13 November 2020: Clinical Research
Protective Effect of Curcumin on Bone Trauma in a Rat Model via Expansion of Myeloid Derived Suppressor Cells
Futian Zhang1BCDE, Fu Liu1BCDE, Shaofen Yu1BD, Guihong Zhang1BCD, Jie Li1BCDE, Xinjun Sun1ADEF*DOI: 10.12659/MSM.924724
Med Sci Monit 2020; 26:e924724
Abstract
BACKGROUND: Bone fracture, a common injury to bones leads to various biophysiological changes and pathological responses in the body. The current study investigated curcumin for treatment of bone fracture in a rat model of bone trauma, and evaluated the related mechanism.
MATERIAL AND METHODS: The rats were separated randomly into 3 groups; sham, model, and curcumin treatment groups. The fracture rat model was established by transverse osteotomy in the right femur bone at the mid-shaft. The osteoblast count was determined using hematoxylin and eosin staining. Vascular endothelial growth factor (VEGF) and proliferating cell nuclear antigen (PCNA) expression were measured by western blotting.
RESULTS: The rpS6-phosphorylation was suppressed and light chain 3 (LC3II) expression elevated in the curcumin treated group of the fracture rat model. In the curcumin-treated group, mineralization of fracture calluses was markedly higher on day 14 of fracture. The formation of osteoblasts was observed at a greater rate in the curcumin treated group compared to the model rat group. Treatment of rats with curcumin significantly (P<0.05) promoted expression of PCNA and VEGF. The decrease in CD11b+/Gr-1+ cell expansion in rats with bone trauma was alleviated significantly by curcumin treatment. A marked increase in arginase-1 expression in rats with bone trauma was caused by curcumin treatment.
CONCLUSIONS: In summary, curcumin activates autophagy and inhibits mTOR activation in bone tissues of rats with trauma. The curcumin promoted myeloid-derived suppressor cell (MDSC) proliferation and increased expansion of MDSCs in a rat model of trauma. Therefore, curcumin may have beneficial effect in patients with bone trauma and should be evaluated further for development of treatment.
Keywords: Barotrauma, Chemoradiotherapy, Adjuvant, Arginase, Bone and Bones, Bony Callus, CD11b Antigen, Curcumin, Femoral Fractures, Microtubule-Associated Proteins, myeloid-derived suppressor cells, Osteoblasts, Phosphorylation, Proliferating Cell Nuclear Antigen, Protective Agents, Ribosomal Protein S6, Vascular Endothelial Growth Factor A, Wounds and Injuries
Background
Bone fracture, a common injury to bones, leads to various biophysiological changes and pathological responses in the body [1]. These responses initiate healing of fractures but are also accompanied by damage to tissues. The development of devices for internal fixation and the use of newly discovered biological materials have greatly improved the healing process but in ~10% of the cases nonunion or delayed union has been reported [2]. Therefore, to improve patient morbidity and minimize expenditure due to bone fractures novel therapies need to be discovered for enhancement of healing fractures [3]. Autophagy plays vital role in destroying the non-functional and damaged macromolecules besides regulating the cellular energy and nutrients [4,5]. The failure of autophagy leads to abnormal gene expression at a cellular level thereby causing the death of cells [6]. Additionally, autophagy failure is associated with the neuronal degeneration, premature death, development of abnormal skeleton and cardiomyopathies [7–9]. The autophagy activation is primarily indicated by enhanced expression of light chain 3 (LC3-II) at the cellular level [10]. An important autophagy suppressor is the mammalian target of rapamycin (mTOR) which acts as upstream effecter of proteins linked to autophagy [11]. The mTOR complex is regulated by many pathways like phosphatidylinositol-3-kinase (PI-3K)/Akt and AMP-activated protein kinases [11]. The dysfunction of mTOR is associated with the development of inflammatory disorders, cardiac diseases, diabetes, etc. [12]. Thus, the mTOR pathway is believed to be potential target for treatment of multiple disorders [12]. Inhibition of ribosomal protein S6 activation which is an mTOR complex-1 downstream factor by chemotherapeutic agents leads to autophagy activation [13,14].
Curcumin has been used for a very long time in traditional medicine because of several pharmacological properties [15]. Multiple properties of curcumin include its role as an anti-inflammatory, antioxidant, and anti-fibrotic agent [15–17]. Additionally, studies have shown tumor growth inhibitory effect and myocardial injury protecting property of curcumin [15–17]. Although studies on the use of curcumin in bone trauma treatment are limited, reports suggest that curcumin may have a positive effect in bone remodeling [18]. The current study investigated curcumin for bone trauma treatment in a rat model and evaluated the related mechanism.
Material and Methods
ANIMALS GROUPING AND TREATMENT:
Thirty adult male Sprague Dawley rats (6 to 8 weeks old; weight; 245 to 265 g) were obtained from the Animal Center of Hubei Medical University (Shiyan, China). The rats were maintained at constant temperature of 25°C and 45% to 50% humidity in the laboratory. All rats were provided food and water
BONE TRAUMA AND TREATMENT:
The rats were separated randomly into sham, model bone trauma, and model curcumin treatment groups. The fracture rat model was established by transverse osteotomy in the right femur bone at the mid-shaft. Surgical procedures on rats were conducted after intraperitoneal injection of ketamine (50 mg/kg) and xylazine hydrochloride (10 mg/kg). The lower limb on right side of rats after shaving was disinfected by povidone-iodine to carry out surgery under sterilized conditions. An incision was made carefully to expose the femurs and an oscillating microsaw was used for inducing transverse femur shaft fracture. Then intramedullary fixation was made using a 1-mm Kirschner wire and standard procedure was followed to close subcutaneous tissues and the skin. Curcumin was injected into rats in the treatment group at 8 mg/kg doses in dimethyl sulfoxide (DMSO) daily for 10 days from the day of fracture via intraperitoneal administration route. The rats in the untreated group received normal saline at equal doses. The approval for conducting the study was obtained from Animal Care and Use Committee National Institute of Health, China. The experimental procedures on rats were carried out according to guidelines from the Institutional Animal Care Committee. Five rats from each group were sacrificed on day 14, 28, and 42 of fracture using 10% chloral hydrate anesthesia at 3 mL/kg dose. For each rat, the right femur bone was exposed to perform middle transverse fracture, using a wire saw, then the fracture was stabilized by Kirschner wire [19].
OSTEOBLAST COUNT ANALYSES:
The right femur bone of rats was harvested and subsequently treated for 24 hours with 4% formalin for fixation. The decalcification of bones was performed by treatment with 10% ethylenediaminetetraacetic acid (EDTA) solution for 1 month and then the bones were embedded in paraffin. The 3–5 μm sections of decalcified bones were cut longitudinally, stained with hematoxylin and eosin (H&E) and analyzed for osteoblast count in each callus section.
IMMUNOHISTOCHEMISTRY:
The samples embedded in paraffin were deparaffinized on treatment with xylene substitute Pro-Par Clearant and subsequently subjected to rehydration in ethanol and water. The samples were treated with sodium citrate buffer (10 mM) and heated for 2 minutes at 88°C in a microwave oven. After cooling the slides at room temperature, the samples were washed with phosphate-buffered saline (PBS) and then blocked for 25 minutes with 5% serum. The samples were incubated with anti-p-rpS6 primary antibodies (1: 100; Abcam) at 4°C for overnight. The sections after PBS washing were subjected to incubation for 1 hour with biotinylated goat anti-mouse secondary antibodies (1: 100; Abcam) at 37°C. The samples were incubated for 25 minutes with Vectastain ABC-alkaline phosphatase, washed in PBS, and incubated again with alkaline phosphatase for 20 minutes.
IMMUNOFLUORESCENCE:
The bone samples embedded in paraffin after deparaffinization were rehydrated in graded ethyl alcohol and water system. The sections after PBS washing were blocked on treatment with 5% serum for 25 minutes at 37°C. The samples were incubated with polyclonal antibodies against LC3-II (dilution, 1: 150; Abcam) at 4°C for overnight. Incubation of the sections with Alexa-Fluor IgG secondary antibodies (1: 200; Abcam) was performed for 1 hour at room temperature.
WESTERN BLOT ANALYSIS:
The callus around femora was carefully resected and then frozen at −80°C for western blotting assay. The tissues were homogenized on treatment with 250 μL radioimmunoprecipitation assay (RIPA) buffer consisting of sodium chloride (150 mM), NP-40 (1%), tris-hydrochloric acid (25 mM; pH 7.6), sodium dodecyl sulfate (SDS) (0.1%), sodium deoxycholate (1%), and a protease inhibitor cocktail (Abcam). The tissue samples after incubation for 30 minutes were centrifuged at 13 000×g for 40 minutes on ice to obtain the supernatant. The content of proteins in the supernatant was estimated using bicinchoninic acid (BCA) method and then 35 mg samples were loaded per lane for resolution on 10% SDS gel. The proteins were transferred to polyvinylidene difluoride (PVDF) membranes which were blocked on treatment with 5% skimmed milk solution over 1 hour. Incubation of membranes was carried out with primary antibodies like anti-Vascular endothelial growth factor (VEGF) and anti-proliferating cell nuclear antigen (PCNA) for 2 hours. Visualization of the complexes was carried out using chemiluminescence (ProLong Gold Antifade Reagent).
GR-1+ CELL ISOLATION:
The samples of blood, spleen, and bone marrow that were collected from the rats were converted into single cellular suspensions. The red blood cell lysis buffer (Sigma-Aldrich) was used for depletion of the erythrocytes. The cells, after washing, were put into the MACS buffer containing 1x-PBS along with EDTA (2 mM) and bovine serum albumin (BSA; 0.5%). The MACS magnetic micro-beads were used for sorting the Gr-1+ cells in the purity range of 85 and 95%.
FLOW CYTOMETRIC ANALYSIS:
The cells after harvesting were put into FACS medium containing 1x-PBS along with BSA (0.1%) and sodium azide (0.1%). Then cells were dyed with FITC-labeled anti-mouse CD11b, and PE-labeled GR1 in accordance with the suppliers’ instructions. The FACScan flow cytometer (BD Biosciences) was used for analysis of the stained cells.
STATISTICAL ANALYSIS:
The data expressed are the mean of ± standard deviation (SD) of 3 experiments conducted independently. The statistically significant differences between 2 groups were evaluated using Student’s
Results
CURCUMIN SUPPRESSED RPS6-PHOSPHORYLATION AND PROMOTED LC3-II EXPRESSION IN RAT FRACTURE MODEL:
The changes in rpS6-phosphorylation in the rats after bone fracture were determined for analysis of mTOR pathway activation (Figure 1). The rpS6-phosphorylation showed a marked reduction in the bone tissues of rats with fracture on treatment with curcumin on day 14, 28, and 42 post fractures. The phosphorylation of rpS6 was found to be markedly higher in the untreated group of fracture rat model. In curcumin treated group of the fracture rat model, the expression of LC3-II was found to be markedly elevated compared to untreated rats (Figure 2). The elevated LC3-II expression in curcumin treated rat bone tissues indicated activation of autophagy.
FORMATION OF CALLUS BY CURCUMIN:
Mineralization of the calluses in rats was analyzed on day 14, 28, and 42 of fracture using radiography (Figure 3). The mineralization was not observed in rat calluses of untreated group on day 14 of fracture. However, density of mineralization increased on day 28 of fracture in the calluses in untreated group. The calluses in untreated group were remarkably small in size on day 42 of fracture which showed significant re-sorption. In curcumin-treated group mineralization of fracture calluses was observed on day 14 of fracture. Compared to untreated group calluses in curcumin treated rats were much larger in size on day 42 of the fracture.
CURCUMIN INCREASED THE NUMBER OF OSTEOBLASTS IN THE RAT CALLUS:
The changes in osteoblast proliferation in rats on day 14, 28, and 42 after fracture by curcumin treatment were analyzed in the callus sections (Figure 4). The formation of osteoblasts was observed at greater rate in curcumin treated group compared to model rat group. In model rats, the formation of osteoblasts was significantly (P<0.02) lower compared to the sham group.
THE PCNA AND VEGF LEVELS INCREASED IN CELLS BY CURCUMIN TREATMENT:
The PCNA and VEGF expressions play vital roles in osteoblast proliferation and healing of fractures. The changes in PCNA and VEGF expression by curcumin were analyzed in the rats using western blot assays (Figure 5). In the model group PCNA and VEGF expression was much lower compared to the sham group of rats. Treatment of rats with curcumin significantly (P<0.05) promoted expressions of PCNA and VEGF on day 14 after fracture.
CURCUMIN ALLEVIATED TRAUMA INDUCED REDUCTION OF CD11B+/GR-1+ CELL EXPANSION IN TRAUMA RAT MODEL:
In rats with bone trauma, the CD11b+/Gr-1+ cell expansion to spleen, bone marrow, and blood was significantly decreased (Figure 6). The decrease in CD11b+/Gr-1+ cell expansion in rats with bone trauma was alleviated significantly by curcumin treatment. In 8 mg/kg curcumin treated rats the trauma mediated decrease in CD11b+/Gr-1+ cell expansion was completely alleviated.
CURCUMIN INCREASED EXPRESSION OF ARGINASE-1 IN GR-1+ CELLS OF TRAUMA RAT MODEL:
Western blotting was employed to assess expression of arginase-1 in Gr-1+ cells of rats with bone trauma following curcumin treatment (Figure 7). A marked increase in arginase-1 expression in rats with bone trauma was caused by curcumin treatment.
Discussion
The rpS6-phosphorylation integrates translation of proteins with the process of cellular proliferation and growth [19]. The inhibition of rpS6-phosphorylation indicates suppression of mTOR pathway [19]. The formation of bones and their degeneration is affected by the activation of autophagy [20,21]. It is reported that autophagy suppression and activation of mTORC1 are involved in the bone loss and other osteoarthritis diseases induced by ageing [22,23]. However, an increase in autophagy and deactivation of mTORC1 have been found to extend the life span of people [24,25]. In the present study, curcumin treatment of the trauma rat model effectively suppressed rpS6-phosphorylation in the bone tissues. The curcumin mediated rpS6-phosphorylation inhibition in the rat bone tissues indicated inhibition of the mTOR pathway. In curcumin treated bone trauma rats, the autophagosomal marker LC3-II expression was markedly promoted compared to untreated rats. Therefore, in the bone trauma rat model, the treatment with curcumin regulated the mTOR pathway and the activation of autophagy.
Bone repair is characterized by formation of calluses and subsequent mineralization. The present study showed rapid formation of calluses and mineralization in the rat model of bone trauma on treatment with curcumin. The VEGF expression has been shown to promote osteocyte survival, extracellular matrix (ECM) calcification, formation of new bone tissues and blood vessels [26]. The osteoprogenitor cells are influenced directly by VEGF thereby increasing the differentiation of osteoblasts and promoting the bone mineralization [27]. The content of PCNA has been shown to be promoted markedly by mTOR pathway inhibition in the calluses of a rat bone trauma model [28]. The present study demonstrated that curcumin treatment in a bone trauma rat model markedly enhanced the levels of VEGF and PCNA expression in bone tissues. Thus, curcumin improved repair of bone fracture in rats by promoting the expression of VEGF and PCNA. The present study demonstrated that curcumin prevents RU486 induced suppression of myeloid-derived suppressor cell (MDSC) expansion to spleen, peripheral blood, and in bone marrow in our trauma rat model. It has been reported that MDSC aggregation is promoted during trauma in various organs including the spleen [29,30]. The data from our present study showed a marked increase in MDSCs in the bone trauma rat model on treatment with curcumin. In an immunosuppressed rat model induced by endotoxin administration, MDSC proliferation was increased by glucocorticoid treatment [31]. In our present study, bone trauma suppressed MDSC proliferation which was evident by Gr-1+ cell accumulation in peripheral blood, spleen, as well as in bone of rats with trauma. However, the suppression in CD11b+/Gr-1+ cell expansion by trauma in rats with bone trauma was alleviated significantly by curcumin treatment. This suggests that curcumin prevents inhibition of MDSC proliferation induced by bone trauma. T-cell functioning is reduced by MDSCs via promotion of arginase-1 expression which leads to arginine depletion in the immune microenvironment [32,33]. The expansion of MDSCs is influenced by many other trauma related molecules such as PGE2 and S100A9/8 [34,35]. These factors are also believed to increase expansion of MDSCs in the trauma patients. Many studies for making clear the bone trauma mechanism is being continuously conducted [36,37]. The present study evaluated changes in arginase-1 expression by curcumin and trauma in a rat model of trauma. Our study data showed that bone trauma caused no significant changes in expression of arginase-1 in rats. However, arginase-1 expression in curcumin-treated rat model of trauma was markedly upregulated compared to the untreated group. Although our present study demonstrated that curcumin exhibits beneficial effect on bone fracture healing, more studies should be designed to confirm the effect using biochemical tests and micro computed tomography (CT) evaluation techniques.
Conclusions
Our study found that curcumin activated autophagy and inhibited mTOR activation in bone tissues of rats with bone trauma. The curcumin promoted MDSC proliferation and increased expansion of MDSCs in the rat model of trauma. Therefore, curcumin might have a beneficial effect in patients with bone trauma and should be evaluated further for development of treatment.
Figures
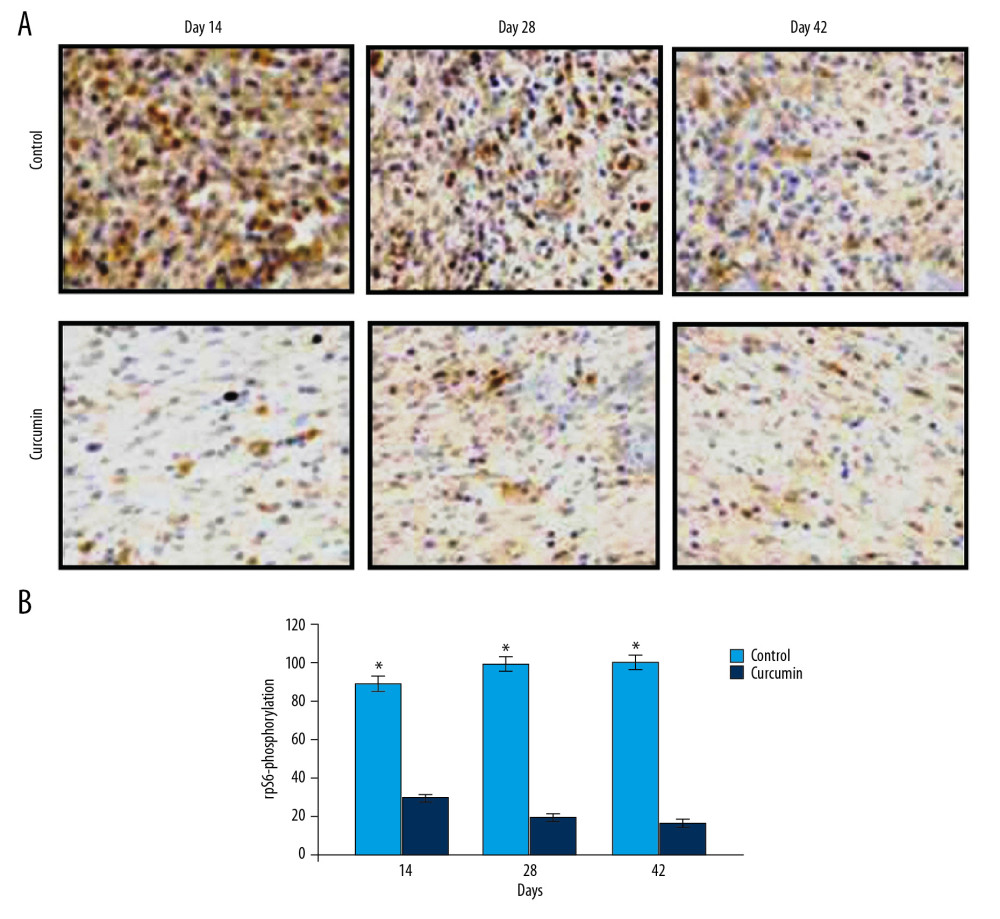 Figure 1. (A, B) Effect of curcumin on rpS6-phosphorylation in fracture rat model. The rat calluses from curcumin treated or untreated groups were collected on day 14, 28, and 42 after fracture. Immunohistochemical analysis was performed for detection of rpS6-phosphorylation. * P<0.05, ** P<0.02 versus model (control) group. Magnification, 200×.
Figure 1. (A, B) Effect of curcumin on rpS6-phosphorylation in fracture rat model. The rat calluses from curcumin treated or untreated groups were collected on day 14, 28, and 42 after fracture. Immunohistochemical analysis was performed for detection of rpS6-phosphorylation. * P<0.05, ** P<0.02 versus model (control) group. Magnification, 200×. 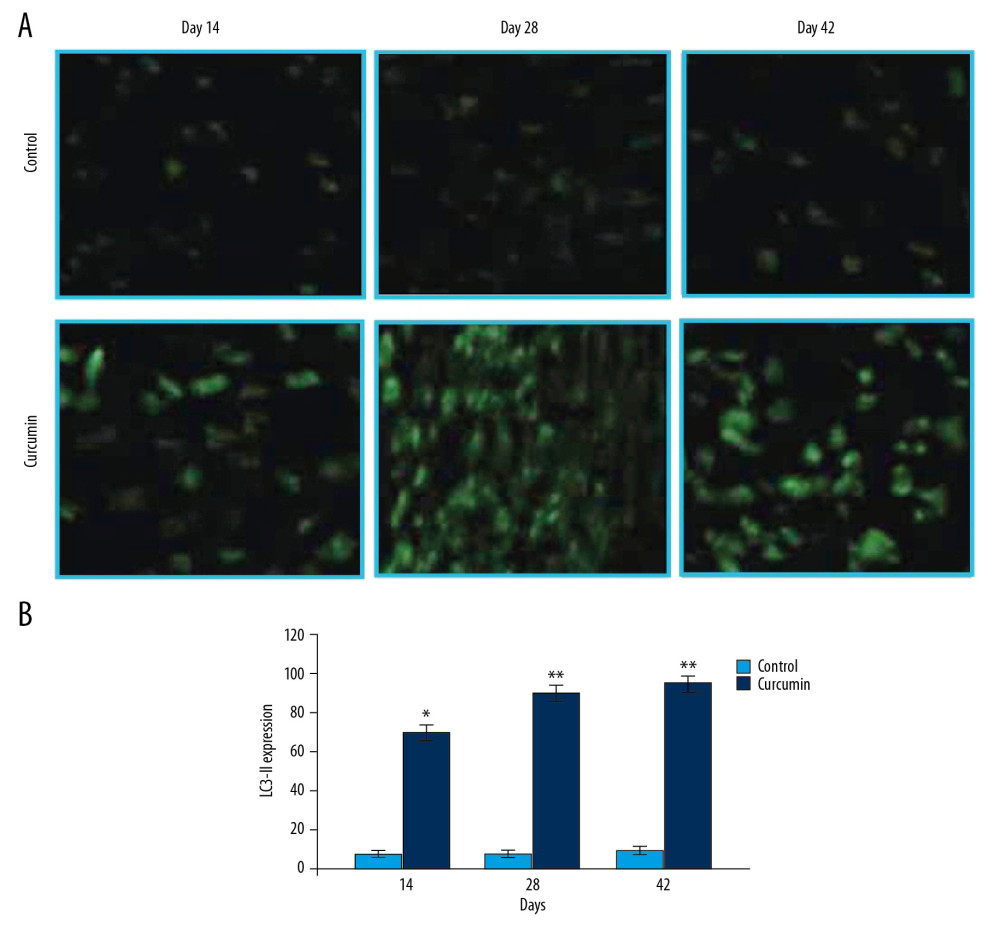 Figure 2. (A, B) Effect of curcumin on LC3-II expression in fracture rat model. The rat calluses from curcumin treated or untreated groups were collected on day 14, 28, and 42 after fracture. The LC3-II expression in the bone tissues was detected using immunofluorescence. * P<0.05, ** P<0.02 versus model (control) group. Magnification, 200×.
Figure 2. (A, B) Effect of curcumin on LC3-II expression in fracture rat model. The rat calluses from curcumin treated or untreated groups were collected on day 14, 28, and 42 after fracture. The LC3-II expression in the bone tissues was detected using immunofluorescence. * P<0.05, ** P<0.02 versus model (control) group. Magnification, 200×. 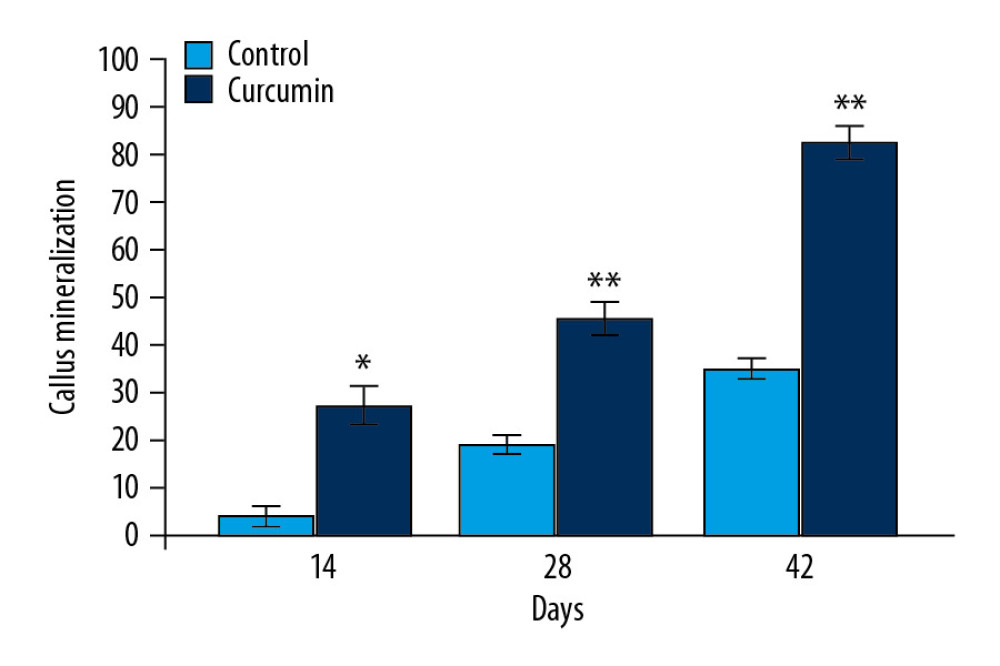 Figure 3. Effect of curcumin on fracture repair in rats. The callus mineralization as well as size in fracture rat model was observed on day 14, 28, and 42 of fracture using x-ray imaging. * P<0.05, ** P<0.02 versus model (control) group.
Figure 3. Effect of curcumin on fracture repair in rats. The callus mineralization as well as size in fracture rat model was observed on day 14, 28, and 42 of fracture using x-ray imaging. * P<0.05, ** P<0.02 versus model (control) group. 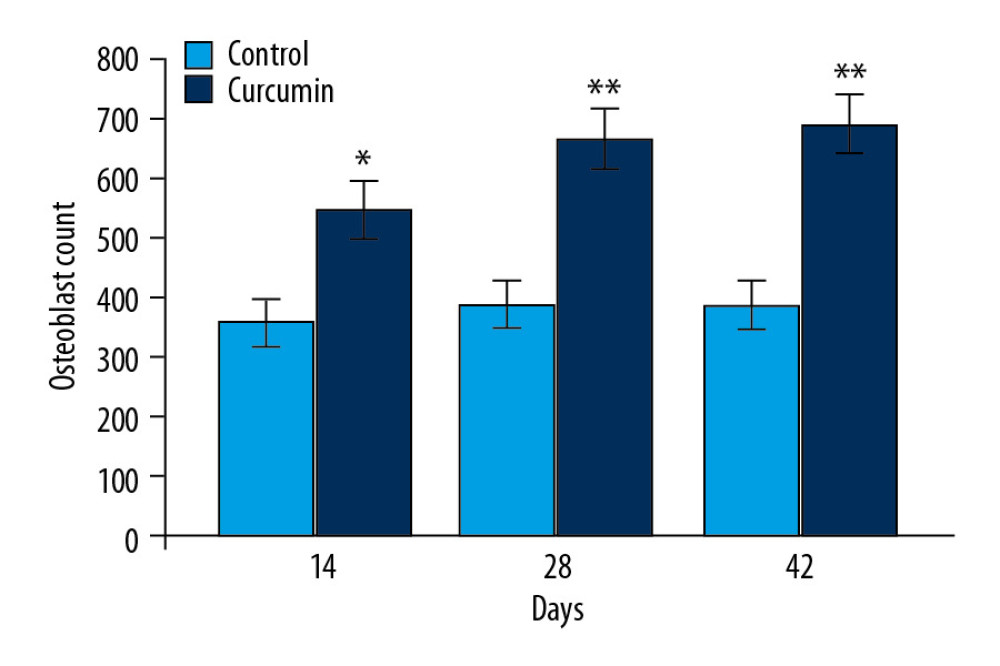 Figure 4. Effect of curcumin on osteoblast proliferation. The callus sections of sham, model, and curcumin treatment groups were analyzed for osteoblast proliferation on day 14, 28, and 42 after fracture using hematoxylin and eosin dyes. * P<0.05, ** P<0.02 versus model (control) group.
Figure 4. Effect of curcumin on osteoblast proliferation. The callus sections of sham, model, and curcumin treatment groups were analyzed for osteoblast proliferation on day 14, 28, and 42 after fracture using hematoxylin and eosin dyes. * P<0.05, ** P<0.02 versus model (control) group. 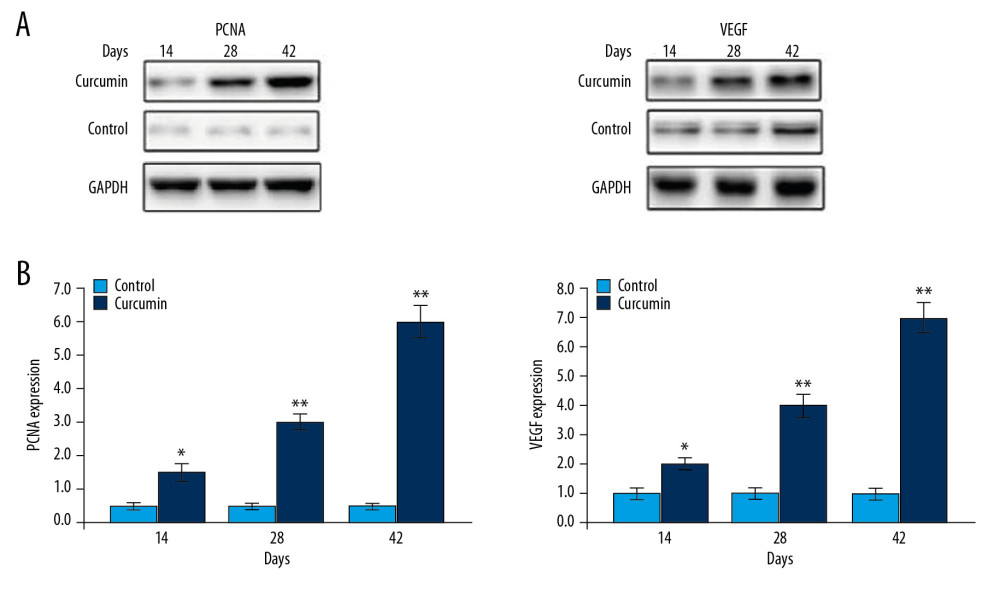 Figure 5. Curcumin promotes PCNA and VEGF expression. (A) The PCNA and VEGF levels in calluses of rats were assessed by western blotting on day 14, 28, and 42 post fractures. (B) The quantification of PCNA and VEGF levels was made taking GAPDH as loading control. * P<0.05, ** P<0.02 versus model (control) group. PCNA – proliferating cell nuclear antigen; VEGF – vascular endothelial growth factor; GAPHD – glyceraldehyde 3-phosphate dehydrogenase.
Figure 5. Curcumin promotes PCNA and VEGF expression. (A) The PCNA and VEGF levels in calluses of rats were assessed by western blotting on day 14, 28, and 42 post fractures. (B) The quantification of PCNA and VEGF levels was made taking GAPDH as loading control. * P<0.05, ** P<0.02 versus model (control) group. PCNA – proliferating cell nuclear antigen; VEGF – vascular endothelial growth factor; GAPHD – glyceraldehyde 3-phosphate dehydrogenase. 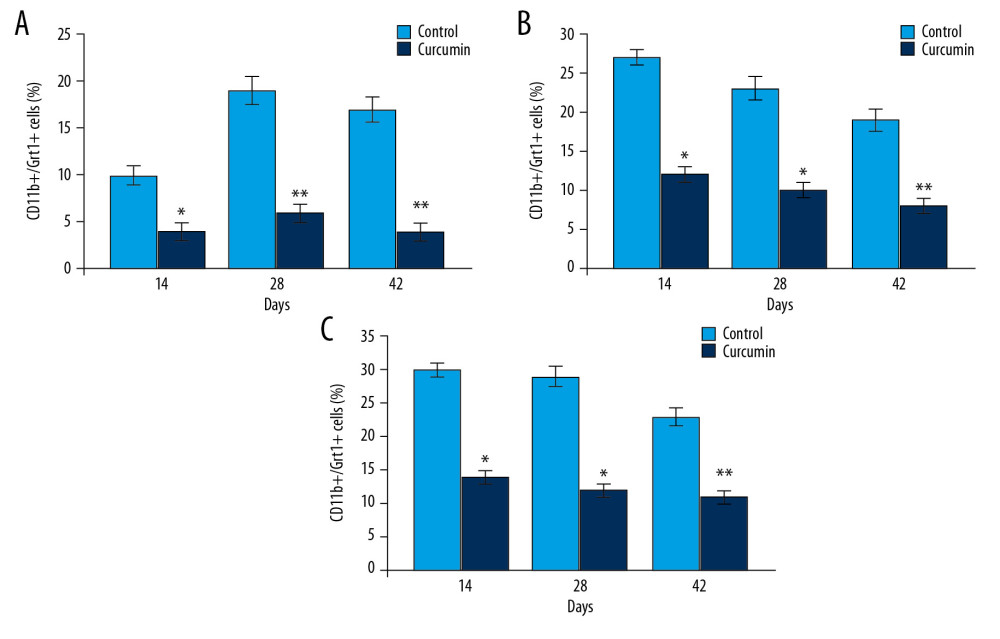 Figure 6. Effect of curcumin on expansion of CD11b+/Gr1+ cells. The rats with bone trauma were treated with curcumin or left untreated. The expansion of CD11b+/Gr1+ cells to (A) spleen, (B) bone marrow, and (C) blood was detected by flow cytometry. * P<0.5 and ** P<0.02 versus model (control).
Figure 6. Effect of curcumin on expansion of CD11b+/Gr1+ cells. The rats with bone trauma were treated with curcumin or left untreated. The expansion of CD11b+/Gr1+ cells to (A) spleen, (B) bone marrow, and (C) blood was detected by flow cytometry. * P<0.5 and ** P<0.02 versus model (control). 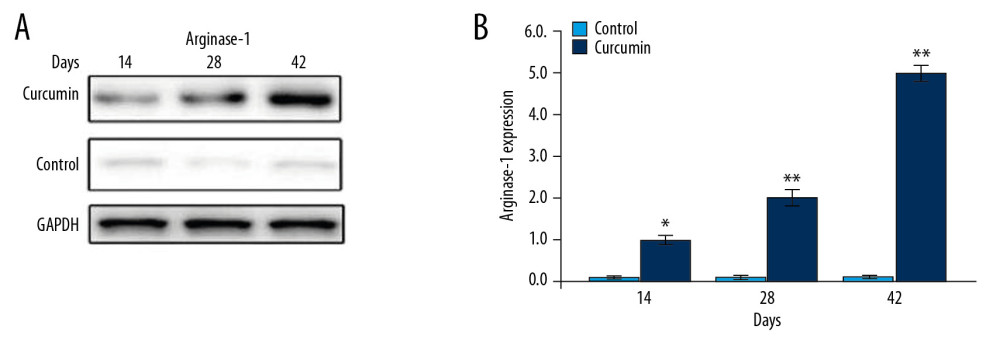 Figure 7. Effect of curcumin on expression of arginase-1 in Gr-1+ cells. The rats were treated with curcumin immediately following trauma. (A) The expression of arginase-1 was measured by western blotting. (B) Quantification of western blotting data. * P<0.05 and ** P<0.02 versus model (control).
Figure 7. Effect of curcumin on expression of arginase-1 in Gr-1+ cells. The rats were treated with curcumin immediately following trauma. (A) The expression of arginase-1 was measured by western blotting. (B) Quantification of western blotting data. * P<0.05 and ** P<0.02 versus model (control). References
1. Yang G, Duan X, Lin D, Rapamycin-induced autophagy activity promotes bone fracture healing in rats: Expt Ther Med, 2015; 10; 1327-33
2. Einhorn T, Enhancement of fracture-healing: J Bone Joint Surg Am, 1995; 77; 940-56
3. O’Neill KR, Stutz CM, Mignemi NA, Micro-computed tomography assessment of the progression of fracture healing in mice: Bone, 2012; 50; 1357-67
4. Mizushima N, Levine B, Cuervo AM, Klionsky DJ, Autophagy fights disease through cellular self-digestion: Nature, 2008; 451; 1069-75
5. Levine B, Kroemer G, Autophagy in the pathogenesis of disease: Cell, 2008; 132; 27-42
6. Mathew R, Karp CM, Beaudoin B, Autophagy suppresses tumorigenesis through elimination of p62: Cell, 2009; 137; 1062-75
7. Hara T, Nakamura K, Matsui M, Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice: Nature, 2006; 441; 885-89
8. Komatsu M, Waguri S, Ueno T, Impairment of starvation-induced and constitutive autophagy in Atg7-deficient mice: J Cell Biol, 2005; 169; 425-34
9. Shibata M, Lu T, Furuya T, Regulation of intracellular accumulation of mutant Huntingtin by Beclin 1: J Biol Chem, 2006; 281; 14474-85
10. Su JC, Tseng PH, Hsu CY, RFX1-dependent activation of SHP-1 induces autophagy by a novel obatoclax derivative in hepatocellular carcinoma cells: Oncotarget, 2014; 5; 4909-19
11. Wullschleger S, Loewith R, Hall MN, TOR signaling in growth and metabolism: Cell, 2006; 124; 471-84
12. Dann SG, Selvaraj A, Thomas G, mTOR complex 1-S6K1 signaling: At the crossroads of obesity, diabetes and cancer: Trends Mol Med, 2007; 13; 252-59
13. Shigemitsu K, Tsujishita Y, Hara K, Regulation of translational effectors by amino acid and mammalian target of rapamycin signaling pathways. Possible involvement of autophagy in cultured hepatoma cells: J Biol Chem, 1999; 274; 1058-65
14. Sabers CJ, Martin MM, Brunn GJ, Isolation of a protein target of the FKBP12-rapamycin complex in mammalian cells: J Biol Chem, 1995; 270; 815-22
15. Qian Y, Zhong P, Liang D, A newly designed curcumin analog Y20 mitigates cardiac injury via anti inflammatory and anti oxidant actions in obese rats: PLoS One, 2015; 10; e0120215
16. Patel PB, Thakkar VR, Patel JS, Cellular effect of curcumin and citral combination on breast cancer cells: Induction of apoptosis and cell cycle arrest: J Breast Cancer, 2015; 18; 225-34
17. Jang EM, Choi MS, Jung UJ, Beneficial effects of curcumin on hyperlipidemia and insulin resistance in high fat fed hamsters: Metabolism, 2008; 57; 1576-83
18. Selim S, Bahattin KA, Alaaddin N, Effect of curcumin on bone healing: An experimental study in a rat model of femur fracture: Injury, 2019; 11; 1915-20
19. Zoncu R, Efeyan A, Sabatini DM, mTOR: From growth signal integration to cancer, diabetes and ageing: Nat Rev Mol Cell Biol, 2011; 12; 21-35
20. Lin NY, Stefanica A, Distler JH, Autophagy: A key pathway of TNF-induced inflammatory bone loss: Autophagy, 2013; 9; 1253-55
21. Zhang L, Guo YF, Liu YZ, Pathway-based genome-wide association analysis identified the importance of regulation-of-autophagy pathway for ultradistal radius BMD: J Bone Miner Res, 2010; 25; 1572-80
22. Chen K, Yang YH, Jiang SD, Jiang LS, Decreased activity of osteocyte autophagy with aging may contribute to the bone loss in senile population: Histochem Cell Biol, 2014; 142; 285-95
23. Stanfel MN, Shamieh LS, Kaeberlein M, Kennedy BK, The TOR pathway comes of age: Biochim Biophys Acta, 2009; 1790; 1067-74
24. Sciarretta S, Volpe M, Sadoshima J, Mammalian target of rapamycin signaling in cardiac physiology and disease: Circ Res, 2014; 114; 549-64
25. Mirzaei H, Longo VD, Acetyl-CoA synthetase is a conserved regulator of autophagy and life span: Cell Metab, 2014; 19; 555-57
26. Yu X, Guo Y, Kang Q, Luo C, Effects and mechanisms of mechanical stress on secondary fracture healing: Front Biosci (Landmark Ed), 2013; 18; 1344-48
27. Keramaris NC, Calori GM, Nikolaou VS, Fracture vascularity and bone healing: A systematic review of the role of VEGF: Injury, 2008; 39(Suppl 2); S45-57
28. Morrow PW, Tung HY, Hemmings HC, Rapamycin causes activation of protein phosphatase-2A1 and nuclear translocation of PCNA in CD4+ T cells: Biochem Biophys Res Commun, 2004; 323; 645-51
29. Barrera G, Landoni V, Martire-Greco D, Model of polymicrobial peritonitis that induces the proinflammatory and immunosuppressive phases of sepsis: Infect Immun, 2011; 79; 1280-88
30. Schwacha MG, Thobe BM, Daniel T, Hubbard WJ, Impact of thermal injury on wound infiltration and the dermal inflammatory response: J Surg Res, 2010; 158; 112-20
31. Rearte B, Maglioco A, Balboa L, Mifepristone (RU486) restores humoral and T cell-mediated immune response in endotoxin immunosuppressed mice: Clin Exp Immunol, 2010; 162; 568-77
32. Makarenkova VP, Bansal V, Matta BM, CD11b+/Gr-1+ myeloid suppressor cells cause T cell dysfunction after traumatic stress: J Immunol, 2006; 176; 2085-94
33. Bryk JA, Popovic PJ, Zenati MS, Nature of myeloid cells expressing arginase 1 in peripheral blood after trauma: J Trauma, 2010; 68; 843-52
34. Ichikawa M, Williams R, Wang L, S100A8/A9 activate key genes and signaling pathways in colon tumor progression: Mol Cancer Res, 2011; 9; 133-48
35. Cheng P, Corzo CA, Luetteke N, Inhibition of dendritic cell differentiation and accumulation of myeloid-derived suppressor cells in cancer is regulated by S100A9 protein: J Exp Med, 2008; 205; 2235-49
36. Jianping Z, Fengxue Y, Xiaolin X: Med Sci Monit, 2018; 24; 8306-14
37. Wan J, Feng J, Li F, Therapeutic advantages of internal fixation with Kirshner wire and bone grafting via limited tarsal sinus incision approach for displaced intra-articular calcaneal fractures of children: Med Sci Monit, 2018; 24; 7862-68
Figures
 Figure 1. (A, B) Effect of curcumin on rpS6-phosphorylation in fracture rat model. The rat calluses from curcumin treated or untreated groups were collected on day 14, 28, and 42 after fracture. Immunohistochemical analysis was performed for detection of rpS6-phosphorylation. * P<0.05, ** P<0.02 versus model (control) group. Magnification, 200×.
Figure 1. (A, B) Effect of curcumin on rpS6-phosphorylation in fracture rat model. The rat calluses from curcumin treated or untreated groups were collected on day 14, 28, and 42 after fracture. Immunohistochemical analysis was performed for detection of rpS6-phosphorylation. * P<0.05, ** P<0.02 versus model (control) group. Magnification, 200×. Figure 2. (A, B) Effect of curcumin on LC3-II expression in fracture rat model. The rat calluses from curcumin treated or untreated groups were collected on day 14, 28, and 42 after fracture. The LC3-II expression in the bone tissues was detected using immunofluorescence. * P<0.05, ** P<0.02 versus model (control) group. Magnification, 200×.
Figure 2. (A, B) Effect of curcumin on LC3-II expression in fracture rat model. The rat calluses from curcumin treated or untreated groups were collected on day 14, 28, and 42 after fracture. The LC3-II expression in the bone tissues was detected using immunofluorescence. * P<0.05, ** P<0.02 versus model (control) group. Magnification, 200×. Figure 3. Effect of curcumin on fracture repair in rats. The callus mineralization as well as size in fracture rat model was observed on day 14, 28, and 42 of fracture using x-ray imaging. * P<0.05, ** P<0.02 versus model (control) group.
Figure 3. Effect of curcumin on fracture repair in rats. The callus mineralization as well as size in fracture rat model was observed on day 14, 28, and 42 of fracture using x-ray imaging. * P<0.05, ** P<0.02 versus model (control) group. Figure 4. Effect of curcumin on osteoblast proliferation. The callus sections of sham, model, and curcumin treatment groups were analyzed for osteoblast proliferation on day 14, 28, and 42 after fracture using hematoxylin and eosin dyes. * P<0.05, ** P<0.02 versus model (control) group.
Figure 4. Effect of curcumin on osteoblast proliferation. The callus sections of sham, model, and curcumin treatment groups were analyzed for osteoblast proliferation on day 14, 28, and 42 after fracture using hematoxylin and eosin dyes. * P<0.05, ** P<0.02 versus model (control) group. Figure 5. Curcumin promotes PCNA and VEGF expression. (A) The PCNA and VEGF levels in calluses of rats were assessed by western blotting on day 14, 28, and 42 post fractures. (B) The quantification of PCNA and VEGF levels was made taking GAPDH as loading control. * P<0.05, ** P<0.02 versus model (control) group. PCNA – proliferating cell nuclear antigen; VEGF – vascular endothelial growth factor; GAPHD – glyceraldehyde 3-phosphate dehydrogenase.
Figure 5. Curcumin promotes PCNA and VEGF expression. (A) The PCNA and VEGF levels in calluses of rats were assessed by western blotting on day 14, 28, and 42 post fractures. (B) The quantification of PCNA and VEGF levels was made taking GAPDH as loading control. * P<0.05, ** P<0.02 versus model (control) group. PCNA – proliferating cell nuclear antigen; VEGF – vascular endothelial growth factor; GAPHD – glyceraldehyde 3-phosphate dehydrogenase. Figure 6. Effect of curcumin on expansion of CD11b+/Gr1+ cells. The rats with bone trauma were treated with curcumin or left untreated. The expansion of CD11b+/Gr1+ cells to (A) spleen, (B) bone marrow, and (C) blood was detected by flow cytometry. * P<0.5 and ** P<0.02 versus model (control).
Figure 6. Effect of curcumin on expansion of CD11b+/Gr1+ cells. The rats with bone trauma were treated with curcumin or left untreated. The expansion of CD11b+/Gr1+ cells to (A) spleen, (B) bone marrow, and (C) blood was detected by flow cytometry. * P<0.5 and ** P<0.02 versus model (control). Figure 7. Effect of curcumin on expression of arginase-1 in Gr-1+ cells. The rats were treated with curcumin immediately following trauma. (A) The expression of arginase-1 was measured by western blotting. (B) Quantification of western blotting data. * P<0.05 and ** P<0.02 versus model (control).
Figure 7. Effect of curcumin on expression of arginase-1 in Gr-1+ cells. The rats were treated with curcumin immediately following trauma. (A) The expression of arginase-1 was measured by western blotting. (B) Quantification of western blotting data. * P<0.05 and ** P<0.02 versus model (control). In Press
08 Mar 2024 : Animal Research
Modification of Experimental Model of Necrotizing Enterocolitis (NEC) in Rat Pups by Single Exposure to Hyp...Med Sci Monit In Press; DOI: 10.12659/MSM.943443
18 Apr 2024 : Clinical Research
Comparative Analysis of Open and Closed Sphincterotomy for the Treatment of Chronic Anal Fissure: Safety an...Med Sci Monit In Press; DOI: 10.12659/MSM.944127
08 Mar 2024 : Laboratory Research
Evaluation of Retentive Strength of 50 Endodontically-Treated Single-Rooted Mandibular Second Premolars Res...Med Sci Monit In Press; DOI: 10.12659/MSM.944110
11 Mar 2024 : Clinical Research
Comparison of Effects of Sugammadex and Neostigmine on Postoperative Neuromuscular Blockade Recovery in Pat...Med Sci Monit In Press; DOI: 10.12659/MSM.942773
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








