05 October 2020: Clinical Research
Are We Meeting the Current Standards of Consent for Anesthesia? An International Survey of Clinical Practice
Tomas Jovaisa1ACDEF, Ieva Norkiene2ABCEF*, Juri Karjagin3AB, Iveta Golubovska4BC, Lukas Gambickas2BCE, Migle Kalinauskaite2BCE, Evaldas Kauzonas2BC, Dhuleep Wijayatilake5ABDEDOI: 10.12659/MSM.925905
Med Sci Monit 2020; 26:e925905
Abstract
BACKGROUND: International application of existing guidelines and recommendations on anesthesia-specific informed consent is limited by differences in healthcare and legal systems. Understanding national and regional variations is necessary to determine future guidelines.
MATERIAL AND METHODS: Anonymous paper surveys on their practices regarding anesthesia-specific patient informed consent were sent to anesthesiologists in Estonia, Latvia, and Lithuania.
RESULTS: A total of 233 responses were received, representing 36%, 26%, and 24% of the practicing anesthesiologists in Lithuania, Latvia, and Estonia, respectively. Although 85% of responders in Lithuania reported using separate forms to secure patient informed consent for anesthesia, 54.5% of responders in Estonia and 50% in Latvia reported using joint forms to secure patient informed consent for surgery and anesthesia. Incident rates were understated by 14.2% of responders and overstated by 66.4% (P<0.001), with the latter frequently quoting incident rates that are several to tens of times higher than those published internationally. Physicians obtaining consent in the outpatient setting were more satisfied with the process than those obtaining consent on the day of surgery, with 62.5% and 42.6%, respectively, agreeing that the informed consent forms provided a satisfactory description of complications (P=0.03). Patients were significantly less likely to read consent information when signing forms on the day of surgery than at earlier times (8.5% vs. 67.5%, P<0.001). Only 46.2% of respondents felt legally protected by the current consent process.
CONCLUSIONS: Anesthesia-specific informed patient consent practices differ significantly in the 3 Baltic states, with these practices often falling short of legal requirements. Efforts should be made to improving information accuracy, patient autonomy, and compliance with existing legal standards.
Keywords: Anesthesia, Consent Forms, Informed Consent, Anesthesiology, Estonia, Female, Humans, Latvia, Lithuania, Male, Physicians
Background
The medicolegal aspects of anesthesia practice are becoming increasingly challenging, with anesthesia-specific informed patient consent (ASIC) becoming more frequent. Different traditions, practices, and opinions result in significant differences in obtaining consent worldwide. Although the International Standards for a Safe Practice of Anesthesia, recently published by the World Health Organization and the World Federation of Societies of Anesthesiologists, highly recommend written ASIC, these recommendations do not specify a satisfactory standard of consent [1]. Guidelines and recommendations are available at the national or regional levels [2], but their broader applicability is limited by differences in legal systems, traditions, and logistics dictated by different healthcare models. Major differences are apparent even within the European Union, with the starkest example being the need for consent itself, with some countries accepting the concept of ‘implied consent’ and do not require formal and separate ASIC. Although most surgical procedures require anesthesia, making consent to anesthesia an inseparable part of patient agreement to undergo these procedures, this somewhat paternalistic approach ignores the multiple choices and combinations of anesthesia, each of which has advantages and disadvantage. This approach also limits patient autonomy, and leaves healthcare professionals open to legal challenges. The situation is not helped by evidence showing that patients may not perceive complications of anesthesia as being significant and are likely to recollect less information related to anesthesia than to surgical risks [3]. Nevertheless, the majority of patients prefer to be well informed of the risks of anesthesia, even at the cost of potentially increased anxiety [4]. Even when separate ASIC is obtained, the process remains controversial. For example, consent obtained on the day of surgery could be challenged at a later date, as it was obtained while the patient is under stress, and the patient did not have sufficient time to determine all the options, ask questions, or review or withdraw consent [5]. This is also mirrored by patient preferences, as fewer than half of patients would prefer to be informed on the day of surgery [4]. Unsurprisingly, concerns have arisen about the medicolegal implications of the current state of affairs, as anesthesiologists are exposed to multiple challenges due to variations in practice and lack of a unified approach [2]. Such variations are noted even within the same country [6].
To the best of our knowledge, no study to date in the Baltic countries has assessed the methodology and conditions under which circumstances ASIC is obtained, how complications are described and recorded during the consent process, and whether established processes meet legal requirements. The present study therefore evaluated the practices and opinions of anesthesiologists in Estonia, Latvia, and Lithuania regarding ASIC.
Material and Methods
A voluntary and anonymous survey was sent to anesthesiologists in Estonia, Latvia, and Lithuania. The survey sample size was calculated at 226 respondents, based on the estimated number of anesthesiologists in the 3 countries, a 95% confidence level, a 5% confidence interval, and an average 70/30 opinion split to index questions based on previously published data [6]. The number of practicing anesthesiologists in the 3 countries was estimated to be 750 using data from national specialist registries and membership registries of the national societies of anesthesiology. The numbers were proportionally reduced to account for those working solely in intensive care medicine. The survey consisted of 3 parts: core demographic data, information related to common and severe complications of general anesthesia, and facts and opinions about the process of consent in their hospitals. Results were compared with a predetermined range of incidence rates of complications based on recently published data (Table 1) [7–13].
Survey responses were collected using anonymised paper forms. Data were processed and analysed using Microsoft Excel (Excel 2019, v16.30) and SPSS Statistics (v23; IBM, Armonk, New York, USA). Categorical data were analysed using the cross-tabulation z-test and χ2 tests, whereas continuous variables were compared using analyses of variance (ANOVA) and Student’s t-tests, followed by Bonferroni adjustment for the 3 pairwise comparisons.
Based on the definitions stipulated by the Republic of Lithuania Law on Ethics of Biomedical Research, this project was not classified as biomedical research; therefore, ethics approval for this project was not necessary. The survey protocol, however, was submitted for further analysis by the institutional review board of the Department of Bioethics of Lithuanian University of Health Sciences. This analysis confirmed that, as this project did not involve patients and vulnerable individuals from the general public, did not collate data on individual health, and was anonymised, ethics approval was not required. Choosing to fill in the questionnaire form was considered an implied consent to take part in the survey.
Results
Responses were received from 233 physicians, 146 from Lithuania, 54 from Latvia, and 33 from Estonia, representing an estimated 36%, 26%, and 24%, respectively, of the practicing anesthesiologists in these countries. The gender distribution of respondents was comparable to that of the memberships of the national societies. Doctors in training and younger doctors were overrepresented in this survey (Table 2).
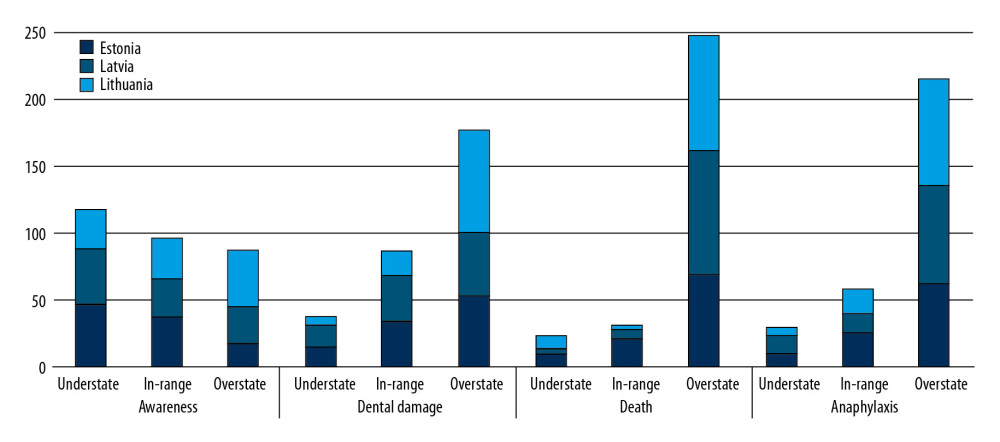
Dedicated ASIC forms were found to be prevalent in Lithuania, whereas approximately half the respondents from Estonia and Latvia used combined consent forms for surgery and anesthesia. However, the reported incidence of surgeons obtaining consent or consent being obtained and recorded as a free text entry was low (Table 3). More than half the respondents from Latvia saw their patients well in advance of surgery, whereas same-day consent was more frequent in Estonia and almost universal in Lithuania (p <0.001). Consent forms varied in describing the potential risks of anesthesia. Risks were not described in a significant proportion of consent forms in Estonia and Latvia, whereas most consent forms in Lithuania contained some information on the risks of anesthesia. However, this information was usually limited to a generic statement on anesthesia risks, rather than specifying each of them individually (Table 3). We found that 79% of respondents either did not record the risks on ASIC forms or used generic risk statements without identifying those risks. Of the 233 respondents, 155 (66.5%) stated that they were aware of the incidence rates of frequent and major complications of general anesthesia. A higher percentage of respondents in Lithuania (73%) were confident about their knowledge than respondents in Estonia (66%) and Latvia (53%) (p<0.001). However, this did not correlate with the accuracy of information provided to the patients. Exception for intra-operative awareness, risks were found to be overstated by 64.4% of anesthesiologists in the Baltic states, who frequently quoted incidence rates that were several to tens of times higher than those published internationally (Figure 1). In contrast, only 19.4% of respondents correctly stated the incidence of risks within the predefined range, whereas 14.2% understated these risks (P<0.001). This tendency was less evident in Estonia, in which quoted incident rates were closer to those published internationally, but the difference was statistically significant only for dental damage (Table 1). While anesthesiologists of both sexes reported very broad ranges, men tended to report lower median incidence rates for all 4 complications included in the questionnaire than women, with the difference being statistically significant only for dental damage: (0.2% [IQR 0.1–1.0%]
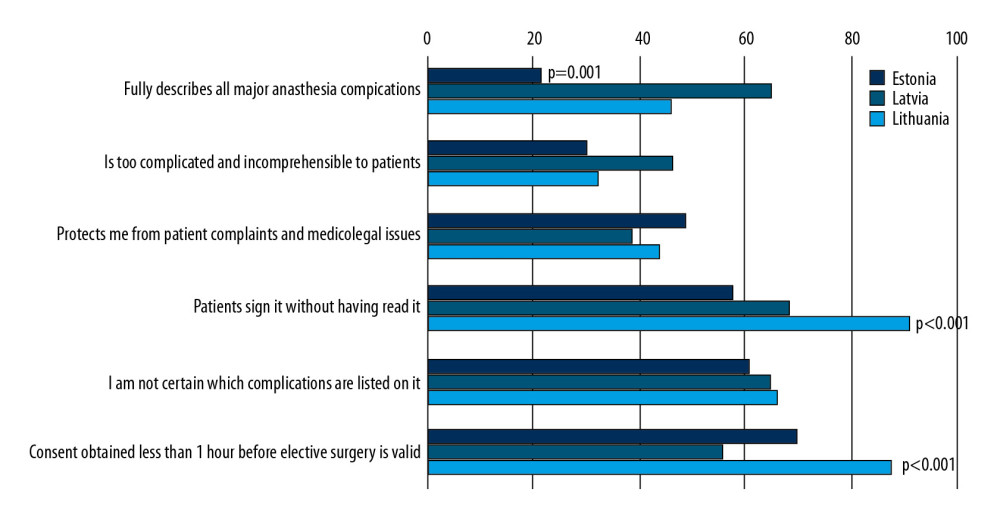
Respondents from all 3 countries were critical about the process of obtaining consent and the quality of information provided by the consent forms (Figure 2). Anesthesiologists in Estonia were significantly less satisfied with this information than anesthesiologists in Latvia and Lithuania (P=0.001). A large proportions of patients in all 3 countries signed consent forms without reading them, with this proportion being significantly higher in Lithuania (P<0.001), with analysis indicating that signing consent forms without reading them was significantly more likely when separate anesthesia consent forms were used (P<0.001). Respondents who used separate ASIC forms were more likely to believe that a consent form signed less than 1 hour before surgery was legally valid (P<0.001), but fewer than half the respondents from all 3 countries felt legally protected by current consent practices.
The process of obtaining ASIC also influenced the opinions of respondents. Generally, 62.5% of respondents who obtained ASIC forms during outpatient pre-assessment agreed that these forms described all the major complications of anesthesia, compared with 42.6% of respondents who obtained consent on the day of surgery (P=0.03). Patients were significantly less likely to read ASIC information when the forms were signed on the day of surgery than during pre-assessment (8.5%
Discussion
Obtaining informed patient consent prior to any diagnostic or interventional procedure is vital in modern medicine [14]. This process helps to ensure the patient’s autonomy, i.e., the ability to independently make important personal decisions [15]. The main principles of informed patient consent include adequate provision of information, a competent patient, voluntary consent, and the ability to withdraw consent at any time [14]. However, many patients are often unable to describe the risks described by their doctors [3]. The results of this survey correlate well with previously published data suggesting significant variations in practice [16]. Physicians in Latvia have established effective systems of early pre-operative assessment, which is reflected in a large proportion of patients being seen in the outpatient setting well before the surgery. In addition, more than two-thirds of responding anesthesiologists in Latvia reported that the forms they used fully describe all possible complications of anesthesia. Lithuanian respondents were less satisfied with the information provided by their forms, and Estonians were even less satisfied, which may be related to the predominant practice of same-day consent or even surgeons receiving consent to anesthesia. Interestingly, completeness of information may come at a cost of clarity and simplicity, as a higher proportion of anesthesiologists in Latvia than in the other 2 countries regarded their consent forms as being too complex for their patients. This survey revealed a behavioural pattern affecting both physicians and patients in all 3 countries, in that most physicians believed that their patients signed consent forms without reading them, with a similar proportion being unable to recall how complications were described on the standard consent forms. The combination of these 2 factors suggests that neither physicians nor patients regard ASIC as an important part of perioperative care. This was supported by the phenomenon of ‘tick-box-exercise consent’, which is a lot more prevalent with the use of separate ASIC forms and same-day consent. Models of current practice rarely allow sufficient time for the consent process, thus re-enforcing the belief that ASIC is nothing more than an unavoidable medicolegal burden. Unsurprisingly, fewer than half the respondents believed that these consent forms provide protection against complaints or legal challenges, and nearly a third question the validity of such consent. The proper amount of information and its manner of presentation remain unclear, as providing too much information may increase the level of pre-operative anxiety in patients [17], whereas providing insufficient information may affect patient expectations and lead to worse subjective outcomes [5,18]. Except for intra-operative awareness, anesthesiologists tend to overstate the risks of anesthesia. Our survey methodology was not sensitive enough to establish the reasons for this practice, which could be related to major regional variations in surgical outcomes, as demonstrated by the European Surgical Outcomes Study [19] or the use of different sources of information and outcome data specific to institutions. Alternatively, this practice could be related to perceived self-protection or even insufficient knowledge of up-to-date complication rates. Our survey found that anesthesiologists were not inclined to present all possible complications of procedures and use very broad ranges when describing their likelihood. All 3 countries have laws protecting the rights of patients and defining the practice and requirements for informed consent. Independent legal advice obtained when designing this survey indicated that current consent practice does not comply with the standards described in the Lithuanian Law on the Rights of Patients and Compensation of the Damage to Their Health [20], in the Estonian Law of Obligations Act [21], and in the Latvian Law on the Rights of Patients [22]. These laws obligate physicians to provide information regarding both typical and rare complications [20–22], whereas our survey found that these obligations remain unfulfilled. Thus, there is a need for a more structured system, which could assist in providing patients with relevant information and ensure adequate legal protection of doctors. We believe that the following recommendations will improve the ASIC process:
Conclusions
ASIC practices vary significantly among the 3 Baltic states, with these practices often falling short of legally required standards. Recommendations have been made to improve the accuracy of information, patient autonomy, and compliance with existing legal frameworks.
References
1. Gelb AW, Morriss WW, Johnson W, Merry AFInternational Standards for a Safe Practice of Anesthesia Workgroup, World Health Organization-World Federation of Societies of Anesthesiologists (WHO-WFSA) International Standards for a Safe Practice of Anesthesia: J Can Anaesth, 2018; 65(6); 698-708
2. Yentis SM, Hartle AJ, Barker IR, AAGBI: Consent for anaesthesia 2017: Association of Anaesthetists of Great Britain and Ireland: Anaesthesia, 2017; 72(1); 93-105
3. Zarnegar R, Brown MRD, Henley M, Patient perceptions and recall of consent for regional anaesthesia compared with consent for surgery: J R Soc Med, 2015; 108(11); 451-56
4. Burkle CM, Pasternak JJ, Armstrong MH, Keegan MT, Patient perspectives on informed consent for anaesthesia and surgery: American attitudes: Acta Anaesthesiol Scand, 2013; 57(3); 342-49
5. White SM, Baldwin TJ, Consent for anaesthesia: Anaesthesia, 2003; 58(8); 760-74
6. Cafferkey A, Lyons B, Cross-sectional analysis of the standards of consent applied to anaesthesia in Ireland: Are anaesthetists aware of their legal and ethical obligations?: Ir Med J, 2018; 111(1); 668
7. Tan Y, Loganathan N, Thinn KK, Dental injury in anaesthesia: A tertiary hospital’s experience: BMC Anesthesiol, 2018; 18(1); 108
8. Gaudio RM, Feltracco P, Barbieri S, Traumatic dental injuries during anaesthesia: Part I: Clinical evaluation: Dent Traumatol, 2010; 26(6); 459-65
9. Freundlich RE, Duggal NM, Housey M, Intraoperative medications associated with hemodynamically significant anaphylaxis: J Clin Anesth, 2016; 35; 415-23
10. Sandin RH, Enlund G, Samuelsson P, Lennmarken C, Awareness during anaesthesia: A prospective case study: Lancet, 2000; 355(9205); 707-11
11. Cook TM, Andrade J, Bogod DGRoyal College of Anaesthetists and the Association of Anaesthetists of Great Britain and Ireland: Anaesthesia, 2014; 69(10); 1102-16
12. Jenkins K, Baker AB, Consent and anaesthetic risk: Anaesthesia, 2003; 58(10); 962-84
13. Haller G, Laroche T, Clergue F, Morbidity in anaesthesia: Today and tomorrow: Best Pract Res Clin Anaesthesiol, 2011; 25(2); 123-32
14. Bowman D, Spicer J, Iqbal R: Informed consent: A primer for clinical practice, 2011, Cambridge, UK, Cambridge University Press
15. Delany C, Making a difference: Incorporating theories of autonomy into models of informed consent: J Med Ethics, 2008; 34(9); e3
16. Lühnen J, Mühlhauser I, Steckelberg A, The quality of informed consent forms – a systematic review and critical analysis: Dtsch Arztebl Int, 2018; 115(22); 377-83
17. Cyna AM, Simmons SW, Guidelines on informed consent in anaesthesia: Unrealistic, unethical, untenable: Br J Anaesth, 2017; 119(6); 1086-89
18. Fortunato JT, Wasserman JA, Menkes DL, When respecting autonomy is harmful: A clinically useful approach to the nocebo effect: Am J Bioeth, 2017; 17(6); 36-42
19. Pearse RM, Moreno RP, Bauer PEuropean Surgical Outcomes Study (EuSOS) group for the Trials groups of the European Society of Intensive Care Medicine and the European Society of Anaesthesiology, Mortality after surgery in Europe: A 7 day cohort study: Lancet, 2012; 380(9847); 1059-65
20. : I-1562 Lithuanian Law on the Rights of Patients and Compensation of the Damage to Their Health Available from: URL: https://www.e-tar.lt/portal/lt/lgalAct/TAR.C6E4170DB704/asr
21. : Law of Obligations Act Available from: https://www.riigiteataja.ee/en/eli/ee/Riigikogu/act/506112013011/consolide
22. : Law on the Rights of Patients Available from: https://likumi.lv/doc.php?id=203008
Figures
Tables
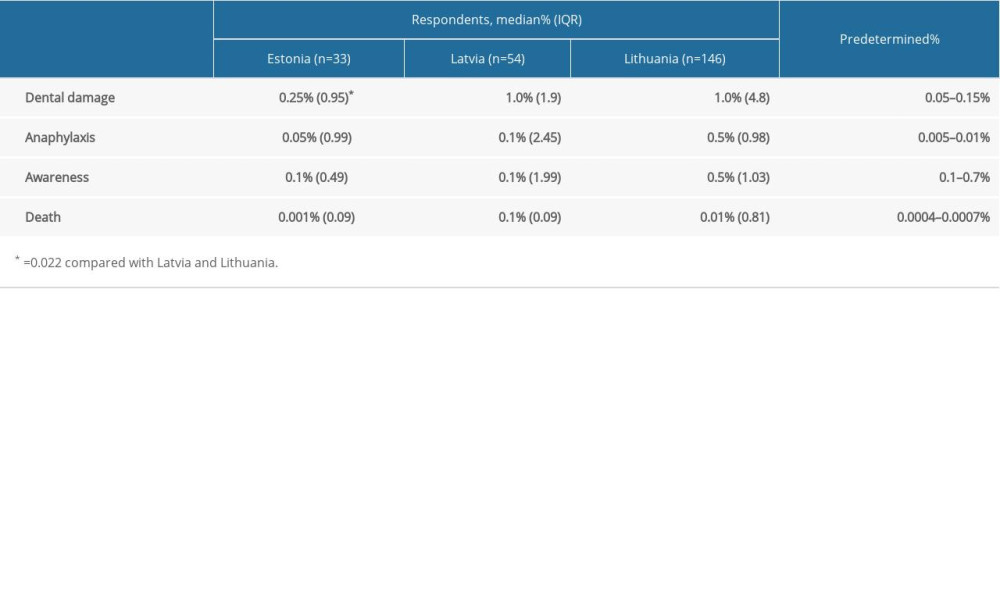 Table 1. Anesthesia complication rates reported by the respondents in Estonia, Latvia, and Lithuania and their predetermined ranges.
Table 1. Anesthesia complication rates reported by the respondents in Estonia, Latvia, and Lithuania and their predetermined ranges.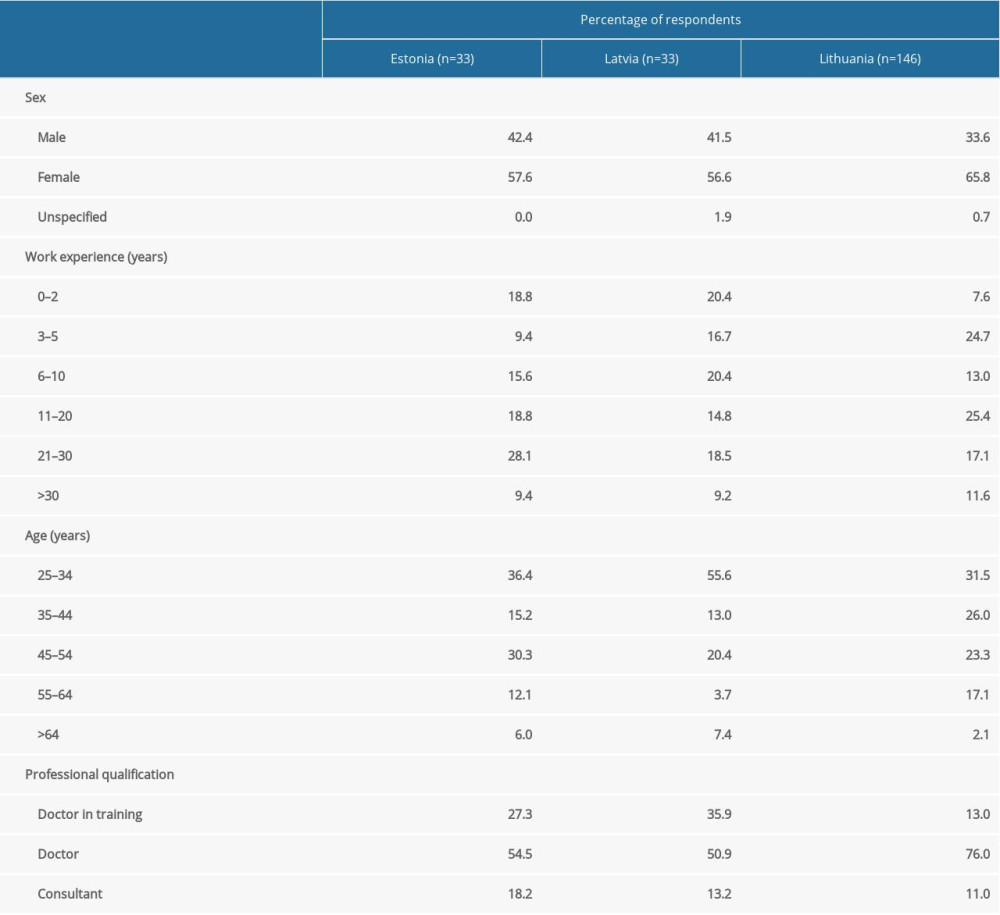 Table 2. Demographic characteristics of survey respondents.
Table 2. Demographic characteristics of survey respondents.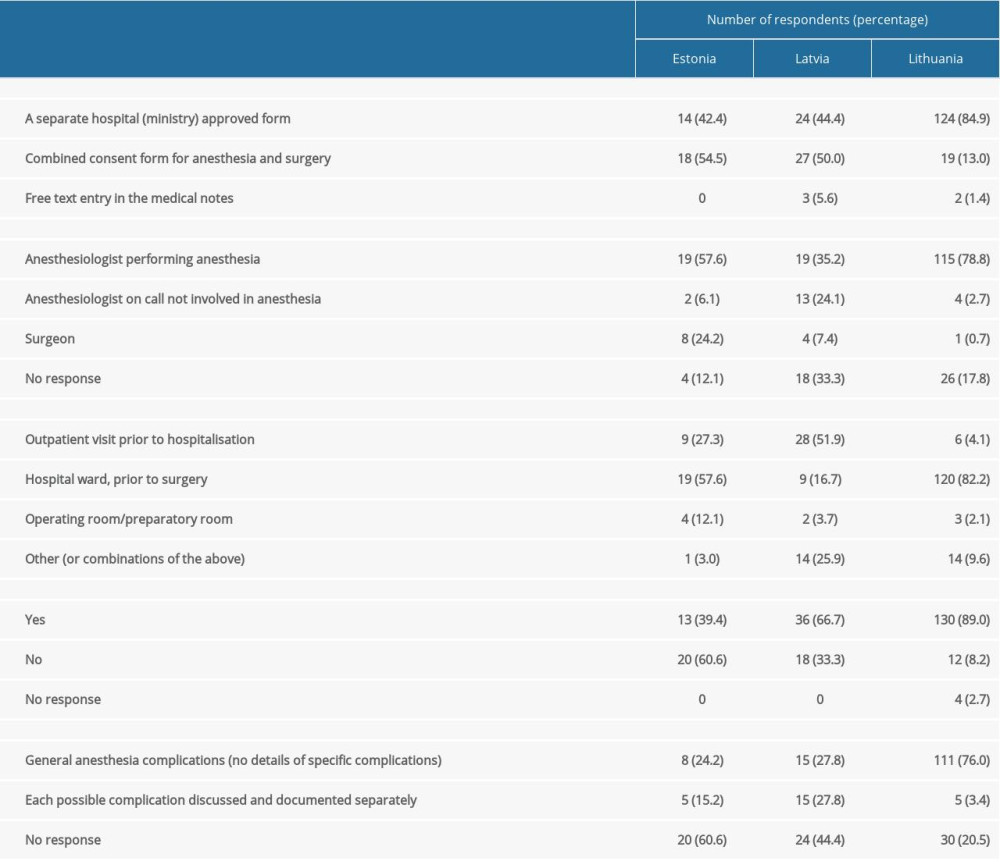 Table 3. Respondent practices of obtaining patient consent.
Table 3. Respondent practices of obtaining patient consent. Table 1. Anesthesia complication rates reported by the respondents in Estonia, Latvia, and Lithuania and their predetermined ranges.
Table 1. Anesthesia complication rates reported by the respondents in Estonia, Latvia, and Lithuania and their predetermined ranges. Table 2. Demographic characteristics of survey respondents.
Table 2. Demographic characteristics of survey respondents. Table 3. Respondent practices of obtaining patient consent.
Table 3. Respondent practices of obtaining patient consent. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387