18 April 2023: Clinical Research
Quantitative Flow Ratio for Assessment of Non-Culprit Coronary Artery Lesions During Percutaneous Coronary Intervention (PCI) in 79 Patients Diagnosed with ST-Elevation Myocardial Infarction (STEMI): A Study from a Single Center in Lithuania
Mindaugas Barauskas1ABCDEF, Greta Žiubrytė2CDEF*, Nojus Jodka1BEF, Ramūnas Unikas1ADDOI: 10.12659/MSM.939360
Med Sci Monit 2023; 29:e939360
Abstract
BACKGROUND: Approximately half of the patients requiring percutaneous coronary intervention (PCI) for ST-elevation myocardial infarction (STEMI) have additional stenotic coronary artery (CA) lesions in non-infarct-related arteries (non-IRA). This study from a single center in Lithuania aimed to evaluate the use of the quantitative flow ratio (QFR) in assessing non-IRA lesions during PCI in 79 patients diagnosed with STEMI.
MATERIAL AND METHODS: We prospectively included 105 vessels of 79 patients with worldwide STEMI criteria and ≥1 intermediate (35-75%) lesion in non-IRA between July 2020 and June 2021. For all included patients, QFR analyses were performed twice, during the index PCI (QFR 1) and during a staged procedure ≥3 months later (QFR 2). The QFR analyses were performed with the QAngio-XA 3D and £0.80 were used as cut-off values for PCI. The primary endpoint was a head-to-head numerical agreement between 2 measurements.
RESULTS: An excellent numerical agreement was found in all investigated lesions, r=0.931, p<0.001, left anterior descending (LAD) r=0.911, p<0.001, left circumflex (LCx) r=0.977, p<0.001, and right coronary artery (RCA) 0.946, p<0.001. Clinical treatment decision-making showed amazing agreement between the 1st and the 2nd QFR analyses, r=0.980, p<0.001. There was 1 disagreement between QFR 1 and QFR 2.
CONCLUSIONS: The findings from this support previous studies and showed that the QFR is a practical quantitative method to evaluate non-IRA lesions, which in this study included STEMI patients during PCI following occlusive CA stenosis.
Keywords: Fractional Flow Reserve, Myocardial, ST elevation myocardial infarction, acute coronary syndrome, percutaneous coronary intervention, Humans, Coronary Vessels, Lithuania, Coronary Stenosis, Coronary Angiography, Treatment Outcome
Background
Although approximately 30–50% of patients presenting with ST-elevation myocardial infarction (STEMI), when the culprit’s vessel is occluded or sub-occluded, are diagnosed with multiple-vessel disease (≥50% diameter stenosis of ≥2 coronary arteries [1]) requiring additional invasive treatment [2]. However, not all the 50–70% stenoses are functionally significant [3]. It is important when considering non-infarct-related arteries (sometimes called non-culprit artery) percutaneous coronary interventions (PCIs). The actual need for revascularization remains an issue despite the increasing evidence of physiology-guided PCIs in stable patients [4]. Moreover, Juni et al did not show benefits of fractional flow reserve (FFR)-guided PCIs in STEMI patients [2]. Due to conflicting results, in real clinical practice, the treatment of non-culprit lesions (NCLs) still depends mainly upon the treating cardiologists’ decision [4].
After an emergency angiography and culprit lesion PCI [5,6], NCLs treatment might include: (1) medical treatment only with possible revascularization at the time of symptoms recurrence, (2) complete revascularization based on visual estimation only [4], but this might cause unnecessary stent deployment in non-significant lesions [6,7], and lastly (3) physiology-guided PCI in NCLs [4].
A meta-analysis of 5 large randomized clinical trials (RCTs) [7–11] support complete revascularization over culprit-lesion-PCI only [4–8] due to total major adverse cardiovascular events (MACE) risk reduction associated with lower rate of repeated myocardial infarction (MI) and revascularization [5–8]. This same meta-analysis proved the benefits of physiology evaluation [4], as in certain cases angiographical evaluation is unable to correctly predict the physiological significance of NCLs [12]. Thus, physiology-guided PCIs are widely preferred [4–6].
It is known that acute coronary syndrome (ACS) significantly effects the hemodynamic of epicardial and microvascular blood flow [4,13,14]; therefore, it is still unclear when and which physiology evaluation method is the most reliable and appropriate for physiology evaluation, especially once it is recommended to provide the complete revascularization during index procedure or index hospitalization [5,6]. Changes in microvascular flow in the acute MI phase may have different impacts on hyperemic and resting indices, not only in the infarct-related region, but also in the myocardium globally [13–15]. Moreover, for hyperemic indices adenosine is used, which might cause certain adverse effects even in stable patients, including chest discomfort, bradycardia, asystole, and death [16]. At this point, resting indices are superior, but during the acute phase of MI they might underrate the significance of coronary lesions [15]. In contrast, hyperemic indices such as FFR results might be slightly increased during the acute phase, simulating a non-significant coronary lesion [14]. The deviation, rather than a constant, is a varying measure per case for both hyperemic and resting indices groups [15]; therefore, any established modification can be applied to the results.
A novel minimally invasive coronary artery physiology method based on three-dimensional coronary artery anatomy reconstruction from ordinary quantitative angiogram images and computerized fluids dynamics computation, known as quantitative flow ratio (QFR), eliminates hyperemic agents and provides reliable results in stable patients [17]. Moreover, several recent studies proved its suitability for NCLs evaluation in STEMI patients [18–20]. Acute QFR has higher diagnostic performance than FFR or instantaneous wave-free ratio (iFR) [18] and acute QFR-based decision-making decreases patients’ symptoms and improves prognosis [20].
Despite many advantages of physiology-guided PCIs in chronic coronary syndromes (CCS), the use of physiology evaluation itself and the method of choice remains debatable in ACS patients, especially those with STEMI and multiple NCLs. Therefore, this study from a single center in Lithuania aimed to evaluate use of the QFR in assessing non-culprit coronary artery lesions during PCI in 79 patients diagnosed with STEMI.
Material and Methods
ETHICS STATEMENT:
The study was approved by Regional Bioethics Committee and was conducted under the principles of the Helsinki Declaration and local laws and regulations. All patients in the study were invited to the study during the index PCI hospitalization and patient consent to be involved in the study was obtained before QFR analyses.
PATIENT SELECTION:
In total, 105 vessels of 79 patients, who met the worldwide STEMI criterion [5] and had at least 1 focal intermediate (with diameter stenosis of 35–75%) lesion in other than the culprit artery between July 2020 and June 2021 were prospectively included into this single-center per-vessel study.
INCLUSION CRITERIA: Inclusion criteria included STEMI patients who came to the PCI center within 120 minutes after onset of symptoms, for whom multivessel disease (at least 1 non-culprit lesion with intermediate 35–75% lumen diameter stenosis) was diagnosed after coronary artery angiography (Figure 1), who gave their written consent before inclusion. Patients who did not meet at least 1 of the inclusion criteria were excluded from the study.
DIAGNOSIS OF STEMI:
ST-segment elevation myocardial infarction was diagnosed by the European Society of Cardiology (ESC) guidelines for management of patients presenting with ST-segment elevation, which was released in 2017 [5].
METHOD OF PCI:
Percutaneous coronary interventions were performed as a staged procedure at least 3 months after the index PCI. If the QFR value on the non-culprit lesion during staged PCI was ≤0.80, the stenosis was stented.
CORONARY ARTERY ANGIOGRAPHY:
All coronary artery angiographies (CAGs) and analyses themselves were performed in compliance with the recommendation for QFR analysis as described in previous publications [21].
METHOD OF QFR:
Quantitative flow ratio analyses were performed using the certified software QAngio-XA 3D, version 2.0 (Medis Medical Imaging Systems, Leiden, The Netherlands) from coronary artery (CA) angiograms. For the quality QFR results it is important to use at least 25° apart 2 angiographic projections and other recommendations, which were described in a previous publication [17]. In our study, QFR for all included patients were performed twice (Figure 2): during STEMI culprit artery index PCI (QFR 1) and during staged procedure 3 months later (QFR 2). Both QFR analyses (QFR1 and QFR2) were performed by experienced and internationally certified QFR observed and averaged. If those 2 values of the same evaluation were not matching (the difference between the 2 measurements was >0.02), a third measurement was performed and then averaged from the 3. For the clinical decision-making, a cut-off value of ≤0.80 was used in this study, as it is recommended in description of the methods to use [21].
PATIENT OUTCOMES:
This study does not involve patient outcomes. The primary endpoint of this study was the head-to-head numerical agreement between 2 measurements (QFR1 vs QFR2). The second endpoint was the difference in treatment decision between those measurements (QFR1 vs QFR2). The patient outcomes will be published in our next publications.
STATISTICAL ANALYSIS:
Statistical analysis was performed using SPSS 28.0 software. For 2 dependent measurements, Pearson correlation was calculated. The Bland-Altman plot analysis was performed to test head-to-head agreement between the acute phase QFR measurement (QFR 1) and stable phase QFR re-measurement (QFR 2). Cohen’s kappa was used to investigate the treatment decision agreement between those 2 independent measurements (QFR 1 vs QFR 2). The chosen level of significance was p<0.001.
Results
NUMERICAL CORRELATION BETWEEN QFR 1 AND QFR 2 VALUES:
A strong correlation between numerical values was found in all investigated lesions: r=0.931, p<0.001 (Figure 3). After dividing all lesions according to coronary artery, the following correlation coefficients were found: LAD r=0.911, p<0.001, LCx r=0.977, p<0.001 and RCA 0.946, p<0.001 (Figure 3). Agreement analysis using Bland-Altman plot analysis (Figure 4) showed non-significant differences between the acute phase and stable phase QFR measurements (QFR 1 vs QFR 2) with the mean difference of 0.019 between QFR 1 and QFR 2, p<0.001.
QFR 1 AND QFR 2 AGREEMENT FOR TREATMENT DECISION-MAKING:
The secondary endpoint of the study was differences of clinical treatment decision-making, which in Cohen’s kappa analysis also showed strong agreement between the acute phase and stable phase QFR measurements (QFR 1 vs QFR 2) K=0.98, p<0.001. Additionally, the correlation coefficients between the 1st and the 2nd QFR analyses were also strong in general (r=0.980, p<0.001), and after dividing according to CA, they were LAD r=1.0, p<0.001; LCx r=1.0, p<0.001; RCA r=0.843, p<0.001. The only disagreement between the 1st and the 2nd QFR analysis was in RCA. In this case, the 1st QFR was non-significant (0.85) and the second was marginal (0.80).
Discussion
This study is unique because it is the first study evaluating agreement between the 2 QFR measurements in STEMI patients, the first of which was performed during index culprit lesion PCI procedure. In our center, we discussed good QFR suitability and comfortability for patients with STEMI but did not know that the correlation between index PCI QFR and staged PCI QFR would be very high. Nevertheless, only 1 of the 105 acute QFRs disagreed with clinical treatment decision-making, and it is perfect because interventional cardiologists are increasingly discussing non-culprit PCI during infarct-related artery PCI [22]. Our results demonstrated that it is reliable and Ullrich et al showed that it decreases angina symptoms and improves the patient’s prognosis.
The comparison of physiology-guided PCI versus angiography alone-guided PCI in the settings of MI with multivessel disease remains questionable despite numerous clinical trials. A recent multicenter randomized clinical trial addressing superiority of FFR-guided PCIs failed to prove physiology-guided PCI benefits over angiography alone-guided PCI [23]. Contrary to the situation of stable patients, hyperemic indices underestimate the severity of coronary lesions in acute-phase MI [14]. This might be the main reason for non-superiority of FFR-guided PCI in STEMI patients in the FLOWER-MI clinical trial. Musto et al found lower rates of adverse events in iFR-guided PCIs, but since it was a quite small population study, further randomized clinical trials are necessary to confirm the findings [24].
QFR reliability in stable patients are non-negotiable and has been previously repeatedly proved in various studies in comparison to both hyperemic and resting indices [25–29]. Sejr-Hansen et al demonstrated that acute QFR compared with staged QFR has higher diagnostic performance than acute FFR and staged FFR or acute iFR and staged iFR [18], which is why we did not double the QFR analyses with hyperemic or resting indices methods. The current study showed excellent numerical agreement between QFR measured during STEMI culprit lesion index PCI and during a staged procedure at least 3 months later. This means that our study proved both numerical and clinical decision-making agreement between measurements acquired during the acute phase of MI and at a later time during the stable phase. Like several other studies focused on QFR [18,19], this study proved its suitability for physiological evaluation of NCLs during STEMI culprit lesion index PCI.
Despite its novelty and statistical significance, this study has several limitations: this was a single-center and quite small study. We also could not show results about patients’ symptoms or prognosis, as did Ullrich et al [20]. However, we keep collecting data and hope to provide even more reliable results and more orientated to patients’ symptoms in the future.
Conclusions
The findings from this study support those of previous studies and showed that the QFR was a practical quantitative method to evaluate non-culprit coronary artery lesions, which in this study included STEMI patients during PCI following occlusive coronary artery stenosis.
Figures
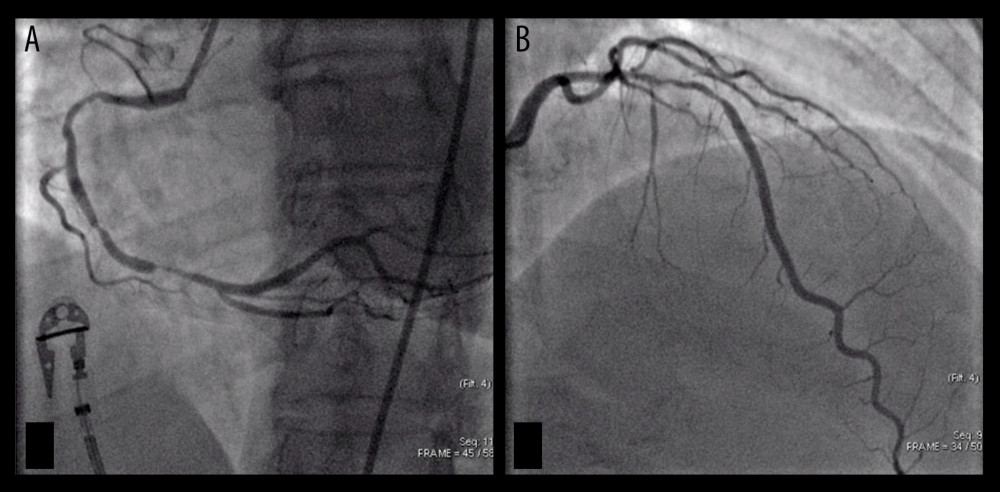 Figure 1. Subtotal occlusion of the right coronary artery (A) – infarct-related artery or culprit vessel. Non-culprit lesion on the left anterior descending artery (B). Both angiograms were performed during index PCI procedure.
Figure 1. Subtotal occlusion of the right coronary artery (A) – infarct-related artery or culprit vessel. Non-culprit lesion on the left anterior descending artery (B). Both angiograms were performed during index PCI procedure. 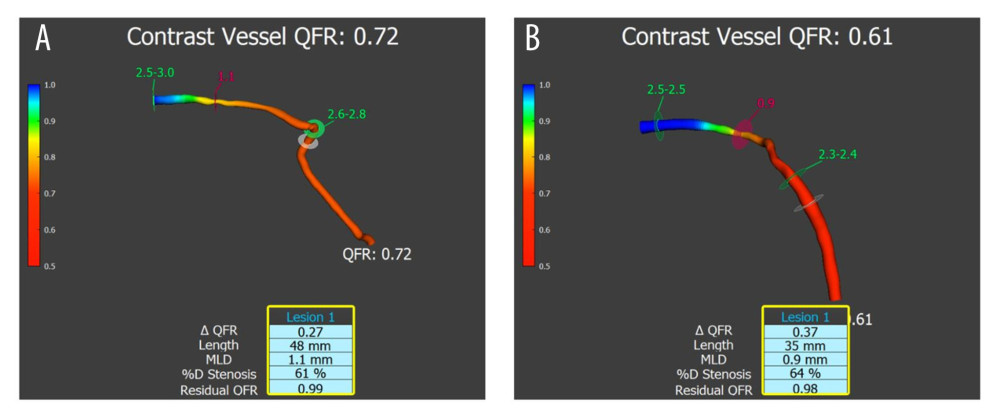 Figure 2. Functionally significant stenosis on the left anterior descending artery. (A) QFR evaluation during the index PCI procedure. (B) QFR evaluation was performed 3 months and 14 days after index PCI (through the staged PCI procedure). The case showed the highest difference between QFR 1 and QFR 2 values in our study. However, it did not change the clinical treatment decision.
Figure 2. Functionally significant stenosis on the left anterior descending artery. (A) QFR evaluation during the index PCI procedure. (B) QFR evaluation was performed 3 months and 14 days after index PCI (through the staged PCI procedure). The case showed the highest difference between QFR 1 and QFR 2 values in our study. However, it did not change the clinical treatment decision. 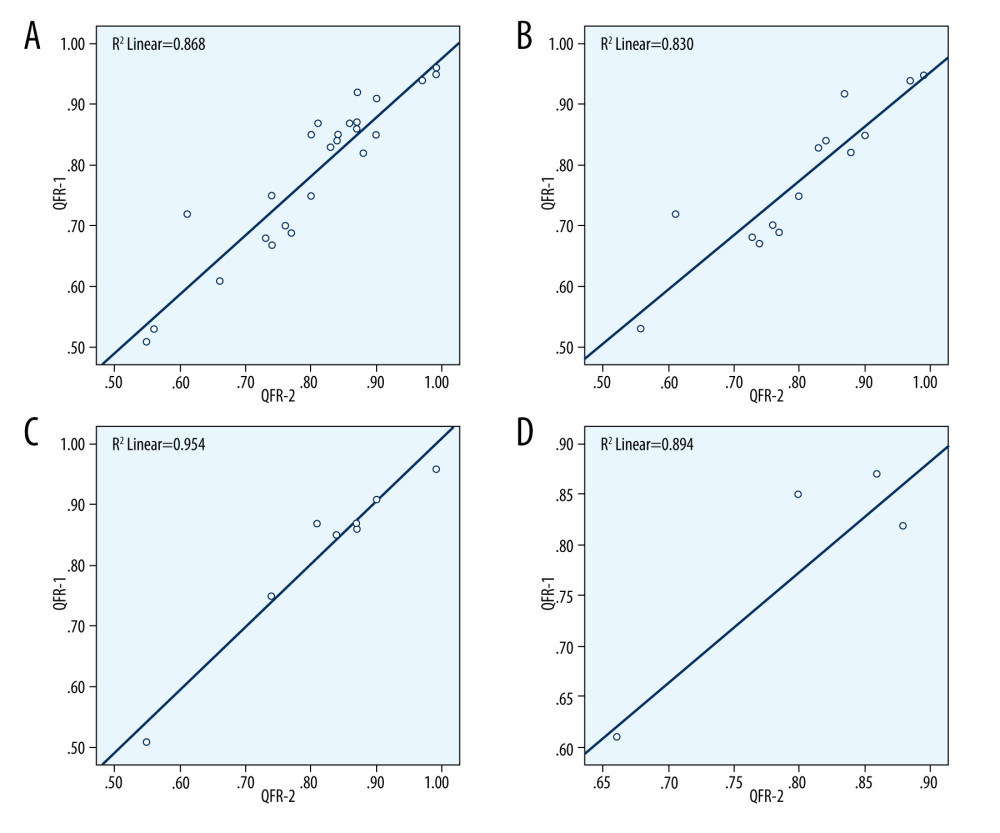 Figure 3. Four scatter diagrams, which show the strong positive correlations between quantitative flow ratio (QFR) 1 and QFR 2 numerical data in all lesions (A) and separate coronary arteries: (B) – left anterior descending artery (LAD); (C) – left circumflex artery (LCx); (D) – right coronary artery (RCA).
Figure 3. Four scatter diagrams, which show the strong positive correlations between quantitative flow ratio (QFR) 1 and QFR 2 numerical data in all lesions (A) and separate coronary arteries: (B) – left anterior descending artery (LAD); (C) – left circumflex artery (LCx); (D) – right coronary artery (RCA). 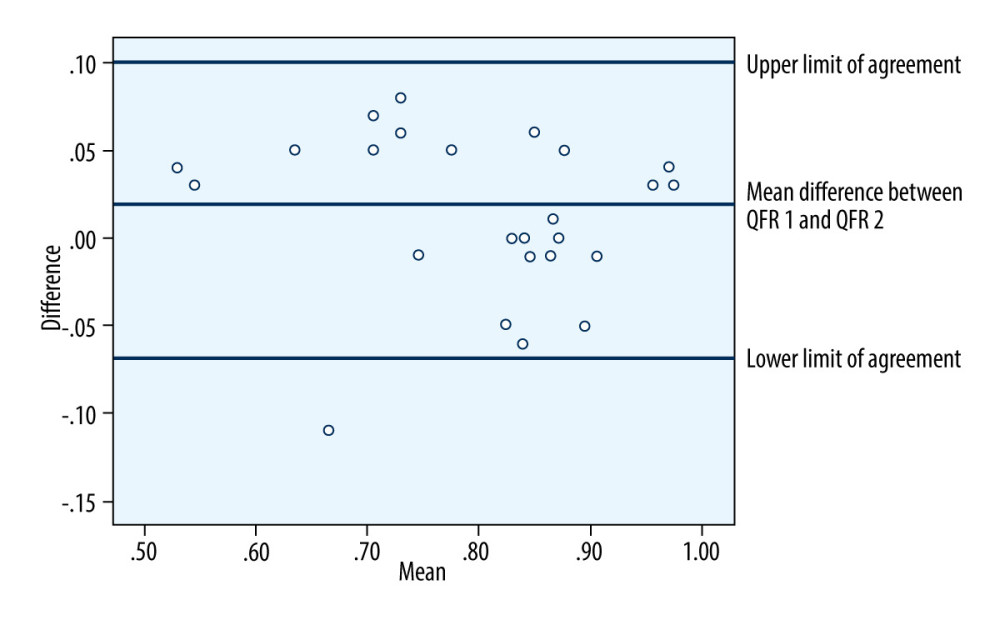 Figure 4. Bland-Altman plot analysis shows the agreement among quantitative flow ratio (QFR) 1 and QFR 2 measurements. There are non-significant differences between the acute phase QFR and staged procedure QFR in the same lesions.
Figure 4. Bland-Altman plot analysis shows the agreement among quantitative flow ratio (QFR) 1 and QFR 2 measurements. There are non-significant differences between the acute phase QFR and staged procedure QFR in the same lesions. References
1. Brener SJ, Milford-Beland S, Roe MT, Culprit-only or multivessel revascularization in patients with acute coronary syndromes: An American College of Cardiology National Cardiovascular Database Registry report: Am Heart J, 2008; 155(1); 140-46
2. Jüni P, PCI for nonculprit lesions in patients with STEMI – no role for FFR: N Engl J Med, 2021; 385(4); 370-71
3. Niccoli G, Indolfi C, Davies JE, Evaluation of intermediate coronary stenoses in acute coronary syndromes using pressure guidewire: Open Heart, 2017; 4(2); e00431
4. Thim T, van der Hoeven NW, Musto C, Evaluation and management of nonculprit lesions in STEMI: JACC Cardiovasc Interv, 2020; 13(10); 1145-54
5. Ibanez B, James S, Agewall S, 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Socie: Eur Heart J, 2018; 39(2); 119-77
6. Levine GN, Bates ER, Blankenship JC, 2015 ACC/AHA/SCAI focused update on primary percutaneous coronary intervention for patients with ST-elevation myocardial infarction: An update of the 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention and the 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction: J Am Coll Cardiol, 2016; 67(10); 1235-50
7. Wald DS, Morris JK, Wald NJ, Randomized trial of preventive angioplasty in myocardial infarction: N Engl J Med, 2013; 369(12); 1115-23
8. Gershlick AH, Khan JN, Kelly DJ, Randomized trial of complete versus lesion-only revascularization in patients undergoing primary percutaneous coronary intervention for STEMI and multivessel disease: The CvLPRIT trial: J Am Coll Cardiol, 2015; 65(10); 963
9. Engstrøm T, Kelbæk H, Helqvist S, Complete revascularisation versus treatment of the culprit lesion only in patients with ST-segment elevation myocardial infarction and multivessel disease (DANAMI-3 – PRIMULTI): An open-label, randomised controlled trial: Lancet (London, England), 2015; 386(9994); 665-71
10. Smits PC, Abdel-Wahab M, Neumann FJ, Fractional flow reserve – guided multivessel angioplasty in myocardial infarction: N Engl J Med, 2017; 376(13); 1234-44
11. Mehta SR, Wood DA, Storey RF, Complete revascularization with multivessel PCI for myocardial infarction: N Engl J Med, 2019; 381(15); 1411-21
12. Tonino PAL, Fearon WF, De Bruyne B, Angiographic versus functional severity of coronary artery stenoses in the FAME study fractional flow reserve versus angiography in multivessel evaluation: J Am Coll Cardiol, 2010; 55(25); 2816-21
13. Thim T, Götberg M, Fröbert O, Nonculprit stenosis evaluation using instantaneous wave-free ratio in patients with ST-segment elevation myocardial infarction: JACC Cardiovasc Interv, 2017; 10(24); 2528-35
14. Van Der Hoeven NW, Janssens GN, De Waard GA, Temporal changes in coronary hyperemic and resting hemodynamic indices in nonculprit vessels of patients with ST-segment elevation myocardial infarction: JAMA Cardiol, 2019; 4(8); 736-44
15. De Waard GA, Hollander MR, Teunissen PFA, Changes in coronary blood flow after acute myocardial infarction: Insights from a patient study and an experimental porcine model: JACC Cardiovasc Interv, 2016; 9(6); 602-13
16. Johnston DL, Daley JR, Hodge DO, Hemodynamic responses and adverse effects associated with adenosine and dipyridamole pharmacologic stress testing: A comparison in 2,000 patients: Mayo Clin Proc, 1995; 70(4); 331-36
17. Tu S, Westra J, Yang J, Diagnostic accuracy of fast computational approaches to derive fractional flow reserve from diagnostic coronary angiography: The international multicenter FAVOR pilot study: JACC Cardiovasc Interv, 2016; 9(19); 2024-35
18. Sejr-Hansen M, Westra J, Thim T, Quantitative flow ratio for immediate assessment of nonculprit lesions in patients with ST-segment elevation myocardial infarction – an iSTEMI substudy: Catheter Cardiovasc Interv, 2019; 94(5); 686-92
19. Lauri FM, Macaya F, Mejía-Rentería H, Angiography-derived functional assessment of non-culprit coronary stenoses in primary percutaneous coronary intervention: Eurointervention, 2020; 15(18); E1594-601
20. Ullrich H, Olschewski M, Belhadj KA, Quantitative flow ratio or angiography for the assessment of non-culprit lesions in acute coronary syndromes: Protocol of the randomized trial QUOMODO: Front Cardiovasc Med, 2022; 9; 815434
21. Ziubryte G, Jarusevicius G, Fractional flow reserve, quantitative flow ratio, and instantaneous wave-free ratio: A comparison of the procedure-related dose of ionising radiation: Postepy Kardiol Interwencyjnej, 2021; 17(1); 33-38
22. Feng Y, Li S, Hu S, The optimal timing for non-culprit percutaneous coronary intervention in patients with multivessel coronary artery disease: A pairwise and network meta-analysis of randomized trials: Front Cardiovasc Med, 2022; 9; 1000664
23. Puymirat E, Cayla G, Simon T, Multivessel PCI guided by FFR or Angiography for Myocardial Infarction: N Engl J Med, 2021; 385(4); 297-308
24. Musto C, Scappaticci M, Biondi-Zoccai G, Instantaneous wave-free ratio-guided revascularization of nonculprit lesions in STEMI patients with multivessel coronary disease: The WAVE registry: Catheter Cardiovasc Interv, 2022; 100(3); 351-59
25. Zaleska M, Koltowski L, Maksym J, Quantitative flow ratio and fractional flow reserve mismatch – clinical and biochemical predictors of measurement discrepancy: Postepy Kardiol Interwencyjnej, 2019; 15(3); 301-7
26. Westra J, Andersen BK, Campo G, Diagnostic performance of in-procedure angiography-derived quantitative flow reserve compared to pressure-derived fractional flow reserve: The FAVOR II Europe-Japan study: J Am Heart Assoc, 2018; 7(14); e009603
27. Westra J, Tu S, Campo G, Diagnostic performance of quantitative flow ratio in prospectively enrolled patients: An individual patient-data meta-analysis: Catheter Cardiovasc Interv, 2019; 94(5); 693-701
28. Erbay A, Steiner J, Lauten A, Assessment of intermediate coronary lesions by fractional flow reserve and quantitative flow ratio in patients with small-vessel disease: Catheter Cardiovasc Interv, 2020; 96(4); 743-51
29. Davies JE, Sen S, Dehbi HM, Use of the instantaneous wave-free ratio or fractional flow reserve in PCI: N Engl J Med, 2017; 376(19); 1824-34
Figures
 Figure 1. Subtotal occlusion of the right coronary artery (A) – infarct-related artery or culprit vessel. Non-culprit lesion on the left anterior descending artery (B). Both angiograms were performed during index PCI procedure.
Figure 1. Subtotal occlusion of the right coronary artery (A) – infarct-related artery or culprit vessel. Non-culprit lesion on the left anterior descending artery (B). Both angiograms were performed during index PCI procedure. Figure 2. Functionally significant stenosis on the left anterior descending artery. (A) QFR evaluation during the index PCI procedure. (B) QFR evaluation was performed 3 months and 14 days after index PCI (through the staged PCI procedure). The case showed the highest difference between QFR 1 and QFR 2 values in our study. However, it did not change the clinical treatment decision.
Figure 2. Functionally significant stenosis on the left anterior descending artery. (A) QFR evaluation during the index PCI procedure. (B) QFR evaluation was performed 3 months and 14 days after index PCI (through the staged PCI procedure). The case showed the highest difference between QFR 1 and QFR 2 values in our study. However, it did not change the clinical treatment decision. Figure 3. Four scatter diagrams, which show the strong positive correlations between quantitative flow ratio (QFR) 1 and QFR 2 numerical data in all lesions (A) and separate coronary arteries: (B) – left anterior descending artery (LAD); (C) – left circumflex artery (LCx); (D) – right coronary artery (RCA).
Figure 3. Four scatter diagrams, which show the strong positive correlations between quantitative flow ratio (QFR) 1 and QFR 2 numerical data in all lesions (A) and separate coronary arteries: (B) – left anterior descending artery (LAD); (C) – left circumflex artery (LCx); (D) – right coronary artery (RCA). Figure 4. Bland-Altman plot analysis shows the agreement among quantitative flow ratio (QFR) 1 and QFR 2 measurements. There are non-significant differences between the acute phase QFR and staged procedure QFR in the same lesions.
Figure 4. Bland-Altman plot analysis shows the agreement among quantitative flow ratio (QFR) 1 and QFR 2 measurements. There are non-significant differences between the acute phase QFR and staged procedure QFR in the same lesions. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








