11 February 2023: Clinical Research
A Study of 42 Partially Edentulous Patients with Single-Crown Restorations and Implants to Compare Bone Loss Between Crestal and Subcrestal Endosseous Implant Placement
Shailesh JainDOI: 10.12659/MSM.939225
Med Sci Monit 2023; 29:e939225
Abstract
BACKGROUND: The purpose of the study was to evaluate the influence of dental implant placement at different bone levels upon the resultant postoperative peri-implant bone loss.
MATERIAL AND METHODS: Forty-two partially edentulous patients seeking implant-supported single-crown restorations were screened followed by segregation into 2 groups (GP), GP E (equicrestal) and GP S (subcrestal) (n=21 each). Sixty endosseous implants (30 each) (Adin Tourage-S, Israel), size 3.5/8 and 4/10 mm for mandibles, were placed using a 2-stage surgical procedure. At 4 to 6 months, straight abutments were attached followed by restoration (Vita Zahnfabrik, Germany). Crestal bone levels (mesial/distal) of implant fixtures were assessed at 5 time intervals (after surgery, and at 3, 6, 9, and 12 months) using digital radiography. Means and standard deviations were calculated, following which the differences were statistically analyzed using ANOVA at P value of <0.05.
RESULTS: The mean annual bone loss for GP S (1.96 mm) was higher than GP E (1.10 mm). At all studied time intervals, the bone loss for implants in GP S was higher than in GP E (P<0.05). Between time intervals, lowest bone loss was observed on the distal side in GP E (0.11 mm/6-9 month) and the highest bone loss was observed on the distal side of GP S (0.6 mm/9-12 month). Differences in the means between the 2 groups on mesial and distal sides were statistically significant at all time intervals (P<0.05).
CONCLUSIONS: Subcrestal implant placement was associated with more bone loss than when implants are placed at the crestal level.
Keywords: Alveolar Bone Loss, Dental Implant-Abutment Design, Dental Implants, Single-Tooth, Osseointegration, Peri-Implantitis, Humans, Prostheses and Implants, Mandible, Biometry, Bone Diseases, Metabolic, Crowns, Dental Implants
Background
Loss of natural teeth initiates an imbalance within orofacial structures, which subsequently can progress into inefficient masticatory dynamics, thereby causing occlusal discrepancy that may further deteriorate to compromised esthetics. However, restoring the missing teeth reduces these adverse effects. Within the last 2 decades, available prosthetic treatment options for replacing lost teeth have witnessed a prodigious change, especially with introduction of dental implants, which in turn have improved quality of the patient’s life [1]. Formerly, removable partial dentures (RPD) were commonly used to substitute missing teeth, which changed with the advent of fixed partial dentures (FPD) (tooth-supported). FPDs are still used widely, as they improve masticatory function, esthetics, and comfort. As science and technology progressed, newer options to replace teeth came into effect. Presently, implants as a treatment preference are being widely advocated for replacing teeth and lost structures, the biological basis of which is osseointegration, a concept introduced in 1969 [2]. A major advantage of dental implants over conventional tooth-supported FPD is that it is independent of the adjacent natural teeth, thus preserving and fulfilling the basic principle of tooth preservation. During the last few decades of clinical advances in material science and design for endosseous implants, replacement of the tooth/teeth has evolved, which has increased prosthetic treatment options within implant dentistry [3]. Osseointegration as per Branemark’s theoretical concept implies that a titanium screw could be drilled into a viable, vital bone and permitted to integrate with the bone, which in time could support the dental prosthesis [2]. With the advent and support of precise computer-aided diagnosis and computer-aided machining (CAD/CAM), implant-supported restorations have developed to be a commonly used treatment modality for restoring lost dentition, as it satisfies the psychological, functional, and esthetic needs of partially edentulous patients [4]. Dental implants have progressed in shape, size, surface treatment, and clinical loading, thus making it one of the most desired treatment options for replacing partially or completely edentulous arches.
Osseointegration is the basis of dental implant success, and depends largely on the quality and quantity of the surrounding bone [5]. Conservation of crestal bone in and around the submerged implant fixture is a vital clinical criterion to be considered for successful implant therapy. The bone loss around an implant also influences the esthetic outcome of the prescribed treatment. Consequently, estimation of the crestal bone levels in patients treated with implants is crucial for the final outcome of the prosthesis. Various modalities have been proposed for measuring crestal bone loss and all of these are well documented in the scientific literature [6]. Radiographic assessment using intra-oral peri-apical radiographs (long-cone paralleling technique) has been widely used for its simplicity, availability, and safety. Orthopantomograph and digital radiography have been found to be reliable methods (tools) to evaluate the clinical crestal bone changes [6]. The benchmark for implant success (long-term) as proposed by Alberktsson demands that the bone loss (vertical) around implant fixtures should be less than 0.2 mm per year after the first year of implant loading, which is still the standard [7]. The indicator for implant success remains the same for all prosthetic implant treatment options, whether implants are restoring a complete or a partial edentulous situation [8], or whether one uses a fixed or a removable implant prosthetic option. The collective configuration of bone loss observed generally is from the alveolar crest, which progresses toward the apical region. The cause of loss of bone around the implant could be either from local or systemic causes. While systemically compromised patients are relatively contraindicated for implant treatments, it is the local factors like infections and mechanical stresses (offset occlusal load) that play more significant roles in developing or preventing peri-implantitis. To achieve less bone loss, various investigators have proposed different implant placement positions in relation to the crestal bone. Two positions – equicrestal and subcrestal – have been reported, with conflicting results. Most studies have been used a mixed methodology in terms of implant numbers, implant-specific treatment option, and implant placement within the arch (anterior or posterior) and within the mouth (maxillary or mandibular). Since bone types are different within each arch, no study has been conducted exclusively studying the bone loss pattern in a specific area of the mouth. Furthermore, the follow-up evaluation period of presence/absence of crestal bone loss in and around the implant has been debated, and different authors have evaluated bone around implants at 6 months [9] and at 12 months [10]. Others have supported these views and have stated that follow-up of the patient at 6 and 9 months is appropriate for evaluating post-surgical and post-loading changes. However, an overall 10% of implants (n=10,000 implants) have been reported to still fail in clinically ideal conditions from multiple studies [11]. A retrospective study of 8,528 patients treated with 39,077 implants over a period of 27 years reported that the incidence of failure was higher within the first year of surgery (69%) (n=857), especially for the maxillary arch [12]. Implant site inflammation during the first year of implant placement was found to be a high risk for late implant failure [12]. Implant failures have been found to be associated with multiple patient, surgery, and prosthesis-related risk factors. Within the first year after surgery, poor patient compliance (eg, maintaining improper oral hygiene, smoking, not attending follow-up visits) has been reported to be a risk factor for implant treatment outcome. However, clinical study (n=84 implants) that assessed the effect of various predictor variables (demographic, health status, anatomic variables, implant-specific, and operative factors) found no significant differences of these variables that would increase the risk of crestal bone loss within 1 year after placement [13]. Studies have also investigated placing implants at various levels in relation to the clinically visible crest of the bone (crestal and subcrestal). While the crestal implant placement has been shown to have greater stability within the first year [14], subcrestal implant placement has been found to be associated with greater stability and less bone loss in the long term [15].
In the context of these 2 backgrounds, we conducted this study to evaluate two different implant placement positions (equicrestal and subcrestal). We recorded marginal bone loss in a specific area of the arch (mandibular posterior) with strict inclusion/exclusion criteria, thereby minimizing the effect of other variables observed in most previous studies. To assess the need for extra follow-up for implant restoration, we also added 2 additional follow-up visits (at 3 and 9 months). These time periods are significant in evaluating the initiation or absence of initiation of bone loss irrespective of implant placement positions. We hypothesized that subcrestal implant placement provides an ideal environment for bacterial growth, especially in the mandibular arch; therefore, more bone loss will be observed after second-stage surgery. Therefore, this study included 42 partially edentulous patients with single-crown restorations and implants and aimed to compare bone loss between crestal and subcrestal endosseous implants. Evaluating the differences in bone loss at 4 different intervals of time within the first year would fulfill the objective of the study, which is to recommend increased follow-up visits for all patients receiving implants.
Material and Methods
ETHICS:
The proposed study was conceptualized after reviewing the current literature and a research proposal was submitted to the Ethics Approval Committee of the college and its affiliated university. After approval from the Ethics Committee (approval number SU/SDS/74-A/2019/02), an informed consent form was customized as per the need of this study.
STUDY DESIGN:
This clinical study was conducted in one of the recognized postgraduate institutes of North India, between the second quarter of 2019 and the first quarter of 2021. The study design was that of a prospective study in which intervention in the form of dental implants was performed at 2 different clinical depths in the posterior mandibular arch, followed by the radiographic evaluation of bone loss around implants at 4 different time intervals, excluding baseline.
SAMPLE PREPARATION, SELECTION, AND GROUPING:
The patient sample for the study was cross-sectional, which was standardized by following strict inclusion and exclusion criteria. Patients who were included had to be 18 to 40 years (single implant restorations), any gender, willing and consenting for implant placement, practicing good oral hygiene, partially edentulous with well-healed mandibular edentulous posterior areas (to exclude the need for bone augmentation), sufficient bone width and height to accommodate a 3.5- or 4-mm by 8- or 10-mm implant, and did not have any type of occlusal problem. Any patient with a history of smoking, alcohol or drug consumption, medically compromised, evidence of parafunctional habit, inaccessible posterior area, or other absolute or relative contraindications for implant placement were excluded from the study. An additional criterion for exclusion of females was currently being pregnant. All patients were registered in the study after a detailed clinical and radiographic examination, assessment of diagnostic casts, bone mapping, orthopantomograph evaluation, and patient education and motivation – 42 patients (31 males and 11 females) were finalized to receive 60 endosseous implants in the mandibular posterior area. These patients were allotted (convenience sampling) to 2 equal groups (GP) – GP E (Equicrestal placement) and GP S (Subcrestal placement) – based on implant fixture relation to the adjacent bone. A total of 30 implants were placed on either right, left, or same/both sides of the mandibular arch. Allotment was done by convenience sampling, so that a uniform distribution of implants on either side was achieved. For each GP, a total of 22 implants in the distribution of 15 and 7 implants on either right or left side was achieved with equal distribution of 4 patients with 8 implants (2 on the same side/1 on the right and 1 on the left). The position of implant placement in the subcrestal group was 2 mm below the level of visible clinical bone crest. Patients were thoroughly informed about the significance of the study and possible risks during the surgical procedure. Written informed consent from each patient was obtained at this stage of the study.
SURGICAL PLACEMENT OF IMPLANTS:
For all patients in both groups, a thorough diagnostic evaluation for the feasibility of placing an implant in terms of determining tentative implant size, position, alignment, and relation to crestal bone was performed using a combination of digital intra-oral peri-apical radiographs (IOPA) (Kodak Carestream, IOPA2 Size Q, XJAM530), orthopantomography (OPG) (Gendex GXDP-700 Series OPG System, KaVo, Germany), (Figure 1A) cone beam computed tomography (CBCT) (Vera view epocs 3D R100; J. Morita, Kyoto, Japan) (Figure 1B, 1C), mounted diagnostic casts on programmed semi-adjustable articulator (Whip Mix series 3000; Elite Dental Services, Inc., Orlando, FL), and customized diagnostic splints. A standard 2-stage surgical protocol was followed for all patients. All patients were supervised by a 3-member multidisciplinary team (prosthodontist, oral surgeon, and periodontist), with more than 8 years of experience in implant treatment. A local anesthetic agent (Lox 2%, Neon Pharmaceuticals, India) was administered and verified for numbness. A mid-crestal incision was made (Figure 2A) and a full-thickness flap was elevated buccally and lingually to the level of the mucogingival junction (Figure 2B), exposing the alveolar ridge. The customized surgical stent/guide was placed intraorally, which aided in proper orientation of the implant (Figure 2C). An initial pilot drill was used, which corresponded to 0.5 mm more than the final length of the chosen implant (Figure 2D). Then, osteotomy was done as per the standard operating protocol for bone type (soft, medium, hard) using different drill types (D1–2 for hard bone and D3–4 for soft and medium bone). After the drilling protocols, the implants (Touareg STM, Adin Dental Implants System Ltd., Afula, Israel) were placed in their predesignated group positions and torque in accordance with the manufacturer’s guidelines. Implants in GP E were placed at the level of the clinically visible bone crest (Figure 2E), while implants in the GP S were placed 2 mm below the clinically visible alveolar bone crest (Figure 2F). Primary stability was achieved up to 25 Ncm (verified and measured by Osstell) in all the subjects, and cover screws were placed (using a 0.05″ hex driver) over the implants and the flap was advanced for the primary closure using interrupted sutures (Ethicon, nonabsorbable surgical suture, Johnson and Johnson). Intra-oral peri-apical radiographs were taken for all patients immediately after placement of implants using the parallel-cone technique with the help of a sensor-positioning device (Rinn XCP, Dentsply). Postoperative care instructions and possible complications were explained to the patients and each patient received written instructions for enhancing patient compliance. Medications prescribed included routinely administering antibiotics and analgesics (5 days for all patients) that were individualized for each patient according to the clinical situation and demand. Patients were also recommended to use chlorhexidine mouthwash twice daily until the next appointment. All patients were recalled after 1 week for suture removal. No postoperative complications were reported by any patients in either group.
PROSTHETIC PHASE:
All patients were respectively recalled for second-stage surgery after 4 months, when the cover screws were exposed and removed. Gingival formers of the appropriate height were chosen according to the clinical situation and placed on the implant for 2 weeks in all patients. For all patients, the implants were used as abutments that supported a single-crown restoration in the posterior mandibular area. Once a satisfactory gingival collar was obtained, the gingival formers were removed and an implant-level impression was made with the help of closed-tray impression copings. The impression material of choice was addition polyvinyl siloxane material (Reprosil, Dentsply/Caulk; Milford, DE, USA), and a dual-mix technique was employed. The implant analogs were screwed and master casts were poured with die stone (Ultrarock, Kalabhai Dental, India). The casts were retrieved from their respective impressions, following which the abutments were chosen and fitted onto the implant analog. The final feldspathic porcelain (VMK-95 Metall Keramik; Vita Zahnfabrik, Bad Sackingen, Germany) restorations were fabricated and were cemented with zinc phosphate cement (Harvard, Germany) onto the implant abutments.
MEASUREMENTS AND DATA EVALUATION, COLLECTION, AND ANALYSIS:
All measurements were made through radiographic evaluation at various follow-up time intervals (3, 6, 9, and 12 months). A digital radiographic sensor (Sopix, Action India) was used, where the exposure parameters were kept standardized at 60 KVp, 10 MA, and 0.05 seconds. The radiographic technique used for IOPA was a parallel-cone technique with the help of a sensor-positioning device (Rinn XCP, Dentsply) [16]. All images were calibrated before measuring on the computer using dental imaging software (6.14.7.3, Carestream Health, Inc., 2014). Metric analysis was performed on a millimeter scale using the measuring tool available in the software. The radiographic evaluation of patients in GP E and GP S was conducted at 5 different intervals of time: after placement (baseline) (Figures 3A, 4A), 3rd month (Figures 3B, 4B), 6th month (Figures 3C, 4C), 9th month (Figures 3D, 4D), and 12th month (Figures 3E, 4E). Following the exposure, images were captured using the software and stored. The 2 reference points (A and B) for the measurement were selected as the most coronal portion of the implant abutment of the measurable marginal bone level of the mesial and distal ends [17]. The determined values of each fixture were collected and compared over the follow-up period of 1 year separately for the mesial and the distal aspects.
STATISTICAL ANALYSIS:
The collected data were first curated and then formally analyzed for normality, then means and standard deviations were calculated and tested (parametric). Analysis of variance (ANOVA) was used to assess statistically significant differences between the groups. Differences between 2 parameters and or groups were considered to be statistically significant if the value (
Results
DEMOGRAPHIC CHARACTERISTICS:
Group characteristics of patients are presented in Table 1, showing that the participants in each group in terms of gender, implant site and implant numbers (gender and site) were homogenous in distribution and did not have any significant differences that would confound the effect on the study results.
DISTRIBUTION OF BONE LOSS OVER OBSERVED TIME INTERVALS:
In GP E, the mean difference of the crestal bone loss was between 1.02 mm (1.02 mm at 12 months minus 0.00 after placement) on the medial side and 1.10 mm (1.10 mm at 12 months minus 0.00 after placement) on the distal side, while for GP S the mean difference was between 1.96 mm (2 mm/0.04 mm at baseline minus 0.04 at 12 months) on both sides (Table 2). For GP S, the mean difference was calculated by subtracting the bone level at 12 months from the bone level at placement. There was relatively more bone loss observed in implants belonging to GP S at the 3rd month (mean 0.48 mm on both sides), 6th month (mean 0.45 mm-mesial, 0.44 mm – distal), 9th month (mean 0.45 mm – mesial, 0.44 – distal), and 12 months (mean 0.58 mm – mesial; 0.60 mm – distal) than in implants belonging to GP E, at all intervals of time (Table 2). The highest amount of bone loss (mean 0.60mm) was observed on the distal side of subcrestal implants between 9 and 12 months, while the least amount (mean 0.11mm) of bone loss was observed on the distal side of the implant in the equicrestal group between the 6th and 9th months (Figure 5).
BONE LOSS PATTERN:
On average, the implants in subcrestal groups on both mesial and distal sides showed a consistent pattern in bone loss (mean range, 0.44 to 0.6 mm) at all studied time intervals (Table 2, Figure 5). In GP E, an inconsistent bone loss pattern was observed with periods of low bone loss in between [mean 0.28 (mesial) and 0.34 (distal) at 3rd month, mean 0.12 (mesial) and 0.36 (distal) at 6th month, mean 0.39 (mesial) and 0.11 (distal) at 9th month, mean 0.23 (mesial) and 0.29 (distal) at 12th month]. When compared with the baseline the implants in GP S showed higher bone loss on both mesial and distal sides (Figure 6). In GP S at the end of 12 months, 0.04 mm of bone were still present on both sides of the implants. Graphic representation of the amount of bone loss shows a consistent and regular bone loss pattern being observed with subcrestal implant placement, while there was a periodical bone loss pattern in equicrestal implant placement.
DIFFERENCES BETWEEN THE 2 STUDIED GROUPS:
ANOVA was done to analyze the significance of differences in the means of bone loss observed at 4 different prospective time intervals from baseline. The differences between the 2 groups on mesial and distal sides were found to be statistically significant at the observed
Discussion
STRENGTH AND LIMITATIONS:
Our study shows that under stringent patient selection and in an exclusive posterior mandibular location, the equicrestal implant placement produces a significant reduction in bone loss as compared to subcrestal implant placement. This study could be the basis for developing a methodology approach for investigating implant prognosis in terms of bone loss. This study is, however, limited by the use of radiography for bone measurements, which are 2-dimensional images and a distortion factor is inherited in such an approach. These could be overcome by measurements done with CT scan or CBCT, which carry the risk of increased radiation exposure. Another limitation of the study is that the bone varies from individual to individual and from one population to another. This is particularly applicable to mandibular and maxillary posterior areas. The accessibility for oral hygiene maintenance tools such as brush and floss are also limited in mandibular posterior areas. The study does also possess the routine limitations of a cross-sectional study, including the smaller sample size.
Conclusions
The results of this study must be interpreted with caution and apply only to the selected patients and clinical conditions used during the study. In similar conditions, we conclude that equicrestal implant placement is better in terms of decreasing crestal bone loss during the first postoperative and post-loading year. We found that that subcrestal implant placement results in more bone loss than crestal implant placements during the first year. The bone loss did not follow a regular pattern, as compared to subcrestal implant placement, where there was increased and uniform bone loss observed at all time intervals.
Figures
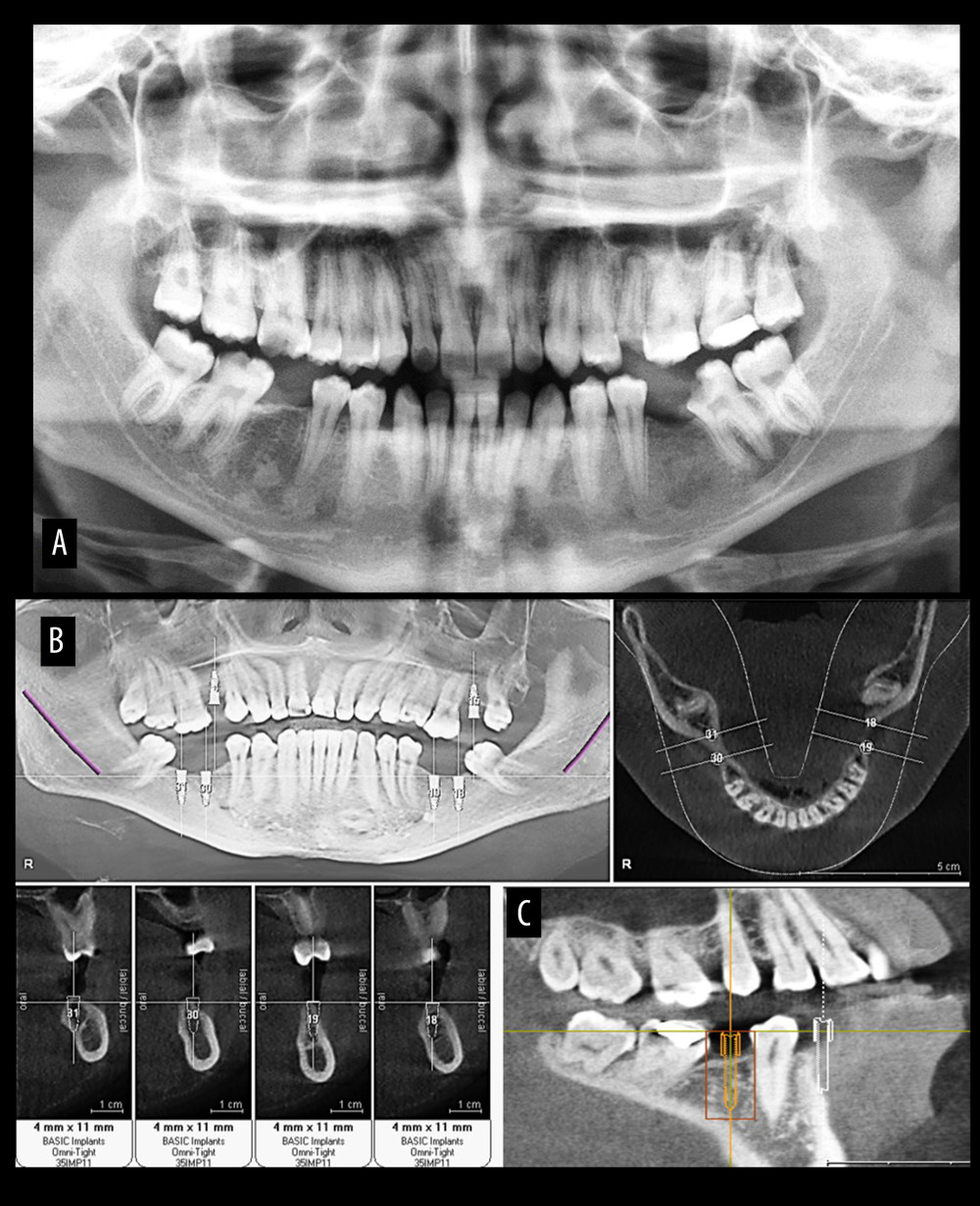 Figure 1. Diagnostic evaluation for implant selection using conventional digital orthopantomograph (A) and cone beam computed tomography (CBCT) (B) and (C). Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 1. Diagnostic evaluation for implant selection using conventional digital orthopantomograph (A) and cone beam computed tomography (CBCT) (B) and (C). Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). 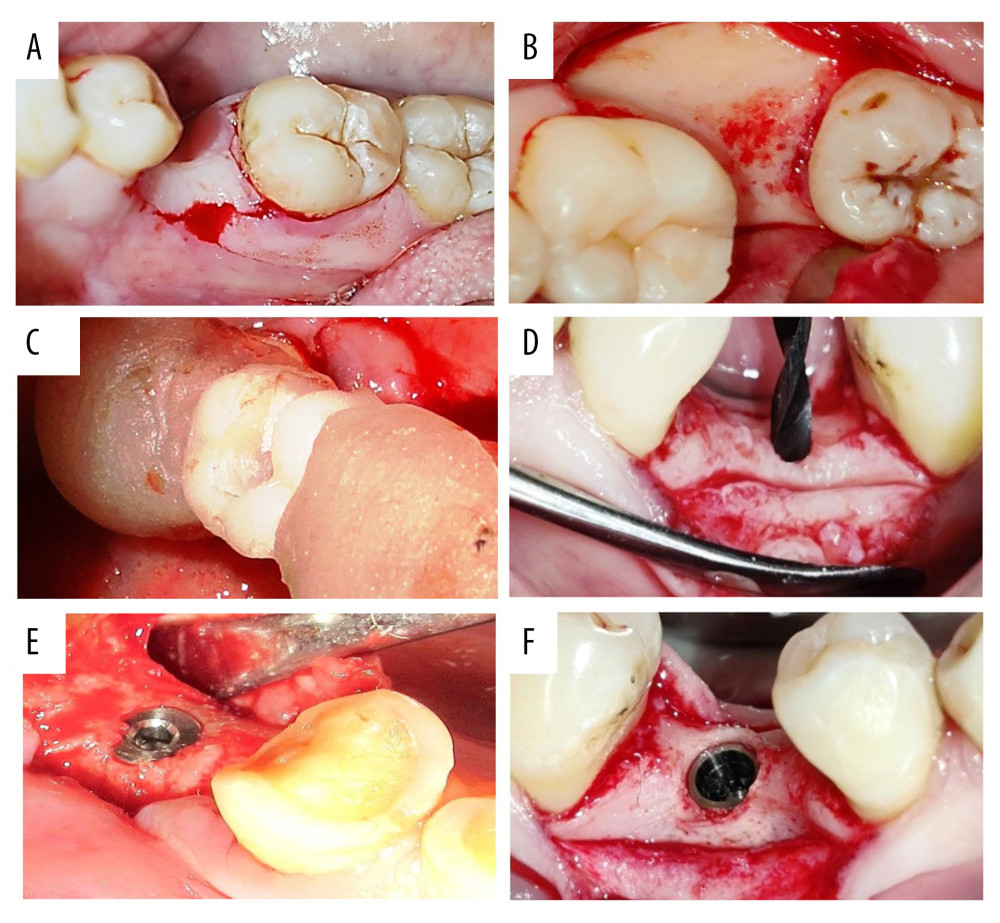 Figure 2. Sequence of implant placement showing crestal incision (A), flap reflection (B), surgical guide placement (C), pilot drilling (D), equicrestal implant fixture placement (E), and Subcrestal implant placement (F). Photographs taken using a digital single-lens reflex (DSLR) (Canon EOS 700D) with 100 mm macro lens) with/without ring flash. Compiled figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 2. Sequence of implant placement showing crestal incision (A), flap reflection (B), surgical guide placement (C), pilot drilling (D), equicrestal implant fixture placement (E), and Subcrestal implant placement (F). Photographs taken using a digital single-lens reflex (DSLR) (Canon EOS 700D) with 100 mm macro lens) with/without ring flash. Compiled figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). 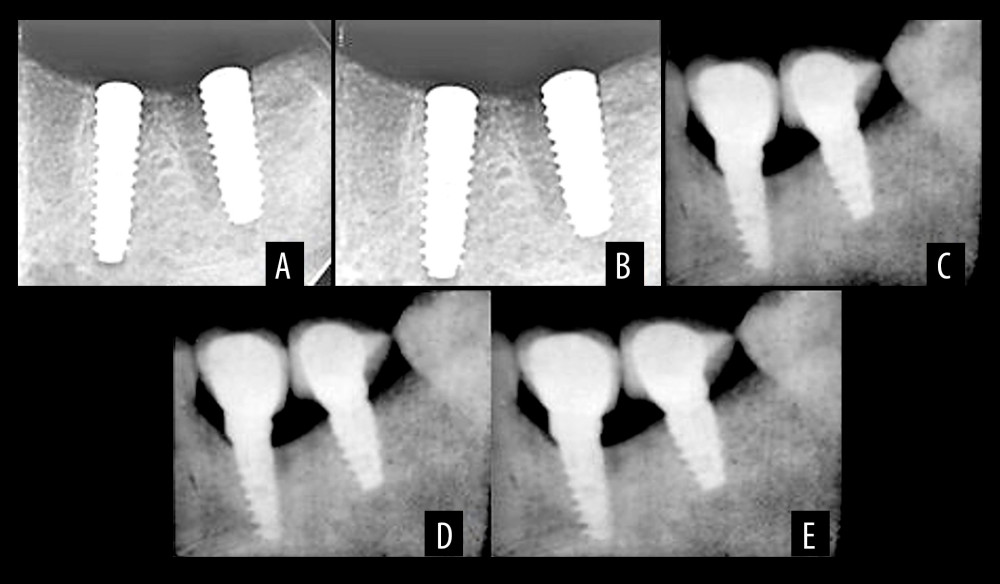 Figure 3. Radiographic interpretation of equicrestal implant placement (A) At the time of placement (B) At 3 months (C) At 6 months (D) At 9 months (E) At 12 months. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 3. Radiographic interpretation of equicrestal implant placement (A) At the time of placement (B) At 3 months (C) At 6 months (D) At 9 months (E) At 12 months. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). 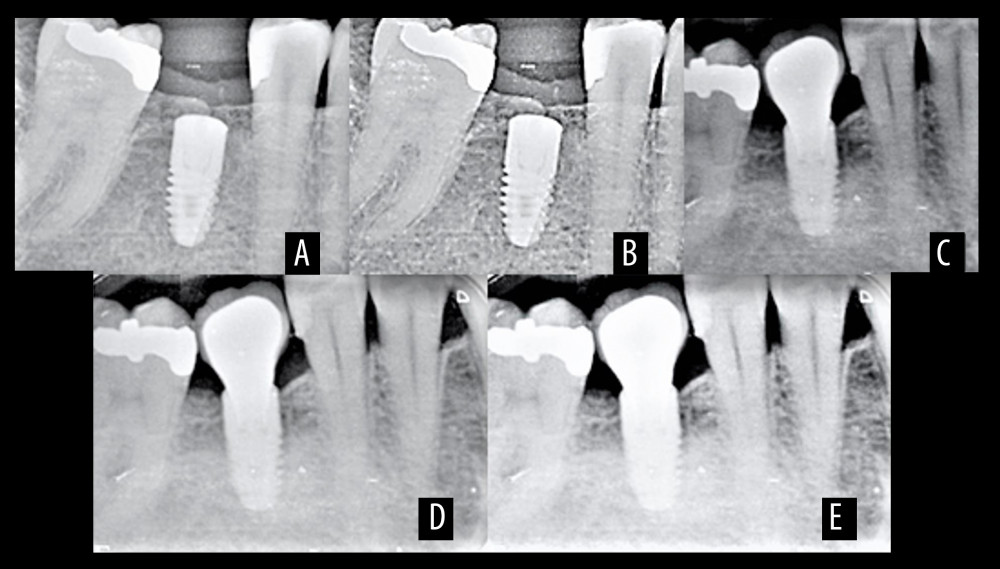 Figure 4. Radiographic interpretation of subcrestal implant placement (A) At the time of placement (B) At 3 months (C) At 6 months (D) At 9 months (E), At 12 months. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 4. Radiographic interpretation of subcrestal implant placement (A) At the time of placement (B) At 3 months (C) At 6 months (D) At 9 months (E), At 12 months. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). 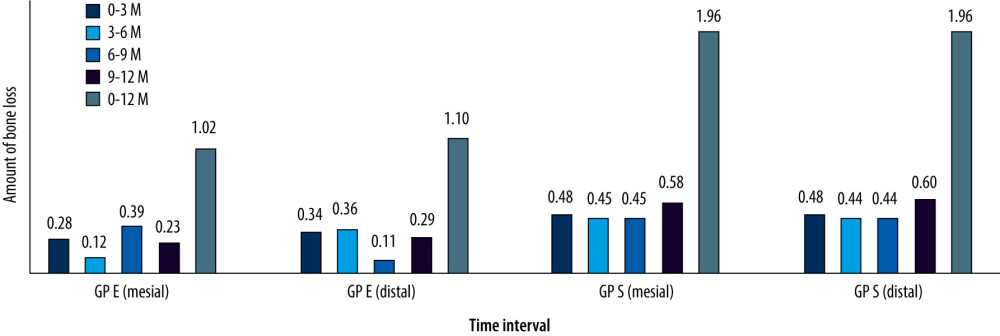 Figure 5. Graphical presentation showing comparative differences in crestal bone loss (millimeters) at different time intervals between equicrestal and subcrestal implant placement. Figure created using MS Excel, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 5. Graphical presentation showing comparative differences in crestal bone loss (millimeters) at different time intervals between equicrestal and subcrestal implant placement. Figure created using MS Excel, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). 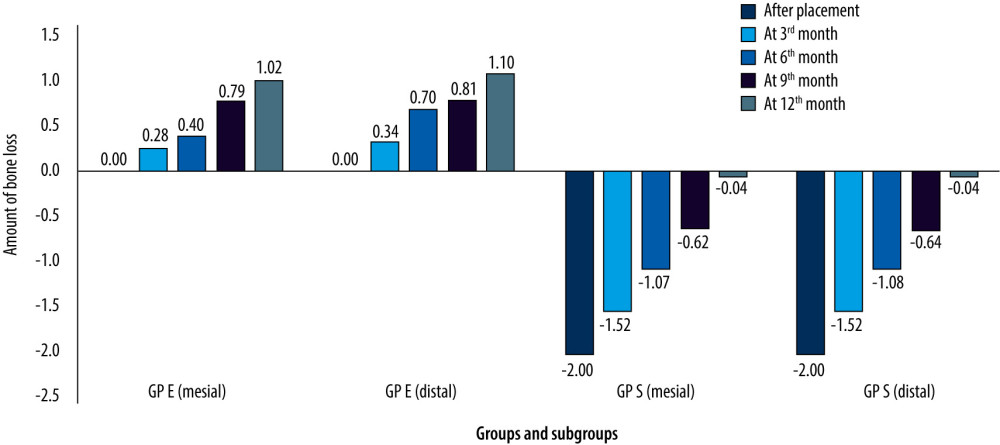 Figure 6. Graphical presentation of bone loss on either side of implants at 5 different time intervals between equicrestal and subcrestal implant placement. Figure created using MS Excel, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 6. Graphical presentation of bone loss on either side of implants at 5 different time intervals between equicrestal and subcrestal implant placement. Figure created using MS Excel, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). Tables
Table 1. Demographic characteristics of study participants and their distribution status between the groups.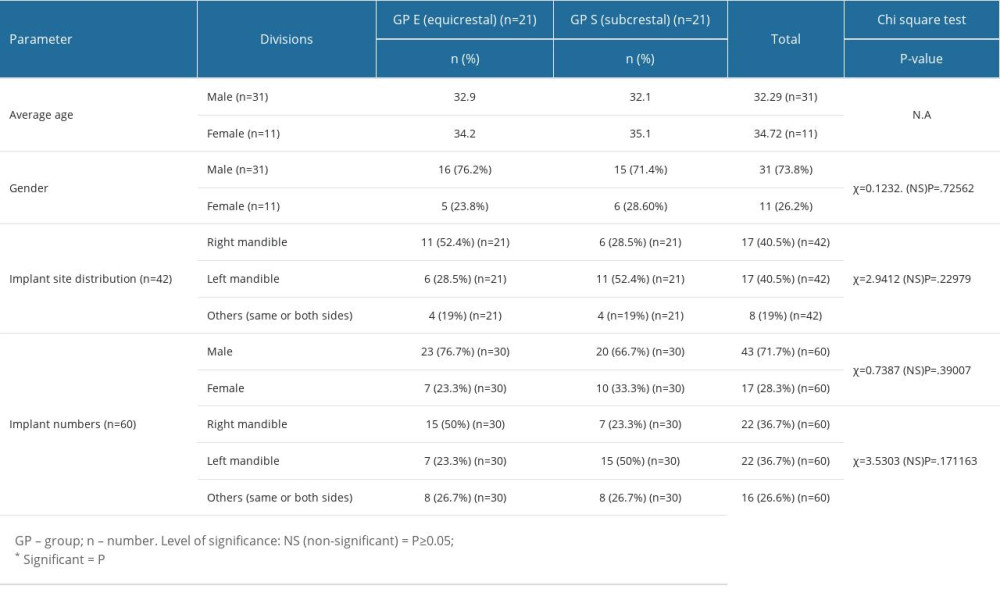 Table 2. Comparative means scores of clinical parameters among patients in various groups and the respective level of significance between various groups at different intervals of time.
Table 2. Comparative means scores of clinical parameters among patients in various groups and the respective level of significance between various groups at different intervals of time.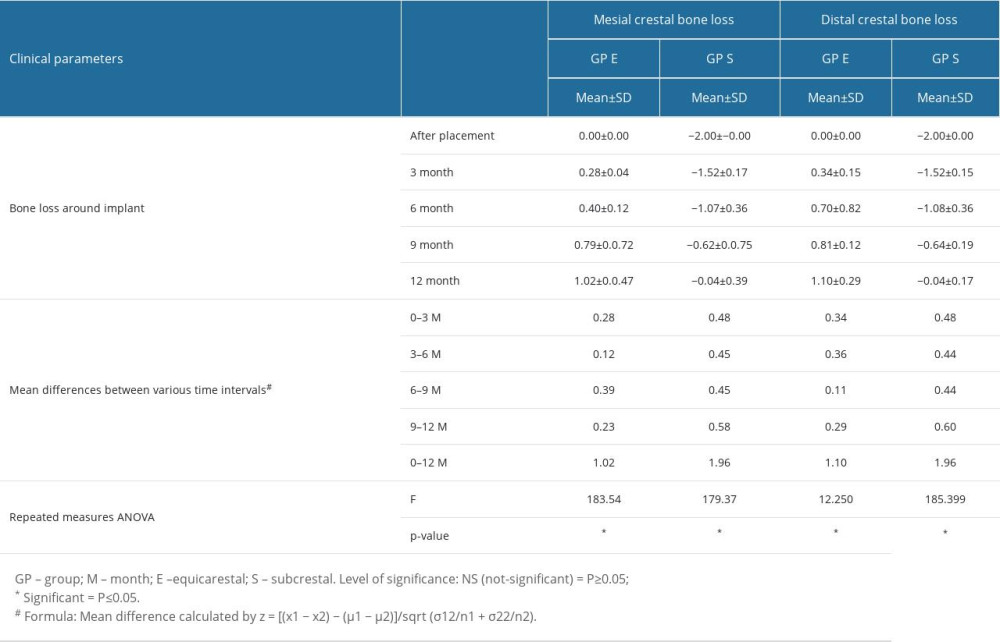
References
1. García-Minguillán G, Preciado A, Romeo M, Differences in self-perceived OHRQoL between fully dentate subjects and edentulous patients depending on their prosthesis type, socio-demographic profile, and clinical features: J Dent, 2021; 114; 103756
2. Singh M, Mattoo KA, Jain S, Replacement of a mandibular molar with implant retained single crown: Oral Surgery, Oral Medicine, Oral Radiology, 2014; 2(2); 25-27
3. Kachalia PR, Ceramic materials in implant dentistry Chapter 11: Evidence-based implant treatment planning and clinical protocols, 2016; 123, Oxford UK, Wiley, Blackwell
4. Zulkarnaen F, Indrasari M, Dewi RSEvaluation of masticatory performance and patient satisfaction before and after posterior implant restoration: Pesqui Bras Odontopediatria Clín Integr, 2021; 21; e5894 [in Portuguese]
5. Atieh MA, Alsabeeha NH, Faggion CM, Duncan WJ, The frequency of peri-implant diseases: A systematic review and meta-analysis: J Periodontol, 2013; 84; 1586-98
6. Naveau A, Shinmyouzu K, Moore C, Etiology and measurement of peri-implant crestal bone loss (CBL): J Clin Med, 2019; 8(2); 166
7. Fuglsig JM, Thorn JJ, Ingerslev J, Long term follow-up of titanium implants installed in block-grafted areas: A systematic review: Clin Implant Dent Relat Res, 2018; 20(6); 1036-46
8. Minocha T, Mattoo K, Rathi N, An 2/2 implant overdenture: J Clin Res Dent, 2020; 3(1); 1-3
9. Nagarajan B, Murthy V, Livingstone D, Evaluation of crestal bone loss around implants placed at Equicrestal and subcrestal levels before loading: A prospective clinical study: J Clin Diagn Res, 2015; 9(12); ZC47
10. Ajanović M, Hamzić A, Sead Redžepagić S, Radiographic evaluation of crestal bone loss around dental implants in maxilla and mandible: One-year prospective clinical study: Acta Stomatol Croat, 2015; 49(2); 128-36
11. Paquette DW, Brodala N, Williams RC, Risk factors for endosseous dental implant failure: Dent Clin, 2006; 50(3); 361-74
12. Jemt T, Olsson M, Franke Stenport V, Incidence of first implant failure: A retroprospective study of 27 years of implant operations at one specialist clinic: Clin Implant Dent Relat Res, 2015; 17; e501-10
13. Kim SY, Dodson TB, Do DT, Factors associated with crestal bone loss following dental implant placement in a longitudinal follow-up study: J Oral Implantol, 2015; 41(5); 579-85
14. Gatti C, Gatti F, Silvestri M, A prospective multicenter study on radiographic crestal bone changes around dental implants placed at crestal or subcrestal level: One-year findings: Int J Oral Maxillofac Implants, 2018; 33(4); 913-18
15. Chatterjee P, Shashikala R, Navneetham A, Comparative study of the crestal vs subcrestal placement of dental implants via radiographic and clinical evaluation: J Contemp Dent Pract, 2022; 23(6); 623-27
16. Pharoah MJ, Imaging techniques and their clinical significance: Int J Prosthodont, 1993; 6; 176-79
17. Afrashtehfar KI, Brägger U, Hicklin SP, Reliability of interproximal bone height measurements in bone-and tissue-level implants: A methodological study for improved calibration purposes: Int J Oral Maxillofac Implants, 2020; 35(2); 289-96
18. Rasouli Ghahroudi AAR, Talaeepour AR, Mesgarzadeh A, Radiographic vertical bone loss evaluation around dental implants following one year of functional loading: Clin Oral Implants Res, 2014; 25(9); 1056-64
19. Albrektsson T, Zarb G, Worthington P, Eriksson AR, The long-term efficacy of currently used dental implants: A review and proposed criteria of success: Int J Oral Maxillofac Implants, 1986; 1(1); 11-25
20. Mayta-Tovalino F, Mendoza-Martiarena Y, Romero-Tapia P, An 11-year retrospective research study of the predictive factors of peri-implantitis and implant failure: Analytic-multicentric study of 1279 implants in Peru: Int J Dent, 2019; 2019; 3527872
21. Mattoo K, Garg R, Bansal V, Designing the occlusion for a single tooth implant in a compromised occlusion: Journal of Medical Science and Clinical Research, 2014; 2(11); 2996-3000
22. Sunitha RV, Ramakrishnan T, Kumar S, Emmadi P, Soft tissue preservation, crestal bone loss around implants around single tooth implants: J Oral Implantol, 2008; 34; 223-29
23. Hammerle CH, Bragger U, Bürgin W, Lang NP, The effect of subcrestal placement of the polished surface of ITI implants on marginal soft and hard tissues: Oral Implants Res, 1996; 7(2); 111-19
24. Nagarajan B, Murthy V, Livingstone D, Evaluation of crestal bone loss around implants placed at equicrestal and subcrestal levels before loading: A prospective clinical study: J Clin Diagn Res, 2015; 9(12); ZC47-50
25. Ghahroudi AR, Talaeepour AR, Mesgarzadeh A, Radiographic vertical bone loss evaluation around dental implants following one year of functional loading: J Dent (Tehran), 2010; 7(2); 89
26. Rasouli R, Barhoum A, Uludag H, A review of nanostructured surfaces and materials for dental implants: Surface coating, patterning and functionalization for improved performance: Biomater Sci, 2018; 6(6); 1312-38
27. Johansson LA, Ekfeldt A, Implant-supported fixed partial prostheses: A retrospective study: Int J Prosthodont, 2003; 16(2); 172-76
28. Fickl S, Zuhr O, Stein JM, Hurzeler MB, Peri Implant bone levels around implants with platform switched abutments: Int J Oral Maxillofac Implants, 2010; 25; 577-81
29. Rani I, Shetty J, Reddy V, A comparison of peri-implant strain generated by different types of implant supported prostheses: J Indian Prosthodont Soc, 2017; 17; 142-48
30. Linkevicius T, Puisys A, Linkevicius R, The influence of submerged healing abutment or subcrestal implant placement on soft tissue thickness and crestal bone stability. A 2-year randomized clinical trial: Clin Implant Dent Relat Res, 2020; 22(4); 497-506
31. Pellicer-Chover H, Díaz-Sanchez M, Soto-Peñaloza D, Impact of crestal and subcrestal implant placement upon changes in marginal peri-implant bone level. A systematic review: Med Oral Patol Oral Cir Bucal, 2019; 24(5); e673
32. Rathi N, Goswami R, Mattoo KA, Implant supported mandibular overdenture – case report: Journal of Advanced Medical and Dental Science Research, 2019; 7(11); 128-30
33. Mattoo K, Singh SP, Brar A, A simple technique to align attachment components in implant supported mandibular overdenture: Int J Res Med Sci Tech, 2014; 1(1); 6-8
34. Nickenig HJ, Wichmann M, Schlegel KA, Radiographic evaluation of marginal bone levels adjacent to parallel- screw cylinder machined-neck implants and rough-surfaced microthreaded implants using digitized panoramic radiographs: Clin Oral Implants Res, 2009; 20(6); 550-54
35. Danza M, Tortora P, Quaranta A, Randomised study for the 1-year crestal bone maintenance around modified diameter implants with different loading protocols: A radiographic evaluation: Clin Oral Investig, 2010; 14(4); 417-26
36. Thapliyal GK, Pawar VR, A comparative analysis of periimplant bone levels of immediate and conventionally loaded implants: Med J Armed Forces India, 2013; 69(1); 41-47
37. Nandal S, Ghalaut P, Shekhawat H, A radiological evaluation of marginal bone around dental implants: An in-vivo study: Natl J Maxillofac Surg, 2014; 5(2); 126-37
Figures
 Figure 1. Diagnostic evaluation for implant selection using conventional digital orthopantomograph (A) and cone beam computed tomography (CBCT) (B) and (C). Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 1. Diagnostic evaluation for implant selection using conventional digital orthopantomograph (A) and cone beam computed tomography (CBCT) (B) and (C). Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). Figure 2. Sequence of implant placement showing crestal incision (A), flap reflection (B), surgical guide placement (C), pilot drilling (D), equicrestal implant fixture placement (E), and Subcrestal implant placement (F). Photographs taken using a digital single-lens reflex (DSLR) (Canon EOS 700D) with 100 mm macro lens) with/without ring flash. Compiled figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 2. Sequence of implant placement showing crestal incision (A), flap reflection (B), surgical guide placement (C), pilot drilling (D), equicrestal implant fixture placement (E), and Subcrestal implant placement (F). Photographs taken using a digital single-lens reflex (DSLR) (Canon EOS 700D) with 100 mm macro lens) with/without ring flash. Compiled figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). Figure 3. Radiographic interpretation of equicrestal implant placement (A) At the time of placement (B) At 3 months (C) At 6 months (D) At 9 months (E) At 12 months. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 3. Radiographic interpretation of equicrestal implant placement (A) At the time of placement (B) At 3 months (C) At 6 months (D) At 9 months (E) At 12 months. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). Figure 4. Radiographic interpretation of subcrestal implant placement (A) At the time of placement (B) At 3 months (C) At 6 months (D) At 9 months (E), At 12 months. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 4. Radiographic interpretation of subcrestal implant placement (A) At the time of placement (B) At 3 months (C) At 6 months (D) At 9 months (E), At 12 months. Figure created using MS PowerPoint, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). Figure 5. Graphical presentation showing comparative differences in crestal bone loss (millimeters) at different time intervals between equicrestal and subcrestal implant placement. Figure created using MS Excel, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 5. Graphical presentation showing comparative differences in crestal bone loss (millimeters) at different time intervals between equicrestal and subcrestal implant placement. Figure created using MS Excel, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). Figure 6. Graphical presentation of bone loss on either side of implants at 5 different time intervals between equicrestal and subcrestal implant placement. Figure created using MS Excel, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation).
Figure 6. Graphical presentation of bone loss on either side of implants at 5 different time intervals between equicrestal and subcrestal implant placement. Figure created using MS Excel, version 20H2 (OS build 19042,1466), Windows 11 Pro, Microsoft corporation). Tables
 Table 1. Demographic characteristics of study participants and their distribution status between the groups.
Table 1. Demographic characteristics of study participants and their distribution status between the groups. Table 2. Comparative means scores of clinical parameters among patients in various groups and the respective level of significance between various groups at different intervals of time.
Table 2. Comparative means scores of clinical parameters among patients in various groups and the respective level of significance between various groups at different intervals of time. Table 1. Demographic characteristics of study participants and their distribution status between the groups.
Table 1. Demographic characteristics of study participants and their distribution status between the groups. Table 2. Comparative means scores of clinical parameters among patients in various groups and the respective level of significance between various groups at different intervals of time.
Table 2. Comparative means scores of clinical parameters among patients in various groups and the respective level of significance between various groups at different intervals of time. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








