10 July 2020: Database Analysis
Prognostic Determinants Analysis and Nomogram for Bone Malignant Vascular Tumors: A Surveillance, Epidemiology and End Results (SEER) Analysis
Sunli Hu12ABCDEFG, Xiangyang Wang1AB*DOI: 10.12659/MSM.923305
Med Sci Monit 2020; 26:e923305
Abstract
BACKGROUND: The aim and objective of our investigations were to explore the prognostic value of various clinical and pathological factors of bone malignant vascular tumors and establish a nomogram for their outcome predictions.
MATERIAL AND METHODS: All data of primary bone malignant vascular tumors (MVTs) patients were randomly selected from the Surveillance, Epidemiology and End Results (SEER) database. However, selected patients were clinically diagnosed with various cancers during 1988–2015. The potential prognostic factors were analyzed using SPSS (Windows, version 22.0). All prognostic factors were combined to formulate a nomogram to predict the overall survival (OS).
RESULTS: A total of 266 selected patients were included in our study. In the univariate model, age (P<0.001), sex (p=0.0255), primary site (P<0.001), surgery (P<0.001), histologic type (P<0.001), metastasis (p=0.000), and pathological grade (P<0.001) were statistically significant for patient survival. The results of Cox analysis revealed that age (≥64) HR: 3.636, 95% CI [1.955–6.762], p=0.000, the primary site in skull HR: 2.6, 95% CI [1.584–4.268], p<0.001], without surgery HR: 1.473 95% CI [1.239–1.751], p<0.001, metastasis HR: 3.076 95% CI [1.983–4.771] p=0.000, man HR 1.802, 95% CI [1.032–3.004], p=0.045, and high malignant grade HR: 3.029, 95% CI [2.101–4.366], p=0.003 were independent unfavorable prognostic factors. Angiosarcoma had highest mortality rate among all vascular malignancies. The nomogram predicting overall survival achieved a C-index of 0.694 (95% CI 0.631, 0.745) in the SEER cohort.
CONCLUSIONS: Surgery can significantly increase OS survival time for bone MVTs, and low-grade malignancy is a significant factor for OS. However, advanced age, tumor metastasis, primary site in skull, AS, and male sex are predictors of poor prognosis.
Keywords: Bone Neoplasms, nomograms, SEER Program, Age Factors, Databases, Genetic, Proportional Hazards Models, Vascular Neoplasms
Background
Vascular malignancy is rare and can occur in the liver, lung, skin, bone, spleen, pleura, and lymph nodes [1–4]. Due to the presence of complex vessels within bone, vascular tumors have a higher incidence rate. Due to its low incidence rate, little is known about the pathogenesis and progression of this tumor, and further investigations are urgently needed to clarify the possible risk factors, pathogenesis, and progression of this tumor [5]. Vascular malignancy can be classified into various pathological types, including angiosarcoma (AS), hemangioendothelioma (HE) and its epithelioid variant (EHE), or hemangiopericytoma (HP) [6,7]. EHE is regarded as the most common type. In1982, the concept of vascular malignant tumor EHE was first proposed by Enzinger and Weiss. EHE has characteristic pathological features and progresses faster than angiosarcoma [8,9]. EHE is the most common malignant vascular tumor, accounting for 2% of soft-tissue tumors. Although there is a higher incidence of AS in vascular tumors, these patients generally have a better prognosis. The most common primary sites of AS are the neck (37%) and head (52%), followed by the breasts and extremities. In addition, AS in bone is extremely rare and accounts for only 1% of ASs, especially in the vertebral column [10]. AS consists of vascular channels with histological features arranged by endothelial cells with huge nucleoli, nuclei, and incremental mitosis [11]. The cell of EHE is fusion form and ovoid, and the cells are surrounded by irregular, anastomotic, fissured, or branching vascular lumen [12]. Histopathological examination of EHE substantially revealed 2 types of tissue structures – dilated blood vessels and spindle cells. Reticular sclerosin staining clearly showed that individual cells and groups of cells were distinctly surrounded by reticular fibers [13]. HP is considered to be tightly packaged, round-to-fusiform cells, around a well-developed, elaborate, branching “staghorn” vasculature [14]. The staghorn appearance is histologically characterized by irregular vascular channels with branches of different sizes. Nucleoli are usually single and have well-dispersed chromatin formation. Given there is no typical clinical symptom or imaging feature in the early-stage of MVTs in bone, most patients are clinically diagnosed in the end-stage and have poor outcomes. There is no clear evidence or studies systematically describing the clinical characteristics of the disease. Many researchers and clinicians have insufficient knowledge and more data is needed on the possible causes, risk factors, and clinical treatment options for patients [15–17]. The current data volume is actually too small to make meaningful statistical descriptions. The SEER database has been widely used for clinical studies of rare tumors, including chondrosarcoma. Therefore, we used the largest and most accurate tumor database in the United States to collect more data.
Material and Methods
DATA SOURCE AND SELECTION:
The data were gathered from the Surveillance, Epidemiology, and End Results (SEER) database, the world’s largest publicly accessible cancer registry, maintained by the National Cancer Institute [18]. Pathology reports are an important source of SEER data. The extracted pathological data were combined with data from other sources into the final case record. These sources included patient records, reports collected from independent diagnostic imaging and chemotherapy clinics, and death certificates. We used the incidence rate of SEER in the version 18 Registration Research Database. The end date of follow-up for this version of the database was the end of 2015. We found 266 cases of angiocentric vascular tumor of bone based on ICD-0-3. Each patient’s demographic features and the clinicopathological characteristics were carefully selected, including age, sex, race, metastasis, primary site, histologic type, marital status, tumor grade, surgery, and chemotherapy and radiation performed. In addition, the SEER database uses a 4-grade system to define the grading of the lesion: grade I (well-differentiate), grade II (moderately differentiated), grade III (poorly differentiated), and grade IV (undifferentiated). The SEER database also reports OS, defined as the period from first diagnosis until death. The X-tile program (Yale University, New Haven, CT, USA) was used to identify the best cutoff of age, and it determined the proper cutoff value based on the minimum p values obtained from log-rank chi-square statistics for continuous age. Survival curves were constructed using Kaplan-Meier analysis, and their deviations were carefully evaluated by log-rank test. The Pearson chi-square test was performed to analyze categorical variables, and the 2-sample t test was performed to analyze continuous variables. The survival curves of OS were compared by log-rank test in various groups with the Kaplan-Meier method. For adjustment for baseline variables during the comparison, we generally applied a Cox proportional hazard model, which included all potential prognostic factors obtained from the SEER database. All statistical analyses were performed using the statistical software package SPSS (Windows, version 22.0, BM Corp., Armonk, NY, USA), and p values <0.05 were considered statistically significant.
Results
PATIENT DESCRIPTIONS:
The comprehensive clinical features of all 266 patients are shown in Table 1. We included 160 (60.1%) men and 106 (39.9%) women, with a mean age of 58.45 years (median 61 years). There were 230 whites, 23 blacks, and 13 people of other ethnic groups. There were 140 patients clinically diagnosed with primary MVTs in bones of the extremities 26 in the skull, 71 in the spine, and 13 in bones of the trunk. For the histological type, AS and EHE accounted for 74.3% of all patients, of which AS accounted for 57.1%. There were 112 (42.1%) cases who received radiation and 154 (57.9%) who did not, 61 (22.9%) received chemotherapy and 205 (77.1%) did not, 153 (57.5%) received surgery and 113 (42.5%) did not. A total of 105 (39.5%) patients were clinically diagnosed with tumors which spread to another organ in the body. There were 201 patients who were married when diagnosed with a malignant vascular tumor and 65 patients were not married. Of all patients, 56 patients were selectively identified as having a low-grade tumor, and 69 patients were diagnosed as having a high-grade tumor. The mean follow-up time was 46.95 months (median 14.0 months; range 1–335 months). X-Plots were constructed, identifying 64 years old as the optimal cutoff point to divide the tested cohort into low and high risk based on OS (Figure 1). Univariate and multivariate analyses were used to identify the potential prognostic factors for overall survival. Univariate analysis results showed that radiotherapy, marital status, and chemotherapy are associated with age (p=0.000), sex (p=0.0255), histological subtype (p=0.000), tumor grade (p=0.000), metastasis (p=0.000), tumor site (p=0.000), and surgery (p=0.000), meaning that these characteristics were strongly associated with OS in the univariate analysis (p<0.05) (Table 1). Then, significant prognostic factors from univariate analysis were carefully assessed for further multivariate analysis. These prognostic factors extracted by univariate analysis were subjected to Cox proportional hazards analysis (Table 2), showing that age <64 years, low-grade malignancy, female sex, diagnosis without metastasis, and receiving surgery were predictors for OS. However, characteristics of the site of onset and pathological types were risk factors for OS, including angiosarcoma and a vascular malignancy of the skull. These findings indicate that people older than 64 years were 3.636 times more likely to be diagnosed than those younger than 64 years (RR=3.636). The relative risk of not receiving surgery was 1.473 times higher than in patients who received surgery (RR=1.473). Metastasis was associated with significantly worsen of OS (No vs. Yes, p=0.000). The relative risk of metastasis was 1.473 times higher in patients without metastasis (RR=3.075). In addition, the data showed the stratification of prognostic factors. The statistical results were age (χ2=18.997, P=0.000), metastasis (χ2=25.552, P=0.000), primary site (χ2=20.231, p=0.0035), sex (χ2=4.033, P=0.0225), and histological type (χ2=69.2, p=0.0000), surgery (χ2=20.195, p=0.0008), and grade (χ2=51.293, p=0.0016), as shown in Table 2. Kaplan-Meier curves were used to show OS for the full cohort (Figure 2). A nomogram predicting 3- and 5-year overall survival was constructed by incorporating the above independent prognostic factors (Figure 3). The C-index of the nomogram for predicting overall survival was about 0.694 in the SEER cohort.
Discussion
MVTs are rare primary cancer in the skeleton, with low diagnosis rate and poor prognosis [5,19]. There are no clear investigations for statistical analysis of clinical features; the possible prognostic factors have been unknown until now. A retrospective analysis of 46 patients with malignant vascular tumors in the spine suggested that age, surgery, metastasis, and malignant grade were prognostic factors for overall survival [20]. A multicenter retrospective study of Japanese patients with epithelioid hemangioendothelioma showed that tumor size >3.0 cm is an independent indicator of poor prognosis [21].
In the present study, we applied univariate and multivariate analysis to identify the potential independent prognostic factors based on the SEER database, which are related to OS in patients with bone MVTs. We established a nomogram clinical prediction model based on these significant prognostic factors. The survival rate of specific tumor patients can be calculated individually, having a great value in clinical practice, which is the main advantage of this model. We found the mean age of patients was 58.4 years, which is consistent with previous studies [5]. Furthermore, our results showed that age was a significant independent prognostic factor for OS of MVTs in bone. We applied the X-tile model to identify optimal cutoff values of age. Our analysis strongly indicated that 64 years of age is an important watershed in determining the prognosis of patients. Therefore, the same findings were obtained including sex, primary site, histologic type, surgery, grade, year of diagnosis in our study. We collected data on race, marital status, and chemotherapy or radiation. Therefore, we hypothesized that radiotherapy and chemotherapy are independent risk factors for survival in patients with vascular tumors undergoing radiotherapy and chemotherapy [22–24]. Surprisingly, the potential effects of chemotherapy and radiation on patient survival were not statistically significant, although whether chemotherapy and radiotherapy should be included in conventional cancer treatments for MVTs still remains controversial [25].
MVTs include a range of pathological types. In addition to its unique histopathological features, some important molecular markers can also be used for early diagnosis and prognostic definition of MVTs. FISH and molecular analysis techniques are widely used to detect the molecular targets of various tumors, which play an important role in clinical and scientific studies. Nevertheless, a clinicopathological and molecular study of 21 cases of epithelioid hemangioma clearly suggested that all cases were positive for ERG and CD31, which is primarily used to demonstrate the presence of endothelial tissues and to evaluate tumor angiogenesis [26]. FOSB nuclear positivity was displayed in some lesioned endothelial cells. FOS gene rearrangements are most frequently found in EH in the bone by FISH analysis [27–29]. In addition, positive nuclear CAMTA1 is a key tool for diagnosis of epithelioid hemangioendothelioma [30].
The patients with high-grade malignancy also differ in appearance from the less malignant patients, for instance necrosis and invasion of bone tissues. Previous investigations showed that the degree of malignancy is also an important factor affecting prognosis [24], which was also shown in the present study. Patients diagnosed with cancer of low-grade malignancy often live longer and have better quality of life than those with high-grade malignancy. Moreover, surgery is mostly considered as a conventional methods of tumor treatment. Research shows surgical management of MVTs patients has a significant effect on patient prognosis. The OS of malignant vascular tumor patients in other organs are more or less prolonged after surgical treatment [31]. Complete resection of the primary tumor site can considerably improve the patient’s prognosis and prolong survival rates [32]. Consequently, curative-intent surgery to removal a primary tumor for patients diagnosed with cutaneous angiosarcoma could become a promising treatment option in the near future [33]. However, several investigations obviously demonstrate that the emphasis on primary hepatic epithelioid hemangioendothelioma is the most beneficial surgical treatment of this tumor [34]. Since blood vessels in bone and surrounding tissues are distinct in different parts, the possibility of vascular malignancy is also diverse. We found malignant hemangiomas in all bones in the SEER database and roughly divided them into the following sections: skull – bone of skull and face and associated joints (includes mandible); trunk bones – rib, sternum, clavicle, and associated joint; spine – vertebral column (includes sacrum and coccyx); and bones of the extremities – bone, joint, and articular cartilage of limbs. According to the statistical results, patients with malignant hemangioma growing in the skull have the shortest survival time, which may be related to the complex vascular network in the brain. External validation is a vital method to address overfitting and to explore the external relevance of the nomogram. Therefore, our remarkable findings can be beneficial to create a new clinical prediction model called a nomogram in the near future.
Our investigations also contain the largest number of cases and most complete clinical features of patients for MVTs in bone. All patients’ data were carefully obtained from the same database; there are unified standards and procedures for evaluation of the patients. However, there are still some limitations in our study. This large-scale retrospective study of 266 patients found that radiotherapy, chemotherapy, and surgery improved survival rates. However, the SEER database does not have complete records on specific surgical methods and other combined treatment measures, leading to the incomplete selection of potential prognostic factors.
Conclusions
We designed and evaluated a prognostic nomogram for patients with primary malignant vascular tumors of bone. The nomogram showed good discrimination ability. As a novel nomogram specific to primary malignant vascular tumors of bone, our model needs to be improved by further research. Our results may be of use in developing bone MVT patient health education and to provide a basis for further investigations.
Figures
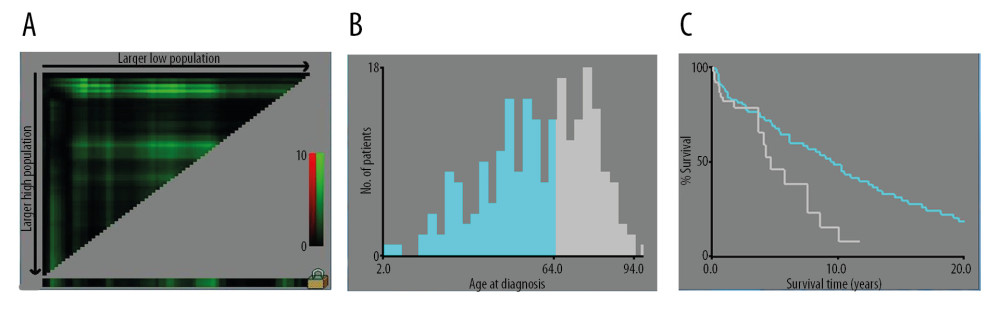 Figure 1. (A–C) The image show defining the optimal cutoff values of age via X-tile analysis. (A) The black dot indicates that optimal cutoff values of age have been identified. (B) A histogram and (C) Kaplan-Meier curve were constructed based on the identified cutoff values. Optimal cutoff values of age were identified as 64 years based on survival.
Figure 1. (A–C) The image show defining the optimal cutoff values of age via X-tile analysis. (A) The black dot indicates that optimal cutoff values of age have been identified. (B) A histogram and (C) Kaplan-Meier curve were constructed based on the identified cutoff values. Optimal cutoff values of age were identified as 64 years based on survival. 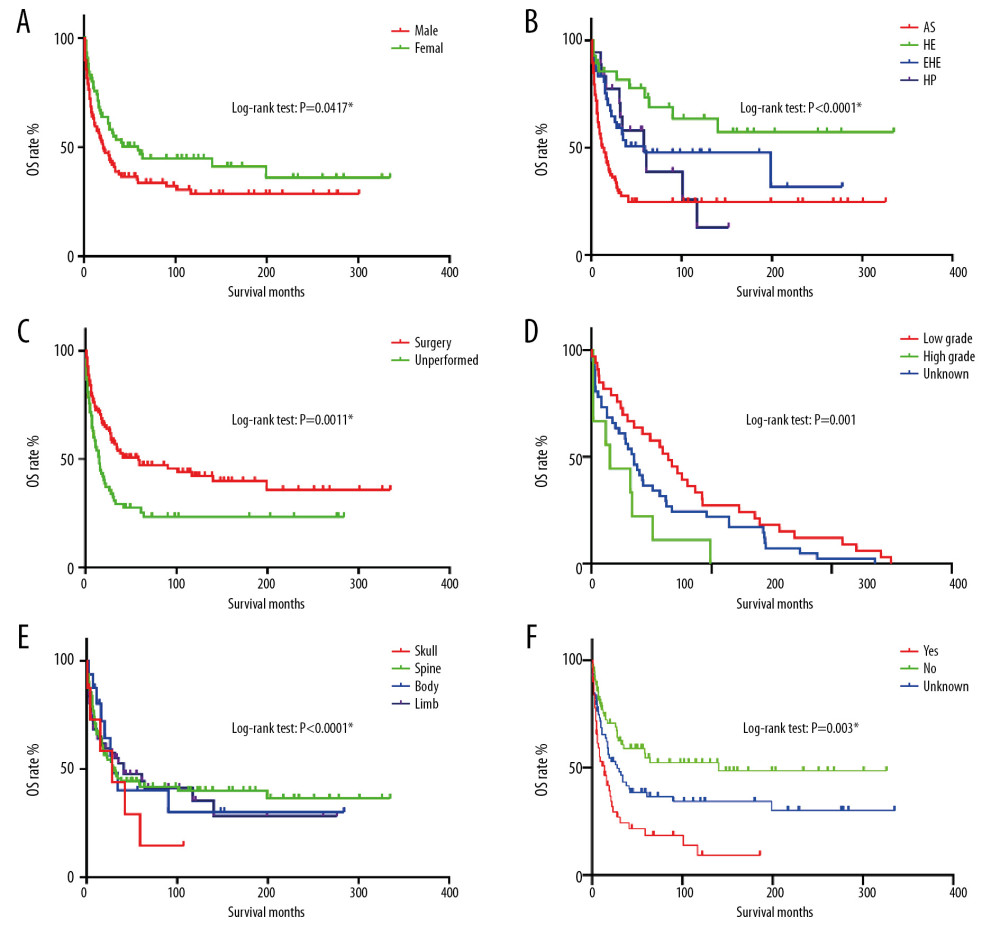 Figure 2. OS for patients with bone MVT Notes: (A) OS for patients stratified by sex. (B) OS for patients stratified by histological classification. (C) OS for patients with/without surgery. (D) OS for patients separated by grade malignancy. (E) OS for patients stratified by primary sites. (F) OS for patients stratified by metastasis status.
Figure 2. OS for patients with bone MVT Notes: (A) OS for patients stratified by sex. (B) OS for patients stratified by histological classification. (C) OS for patients with/without surgery. (D) OS for patients separated by grade malignancy. (E) OS for patients stratified by primary sites. (F) OS for patients stratified by metastasis status. 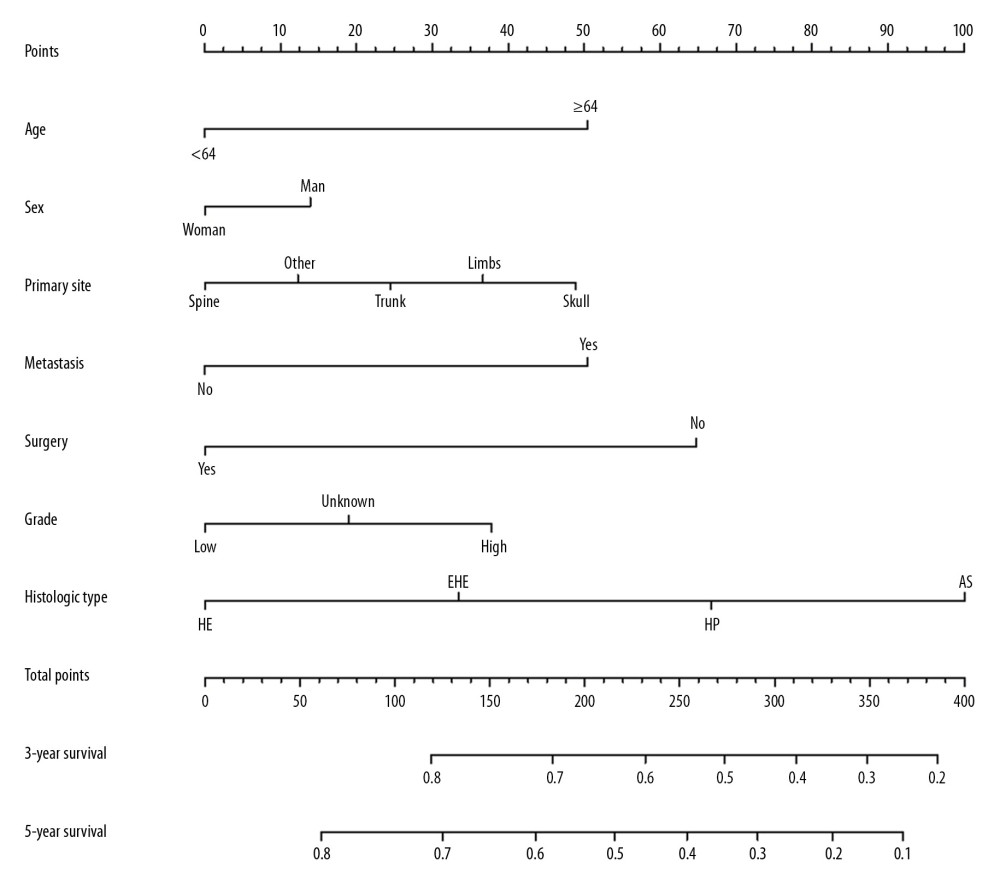 Figure 3. Cancer-specific survival nomogram for patients with malignant vascular tumors of bone.
Figure 3. Cancer-specific survival nomogram for patients with malignant vascular tumors of bone. References
1. Deshpande V, Rosenberg AE, O’Connell JX, Epithelioid angiosarcoma of the bone: A series of 10 cases: Am J Surg Pathol, 2003; 27; 709-16
2. Deyrup AT, Tighiouart M, Montag AG, Epithelioid hemangioendothelioma of soft tissue: A proposal for risk stratification based on 49 cases: Am J Surg Pathol, 2008; 32; 924-27
3. Xu W-N, Yang R-Z, Zheng H-L, PGC-1{a acts as an mediator of Sirtuin2 to protect annulus fibrosus from apoptosis induced by oxidative stress through restraining mitophagy: Int J Biol Macromol, 2019; 136; 1007-17
4. Maddox JC, Evans HL, Angiosarcoma of skin and soft tissue: A study of forty-four cases: Cancer, 1981; 48; 1907-21
5. Leowardi C, Hinz U, Hormann Y, Malignant vascular tumors: Clinical presentation, surgical therapy, and long-term prognosis: Ann Surg Oncol, 2005; 12; 1090-101
6. van IJzendoorn DGP, Bovée JVMG, Vascular tumors of bone: The evolvement of a classification based on molecular developments: Surg Pathol Clinics, 2017; 10; 621-35
7. Rosenberg AE, WHO classification of soft tissue and bone, fourth edition: Summary and commentary: Curr Opin Oncol, 2013; 25; 571-73
8. Weiss SW, Enzinger FM, Epithelioid hemangioendothelioma: A vascular tumor often mistaken for a carcinoma: Cancer, 1982; 50; 970-81
9. Weiss SW, Ishak KG, Dail DH, Epithelioid hemangioendothelioma and related lesions: Semin Diagn Pathol, 1986; 3; 259-87
10. Young RJ, Brown NJ, Reed MW, Angiosarcoma: Lancet Oncol, 2010; 11; 983-91
11. Wenger DE, Wold LE, Malignant vascular lesions of bone: Radiologic and pathologic features: Skeletal Radiol, 2000; 29; 619-31
12. Requena L, Kutzner H, Hemangioendothelioma: Semin Diagn Pathol, 2013; 30; 29-44
13. Balansay BE, Zhang X, Loftus PD, Diagnosing epithelioid hemangioendothelioma with pericardial involvement: Ann Thorac Surg, 2018; 106; e173-75
14. Koch M, Nielsen GP, Yoon SS, Malignant tumors of blood vessels: Angiosarcomas, hemangioendotheliomas, and hemangioperictyomas: J Surg Oncol, 2008; 97; 321-29
15. Puri S, Gupta N, Asotra S, Hemangioma of rib masquerading a malignancy: J Cancer Res Ther, 2018; 14; S809-11
16. Zhou Q, Lu L, Fu Y, Epithelioid hemangioma of bone: A report of two special cases and a literature review: Skeletal Radiol, 2016; 45; 1723-27
17. Zhu Y, Fan M, Pandey S, Multiple epithelioid hemangioedothelioma of the skull in a child: A case report: Medicine, 2016; 95; e4081
18. Decullier E, Lhéritier V, Chapuis F, The activity of French research ethics committees and characteristics of biomedical research protocols involving humans: A retrospective cohort study: BMC Med Ethics, 2005; 6; E9
19. Das JM, Louis R, Sunilkumar BS, Primary epithelioid hemangioendothelioma of the dorsal spine: A review: Neurol India, 2017; 65; 1180-86
20. Xu K, Liu Y, Li B, Prognostic factors of patients with malignant epithelioid vascular tumors in the spine: Retrospective analysis of 46 patients in a single center: Spine (Phila Pa 1976), 2018; 43; E1218-24
21. Shiba S, Imaoka H, Shioji K, Clinical characteristics of Japanese patients with epithelioid hemangioendothelioma: A multicenter retrospective study: BMC Cancer, 2018; 18; 993
22. Jeong Y, Shin MH, Yoon SM, Liver transplantation after transarterial chemoembolization and radiotherapy for hepatocellular carcinoma with vascular invasion: J Gastrointest Surg, 2017; 21; 275-83
23. Sundby Hall K, Bruland ØS, Bjerkehagen B, Adjuvant chemotherapy and postoperative radiotherapy in high-risk soft tissue sarcoma patients defined by biological risk factors-A Scandinavian Sarcoma Group study (SSG XX): Eur J Cancer, 2018; 99; 78-85
24. Yin H, Yang X, Xu W, Treatment and outcome of primary aggressive giant cell tumor in the spine: Eur Spine J, 2015; 24; 1747-53
25. Mark RJ, Poen JC, Tran LM, Angiosarcoma. A report of 67 patients and a review of the literature: Cancer, 1996; 77; 2400-6
26. Fetsch JF, Sesterhenn IA, Miettinen M, Epithelioid hemangioma of the penis: a clinicopathologic and immunohistochemical analysis of 19 cases, with special reference to exuberant examples often confused with epithelioid hemangioendothelioma and epithelioid angiosarcoma: Am J Surg Pathol, 2004; 28; 523-33
27. Antonescu CR, Chen HW, Zhang L, ZFP36-FOSB fusion defines a subset of epithelioid hemangioma with atypical features: Genes Chromosomes Cancer, 2014; 53; 951-59
28. Huang SC, Zhang L, Sung YS, Frequent FOS gene rearrangements in epithelioid hemangioma: A molecular study of 58 cases with morphologic reappraisal: Am J Surg Pathol, 2015; 39; 1313-21
29. van IDG, de Jong D, Romagosa C, Fusion events lead to truncation of FOS in epithelioid hemangioma of bone: Genes Chromosomes Cancer, 2015; 54; 565-74
30. Doyle LA, Fletcher CD, Hornick JL, Nuclear expression of CAMTA1 distinguishes epithelioid hemangioendothelioma from histologic mimics: Am J Surg Pathol, 2016; 40; 94-102
31. Wang B, Meng N, Zhuang H, The role of radiotherapy and surgery in the management of aggressive vertebral hemangioma: A retrospective study of 20 patients: Med Sci Monit, 2018; 24; 6840-50
32. Sasaki R, Soejima T, Kishi K, Angiosarcoma treated with radiotherapy: Impact of tumor type and size on outcome: Int J Radiat Oncol Biol Phys, 2002; 52; 1032-40
33. Oashi K, Namikawa K, Tsutsumida A, Surgery with curative intent is associated with prolonged survival in patients with cutaneous angiosarcoma of the scalp and face – a retrospective study of 38 untreated cases in the Japanese population: Eur J Surg Oncol, 2018; 44; 823-29
34. Mehrabi A, Kashfi A, Fonouni H, Primary malignant hepatic epithelioid hemangioendothelioma: A comprehensive review of the literature with emphasis on the surgical therapy: Cancer, 2006; 107; 2108-21
Figures
 Figure 1. (A–C) The image show defining the optimal cutoff values of age via X-tile analysis. (A) The black dot indicates that optimal cutoff values of age have been identified. (B) A histogram and (C) Kaplan-Meier curve were constructed based on the identified cutoff values. Optimal cutoff values of age were identified as 64 years based on survival.
Figure 1. (A–C) The image show defining the optimal cutoff values of age via X-tile analysis. (A) The black dot indicates that optimal cutoff values of age have been identified. (B) A histogram and (C) Kaplan-Meier curve were constructed based on the identified cutoff values. Optimal cutoff values of age were identified as 64 years based on survival. Figure 2. OS for patients with bone MVT Notes: (A) OS for patients stratified by sex. (B) OS for patients stratified by histological classification. (C) OS for patients with/without surgery. (D) OS for patients separated by grade malignancy. (E) OS for patients stratified by primary sites. (F) OS for patients stratified by metastasis status.
Figure 2. OS for patients with bone MVT Notes: (A) OS for patients stratified by sex. (B) OS for patients stratified by histological classification. (C) OS for patients with/without surgery. (D) OS for patients separated by grade malignancy. (E) OS for patients stratified by primary sites. (F) OS for patients stratified by metastasis status. Figure 3. Cancer-specific survival nomogram for patients with malignant vascular tumors of bone.
Figure 3. Cancer-specific survival nomogram for patients with malignant vascular tumors of bone. Tables
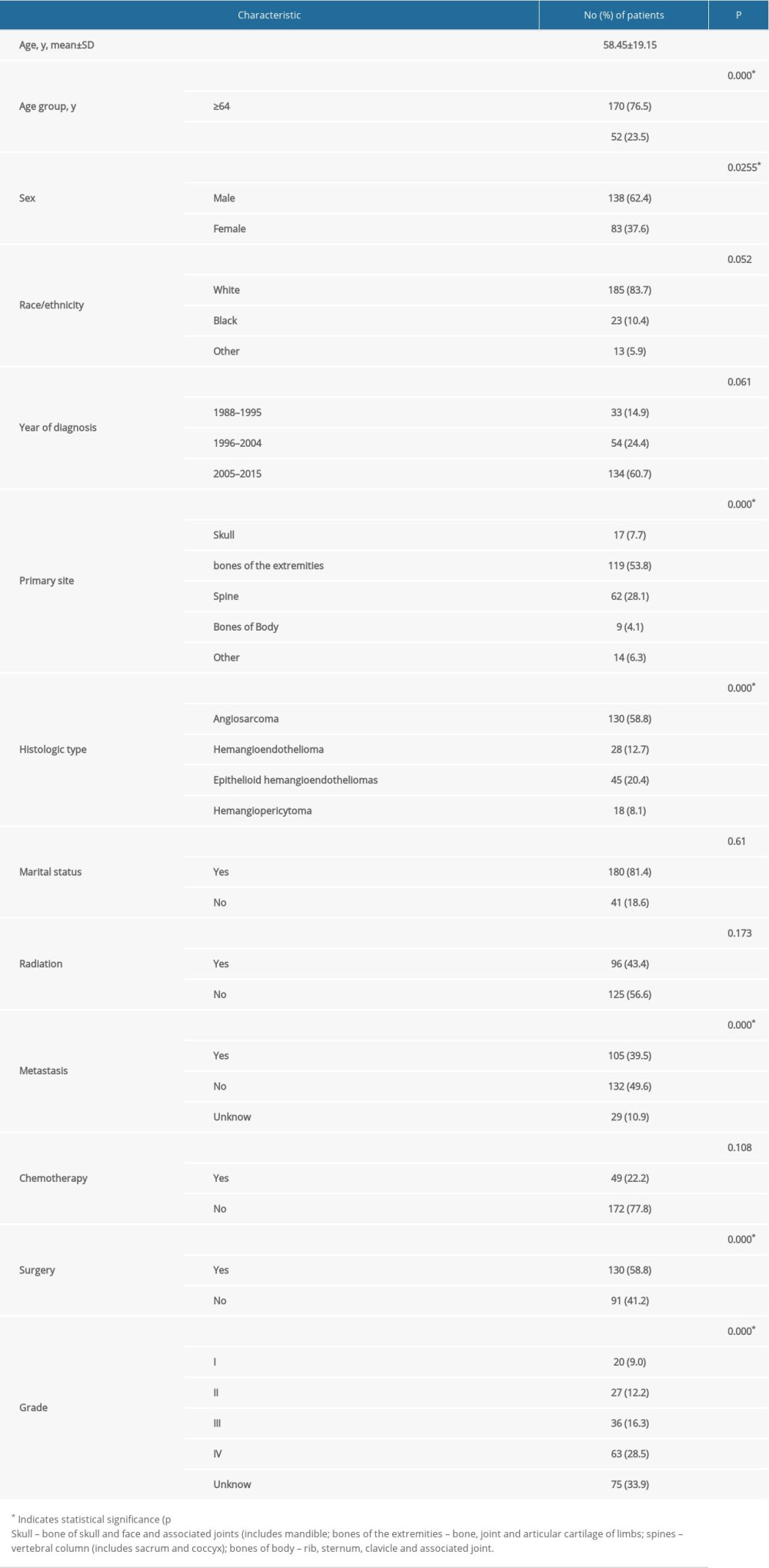 Table 1. Patient characteristics and univariate analysis of prognostic factors affecting overall survival.
Table 1. Patient characteristics and univariate analysis of prognostic factors affecting overall survival.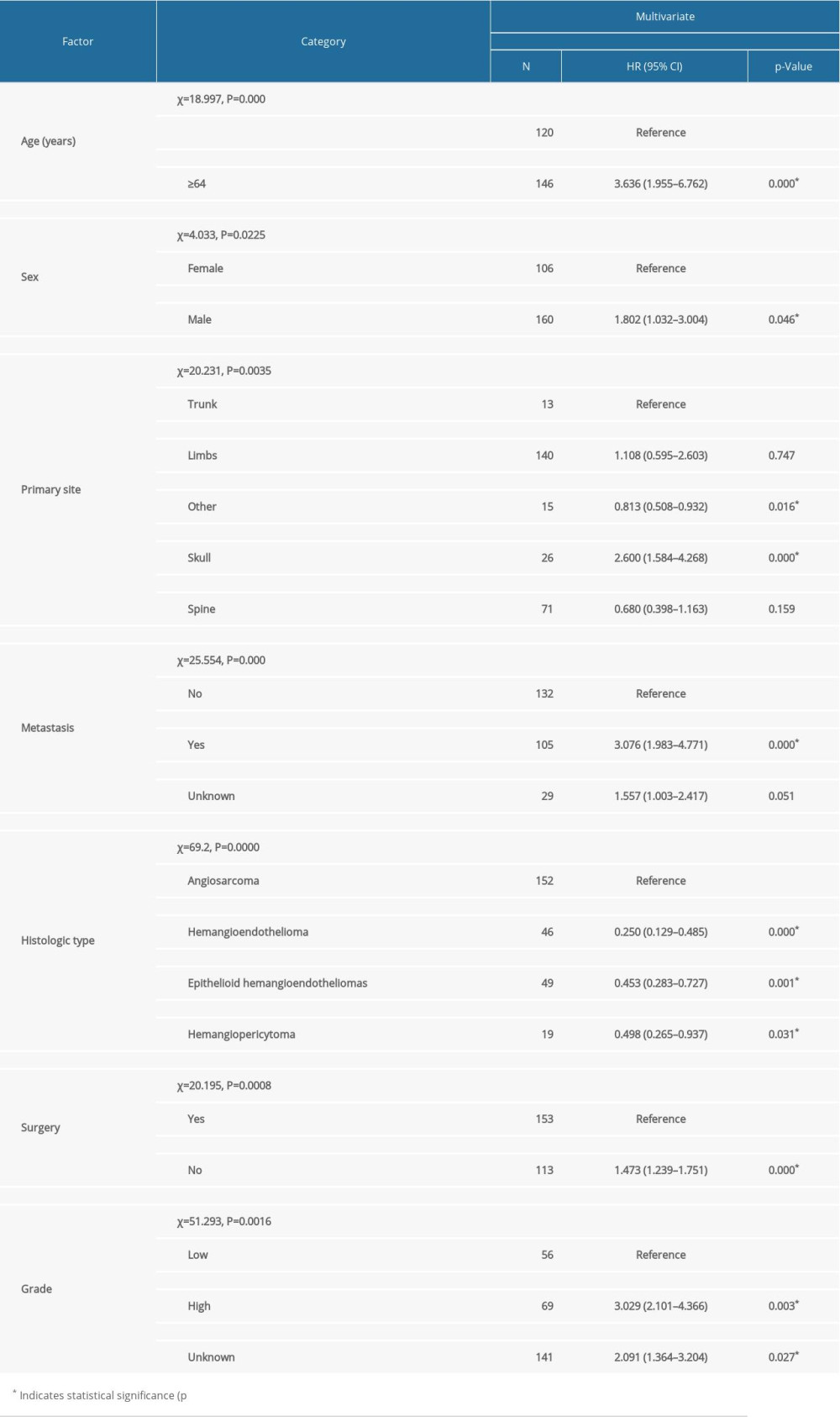 Table 2. Multivariate analysis of prognostic factors in patients with primary malignant vascular tumors of bone.
Table 2. Multivariate analysis of prognostic factors in patients with primary malignant vascular tumors of bone. Table 1. Patient characteristics and univariate analysis of prognostic factors affecting overall survival.
Table 1. Patient characteristics and univariate analysis of prognostic factors affecting overall survival. Table 2. Multivariate analysis of prognostic factors in patients with primary malignant vascular tumors of bone.
Table 2. Multivariate analysis of prognostic factors in patients with primary malignant vascular tumors of bone. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








