30 July 2020: Meta-Analysis
Outcomes of VA-ECMO with and without Left Centricular (LV) Decompression Using Intra-Aortic Balloon Pumping (IABP) versus Other LV Decompression Techniques: A Systematic Review and Meta-Analysis
Pan PanABCE, Peng YanB, Dawei LiuD, Xiaoting WangD, Xiang ZhouD, Yun LongD, Kun XiaoC, Weiguo ZhaoD, Lixin XieA, Longxiang SuACEDOI: 10.12659/MSM.924009
Med Sci Monit 2020; 26:e924009
Abstract
BACKGROUND: Left ventricular decompression is the primary method for solving VA-ECMO-induced LV afterload increase, but the effect of specific methods on patient outcomes and complications is unknown.
MATERIAL AND METHODS: We searched for all published reports conducted in patients undergoing ECMO combined with LVD. Statistical analyses were performed using Stata 12.0.
RESULTS: The results showed that the risk of death with ECMO combined with LVD was 29% lower than that with ECMO alone (OR=0.71, 95% CI: 0.56–0.89, I²=59.5%, P<0.001). Although the risk of death with ECMO combined other LV decompression techniques was higher than that with ECMO combined with IABP, the difference was not statistically significant (OR=1.27, 95% CI: 0.86–1.87, I²=44.0%, P=0.057). In addition, the ORs values of hemorrhage, stroke/acute episodes, lower-limb ischemia, and hemolysis for ECMO combined with LVD were 0.69 (0.66–0.71), 0.82 (0.78–0.89), 0.71 (0.30–1.66), and 0.48 (0.16–1.39), respectively. The risk of complications, such as stroke/TIA, limb ischemia, and hemolysis, of ECMO combined with IABP was lower than that of ECMO combined other LV decompression techniques, and the risk of bleeding was higher for ECMO combined with IABP.
CONCLUSIONS: ECMO combined with LVD is more beneficial than using ECMO alone and helps to lower patient mortality.
Keywords: Extracorporeal Membrane Oxygenation, Intra-Aortic Balloon Pumping, Mortality, Postoperative Complications, Ventricular Outflow Obstruction, Heart Ventricles, Heart-Assist Devices, Models, Statistical, Risk Factors, Shock, Cardiogenic, Ventricular Dysfunction, Left
Background
Cardiogenic shock is a clinical syndrome resulting from left, right, or biventricular dysfunction that eventually leads to circulatory failure and multiple organ dysfunction [1]. Acute myocardial infarction (AMI), especially ST-elevation myocardial infarction (STEMI), is the most common cause of cardiogenic shock. In addition, the causes of non-acute coronary syndrome can also result in cardiogenic shock [2]. Cardiogenic shock is now considered an acute and lethal disorder in the ICU. The main presentation of cardiogenic shock is hemodynamic instability, and it can even progress to refractory shock, which is associated with a mortality rate of approximately 50% [3].
In cardiogenic shock, ventricular abnormalities mainly manifest as irreversible contraction and diastolic dysfunction, leading to reduced cardiac output, increased ventricular diastolic pressure, and decreased coronary perfusion pressure. Left ventricular (LV) dysfunction and ischemia are the most common complications, elevating left atrial pressure, leading to pulmonary edema, hypoxia, pulmonary blood vessel convulsion, and worsening ischemia [4,5]. Fluids, vasopressors, and inotropes are usually used to assure cardiac output and oxygen delivery. However, the effects of treatment are often not ideal because escalating doses of vasopressors and inotropes are related to higher mortality [6,7]. Thus, mechanical circulatory support may improve the management of refractory cardiogenic shock.
Currently, the most popular and useful method for restoring cardiac function and improving cardiac output is extracorporeal membrane oxygenation (ECMO). Veno-arterial ECMO (VA-ECMO) provides cardiopulmonary support for patients whose heart and lungs are unable to survive. It is also a bridge to myocardial recovery or heart transplantation in cases of refractory cardiogenic shock [8]. Currently, VA-ECMO is widely recognized as a first-line treatment in the clinic. However, from the clinical perspective, ECMO does not improve cardiac function or promote cardiac output as much as expected. In fact, ECMO can only help cardiogenic shock patients achieve LV function but cannot improve LV function during left ventricular (LV) failure. However, VA-ECMO often increases left ventricular afterload and increases the stress on an already dysfunctional left ventricle [9,10]. This phenomenon results in retrograde aortic flow that can increase left ventricular end-diastolic pressure (LVEDP), which leads to severe pulmonary edema and increased wall stress and myocardial oxygen consumption, ultimately impairing myocardial recovery and increasing mortality. As ECMO appears to be the most appropriate way to replace cardiac function, determining how to unload the left ventricle during ECMO administration is essential.
In a recent study, researchers found that left ventricular decompression, such as ECMO combined IABP and ECMO combined other LV decompression techniques (like ventricular assist device, surgical atrial septostomy, or left ventricular drainage), might be useful in decreasing LV afterload [11,12]. The combination of ECMO and left ventricular decompression (LVD) may be a potential treatment for patients on ECMO who have LV dysfunction. However, studies have shown wide variations in clinical results regarding IABP and surgical methods during ECMO [13,14]. The combination of ECMO and LVD has been reviewed, but, to date, no study has been conducted to determine which method is best for reducing LV afterload. Therefore, we performed this systematic review and meta-analysis to assess the methodology of IABP and other left ventricular decompression methods during VA-ECMO and to evaluate the efficacy, feasibility, and safety of the combination of these methodologies.
Material and Methods
SEARCH STRATEGY AND SELECTION CRITERIA:
We performed a systematic review of published randomized controlled trials (RCTs), quasi-RCTs, and other comparative studies performed in patients undergoing ECMO plus left ventricular decompression. We defined left ventricular decompression as mechanical support including intra-aortic balloon pumping (IABP) and other left ventricular decompression methods, including left ventricular assist devices (VADs) and surgical methods such as surgical atrial septostomy and left ventricular drainage.
We searched the following medical bibliographical databases: PubMed, EMBASE, Web of Knowledge, and the Cochrane Library. We also searched trial registries for ongoing trials. We used text words and MeSH headings containing “VA-extracorporeal membrane oxygenation”, “mechanical support”, “intra-aortic balloon pumping”, “left ventricular assist devices”, “atrial septostomy”, “left ventricular drainage”, “cardiogenic shock”, “left ventricular distension”, “left ventricular vent”, and “left ventricular unloading” in the search. The PubMed search strategy is presented in Figure 1.
Reviews, commentaries, letters, correspondences, conference abstracts, case reports, expert opinions, editorials, and animal experiments were excluded. Articles involving pediatric patients were excluded. Three investigators (PP, PY, and KX) independently performed the search and selection of the articles. Any disagreement was resolved by a third party (DL, XZ, XW, LY, and WZ). The date range for the search was from the date of the first references available to May 31, 2019.
DATA EXTRACTION AND GROUP COMPARISONS:
The following data were extracted for each trial: the author, year of publication, study type, study population and number, technical parameters, indicators of ECMO, main outcomes, and complications. The primary outcome was hospital mortality, and the secondary outcome was the incidence of complications. We tried to contact the corresponding author(s) if the data were not presented or needed clarification.
When studying left heart decompression, we first divided the studies into 2 groups: the ECMO alone group and ECMO combined with the left ventricular decompression group. The left ventricular decompression group included studies on mechanical assistance (IABP and VAD) and assisted surgery (surgical atrial septostomy and left ventricular drainage). In addition, because there were many studies on IABP and fewer articles on VAD and other assisted surgery, we compared the mortality rate and complications of the ECMO+IABP group with that of the VAD and ECMO-assisted surgery together, defined as the ECMO combined other LV decompression techniques group.
STATISTICAL AND META-ANALYSIS:
The relative risk (RR) and its confidence interval were calculated by extracting positive and negative numbers from the 2 groups. The meta-analysis results in this study are represented by forest maps. The heterogeneity test between different studies was evaluated by I2 statistics. When
Results
Systematic review
THE RISK OF DEATH WITH ECMO COMBINED WITH LEFT VENTRICULAR DECOMPRESSION THERAPY COMPARED WITH ECMO ALONE: A total of 23 studies were included in the meta-analysis. The results showed that the risk of death with ECMO combined with left ventricular decompression therapy was 29% lower than that with ECMO alone. This difference was statistically significant (OR=0.71, 95% CI: 0.56–0.89), but I2=59.5% (P<0.001) indicated that the studies exhibited strong heterogeneity, and further analysis is needed to find the source of heterogeneity (Figure 2).
SUBGROUP ANALYSIS FOR THE COMPARISON OF THE RISK OF DEATH WITH ECMO COMBINED WITH LEFT VENTRICULAR DECOMPRESSION THERAPY TO THAT WITH ECMO ALONE: Subgroup analysis was performed based on whether the sample size was greater than 50. The results showed that, in studies with a sample size smaller than 50, the risk of death with ECMO combined with left ventricular decompression therapy was 49% lower than that with ECMO alone. The difference was statistically significant (OR=0.51, 95% CI: 0.33–0.81, I2=0.0%, P=0.859), suggesting low heterogeneity. In studies with a sample size greater than 50, the risk of death with ECMO combined with left ventricular decompression therapy was still less than 22% and was lower than that with ECMO alone, but the difference was not statistically significant (OR=0.78, 95% CI: 0.61–1.01, I2=69.0%, P<0.001). This result indicated high heterogeneity (see Figure 3A), which may have been because the samples from study 36 and study 39 were much larger than those from the other studies, making the results unstable.
After study 36 and study 39 were excluded, studies with a sample size greater than 50 showed that the risk of death with ECMO combined with left ventricular decompression therapy was still lower than that with ECMO alone (OR=0.79, 95% CI: 0.64–0.99, I2=0.0%, P=0.607). The heterogeneity for the entire meta-analysis was small and acceptable (I2=0.0%, P=0.732), indicating that the heterogeneity was likely derived from the especially large sample sizes of studies 36 and 39 (see Figure 3B).
COMPARISON OF THE RISK OF DEATH WITH ECMO COMBINED WITH IABP TO THAT OF ECMO COMBINED OTHER LV DECOMPRESSION TECHNIQUES: To further analyze the effect of different methods of left ventricular decompression on mortality, a total of 11 articles were included in the meta-analysis for the risk of death with ECMO combined with IABP group compared to that of ECMO combined with other LV decompression techniques group (Table 2). The results showed that although the risk of death with ECMO combined other LV decompression techniques group was higher than that in the ECMO+IABP group, the difference was not statistically significant (OR=1.27, 95% CI: 0.86–1.87, I2= 44.0%, P=0.057), as shown in Figure 4.
Subgroup analysis based on different sample sizes (Figure 5A) showed that the risk of death in the ECMO combined other LV decompression techniques group was higher than that in the ECMO+IABP group in studies with a sample size smaller than 50, and the difference was statistically significant (OR=1.99, 95% CI: 1.00–3.98, I2=0.0%, P=0.444). In studies with a sample size greater than 50, although the risk of death in the ECMO combined other LV decompression techniques group was higher than that in the ECMO+IABP group, the difference was not statistically significant (OR=1.10, 95% CI: 0.70–1.71, I2=50.6%, P=0.059). This result indicates that there was no statistically significant difference in the risk of death between the 2 in the studies with large sample sizes. Therefore, large-sample cohort studies and clinical randomized controlled trials are needed to further validate the therapeutic effects of these 2 methods.
Subgroup analysis based on the different regions (Figure 5B) showed as that in Asia, the risk of death in the ECMO combined other LV decompression techniques group was higher than that in the ECMO+IABP group, but the difference was not statistically significant (OR=1.24, 95% CI: 0.61–2.52, I2=63.4%, P=0.042), suggesting that the results of the included studies from Asia were heterogeneous, probably due to the significant differences in sample sizes among these studies; similar results were found for Europe and North America. There was no significant difference in mortality between the 2 groups (Europe: OR=1.17, 95 CI%: 0.66–2.09, I2=38.5, P=0.164; North America: OR=2.18, 95% CI: 0.53–8.98, I2=44%, P=0.057), suggesting that the studies from Europe were less heterogeneous. The heterogeneity of the studies from North America was small, but only 2 articles were included.
COMPARISON OF THE RISK OF COMPLICATIONS AMONG ECMO COMBINED WITH LEFT VENTRICULAR DECOMPRESSION THERAPY VS. ECMO ALONE AND ECMO COMBINED WITH IABP VS. ECMO COMBINED WITH OTHER LV DECOMPRESSION TECHNIQUES: The results showed that the risk of 4 complications of ECMO combined with left ventricular decompression therapy was lower than that of ECMO alone. The ORs for hemorrhage, stroke/acute episodes, lower-limb ischemia, and hemolysis were 0.69 (0.66–0.71), 0.82 (0.78–0.89), 0.71 (0.30–1.66), and 0.48 (0.16–1.39), respectively, as shown in Figure 6A. In addition, the risk of stroke/transient ischemic attack (TIA), limb ischemia, and hemolysis complications in the ECMO combined with IABP group was lower than that of the ECMO combined other LV decompression techniques group, and the risk of bleeding in the ECMO combined with IABP group was higher than that in the ECMO combined other LV decompression techniques group, as shown in Figure 6B.
PUBLICATION BIAS AND SENSITIVITY ANALYSIS: Figures 7 and 8 show the Egger’s test and sensitivity analysis results for risk of death with ECMO combined with left ventricular decompression therapy compared to that with ECMO alone and the risk of death with ECMO combined other LV decompression techniques compared to that with ECMO combined with IABP. Figure 7A shows that there was no publication bias (P=0.175), and the funnel plot shows that the studies were evenly distributed on the bottom sides of the funnel. Sensitivity analysis showed that after the elimination of studies 36 and 39, the results were more stable, as shown in Figure 8A. The results of Begg’s test and Egger’s test, shown in Figure 7B, indicated that there was no publication bias, and the P values were 0.484 and 0.241, respectively. The funnel plots showed that the studies were evenly distributed on both sides of the funnel. Sensitivity analysis showed that the results were stable, as shown in Figure 8B.
Discussion
As we often see, central venous pressure (CVP) decreases significantly during ECMO in patients with severe LV dysfunction. However, the left atrial pressure remains high as the left ventricle contracts, and diastolic function is seriously impaired, resulting in excessive LV preload and LVEDP [15]. Elevating ECMO flow is not a good strategy for decreasing preload because a high blood flow rate can injure blood cells and the increase in retrograde perfusion flow further increases the LV afterload [9,16]. At this time, ECMO combined with decompression therapy such as IABP or ECMO combined other LV decompression techniques can effectively reduce LV afterload and balance the left and right heart filling. In this study, we performed a meta-analysis and found that the risk of death with ECMO combined with left ventricular decompression therapy was lower than that with ECMO alone. Through subgroup analysis, we found that a difference in sample sizes was the source of study heterogeneity. By dividing the studies into those with a sample size greater than 50 cases and those with a sample size smaller than 50 cases and excluding studies 36 and 39, we confirmed this conclusion.
The intra-aortic balloon pump (IABP), a counter-pulsation pump placed in the descending aorta, is the most commonly used assist device worldwide. The principle of IABP is inflation of the balloon in diastole and active deflation in systole, which induces higher diastolic perfusion pressure in the coronary arteries and unloads the diseased heart by reducing left ventricular afterload during systole [17]. According to its mechanistic features, IABP can neutralize some of the adverse effects of VA-ECMO; IABP can reduce the increased afterload of retrograde flow and increase myocardial oxygen supply by lowering myocardial oxygen consumption [13]. Furthermore, in some studies, IABP was found to increase coronary perfusion [17]. The counteracting effect of IABP helps ECMO better reduce the work of the heart and help the heart recover. Our statistical results show that ECMO combined with LVD is more beneficial than using ECMO alone and helps to lower patient mortality. Similar to our results, another team illustrated that ECMO and IABP have synergistic effects, play complementary roles in the treatment of acute cardiac failure, and can improve treatment outcomes [11], and IABP combined with VA-ECMO can also improve the success rate of weaning from VA-ECMO. However, the advantages of ECMO combined with IABP are largely based on theory and small-sample studies. When we conducted a meta-analysis, the heterogeneity of the results was mainly due to the number of samples and regional differences. Thus, in the future, we plan to carry out large-scale, multi-regional, and multi-center RCT studies to assess changes in hemodynamics and microcirculation with ECMO+IABP.
ECMO combined other LV decompression techniques is increasingly used in patients with left ventricular venting. In the ICU, we prefer a simple and rapid method of decreasing LV afterload. The most common method of LV decompression during ECMO is Impella. As a left ventricular assist device, Impella is a micro-axial pump that unloads the left ventricle and reduces left ventricular wall stress [18]. The left ventricular wall tension can be reduced by 80%, and myocardial oxygen demand is reduced by 40%; thus, it is an ideal treatment for correcting refractory heart failure and heart transplantation problems. In recent years, many experiments have proven that Impella combined with ECMO, termed ECMPELLA, can better replace cardiac function [19,20]. Besides Impella, other methods, such as intraoperative left atrial decompression, can be performed by placing a left ventricular drainage tube to improve heart function. In our meta-analysis, our results showed that patients with ECMO combined other LV decompression techniques exhibited lower mortality than those undergoing ECMO alone. Subgroup analysis indicated that, as a method for left ventricular assist, IABP seems to have more advantages than ECMO combined other LV decompression techniques. Although there was no significant difference between the ECMO+IABP group and the ECMO combined other LV decompression techniques group, subgroup analysis showed that the risk of death with ECMO combined other LV decompression techniques as higher than that with ECMO+IABP in studies with a sample size smaller than 50. In addition, high costs and risk of complications should be considered with the use of Impella [19]. Nevertheless, the results are based on retrospective data, and randomized controlled trials should be performed in the future.
When using ECMO and related assist devices, complications, which are decisive factors in the prognosis of patients, are the focus of attention. Many experiments have shown that the cause of death in refractory cardiac shock patients is not heart failure, but is instead incurable complications due to the use of mechanical support [21,22]. To our surprise, the incidence of complications in patients undergoing ECMO combined with assist devices was lower than that in patients undergoing ECMO alone. Similarly, another analysis also showed that VA-ECMO plus IABP is related to decreased in-hospital deaths of patients with extracorporeal cardiopulmonary resuscitation, postcardiotomy cardiogenic shock, and ischemic heart disease [23]. This does not mean that the use of an assist device increases complications. When we use various mechanical devices, we will be more vigilant and fully prepared to prevent or reduce the occurrence of bleeding, ischemia, and hemolysis. In other words, when regular treatments cannot support a patient’s life and mechanical devices are needed to replace heart function, complications are by no means an excuse for refusing to use the device.
The present study has several limitations. First, this was a meta-analysis that incorporated a series of related studies, including retrospective studies and prospective studies, from different regions and with different sample sizes, which may have been the source of heterogeneity and may have affected the final conclusions. Second, we compared ECMO+IABP and ECMO combined other LV decompression techniques. Due to the limited literature, we did not discuss the different methods of left ventricular decompression in more detail. It is possible that other methods, such as different surgical methods, affect the dominant effect of ECMO-assisted surgery and other interventions. Future research should specifically distinguish the different effects of different left ventricular decompression methods in addition to IABP in patients with cardiogenic shock.
Conclusions
ECMO is useful in cardiogenic shock and is associated with lower mortality. Left ventricular decompression is more important for VA-ECMO than for ECMO alone and helps to improve patient outcomes without increasing the risk of ECMO-related complications. Our statistical analysis did not find better outcomes for ECMO combined other LV decompression techniques than for ECMO+IABP. A prospective multi-center study would help determine the potential of this technique to improve the outcomes of critically ill patients. In particular, the hemodynamic effects of different left heart decompression methods should be clearly defined.
Figures
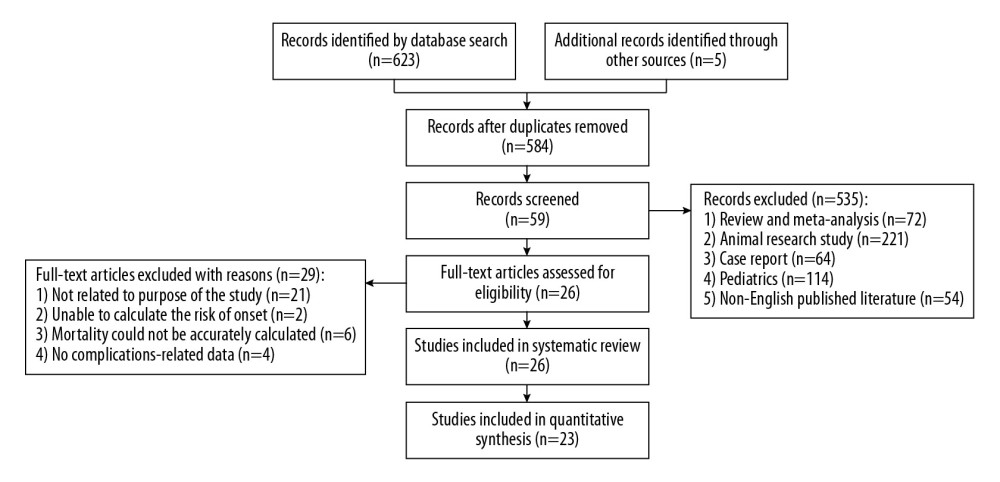 Figure 1. Process for the identification of the included studies.
Figure 1. Process for the identification of the included studies. 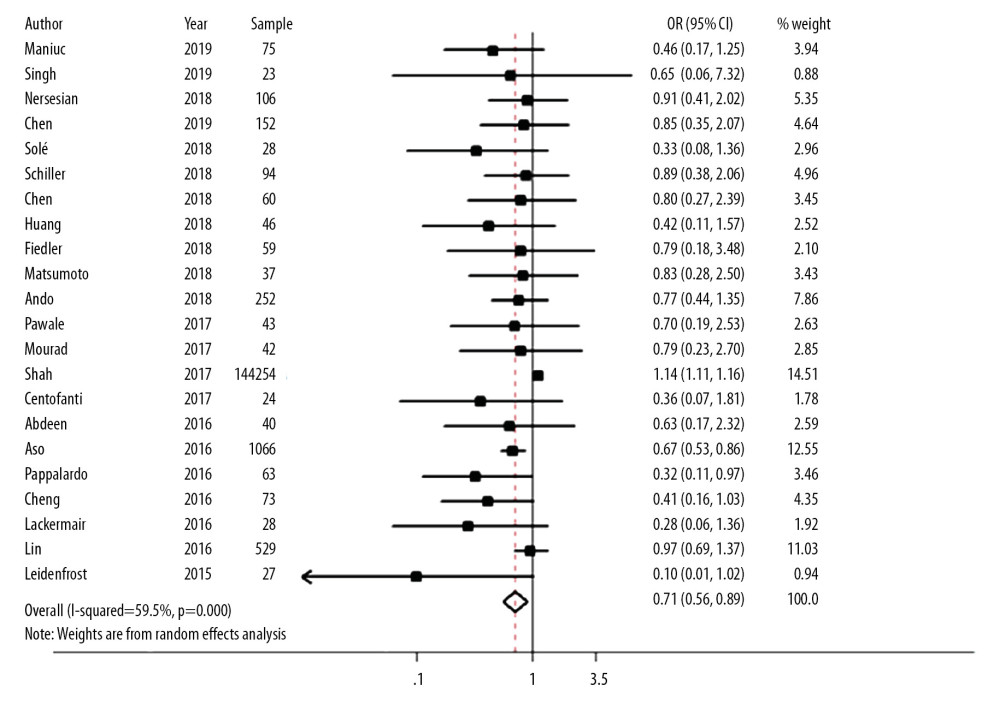 Figure 2. Forest plot of comparing the risk of death between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone.
Figure 2. Forest plot of comparing the risk of death between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone. ![Forest plot comparing the risk of death between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A represents all the included studies; B represents the studies excluding ref. [14] and [17]).](https://jours.isi-science.com/imageXml.php?i=medscimonit-26-e924009-g003.jpg&idArt=924009&w=1000) Figure 3. Forest plot comparing the risk of death between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A represents all the included studies; B represents the studies excluding ref. [14] and [17]).
Figure 3. Forest plot comparing the risk of death between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A represents all the included studies; B represents the studies excluding ref. [14] and [17]). 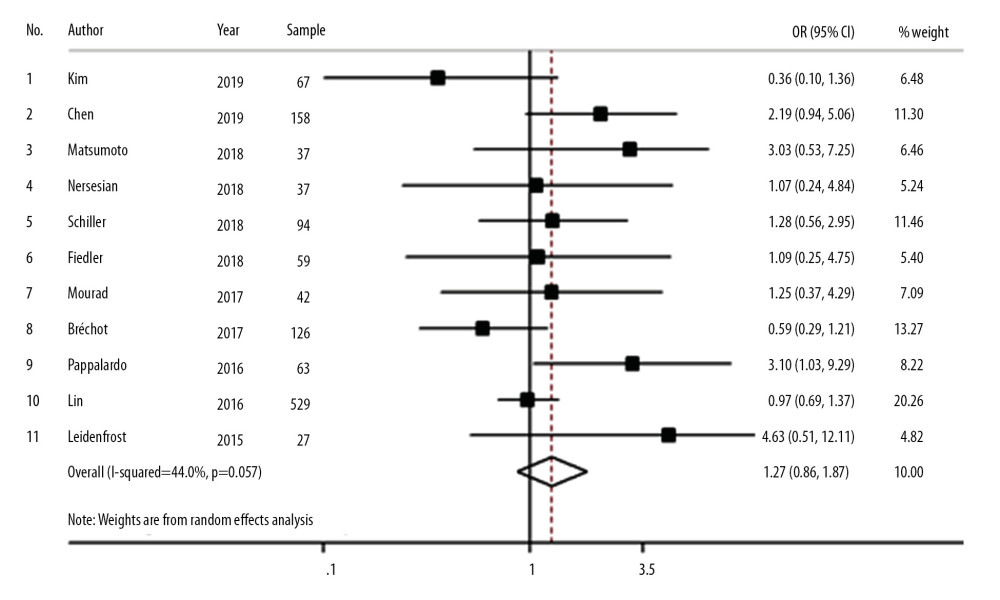 Figure 4. Forest plot comparing the risk of death between ECMO combined with ECMO-assisted surgery and ECMO combined with IABP.
Figure 4. Forest plot comparing the risk of death between ECMO combined with ECMO-assisted surgery and ECMO combined with IABP. 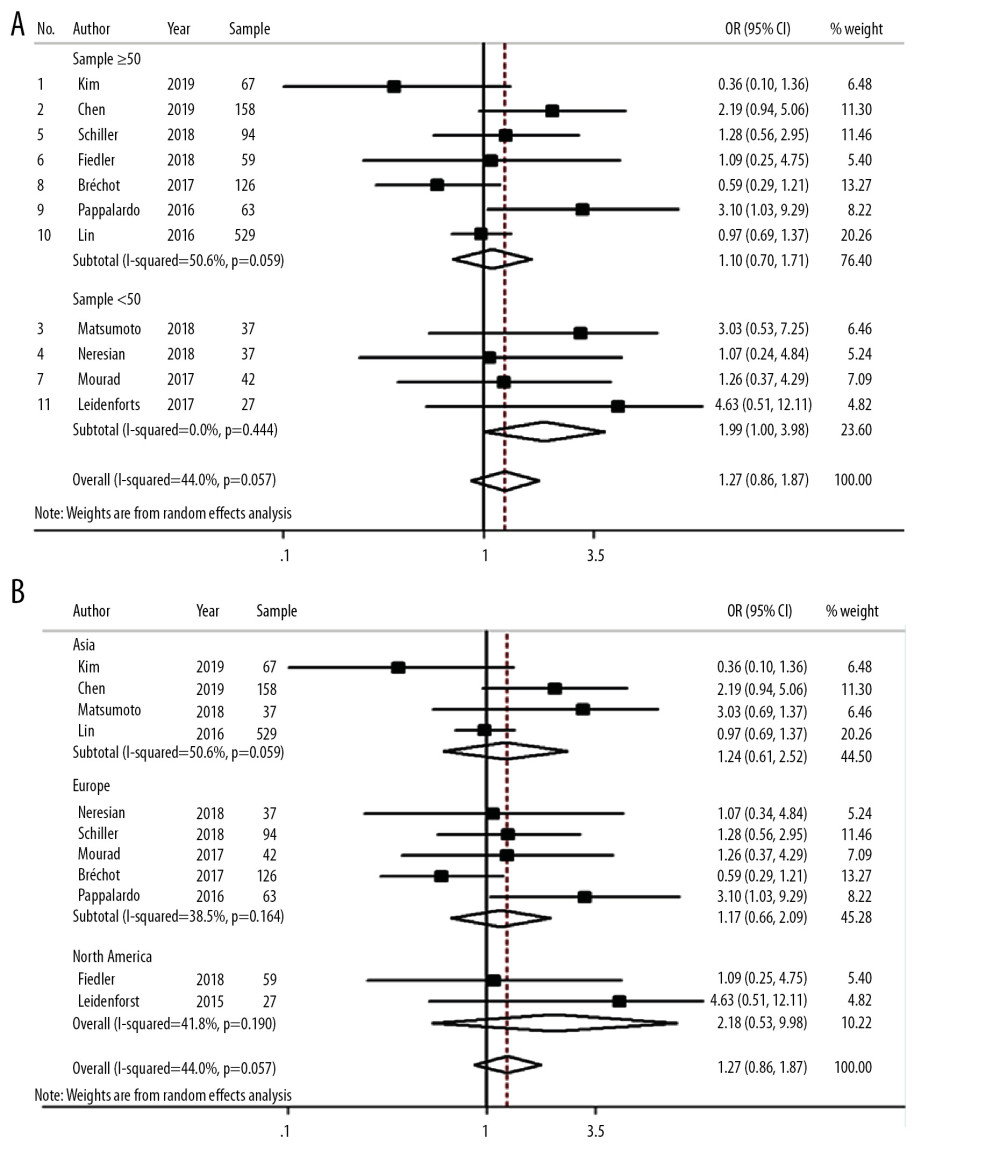 Figure 5. Forest plot comparing the risk of death between ECMO-assisted surgery and ECMO combined with IABP (A represents grouping by sample size; B represents grouping by region).
Figure 5. Forest plot comparing the risk of death between ECMO-assisted surgery and ECMO combined with IABP (A represents grouping by sample size; B represents grouping by region). 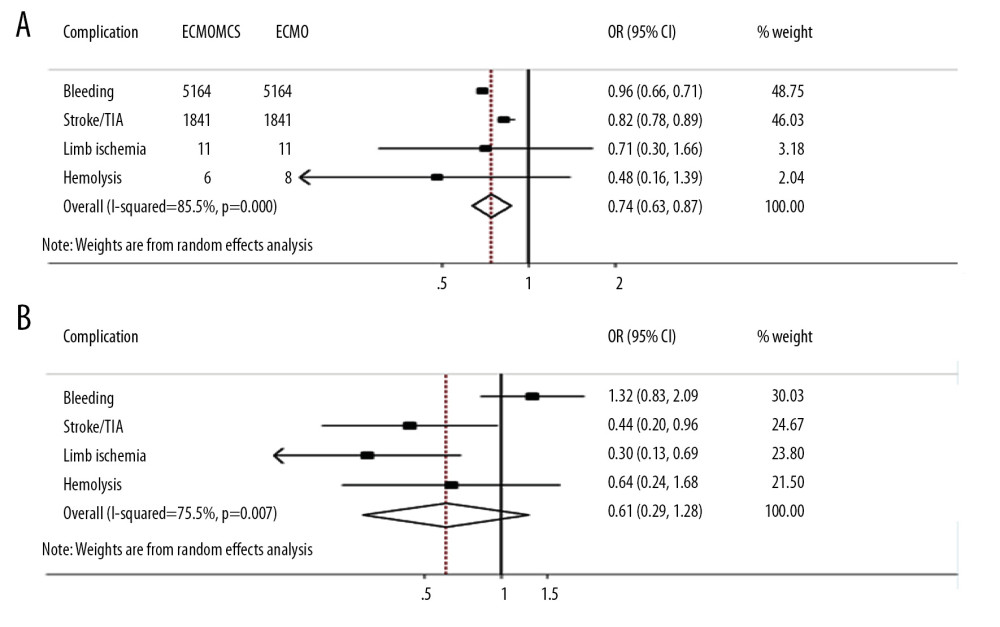 Figure 6. Comparison of the risk of complication between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B).
Figure 6. Comparison of the risk of complication between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B). 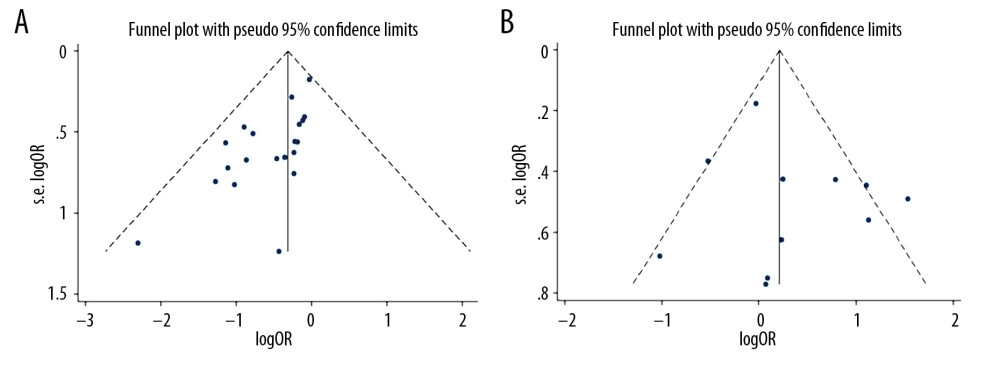 Figure 7. Funnel plot for ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B).
Figure 7. Funnel plot for ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B). 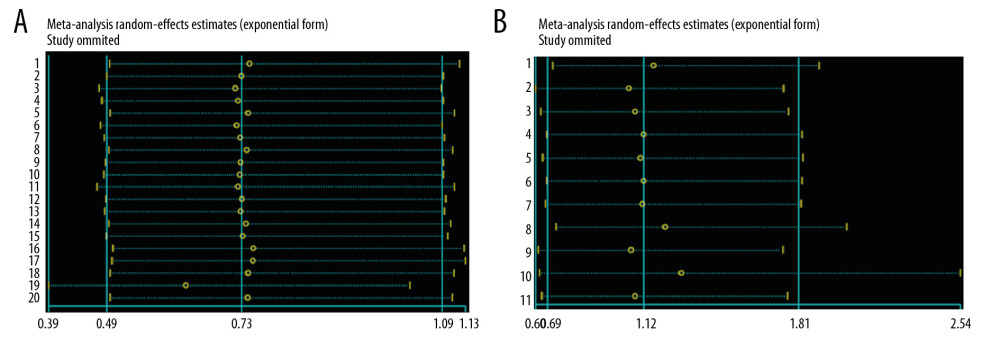 Figure 8. Sensitivity analysis for ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B).
Figure 8. Sensitivity analysis for ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B). References
1. Mebazaa A, Combes A, van Diepen S, Management of cardiogenic shock complicating myocardial infarction: Intensive Care Med, 2018; 44(6); 760-73
2. Harjola VP, Lassus J, Sionis A, Clinical picture and risk prediction of short-term mortality in cardiogenic shock: Eur J Heart Fail, 2015; 17(5); 501-9
3. Mandawat A, Rao SV, Percutaneous mechanical circulatory support devices in cardiogenic shock: Circ Cardiovasc Interv, 2017; 10(5); e004337
4. Hajjar LA, Teboul JL, Mechanical circulatory support devices for cardiogenic shock: State of the art: Crit Care, 2019; 23(1); 76
5. Bellumkonda L, Gul B, Masri SC, Evolving concepts in diagnosis and management of cardiogenic shock: Am J Cardiol, 2018; 122(6); 1104-10
6. Hamzaoui O, Jozwiak M, Geffriaud T, Norepinephrine exerts an inotropic effect during the early phase of human septic shock: Br J Anaesth, 2018; 120(3); 517-24
7. Toscani L, Aya HD, Antonakaki D, What is the impact of the fluid challenge technique on diagnosis of fluid responsiveness? A systematic review and meta-analysis: Crit Care, 2017; 21(1); 207
8. Keebler ME, Haddad EV, Choi CW, Venoarterial extracorporeal membrane oxygenation in cardiogenic shock: JACC Heart Fail, 2018; 6(6); 503-16
9. Gokalp O, Donmez K, Iner H, Should ECMO be used in cardiogenic shock?: Crit Care, 2019; 23(1); 174
10. Truby LK, Takeda K, Mauro C, Incidence and implications of left ventricular distention during venoarterial extracorporeal membrane oxygenation support: ASAIO J, 2017; 63(3); 257-65
11. Ma P, Zhang Z, Song T, Combining ECMO with IABP for the treatment of critically Ill adult heart failure patients: Heart Lung Circ, 2014; 23(4); 363-68
12. Shinar Z, Is the “Unprotected Heart” a clinical myth? Use of IABP, Impella, and ECMO in the acute cardiac patient: Resuscitation, 2019; 140; 205-6
13. Nuding S, Werdan K, IABP plus ECMO – is one and one more than two?: J Thorac Dis, 2017; 9(4); 961-64
14. Li Y, Yan S, Cai L, Zhang Q, Does VA-ECMO plus Impella work in refractory cardiogenic shock?: JACC Heart Fail, 2019; 7(4); 364
15. Aiyagari RM, Rocchini AP, Remenapp RT, Graziano JN, Decompression of the left atrium during extracorporeal membrane oxygenation using a transseptal cannula incorporated into the circuit: Crit Care Med, 2006; 34(10); 2603-6
16. Muller G, Flecher E, Lebreton G, The ENCOURAGE mortality risk score and analysis of long-term outcomes after VA-ECMO for acute myocardial infarction with cardiogenic shock: Intensive Care Med, 2016; 42(3); 370-78
17. Kapelios CJ, Terrovitis JV, Nanas JN, Current and future applications of the intra-aortic balloon pump: Curr Opin Cardiol, 2014; 29(3); 258-65
18. Schrage B, Burkhoff D, Rubsamen N, Unloading of the left ventricle during venoarterial extracorporeal membrane oxygenation therapy in cardiogenic shock: JACC Heart Fail, 2018; 6(12); 1035-43
19. Pappalardo F, Schulte C, Pieri M, Concomitant implantation of Impella((R)) on top of veno-arterial extracorporeal membrane oxygenation may improve survival of patients with cardiogenic shock: Eur J Heart Fail, 2017; 19(3); 404-12
20. Moller-Helgestad OK, Hyldebrandt JA, Banke A, Impella CP or VA-ECMO in profound cardiogenic shock: Left ventricular unloading and organ perfusion in a large animal model: EuroIntervention, 2019; 14(15); e1585-92
21. Le Guennec L, Cholet C, Huang F, Ischemic and hemorrhagic brain injury during venoarterial-extracorporeal membrane oxygenation: Ann Intensive Care, 2018; 8(1); 129
22. Vaquer S, de Haro C, Peruga P, Systematic review and meta-analysis of complications and mortality of veno-venous extracorporeal membrane oxygenation for refractory acute respiratory distress syndrome: Ann Intensive Care, 2017; 7(1); 51
23. Li Y, Yan S, Gao S, Effect of an intra-aortic balloon pump with venoarterial extracorporeal membrane oxygenation on mortality of patients with cardiogenic shock: A systematic review and meta-analysisdagger: Eur J Cardiothorac Surg, 2019; 55(3); 395-404
24. Maniuc O, Salinger T, Anders F, Impella CP use in patients with non-ischaemic cardiogenic shock: ESC Heart Fail, 2019; 6(4); 863-66
25. Avtaar Singh SS, Das De S, Nappi F, Mechanical circulatory support for refractory cardiogenic shock post-acute myocardial infarction-a decade of lessons: J Thorac Dis, 2019; 11(2); 542-48
26. Nersesian G, Hennig F, Muller M, Temporary mechanical circulatory support for refractory heart failure: The German Heart Center Berlin experience: Ann Cardiothorac Surg, 2019; 8(1); 76-83
27. Chen K, Hou J, Tang H, Hu S, Concurrent initiation of intra-aortic balloon pumping with extracorporeal membrane oxygenation reduced in-hospital mortality in postcardiotomy cardiogenic shock: Ann Intensive Care, 2019; 9(1); 16
28. Ariza-Sole A, Sanchez-Salado JC, Sbraga F, The role of perioperative cardiorespiratory support in post infarction ventricular septal rupture-related cardiogenic shock: Eur Heart J Acute Cardiovasc Care, 2020; 9(2); 128-37
29. Schiller P, Hellgren L, Vikholm P, Survival after refractory cardiogenic shock is comparable in patients with Impella and veno-arterial extracorporeal membrane oxygenation when adjusted for SAVE score: Eur Heart J Acute Cardiovasc Care, 2019; 8(4); 329-37
30. Chen K, Hou J, Tang H, Hu S, Concurrent implantation of intra-aortic balloon pump and extracorporeal membrane oxygenation improved survival of patients with postcardiotomy cardiogenic shock: Artif Organs, 2019; 43(2); 142-49
31. Huang CC, Hsu JC, Wu YW, Implementation of extracorporeal membrane oxygenation before primary percutaneous coronary intervention may improve the survival of patients with ST-segment elevation myocardial infarction and refractory cardiogenic shock: Int J Cardiol, 2018; 269; 45-50
32. Fiedler AG, Dalia A, Axtell AL, Impella placement guided by echocardiography can be used as a strategy to unload the left ventricle during peripheral venoarterial extracorporeal membrane oxygenation: J Cardiothorac Vasc Anesth, 2018; 32(6); 2585-91
33. Matsumoto M, Asaumi Y, Nakamura Y, Clinical determinants of successful weaning from extracorporeal membrane oxygenation in patients with fulminant myocarditis: ESC Heart Fail, 2018; 5(4); 675-84
34. Ando M, Garan AR, Takayama H, A continuous-flow external ventricular assist device for cardiogenic shock: Evolution over 10 years: J Thorac Cardiovasc Surg, 2018; 156(1); 157-165.e151
35. Pawale A, Schwartz Y, Itagaki S, Selective implantation of durable left ventricular assist devices as primary therapy for refractory cardiogenic shock: J Thorac Cardiovasc Surg, 2018; 155(3); 1059-68
36. Mourad M, Gaudard P, De La Arena P, Circulatory support with extracorporeal membrane oxygenation and/or impella for cardiogenic shock during myocardial infarction: ASAIO J, 2018; 64(6); 708-14
37. Shah M, Patnaik S, Patel B, Trends in mechanical circulatory support use and hospital mortality among patients with acute myocardial infarction and non-infarction related cardiogenic shock in the United States: Clin Res Cardiol, 2018; 107(4); 287-303
38. Centofanti P, Attisani M, La Torre M, Left ventricular unloading during peripheral extracorporeal membrane oxygenator support: A bridge to life in profound cardiogenic shock: J Extra Corpor Technol, 2017; 49(3); 201-5
39. Abdeen MS, Albert A, Maxhera B, Implanting permanent left ventricular assist devices in patients on veno-arterial extracorporeal membrane oxygenation support: Do we really need a cardiopulmonary bypass machine?: Eur J Cardiothorac Surg, 2016; 50(3); 542-47
40. Aso S, Matsui H, Fushimi K, Yasunaga H, The effect of intraaortic balloon pumping under venoarterial extracorporeal membrane oxygenation on mortality of cardiogenic patients: An analysis using a Nationwide Inpatient Database: Crit Care Med, 2016; 44(11); 1974-79
41. Cheng R, Ramzy D, Azarbal B, Device strategies for patients in INTERMACS profiles 1 and 2 cardiogenic shock: Double bridge with extracorporeal membrane oxygenation and initial implant of more durable devices: Artif Organs, 2017; 41(3); 224-32
42. Lackermair K, Sattler S, Huber BC, Retrospective analysis of circulatory support with the Impella CP(R) device in patients with therapy refractory cardiogenic shock: Int J Cardiol, 2016; 219; 200-3
43. Lin LY, Liao CW, Wang CH, Effects of additional intra-aortic balloon counter-pulsation therapy to cardiogenic shock patients supported by extra-corporeal membranous oxygenation: Sci Rep, 2016; 6; 23838
44. Leidenfrost J, Prasad S, Itoh A, Right ventricular assist device with membrane oxygenator support for right ventricular failure following implantable left ventricular assist device placement: Eur J Cardiothorac Surg, 2016; 49(1); 73-77
45. Kim Y, Cho YH, Yang JH, Outcomes of coronary artery bypass grafting after extracorporeal life support in patients with cardiac arrest or cardiogenic shock: Korean J Thorac Cardiovasc Surg, 2019; 52(2); 70-77
46. Brechot N, Demondion P, Santi F, Intra-aortic balloon pump protects against hydrostatic pulmonary oedema during peripheral venoarterial-extracorporeal membrane oxygenation: Eur Heart J Acute Cardiovasc Care, 2018; 7(1); 62-69
Figures
 Figure 1. Process for the identification of the included studies.
Figure 1. Process for the identification of the included studies. Figure 2. Forest plot of comparing the risk of death between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone.
Figure 2. Forest plot of comparing the risk of death between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone.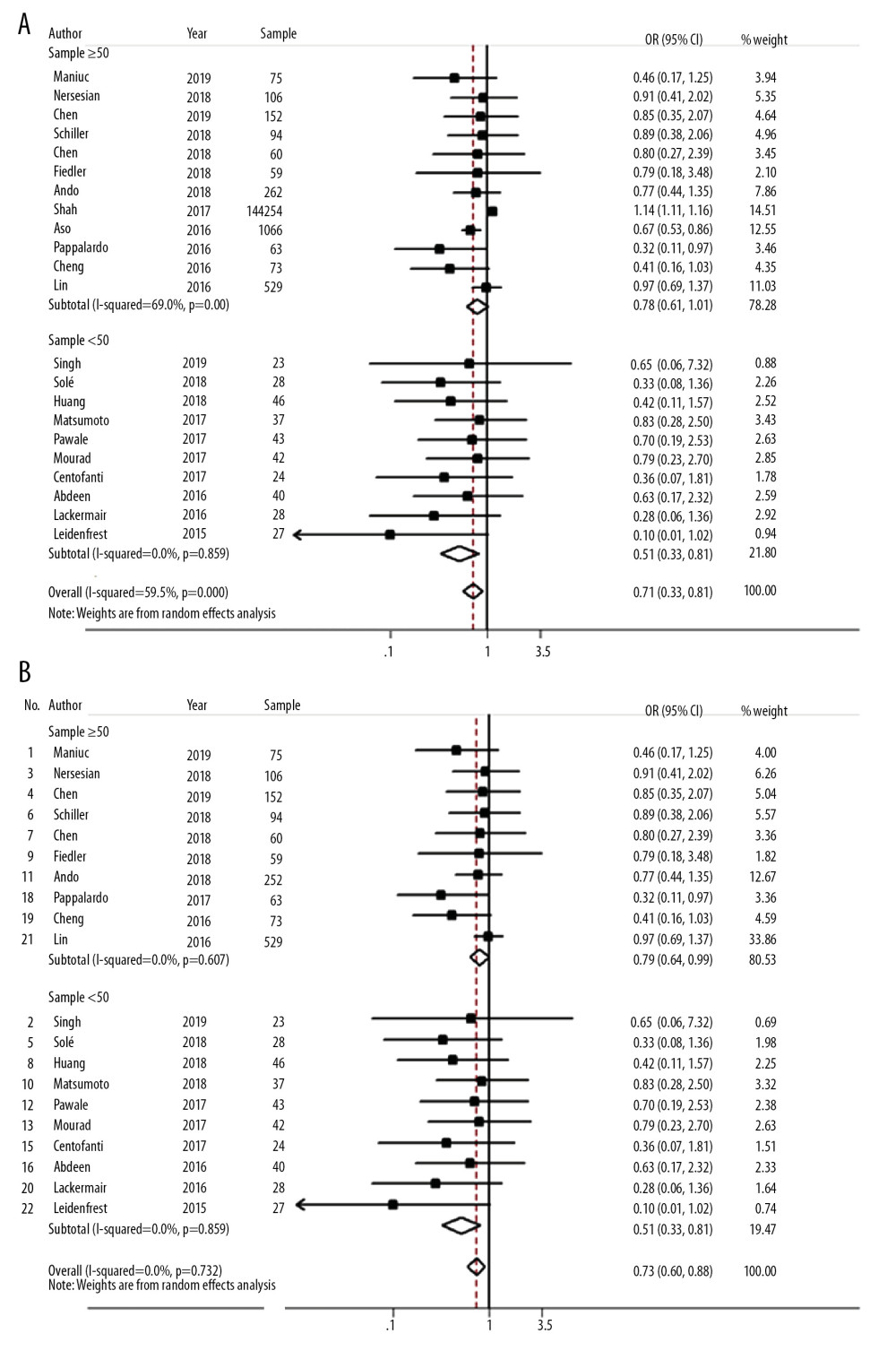 Figure 3. Forest plot comparing the risk of death between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A represents all the included studies; B represents the studies excluding ref. [14] and [17]).
Figure 3. Forest plot comparing the risk of death between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A represents all the included studies; B represents the studies excluding ref. [14] and [17]). Figure 4. Forest plot comparing the risk of death between ECMO combined with ECMO-assisted surgery and ECMO combined with IABP.
Figure 4. Forest plot comparing the risk of death between ECMO combined with ECMO-assisted surgery and ECMO combined with IABP. Figure 5. Forest plot comparing the risk of death between ECMO-assisted surgery and ECMO combined with IABP (A represents grouping by sample size; B represents grouping by region).
Figure 5. Forest plot comparing the risk of death between ECMO-assisted surgery and ECMO combined with IABP (A represents grouping by sample size; B represents grouping by region). Figure 6. Comparison of the risk of complication between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B).
Figure 6. Comparison of the risk of complication between ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B). Figure 7. Funnel plot for ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B).
Figure 7. Funnel plot for ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B). Figure 8. Sensitivity analysis for ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B).
Figure 8. Sensitivity analysis for ECMO combined with left ventricular decompression (LVD) therapy and ECMO alone (A) and ECMO-assisted surgery and ECMO combined with IABP (B). Tables
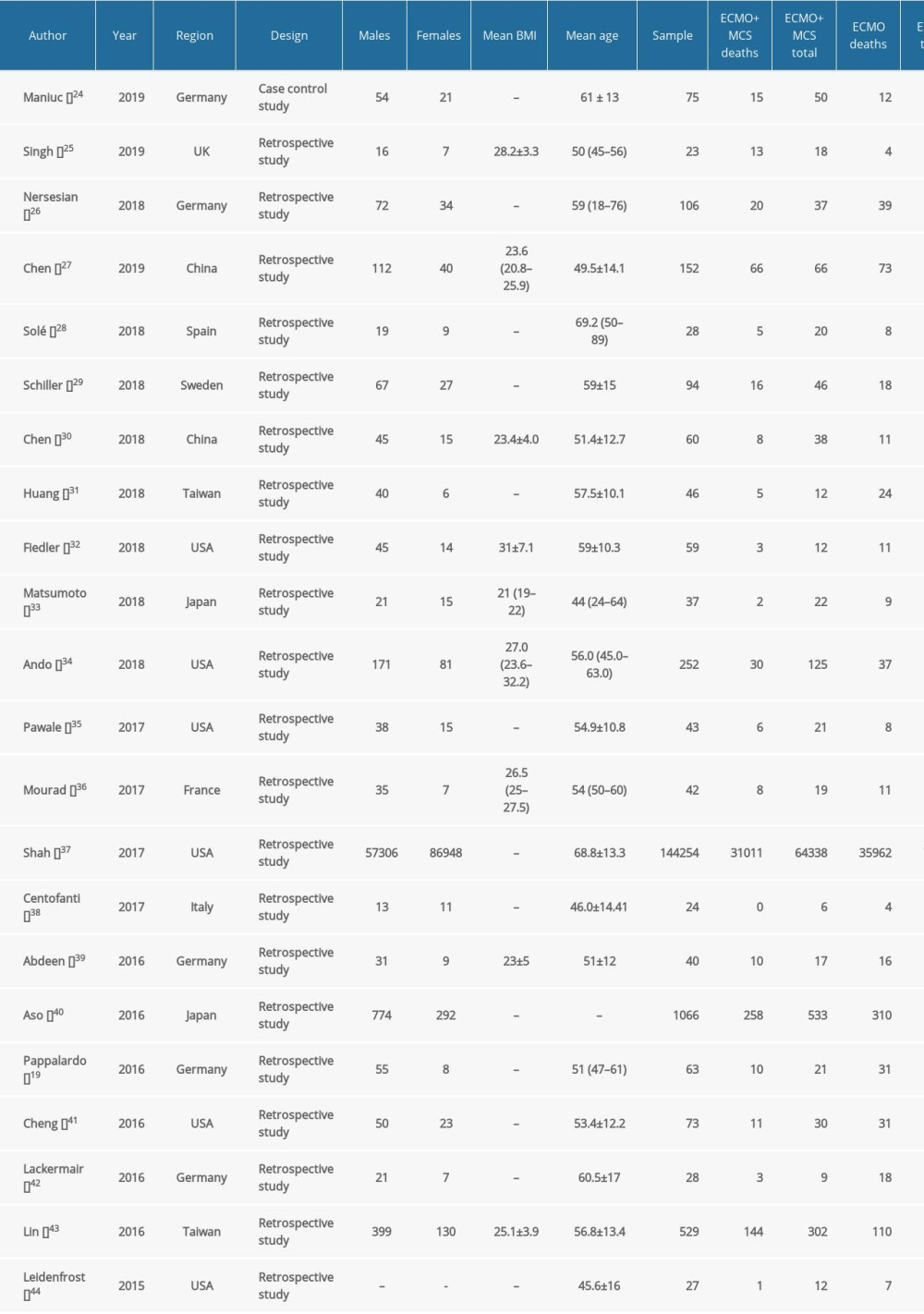 Table 1. Features of the studies included in this systematic review comparing ECMO combined with left ventricular decompression and ECMO alone.
Table 1. Features of the studies included in this systematic review comparing ECMO combined with left ventricular decompression and ECMO alone.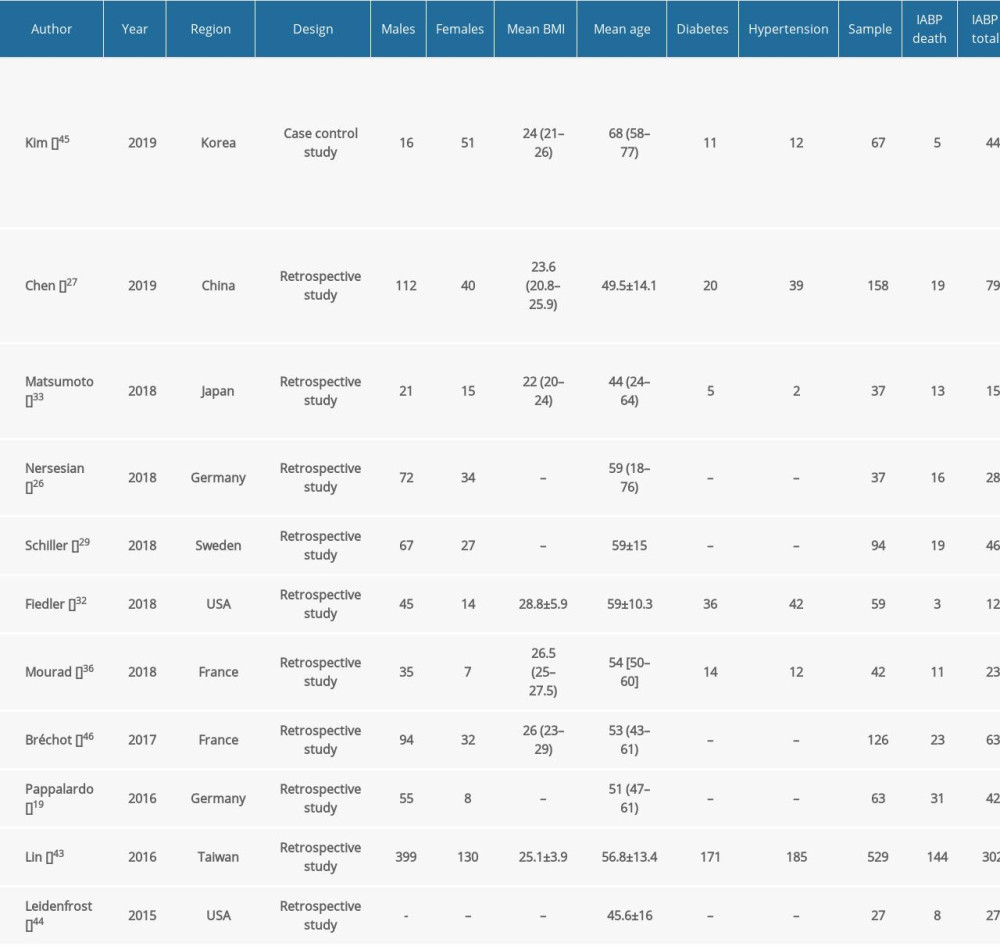 Table 2. Features of the studies included in this systematic review comparing ECMO combined with IABP, ECMO-assisted surgery.
Table 2. Features of the studies included in this systematic review comparing ECMO combined with IABP, ECMO-assisted surgery. Table 1. Features of the studies included in this systematic review comparing ECMO combined with left ventricular decompression and ECMO alone.
Table 1. Features of the studies included in this systematic review comparing ECMO combined with left ventricular decompression and ECMO alone. Table 2. Features of the studies included in this systematic review comparing ECMO combined with IABP, ECMO-assisted surgery.
Table 2. Features of the studies included in this systematic review comparing ECMO combined with IABP, ECMO-assisted surgery. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








