18 September 2023: Clinical Research
Enhancing Functional Efficiency and Quality of Life through Revascularization Surgery in Peripheral Arterial Disease: A Comparative Analysis of Objective and Subjective Indicators
Anna NowaczykDOI: 10.12659/MSM.941673
Med Sci Monit 2023; 29:e941673
Abstract
BACKGROUND: This study aimed to compare the ankle-brachial index (ABI), maximal claudication distance (MCD), pain-free walking distance (PFWD), claudication pain, and quality of life (intermittent claudication questionnaire [ICQ]) before and 3 months after revascularization surgery in 98 patients diagnosed with peripheral arterial disease (PAD) at a single center in Poland.
MATERIAL AND METHODS: Ninety-eight patients were examined (77% men, 23% women, 65.65±7.27 years old), diagnosed with PAD, and qualified for revascularization. The diagnosis of PAD was made on the basis of ABI ≤0.9 and medical records. The patients underwent a noninvasive examination, including measurement of ABI (by Doppler with the EZ8 probe), assessment of the quality of life by ICQ, distance of intermittent claudication on a treadmill using the Gardner-Skinner protocol (including PFWD and MCD), and pain intensity during walking (numeric rating scale [NRS11]). The assessment was carried out twice: 1 to 5 days before surgery and 3 months after surgery.
RESULTS: There was an increase of ABI (0.4 vs 0.62, P<0.001), PFWD (26.64 vs 80.21, P<0.001), MCD (60.08 vs 181.85, P<0.001), and ICQ (79.92 vs 60.23, P<0.001) and reduction of PFWD pain (7.26 vs 6.05, P<0.001) and MCD pain (9.24 vs 8.11, P<0.001).
CONCLUSIONS: Revascularization surgery improved the ABI and patients functional efficiency expressed in the improvement of subjective indicators PFWD, MCD, NRS11, and ICQ. Patients who had a longer duration of disease had worse outcomes after revascularization. More attention should be paid to increasing access to preventive examinations aimed at early detection of PAD and the possibility of implementing conservative treatment.
Keywords: Angioplasty, Ankle Brachial Index, peripheral arterial disease, atherosclerosis, Quality of Life, Male, Humans, Female, Middle Aged, Aged, Intermittent Claudication, conservative treatment, Pain
Background
Increased attention is being given to assessing the distance of intermittent claudication and quality of life in patients with peripheral arterial disease (PAD), which is affected by the rising incidence of atherosclerosis in society [1]. PAD is increasingly recognized as an important cause of cardiovascular morbidity and mortality, which affects more than 230 million people worldwide [2]. According to the National Health and Nutrition Examination Survey, the prevalence of PAD is about 15% in individuals aged ≥70 years, but about 1% in those aged 40 to 49 years [2,3]. PAD is strongly linked with a heightened risk of death. According to statistics published in 2019, PAD was listed as the leading cause of death among 11 753 people out of a total of 58 210 recorded deaths [3]. Risk factors for atherosclerosis include hypertension, smoking, low activity level, and improper diet [4–11]. PAD is a narrowing and eventual complete occlusion of the main arteries that supply blood to the lower extremities. This causes pain and impaired gait economy, which limits the ability to move and reduces the level of physical activity of patients. Restricted blood flow to the limbs can cause pain in the thigh, buttock, or calf when walking, which is called intermittent claudication. Muscle pain, which is caused by reduced blood flow in the lower extremities, negatively affects the quality of life of patients with PAD [12]. PAD diagnostics include measurement of the ankle-brachial index (ABI), physical examination (pulse assessment, lower limb temperature), Doppler ultrasound, computed tomography and magnetic resonance angiography (MRA) [13]. The main goals of treating patients with PAD are to reduce cardiovascular risk and improve walking ability [14]. The basis of PAD treatment is lifestyle modification and eliminating risk factors, such as smoking, obesity, diabetes, hypertension, and hypercholesterolemia [15]. Treatment options for PAD depend on the stage of the disease and manifested symptoms. In the first place, treatment with exercise therapy is recommended, namely walking until moderate pain is achieved, with breaks for rest. It is recommended to train 3 to 4 times a week for 30 to 45 min [16]. In the treatment of PAD, pharmacotherapy is also used, including cilostazol and pentoxifylline [13]. In patients who do not improve with exercise and pharmacological treatment, surgical treatment should be considered [17]. The aim of invasive treatment is to extend the distance of intermittent claudication, reduce pain, improve the quality of life, and reduce the risk of cardiovascular complications [18–21]. Numerous randomized clinical trials and meta-analyses indicate also the effectiveness of supervised exercise programs in improving the functioning and quality of life of patients with intermittent claudication [22–28]. In the intermediate stages of the disease (Fonteine stage IIa), the primary intervention consists of physical training [29]. Endovascular or invasive surgical treatment is indicated only in some patients with PAD, in whom physical rehabilitation and conservative treatment have not brought the expected improvement and there is a high risk of developing critical limb ischemia, including the risk of amputation or even death of the patient [30,31]. It is assumed that invasive treatment is undertaken when the claudication distance is 100 m or less, and when the patient has pain at rest and necrotic lesions (III and IV, according to the Fontaine classification) [32–35]. We hypothesize that the improvement in objective parameters, such as ABI, maximal claudication distance (MCD), and pain-free walking distance (PFWD) associated with revascularization, improves subjective parameters, such as quality of life and pain level. Therefore, this study aimed to compare the ABI, MCD, PFWD, claudication pain, and quality of life (intermittent claudication questionnaire [ICQ]) before and 3 months after revascularization surgery in 98 patients diagnosed with PAD at a single center in Poland.
Material and Methods
ETHICS STATEMENT:
The study protocol was approved by the Local Bioethics Committee of the Nicolaus Copernicus University in Toruń and Ludwik Rydygier Collegium Medicum in Bydgoszcz (No. KB 331/2019) and conducted in accordance with the Helsinki Declaration of 1975. Written informed consent was obtained from the enrolled patients.
INCLUSION AND EXCLUSION CRITERIA:
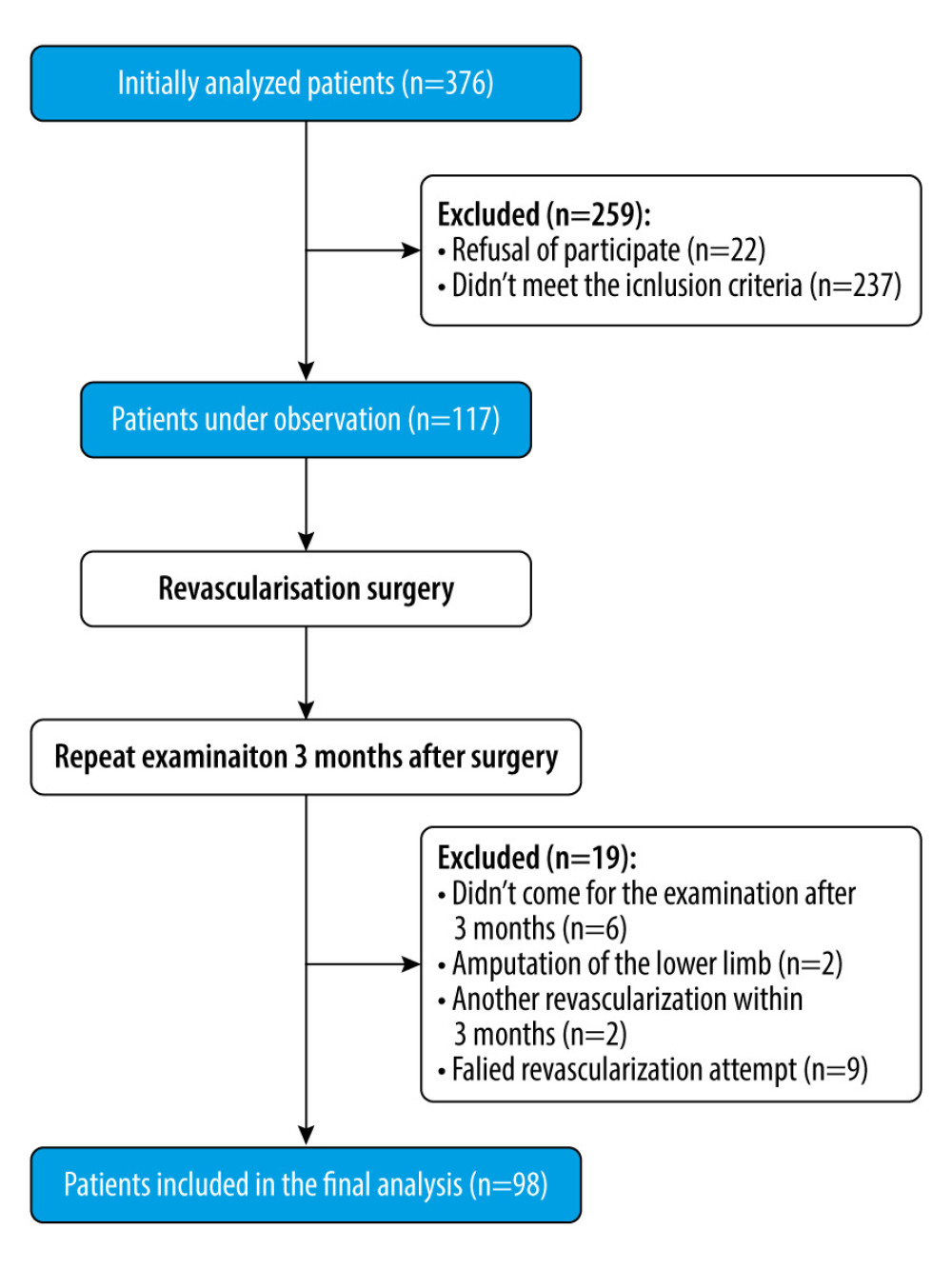
This prospective observational study included 98 adult patients with symptomatic PAD, treated with endovascular procedures (balloon angioplasty) and classic surgery (age ≥18 years <85, Fontaine stage IIb, claudication distance <100 m). The recruitment took place in the Clinic of Vascular Surgery and Angiology, University Hospital No. 1 in Bydgoszcz, Poland, between April 2019 and January 2022. Medical records were reviewed 1 to 5 days before planned revascularization to acquire the following data: type of planned revascularization, age, cardiovascular and non-cardiovascular comorbidities, laboratory results, previous course of the disease, and PAD-specific data (symptoms, Fontaine score, results of ultrasonography and MRA). Patients were qualified for the procedure on the basis of a clinical and angiographic examination (performed by a vascular surgeon), which was obtained from medical records. Clinical evaluation included Fontaine scale and ABI (diagnosis of PAD if rest ABI ≤0.9). Patients were eligible for the study if they had been diagnosed with PAD and had scheduled revascularization surgery (endovascular treatment with balloon angioplasty and classic surgery) and previous conservative therapy had failed. Initially, 117 patients with PAD were enrolled in the study, and 98 patients were included in the final analysis (Figure 1).
Inclusion criteria were (1) patients aged ≥18 years <85 years; (2) diagnosis of PAD; (3) qualification for endovascular or classic surgery treatment; (4) waiting for planned revascularization 1 to 5 days before the procedure; and (5) claudication distance < 100 m;
Exclusion criteria were (1) lower limb amputation, (2) patients with known and treated diabetes as well as those with a current fasting blood-glucose level >125 mg/dL, (3) pain in limbs at rest, (4) acute limb ischemia, (5) leg necrosis or ulcers, (6) disability and other factors preventing the treadmill test (including the use of assistive devices while moving, acute cardiovascular conditions, recent myocardial infarction, III–IV degree heart failure according to NYHA, diseases of the musculoskeletal system and the osteoarticular system) (7) sensory disorders, and (8) neurological and autoimmune diseases that can interfere with the interpretation of test results.
ASSESSMENT METHODS:
In this study, we assessed the following parameters: ABI value, claudication distance (both PFWD and MCD), and pain intensity on an 11-point numeric rating scale (NRS11) scale. To assess health-related quality of life, we used the intermittent claudication questionnaire (ICQ) specific for patients with claudication. Dyslipidemia and arterial hypertension were defined by prior medical diagnosis. Smoking, age, and disease duration were defined at the time of interview. The patients were examined twice: just before the surgery (1–5 days before) and 3 months after.
ABI MEASUREMENTS:
We used a non-directional Doppler with the EZ8 probe (MD2 Multi Dopplex® II, Huntleigh, Arjo, Inc., Addison, IL, USA) to measure the ABI. Preparation for the examination included prohibition of drinking coffee and smoking 2 h before the examination and 20 min of rest in the supine position on a rehabilitation table in a room with a temperature in the range of 21 to 24°C. Blood pressure measurements were always taken in the following order: right brachial artery, right dorsal artery of the foot, right posterior tibial artery, left dorsal pedis artery, left posterior tibial artery, left brachial artery, right brachial artery again (measurements were repeated to exclude false hypertension resulting from emotions and white coat syndrome) [36]. The results of the measurements were obtained by dividing the highest systolic blood pressure measured in the arteries of the lower extremities (dorsal pedis and posterior tibial arteries) by the highest systolic blood pressure measured in the brachial arteries [37]. During the study, the Doppler probe was placed in specific locations: to the posterior region of the medial malleolus when examining the posterior tibial artery; on the dorsum of the foot to verify the dorsal artery of the foot; and in the area of the cubital fossa of the arm to assess the flow wave of the brachial artery. The obtained ratio of measurements considered normal for adults ranged from 0.90 to 1.30, while ratios less than 0.9 indicated the presence of PAD [38,39]. The lower the ABI value, the more clinically advanced PAD and more advanced ischemia of the lower limb [40,41].
ASSESSMENT OF INTERMITTENT CLAUDICATION DISTANCE (PFWD AND MCD) AND PAIN INTENSITY (NRS11):
The examination was performed on a treadmill (Schwinn Fitness 810, Nautilus, Inc., Vancouver, WA, USA), according to the Gardner-Skinner protocol. The running speed of the treadmill was constant at 3.2 km/h. Every 2 min of walking, the inclination angle of the treadmill was increased by 2%, until it reached a maximum value of 12% [42,43]. Patients were instructed how to signal the first pain in the limb while walking or possible deterioration of well-being, which required the test to be stopped. During the walking test, the PFWD was assessed and measured in meters, until the first pain in the lower limb appeared (calf, foot, buttock, thigh). The patient assessed the intensity of pain for the first time, and then continued walking until severe pain appeared, preventing further activity and requiring rest. On this basis, the MCD was assessed. The patient assessed the intensity of maximum pain. We used the NRS11 to assess pain intensity, with 0 representing no pain, and 10 representing worst pain imaginable [44]. Patients were asked not to take analgesics 5 h before the scheduled examination.
QUALITY OF LIFE ASSESSMENT WITH ICQ:
The ICQ contained 16 questions about the severity of the problem, severity of symptoms, and the impact of intermittent claudication on the patient’s daily life. The answers were scored in the range from 0 to 5 points, and then summed. A higher score indicated a significant severity of the problem and greater limitations caused by intermittent claudication. The total sum of the test scores ranged from 0 (indicating good quality of life) to 80 (indicating poor quality of life). The final results were converted into percentages, where the maximum score was 80 points (meant 100%). The questionnaire contained written instructions for the patients to answer the questions on their own. Before returning the questionnaires, the completeness of the answers provided by the patients was checked. Only completely and correctly completed questionnaires were included in the final analysis.
STATISTICAL ANALYSIS:
The sample size was estimated based on the Central Statistical Office report from 2018. Estimation was calculated according to the following formula:
STATISTICAL METHODS:
Equity of variance and normality of data distribution were tested with Levene and Kolmogorov-Smirnov tests, respectively. All results presented in the text and tables are expressed as means±standard deviation or number and percentage. ABI, PFWD, MCD, NRS, and ICQ score parameters were compared using
Results
CHARACTERISTICS OF THE STUDY GROUP:
The study group included 98 patients with PAD (65.77±7 years old, male/female: 81.63%/18.37%), who accepted the invitation for the interview and the examination before and after revascularization surgery. The duration of disease was an average of 3.94±2 years. Patients at high cardiovascular risk participated in the study. Risk factors included dyslipidemia (100%), hypertension (76%), and active smoking (80.61%). The common comorbidities in patients with PAD were myocardial infraction (22.45%), cerebrovascular disease (14.29%), and any tumor without metastasis (7.14%).
MEASUREMENTS OF ABI, PFWD, MCD AND PAIN LEVEL:
The follow-up visit 3 month after revascularization showed that patients with PAD had significantly higher ABI, PFWD, and MCD compared with measurements before surgery. Statistically significantly lower pain levels were demonstrated during PFWD and MCD. The average ABI value increased by 0.22±0.09 (P<0.001). The average PFWD increased by 53.57±20.13 m (P<0.001) after surgery. The average MCD increased by 121.77±78.34 m (P<0.001). The level of pain intensity after surgery decreased on average by 1.21±0.87 (P<0.001) during PFWD and by 1.13±0.66 (P<0.001) during MCD on the NRS11 scale. The differences were statistically significant (Table 1).
CORRELATION OF ABI, PFWD, MCD, ICQ, AND PAIN LEVELS WITH DISEASE DURATION AND AGE:
The longer the duration of the disease, the lower was the improvement of the following parameters: PFWD (
RESULTS OF QUALITY OF LIFE AND FUNCTIONAL EFFICIENCY MEASUREMENTS (ICQ):
The results of the ICQ showed that the greatest limitations in the daily activity of patients with PAD resulted from the need to stop and rest many times (more than 3 times a day) (4.99±0.10) and the high intensity of pain (4.80±0.39). Intermittent claudication also limited walking more than 1500 m (4.78±0.51), climbing stairs (4.74±0.53), and using public transport (4.56±0.78). Patients also reported limited ability to work and to deal with everyday matters, such as shopping and going to the post office or bank (4.30±0.76). Intermittent claudication affected social activity (2.73±1.00) and feelings of depression and worrying, but to moderate degree (2.68±0.94). There was an improvement in all areas of the questionnaire (on average by 0.55–1.20 points), except for the need to stop during activity (the difference was 0.00±0.16). The revascularization procedure contributed to a statistically significant improvement in the patients’ total functional capacity and total ICQ score (79.92±8.29 vs 60.23±12.36, P<0.001; Table 2).
Discussion
LIMITATIONS OF THE STUDY:
The findings of this study should be interpreted after consideration of the strengths and weaknesses of the investigation. We did not include BMI data in the study, and there are studies that indicate the influence of body weight on the length of the claudication distance [74,75]. The study did not differentiate patients according to the type of revascularization performed (classic, endovascular, hybrid). Endovascular therapy is associated with a lower risk of complications, while the classic procedure is more effective in long-term studies [76]. We excluded patients with diabetes from the study, but it should be remembered that a very high percentage of patients with PAD also have diabetes [77]. We decided to exclude these patients because of possible abnormal ABI, abnormal pain sensation due to neuropathy, and high incidence of ulceration. Our study also did not assess patients’ physical activity levels within the 3 months after surgery. The beneficial effect of physical activity on the length of the claudication distance is confirmed by numerous studies [78–80]. However, our study included patients who were qualified for revascularization due to the fact that conservative therapy did not bring the expected results (Fontaine IIb and claudication distance <100 m). Revascularization is currently the best form of treatment in very advanced clinical stages of PAD (Fontaine IIb, III, and IV), when treatment with physical exercise does not bring the expected results [81]. Our study included the assessment of parameters 3 months after the procedure. However, long-term studies show a gradual deterioration of the assessed parameters, ie, ABI and claudication distance [81–83]. Owing to the gradual management of PAD, in addition to the necessary revascularization, special attention should be paid to the implementation of conservative treatment methods that will maintain the benefits of the performed revascularization [84,85].
Conclusions
The revascularization surgery performed in the course of PAD significantly improves the blood supply to the lower limbs, expressed as the ABI index. The PFWD and MCD significantly increase within 3 months of revascularization surgery. The improvement of objective parameters, such as ABI and intermittent claudication distance, is associated with a significant improvement in subjective parameters, such as quality of life and pain level.
Patients who have had PAD for a shorter period of time achieve greater improvement in the analyzed parameters. In patients with a significantly advanced clinical stage of PAD, revascularization should be chosen as the form of treatment.
Tables
Table 1. Comparison of the values of the assessed parameters (pain-free walking distance, maximal claudication distance, ankle-brachial index, pain level, duration of disease and age of patients) before and after the revascularization surgery.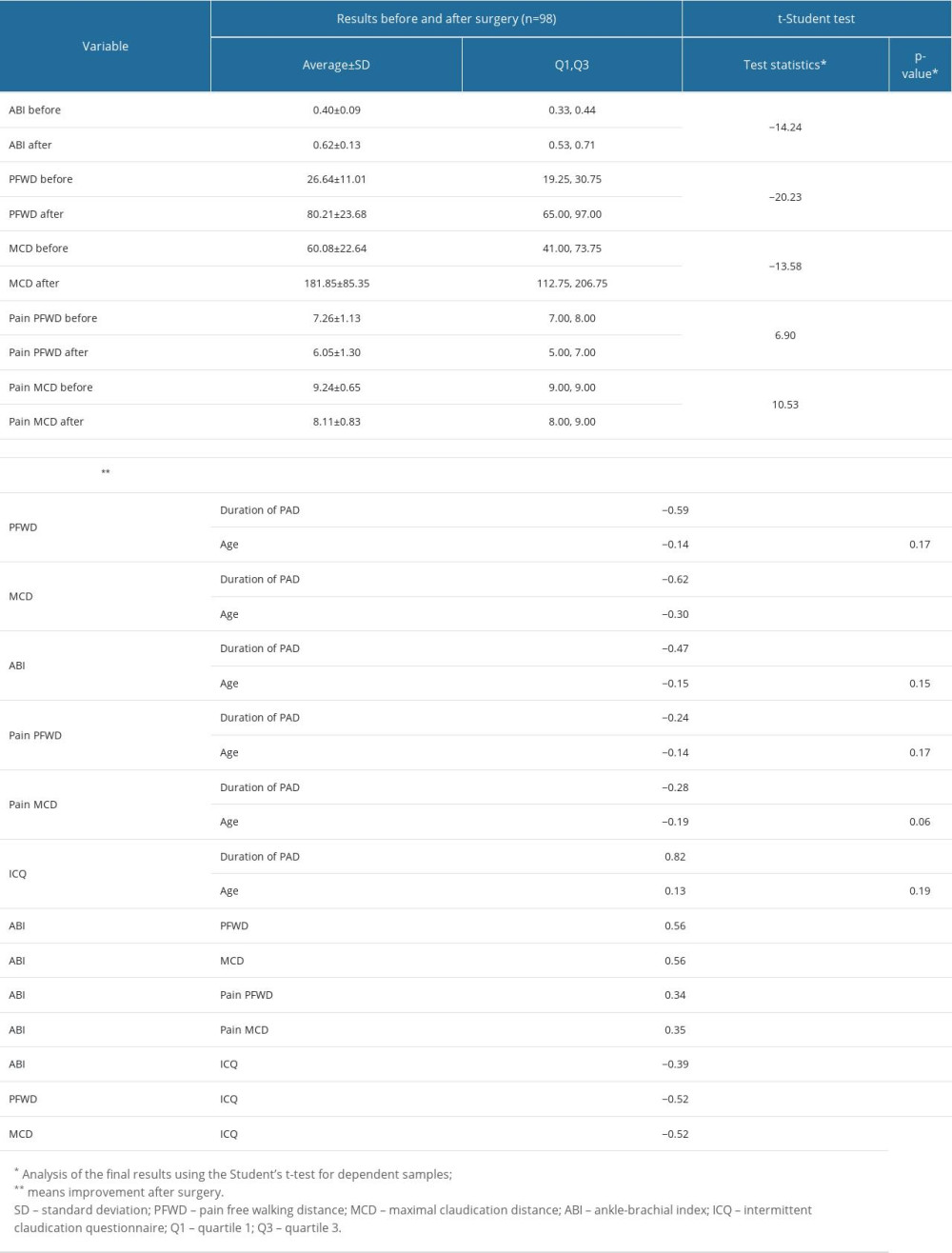 Table 2. Comparison of changes in specific questions and intermittent claudication questionnaire (ICQ) total score in patients with peripheral arterial disease (PAD) before and after revascularization surgery.
Table 2. Comparison of changes in specific questions and intermittent claudication questionnaire (ICQ) total score in patients with peripheral arterial disease (PAD) before and after revascularization surgery.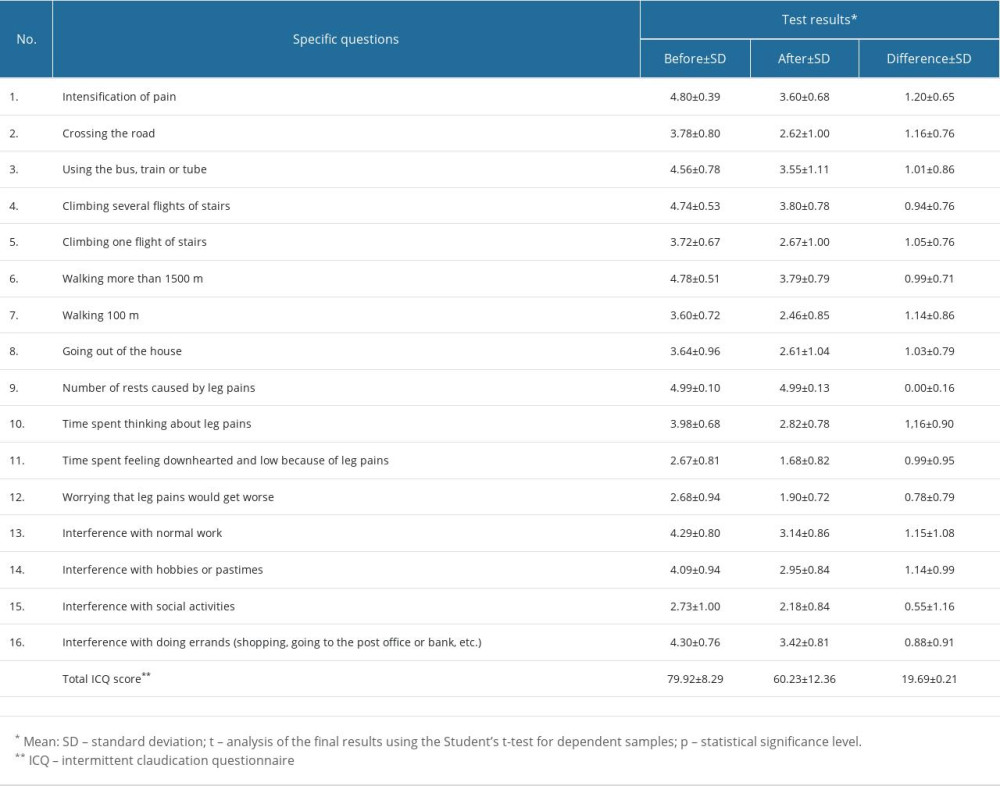
References
1. Signorelli SS, Marino E, Scuto S, Di Raimondo D, Pathophysiology of peripheral arterial disease (PAD): A review on oxidative disorders: Int J Mol Sci, 2020; 21(12); 4393
2. Song P, Rudan D, Zhu Y, Global, regional, and national prevalence and risk factors for peripheral artery disease in 2015: An updated systematic review and analysis: Lancet Glib Health, 2019; 7(8); e1020-e30
3. Aday A, Matsushita K, Epidemiology of peripheral artery disease and polyvascular disease: Circulation, 2021; 128(12); e1818-e32
4. Criqui M, Aboyans V, Epidemiology of peripheral artery disease: Circ Res, 2015; 116(9); e1509-e26
5. Virani S, Alonso VA, Aparicio HJ, Heart disease and stroke statistics – 2021 update. A report from the American Heart Association: Circulation, 2021; 143(8); e254-e743
6. Olin J, Sealove B, Peripheral artery disease: Current insight into the disease and its diagnosis and management: Mayo Clin Proc, 2010; 85(7); e678-e92
7. Bø E, Hisdal J, Cvancarova M, Twelve-months follow-up of supervised exercise after percutaneous transluminal angioplasty for intermittent claudication: A randomised clinical trial: Int J Environ Res Public Health, 2013; 10(11); e5998-e6014
8. Joosten M, Pai J, Bertoia M, Associations between conventional cardiovascular risk factors and risk of peripheral artery disease in men: JAMA, 2012; 308(16); e1660-67
9. Hiatt W, Goldstone J, Smith S, Atherosclerotic Peripheral Vascular Disease Symposium II: Nomenclature for vascular diseases: Circulation, 2008; 118(25); e2826-29
10. Nehler M, Duval S, Diao L, Epidemiology of peripheral arterial disease and critical limb ischemia in an insured national population: J Vasc Surg, 2014; 60(3); e686-95
11. Criqui M, Peripheral arterial disease – epidemiological aspects: Vasc Med, 2001; 6(3); 3-7
12. Kim M, Kim Y, Ryu G, Choi M, Functional status and health-related quality of life in patients with peripheral artery disease: A cross-sectional study: Int J Environ Res Public Health, 2021; 18(20); e109-41
13. Zemaitis MR, Boll JM, Dreyer MA, Peripheral arterial disease. [Updated 2023 May 23]: StatPearls [Internet], 2023, Treasure Island (FL), StatPearls Publishing Available from:https://www.ncbi.nlm.nih.gov/books/NBK430745/
14. Debus ES, Kriston L, Schwaneberg T, Rationale and methods of the IDOMENEO health outcomes of the peripheral arterial disease revascularization study in the GermanVasc registry: Vasa, 2018; 47(6); 499-505
15. Jin J, Screening for peripheral artery disease with Ankle-Brachial Index: JAMA, 2018; 320(2); 212
16. Shabani Varaki E, Gargiulo GD, Penkala S, Breen PP, Peripheral vascular disease assessment in the lower limb: A review of current and emerging non-invasive diagnostic methods: Biomed Eng Online 11, 2018; 17(1); 61
17. Cooper K, Majdalany BS, Kalva SPExpert Panel on Vascular Imaging: J Am Coll Radiol, 2018; 15(5S); S104-S15
18. Malgor R, Alalahdab F, Elraiyah T, A systematic review of treatment of intermittent claudication in the lower extremities: J Vasc Surg, 2015; 61(3); e154-73
19. Kojima I, Ninomiya T, Hata J, A low ankle brachial index is associated with an increased risk of cardiovascular disease: the Hisayama study: J Atheroscler Thromb, 2014; 21(9); e966-73
20. Benjamin E, Blaha M, Chiuve S, Heart disease and stroke statistics – 2017 update: A report from the American Heart Associatione: Circulation, 2017; 135(10); e146-e603
21. Sampson U, Fowkes F, McDermott M, Global and regional burden of death and disability from peripheral artery disease: 21 world regions, 1990 to 2010: Glob Heart, 2014; 9(1); 145-58
22. Fakhry F, Spronk S, van der Laan L, Endovascular revascularization and supervised exercise for peripheral artery disease and intermittent claudication: A randomized clinical trial: JAMA, 2015; 314(18); 1936-44
23. Ehrman J, Gardner A, Salisbury D, Lui K, Treat-Jacobson D, Supervised exercise therapy for symptomatic peripheral artery disease: A review of current experience and practice-based recommendations: J Cardiopulm Rehabil Prev, 2023; 41(1); 15-21
24. Piepoli M, Hoes A, Agewal SSixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice, Developed with the special contribution of the European Association for Cardiovascular Prevention Rehabilitation (EACPR): Eur Heart J, 2016; 37(29); e2315-81
25. National Coverage Analysis: Supervised exercise therapy (SET) for symptomatic peripheral artery disease (PAD) Oct, 2018, Center for Medicare Medicaid Services Available from: https://wwwcmsgov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=N&NCAId=287
26. Fokkenrood H, Bendermacher B, Lauret G, Supervised exercise therapy versus non-supervised exercise therapy for intermittent claudication: Cochrane Database Syst Rev, 2013; 23(8); CD005263
27. Fakhry F, Luijtgaarden K, Bax L, Supervised walking therapy in patients with intermittent claudication: J Vasc Surg, 2012; 56(4); 1132-42
28. Gommans L, Saarloos R, Scheltinga M, Editor’s choice: The effect of supervision on walking distance in patients with intermittent claudication: A meta-analysis: Eur J Vasc Endovasc Surg, 2014; 48(2); 169-84
29. Thukkani AK, Kinlay S, Endovascular intervention for peripheral artery disease: Circ Res, 2015; 116(9); 1599-613
30. Aboyans V, Ricco J, Bartelink M, 2017 ESC guidelines on the diagnosis and treatment of peripheral arterial diseases, in collaboration with the European Society for Vascular Surgery (ESVS): Kardiol Pol, 2017; 75(11); 1065-60
31. Jakubseviciene E, Vasiliauskas D, Velicka L, Effectiveness of a new exercise program after lower limb arterial blood flow surgery in patients with peripheral arterial disease: A randomized clinical trial: Int J Environ Res Public Health, 2014; 11(8); 7961-76
32. Rowe W, Lee F, Weaver D, Etzioni D, Patterns of treatment for peripheral arterial disease in the United States: 1996–2005: J Vasc Surg, 2009; 49(4); 910-17
33. Stroke O, Tendera M, Aboyans V, ESC guidelines on the diagnosis and treatment of peripheral artery diseases: Eur Heart J, 2011; 32(22); 2851-906
34. Crawford F, Welch K, Andras A, Chappell FM, Ankle brachial index for the diagnosis of lower limb peripheral arterial disease: Cochrane Database Syst Rev, 2016; 9(9); CD010680
35. Karpińska A, Szewczyk M, Cwajda-Białasik J, Mościcka P, Assessment of functional efficiency and quality of life in patients with lower limb ischemia after revascularization surgery – preliminary results: Surg Vasc Nurs, 2020; 4(14); 170-77
36. Freitas D, Toneti AN, Cesarino EJ, Desidério VL, Cardiovascular risk in white coat hypertension: An evaluation of the ankle brachial index: J Vasc Nurs, 2014; 32(2); 38-45
37. Hammad TA, Smolderen KG, Spertus JA, Associations of exercise ankle-brachial index, pain-free walking distance and maximum walking distance with the Peripheral Artery Questionnaire: Finding from the PORTRAIT PAD Registry: Vasc Med, 2019; 24(1); 32-40
38. McDermott M, Liu K, Criqui M, Ankle-brachial index and subclinical cardiac and carotid disease: the multi-ethnic study of atherosclerosis: Am J Epidemiol, 2005; 162(1); 33-41
39. Schroder F, Diehm N, Kareem S, A modified calculation of ankle-brachial pressure index is far more sensitive in the detection of peripheral arterial disease: J Vasc Surg, 2006; 44(3); 531-36
40. Aboyans V, Criqui M, Abraham P, Measurement and interpretation of the ankle-brachial index: A scientific statement from the American Heart Association: Circulation, 2012; 126(24); 2890-909
41. McDermott M, Criqui M, Liu K, Lower ankle/brachial index, as calculated by averaging the dorsalis pedis and posterior tibial arterial pressures, and association with leg functioning in peripheral arterial disease: J Vasc Surg, 2000; 32(6); 1164-71
42. Fokkenrood H, Houten M, Houtermann S, Agreements and discrepancies between the estimated walking distance, nongraded and graded treadmill testing, and outside walking in patients with intermittent claudication: Ann Vasc Surg, 2015; 29(6); 1218-24
43. Mika P, Andrzejczak A, Konik A, Clinical methods of evaluating patients with intermittent claudication: Med Rehabil, 2011; 15(1); 10-20
44. Rodriguez C, Pain measurement in the elderly: A review: Pain Manage Nurs, 2001; 2(2); 38-46
45. Migdalski A, Jawień A, Clinical value of ankle brachial pressure index: Surg Vasc Nurs, 2007; 1(2); 81-86
46. Nordanstig J, Taf C, Hensäter M, Two-year results from a randomized clinical trial of revascularization in patients with intermittent claudication: Br J Surg, 2016; 103(10); 1290-99
47. Spannbauer A, Berwecki A, Chwała M, Maximal walking distance in patients with PAD after aorto-bifemoral bypass (ABF) and rehabilitation treatment: Physiother Prev Civiliz Dis Treat, 2016; 1(1); 373-91
48. Fakhry F, Fokkenrood HJ, Spronk S, Endovascular revascularisation versus conservative management for intermittent claudication: Cochrane Database Syst Rev, 2018; 3(3); CD010512
49. Jaquinandi V, Picquet J, Saumet J, Functional assessment at the buttock level of the effect of aortobifemoral bypass surgery: Ann Surg, 2008; 247(5); 869-76
50. Ferrari R, Vidotto G, Ferraro T, Recovery expectations and quality of life after revascularization treatments: Br J Med Med Res, 2015; 5(11); 1431-40
51. Hogan SE, Nehler MR, Anand S, Improvement in walking impairment following surgical and endovascular revascularisation: Insights from VOYAGER PAD: Vasc Med, 2022; 27(4); 343-49
52. Koelemay MJ, Reijen NS, Dieren S, Editor’s choice: Randomised clinical trial of supervised exercise therapy vs. endovascular revascularisation for intermittent claudication caused by iliac artery obstruction: The SUPER study: Eur J Vasc Endovasc Surg, 2022; 63(3); 421-29
53. Klaphake S, Buettner S, Ultee KH, Combination of endovascular revascularisation and supervised exercise therapy for intermittent claudication: A systematic review and meta-analysis: J Cardiovasc Surg (Torino), 2018; 59(2); 150-57
54. Nordanstig J, Taft C, Hensater M, Improved quality of life after 1 year with an invasive versus a noninvasive treatment strategy in claudicants: One-year results of the Invasive Revascularisation or Not in Intermittent Claudicaction (IRONIC) Trial: Circularion, 2014; 139(12); 939-47
55. Momsen AM, Jensen MB, Norager CB, Quality of life and functional status after revascularisation or conservative treatment in patients with intermittent claudication: Vasc Endovascular Surg, 2011; 45(2); 122-29
56. Wiśniewska A, Szewczyk MT, Cwajda-Białasik J, Beliefs about pain control in patients with a chronic ischemia of lower limb: Surg Vasc Nurs, 2009; 3(3); 113-21
57. Hallin A, Bergqvist D, Fugl-Meyer K, Holmberg L, Areas of concern, quality of life satisfaction in patients with peripheral vascular disease: Eur J Vasc Endovasc Surg, 2002; 24(3); 255-63
58. Hernandez-Osma E, Cairols MA, Marti XE, Impact of treatment on the quality of life in patients with critical limb ischaemia: Eur J Vasc Endovasc Surg, 2002; 23(6); 491-94
59. Golledge J, Leicht AS, Yip L, Relationship between disease specific quality of life measures, physical performance, and activity in people with intermittent claudication caused by peripheral artery disease: Eur J Vasc Endovasc Surg, 2020; 59(6); 957-64
60. Tu HP, Lin CH, Hsieh HM, Prevalence of anxiety disorder in patients with type 2 diabetes: A nationwide population-based study in Taiwan 2000–2010: Psychiatr Q, 2017; 88(1); 75-91
61. Andrade-Lima A, Junior NS, Chehuen M, Walking training improves systemic and local pathophysiological processes in intermittent claudication: Eur J Vasc Endovasc Surg, 2021; 61(6); 954-63
62. Remes L, Isoaho R, Vahlberg T, Quality of LiFE among lower extremity peripheral arterial disease patients who have undergone endovascular or surgical revascularisation: A case-control study: Eur J Vasc Endovasc Surg, 2010; 40(5); 618-25
63. Vlajinac H, Marinkovic J, Tanaskovic S, Quality of life after peripheral bypass surgery: A 1 year follow-up: Wien Klin Wochenschr, 2015; 127(5–6); 210-17
64. Peterson S, Remaekers BL, Olie RH, Comparsion of three generic quality-of-life metrics in peripheral aarterial disease patients undergoing conservative and invasive treatments: Qual Life Res; 28(8); 2257-79 209
65. Wohlgemuth WA, Safanova O, Engelhardt M, Improvement of the quality of life concerning the health of patients with peripheral arterial disease (PAD) after successful bypass surgery: Vasa, 2008; 37(4); 338-44
66. Long J, Modrall JG, Parker BJ, Correlation between ankle-brachial index, symptoms, health-related quality of life in patients with peripheral vascular disease: J Vasc Surg, 2004; 39(4); 723-27
67. Poredos P, Boc V, Zlajpah U, Walking impairment questionnaire and walking tests are reliable indicatiors of success of treatment of peripheral artery disease: Angiology, 2022; 73(4); 331-37
68. Henni S, Ammi M, Semporé Y, Treadmill measured vs. questionnaire estimated changes in walking ability in patients with peripheral artery disease: Eur J Vasc Endovasc Surg, 2019; 57(5); 676-84
69. Chong PF, Garratt AM, Golledge J, The intermittent claudication questionnaire: A patient-assessed condition-specific health outcome measure: J Vasc Surg, 2002; 36(4); 764-71
70. McDermott MM, Guralnik JM, Tian L, Incidence and prognostic significance of depressive symptoms in peripheral artery disease: J Am Heart Assoc, 2016; 5(3); 29-59
71. Kieback AG, Espinola-Klein C, Lamina C, One simple claudication question as first step in Peripheral Arterial Disease (PAD) screening: A metaanalysis of the association with reduced Ankle Brachail Index (ABI) in 27,945 subjects: PLoS One; 14(11); e0224608 209
72. Curry SJ, Krist AH, Owens DK, Screening for peripheral artery disease and cardiovascular disease risk assessment with the ankle-brachial index: US Preventive Services Task Force Recommendation Statement: JAMA, 2018; 320(2); 177-83
73. Firnhaber JM, Powell CS, Lower extremity peripheral artery disease: Diagnosis and treatment: Am Fam Physician, 2019; 99(6); 362-69
74. Jansen SC, Hoorweg BB, Hoeks SE, A systematic review and meta-analysis of the effects of supervised exercise therapy on modifiable cardiovascular risk factors in intermittent claudication: J Vasc Surg, 2019; 69(4); 1293-308
75. Madden J, Brunner A, Dastur ND, Fish oil included increase in walking distance, but not ankle brachial pressure index, in peripheral arterial disease is dependent on both body mass index and inflammatory genotype: Prostaglandins Leukot Essent Fatty Acids, 2007; 76(6); 331-40
76. Abouhamda A, Alturkstani M, Jan Y, Lower sensitivity of ankle-brachial index measurements among people suffering with diabetes-associated vascular disorders: A systematic review: SAGE Open Med, 2019; 7; 2050312119835038
77. Pymer S, Tew G, Palmer J, Home-based exercise programmes for individuals with intermittent claudication: A protocol for an updated systematic review and meta-analysis: SAGE Open Med, 2018; 6; 2050312118818295
78. McDermott M, Ades P, Guralnik J, Treadmill exercise and resistance training in patients with peripheral arterial disease with and without intermittent claudication: A randomized controlled trial: JAMA, 2009; 301(2); 165-74
79. Mika P, Konik A, Januszek R, Comparison of two treadmill training programs on walking ability and endothelial function in intermittent claudication: Int J Cardiol, 2013; 168(2); 838-42
80. Biswas M, Capell W, McDermott M, Exercise training and revascularization in the management of symptomatic peripheral artery disease: JACC Basic Trans Sci, 2021; 6(2); 174-88
81. Surowiec SM, Davies MG, Eberly SW, Percutaneous angioplasty and stenting of the superficial femoral artery: J Vasc Surg, 2005; 41(2); 269-78
82. Giustino G, Serruys PW, Sabik JF, Mortality after repeat revascularization following PCI or CABG for left main disease: the EXCEL trial: JACC Cardiovasc Interv, 2020; 13(3); 375-87
83. Juszkat R, Pukacki F, Żabicki B, Results of percutaneous angioplasty of the femoral-popliteal segment in lesions type A disease according to the TASC classification: Pol Surg, 2007; 9(4); 193-201
84. Steunenberg SL, de Vries J, Raats JW, Quality of life and mortality after endovascular, surgical, or conservative treatment of elderly patients suffering from critical limb ischemia: Ann Vasc Surg, 2018; 51; 95-105
85. McDermott MM, Exercise training for intermittent claudication: J Vasc Surg, 2017; 66(5); 1612-20
Tables
 Table 1. Comparison of the values of the assessed parameters (pain-free walking distance, maximal claudication distance, ankle-brachial index, pain level, duration of disease and age of patients) before and after the revascularization surgery.
Table 1. Comparison of the values of the assessed parameters (pain-free walking distance, maximal claudication distance, ankle-brachial index, pain level, duration of disease and age of patients) before and after the revascularization surgery. Table 2. Comparison of changes in specific questions and intermittent claudication questionnaire (ICQ) total score in patients with peripheral arterial disease (PAD) before and after revascularization surgery.
Table 2. Comparison of changes in specific questions and intermittent claudication questionnaire (ICQ) total score in patients with peripheral arterial disease (PAD) before and after revascularization surgery. Table 1. Comparison of the values of the assessed parameters (pain-free walking distance, maximal claudication distance, ankle-brachial index, pain level, duration of disease and age of patients) before and after the revascularization surgery.
Table 1. Comparison of the values of the assessed parameters (pain-free walking distance, maximal claudication distance, ankle-brachial index, pain level, duration of disease and age of patients) before and after the revascularization surgery. Table 2. Comparison of changes in specific questions and intermittent claudication questionnaire (ICQ) total score in patients with peripheral arterial disease (PAD) before and after revascularization surgery.
Table 2. Comparison of changes in specific questions and intermittent claudication questionnaire (ICQ) total score in patients with peripheral arterial disease (PAD) before and after revascularization surgery. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387