11 September 2020: Clinical Research
Decreased Synovial Fluid Biomarkers Levels Are Associated with Rehabilitation of Function and Pain in Rotator Cuff Tear Patients Following Electroacupuncture Therapy
Jian Guan1ABDEF, Wei-Qiang Geng2AF, Yao Li1A, Guang-Yuan Liu1B, Luo-Bin Ding1B, You-Jie Liu3C, Wei Xue4C, Huajun Wang3E*, Xiao-Fei Zheng3EDOI: 10.12659/MSM.923240
Med Sci Monit 2020; 26:e923240
Abstract
BACKGROUND: The aim of this study was to assess inflammatory cytokines levels in synovial fluid (SF) before and after electroacupuncture (EA) treatment and to explore whether these biomarkers are associated with function of rotator cuff tear (RCT) patients.
MATERIAL AND METHODS: We recruited 54 patients with RCT and separated them into an EA group and a control group. The SF biomarker levels were detected at baseline and at 6-week and 6-month follow-up. The symptomatic severity was evaluated by visual analog scale (VAS), Constant-Murley score, and American Shoulder and Elbow Surgeons score (ASES). We also investigated the correlation between symptomatic severity and biomarker levels in SF of the shoulder joint.
RESULTS: The reductions in VAS and improved functional score (ASES and Constant-Murley score) were significantly different between the 2 groups, and SF biomarker concentrations were significantly lower in the EA group. IL-1β levels were significantly negatively correlated with Constant-Murley score (r=–0.73, P=0.04) and ASES score (r=–0.59, P<0.001) and positively correlated with VAS scores (r=0.81, P=0.004). IL-6 levels were significantly negatively correlated with Constant-Murley score (r=–0.67, P=0.03) and positively correlated with VAS score (r=0.7, P=0.01). MMP-1 levels were significantly negatively correlated with ASES score (r=–0.57, P<0.001).
CONCLUSIONS: The biomarkers in SF were directly associated with shoulder pain and shoulder function in rotator cuff tear. EA, as a safe and effective conservative therapy, obviously decreased the level of inflammatory cytokines in RCT patients, accompanied by a reduction in shoulder pain and improved function.
Keywords: Biological Markers, Electroacupuncture, rotator cuff, Synovial Fluid, Cytokines, Recovery of Function, Rotator Cuff Injuries
Background
Rotator cuff tear (RCT) is one of the most common musculoskeletal disorders, representing almost 70% of shoulder injuries and affecting more than 17 million persons in the USA [1–3]. Rotator cuff tear mainly impairs active motion and decreases muscle strength, which results in low quality of life and impaired work performance [4,5]. Etiologically, extrinsic factors like impingement and traumatic force cause rotator cuff injuries. Moreover, intrinsic factors play an equally important role in rotator cuff tear [6,7]. Changes in biomarkers of synovial fluid (SF) have been proposed as a contributing intrinsic factor in pathogenesis of the disease [6,7]. There have been many studies on biomarkers of SF in knee disorders and they demonstrate the close correlation between biomarker levels in SF and the pathological conditions of osteoarthritis (OA) [9–13]. However, there have been few studies regarding the correlation between biomarkers in SF and severity of shoulder diseases. Some of these studies indicate the molecule levels in SF are associated with shoulder pain and impaired function in patients with shoulder diseases [14–17]. Studies reported that cytokine levels varied significantly according to the severity of rotator cuff tears [15–17]. Interleukins play an important role in shoulder pain and impaired shoulder range of motion [15], but other studies found that specific interleukins were not correlated with pain in RCT [17,18]. Some researchers reported that upregulation of metalloproteinases levels is common in RCT [18–21]; they found that matrix metalloproteinases levels increased with rotator cuff tear severity and might be the most important biomarkers distinguishing between non-rotator cuff tear groups and rotator cuff tear patients [21–24]. Although previous studies tried to determine the intrinsic mechanism of RCT, their findings were not robust [16–20]. All these previous studies were single-time-point investigations with small samples, and lack of control groups also contributed to their limited strength [15–21]. More reliable conclusions need to be provided by controlled trials with longer follow-up periods.
Electroacupuncture (EA) [25] has been proved to be an effective therapy for managing musculoskeletal disorders of the extremities. A randomized controlled trial suggested that EA accelerated bone mineralization radiographically and biomechanically during the healing stage [26]. The use of EA to treat Achilles tendon rupture increased the concentration of growth factors and enhanced the physical strength of the healing tendon compared with a control group [27]. Accordingly, we hypothesized that EA is an effective method for management of RCT.
The effects of changes in inflammatory cytokine levels on RCT progression have been unclear. The present study aimed to: 1) measure the levels of inflammatory cytokines in SF before and after EA treatment and to determine whether these biomarkers are associated with RCT function, and 2) evaluate the effectiveness and safety of EA for treating rotator cuff tear.
Material and Methods
STUDY DESIGN AND PARTICIPANTS:
After Institutional Review Board approval (No. 2017-003), we retrospectively recruited out-patients who were diagnosed as having a full-thickness rotator cuff tear by magnetic resonance imaging (MRI) and finished the follow-up from January 2017 to January 2019 in the Third Hospital of Shijiazhuang. For all patients, we performed standard detailed history collecting, physical examination, and MRI assessment. We recorded patient’s characteristics that could affect the study results, such as age, sex, duration of injury, size of lesion, diabetes, smoking, body mass index (BMI), workload, and tear on dominant side. Inclusion criteria were: 1) patients diagnosed as having an MRI-proven full-thickness rotator cuff tear; 2) patients who received conservative treatment; 3) with at least 6 months of follow-up after conservative treatment; 4) tear size is small or medium (DeOrio and Cofield classification [28]). According to the DeOrio and Cofield classification, rotator cuff tear size is classified into 4 grades (small to massive) according to the oblique-sagittal views: for small, the greatest dimension is <1 cm; for medium, 1–3 cm; for large, 3–5 cm; and for massive, >5 cm. Tear size was evaluated and recorded based on MRI at first-time consultancy by the same senior surgeon. The following exclusion criteria were used: 1) previous operative history on the same shoulder; 2) with other shoulder diseases such as frozen shoulder, arthritis, or fracture; 3) received any recent pharmacotherapy or shoulder injections with non-steroidal anti-inflammatory drugs or pure pain-killers during the last month prior to beginning the study; and 4) shoulder pain caused by other diagnosed shoulder disorders. We included patients into the study based on inclusion and exclusion criteria strictly applied by 2 experienced experts; when there was disagreement, a third expert was consulted before a final judgment was made. Included patients were separated into the EA group and the control group according the type of conservative treatment applied. To establish the best possible matching of patients in the EA group and control group, propensity score analysis was used with patients matched by factors thought to influence the outcome, including age, sex, BMI, workload, size of lesion, symptom duration, diabetes, smoking, and tear on dominant side. Patients who were unmatched by the propensity score analysis were excluded. We collected all baseline demographic data (Table 1).
REHABILITATION AND EA TREATMENT PROCEDURE:
All participants were treated with conservative treatment. Patients in the control group received rehabilitation only, and patients in the EA group received rehabilitation plus EA treatment. The rehabilitation program included range of motion, flexibility, and strengthening, and was derived from a prospective multicenter study that demonstrated physical therapy was effective in curing atraumatic full-thickness rotator cuff tears [29]. Specifically, range of motion consists of active range of motion, active-assisted motion, active training of scapular muscle, and postural exercises; daily flexibility consists of anterior and posterior shoulder stretching. Rehabilitation was performed 3 times per week for 6 consecutive weeks.
All patients received rehabilitation and the EA group received the same rehabilitation plus EA stimulation. The detailed procedures of EA are described in the STRICTA checklist [30]. All patients in the EA group received an identical procedure. Low-frequency EA treatment was performed 2 times per week with a stimulation frequency of 10 Hz and a current intensity at pulse duration of 200 us for 30 minutes. EA treatment was practiced for 12 sessions over a 6-week period by a licensed senior physiotherapist. In each session, patients in the EA group were required to lie in a lateral decubitus position. The skin in the treatment area was prepared with isopropyl alcohol, as were the sterile single-use needles (40 mm in length, 0.25 mm in diameter, Huatuo®, Suzhou Medical Co., Suzhou, China). After disinfection, patients received acupuncture at 3 local points (LI15 Jian yu, SI9 Jian zhen, and EX-UE12 Jian qian) located around the deltoid muscle and at 1 distal healthy side point (S38 Tiao kou) by inserting the needle intramuscularly to a depth of 20 mm [31]. The locations of each acupoint conformed to the standard defined by the World Health Organization [32]. All needles in the points were twisted approximately 180° clockwise and counterclockwise at increasing frequency from 0.3 to 1 Hz to induce the de qi sensation (a feeling of warmth, tingling, and paresthesia) for 30 seconds. When the patients felt the sensation of de qi in the acupuncture region, the needle handles were attached to the paired electrodes from the SDZ-V electroacupuncture apparatus (Huatuo®, Suzhou Medical Co., Suzhou, China) to start stimulating (LI15 and SI9 were connected to a pair of electrodes, and EX-UE12 and S38 were connected to a pair of electrodes), eliciting light muscular twitching. The rehabilitation program and EA treatment were practiced by the same licensed physiotherapist.
CLINICAL MEASUREMENTS AND FOLLOW-UP:
American Shoulder and Elbow Surgeons (ASES) score, Constant-Murley score, and visual analog scale (VAS) score were obtained by the senior physiotherapist at baseline, 6 weeks, and 6 months after treatment.
The American Shoulder and Elbow Surgeons (ASES) score is a commonly-used system that is appropriate for evaluating the severity of rotator cuff tears. The system focuses equally on self-reported patient assessment of pain (50%) and on self-reported ability to perform activities of daily living (50%). The Constant-Murley scoring is another commonly applied evaluation system that consists of 4 parts: self-reported evaluation of pain (15%), activities of daily living (20%), range of shoulder joint motion (40%), and muscle strength (25%). In both scoring systems, a higher score indicates less pain and better physical function, while lower scores represent more intense pain and worse physical function. The visual analog scale (VAS) is a numerical scale with a maximum of 10 points and is widely used to measure pain intensity. The patients were instructed to subjectively indicate the extent of pain, with higher values indicating more severe pain.
LABORATORY EXAMINATION OF SF:
We collected and stored SF samples at each visit. Treated shoulder joint SF samples were subjected to ultrasound (US)-guided percutaneous aspiration before rehabilitation with EA treatment and at each follow-up. All samples were immediately separated by centrifugation. After debris was removed from samples, and the remaining SF was stored at −80°C until needed for enzyme-linked immunosorbent assay (ELISA). Collection, storage, and measurement were performed using the same protocol. Interleukin (IL) −1β, IL-6, matrix metalloproteinase (MMP)-1, and MMP-13 levels were calculated in SF using ELISA kits.
STATISTICAL ANALYSIS:
All outcome measures were evaluated at baseline before treatment, at 6-week follow-up, and at 6-month follow-up. Data were assessed for normal distribution using the Shapiro-Wilk test. Descriptive data are presented as mean±standard deviation (SD). At baseline, chi-squared tests were performed for categorical variables and the unpaired
Result
DEMOGRAPHIC DATA:
Finally, 54 patients (27 in the EA group and 27 in the control group) with rotator cuff tears were matched and recruited into our study. The mean age was 53.4±6.3 years in the EA group and 54.5±7.7 years in the control group. The mean duration of symptoms was 9±4 months in the EA group and 10±5 months in the control group. The mean lesion size was 2.1±0.6 cm in the EA group and 1.9±0.7cm in the control group. Patients in the EA group were classified into 2 stages based on DeOrio and Cofield classification: 14 cases were classified as small lesions and 13 cases were classified as medium lesions. In the control group, DeOrio and Cofield classification showed 12 cases had small lesions and 15 cases had medium lesions. The demographic data of the EA group and control group are documented in Table 1. There were no significant differences in age, sex, BMI, workload, size of lesion, symptoms duration, dominant side, diabetes, smoking status, or DeOrio and Cofield classification between the 2 groups.
PRE- AND POST-TREATMENT CLINICAL MEASUREMENTS:
Table 2 documents the mean baseline and post-treatment scores. There were no significant differences in ASES, Constant-Murley score, or VAS prior to treatment between the 2 groups. After treatment, the EA group showed a greater improvement in clinical measurements and pain compared with the control group, evaluated as differences in ASES, Constant-Murley score, and VAS score after 6 weeks of intervention. ASES and VAS scores changed by 126.9% and 62.5%, respectively, in the EA group contrasted with 61% and 35.3%, respectively, in the control group. Furthermore, the within-group repeated-measures analysis showed significant difference in ASES, Constant-Murley, and VAS scores among the 2 groups at baseline, after 6 weeks, and after 6 months of conservative treatment. The effects were maintained for up to 6 months after start of treatment.
PRE- AND POST-TREATMENT SF BIOMARKERS MEASUREMENTS:
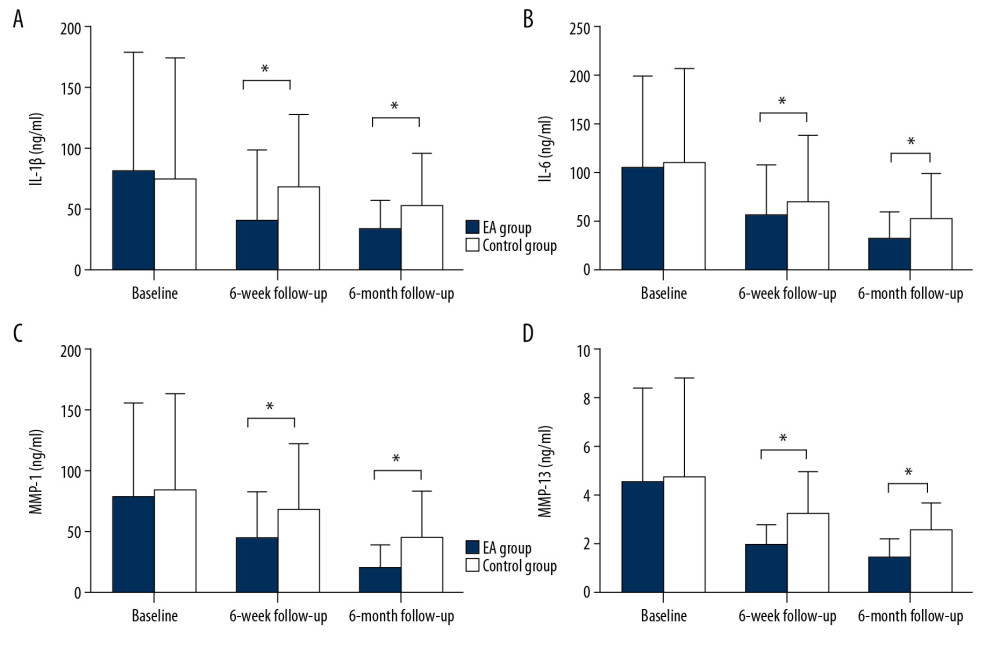
Figure 1 shows the average biomarker levels measured at different time-points in the 2 groups. There were no significant differences between the 2 groups before treatment, but we found significant differences between the EA group and control group following treatment, with biomarker levels being lower at 6-week and 6-month follow-ups.
For IL-1β, the average concentration in the EA group was 40.7±31.3 ng/ml at 6-week follow-up and 34.5±22.8 ng/ml at 6-month follow-up. The average concentration in the control group was 68.1±52.4 ng/ml at 6-week follow-up and 52.9±46.1 ng/ml at 6-month follow-up (Figure 1A).
The mean concentrations of IL-6 were significantly higher in the control group (70.4±59.3 ng/ml) than in the EA group (56.2±52.3 ng/ml) at 6-week follow-up, and significant differences remained at 6-month follow-up (32.1±27.5 ng/ml vs. 53.2±42.7 ng/ml) (Figure 1B).
The average concentrations of MMP-1 were significantly different within the EA group and control group at 6-week follow-up (45.6±37.9 ng/ml vs. 68.4±55.3 ng/ml). Subsequently, the results at 6-month follow-up indicated significant differences within the 2 groups (21.2±18.1 ng/ml vs. 44.8±37.7 ng/ml) (Figure 1C);
For MMP-13, the average concentrations in the EA group were 1.9±0.9 ng/ml at 6-week follow-up and 1.4±0.8 ng/ml at 6-month follow-up. The average of concentrations in the control group were 3.2±1.8 ng/ml at 6-week follow-up and 2.5±1.2 ng/ml at 6-month follow-up (Figure 1D).
CORRELATION OF SF INFLAMMATORY PARAMETERS WITH FUNCTIONAL OUTCOMES:
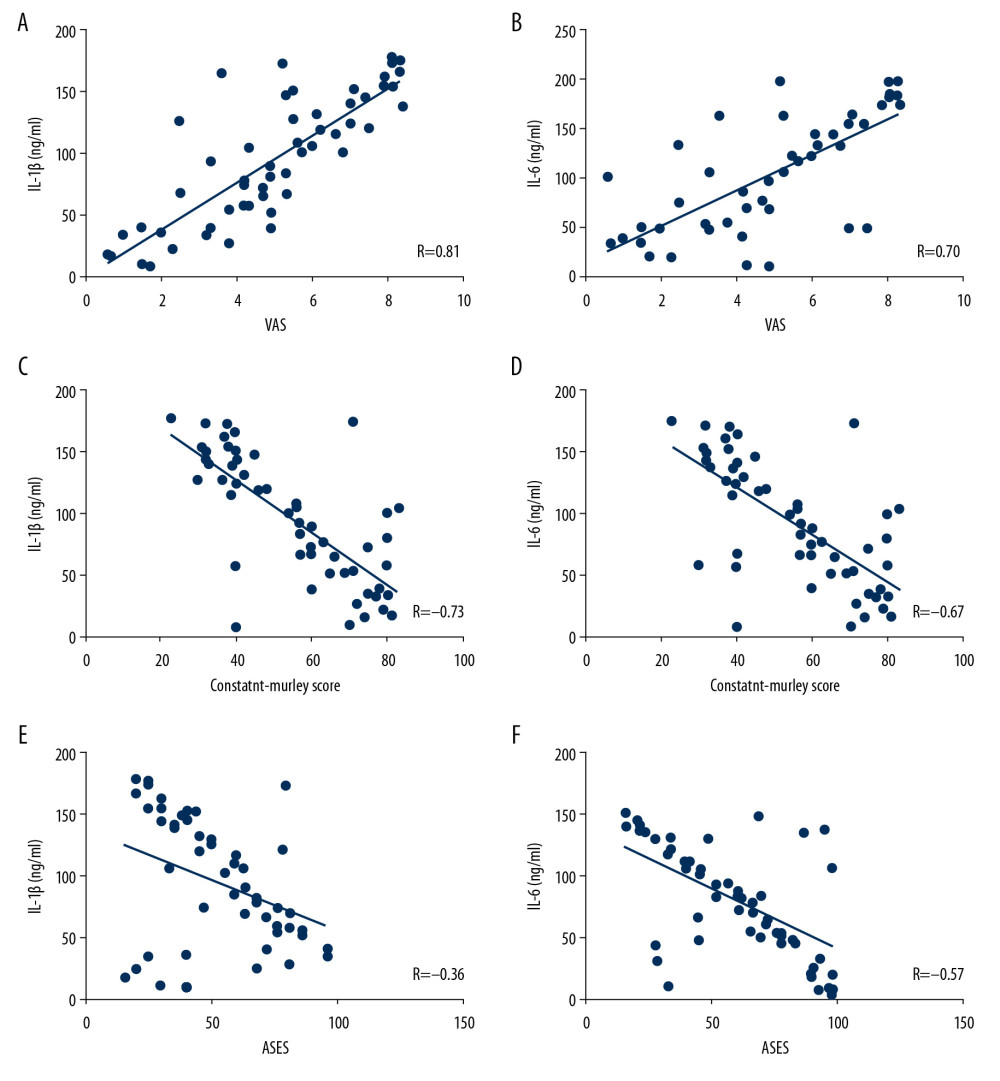
We enrolled all functional outcomes and SF biomarkers throughout the study to analyze the correlations between these data. SF IL-1β levels were significantly negatively correlated with Constant-Murley score (r=−0.73, P=0.04) (Figure 2A; Table 3), ASES score (r=−0.59, P<0.001) (Figure 2B) and positively correlated with VAS scores (r=0.81, P=0.004) (Figure 2C). SF IL-6 levels were significantly negatively correlated with Constant-Murley score (r=−0.67, P=0.03) (Figure 2D) and positively correlated with VAS score (r=0.7, P=0.01) (Figure 2E). SF MMP-1 levels were significantly negatively correlated with ASES score (r=−0.57, P<0.001) (Figure 2F).
ADVERSE REACTIONS OR COMPLICATIONS:
There were no serious adverse reactions or complications resulting from the treatment in the 2 groups. Only a few minor adverse events were reported, including temporary ecchymosis due to small-vessel damage (n=2) and transient dizziness (n=3).
Discussion
By 6-month follow-up, we found for the first time that biomarker levels of SF were decreased in patients with rotator cuff tears following conservative treatment. With further research, more significantly, the results showed that SF IL-1β was correlated with functional outcomes and pain intensity in follow-up. These findings suggested that biomarkers in SF were potentially helpful for predicting or curing RCT. In addition, the EA group showed significantly better measured outcomes than the control group for nearly all outcomes (Constant-Murley score, ASES, and VAS) at each follow-up visit, without serious adverse complications. Our results strongly suggest that EA treatment is an effective complementary method for treating rotator cuff tears.
The pathogenesis of rotator cuff tear is still unclear, but some studies have implicated inflammation caused by interleukins [14–16]. A previous study reported that IL-1β and IL-6 are expressed at higher levels in the shoulder joint synovium and subacromial bursa and are associated with more severe pain behaviors in a rat unstabilized rotator cuff defect model [33]. Clinical studies by Siu et al. [34] and Nakama et al. [35] also found that IL-1β and IL-6 levels are significantly correlated with the degree of pain and range of motion. However, other studies found no significant correlations between mean IL-1β and IL-6 levels and symptoms [17,36]. Chaudhury even reported that the gene expression of IL-1 was significantly downregulated in the RCT group [37]. In our opinion, the conflicting results are due to use of different ethnic groups and small sample sizes, especially in cross-sectional studies. For this reason, we designed this long-term follow-up clinical research to better understand how interleukins are involved in the RCT recovery process. Our results demonstrated that IL-1β and IL-6 levels were gradually and significantly decreased at each follow-up time-point and were strongly associated with pain relief and shoulder joint function rehabilitation at 6-month follow-up. Of particular importance, the concentration of IL-1 in synovial fluid was negatively correlated with Constant-Murley score and ASES (r=−0.59, P<0.001; r=−0.73, P=0.04), and IL-1β in SF was positively correlated with VAS (r=0.81, P=0.004). Our results suggest that IL-1β is associated with inflammation-related pain intensity and impaired function in patients with rotator cuff tears.
In addition to interleukins, matrix metalloproteinases are known to participate in degrading various kinds of extracellular matrix proteins, such as collagens, proteoglycans, fibronectin, and many others. In musculoskeletal medicine, MMPs are significantly involved in degeneration of the tendon and intervertebral disc, and loosening of hip joint replacements [19]. In previous studies, MMP-1 and MMP-13 were found to be capable of cleaving almost all kinds of collagens, which indicates that MMPs contribute to deterioration of shoulder joint function [20,21]. Previous studies on this topic have several limitations, including very small sample sizes, single-time-point data, and obviously unbalanced sex ratios between the experimental group and control group, which reduce the quality of evidence. Controlled clinical trials with sufficiently long follow-up periods are needed to determine the correlation between MMPs and RCT. Furthermore, several controlled studies of shoulder synovial fluid reported nonsignificant differences in MMP-1, MMP-3, and MMP-13 levels in patients with and without RCT [22,23]. The discrepancies among these studies suggest that the role of MMPs in the pathogenesis of RCT requires further research. At 6-month follow-up, we found that MMP-1 was markedly correlated with and shoulder joint motion after treatment, and that the concentration of MMP-1 in SF was negatively associated with ASES (r=−0.57,
EA has been applied world-wide to cure a variety of diseases and has achieved good results, and increasing evidence shows that EA accelerates the healing process and relieves symptoms in musculoskeletal conditions. In previous studies, EA has achieved good therapeutic effects in fractures, osteoporosis, and tendon injuries [38–40]. For the majority of RCT patients, conservative treatment is preferred due to its safety and effectiveness. The concept of Enhanced Recovery After Surgery (ERAS) has become widely used to promote recovery from diseases, and EA treatment may be an important alternative method for managing RCT. Based on the effectiveness of EA in treating musculoskeletal diseases, we used EA to treat RCT and achieved good results. In our study, there were no serious adverse events resulting from treatment in the EA group; only 2 patients had small-vessel damage and 3 patients had transient dizziness. By 6-week follow-up, both groups had significant improvement of all outcomes (Constant-Murley score, ASES, and VAS) compared to baseline, and the EA group had better treatment effects than in the control group treated by the rehabilitation program alone. Thus, these results consistently suggest that electroacupuncture had a clinically meaningful benefit in improving symptoms and relieving pain in patients with RCT. A previous study showed the positive effects of EA in eliminating inflammation, activating growth factors, and regulating bone metabolism [41]. Wu et al. found that IL-1β and IL-6 levels in SF and cartilage in the EA-treated group were significantly decreased in the OA rat model [42]. Furthermore, Bao et al. found that acupuncture stimulation performs better than medication with Diclofenac cream in reducing the expression of MMP-1 in cartilage [43], and Zhou et al. reported that EA downregulated the expression of MMP-13 [44]. However, there has been little research on the ability of EA to regulate SF biomarkers in tendon tissue. In the present study, we analyzed changes in the levels of biomarkers in SF continuously before and after treatment of EA. The results showed that SF IL-1β, IL-6, and MMP-13 levels in the EA group were lower than in the control group at follow-up. These suggest that anti-inflammation plays a critical role in the mechanism of EA stimulation treatment for RCT. Therefore, we suggest that EA treatment is effective in relieving pain and improving function by modulating levels of cytokines and matrix metalloproteinases.
The present study has several limitations. First, it was difficult to obtain a sufficient volume of joint fluid from every patient at every follow-up time-point. Sometimes, we aspirated too little fluid to test, and this sometimes occurred before and after treatment, but we were fortunately able to collect enough data to complete the analysis of the results. Second, this was a single-center study with a relatively small number of patients, leading to limited statistical power, but the results were clear and encouraging. Multicenter or longitudinal studies with larger populations are needed to verify our results. Third, we did not perform post-treatment MRI for every patient and thus were unable to reach a final conclusion about rotator cuff recovery. Last, although we used inclusion and exclusion criteria, this study was retrospective and data and samples were collected before setting up the experiment.
Conclusions
In conclusion, the results of the present study suggest that biomarkers in SF are directly associated with shoulder pain and shoulder function in patients with rotator cuff tear. SF biomarkers may be useful as predictors of RCT patient outcomes. Additionally, EA obviously decreased the level of inflammatory cytokines in RCT, accompanied by a reduction in shoulder pain and improved function. This evidence shows that electroacupuncture is a safe and effective conservative therapy, but more research is needed to provide more detailed information on the effect of EA on RCT healing.
References
1. Mather RC, Koenig L, Acevedo D, The societal and economic value of rotator cuff repair: J Bone Joint Surg Am, 2013; 95; 1993-2000
2. Deprés-Tremblay G, Chevrier A, Snow M, Rotator cuff repair: A review of surgical techniques, animal models, and new technologies under development: J Shoulder Elbow Surg, 2016; 25(12); 2078-85
3. McElvany MD, McGoldrick E, Gee AO, Rotator cuff repair: Published evidence on factors associated with repair integrity and clinical outcome: Am J Sports Med, 2015; 43(2); 491-500
4. Jain NB, Ayers GD, Fan R, Comparative effectiveness of operative versus nonoperative treatment for rotator cuff tears: A propensity score analysis from the ROW Cohort: Am J Sports Med, 2019; 47(13); 3065-72
5. Lewis J, Rotator cuff related shoulder pain: Assessment, management and uncertainties: Man Ther, 2016; 23; 57-68
6. Abrams GD, Luria A, Carr RA, Association of synovial inflammation and inflammatory mediators with glenohumeral rotator cuff pathology: J Shoulder Elbow Surg, 2016; 25(6); 989-97
7. Bedi A, Maak T, Walsh C, Cytokines in rotator cuff degeneration and repair: J Shoulder Elbow Surg, 2012; 21; 218-27
9. Mobasheri A, Bay-Jensen AC, van Spil WE, Osteoarthritis Year in Review 2016: Biomarkers (biochemical markers): Osteoarthritis Cartilage, 2017; 25(2); 199-208
10. Emery CA, Whittaker JL, Mahmoudian A, Establishing outcome measures in early knee osteoarthritis: Nat Rev Rheumatol, 2019; 15(7); 438-48
11. Li H, Wang D, Yuan Y, New insights on the MMP-13 regulatory network in the pathogenesis of early osteoarthritis: Arthritis Res Ther, 2017; 19; 248
12. Gomez-Urena EO, Tande AJ, Osmon DR, Diagnosis of prosthetic joint infection: Cultures, biomarker and criteria: Infect Dis Clin North Am, 2017; 31; 219-35
13. Radojcic MR, Thudium CS, Henriksen K, Biomarker of extracellular matrix remodelling C1M and proinflammatory cytokine interleukin 6 are related to synovitis and pain in end-stage knee osteoarthritis patients: Pain, 2017; 158; 1254-63
14. Osawa T, Shinozaki T, Takagishi K, Multivariate analysis of biochemical markers in synovial fluid from the shoulder joint for diagnosis of rotator cuff tears: Rheumatol Int, 2005; 25; 436-41
15. Okamura K, Kobayashi T, Yamamoto A, Shoulder pain and intra-articular interleukin-8 levels in patients with rotator cuff tears: Int J Rheum Dis, 2017; 20; 177-81
16. Ko JY, Wang FS, Huang HY, Increased IL-1beta expression and myofibroblast recruitment in subacromial bursa is associated with rotator cuff lesions with shoulder stiffness: J Orthop Res, 2008; 26; 1090-97
17. Shih CA, Wu KC, Shao CJ, Synovial fluid biomarkers: Association with chronic rotator cuff tear severity and pain: J Shoulder Elbow Surg, 2018; 27; 545-52
18. Tajana MS, Murena L, Valli F, Correlations between biochemical markers in the synovial fluid and severity of rotator cuff disease: Chir Organi Mov, 2009; 93(Suppl 1); S41-48
19. Garofalo R, Cesari E, Vinci E, Role of metalloproteinases in rotator cuff tear: Sports Med Arthrosc Rev, 2011; 19; 207-12
20. Lakemeier S, Braun J, Efe T, Expression of matrix metalloproteinases 1, 3, and 9 in differing extents of tendon retraction in the torn rotator cuff: Knee Surgery Sports Traumatol Arthrosc, 2011; 19; 1760-65
21. Bramono DS, Richmond JC, Weitzel PP, Matrix metalloproteinases and their clinical applications in orthopaedics: Clin Orthop Relat Res, 2004; 428; 272-85
22. Lehmann LJ, Schollmeyer A, Stoeve J, Biochemical analysis of the synovial fluid of the shoulder joint in patients with and without rotator cuff tears: Z Orthop Unfall, 2010; 148; 90-94
23. Leal MF, Caires Dos Santos L, Martins de Oliveira A, Epigenetic regulation of metalloproteinases and their inhibitors in rotator cuff tears: PLoS One, 2017; 12; e0184141
24. Lo IK, Marchuk LL, Hollinshead R, Matrix metalloproteinase and tissue inhibitor of matrix metalloproteinase mRNA levels are specifically altered in torn rotator cuff tendons: Am J Sports Med, 2004; 32; 1223-29
25. Cox J, Varatharajan S, Cote P, Effectiveness of acupuncture therapies to manage musculoskeletal disorders of the extremities: A systematic review: J Orthop Sports Phys Ther, 2016; 46; 409-29
26. Nakajima M, Inoue M, Hojo T, Effect of electroacupuncture on the healing process of tibia fracture in a rat model: A randomised controlled trial: Acupunct Med, 2010; 28; 140-43
27. Inoue M, Nakajima M, Oi Y, The effect of electroacupuncture on tendon repair in a rat Achilles tendon rupture model: Acupunct Med, 2015; 33; 58-64
28. Davidson J, Burkhart SS, The geometric classification of rotator cuff tears: A system linking tear pattern to treatment and prognosis: Arthroscopy, 2010; 26(3); 417-24
29. Kuhn JE, Dunn WR, Sanders R, Effectiveness of physical therapy in treating atraumatic full-thickness rotator cuff tears: A multicenter prospective cohort study: J Shoulder Elbow Surg, 2013; 22; 1371-79
30. MacPherson H, Altman DG, Hammerschlag R, Revised STandards for Reporting Interventions in Clinical Trials of Acupuncture (STRICTA): Extending the CONSORT statement: J Evid Based Med, 2010; 3(3); 140-55
31. Tukmachi ES, Frozen shoulder: A comparison of Western and traditional Chinese approaches and a clinical study of its acupuncture treatment: Acupuncture in Medicine, 1999; 17; 9-21
32. Lim S, WHO standard acupuncture point locations: Evid Based Complement Alternat Med, 2010; 7(2); 167-68
33. Yamazaki H, Ochiai N, Kenmoku T, Assessment of pain-related behavior and pro-inflammatory cytokine levels in the rat rotator cuff tear model: J Orthop Res, 2014; 32; 286-90
34. Siu KK, Zheng LB, Ko JY, Increased interleukin 1β levels in the subacromial fluid in diabetic patients with rotator cuff lesions compared with nondiabetic patients: J Shoulder Elbow Surg, 2013; 22(11); 1547-51
35. Nakama K, Gotoh M, Yamada T, Interleukin-6-induced activation of signal transducer and activator of transcription-3 in ruptured rotator cuff tendon: J Int Med Res, 2006; 34(6); 624-31
36. Gotoh M, Hamada K, Yamakawa H, Interleukin-1 – induced glenohumeral synovitis and shoulder pain in rotator cuff diseases: J Orthop Res, 2002; 20; 1365-71
37. Chaudhury S, Xia Z, Thakkar D, Gene expression profiles of changes underlying different-sized human rotator cuff tendon tears: J Shoulder Elbow Surg, 2016; 25(10); 1561-70
38. Inoue M, Nakajima M, Hojo T, The effect of electroacupuncture on osteotomy gap healing in a rat fibula model: Acupunct Med, 2013; 31; 222-27
39. dos Santos de Almeida M, de Aro AA, Da Ré Guerra F, Electroacupuncture increases the concentration and organization of collagen in a tendon healing model in rats: Connect Tissue Res, 2012; 53; 542-47
40. Zheng X, Nie Y, Sun C, Long-term electroacupuncture stimulation prevents osteoporosis in ovariectomised osteopaenic rats through multiple signalling pathways: Acupunct Med, 2018; 36; 176-82
41. Song MJ, Wang YQ, Wu GC, Additive anti-hyperalgesia of electroacupuncture and intrathecal antisense oligodeoxynucleotide to interleukin-1 receptor type I on carrageenan-induced inflammatory pain in rats: Brain Res Bull, 2009; 78; 335-41
42. Wu GW, Chen J, Huang YM, Electroacupuncture delays cartilage degeneration by modulating nuclear factor-κB signaling pathway: Chin J Integr Med, 2019; 25(9); 677-83
43. Bao F, Sun H, Wu ZHEffect of acupuncture on expression of matrix metalloproteinase and tissue inhibitor in cartilage of rats with knee osteoarthritis: Zhong Guo Zhen Jiu, 2011; 31(3); 241-46 [in Chinese]
44. Zhou J, Zhong P, Liao Y, Electroacupuncture ameliorates subchondral bone deterioration and inhibits cartilage degeneration in ovariectomised rats: Acupunct Med, 2018; 36; 37-43
Figures
Tables
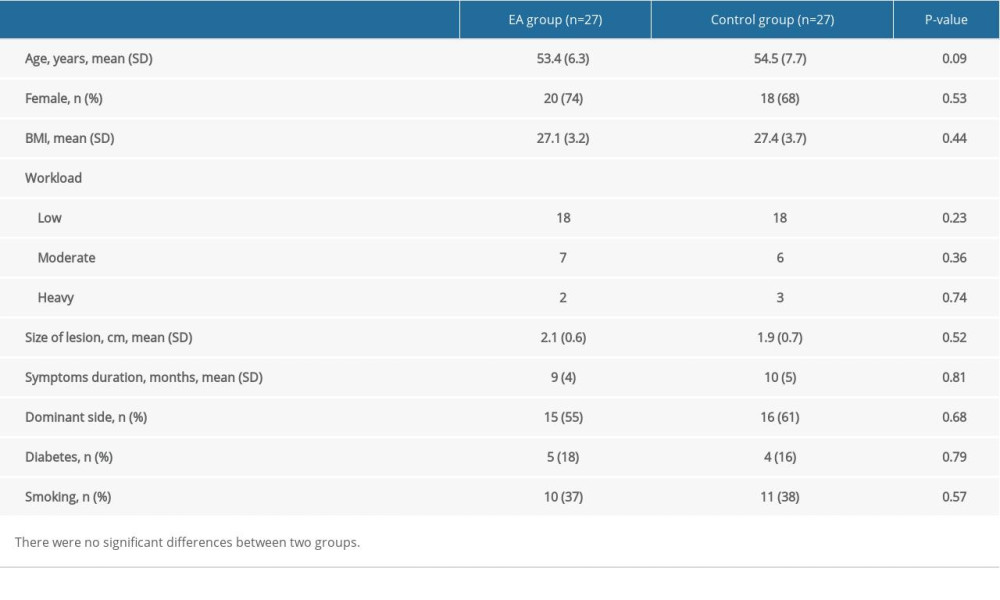 Table 1. Characteristics of patients in EA group and control group.
Table 1. Characteristics of patients in EA group and control group.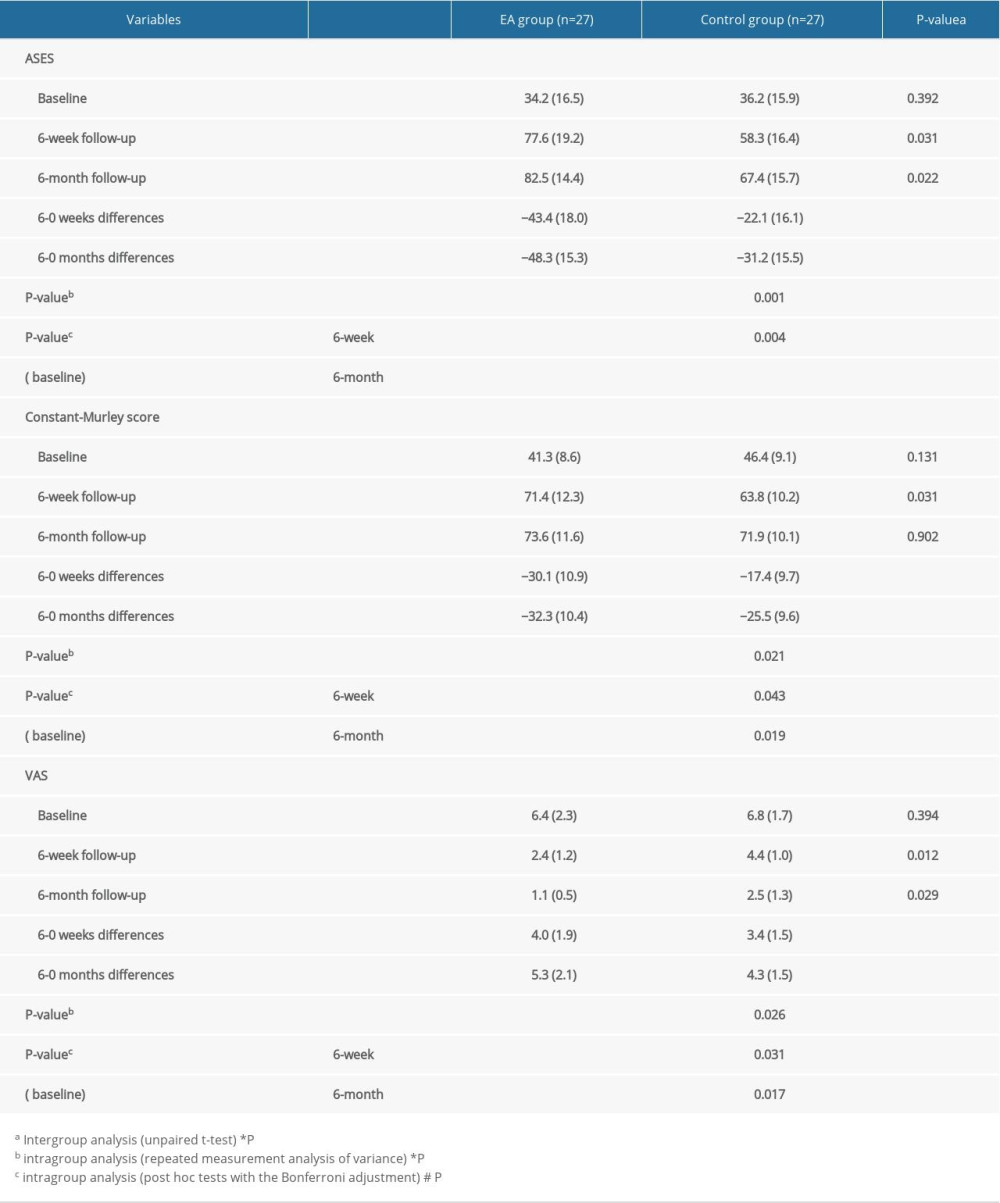 Table 2. Outcome measurements of patients in EA group and control group.
Table 2. Outcome measurements of patients in EA group and control group.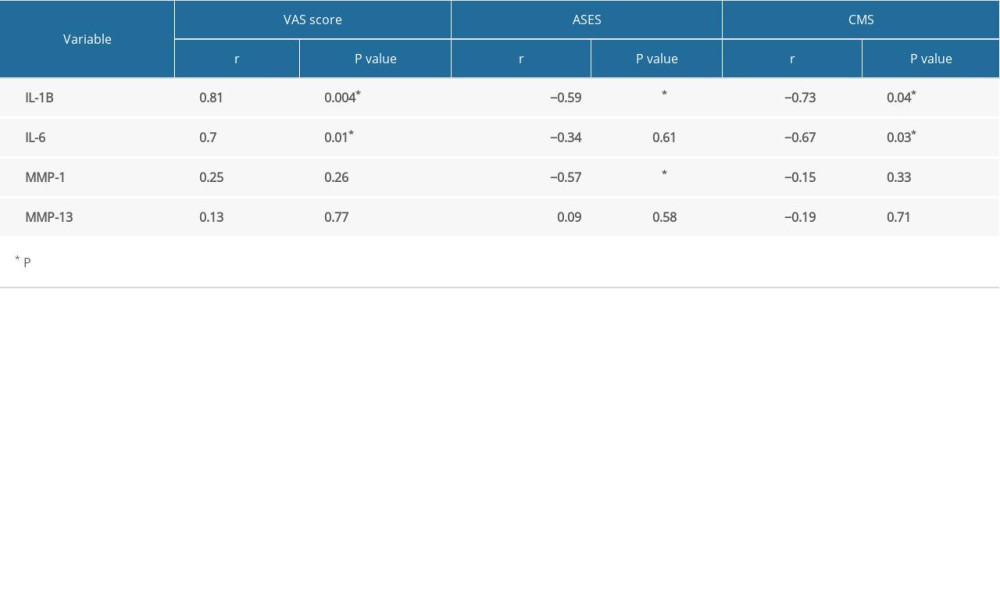 Table 3. Correlation between IL-1, IL-6, MMP-1 and MMP-13 levels and the VAS score CMS and ASES in all patients.
Table 3. Correlation between IL-1, IL-6, MMP-1 and MMP-13 levels and the VAS score CMS and ASES in all patients. Table 1. Characteristics of patients in EA group and control group.
Table 1. Characteristics of patients in EA group and control group. Table 2. Outcome measurements of patients in EA group and control group.
Table 2. Outcome measurements of patients in EA group and control group. Table 3. Correlation between IL-1, IL-6, MMP-1 and MMP-13 levels and the VAS score CMS and ASES in all patients.
Table 3. Correlation between IL-1, IL-6, MMP-1 and MMP-13 levels and the VAS score CMS and ASES in all patients. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387