous infusion. In addition to the initial dose, the patient received IFX at a dose of 5 mg/kg in weeks 2 and 6 after the first dose. This was followed by a maintenance phase in which the patient received an IFX infusion of 5 mg/kg every 8 weeks (the first maintenance infusion was therefore given 14 weeks after the initial dose).
ETHICAL ASPECTS:
All patients consented to the study with their signature. All information obtained were anonymous and not personally identifiable. The National Ethics Committee of the Republic of Slovenia approved the study (no. 45/5/14).
DETERMINATION OF IFX SERUM CONCENTRATIONS:
The lowest serum concentrations of IFX (TL IFX) were measured for each patient. The measurements were taken just before the next drug application. According to the research, IFX levels are currently considered to be associated with the best clinical outcome within the interval of 3 to 7 μg/mL. Monitoring of IFX levels thus allows the optimization of the treatment [12]. The quantitative concentrations of serum IFX (Remicade®, anti-TNF-α) were determined with 2 commercially available ELISA tests: apDia Infliximab ELISA (apDia bvba, Turnhout, Belgium) and Lisa-Tracker Duo Infliximab (Theradiag®, Croissy Beaubourg, France). The results were then compared with each other and with the in-house test from the University of Leuven (UHL test). The UHL test has been taken as a reference, since has been developed by University of Leuven and proven in several scientific and clinical reports and serves as a guiding treatment algorithm for patients with IBD [12].
UHL TEST FOR INFLIXIMAB AND ANTI-INFLIXIMAB:
The UHL test is based on a methodology of a simple ELISA. Meanwhile, ATI are measured by double-antigen ELISA capturing IFX-coated microplates and detection by peroxidase-conjugated IFX. This assay was standardized using a mouse mAb against human immunoglobulin G [13,14]. IFX levels were measured according to the following. Micro-plate wells were coated with TNF-α. The wells in the plates were initially blocked for 2 h at room temperature with a phosphate buffered saline (PBS) solution containing 1% bovine serum albumin (BSA). Samples were also diluted in a mixture of PBS/1% BSA and incubated for 2 h at 37°C. The secondary antibody that was used was a monospecific rabbit polyclonal antibody conjugated to horseradish peroxidase (HRP), followed by incubation at room temperature for 1 h. Plates were developed using 400 lg/mLo-phenylenediamine (Acros Organics, Geel, Belgium) and 0.003% (v/v) H2O2 in 0.1 M sodium citrate 0.2 M disodium phosphate buffer pH 5. The reaction was stopped with 2 M H2SO4. Absorption at 490 nm was measured. Results were related to a titration curve of IFX on each plate. The cut off for an IFX-positive sample was 0.3 mg/L.
Moreover, to measure antibodies to IFX, an in-house-developed ELISA was used. High binding 96-well plates were coated for 72 h with IFX (Janssen Biologics, Leiden, Netherlands) at 4°C. Plates were blocked with PBS/1% BSA for 2 h at room temperature, and samples were diluted in PBS/0.1% BSA/0.002% (v/v) Tween 80 and incubated overnight at 4°C. Detecting antibody HRP-linked IFX (made in house) was added into the wells, and plates were incubated at room temperature for 2 h. Plates were developed using 400 lg/mLo-phenylenediamine (Acros Organics) and 0.003% (v/v) H2O2 in 0.1 M sodium citrate 0.2 M disodium phosphate buffer pH 5. The reaction was stopped with 4 M H2SO4. Absorption at 490 nm was measured. Results were related to a titration curve of monospecific rabbit polyclonal anti-body to IFX in each plate. The cutoff for an ATI-positive sample was 1 mg/L.
TEST APDIA INFLIXIMAB ELISA:
The test was performed according to the manufacturers’ instructions. Microtiter strips coated with TNF-α were incubated with calibrators, controls, and diluted patient samples. During this incubation step, IFX bound specifically to the TNF-α on the solid phase. After removal of the unbound serum proteins by a washing procedure, the antigen-antibody complex in each well was detected with specific peroxidase-conjugated monoclonal antibody (clone 6B7, developed at the KU Leuven) directed to IFX. After removal of the unbound conjugate, the strips were incubated with a chromogenic solution containing tetramethylbenzidine and hydrogen peroxide: a blue color developed in proportion to the amount of immunocomplex bound to the wells of the strips. The enzymatic reaction was stopped by the addition of 0.5 M H2SO4, and the absorbance values at 450 nm were determined. The absorbances of 5 calibrators were measured and plotted on the y-axis. Corresponding concentrations were plotted on the x-axis to obtain a standardized curve. The absorbance value of the sample (y-axis) was used to determine IFX concentration in the sample and was interpolated to the x-axis according to the calibration curve. The value was then multiplied by the dilution factor. In order for the test results to be valid, criteria according to the manufacturer’s instructions had to be met: zero calibrator absorption value (CAL0) <0.080; highest calibrator absorbance (CAL5) value >1.400; first positive control (CTL1) concentration value=3 μg/mL, range 2 to 4 μg/mL; second positive control (CTL2) concentration value=7 μg/mL, range 5 to 10 μg/mL.
TEST LISA-TRACKER DUO INFLIXIMAB:
The test was performed according to the manufacturers’ instructions. The test has been validated to monitor drug levels and anti-drug antibody levels of any biological drug that contains the active substance IFX, that is the original drug Remicade, and any biosimilar drug, such as CT-P13 (Remsima or Inflectra) and SB2 (Flixabi or Renflexis). Thus, this test was used to measure trough levels of IFX and levels of anti-IFX antibodies. The TNF-α was coated onto a polystyrene microtiter plate. First, the diluted sample was added to the TNF-α-coated well, which allowed it to bind. After incubation, unbound proteins were removed by washing. Biotinylated polyclonal rabbit anti-IFX antibody was added. After incubation, unbound antibodies were removed by washing. Then horseradish peroxidase-labeled streptavidin was added. The streptavidin binds to the complex formed with biotinylated anti-IFX antibodies. After incubation, the wells were washed again to eliminate any excess of conjugate. The bound enzyme was revealed by addition of substrate TMB (3,3′,5,5′ tetramethylbenzidine). The color intensity was proportional to the amount of IFX. Adding H2SO4 stops the enzymatic reaction. After stopping the reaction by H2SO4, the optical density was read by a spectrophotometer at 450 nm. A range of calibration allows us to define the quantity of IFX of each patient sample, expressed in μg/mL. The absorbance values of the 5 standards were measured and plotted on the y-axis. Corresponding concentrations (μg/mL) were plotted on the x-axis. Using the standards, a polynomial curve of degree 4 was determined, from which, according to the measured absorbance, the concentration of IFX in the sample was read directly. The obtained value was multiplied by the dilution factor. The test can detect IFX concentrations between 0.1 and 8 μg/mL. In order for the test results to be valid, criteria according to the manufacturer’s instructions had to be met: absorption value of standard 1 ≥0.8 and the IFX concentration in the positive control should be within the range recorded on the vial.
Moreover, undetectable sample concentrations were set as follows: undetectable TL IFX from the UHL test was defined as TL IFX <0.30 μg/mL; undetectable TL IFX from the apDia test was defined as TL IFX <0.50 μg/mL and undetectable TL IFX from the Lisa-Tracker test was defined as TL IFX <0.10 μg/mL.
DETERMINATION OF ANTI-IFX SERUM CONCENTRATIONS:
In clinical practice, ATI are determined for patients who have undetectable serum IFX concentrations (<0.30 μg/mL). One of the reasons for low serum IFX concentrations are anti-IFX antibodies that accelerate drug clearance and neutralize IFX function [15]. Quantitative concentrations of ATI were determined using the Lisa-Tracker Duo Infliximab assay. The results were compared with those measured with the in-house UHL test.
TEST LISA-TRACKER DUO INFLIXIMAB:
The test was performed according to the manufacturers’ instructions. The IFX was coated onto a polystyrene microtiter plate. First, the diluted sample was added to the antibody-coated well, which allowed it to bind. After incubation, unbound proteins were removed by washing. Biotinylated IFX was added. After incubation, unbound antibodies were removed by washing. Then horseradish peroxidase-labeled streptavidin was added. The streptavidin binds to the complex formed with biotinylated IFX. After incubation, the wells were washed again to eliminate any excess of conjugate. The bound enzyme was revealed by addition of substrate TMB (3,3′,5,5′ tetramethylbenzidine). The color intensity is proportional to the amount of anti-IFX antibodies. Adding H2SO4 stops the enzymatic reaction. After stopping the reaction by H2SO4, the optical density was read by a spectrophotometer at 450 nm. A range of calibration allows us to define the quantity of anti-IFX antibodies of each patient sample, expressed in ng/mL. Due to the high sensitivity of the test, but a narrow range for ATI determination (detecting range of 10–200 ng/mL), the samples were diluted. Samples were diluted 1: 100 for Lisa-Tracker. Samples with undetectable ATI (<1 μg/mL) with UHL were diluted 1: 100 and 1: 10. The measurements were executed, and undetectable samples were diluted for the second time. Those samples were then diluted 1: 32 for Lisa-Tracker, and samples with undetectable ATIs (<1 μg/mL) with UHL were diluted 1: 32 and 1: 16, respectively.
The absorbance values of 5 standards were measured and plotted on the y-axis. Corresponding concentrations (ng/mL) were plotted on the x-axis. Using standards, a standard polynomial curve was determined from which the concentration of ATI (ng/mL) in the sample according to the measured absorbance was read. The value was obtained and multiplied by the dilution factor.
Moreover, undetectable sample concentrations were set as follows: undetectable ATI with the UHL test was defined as ATI <1 μg/mL. Low ATI with the UHL test were defined as ATI <8 μg/mL, and high ATI levels were defined as ATI >8 μg/mL. The presence of ATI with the Lisa-Tracker test was defined as ATI >0.000.
STATISTICAL METHODS:
Statistical analyses were performed using SPSS (IBM Corp, Armonk, NY, USA). Series of
Results
Overall, 32 serum samples were analyzed with 3 different tests and included in the study. On average, patients with CD at the time of diagnosis were 23.8 years old (the youngest was 14 years old and the oldest was 57 years old), and patients with UC were an average of 31.9 years at diagnosis (the youngest was 9 years old, the oldest was 58 years old). On average, the first IFX infusion was given 8.3 years after IBD diagnosis.
Concentrations of IFX were measured in the serum of 18 patients with all 3 tests: apDia Infliximab ELISA, Lisa-Tracker Duo Infliximab immunoassay, and in-house UHL test (Table 2). All the tests were valid, as validation criteria were met according to the manufacturers’ instructions.
The results were then divided into 3 subgroups, according to the measured TL IFX; (i) TL IFX <3 μg/mL, (ii) TL IFX 3–7 μg/mL, and (iii) TL IFX >7 μg/mL. These groups can give guidance how to continue with the therapy. Tables 2 and 3 present comparisons according to the subgroups.
Discussion
The determination of IFX and ATI levels is increasingly becoming a part of clinical practice, as with their help we can identify patients with a presence of low IFX and ATI levels and therefore optimize and adjust treatment so that patient’s TL IFX are within the proposed interval of 3 to 7 μg/mL [16,17]. The development of ADAs leads to loss of response in patients on anti-TNF treatment for CD or UC. The reasons for development of ADAs and ATI and heterogeneity in titers vary and are also related to genetics [20–22]. However, the development of ATI guides clinical decisions in patients, such as dose de-escalation or escalation, re-initiation of therapy, the application of combinational therapy with immunomodulator and corticosteroids, or even treatment cessation and switching to another mode of action. Thus, the laboratory evaluations of IFX concentration and ATI are useful biomarkers in detecting loss of response and are important in guidance and eventual optimization of treatment outcome [23,24]. However, their detection and evaluation highly depend on the assay used, as the use of different assays are not always interchangeable [19,23]. The aim of this research was thus to validate and compare 2 commercially available tests for measuring IFX levels (Lisa-Tracker Duo Infliximab test and apDia Infliximab ELISA) with a commercially unavailable test developed at the University of Leuven, the UHL test, and to compare a commercially available ATI test (Lisa-Tracker Duo Infliximab test) with a commercially unavailable ATI assay (UHL test) for detecting levels of ATI.
In our analysis, both commercially available tests, apDia and Lisa-Tracker, showed slightly higher concentrations of IFX in 5 samples when compared to the UHL test (Tables 3, 4). The UHL test showed levels from the second subgroup (3–7 μg/mL), but both apDia and Lisa-Tracker showed levels higher than 7 μg/mL. Therefore, according to the results by apDia and Lisa-Tracker, the treatment decision in these 5 patients would have led to reduced IFX dosing or increased dosing interval, while therapy should have remained the same with respect to the results of the UHL test. When other parameters were considered, in 2 out of 5 sample patients had CRP <5 mg/L and were in endoscopic remission, which suggested the appropriate decision to reduce IFX dosing or increase the IFX dosing interval. For the other 3 samples, no information on the patients’ CRP levels and endoscopic disease activity were obtained, so no conclusion was made, and patients remained on the therapy. When comparing the UHL and Lisa-Tracker tests, there was also 1 discrepancy in the sample having an IFX level of <3 μg/mL with UHL, and IFX level between 3 μg/mL and 7 μg/mL measured with Lisa-Tracker (Table 4). No information on CRP levels and endoscopic activity was obtained, so no actions could have been taken on the appropriate therapy. The results of different assays showed differences in some of the patients; thus, the impact on clinical decision might be different, but according to our opinion, minimal or almost irrelevant. The alignment of patients with commercial tests was high. Namely, the main differences between all tests were found in patients with higher levels of IFX and in remission. The discrepancy could be because of the higher upper quantification limit for the 2 commercial assays when compared with that of UHL. Discrepancies between the tests are common situations and were also obtained by other reports [16,19]. Bertin et al [19] evaluated 3 available tests: Lisa-Tracker Duo Infliximab, Ridascreen IFX Monitoring by R-Biopharm AG, and Promonitor IFX by Progenika Biopharma SA, and reported that the assays were not quantitatively interchangeable due to variations in some results. They also found the differences in group classification that could lead, for some patients, to divergent therapeutic decisions. Overall, from our findings, in 11 (68.8%) out of 16 specimens, the decision to continue patient therapy would be the same based on the results of the UHL and apDia tests. Meanwhile, the decision would be the same for 12 (66.7%) out of 18 samples based on UHL and Lisa-Tracker test results. This is showing almost 70% concordance between our tests. Similar concordance was shown by Schmitz et al [16], who tested Lisa-Tracker, apDia, and Progenika and compared them with their in-house test. The authors showed the highest IFX levels with apDia and showed 70% concordance between the tests.
Linear regression was calculated to determine the correlations between the tests. When comparing the apDia and Lisa-Tracker tests, Pearson’s R correlation coefficient was 0.91 (95% CI: 0.75–0.97;
Moreover, in our study, patients’ serum ATI concentrations were determined by the Lisa-Tracker Duo Infliximab and UHL test (Table 5). Only 14 samples from patients with IBD were obtained and were possible to analyze. The tests were performed twice and were valid on both occasions, as the criteria for validity were met. The determination of ATI with the Lisa-Tracker test has a main obstacle of choosing the right dilution factor so that the samples do not exceed the upper limit of ATI detection (200 ng/mL) or in order not to dilute the sample too much and get undetectable levels of ATI. For instance, in our batch, 9 samples had a dilution of 1: 32, which proved appropriate, but the dilution of 1: 32 was too high for 4 samples, and consequently no detectable ATI were obtained. For 1 of these samples, ATI was determined with dilutions of 1: 10 and 1: 16, which were therefore more appropriate than dilutions of 1: 32. In contrast, for 2 samples, the 1: 32 dilution was still too low, and the results were above the detection limit (excess) (Table 5). In both samples, the 1: 100 dilution was too low to obtain a quantitative concentration of ATI (but in 1 of the samples ATI concentration was only slightly above the detection limit). The manufacturer has, however, ensured that ATI dilution <10 ng/mL means no ATI, while ATI=200 ng/mL is already considered as a strong positive and no additional dilution is required. The Lisa-Tracker test in terms of ATI is therefore more of a qualitative than quantitative method.
As Bertin et al [19] found discrepancies between assays for classification of TL IFX into concentration ranges, their pairwise qualitative comparison showed a perfect correlation between the 3 pairs for ADA detection. In the case of IFX concentration measurements, different tests can be compared directly, since IFX is used as the calibrator. In contrast, the tests used to measure ATI do not have a universal calibrator. First generation ELISAs for the determination of ATI, including UHL, use polyclonal anti-IFX antibodies as the calibrator, while second generation, which includes Lisa-Tracker, use monoclonal anti-IFX antibodies as the calibrator. First generation polyclonal antibody ELISAs are cross-reactive with other anti-TNFs and, because of their polyclonal nature, they bring variability to the assay. The second generation tests are more specific than the first generation tests, as there is no cross-reactivity with other anti-TNFs, and they are also more sensitive, since this method is expected to measure ATI in the presence of IFX, which is not possible with the tests from the first generation [26]. Therefore, due to the lack of standardization between the 2 tests, the tests were compared qualitatively and not quantitatively. Table 6 shows the match between the 2 methods.
The Lisa-Tracker test measured low ATI concentrations in 3 patients who had otherwise undetectable ATI with UHL. The reason could be the greater sensitivity of the Lisa-Tracker test, as the levels were shown as low. However, there might be a concern regarding the Lisa-Tracker test, which in fact could have determined false-positive ATI results. Although it is not reasonable to compare the quantitative tests, it can be observed that the Lisa-Tracker was able to measure approximately 3 times lower concentrations of ATI than the UHL test. On the other hand, the UHL test can capture other, nonspecific antibodies as well and cover a wider range of antibodies than Lisa-Tracker, which might mean a lower specificity for the UHL. Here, we would like to mention a study from Jentzer et al [18], who tested the capability of Lisa-Tracker in detecting TL and ATI levels for biosimilar IFX. They proved that the Lisa-Tracker test can be effectively used in monitoring of patients treated with biosimilar IFX and is not strictly limited to the original molecule. Comparing the methods only qualitatively, the correlation in our study between the 2 tests was good, since no false-negative ATI was measured with Lisa-Tracker and the concordance was almost 79%. Regarding the measurement of ATI, here we would also like to express the opinion that the same assay should always be used during the follow-up of patients. One of the most probable reasons for the occurrence of ADAs is the generation of immune complexes. The ADAs are thus present in both, free and in bound form to the immune complexes. Consequently, the concordance of 79% presents an extremely comparable result, as the assays might be subjected to the interference with the drug itself, whereas the free circulating form of IFX limits the detection of ADAs, for instance showing low levels of ATI, while they are present in much higher serum concentration. Therefore, sticking with one assay is more prone to lead to a clinical decision than is using different assays that are not interchangeable due to the influence of immune complexes in patients with IBD.
The existing knowledge and evidence support the use of reactive TDM in secondary non-responders with CD and UC, but we are still lacking findings if regular/proactive TDM can improve patients’ clinical outcomes. It has already been shown that proactive TDM is associated with fewer surgeries and higher rates of mucosal healing than disease management without TDM [27]. Thus, measurements of TL IFX and ATI may help in better disease management. However, as there will always be discrepancies between different tests, shown in the present study and by other investigators, we recommend using only 1 standardized assay for the follow-up of patients with IBD.
Our analysis had some limitations, including first, its small sample size. We should include more patients with IBD and their serum samples, but it must be emphasized that the execution of all 3 tests were done in 2 separate laboratories and it was almost logistically impossible to have included a significant batch of samples. In addition to the small number of samples, a limitation of ATI comparison methods is also that we were not able to include more samples with detectable levels. Larger sample size could determine if it is truly possible to measure ATI in the presence of IFX with the Lisa-Tracker test.
Conclusions
The commercially available apDia Infliximab ELISA and Lisa-Tracker Duo Infliximab test were comparable to the commercially unavailable Belgian test from the University of Leuven (UHL test). The IFX levels were established as important to decide on further therapy. The study showed that the decision to continue therapy would be the same in approximately 70% of patients according to the practice that IFX levels are within the range of 3 to 7 μg/mL, regardless of the tests. The detectable IFX levels realistically reflect the presence of the drug in the patients’ blood and thus control inflammatory activity. Therefore, all 3 tests performed well in this aspect and can be used in daily clinical practice for IFX quantification. Also, according to the findings from our study, we propose to stick with 1 assay for monitoring the outcome of patients with IBD, and this might be commercial tests, which provided accurate results.
Tables
Table 1. Basic characteristics of patients whose samples were included in analysis. Table 2. Measured IFX levels according to the 3 methods.
Table 2. Measured IFX levels according to the 3 methods.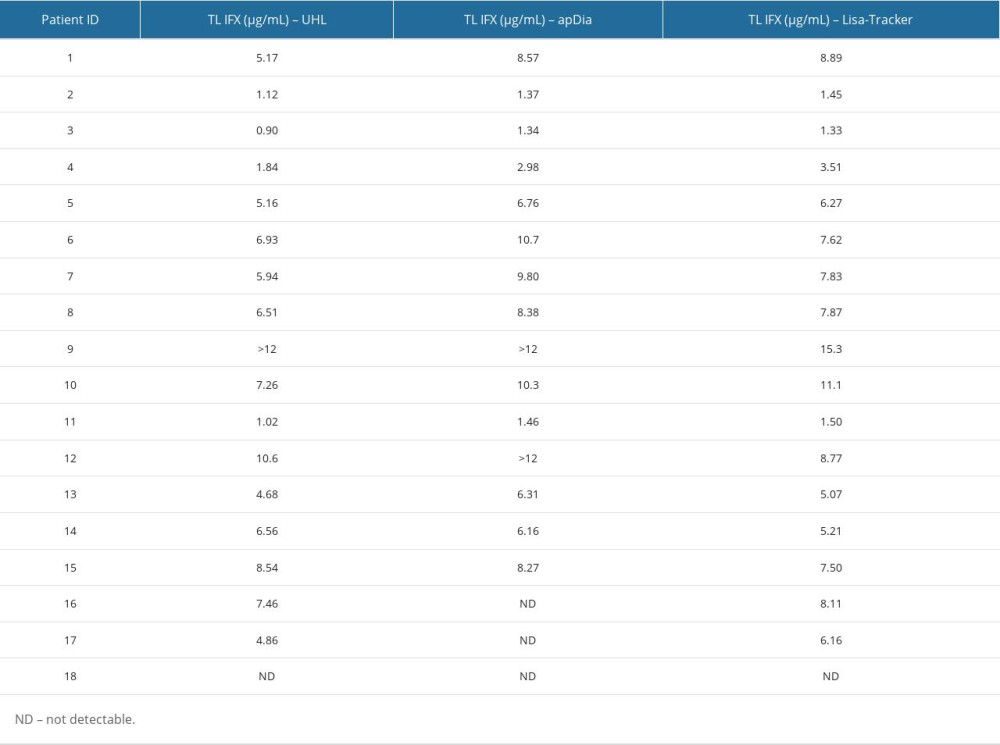 Table 3. Comparison of samples between University of Leuven (UHL) test and apDia test: number of samples in a respective group.
Table 3. Comparison of samples between University of Leuven (UHL) test and apDia test: number of samples in a respective group.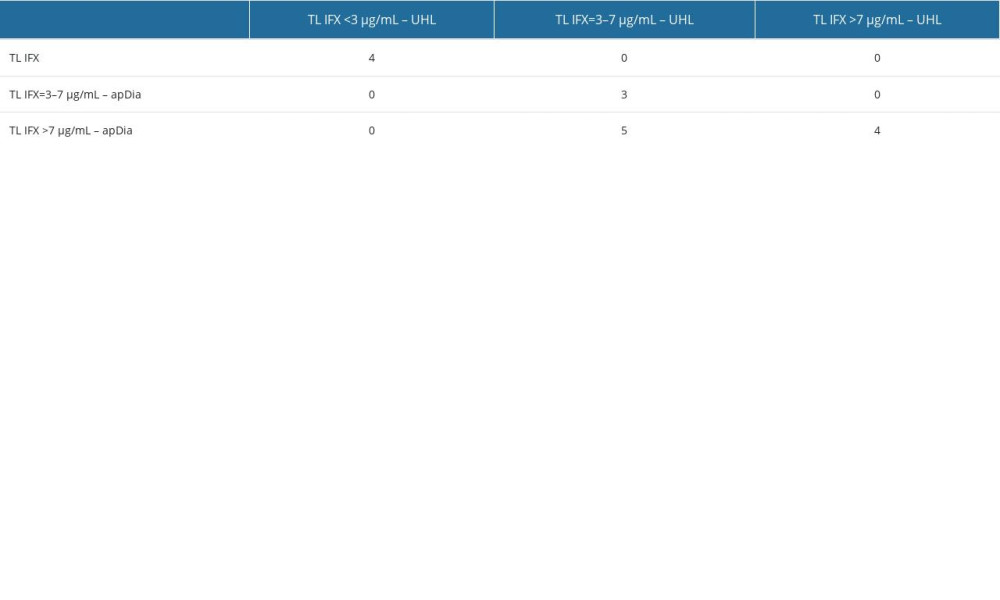 Table 4. Comparison of samples between University of Leuven (UHL) test and Lisa-Tracker test: number of samples in a respective group.
Table 4. Comparison of samples between University of Leuven (UHL) test and Lisa-Tracker test: number of samples in a respective group.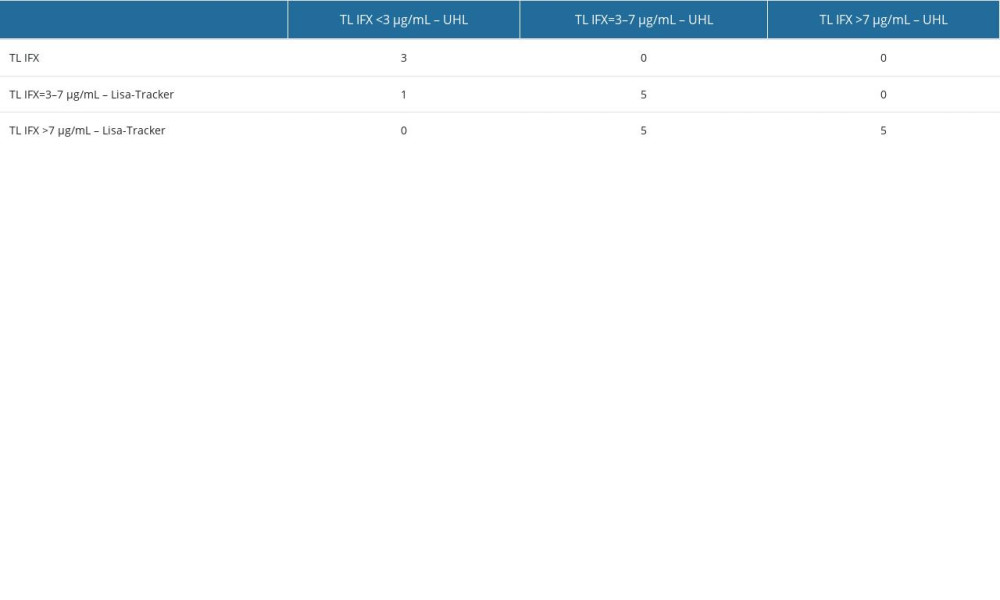 Table 5. Comparison of antibodies to IFX concentrations between University of Leuven (UHL) and Lisa Tracker.
Table 5. Comparison of antibodies to IFX concentrations between University of Leuven (UHL) and Lisa Tracker.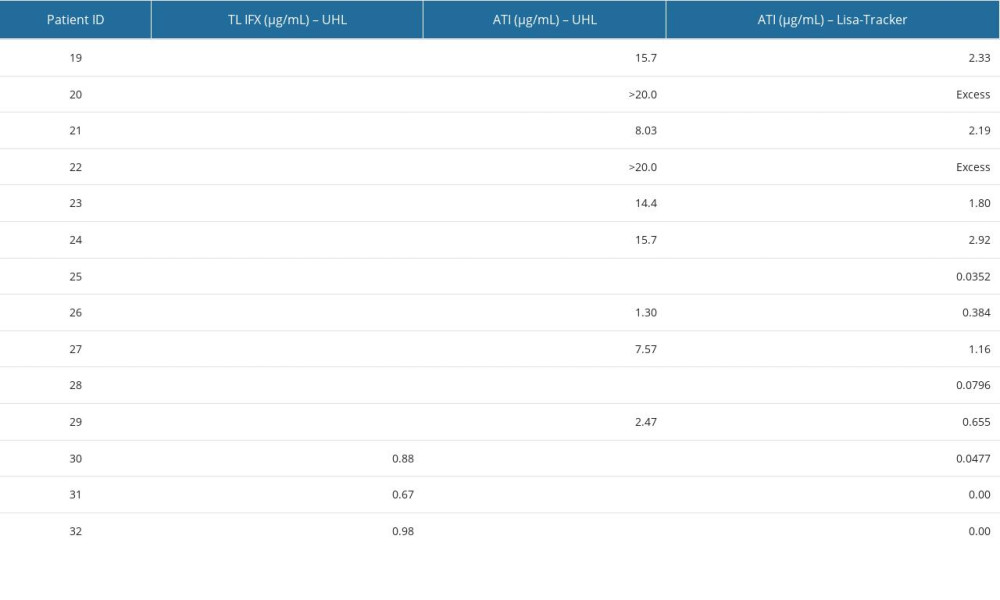 Table 6. Number of samples with detectable (+) and undetectable (−) antibodies to IFX for a respective test.
Table 6. Number of samples with detectable (+) and undetectable (−) antibodies to IFX for a respective test.
References
1. Basso PJ, Fonseca MT, Bonfá G, Association among genetic predisposition, gut microbiota, and host immune response in the etiopathogenesis of inflammatory bowel disease: Braz J Med Biol Res, 2014; 47; 727-37
2. Gils A, Combining therapeutic drug monitoring with biosimilars, a strategy to improve the efficacy of biologicals for treating inflammatory bowel diseases at an affordable cost: Dig Dis, 2017; 35; 61-68
3. Pariente B, Cosnes J, Danese S, Development of the Crohn’s disease digestive damage score, the Lémann score: Inflamm Bowel Dis, 2011; 17; 1415-22
4. Han J, Wang J, Wang JH, How to achieve deep remission in treatment of inflammatory bowel disease: J Tradit Chin Med, 2013; 33; 549-52
5. Ardizzone S, Bianchi Porro G, Biologic therapy for inflammatory bowel disease: Drugs, 2005; 65; 2253-86
6. Kornbluth A, Infliximab approved for use in Crohn’s disease: A report on the FDA GI Advisory Committee conference: Inflamm Bowel Dis, 1998; 4; 328-29
7. Yapali S, Hamzaoglu HO, Anti-TNF treatment in inflammatory bowel disease: Ann Gastroenterol, 2007; 20; 48-53
8. Lichtenstein GR, Yan S, Bala M, Infliximab maintenance treatment reduces hospitalizations, surgeries, and procedures in fistulizing Crohn’s disease: Gastroenterol, 2005; 128; 862-69
9. Nanda KS, Cheifetz AS, Moss AC, Impact of antibodies to infliximab on clinical outcomes and serum infliximab levels in patients with inflammatory bowel disease (IBD): A meta-analysis: Am J Gastroenterol, 2013; 108; 40-47
10. Vande Casteele N, Feagan BG, Gils A, Therapeutic drug monitoring in inflammatory bowel disease: Current state and future perspectives: Curr Gastroenterol Rep, 2014; 16; 378
11. Vande Casteele N, Assays for measurement of TNF antagonists in practice: Frontline Gastroenterol, 2017; 8; 236-42
12. Vande Casteele N, Ferrante M, Van Assche G, Trough concentrations of infliximab guide dosing for patients with inflammatory bowel disease: Gastroenterol, 2015; 148; 1320-29
13. Ternant D, Mulleman D, Degenne D, An enzyme-linked immunosorbent assay for therapeutic drug monitoring of infliximab: Ther Drug Monit, 2006; 28; 169-74
14. Van Assche G, Magdelaine-Beuzelin C, D’Haens G, Withdrawal of immunosuppression in Crohn’s disease treated with scheduled infliximab maintenance: A randomized trial: Gastroenterology, 2008; 134; 1861-68
15. Vande Casteele N, Khanna R, Levesque BG, The relationship between infliximab concentrations, antibodies to infliximab and disease activity in Crohn’s disease: Gut, 2015; 64; 1539-45
16. Schmitz EM, van de Kerkhof D, Hamann D, Therapeutic drug monitoring of infliximab: Performance evaluation of three commercial ELISA kits: Clin Chem Lab Med, 2016; 54; 1211-19
17. Curci D, Lucafò M, Cifù A, Determination of serum infliximab concentration by point-of-care devices in children with inflammatory bowel disease: J Pediatr Gastroenterol Nutr, 2019; 69; 474-79
18. Jentzer A, Berger AE, Labetoulle R, Evaluation of infliximab and anti-infliximab LISA-TRACKER immunoassays for the therapeutic drug monitoring of SB2 infliximab biosimilar: Ther Drug Monit, 2018; 40; 749-53
19. Bertin D, Serrero M, Grimaud JC, Monitoring of infliximab trough levels and anti-infliximab antibodies in inflammatory bowel diseases: A comparison of three commercially available ELISA kits: Cytokine, 2020; 126; 154859
20. Sasson AN, Ananthakrishnan AN, High anti-infliximab antibody titers do not impact response to subsequent adalimumab treatment in inflammatory bowel diseases: Dig Dis Sci, 2021; 67; 3124-28
21. Vermeire S, Dreesen E, Papamichael K, Dubinsky MC, How, when, and for whom should we perform therapeutic drug monitoring?: Clin Gastroenterol Hepatol, 2020; 18; 1291-99
22. Sazonovs A, Kennedy NA, Moutsianas L, HLA-DQA1*05 carriage associated with development of anti-drug antibodies to infiximab and adalimumab in patients with Crohn’s disease: Gastroenterology, 2020; 158; 189-99
23. Facchin S, Buda A, Cardin R, Rapid point-of-care anti-infliximab antibodies detection in clinical practice: Comparison with ELISA and potential for improving therapeutic drug monitoring in IBD patients: Therap Adv Gastroenterol, 2021; 14; 1756284821999902
24. Laserna-Mendieta EJ, Salvador-Martín S, Arias-González L, Comparison of a new rapid method for the determination of adalimumab serum levels with two established ELISA kits: Clin Chem Lab Med, 2019; 57; 1906-14
25. Pérez I, Fernández L, Sánchez-Ramón S, Reliability evaluation of four different assays for therapeutic drug monitoring of infliximab levels: Therap Adv Gastroenterol, 2018; 11; 1756284818783613
26. Van Stappen T, Billiet T, Vande Casteele N, An optimized anti-infliximab bridging enzyme-linked immunosorbent assay for harmonization of anti-infliximab antibody titers in patients with inflammatory bowel diseases: Inflamm Bowel Dis, 2015; 21; 2172-77
27. Fernandes SR, Bernardo S, Simões C, Proactive infliximab drug monitoring is superior to conventional management in inflammatory bowel disease: Inflamm Bowel Dis, 2020; 26; 263-70
Tables
 Table 1. Basic characteristics of patients whose samples were included in analysis.
Table 1. Basic characteristics of patients whose samples were included in analysis. Table 2. Measured IFX levels according to the 3 methods.
Table 2. Measured IFX levels according to the 3 methods. Table 3. Comparison of samples between University of Leuven (UHL) test and apDia test: number of samples in a respective group.
Table 3. Comparison of samples between University of Leuven (UHL) test and apDia test: number of samples in a respective group. Table 4. Comparison of samples between University of Leuven (UHL) test and Lisa-Tracker test: number of samples in a respective group.
Table 4. Comparison of samples between University of Leuven (UHL) test and Lisa-Tracker test: number of samples in a respective group. Table 5. Comparison of antibodies to IFX concentrations between University of Leuven (UHL) and Lisa Tracker.
Table 5. Comparison of antibodies to IFX concentrations between University of Leuven (UHL) and Lisa Tracker. Table 6. Number of samples with detectable (+) and undetectable (−) antibodies to IFX for a respective test.
Table 6. Number of samples with detectable (+) and undetectable (−) antibodies to IFX for a respective test. Table 1. Basic characteristics of patients whose samples were included in analysis.
Table 1. Basic characteristics of patients whose samples were included in analysis. Table 2. Measured IFX levels according to the 3 methods.
Table 2. Measured IFX levels according to the 3 methods. Table 3. Comparison of samples between University of Leuven (UHL) test and apDia test: number of samples in a respective group.
Table 3. Comparison of samples between University of Leuven (UHL) test and apDia test: number of samples in a respective group. Table 4. Comparison of samples between University of Leuven (UHL) test and Lisa-Tracker test: number of samples in a respective group.
Table 4. Comparison of samples between University of Leuven (UHL) test and Lisa-Tracker test: number of samples in a respective group. Table 5. Comparison of antibodies to IFX concentrations between University of Leuven (UHL) and Lisa Tracker.
Table 5. Comparison of antibodies to IFX concentrations between University of Leuven (UHL) and Lisa Tracker. Table 6. Number of samples with detectable (+) and undetectable (−) antibodies to IFX for a respective test.
Table 6. Number of samples with detectable (+) and undetectable (−) antibodies to IFX for a respective test. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387