06 January 2021: Animal Study
Secondary Effects of the Rupture and Reconstruction of the Interosseous Talocalcaneal Ligament on the Peritalar Joints
Chen Liu1AE, Jing-xian Zhu2B, Yue-lin Hu2B, Chen Jiao2C, Qin-wei Guo2C, Ying-fang Ao2A*DOI: 10.12659/MSM.925292
Med Sci Monit 2021; 27:e925292
Abstract
BACKGROUND: The interosseous talocalcaneal ligament (ITCL) is the main soft-tissue contributor to subtalar joint stability. The role of ITCL reconstruction in retaining this stability is minimally reported. Therefore, we conducted this study to investigate the effects of rupture and reconstruction of the ITCL on the subtalar and peritalar joints.
MATERIAL AND METHODS: This experimental study randomly divided 72 rabbits into 3 equal groups of 24 rabbits each. Group I underwent reconstruction surgery, group II underwent resection, and group III was the control group. The cartilages between the talocrural and calcaneocrural joints, and between the subtalar and talonavicular joints on both sides were assessed by gross observation, ink staining, histology, and immunohistochemistry at weeks 4, 8, 16, and 32, postoperatively.
RESULTS: In group II, the quantitative ink staining analysis revealed degeneration of the articular cartilages on the talonavicular joint (T=2.070, P=0.038) and the posterior subtalar joint (T=2.121, P=0.034) compared with the 2 sides of the same rabbit at 4 and 8 postoperative weeks. Comparing the operated sides of all the groups showed the posterior subtalar joints (Hc=9.563, P=0.008) and talonavicular joints (Hc=9.714, P=0.008) had an obvious difference at postoperative week 4; and in the calcaneocrural joints (Hc=6.750, P=0.034), it was noticed at postoperative week 8. Histology and immunohistochemistry findings confirm these observations.
CONCLUSIONS: An ITCL resection can lead to the progressive degeneration of the talonavicular and posterior subtalar joints, while an ITCL reconstruction can be beneficial in restoring the stability of these joints, preventing or postponing their degeneration, and protecting the articular cartilages.
Keywords: Bone Diseases, Finite Element Analysis, Subtalar Joint, Calcaneus, Ligaments, Articular, Reconstructive Surgical Procedures, Rupture, Staining and Labeling
Background
The interosseous talocalcaneal ligament (ITCL) is considered the major stabilizer of subtalar joints [1,2]. It is located in the tarsal sinus between the medial and posterior talocalcaneal joints. It originates from the sulcus talus with an insertion at the sulcus calcaneus, which is located anterior and medial to the posterior talocalcaneal joint capsule [3].
A rupture of the ITCL is often caused by severe injury to the subtalar joints. Although this incident is considered infrequent and minimally reported in literature, such issues are sometimes misdiagnosed as the sinus tarsi syndrome (STS) [4–6]. With an increased understanding of the hindfoot structure along with the recent advances in radiologic and arthroscopic diagnostics, the distinguishing characters of ITCL rupture and STS are gradually being recognized [5,7,8]. Despite the fact that researchers have been investigating other novel treatment options for such conditions recently, their results have not provided clear-cut evidence of their efficacy, with a minimal number of studied individuals [5,9,10].
The long-term outcomes of ITCL rupture are not clearly recognized; this is a key factor influencing the treatment strategy. The aim of the current investigation is to identify the degree of secondary degeneration of the subtalar and peritalar joints in ITCL rupture as well as comprehend the protective effects of ITCL reconstruction. This research was conducted to give helpful insights into novel treatment strategies for ITCL rupture. For the purposes of the present study, we wanted to investigate the effects of ITCL resection and reconstruction on subtalar joint stability. We hypothesized that an ITCL resection would negatively impact the stability of this joint, while an ITCL reconstruction could result in beneficial effects and successfully lead to the restoration of joint stability. The null hypothesis was that there would be no difference between the 2 procedures on restoring subtalar joint stability, while the alternative hypothesis was that there would be a statistically significant difference between the 2 procedures in terms of subtalar stability.
Material and Methods
STATISTICAL ANALYSIS:
All statistical analyses were conducted using the Statistical Package for Social Sciences (IBM SPSS Statistics Version 23, SPSS US Inc., Chicago, Illinois, US). Mankin scores at each stage in all the groups were presented as means. We used the Kruskal-Wallis H test to determine the statistically significant differences of continuous variables between 2 or more groups [15]. We used the non-parametric Rank Sum test (Wilcoxon test) to estimate the statistical differences between the 2 paired groups. P<0.05 was considered statistically significant.
The protocol for this experimental study was reviewed and approved by the Institutional Review Board’s Animal Ethics Committee at our institution (0576).
Results
GROSS FINDINGS:
During the 4-week assessment period, group I (ITCL reconstruction) revealed good tension with no injury to the cartilage. In group II (resection group), obvious hyperemia with some hyperplasia of the synovium was noted; the color was normal on the cartilage surface while fissures and pits were not observed at week 4. Group III (control) revealed no hyperemia or edema of the synovium along with injuries to the cartilage.
During the 8-week assessment period, group I showed similar observations comparable to week 4 while group II showed hyperemia and hyperplasia of the synovia of the joints, and the surfaces of some cartilages revealed fissures. In group III, no hyperemia or edema was observed.
During the 16-week assessment period, group I showed no injury to the cartilage with good tension, while group II revealed slight hyperemia and edema of the synovium, and a spur had formed mainly on the posterior subtalar joint. In group III, no changes were observed.
During the 32-week assessment period, group I revealed no change while group II revealed some hyperemia/edema of the synovium with obvious hyperplasia of the synovium. Moreover, fissures, fibrosis, and defects were visible on the surfaces of some cartilages, while florid spur formation was mainly observed at the posterior subtalar, talocrural, and talonavicular joints (Figure 2A–2C). In group III, no changes were observed.
INK STAINING:
We performed the Wilcoxon test to compare both sides (operated and unoperated) of the same rabbit in group II (resection) and group III (control). The differences in the ink-staining scores presented as Yoshioka scores between the operated side and the contralateral side for all joints did not reach statistical significance in any assessment period (P>0.05). In group II, we noted differences between the talonavicular and posterior subtalar joints during postoperative weeks 4 and 8, respectively. On the other hand, statistically significant differences in the Yoshioka score (ink score) were noted between the talonavicular joint and the anterior subtalar joint (anterior calcaneal surface of talus [aCa]) in the 32nd postoperative week (Table 1). A comparison of the operated sides of all 3 groups by the Kruskal-Wallis test showed results (differences) similar to the ink-staining scores of the examined joints (Table 2).
HISTOLOGY AND IMMUNOHISTOCHEMISTRY:
At all assessment periods, the articular cartilages of the 2 sides of the control group and the non-operated side of groups I and II were mostly normal, their Mankin scores were 0–1. The results of the HE staining revealed a smooth surface layer, well-arranged and well-distributed chondrocytes, the lack of a cluster, a complete tidemark, and an even distribution of the toluidine blue stain implying that the degree of degeneration was minimal.
The immunohistochemistry staining showed that type I collagen was localized only in the subchondral bone, type II collagen was evenly distributed, and type III collagen was absent. The histological results showed the cartilage of the operated side in group I was mostly normal.
In group II, the chondral injuries became more obvious as the postoperative duration increased. The severity of the chondral injuries varied in the different articular surfaces of the leg, among which the injuries in the posterior subtalar joint and the talonavicular joint were the most concerning (high Mankin degeneration score). The alterations observed in the chondral histology of the calcaneus surface of the posterior subtalar joint at every follow-up time point are presented in Table 3.
In week 4, the result of the chondral histology was close to normal or gently injured (Mankin scores 2–5). The surface layer was not flat and showed a small fissure or a little cluster of chondrocytes. A loss of toluidine blue was seen on the surface layer indicating the failure of dye uptake implying cellular death. We noted positive staining of type I collagen at the surface layer; type II collagen was noted inside the cartilage and around the chondrocyte, respectively.
In week 8, the proportion and degree of chondral injury increased (Mankin scores 6–8). The changes noted were that the surface layer was not flat, a small fissure was present; a little cluster of chondrocytes was found. The staining of toluidine blue was not well-distributed. Type I collagen was detected in the surface layer, type II collagen was distributed in the middle and deep layers, and type III collagen was only found in the injured area.
In week 16, the proportion and degree of chondral injuries increased to a greater degree (Mankin scores 9–11). The surface layer of the cartilage began to show fibrosis and the gap in the middle layer, which was a small fissure in week 4, was now deep. The arrangement of cells was disturbed, the tidemark was irregular, and the number of clusters of chondrocytes had increased. The loss of toluidine blue was seen in the surface and middle layers. Type I collagen showed diffuse distribution, type II collagen was mainly distributed in the middle and deep layers, type III collagen was mainly distributed in the moderate and deep layers.
In week 32, in group II, 1 rabbit died due to a rapid intravenous anesthetic injection, and 1 suffered a postoperative joint infection. After eliminating these, another 2 rabbits were selected for supplementation to the study samples. The scores of chondral injuries became higher, and severe chondral injuries were seen (Mankin scores 12–14). In the deep layers of the cartilage, there were large areas of fibrosis. In the same context, the number of cells decreased significantly, the tidemark was not apparent, and the calcified cartilage layer was undistinguishable (Figure 3A–3C). The loss of toluidine blue was observed in a majority of the cartilage (Figure 3B). The staining of collagen types I and III were strongly and diffusely positive; type II collagen was distributed unevenly in the deep layers (Figure 3C–3E).
Discussion
The ITCL resection resulted in a significant degeneration of the articular cartilages of the study specimens during the early postoperative assessment period. The ITCL reconstruction revealed better outcomes in terms of less cartilage degeneration, good tension, and no gross injuries to the cartilage during all the assessment periods.
As yet, there is no agreement on the mechanism of the movement of subtalar joints, and thus, the effect of the ITCL on the stability of subtalar joints is uncertain [10,16]. In a series of
In the present study, the changes in the cartilage could be observed microscopically by ink staining while gross changes emerged after the ink staining. The method was validated in the knee joint [21]. We used it to evaluate the cartilages of the subtalar and peritalar joints, which can reveal the area of chondral injury along with the depth of the injury. The HE and toluidine blue staining display the histological changes in the microstructures of the cartilage (ie collagen fibrils and proteoglycans). The Mankin scoring system assesses the structure, cell, matrix, and tidemark at a cellular level. Immunohistochemistry staining was used to investigate collagen changes in the cartilage.
Clinical studies show that cartilage injuries commonly occur with an ITCL injury in a high proportion of the patients [5,7]. The results of our study showed that the talonavicular joint and the posterior subtalar joint were the first joints to be affected after an ITCL resection. Meanwhile, the severity of the degeneration was associated with the postsurgical duration. No obvious enantiomorphous changes were found. The injury on the 2 surfaces of the same articular side showed unequable behavior. The distal surfaces had a tendency to be more severely injured than the proximal surfaces. The underlying reason for this is still unclear.
The talonavicular joint is an ovoid joint, where the acetabulum pedis sockets the talar head. As defined by Kilikian et al. [22], the motion generated by a male ovoid surface moving on a female surface is that of a slide, roll, and spin. The roll is in the opposite direction of the slide [23]. Therefore, when the talus extends and flexes, the roll of the talar head in the acetabulum pedis changes to a slide. At the same time, in the sagittal vector, the talar displacement gradient changes from up to down in a superior-to-inferior direction. When the talus extends and flexes, the impingement occurs. The 2 surfaces are no longer consistent, which is the reason for faster degeneration of the talonavicular.
The subtalar joint also showed rapid degeneration. It bears 50% of the weight transferred from the crus in humans [24]. In rabbits, it also bears a large part of their weight. The changes in the talar displacement grads in the vertical and sagittal vectors lead to disagreement and degeneration of the 2 surfaces. When the ITCL ruptured, the talus descended and the calcaneocrural joint bore more weight than an intact foot. That could be the reason that the degree of calcaneocrural degeneration was faster than that observed in the talocrural joint. The anterior subtalar joint is in the sagittal plane and barely bears any weight in rabbits. A strong ligamentous capsule around the medial subtalar joint on the sustentaculum tali passes horizontally and ends on the medial aspect of the talus. This ligamentous capsule is similar to but much broader than the plantar calcaneonavicular (spring) ligament in humans. The structure, which shows fibrocartilaginous metaplasia in the area articulating the medial subtalar joint of the talus in a normal rabbit foot, plays a fundamental role in maintaining the talocalcaneonavicular connections. This contributes to sharing a great part of the weight transferred from the talus, and prevents its plantar and medial dislocation. This weakens the impact of the ITCL rupture on the anterior and medial subtalar joints; thus, the 2 joints showed little and delayed degeneration.
Reconstruction of the ITCL with a portion of the Achilles’ tendon inserted into a tubed Leeds-Keio artificial ligament was first reported by Kato [25]. Other reports showed satisfactory results of an ITCL reconstruction [5,26]. Due to the diagnostic difficulties (e.g. clinical measures do not reflect isolated subtalar instability, misdiagnosis, and radiographs have high false positive rates of ITCL ruptures), the ITCL rupture was not thoroughly studied before this. Magnetic resonance imaging (MRI) [27–29] and stressed radiography [9,30] provided valuable evidence to reach a diagnosis of ITCL rupture. Arthroscopy [5,7,8] could be the most reliable method in the diagnosis of this disease so far.
This study provided an animal model for a reconstruction of the ITCL with an autograft of the semitendinosus. The semitendinosus is a regular autograft for tendon transplant [31]. One strand of it is similar to the ITCL in diameter and intensity according to the results of our study for ligamentous biomechanics [32]. A steel-wire button referring to the EndoButton (Smith & Nephew, Andover, MA, USA) [33] was used to fix the talar terminal, which provided adequate tension for fixation and a decreased the risk of talar necrosis compared to the bone-bridge fixation of the calcaneal terminal. In group I, none of the rabbits showed talar necrosis at any postoperative assessment point. The best choice for the autograft is the local tendon. We tried to construct it with 2 strands of the semitendinosus muscle tendon, in order to preserve the distal termination and fix the calcaneal side of the bone bridge. This can reduce the length of the incision on the knee and decrease the risk of infection by avoiding an implant button. We selected the semitendinosus muscle, as a dynamic stabilizer would have been destroyed after the posterior tibial tendon was cut, and it could affect the results of this experiment. It is worth mentioning that several factors could have affected the results of this study. During the arthrotomy of the subtalar joints, bleeding and injury to other structures can result in histological changes noted in the articular cartilage. A large number of cytokines in the effused blood could lead to the degeneration of the knee. The bone-tendon interface had no fixation and there was no healing between the interfaces during the early stage after reconstruction [34]. Therefore, when the foot moved, the graft shifted in the bone tunnel (Bungee’s effect and windshield wiper effect) [35], which affected the reconstruction. Perhaps the reconstruction could not completely simulate the biomechanics of the original ITCL.
Conclusions
An ITCL resection can lead to progressive degeneration of the subtalar and peritalar joints. The reconstruction of the ITCL is an effective option in recovering subtalar stability, protecting the articular cartilages, and preventing or delaying the degeneration of joints. Despite the fact that the biomechanics of human joints are not definitively the same as those of rabbits, this study provides a helpful insight into the importance of the ITCL for the stability of the subtalar and peritalar joints. Therefore, more robust studies on human subjects would provide higher-quality evidence about the role of ITCL reconstruction in retaining the subtalar joint stability.
Figures
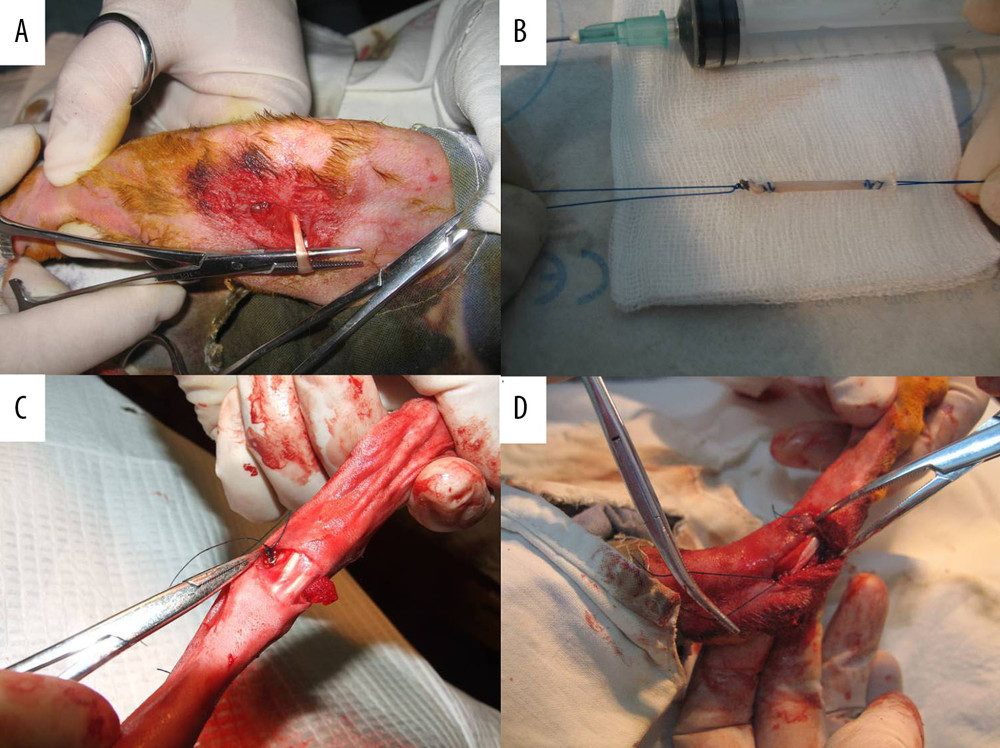 Figure 1. (A) The semitendinosus was located and cut approximately 3 cm from the border of the tendon muscle for attachment to the tibia. (B) The semitendinosus strand was woven with a 3-0 polyester thread at both ends. (C) The talar terminal was fixed with a 1.5 cm-diameter steel-wire button. (D) One strand of a thread from the calcaneal terminal was passed through the tunnel and fastened with another thread to form a bone-bridge fixation while the graft kept tension.
Figure 1. (A) The semitendinosus was located and cut approximately 3 cm from the border of the tendon muscle for attachment to the tibia. (B) The semitendinosus strand was woven with a 3-0 polyester thread at both ends. (C) The talar terminal was fixed with a 1.5 cm-diameter steel-wire button. (D) One strand of a thread from the calcaneal terminal was passed through the tunnel and fastened with another thread to form a bone-bridge fixation while the graft kept tension. 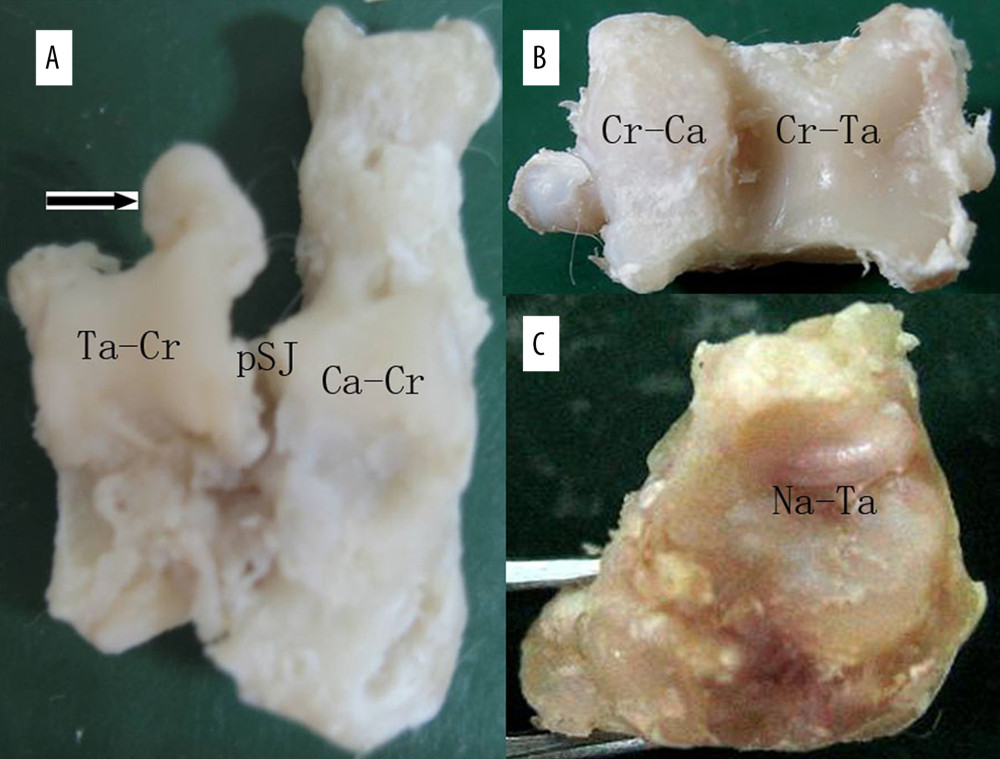 Figure 2. Specimen sacrificed at 32 weeks after resection surgery in group II: (A) A specimen of the talus and calcaneus. The black arrow shows the enormous osteophyte at the posterior subtalar joint. (B) A specimen of the crus. The degeneration of the calcaneal surface of the crus (Cr-Ca) was more severe than the talar surface of the crus (Cr-Ta). (C) A specimen of the talar surface of the navicular (Na-Ta). Ca-Cr – crural surface of the calcaneus; Cr-Ca – calcaneal surface of crus; Cr-Ta – talar surface of the crus; Na-Ta – talar surface of the navicular; pSJ – posterior subtalar joint; Ta-Cr – crural surface of the talus.
Figure 2. Specimen sacrificed at 32 weeks after resection surgery in group II: (A) A specimen of the talus and calcaneus. The black arrow shows the enormous osteophyte at the posterior subtalar joint. (B) A specimen of the crus. The degeneration of the calcaneal surface of the crus (Cr-Ca) was more severe than the talar surface of the crus (Cr-Ta). (C) A specimen of the talar surface of the navicular (Na-Ta). Ca-Cr – crural surface of the calcaneus; Cr-Ca – calcaneal surface of crus; Cr-Ta – talar surface of the crus; Na-Ta – talar surface of the navicular; pSJ – posterior subtalar joint; Ta-Cr – crural surface of the talus. 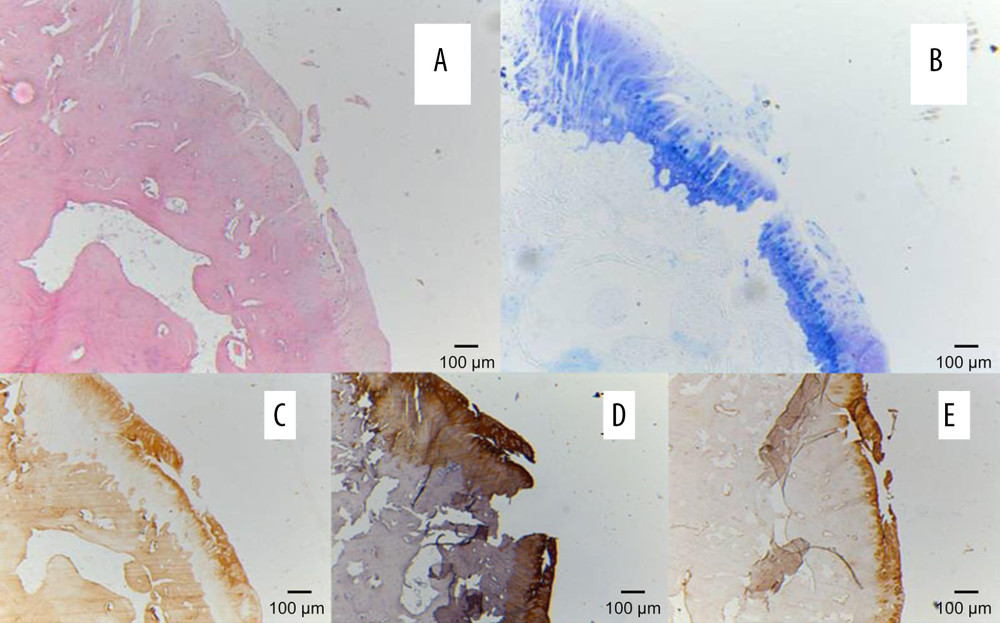 Figure 3. Pathology results of the talar surface of the calcaneus (Ca-Ta) of the posterior subtalar joint in group II: (A) Fibrosis and a cluster of chondrocytes seen in the cartilage of the medial condyle (HE×5). (B) Loss of toluidine blue is seen in majority of the cartilage. (C) Type I collagen strongly and diffusely positive at 32 weeks in group I (immunohistochemistry ×5). (D) Type II collagen was uneven in the deep layers (immunohistochemistry ×5). (E) Type III collagen was strongly and diffusely positive (immunohistochemistry ×5).
Figure 3. Pathology results of the talar surface of the calcaneus (Ca-Ta) of the posterior subtalar joint in group II: (A) Fibrosis and a cluster of chondrocytes seen in the cartilage of the medial condyle (HE×5). (B) Loss of toluidine blue is seen in majority of the cartilage. (C) Type I collagen strongly and diffusely positive at 32 weeks in group I (immunohistochemistry ×5). (D) Type II collagen was uneven in the deep layers (immunohistochemistry ×5). (E) Type III collagen was strongly and diffusely positive (immunohistochemistry ×5). Tables
Table 1. Results of 2 sides of the 12 articular surfaces compared at every stage in group II by the Wilcoxon test (n=6).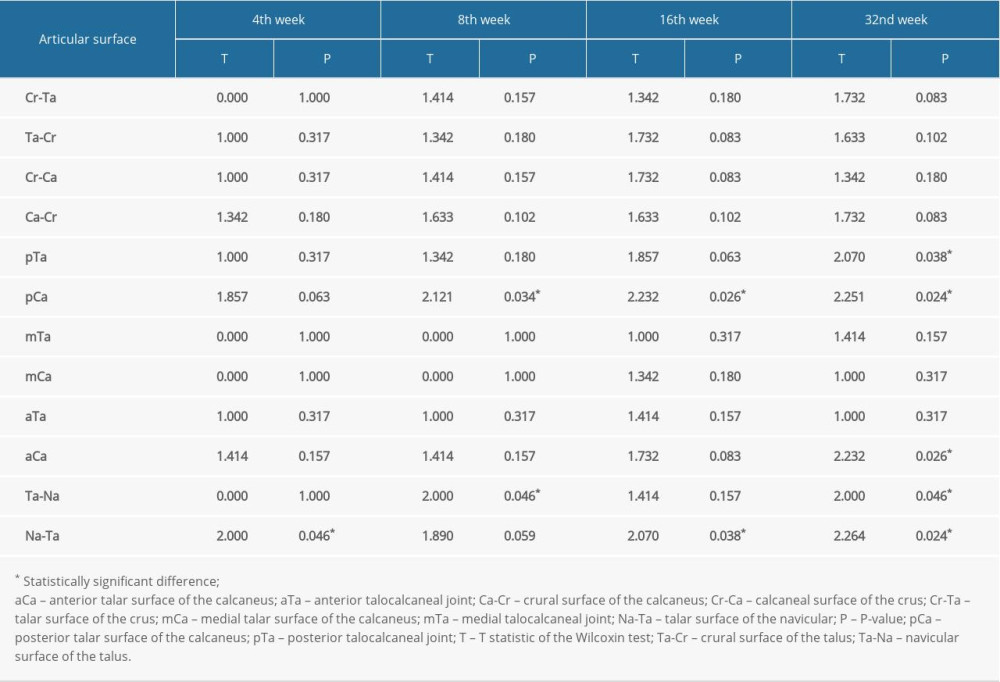 Table 2. Results of the operated side of the 12 articular surfaces compared at every stage in the 3 groups by the Kruskal-Wallis test (n=6).
Table 2. Results of the operated side of the 12 articular surfaces compared at every stage in the 3 groups by the Kruskal-Wallis test (n=6).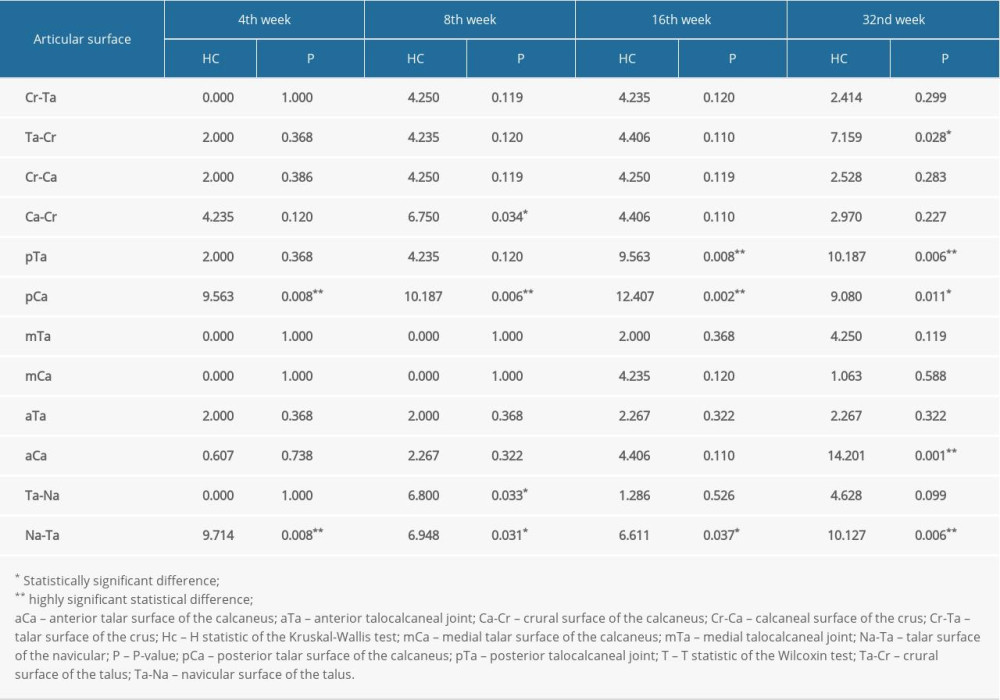 Table 3. Mankin scores for the posterior talar surface of the calcaneus and the talar surface of the navicular at every stage in groups I and II.
Table 3. Mankin scores for the posterior talar surface of the calcaneus and the talar surface of the navicular at every stage in groups I and II.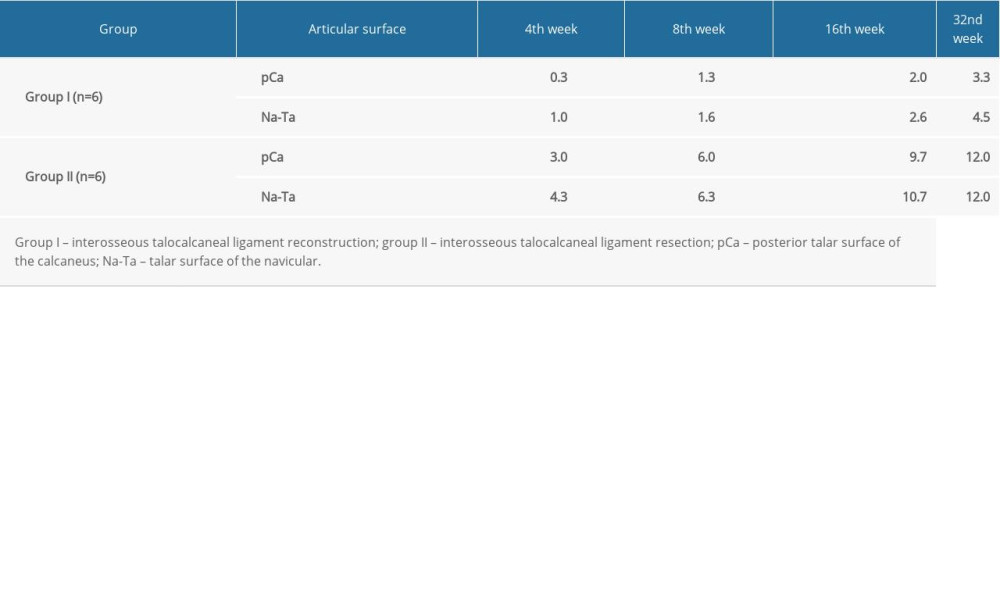
References
1. Poonja AJ, Hirano M, Khakimov D, Anatomical study of the cervical and interosseous talocalcaneal ligaments of the foot with surgical relevance: Cureus, 2017; 9; e1382
2. Tochigi Y, Effect of Arch Supports on Ankle-Subtalar Complex Instability: A biomechanical experimental study: Foot Ankle Int, 2003; 24; 634-39
3. Li S-Y, Hou Z-D, Zhang P, Ligament structures in the tarsal sinus and canal: Foot Ankle Int, 2013; 34; 1729-36
4. Frey C, Feder KS, DiGiovanni C, Arthroscopic evaluation of the subtalar joint: Does sinus tarsi syndrome exist?: Foot Ankle Int, 1999; 20; 185-91
5. Liu C, Jiao C, Hu Y, Interosseous talocalcaneal ligament reconstruction with hamstring autograft under subtalar arthroscopy: Case report: Foot Ankle Int, 2011; 32; 1089-94
6. Pisani G, Pisani PC, Parino E, Sinus tarsi syndrome and subtalar joint instability: Clin Podiatr Med Surg, 2005; 22; 63-77
7. Lee KB, Bai LB, Song EK, Subtalar arthroscopy for sinus Tarsi syndrome: Arthroscopic findings and clinical outcomes of 33 consecutive cases: Arthroscopy, 2008; 24; 1130-34
8. Lee K-B, Bai L-B, Park J-G, Efficacy of MRI versus arthroscopy for evaluation of sinus tarsi syndrome: Foot Ankle Int, 2008; 29; 1111-16
9. Barg A, Tochigi Y, Amendola A, Subtalar instability: diagnosis and treatment: Foot Ankle Int, 2012; 33; 151-60
10. Choisne J, Ringleb SI, Samaan MA, Influence of kinematic analysis methods on detecting ankle and subtalar joint instability: J Biomech, 2012; 45; 46-52
11. Lui TH, Chan KB, Chan LK, Portal safety and efficacy of anterior subtalar arthroscopy: A cadaveric study: Knee Surg Sports Traumatol Arthrosc, 2010; 18; 233-37
12. Yoshioka M, Shimizu C, Harwood FL, The effects of hyaluronan during the development of osteoarthritis: Osteoarthritis Cartilage, 1997; 5; 251-60
13. Mankin HJ, Biochemical and metabolic aspects of osteoarthritis: Orthop Clin North Am, 1971; 2; 19-31
14. Kraus VB, Huebner JL, DeGroot J, Bendele A, The OARSI histopathology initiative – recommendations for histological assessments of osteoarthritis in the guinea pig: Osteoarthritis Cartilage, 2010; 18(Suppl 3); S35-52
15. Kruskal WH, Wallis WA, Use of ranks in one-criterion variance analysis: Journal of the American Statistical Association, 1952; 47; 583-621
16. Bonnel F, Toullec E, Mabit C, Tourné Y, Chronic ankle instability: Biomechanics and pathomechanics of ligaments injury and associated lesions: Orthop Traumatol Surg Res, 2010; 96; 424-32
17. Jung H-G: Foot and ankle disorders: an illustrated reference, 2016, Springer
18. Kjærsgaard-Andersen P, Wethelund J-O, Helmig P, Søballé K, The stabilizing effect of the ligamentous structures in the sinus and canalis tarsi on movements in the hindfoot: An experimental study: Am J Sports Med, 1988; 16; 512-16
19. Tochigi Y, Amendola A, Rudert MJ, The role of the interosseous talocalcaneal ligament in subtalar joint stability: Foot Ankle Int, 2004; 25; 588-96
20. Knudson GA, Kitaoka HB, Lu C-L, Subtalar joint stability: Talocalcaneal interosseous ligament function studied in cadaver specimens: Acta Orthop Scand, 1997; 68; 442-46
21. Choisne J, Hoch MC, Bawab S, Alexander I, Ringleb SI, The effects of a semi-rigid ankle brace on a simulated isolated subtalar joint instability: J Orthop Res, 2013; 31; 1869-75
22. Kelikian AS, Sarrafian SK: Sarrafian’s anatomy of the foot and ankle: Descriptive, topographic, functional, 2011, Lippincott Williams & Wilkins
23. Mansfield PJ, Neumann DA: Essentials of kinesiology for the physical therapist assistant e-book, 2018, Elsevier Health Sciences
24. Krähenbühl N, Horn-Lang T, Hintermann B, Knupp M, The subtalar joint: A complex mechanism: EFORT Open Rev, 2017; 2; 309-16
25. Kato T, The diagnosis and treatment of instability of the subtalar joint: J Bone Joint Surg Br, 1995; 77; 400-6
26. Xu C, Li M, Wang C, Hua L, Biomechanical comparison of tenodesis reconstruction for subtalar instability: A finite element analysis: BMC Musculoskelet Disord, 2020; 21(1); 669
27. Faymonville C, Andermahr J, Seidel U, Compartments of the foot: Topographic anatomy: Surg Radiol Anat, 2012; 34; 929-33
28. Lektrakul N, Chung CB, Lai Y-M, Tarsal sinus: arthrographic, MR imaging, MR arthrographic, and pathologic findings in cadavers and retrospective study data in patients with sinus tarsi syndrome: Radiology, 2001; 219; 802-10
29. Thacker P, Mardis N, Ligaments of the tarsal sinus: Improved detection, characterisation and significance in the paediatric ankle with 3-D proton density MR imaging: Pediatr Radiol, 2013; 43; 196-201
30. Kim TH, Moon SG, Jung H-G, Kim NR, Subtalar instability: Imaging features of subtalar ligaments on 3D isotropic ankle MRI: BMC Musculoskelet Disord, 2017; 18; 475
31. Usuelli FG, D’Ambrosi R, Manzi L, Clinical outcomes and return to sports in patients with chronic Achilles tendon rupture after minimally invasive reconstruction with semitendinosus tendon graft transfer: Joints, 2017; 5; 212-16
32. Goyal D, Yadav S, Vidyasagar J, Clinical experience with woven and parallel hamstring-tendon anterior cruciate ligament reconstruction: Knee Surg Relat Res, 2019; 31; 4
33. Ohnishi Y, Chang A, Utsunomiya H, Arthroscopic technique to reduce suture button migration during anterior cruciate ligament reconstruction procedure: Arthrosc Tech, 2017; 6; e1927-31
34. Lu H, Chen C, Xie S, Tendon healing in bone tunnel after human anterior cruciate ligament reconstruction: A systematic review of histological results: J Knee Surg, 2019; 32; 454-62
35. Giorgio N, Moretti L, Pignataro P, Correlation between fixation systems elasticity and bone tunnel widening after ACL reconstruction: Muscles Ligaments Tendons J, 2016; 6; 467-72
Figures
 Figure 1. (A) The semitendinosus was located and cut approximately 3 cm from the border of the tendon muscle for attachment to the tibia. (B) The semitendinosus strand was woven with a 3-0 polyester thread at both ends. (C) The talar terminal was fixed with a 1.5 cm-diameter steel-wire button. (D) One strand of a thread from the calcaneal terminal was passed through the tunnel and fastened with another thread to form a bone-bridge fixation while the graft kept tension.
Figure 1. (A) The semitendinosus was located and cut approximately 3 cm from the border of the tendon muscle for attachment to the tibia. (B) The semitendinosus strand was woven with a 3-0 polyester thread at both ends. (C) The talar terminal was fixed with a 1.5 cm-diameter steel-wire button. (D) One strand of a thread from the calcaneal terminal was passed through the tunnel and fastened with another thread to form a bone-bridge fixation while the graft kept tension. Figure 2. Specimen sacrificed at 32 weeks after resection surgery in group II: (A) A specimen of the talus and calcaneus. The black arrow shows the enormous osteophyte at the posterior subtalar joint. (B) A specimen of the crus. The degeneration of the calcaneal surface of the crus (Cr-Ca) was more severe than the talar surface of the crus (Cr-Ta). (C) A specimen of the talar surface of the navicular (Na-Ta). Ca-Cr – crural surface of the calcaneus; Cr-Ca – calcaneal surface of crus; Cr-Ta – talar surface of the crus; Na-Ta – talar surface of the navicular; pSJ – posterior subtalar joint; Ta-Cr – crural surface of the talus.
Figure 2. Specimen sacrificed at 32 weeks after resection surgery in group II: (A) A specimen of the talus and calcaneus. The black arrow shows the enormous osteophyte at the posterior subtalar joint. (B) A specimen of the crus. The degeneration of the calcaneal surface of the crus (Cr-Ca) was more severe than the talar surface of the crus (Cr-Ta). (C) A specimen of the talar surface of the navicular (Na-Ta). Ca-Cr – crural surface of the calcaneus; Cr-Ca – calcaneal surface of crus; Cr-Ta – talar surface of the crus; Na-Ta – talar surface of the navicular; pSJ – posterior subtalar joint; Ta-Cr – crural surface of the talus. Figure 3. Pathology results of the talar surface of the calcaneus (Ca-Ta) of the posterior subtalar joint in group II: (A) Fibrosis and a cluster of chondrocytes seen in the cartilage of the medial condyle (HE×5). (B) Loss of toluidine blue is seen in majority of the cartilage. (C) Type I collagen strongly and diffusely positive at 32 weeks in group I (immunohistochemistry ×5). (D) Type II collagen was uneven in the deep layers (immunohistochemistry ×5). (E) Type III collagen was strongly and diffusely positive (immunohistochemistry ×5).
Figure 3. Pathology results of the talar surface of the calcaneus (Ca-Ta) of the posterior subtalar joint in group II: (A) Fibrosis and a cluster of chondrocytes seen in the cartilage of the medial condyle (HE×5). (B) Loss of toluidine blue is seen in majority of the cartilage. (C) Type I collagen strongly and diffusely positive at 32 weeks in group I (immunohistochemistry ×5). (D) Type II collagen was uneven in the deep layers (immunohistochemistry ×5). (E) Type III collagen was strongly and diffusely positive (immunohistochemistry ×5). Tables
 Table 1. Results of 2 sides of the 12 articular surfaces compared at every stage in group II by the Wilcoxon test (n=6).
Table 1. Results of 2 sides of the 12 articular surfaces compared at every stage in group II by the Wilcoxon test (n=6). Table 2. Results of the operated side of the 12 articular surfaces compared at every stage in the 3 groups by the Kruskal-Wallis test (n=6).
Table 2. Results of the operated side of the 12 articular surfaces compared at every stage in the 3 groups by the Kruskal-Wallis test (n=6). Table 3. Mankin scores for the posterior talar surface of the calcaneus and the talar surface of the navicular at every stage in groups I and II.
Table 3. Mankin scores for the posterior talar surface of the calcaneus and the talar surface of the navicular at every stage in groups I and II. Table 1. Results of 2 sides of the 12 articular surfaces compared at every stage in group II by the Wilcoxon test (n=6).
Table 1. Results of 2 sides of the 12 articular surfaces compared at every stage in group II by the Wilcoxon test (n=6). Table 2. Results of the operated side of the 12 articular surfaces compared at every stage in the 3 groups by the Kruskal-Wallis test (n=6).
Table 2. Results of the operated side of the 12 articular surfaces compared at every stage in the 3 groups by the Kruskal-Wallis test (n=6). Table 3. Mankin scores for the posterior talar surface of the calcaneus and the talar surface of the navicular at every stage in groups I and II.
Table 3. Mankin scores for the posterior talar surface of the calcaneus and the talar surface of the navicular at every stage in groups I and II. In Press
12 Mar 2024 : Database Analysis
Risk Factors of Age-Related Macular Degeneration in a Population-Based Study: Results from SHIP-TREND-1 (St...Med Sci Monit In Press; DOI: 10.12659/MSM.943140
12 Mar 2024 : Clinical Research
Preoperative Blood Transfusion Requirements for Hemorrhoidal Severe Anemia: A Retrospective Study of 128 Pa...Med Sci Monit In Press; DOI: 10.12659/MSM.943126
12 Mar 2024 : Clinical Research
Tissue Inhibitors of Metalloproteinase 1 (TIMP-1) and 3 (TIMP-3) as New Markers of Acute Kidney Injury Afte...Med Sci Monit In Press; DOI: 10.12659/MSM.943500
12 Mar 2024 : Review article
Optimizing Behçet Uveitis Management: A Review of Personalized Immunosuppressive StrategiesMed Sci Monit In Press; DOI: 10.12659/MSM.943240
Most Viewed Current Articles
17 Jan 2024 : Review article
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
14 Dec 2022 : Clinical Research
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387
01 Jan 2022 : Editorial
Editorial: Current Status of Oral Antiviral Drug Treatments for SARS-CoV-2 Infection in Non-Hospitalized Pa...DOI :10.12659/MSM.935952
Med Sci Monit 2022; 28:e935952








