30 August 2020: Lab/In Vitro Research
Honokiol Suppression of Human Epidermal Growth Factor Receptor 2 (HER2)-Positive Gastric Cancer Cell Biological Activity and Its Mechanism
Yidan Yan1ABC, Jianmin Xu2CDE, Guoxin Mao3EFG*DOI: 10.12659/MSM.923962
Med Sci Monit 2020; 26:e923962
Abstract
BACKGROUND: The purpose of our study was to determine the effects and mechanisms of honokiol on human epidermal growth factor receptor 2 (HER2)-positive gastric cancer cells by in vitro study.
MATERIAL AND METHODS: We measured HER2 expression in different gastric cancer cell lines by real-time quantitative polymerase chain reaction (RT-qPCR) and western blot (WB) assay. Cell proliferation, apoptosis, and cell cycle were evaluated by cell-counting kit 8 and flow cytometry assays. The invading cell numbers and wound-healing rates were measured by transwell and wound-healing assays. Phosphatidylinositol 3-kinase (PI3K), protein kinase B (AKT), P21, and matrix metalloproteinase (MMP)-9 proteins and messenger ribonucleic acid (mRNA) expression were measured by WB and RT-qPCR assay. HER2 protein expression was evaluated by cellular immunofluorescence.
RESULTS: Honokiol suppressed cell proliferation via increasing cell apoptosis, invasion, and migration with dose dependence. By WB and RT-qPCR assays, compared with the control group, PI3K, AKT, P21, and MMP-9 proteins and mRNA expression were significantly different (P<0.05). By cellular immunofluorescence, HER2 protein expression was significantly depressed in honokiol-treated groups compared with control groups (P<0.05).
CONCLUSIONS: Honokiol has suppressive effects on HER2-positive gastric cancer cell biological activities via regulation of HER2/PI3K/AKT pathways in vitro.
Keywords: Receptor, erbB-2, Biphenyl Compounds, Case-Control Studies, Lignans, Matrix Metalloproteinase 9, phosphatidylinositol 3-kinase, RNA, Messenger, Tumor Cells, Cultured
Background
Gastric cancer is the fourth most common cancer and the third most common cause of cancer-related death [1,2]. At present, the treatment of gastric cancer includes surgery, radiotherapy, chemotherapy, immunotherapy, and targeted therapy. However, radical resection is still the only treatment of early gastric cancer. Preoperative neoadjuvant therapy, perioperative chemotherapy, and postoperative adjuvant chemoradiotherapy can reduce the recurrence rate and improve the disease-free survival of patients. Clinical trials have demonstrated that preoperative neoadjuvant therapy can significantly improve the 5-year survival rate of patients and improve the radical resection rate and the efficacy of surgical treatment in patients with advanced gastric cancer [3–8]. Research on tumor pathogenesis has suggested that the occurrence and metastasis of gastric cancer are related to the overexpression of some specific oncogenes or receptor genes [9]. Human epidermal growth factor receptor 2 (HER2) is one of the proto-oncogenes that has been extensively investigated and has protein overexpression and/or gene amplification at different degrees in gastric cancer, breast cancer, and ovarian cancer, and plays an important role in the occurrence, development, and metastasis of tumors [10–12]. Basic experiments have confirmed that HER2 can affect the biological activity of tumor cells by activating the mitogen-activated protein kinase and phosphatidylinositol 3-kinase/protein kinase B (P13K/AKT) signaling pathways [13].
Honokiol (Hon) is one of the main components of
Material and Methods
MATERIAL SOURCE:
NCI-N87, OE-19, AGS, MGC-803, and SGC-7901 cells were purchased from the Cell Bank of the Chinese Academy of Sciences in Shanghai. Dulbecco’s modified Eagle medium (DMEM) (HyClone, Logan, UT, USA) was used. Hon (purity ≥98%) was purchased from Chengdu Best Reagent Co., Ltd. (Chengdu, China), dimethyl sulfoxide (DMSO, meeting the standard of the American Chemical Society) from Beijing Solarbio Science & Technology Co., Ltd. (Beijing, China), and cell-counting kit 8 (CCK-8) from Invitrogen (Carlsbad, CA, USA). Additionally, HER2 antibody (Abcam, Cambridge, UK), PI3K (CST, Danvers, MA, USA), AKT (CST), P21 (Abcam), matrix metalloproteinase (MMP)-9 (Abcam), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (Abcam) were used.
CELL CULTURE:
Cells were cultured in DMEM (containing 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin) in a 5% CO2 incubator at 37°C.
CCK-8 ASSAY:
Cells at the exponential growth phase were inoculated into a 24-well plate and treated with 5.0, 10.0, and 20.0 μmol/L Hon. After 24 and 48 h, crystal violet staining was performed to detect cell proliferation. The method was as follows: the plate was washed once with phosphate-buffered saline (PBS), saturated crystal violet solution was added (crystal violet in 500 μL of PBS and 10% formaldehyde), and then the plate was incubated at room temperature for 20 min. After washing with PBS three times, the plate was dried at room temperature for scanning. Next, 500 μL of 10% acetic acid was added to dissolve crystal violet, with slight shaking. The absorbance was measured at 490 nm after CCK-8 treatment for 10 min. The method was repeated three times for each group.
FLOW CYTOMETRY:
Well-grown cells were inoculated into a 6-well plate. After the cells adhered to the wells, Hon at different concentrations or an equal volume of DMSO was added. After 48 h, the cells were collected and the cell cycle was analyzed according to the kit instructions, or the cells were digested and collected using trypsin without ethylenediaminetetraacetic acid, and apoptosis and the cell cycle were analyzed according to the kit instructions. The procedure was repeated three times for each group.
RT-QPCR ASSAY: The expression of related genes was detected by RT-qPCR. Total ribonucleic acid (RNA) was obtained using Trizol and complementary deoxyribonucleic acid was obtained by a reverse transcription kit. After electrophoretic identification and quantitation by enzyme-linked immunosorbent assay (ELISA), the reaction system and conditions were set according to Takara (Shiga, Japan) SYBR Green fluorescence quantitative kit. With GAPDH as housekeeping gene, the relative expression of target genes was calculated by the 2−ΔΔCT method. The sequence of primers is seen in Table 1.
WOUND-HEALING ASSAY:
The cells at the logarithmic stage of growth after corresponding treatment were collected, digested with trypsin, resuspended, adjusted to a cell density of 1.0×104/mL, and inoculated into a 6-well plate. After the cells were continuously cultured until refusion under the same conditions, a straight line was drawn in the center of the 6-well plate using a 50-μL sterile tip. The cell fragments at the edge of the straight line were blown gently with buffer to ensure neat edges. The cells were cultured in complete medium for 24 and 48 h, and the cell migration rate was photographed and calculated.
TRANSWELL ASSAY:
The cells at the logarithmic stage of growth in each group were collected and prepared in a single-cell suspension. The cup-shaped transwell chamber was taken, with the filter membrane at the bottom of the chamber evenly coated with artificial medium diluted with RPMI-1640 medium at 1: 10, and put in a constant-temperature incubator for drying. The supernatant of serum-free medium after cell culture for 24 and 48 h was collected and added to the lower chamber. Another single-cell suspension was collected and added to the upper chamber. Under the same conditions, the filter membrane at the bottom of the chamber was cultured for 24 and 48 h, fixed with 4% paraformaldehyde, and stained with crystal violet in the dark. Five visual fields were randomly selected and the number of cells passing through the membrane was counted under a microscope. The experiment was repeated three times.
WESTERN BLOT (WB) ASSAY:
Cells at the logarithmic stage of growth in each group were collected. Total protein was extracted and protein concentration determined by the Bradford method. The extracted total protein was mixed with gel-loading buffer, heated at 100°C for 5 min, and then stored at −70°C. Next, 12.5% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, semi-dry transfer at 1 mA/cm2 for 100 min, was performed, with sealing with skimmed milk powder for2 h. Rabbit anti-mouse PI3K, AKT, P21, MMP-9, and GAPDH primary antibodies at 1: 500 dilution were added and incubated overnight at 4°C. After rewarming at room temperature for 30 min, the mixture was washed with Tris-buffered saline with Tween 20 three times, and horseradish peroxidase-labeled immunoglobulin G secondary antibody at 1: 2000 dilution was added and allowed to react at 37°C for 1 h. The color was developed with chemiluminescence. Scanning and analysis were performed using Quantity One.
CELL IMMUNOFLUORESCENCE:
After washing with PBS, the cells at the logarithmic stage of proliferation in a 35-mm cell culture dish were fixed with tissue fixative, permeabilized with Triton X-100, and sealed with 5% bovine serum albumin. Then, the cells were diluted with HER2 primary antibody at 1: 200 and incubated at room temperature for 2 h. The nuclei were stained with 4′,6-diamidino-2-phenylindole. Observation was under a confocal laser scanning microscope after sealing.
STATISTICAL METHODS:
Statistical analysis was carried out using SPSS 20.0. Enumerated data were expressed as percent and analyzed by the chi-square test. Measurement data were expressed as mean ± standard deviation. Independent sample
Results
HER2 MESSENGER (M)RNA AND PROTEIN EXPRESSIONS IN DIFFERENT CELL LINES:
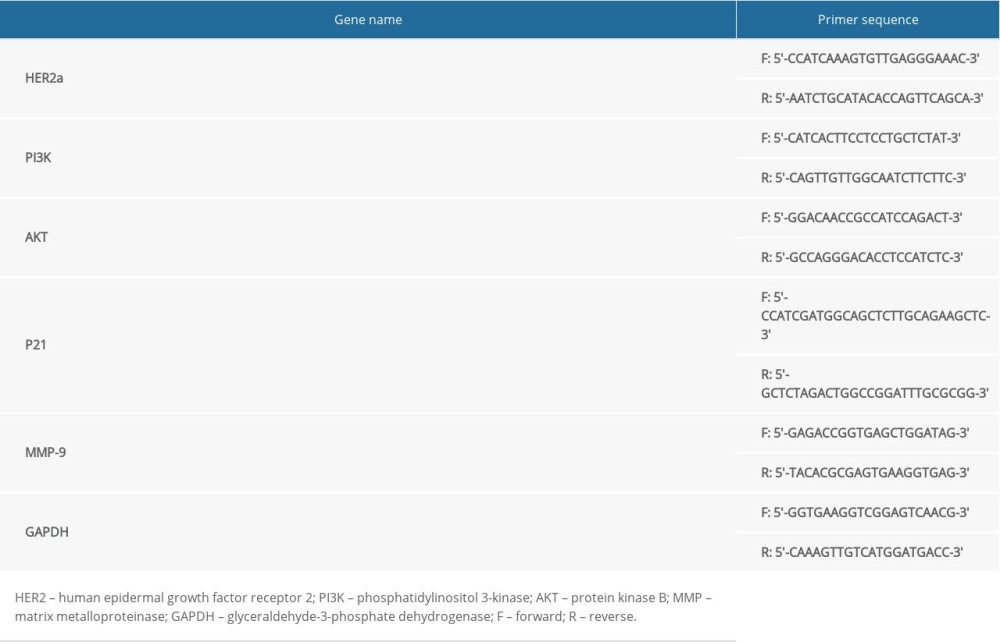
Figure 1 shows that HER2 mRNA expression (A, by RT-qPCR assay) and protein expression (B, by WB assay) in NCI-N87 and OE-19 cells were highest of all the gastric cancer cell lines tested (AGS, NCI-N87, MGC-803, SGC-7901, and OE-19). Because of these results, we used NCI-N87 and OE-19 cells in our study.
HON AFFECTED HER2-POSITIVE CELL PROLIFERATION AND APOPTOSIS:
By CCK-8 assay, the cell viabilities of Hon-treated groups were significantly depressed in NCI-N87 and OE-19 cell lines, with dose dependence at 12, 24, and 48 h (P<0.05, Figure 2A). By flow cytometry, the cell apoptosis rate of Hon-treated groups was significantly increased in NCI-N87 and OE-19 cell lines and was dose dependent (P<0.05, Figure 2B). Thus Hon had dose-dependent suppressive effects on NCI-N87 and OE-19 cell proliferation, with cell apoptosis increasing.
HON AFFECTED CELL CYCLE IN NCI-N87 AND OE-19 CELL LINES BY FLOW CYTOMETRY:
By flow cytometry, Figure 3 shows that G1 phase rates of Hon-treated groups were significantly upregulated and G2 phase rates were significantly downregulated compared with those of the control group, with dose dependence (P<0.05), in NCI-N87 and OE-19 cell lines. These results show that Hon kept the cell cycle in G1 phase, which might be the cause of the cell apoptosis rate increase.
HON AFFECTS CELL INVASION AND WOUND-HEALING RATES IN NCI-N87 AND OE-19 CELL LINES:
By transwell assay, compared with the control group, the number of invading cells of the Hon-treated groups was significantly depressed with dose dependence in NCI-N87 and OE-19 cell lines (P<0.05, Figure 4A, 4B); By wound-healing assay, the wound healing rate of Hon-treated groups was significantly suppressed compared with that of the control group in NCI-N87 and OE-19 cell lines at 24 and 48 h (P<0.05, Figure 4C, 4D). These results show that Hon could suppress cell invasion and migration abilities in NCI-N87 and OE-19 cell lines in a dose-dependent manner.
HON AFFECTED RELATIVE PROTEIN AND MRNA EXPRESSION IN NCI-N87 AND OE-19 CELL LINES:
By WB assay, the PI3K, AKT, and MMP-9 protein and mRNA expression of Hon-treated groups were significantly suppressed compared with those of the control group in a dose-dependent manner in NCI-N87 and OE-19 cell lines (P<0.05, Figure 5A–5D); and P21 protein and mRNA expressions of Hon-treated groups were significantly increased compared with those of the control group with dose dependence in these cell lines (P<0.05, Figure 5A–5D). The results show that Hon could depress PI3K, AKT, and MMP-6 proteins and gene expression, which might be correlated with cell invasion and migration ability, and increase P21 protein and gene expression, which might be correlated with keeping the cell cycle in G1 phase, leading to an increase in cell apoptosis.
HER2 EXPRESSION IN DIFFERENT GROUPS BY CELLULAR IMMUNOFLUORESCENCE:
By cellular immunofluorescence, the HER2 expression of Hon-treated groups was significantly depressed compared with that of the control group in NCI-N87 and OE-19 cell lines, with dose dependence (P<0.05, Figure 6A, 6B). The results of immunofluorescence show that Hon affects antitumor activity by inhibiting Her2 nuclear protein.
Discussion
With continuous research on the pathogenesis of gastric cancer, molecularly targeted therapy is important for treatment, especially for patients with intolerance to surgery or with metastatic gastric cancer. At present, systemic chemotherapy is still the main treatment for these patients. However, there is no standardized treatment for advanced gastric cancer, and the effective rate of systemic chemotherapy is unsatisfactory. Therefore, targeted therapy for growth, proliferation, apoptosis, and metastasis of tumor cells has become an important research direction. Kolyadina et al. [20] found that the occurrence and metastasis of many tumors were related to cell overproliferation caused by overexpression of specific oncogenes or receptor genes. Usually, HER2 protein is expressed only in the fetal period, and in a few tissues at a low level or not expressed in adulthood, but it is overexpressed in many tumor tissues. It has been confirmed that 12–35% of gastric cancers have HER2 overexpression, and HER2 is an independent prognostic factor of gastric cancer [21]. The results of Roy et al. [22] showed that HER2 protein expression in patients with gastric cancer combined with lymph node metastasis was significantly higher than that in those without lymph node metastasis; HER2 protein expression in patients with infiltration reaching beyond the serosa was significantly higher than in those with infiltration within the serosa, and HER2 protein in lymph node metastasis of gastric cancer was highly expressed. Patients with HER2-positive tumors had poorer prognosis and lower 5-year survival rate. Age, TNM stage, and high HER2 expression were all independent factors affecting the survival of the patients. In other words, more studies suggest that HER2 expression is related to the prognosis of gastric cancer, can promote the occurrence and development of tumors, and can be used as an independent factor to evaluate the prognosis of gastric cancer.
The HER2 gene is located on human chromosome 17q21 and encodes a transmembrane protein with a molecular weight of 185 kDa. It has tyrosine protein kinase activity and belongs to the epidermal growth factor receptor (EGFR) family. HER2 is always expressed during embryonic development and plays an important role in the growth and proliferation of normal cells. However, multiple studies have shown that overexpression of HER2 protein can promote the occurrence and development of tumors [23]. HER2 and PI3K are closely related to each other. Usually, they are highly expressed in malignant epithelial tumor cells, and are related to tumor invasion, treatment effect, and survival. They are important indicators of poor prognosis. Although HER2 and PI3K play different roles in the occurrence, development, and metastasis of tumors, there is much evidence that the HER2 and PI3K signaling pathways interact [24]. Basic experimental studies have demonstrated that the activation of the PI3K signaling pathway is caused by HER2/neu activation in various tumors [24].
Currently, the main chemotherapy drugs used in treatment are cytotoxic; thus it is urgent to explore low-toxicity and high-efficiency drugs. It has been found that Hon can inhibit the biological activities of tumor cells [25–27]. Related
Cyclin-dependent kinase (CDK) is a key molecule in the cell cycle. Its overactivation promotes cells to copy deoxyribonucleic acid prematurely, which leads to genome instability and tumorigenesis [29]. Cip-Kip is one of the families of cell cycle inhibitors including P21, P27, and P57. They can bind to various CDK-cell cycle complexes and thereby inhibit their roles [30]. Studies have revealed that low P21 level is always correlated with poor prognosis of tumors [31,32]. In this study, after Hon intervention, P21 protein and gene expression increased significantly, resulting in a large number of cells staying in G1 phase, which led to a significant increase in apoptosis.
MMP-9 is one of the most widely investigated MMPs and plays an important role in the invasion and metastasis of cancer cells. Human MMP-9 gene is located on chromosome 20q13.12 [33]. Related studies have shown that [34,35] MMP-9 overexpression is closely related to tumor invasion and migration. In our study, with Hon intervention, the invasion and migration of HER2-positive gastric cancer cells were significantly inhibited, and MMP-9 protein and mRNA expression were also significantly inhibited. Therefore, we infer that the inhibitory effect of Hon on the invasion and migration of HER2-positive gastric cancer cells may be related to the decrease in MMP-9 expression.
In recent years, nanobiology has attracted more and more attention [36,37]. Hon combined with nanomaterials to treat gastric cancer is a new research idea for the future.
Conclusions
In conclusion, Hon can inhibit the biological activity of HER2-positive gastric cancer cells, and its mechanism may be related to Hon inhibiting HER2 expression and thereby inhibiting the overactivation of the PI3K/AKT signal pathway. However, the specific downstream mechanism is unclear, and still needs further study.
Figures
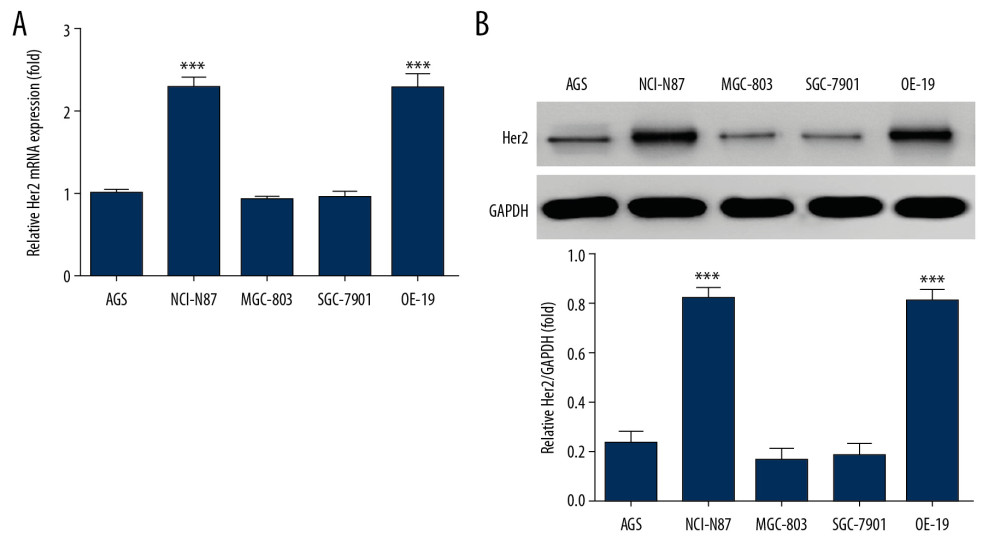 Figure 1. Human epidermal growth factor receptor 2 (HER2) messenger ribonucleic acid (mRNA) and protein expression in different cell lines (A) by real-time quantitative polymerase chain reaction (RT-qPCR) assay and (B) by western blot (WB) assay. *** P<0.001, compared with AGS cell line.
Figure 1. Human epidermal growth factor receptor 2 (HER2) messenger ribonucleic acid (mRNA) and protein expression in different cell lines (A) by real-time quantitative polymerase chain reaction (RT-qPCR) assay and (B) by western blot (WB) assay. *** P<0.001, compared with AGS cell line. 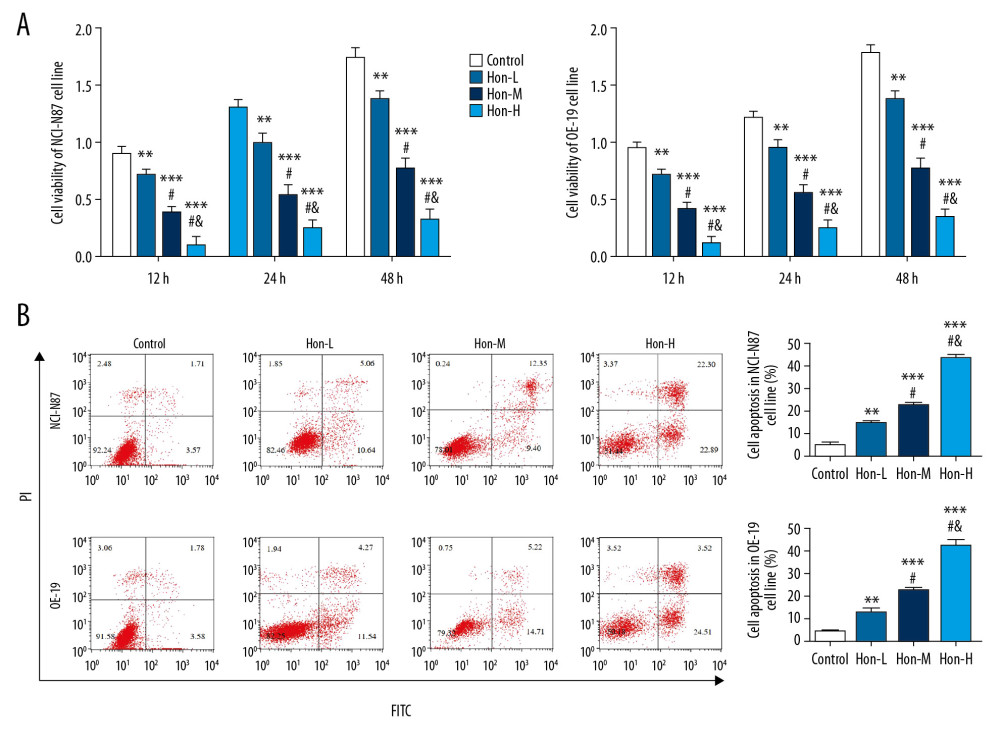 Figure 2. Honokiol (Hon) affected HER2-positive cell proliferation and apoptosis. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. (A) The cell viability of different cell lines at different time points by cell-counting kit 8 (CCK-8) assay. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. (B) Cell apoptosis rate of different cell lines by flow cytometry. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 2. Honokiol (Hon) affected HER2-positive cell proliferation and apoptosis. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. (A) The cell viability of different cell lines at different time points by cell-counting kit 8 (CCK-8) assay. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. (B) Cell apoptosis rate of different cell lines by flow cytometry. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.  Figure 3. Honokiol (Hon) affected cell cycle in NCI-N87 and OE-19 cell lines by flow cytometry. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Cell cycle of different groups in NCI-N87 cell line (A) and in OE-19 cell line (B) by flow cytometry. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 3. Honokiol (Hon) affected cell cycle in NCI-N87 and OE-19 cell lines by flow cytometry. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Cell cycle of different groups in NCI-N87 cell line (A) and in OE-19 cell line (B) by flow cytometry. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. 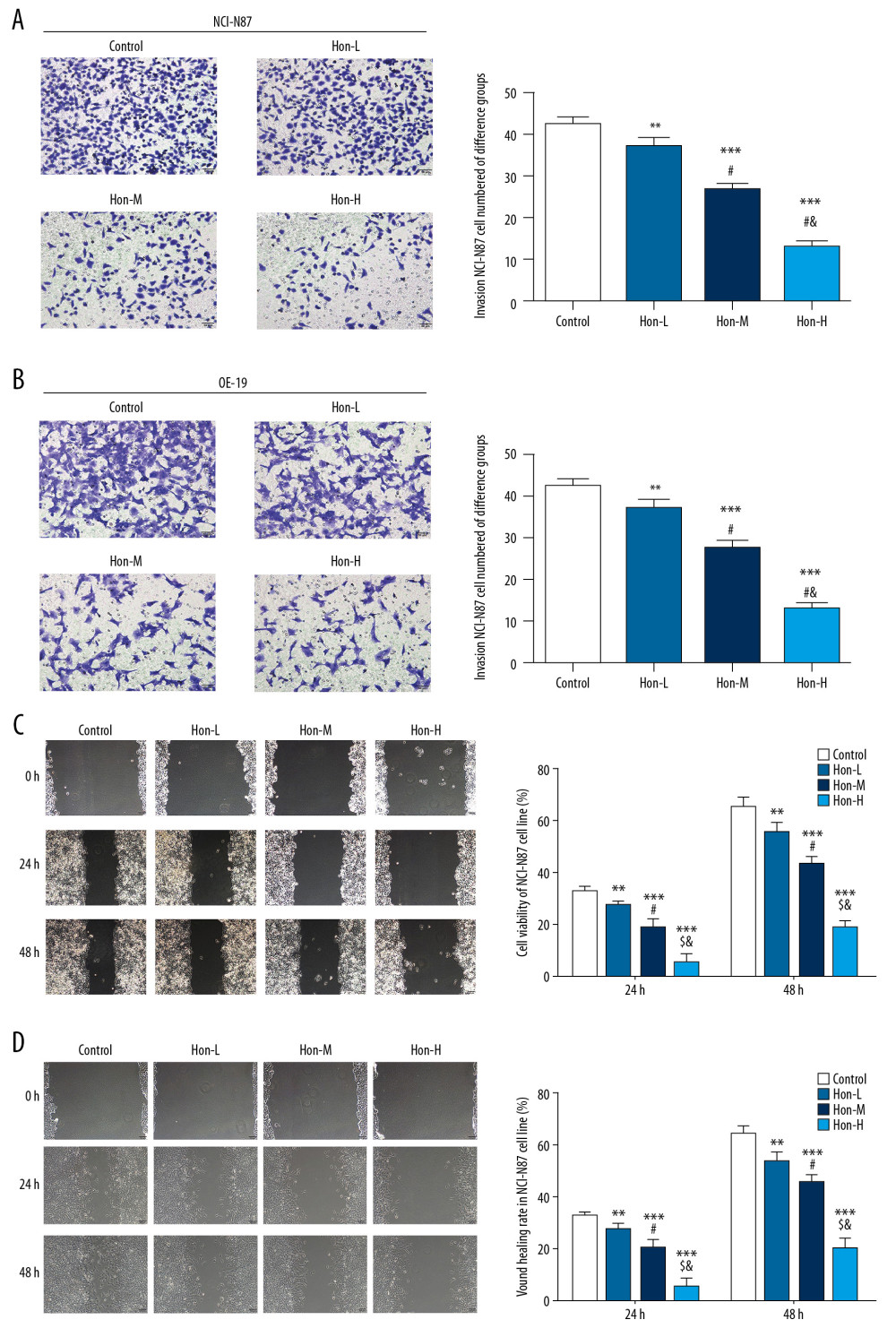 Figure 4. Honokiol (Hon) affects cell invasion and wound-healing rate in NCI-N87 and OE-19 cell lines. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Invading cell numbers of different groups in NCI-N87 cell line (A) and in OE-19 cell line (B) by transwell assay (200×). Wound-healing rate of different groups in NCI-N87 cell line (C) and in OE-19 cell line (D) by wound-healing assay (100×). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 4. Honokiol (Hon) affects cell invasion and wound-healing rate in NCI-N87 and OE-19 cell lines. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Invading cell numbers of different groups in NCI-N87 cell line (A) and in OE-19 cell line (B) by transwell assay (200×). Wound-healing rate of different groups in NCI-N87 cell line (C) and in OE-19 cell line (D) by wound-healing assay (100×). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. 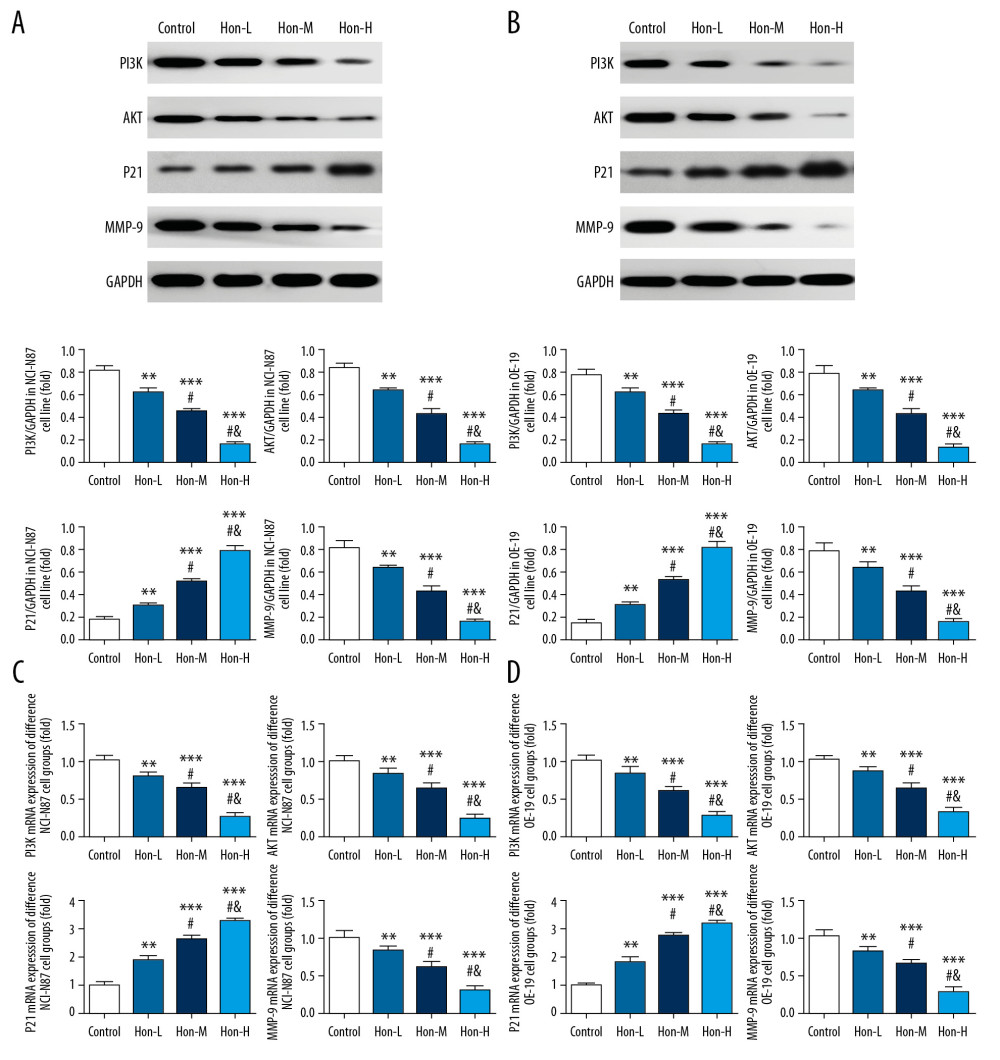 Figure 5. Relative protein and messenger ribonucleic acid (mRNA) expression by western blot (WB) and real-time quantitative polymerase chain reaction (RT-qPCR) assay. Control: Untreated cells. Hon-L: Cells treated with low-dose honokiol. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Relative protein expression by WB assay in NCI-N87 cell line (A) and in OE-19 cell line (B). Relative mRNA expression by RT-qPCR assay in NCI-N87 cell line (C) and in OE-19 cell line (D). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 5. Relative protein and messenger ribonucleic acid (mRNA) expression by western blot (WB) and real-time quantitative polymerase chain reaction (RT-qPCR) assay. Control: Untreated cells. Hon-L: Cells treated with low-dose honokiol. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Relative protein expression by WB assay in NCI-N87 cell line (A) and in OE-19 cell line (B). Relative mRNA expression by RT-qPCR assay in NCI-N87 cell line (C) and in OE-19 cell line (D). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. 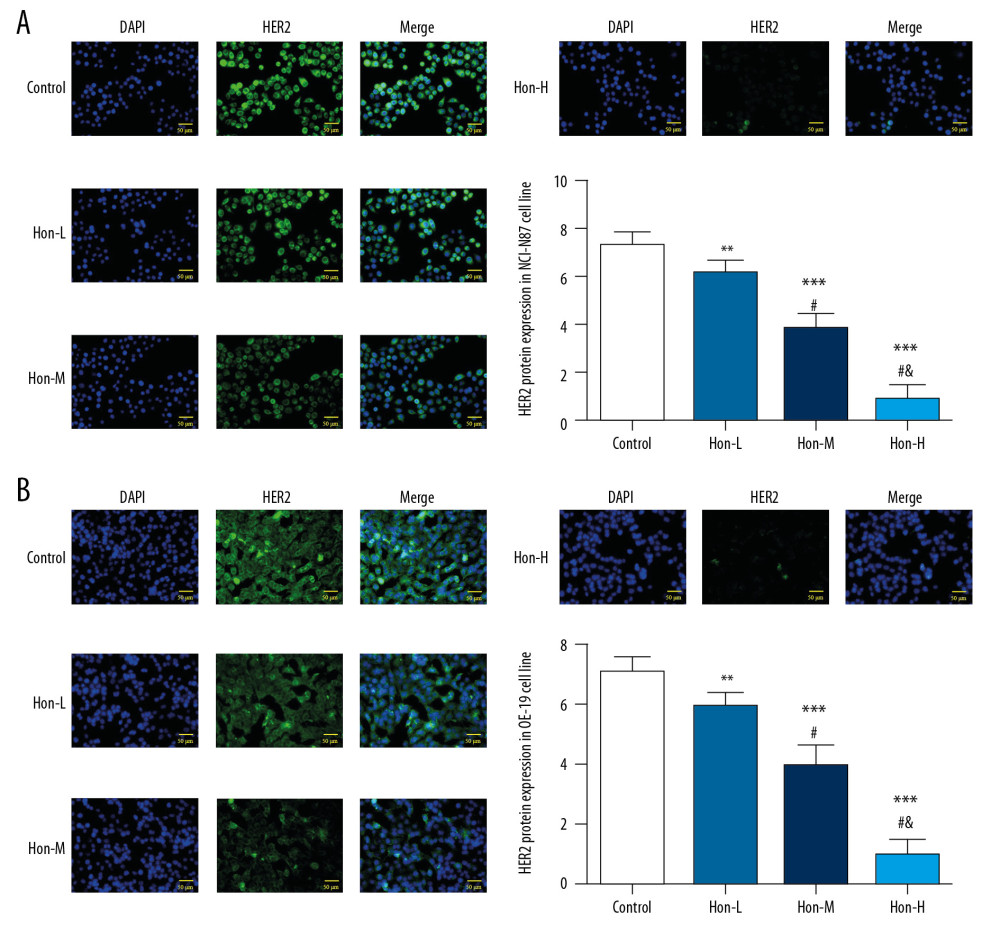 Figure 6. Human epidermal growth factor receptor 2 (HER2) expression of different groups by cellular immunofluorescence. Control: Untreated cells. Hon-L: Cells treated with low-dose honokiol. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. HER2 expression of different groups by cellular immunofluorescence in NCI-N87 cell line (A) and in OE-19 cell line (B). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 6. Human epidermal growth factor receptor 2 (HER2) expression of different groups by cellular immunofluorescence. Control: Untreated cells. Hon-L: Cells treated with low-dose honokiol. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. HER2 expression of different groups by cellular immunofluorescence in NCI-N87 cell line (A) and in OE-19 cell line (B). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. References
1. Schild SE, Vokes EE, Pathways to improving combined modality therapy for stage III nonsmall-cell lung cancer: Ann Oncol, 2016; 27(4); 590-99
2. Ishida H, Lam AK, Pancreatic neuroendocrine neoplasms: The latest surgical and medical treatment strategies based on the current World Health Organization classification: Crit Rev Oncol Hematol, 2019; 145; 102835
3. Cai Z, Yin Y, Zhao Z, Comparative effectiveness of neoadjuvant treatments for resectable gastroesophageal cancer: A network meta-analysis: Front Pharmacol, 2018; 9; 872
4. Fitzgerald TL, Efird JT, Bellamy N, Perioperative chemotherapy versus postoperative chemoradiotherapy in patients with resectable gastric/gastroesophageal junction adenocarcinomas: A survival analysis of 5058 patients: Cancer, 2017; 123(15); 2909-17
5. Bringeland EA, Wasmuth HH, Grønbech JE, Perioperative chemotherapy for resectable gastric cancer – What is the evidence?: Scand J Gastroenterol, 2017; 52(6–7); 647-53
6. Smalley SR, Benedetti JK, Haller DG, Updated analysis of SWOG-directed intergroup study 0116: A phase III trial of adjuvant radiochemotherapy versus observation after curative gastric cancer resection: J Clin Oncol, 2012; 30(19); 2327-33
7. Holowatyj AN, Ulrich CM, Lewis MA, Racial/ethnic patterns of young-onset noncardia gastric cancer: Cancer Prev Res (Phila), 2019; 12(11); 771-80
8. Gao J, Ren W, Xiao C, Involvement of SLC39A6 in gastric adenocarcinoma and correlation of the SLC39A6 polymorphism rs1050631 with clinical outcomes after resection: BMC Cancer, 2019; 19(1); 1069
9. Nakata S, Fujita M, Nakanishi H, Efficacy of afatinib and lapatinib against HER2 gene-amplified trastuzumab-sensitive and -resistant human gastric cancer cells: Anticancer Res, 2019; 39(11); 5927-32
10. Nam AR, Kim JW, Cha Y, Therapeutic implication of HER2 in advanced biliary tract cancer: Oncotarget, 2016; 7(36); 58007-21
11. Aghazadeh S, Yazdanparast R, Mycophenolic acid potentiates HER2-overexpressing SKBR3 breast cancer cell line to induce apoptosis: Involvement of AKT/FOXO1 and JAK2/STAT3 pathways: Apoptosis, 2016; 21(11); 1302-14
12. Liu Y, Majumder S, McCall W, Inhibition of HER-2/neu kinase impairs androgen receptor recruitment to the androgen responsive enhancer: Cancer Res, 2005; 65(8); 3404-9
13. Shepard HM, Brdlik CM, Schreiber H, Signal integration: A framework for understanding the efficacy of therapeutics targeting the human EGFR family: J Clin Invest, 2008; 118(11); 3574-81
14. Kim YJ, Jung UJ, Honokiol improves insulin resistance, hepatic steatosis, and inflammation in type 2 diabetic db/db mice: Int J Mol Sci, 2019; 20(9); E2303
15. Khalid S, Khan A, Shal B: Biomed Pharmacother, 2019; 114; 108777
16. Hong T, Min H, Hui Z, Oral administration of honokiol attenuates airway inflammation in asthmatic mouse model: Pak J Pharm Sci, 2018; 31(4); 1279-84
17. Hsiao CH, Yao CJ, Lai GM, Honokiol induces apoptotic cell death by oxidative burst and mitochondrial hyperpolarization of bladder cancer cells: Exp Ther Med, 2019; 17(5); 4213-22
18. Wang X, Cheng L, Xie HJ, Functional paclitaxel plus honokiol micelles destroying tumour metastasis in treatment of non-small-cell lung cancer: Artif Cells Nanomed Biotechnol, 2018; 46(Suppl 2); 1154-69
19. Chou HC, Lu CH, Su YC, Proteomic analysis of honokiol-induced cytotoxicity in thyroid cancer cells: Life Sci, 2018; 207; 184-204
20. Kolyadina IV, Zavalishina LE, Ganshina IP, Role of clustered HER2/neu amplification as a marker for a special sensitivity to neoadjuvant anti-HER2 therapy with trastuzumab in patients with stage II–III breast cancer: Arkh Patol, 2019; 81(6); 56-62
21. Wang H, Wang Y, Xiao Z: Antibodies (Basel), 2019; 8(1); E25
22. Roy PS, Nyodu T, Hazarika M, Prevalence of HER2 expression and its correlation with clinicopathological parameters in gastric or gastroesophageal junction adenocarcinoma in North-East Indian population: Asian Pac J Cancer Prev, 2019; 20(4); 1139-45
23. Tokumaru Y, Tajirika T, Sugito N, Synthetic miR-143 inhibits growth of HER2-positive gastric cancer cells by suppressing KRAS networks including DDX6 RNA helicase: Int J Mol Sci, 2019; 20(7); E1697
24. Díaz-Serrano A, Angulo B, Dominguez C, Genomic profiling of HER2-positive gastric cancer: PI3K/Akt/mTOR pathway as predictor of outcomes in HER2-positive advanced gastric cancer treated with trastuzumab: Oncologist, 2018; 23(9); 1092-102
25. Liu RX, Ren WY, Ma Y, BMP7 mediates the anticancer effect of honokiol by upregulating p53 in HCT116 cells: Int J Oncol, 2017; 51(3); 907-17
26. Wynn ML, Consul N, Merajver SD, Schnell S, Inferring the effects of honokiol on the notch signaling pathway in SW480 colon cancer cells: Cancer Inform, 2014; 13(Suppl 5); 1-12
27. Hua H, Chen W, Shen L, Honokiol augments the anti-cancer effects of oxaliplatin in colon cancer cells: Acta Biochim Biophys Sin (Shanghai), 2013; 45(9); 773-79
28. Chen F, Wang T, Wu YF, Honokiol: A potent chemotherapy candidate for human colorectal carcinoma: World J Gastroenterol, 2004; 10(23); 3459-63
29. Sato N, Yamabuki T, Takano A, Wnt inhibitor Dickkopf-1 as a target for passive cancer immunotherapy: Cancer Res, 2010; 70(13); 5326-36
30. Wang K, Zhang Y, Li X, Characterization of the Kremen binding site on Dkkl and elucidation of the role of Kremen in Dkk mediated wnt antagonism: J Biol Chem, 2008; 283(34); 23371-75
31. Vandooren J, Van den Steen PE, Opdenakker G, Biochemistry and molecular biology of gelatinase B or matrix metalloproteinase 9 (MMP-9): The next decade: Crit Rev Biochem Mol Biol, 2013; 48(3); 222-72
32. Van den Steen PE, Dubois B, Nelissen I, Biochemistry and molecular biology of gelatinase B or matrix metalloproteinase-9 (MMP-9): Crit Rev Biochem Mol Biol, 2002; 37(6); 375-36
33. Huang H, Matrix metalloproteinase-9 (MMP-9) as a cancer biomarker and MMP-9 biosensors: Recent advances: Sensors (Basel), 2018; 18(10); E3249
34. Song Z, Wang J, Su Q, The role of MMP-2 and MMP-9 in the metastasis and development of hypopharyngeal carcinoma: Braz J Otorhinolaryngol, 2019 [Online ahead of print]
35. Huang L, Lin H, Chen Q, MPPa-PDT suppresses breast tumor migration/invasion by inhibiting Akt-NF-κB-dependent MMP-9 expression via ROS: BMC Cancer, 2019; 19(1); 1159
36. Yang Z, Wang J, Ai S, Self-generating oxygen enhanced mitochondrion-targeted photodynamic therapy for tumor treatment with hypoxia scavenging: Theranostics, 2019; 9(23); 6809-23
37. Zhengyang Y, Jiafeng W, Song L, Tumor-targeting W18O49 nanoparticles for dual-modality imaging and guided heat-shock-response-inhibited photothermal therapy in gastric cancer: Part Part Syst Charact, 2019; 36 1900124
Figures
 Figure 1. Human epidermal growth factor receptor 2 (HER2) messenger ribonucleic acid (mRNA) and protein expression in different cell lines (A) by real-time quantitative polymerase chain reaction (RT-qPCR) assay and (B) by western blot (WB) assay. *** P<0.001, compared with AGS cell line.
Figure 1. Human epidermal growth factor receptor 2 (HER2) messenger ribonucleic acid (mRNA) and protein expression in different cell lines (A) by real-time quantitative polymerase chain reaction (RT-qPCR) assay and (B) by western blot (WB) assay. *** P<0.001, compared with AGS cell line. Figure 2. Honokiol (Hon) affected HER2-positive cell proliferation and apoptosis. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. (A) The cell viability of different cell lines at different time points by cell-counting kit 8 (CCK-8) assay. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. (B) Cell apoptosis rate of different cell lines by flow cytometry. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 2. Honokiol (Hon) affected HER2-positive cell proliferation and apoptosis. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. (A) The cell viability of different cell lines at different time points by cell-counting kit 8 (CCK-8) assay. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. (B) Cell apoptosis rate of different cell lines by flow cytometry. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. Figure 3. Honokiol (Hon) affected cell cycle in NCI-N87 and OE-19 cell lines by flow cytometry. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Cell cycle of different groups in NCI-N87 cell line (A) and in OE-19 cell line (B) by flow cytometry. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 3. Honokiol (Hon) affected cell cycle in NCI-N87 and OE-19 cell lines by flow cytometry. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Cell cycle of different groups in NCI-N87 cell line (A) and in OE-19 cell line (B) by flow cytometry. ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. Figure 4. Honokiol (Hon) affects cell invasion and wound-healing rate in NCI-N87 and OE-19 cell lines. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Invading cell numbers of different groups in NCI-N87 cell line (A) and in OE-19 cell line (B) by transwell assay (200×). Wound-healing rate of different groups in NCI-N87 cell line (C) and in OE-19 cell line (D) by wound-healing assay (100×). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 4. Honokiol (Hon) affects cell invasion and wound-healing rate in NCI-N87 and OE-19 cell lines. Control: Untreated cells. Hon-L: Cells treated with low-dose Hon. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Invading cell numbers of different groups in NCI-N87 cell line (A) and in OE-19 cell line (B) by transwell assay (200×). Wound-healing rate of different groups in NCI-N87 cell line (C) and in OE-19 cell line (D) by wound-healing assay (100×). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. Figure 5. Relative protein and messenger ribonucleic acid (mRNA) expression by western blot (WB) and real-time quantitative polymerase chain reaction (RT-qPCR) assay. Control: Untreated cells. Hon-L: Cells treated with low-dose honokiol. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Relative protein expression by WB assay in NCI-N87 cell line (A) and in OE-19 cell line (B). Relative mRNA expression by RT-qPCR assay in NCI-N87 cell line (C) and in OE-19 cell line (D). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 5. Relative protein and messenger ribonucleic acid (mRNA) expression by western blot (WB) and real-time quantitative polymerase chain reaction (RT-qPCR) assay. Control: Untreated cells. Hon-L: Cells treated with low-dose honokiol. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. Relative protein expression by WB assay in NCI-N87 cell line (A) and in OE-19 cell line (B). Relative mRNA expression by RT-qPCR assay in NCI-N87 cell line (C) and in OE-19 cell line (D). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. Figure 6. Human epidermal growth factor receptor 2 (HER2) expression of different groups by cellular immunofluorescence. Control: Untreated cells. Hon-L: Cells treated with low-dose honokiol. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. HER2 expression of different groups by cellular immunofluorescence in NCI-N87 cell line (A) and in OE-19 cell line (B). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group.
Figure 6. Human epidermal growth factor receptor 2 (HER2) expression of different groups by cellular immunofluorescence. Control: Untreated cells. Hon-L: Cells treated with low-dose honokiol. Hon-M: Cells treated with middle-dose Hon. Hon-H: Cells treated with high-dose Hon. HER2 expression of different groups by cellular immunofluorescence in NCI-N87 cell line (A) and in OE-19 cell line (B). ** P<0.01, *** P<0.001 compared with control group; # P<0.05 compared with Hon-L group; & P<0.05 compared with Hon-M group. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387