27 July 2020: Clinical Research
Clinical Characteristics and Prognosis of Osteoclast-like Giant Cell Tumors of the Pancreas Compared with Pancreatic Adenocarcinomas: A Population-Based Study
Mingfang Xu1ABCDE, Wei Chen2ABCD, Dong Wang1ABCD, Mao Nie3EFG*DOI: 10.12659/MSM.922585
Med Sci Monit 2020; 26:e922585
Abstract
BACKGROUND: The incidence of osteoclast-like giant cell tumor of the pancreas (OGTP) is very low, and relatively little OGTP clinical data is available. The present study, therefore, sought to conduct a more comprehensive analysis of the clinical characteristics and prognosis of OGTP.
MATERIAL AND METHODS: A large population-based cohort analysis was conducted using the Surveillance, Epidemiology and End Results (SEER) registry. We conducted a systematic assessment of the demographic and clinical characteristics of these patients, in addition to assessing available prognostic and therapeutic data corresponding to their disease. We further compared overall survival (OS) in these OGTP and pancreatic adenocarcinoma (PA) patient cohorts, adjusting for sex, grade, stage, and surgical treatment by propensity score matching (PSM).
RESULTS: We included a total of 47 OGTP patients and 73 150 PA patients in the present analysis. The mean ages of PA and OGTP diagnosis were 68.0 and 62.8 years, respectively. Compared with PA patients, OGTP patients were more likely to be female (70.2% versus 48.7%, P<0.01), to have early-stage disease, to have lower rates of lymph node metastasis (17.0% versus 28.8%, P<0.01) and distant metastasis (17.0% versus 45.1%, P<0.01), and to have higher rates of tumor resection (70.2% versus 15.4%, P<0.01). OGTP patients also had a significantly longer median OS than did PA patients (13 months versus 6 months; hazard ratio [HR] 0.55, 95% confidence interval [CI] 0.37–0.57, P<0.0001). No significant differences in tumor site preferences were detected. Our findings also suggested that being female, having early-stage disease, and undergoing surgical resection may be associated with a more favorable prognosis in patients with OGTP.
CONCLUSIONS: OGTP patients had distinctive clinical characteristics and a better prognosis compared with PA patients. Understanding these differences will help clinicians accurately recognize these diseases. Radical resection was beneficial to the survival of OGTP patients.
Keywords: Pancreatic Neoplasms, Population Characteristics, Adenoma, Carcinoma, Pancreatic Ductal, Cohort Studies, Databases, Genetic, Giant Cell Tumors, Osteoclasts, Proportional Hazards Models, SEER Program
Background
Pancreatic cancer can manifest as a wide range of pathological types. It is generally divided into epithelial and non-epithelial tumors according to histological differentiation. Epithelial neoplasms can be exocrine or endocrine, and exocrine neoplasms can be categorized into ductal neoplasms or acinar neoplasms [1]. Ductal adenocarcinoma occupies the first among all types, making up 80% to 90% of pancreatic cancer cases. In contrast, osteoclast-like giant cell tumor of the pancreas (OGTP) is an extremely rare type of pancreatic cancer which was first reported by Juan Rosai in 1968 [2]. These tumors are non-endocrine in origin, with high rates of malignancy and poor differentiation. Similar osteoclast-like giant cell tumors (OGCs) have similarly been detected in many other sites in the body, including in the thyroid, skin, urinary tract, lungs, parotid gland, breast, and in various soft tissues [3–13]. In pancreatic manifestations, such OGCs most often present in the context of ductal adenocarcinoma, with a histological appearance consistent with that of giant cell tumors of the bone. Two primary cell populations, including mononuclear stromal cells and giant multinucleated reactive cells, compose OGTP tumors which appear similar to osteoclasts [14]. The specific origin of these osteoclast-like cells in OGTP remained controversial, with different studies suggesting them to be of epithelial or mesenchymal lineages [15–18].
Given the rarity of this condition, no large-scale clinical studies of OGTP are available at present, with the bulk of relevant publications instead being case reports. This has led to significant speculation regarding this condition. For example, some researchers have proposed that OGTP does not exhibit any sex-bias with respect to its development and that it has a relatively good prognosis as compared to other pancreatic tumors owing to the fact that OGTP typically only exhibits localized spreading, with metastasis to lymph nodes and distant sites being slow to develop and rarely reported [19]. In contrast, however, some researchers have reported OGTP to be more common in females [20] and to have a worse prognosis [21,22]. The site preferences for OGTP tumors similarly remains controversial. As such, there is a clear need for further comprehensive studies of OGTP in order to guide oncologists and to allow them to make correct diagnostic and treatment decisions. To that end, we conducted the present large cohort to compare OGTP and pancreatic adenocarcinoma (PA) using the SEER database.
Material and Methods
ETHICS:
The Research Ethics Committee of Daping Hospital approved the present study. No patient consent was required for the use of data contained in the SEER database, as in the USA cancer is considered to be a reportable disease.
POPULATION:
Both pancreatic adenocarcinoma, NOS (not otherwise specified) and carcinoma with OGTP patient cases contained in the National Cancer Institute SEER database (
STATISTICAL ANALYSIS:
Chi-squared tests were used for comparing categorical data, which were given as numbers and percentages, while continuous data were given as mean (median) range and were compared via Student’s
Results
PATIENTS CHARACTERISTICS:
Through a search of the SEER database, we identified 73 150 total PA patients and 47 OGTP patients who were diagnosed between 2001 and 2016. OGTP patients had a mean age of 62.8 years at the time of diagnosis, whereas in PA patients this age was 68 years at time of diagnosis. Table 1 shows the demographic and clinical characteristics of these 2 patient cohorts before and after PSM. There were significant differences between these OGTP and PA patient cohorts with respect to age at diagnosis, sex, staging, lymph node involvement, distant metastasis, rates of surgical treatment, and survival outcomes before but not after PSM. In contrast, no significant differences in patient race, year of diagnosis, or tumor site preferences were observed between these groups. Significantly more OGTP patients were alive as of last follow-up relative to PA patients (23.4% versus 5.3%, P<0.001).
SURVIVAL ANALYSIS:
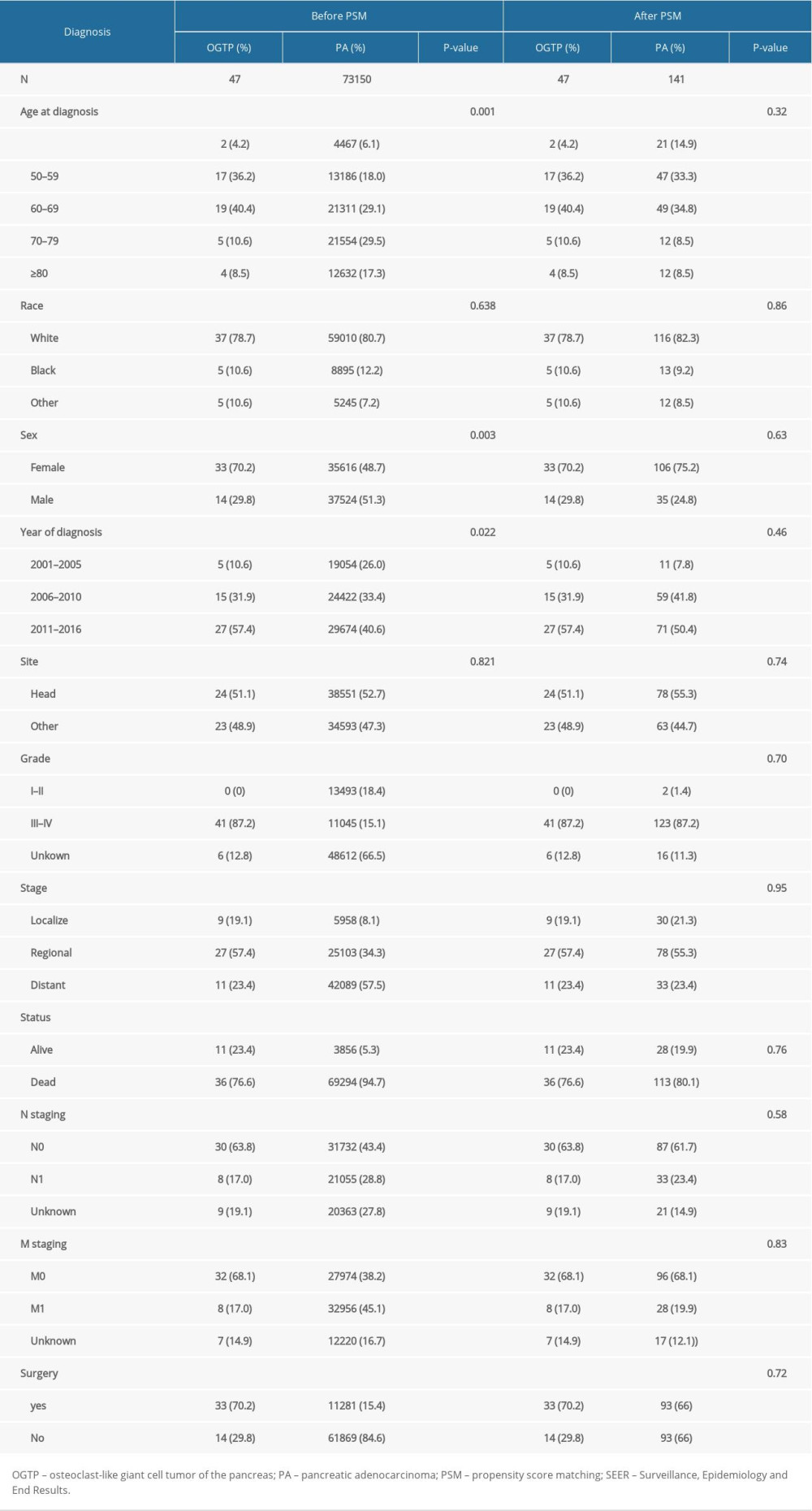
We next compared survival curves for patients with PA and OGTP (Figure 1A), revealing that OGTP patients had a median overall survival (mOS) of 13 months (range: 1–160 months), whereas PA patients had a mOS of 6 months (range: 1–227 months) (HR 0.55, 95% CI 0.37–0.57) before propensity score matching. This indicated a significant difference in mOS between these 2 patient cohorts P<0.0001). However, the survival curves were not significantly different after PSM (Figure 1B). We then compared OGTP patient survival outcomes as a function of whether or not patients underwent tumor resection (Figure 2), revealing that those patients treated via surgical resection had significantly better survival outcomes than those patients who did not undergo surgery (33 months versus 5 months, HR 0.07, 95% CI 0.03–0.21) (P<0.0001). We further compared OGTP patient outcomes as a function of disease stage (Figure 3), revealing localized, regional, and distantly metastasized OGTP to have mOS values of 73, 26, and 7 months, respectively (P<0.01). When OGTP patient survival was compared based upon patient sex (Figure 4), we found females to have a significantly better prognosis than male patients (36 months versus 7 months, HR 0.38, 95% CI 0.17–0.88) (P<0.05). Lastly, we compared OGTP patient survival outcomes according to patient age, race, or tumor site preference (Figure 5), indicating no significant differences in survival as a function of these variables (P>0.05).
Discussion
OGTP is a very rare form of adenocarcinoma that makes up just 1.4% of all pancreatic cancers, and it has been defined by the World Health Organization as a form of undifferentiated carcinoma exhibiting osteoclast-like giant cells [23]. While OGCs are detectable in a wide range of tissue types, the pancreas seems to be the most commonly affected tissue [24]. In the SEER patient cohort analyzed in the present study, we similarly observed higher rates of OGC incidence in the pancreas relative to other organs. At present, no large-scale clinical studies of OGTP have been conducted, with current understanding of this disease being largely restricted to case reports and review articles. As such, we conducted the present study using the SEER database in an effort to better elucidate the characteristics of OGTP.
Previous studies have suggested that OGTP most commonly manifests without any specific sex-bias in individuals between 60 and 70 years of age, although in some cases it has been reported in individuals as young as 32 years old or as old as 82 years [16,25]. We observed OGTP to be the most common in individuals between 50 and 70 years old (range: 35–90 years), consistent with previous studies, although unlike this previous report we observed a significantly higher rate of OGTP among females (70.2% versus 48.7%,
There also remained uncertainty in the literature with respect to the site preferences of OGTP in the pancreas, with PA typically being found to involve the head of the pancreas whereas OGTP most often involves the pancreatic body and tail [26]. As a result, OGTP patients most often initially present with abdominal distension, upper abdominal pain, weight loss, and a palpable mass, whereas PA patients more often first present with jaundice. In contrast to these reports, however, other studies have suggested that OGTP and PA exhibit comparable site preferences within the pancreas [27]. In line with this latter report, we did not detect any significant difference in tumor site preference when comparing OGTP and PA based upon the frequency of tumors involving the head of the pancreas (51.1% versus 52.7%,
Several studies [18,23,28–30] have reported OGTP to be associated lower rates of lymph node and distant metastasis relative to PA, and with better prognosis as a result. However, other reports have suggested the opposite, identifying poorer outcomes among OGTP patients relative to PA patients [21,22]. Given the limited number of OGTP cases reported to date, the prognosis of this condition thus remains unclear. Consistent with the former findings, in this analysis we found OGTP to typically present as an earlier-stage disease, with lower rates of lymph node and distant metastasis and a significantly longer mOS relative to PA (13 months versus 6 months). Importantly, among OGTP patients we found that those patients who had undergone surgical tumor resection had a significantly longer mOS than those patients who had not (33 months versus 5 months). This may be a consequence of the higher rates of early-stage OGTP leading to higher rates of surgical treatment and suggests that radical resection was the optimal approach to curing this form of undifferentiated carcinoma.
We observed no significant differences among OGTP patients as a function of patient age, race, or tumor site. Previous studies have not assessed these issues in depth. Our Kaplan-Meier survival curves suggested that there may be a trend towards a poorer patient prognosis among Caucasians and those with tumors in the pancreatic body and tail regions, however, raising the possibility that we only failed to observe any significant results in these analyses due to the limited sample size of the present study.
Luchini et al. [30] found that genetic alterations observed in OGTP were highly similar to known PA driver mutations, including KRAS, CDKN2A, TP53, and SMAD4 mutations, with no unique phenotypic markers specific to OGTP having been identified in their analysis. Another study detected the expression of PD-L1 on tumor cells in 63% of patient cases and found this expression to be linked with a poorer prognosis [31]. This therefore suggested that OGTP treatment might be benefit from anti-PD-1/PD-L1 monoclonal antibody therapy, although further research will be needed to validate this hypothesis and to identify other effective treatment programs.
On imaging, OGTP usually presents as a large cystic tumor with variable areas of hemorrhage and necrosis [20,25]. Hence, pancreatic cystic lesions such as pancreatic cystic tumors, pancreatic pseudocysts, and solid pancreatic tumors must be considered in the differential diagnosis with this tumor type [25,26]. However, a definitive diagnosis can only be established based upon histopathology or cytology. At the microscopic level, this rare tumor type presents with cytomorphological features including bizarre pleomorphic cells, bland OGCs, and mononuclear cells [26].
Although conducted using population-based data, there were still several limitations to the present study, including potential selection bias due to its retrospective nature. Furthermore, as the SEER database does not include any gene expression data, we were unable to compare patient outcomes based upon such parameters. In addition, this database did not contain sufficient detail regarding patient drug usage, surgical treatment, radiotherapy doses, or comorbidities, and as such, we were unable to explore the relationship between those variables and patient outcomes. Differences in the circumstances surrounding data input into this database also have the potential to introduce significant heterogeneity in these results.
Conclusions
In this population-based study, we investigated the clinical characteristics of OGTP patients, potentially enabling oncologists to better recognize and diagnose this tumor. We found that patients aged 50–70 years had the highest incidence of OGTP, with females being more frequently affected. We also found that rates of lymph node and distant metastasis were lower in OGTP patients relative to PA patients, and that these patients also had longer mOS and earlier stage disease. The definitive diagnosis of OGTP can only be determined by histopathology or cytology, and radical resection is an effective treatment.
Figures
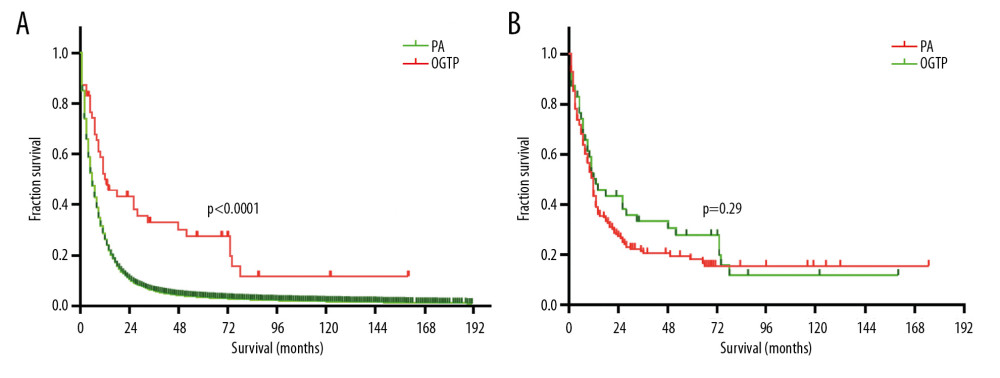 Figure 1. (A) Kaplan Meier curves suggested that OGTP patients had significantly longer survival than PA patients before PSM (13 months versus 6 months; HR 0.55, 95% CI 0.37–0.57, P<0.0001). (B) Kaplan-Meier survival curves demonstrating no significant extension in survival for OGTP patients relative to PA patients after PSM (13 months versus 12 months; HR 0.92, 95% CI 0.63–1.34, P>0.05). OGTP – osteoclast-like giant cell tumor of the pancreas; PA – pancreatic adenocarcinoma; PSM – propensity score matching; HR – hazard ratio; CI – confidence interval.
Figure 1. (A) Kaplan Meier curves suggested that OGTP patients had significantly longer survival than PA patients before PSM (13 months versus 6 months; HR 0.55, 95% CI 0.37–0.57, P<0.0001). (B) Kaplan-Meier survival curves demonstrating no significant extension in survival for OGTP patients relative to PA patients after PSM (13 months versus 12 months; HR 0.92, 95% CI 0.63–1.34, P>0.05). OGTP – osteoclast-like giant cell tumor of the pancreas; PA – pancreatic adenocarcinoma; PSM – propensity score matching; HR – hazard ratio; CI – confidence interval. 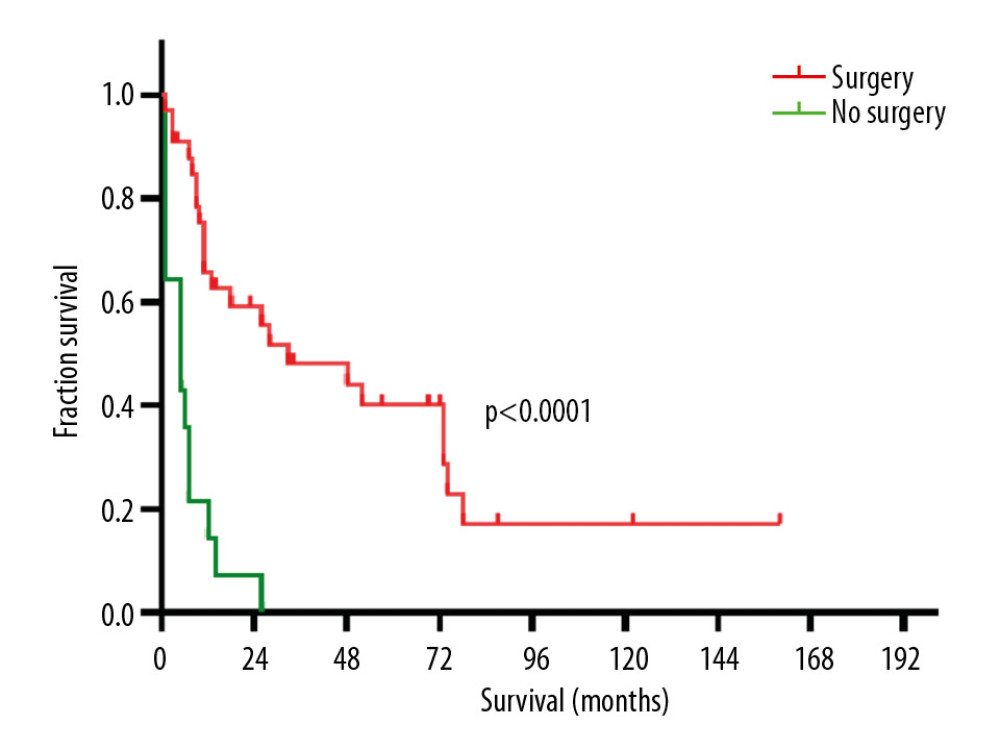 Figure 2. Kaplan-Meier curves indicated that patients with OGTP that underwent surgical resection had significantly longer survival than those who did not undergo resection (33 months versus 5 months; HR 0.07, 95% CI 0.03–0.21, P<0.0001) OGTP – osteoclast-like giant cell tumor of the pancreas; HR – hazard ratio; CI – confidence interval.
Figure 2. Kaplan-Meier curves indicated that patients with OGTP that underwent surgical resection had significantly longer survival than those who did not undergo resection (33 months versus 5 months; HR 0.07, 95% CI 0.03–0.21, P<0.0001) OGTP – osteoclast-like giant cell tumor of the pancreas; HR – hazard ratio; CI – confidence interval.  Figure 3. Kaplan-Meier curves indicated that the mOS of OGTP patients with localized, regional, and distant staging were 73, 26, and 7 months, respectively (P<0.01). mOS – median overall survival; OGTP – osteoclast-like giant cell tumor of the pancreas.
Figure 3. Kaplan-Meier curves indicated that the mOS of OGTP patients with localized, regional, and distant staging were 73, 26, and 7 months, respectively (P<0.01). mOS – median overall survival; OGTP – osteoclast-like giant cell tumor of the pancreas. 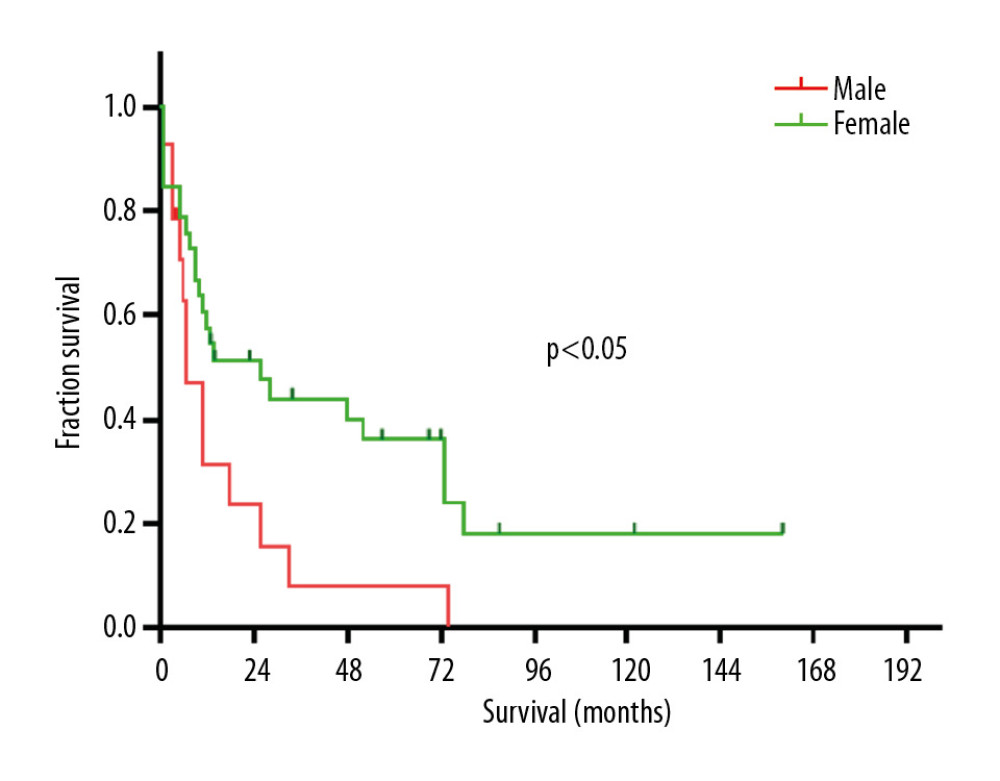 Figure 4. Kaplan-Meier curves indicated that females had a better prognosis than males (36 months versus 7 months, HR 0.38, 95% CI 0.17–0.88, P<0.05). Abbreviations: HR, hazard ratio; CI, confidence interval.
Figure 4. Kaplan-Meier curves indicated that females had a better prognosis than males (36 months versus 7 months, HR 0.38, 95% CI 0.17–0.88, P<0.05). Abbreviations: HR, hazard ratio; CI, confidence interval. 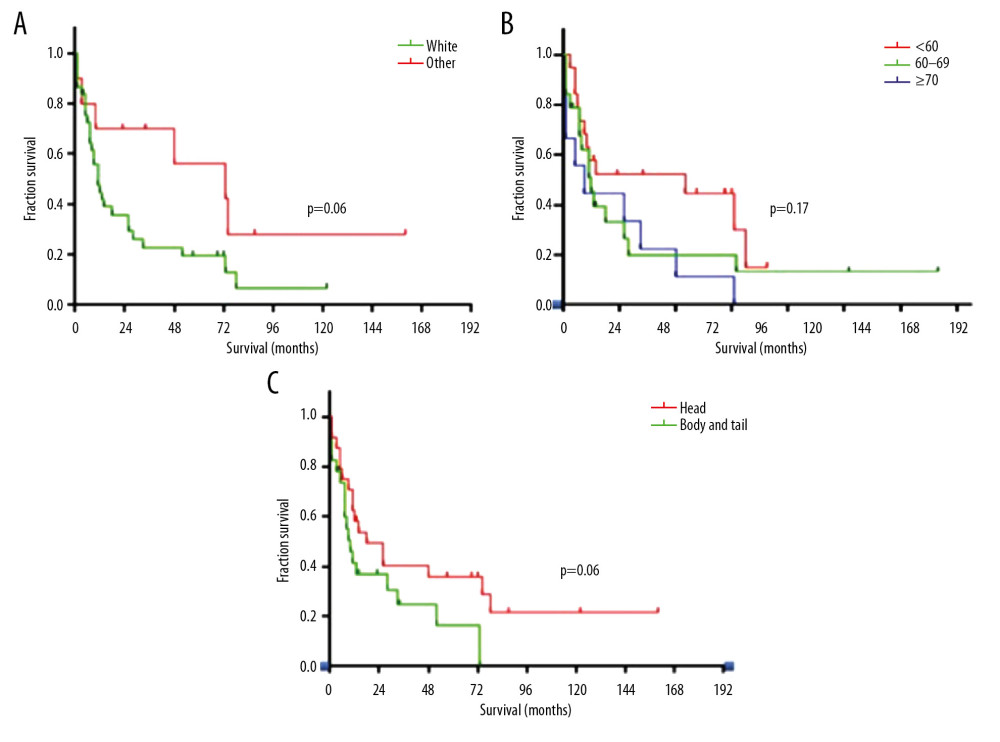 Figure 5. (A–C) Kaplan-Meier curves indicated that there was no significant difference in OGTP patient survival as a function of patient age, site, or race (P>0.05). OGTP – osteoclast-like giant cell tumor of the pancreas.
Figure 5. (A–C) Kaplan-Meier curves indicated that there was no significant difference in OGTP patient survival as a function of patient age, site, or race (P>0.05). OGTP – osteoclast-like giant cell tumor of the pancreas. References
1. Haeberle L, Esposito I, Pathology of pancreatic cancer: Transl Gastroenterol Hepatol, 2019; 4; 50
2. Rosai J, Carcinoma of pancreas simulating giant cell tumor of bone. Electron-microscopic evidence of its acinar cell origin: Cancer, 1968; 22(2); 333-44
3. Xu Z, Gu J, Zhang S, Leiomyosarcoma with osteoclast-like (LMS-OGC) giant cells the breast: A report of a rare case: Thorac Cancer, 2019; 10(10); 2054-56
4. Pena-Jaimes L, Gonzalez-Garcia I, Reguero-Callejas ME, Pleomorphic lobular carcinoma of the breast with osteoclast-like giant cells: A case report and review of the literature: Diagn Pathol, 2018; 13(1); 62
5. Priore SF, Schwartz LE, Epstein JI, An expanded immunohistochemical profile of osteoclast-rich undifferentiated carcinoma of the urinary tract: Mod Pathol, 2018; 31(6); 984-88
6. Chung IA, Petelin K, Pizem J, Luzar B, Cutaneous aneurysmal bone cyst: First report of a case and literature review: J Cutan Pathol, 2019; 46(11); 858-63
7. Shelly D, Gupta D, Mishra S, Bharadwaj R, Osteoclastic variant of anaplastic thyroid carcinoma: A case report of rare entity: J Cancer Res Ther, 2019; 15(3); 704-7
8. Liao X, Houldsworth J, Luo J, Polypoid undifferentiated carcinoma with osteoclast-like giant cells arising in the distal common bile duct: A rare case report: Anticancer Res, 2019; 39(1); 437-41
9. Lindholm KE, Kalhor N, Moran CA, Osteoclast-like giant cell-rich carcinomas of the lung: A clinicopathological, immunohistochemical, and molecular study of 3 cases: Hum Pathol, 2019; 85; 168-73
10. Cheng CH, Su B, Ding DC, Rare case of undifferentiated uterine sarcoma with neuroectodermal differentiation and osteoclast-like giant cells: Taiwan J Obstet Gynecol, 2018; 57(3); 442-46
11. Elhence P, Rao M, Goyal A, Osteoclast-like giant cell tumor of the parotid gland: report of a case diagnosed on fine-needle aspiration cytology with histological and immunohistochemical findings: Diagn Cytopathol, 2016; 44(6); 548-51
12. Saito R, Fujishima F, Nakamura Y, A case of pulmonary adenocarcinoma harboring osteoclast-like giant cells: Its evaluation by immunohistochemical and genetic analyses: Pathol Int, 2016; 66(4); 224-29
13. Righi S, Boffano P, Patetta R, Soft tissue giant cell tumor of low malignant potential with 3 localizations: Report of a case: Oral Surg Oral Med Oral Pathol Oral Radiol, 2014; 118(5); e135-38
14. Watanabe M, Miura H, Inoue H, Mixed osteoclastic/pleomorphic-type giant cell tumor of the pancreas with ductal adenocarcinoma: Histochemical and immunohistochemical study with review of the literature: Pancreas, 1997; 15(2); 201-8
15. Zhang L, Lee JM, Yoon JH, Huge and recurrent undifferentiated carcinoma with osteoclast-like giant cells of the pancreas: Quant Imaging Med Surg, 2018; 8(4); 457-60
16. Joo YE, Heo T, Park CH, A case of osteoclast-like giant cell tumor of the pancreas with ductal adenocarcinoma: Histopathological, immunohistochemical, ultrastructural and molecular biological studies: J Korean Med Sci, 2005; 20(3); 516-20
17. Jalloh SS, Giant cell tumour (osteoclastoma) of the pancreas – an epithelial tumour probably of pancreatic acinar origin: J Clin Pathol, 1983; 36(10); 1171-75
18. Lewandrowski KB, Weston L, Dickersin GR, Giant cell tumor of the pancreas of mixed osteoclastic and pleomorphic cell type: Evidence for a histogenetic relationship and mesenchymal differentiation: Hum Pathol, 1990; 21(11); 1184-87
19. Goldberg RD, Michelassi F, Montag AG, Osteoclast-like giant cell tumor of the pancreas: Immunophenotypic similarity to giant cell tumor of bone: Hum Pathol, 1991; 22(6); 618-22
20. Wada T, Itano O, Oshima G, A male case of an undifferentiated carcinoma with osteoclast-like giant cells originating in an indeterminate mucin-producing cystic neoplasm of the pancreas. A case report and review of the literature: World J Surg Oncol, 2011; 9; 100
21. Singhal A, Shrago SS, Li SF, Giant cell tumor of the pancreas: A pathological diagnosis with poor prognosis: Hepatobiliary Pancreat Dis Int, 2010; 9(4); 433-37
22. Chiarelli M, Guttadauro A, Gerosa M, An indeterminate mucin-producing cystic neoplasm containing an undifferentiated carcinoma with osteoclast-like giant cells: A case report of a rare association of pancreatic tumors: BMC Gastroenterol, 2015; 15; 161
23. Muraki T, Reid MD, Basturk O, Undifferentiated carcinoma with osteoclastic giant cells of the pancreas: Clinicopathologic analysis of 38 cases highlights a more protracted clinical course than currently appreciated: Am J Surg Pathol, 2016; 40(9); 1203-16
24. Nai GA, Amico E, Gimenez VR, Guilmar M, Osteoclast-like giant cell tumor of the pancreas associated with mucus-secreting adenocarcinoma. Case report and discussion of the histogenesis: Pancreatology, 2005; 5(2–3); 279-84
25. Sivanandham S, Subashchandrabose P, Muthusamy KR, FNA diagnosis of osteoclast-like giant cell tumor of the pancreas: J Cytol, 2012; 29(4); 270-72
26. Yepuri N, Pruekprasert N, Ramani N, Osteoclast-like giant cell tumor of the pancreas-an unusual presentation in a patient with large mantle cell lymphoma: J Surg Case Rep, 2018; 2018(12) rjy341
27. Reid MD, Muraki T, HooKim K, Cytologic features and clinical implications of undifferentiated carcinoma with osteoclastic giant cells of the pancreas: An analysis of 15 cases: Cancer Cytopathol, 2017; 125(7); 563-75
28. Jeffrey I, Crow J, Ellis BW, Osteoclast-type giant cell tumour of the pancreas: J Clin Pathol, 1983; 36(10); 1165-70
29. Alguacil-Garcia A, Weiland LH, The histologic spectrum, prognosis, and histogenesis of the sarcomatoid carcinoma of the pancreas: Cancer, 1977; 39(3); 1181-89
30. Luchini C, Pea A, Lionheart G, Pancreatic undifferentiated carcinoma with osteoclast-like giant cells is genetically similar to, but clinically distinct from, conventional ductal adenocarcinoma: J Pathol, 2017; 243(2); 148-54
31. Luchini C, Cros J, Pea A, PD-1, PD-L1, and CD163 in pancreatic undifferentiated carcinoma with osteoclast-like giant cells: Expression patterns and clinical implications: Hum Pathol, 2018; 81; 157-65
Figures
 Figure 1. (A) Kaplan Meier curves suggested that OGTP patients had significantly longer survival than PA patients before PSM (13 months versus 6 months; HR 0.55, 95% CI 0.37–0.57, P<0.0001). (B) Kaplan-Meier survival curves demonstrating no significant extension in survival for OGTP patients relative to PA patients after PSM (13 months versus 12 months; HR 0.92, 95% CI 0.63–1.34, P>0.05). OGTP – osteoclast-like giant cell tumor of the pancreas; PA – pancreatic adenocarcinoma; PSM – propensity score matching; HR – hazard ratio; CI – confidence interval.
Figure 1. (A) Kaplan Meier curves suggested that OGTP patients had significantly longer survival than PA patients before PSM (13 months versus 6 months; HR 0.55, 95% CI 0.37–0.57, P<0.0001). (B) Kaplan-Meier survival curves demonstrating no significant extension in survival for OGTP patients relative to PA patients after PSM (13 months versus 12 months; HR 0.92, 95% CI 0.63–1.34, P>0.05). OGTP – osteoclast-like giant cell tumor of the pancreas; PA – pancreatic adenocarcinoma; PSM – propensity score matching; HR – hazard ratio; CI – confidence interval. Figure 2. Kaplan-Meier curves indicated that patients with OGTP that underwent surgical resection had significantly longer survival than those who did not undergo resection (33 months versus 5 months; HR 0.07, 95% CI 0.03–0.21, P<0.0001) OGTP – osteoclast-like giant cell tumor of the pancreas; HR – hazard ratio; CI – confidence interval.
Figure 2. Kaplan-Meier curves indicated that patients with OGTP that underwent surgical resection had significantly longer survival than those who did not undergo resection (33 months versus 5 months; HR 0.07, 95% CI 0.03–0.21, P<0.0001) OGTP – osteoclast-like giant cell tumor of the pancreas; HR – hazard ratio; CI – confidence interval. Figure 3. Kaplan-Meier curves indicated that the mOS of OGTP patients with localized, regional, and distant staging were 73, 26, and 7 months, respectively (P<0.01). mOS – median overall survival; OGTP – osteoclast-like giant cell tumor of the pancreas.
Figure 3. Kaplan-Meier curves indicated that the mOS of OGTP patients with localized, regional, and distant staging were 73, 26, and 7 months, respectively (P<0.01). mOS – median overall survival; OGTP – osteoclast-like giant cell tumor of the pancreas. Figure 4. Kaplan-Meier curves indicated that females had a better prognosis than males (36 months versus 7 months, HR 0.38, 95% CI 0.17–0.88, P<0.05). Abbreviations: HR, hazard ratio; CI, confidence interval.
Figure 4. Kaplan-Meier curves indicated that females had a better prognosis than males (36 months versus 7 months, HR 0.38, 95% CI 0.17–0.88, P<0.05). Abbreviations: HR, hazard ratio; CI, confidence interval. Figure 5. (A–C) Kaplan-Meier curves indicated that there was no significant difference in OGTP patient survival as a function of patient age, site, or race (P>0.05). OGTP – osteoclast-like giant cell tumor of the pancreas.
Figure 5. (A–C) Kaplan-Meier curves indicated that there was no significant difference in OGTP patient survival as a function of patient age, site, or race (P>0.05). OGTP – osteoclast-like giant cell tumor of the pancreas. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387