26 October 2020: Database Analysis
Small Nucleolar RNAs (snoRNAs)-Based Risk Score Classifier Predicts Overall Survival in Bladder Carcinoma
Rong-Quan He1A, Zhi-Guang Huang2C, Gao-Qiang Zhai3C, Su-Ning Huang4ACDEF, Yong-Yao Gu2B, Yong-Yao Gu2B, Gang Chen2ACDEF, Jie Ma1B, Ji-Wen Cheng3ACE, Hai-Biao Yan3ADEF, Sheng-hua Li5A*DOI: 10.12659/MSM.926273
Med Sci Monit 2020; 26:e926273
Abstract
BACKGROUND: Bladder carcinoma (BLCA) is a leading cause of cancer-related deaths worldwide. The aim of this work was to develop an accurate stratification in predicting the prognosis and directing the treatment of BLCA patients based on small nucleolar RNAs (snoRNAs).
MATERIAL AND METHODS: Expression profiles of snoRNAs were downloaded from the SNORic database. The expression profiles and clinical outcomes of BLCA patients were analyzed. Survival-associated snoRNAs were identified and used to develop a novel risk score classifier. Genes in the whole genome that were significantly correlated with the included prognostic snoRNAs were used for functional enrichment analysis.
RESULTS: The results showed that age, American Joint Committee on Cancer (AJCC) stage, and tumor status were significantly correlated with overall survival (OS) of BLCA patients. We selected 12 survival-associated snoRNAs to build a prognostic signature. Patients were separated into high- and low-risk groups based on the median value of the risk score. Patients in the high-risk group and low-risk group have distinct clinical outcomes. The AJCC TNM stage showed moderate utility as a prognostic indicator for clinical outcome prediction. Then, clinical parameters and risk scores were entered in multivariate Cox analysis. Notably, the prognostic signature remained an independent significant prognostic risk factor. The pathway analysis suggested that these genes were enriched in several types of cancer and “Focal adhesion” pathways.
CONCLUSIONS: The prognostic signature defined by expression profiles of 12 survival-associated snoRNAs appears to be an excellent predictor of the clinical outcome of BLCA patients.
Keywords: Risk Factors, RNA, Small Untranslated, Survival Rate, Biomarkers, Tumor, Carcinoma, RNA, Small Nucleolar
Background
Bladder cancer is a heterogeneous malignancy that is responsible for an estimated 81 400 new cases and 17 980 deaths in the United states in 2020 [1]. Although progress has been made in surgical resection technology and neoadjuvant chemotherapy, survival after surgical resection differs considerably among BLCA patients [2–4]. The predictive accuracy of the current clinical staging system is insufficient for this medically precise era and for the display of the molecular characteristics of BLCA. Hence, cancer researchers have been interested in developing an accurate stratification method that can predict the prognosis and direct treatment of BLCA patients to improve their survival [5–10]. It is also imperative to determine the biological characteristics of BLCA.
Genome researchers have long regarded the non-coding RNAs of the human genome as ‘junk’ DNA [11]. Specifically, the rapid development of high-throughput technology helped scientists to identify a large number of non-coding RNAs, which account for almost 60% of the transcriptional output in human cells [12–16]. This huge number of non-coding RNAs makes it difficult to dismiss them as “junk”. Hence, recent extensive research has identified that non-coding RNAs, especially long non-coding RNAs and micro RNAs, are directly linked to the tumorigenesis and development of cancers [17,18]. Notably, small nucleolar RNA (snoRNAs), a class of small (60–300 nucleotides) non-coding RNAs, has been well documented in rRNA biogenesis. Recently, however, some studies have highlighted that snoRNAs are involved in cancer development and progression [19]. Because these studies dismissed the non-coding RNAs of the human genome as ‘junk’ DNA, the relationships between snoRNAs and BLCA have not been well characterized.
The Cancer Genome Atlas (TCGA) consortium and other public genomic datasets offer high-throughput data for the subclassification of BLCA, as well as predicting diagnosis and prognosis [20–22]. Gong et al. developed a database of snoRNA in cancers to systematically quantify and deposit snoRNA expression profiles in more than 10 000 samples across 31 cancer types based on TCGA, including BLCA [23]. This work greatly promoted the development of snoRNAs analysis in cancers. However, very few other studies have explored the role of snoRNAs in BLCA. Furthermore, no investigative reports on models that predict the survival status of BLCA based on snoRNAs are available.
Based on TCGA data portal, we analyzed the expression profiles and clinical outcomes of BLCA patients. Survival-associated snoRNAs were identified and submitted to develop a novel 12-snoRNAs-based risk score classifier. To leverage the complementary value of molecular and clinical parameters, clinical factors were integrated to build a nomogram, which allowed improved prediction of BLCA patient survival.
Material and Methods
DATA ACQUISITION:
Expression profiles of snoRNAs were downloaded from the SNORic database [23]. Corresponding clinical data of BLCA patients were acquired from the TCGA database (https://cancergenome.nih.gov/).
SURVIVAL ANALYSIS:
To generate a prognostic classifier to predict the outcome of BLCA, we used univariate Cox analysis to explore the relationships between snoRNAs expression levels and the overall survival (OS) of BLCA patients. Patients with OS less than 90 days were removed to obtain more accurate results [24]. Then, multivariate Cox regression analysis was conducted to screen out independent prognostic factors. We calculated a signature involving the single prognostic parameters achieved from the previous step to assess the OS risk based on the individual expression of the prognostic snoRNAs, weighted by the regression coefficient. The data were then divided into high- or low-risk groups by using the median risk score as the threshold value. To leverage the complementary value of the snoRNAs and several indispensable clinical parameters, including age, sex, tumor T stage, tumor N stage, tumor M stage, and histological grade, we integrated them by using a nomogram graph.
FUNCTIONAL CHARACTERIZATION OF THE SNORNAS-BASED RISK SCORE:
To further gain a biological understanding of the snoRNAs-based risk score, we collected genes in the whole genome which were significantly correlated with the included prognostic snoRNAs that were obtained from SNORic. Significantly correlated genes were identified with the |Spearman coefficient| ≥0.4 and FDR <0.05. These significantly correlated genes were submitted to a “clusterProfiler” package in R software for gene functional enrichment analysis. The background of the gene list was measured by “Homo sapiens.” Statistically significant terms were identified when the p-value and the q-value were both less than 0.05.
STATISTICAL ANALYSIS:
All statistical analyses were performed using SPSS (version 22.0, SPSS Inc., Chicago, IL, USA) and R (version 3.3.1;
Results
IDENTIFICATION OF SURVIVAL-ASSOCIATED SNORNAS:
Expression profiles of 412 snoRNAs were acquired from the SNORic database. Finally, a total of 366 BLCA patients with OS more than 90 days were included in the present study. Table 1 shows the clinical information of the patients with BLCA from TCGA. Univariate Cox hazard analyses were performed to assess the relationship between clinical parameters and clinical outcomes of the BLCA patients. Age (HR=1.792, 95% CI: 1.262–2.543; P=0.001) and AJCC stage (HR=2.161, 95% CI: 1.459–3.202; P<0.001) were significantly correlated with OS of BLCA patients. However, no significant correlations were observed between OS and sex or histological grade. All 368 snoRNAs were submitted to a univariate Cox analysis and 58 snoRNAs were identified as survival-associated snoRNAs (Figure 1, Table 2).
CONSTRUCTION OF SNORNAS-BASED PROGNOSTIC SIGNATURE:
Significant factors from univariate selection were kept in the multivariate analysis by using backward selection (Table 3). A multivariate Cox regression analysis was used to build a prognostic signature that selected 12 out of the 58 snoRNAs. Kaplan-Meier (K-M) curves were used to display the prognostic value of each snoRNA (Figure 2). We then derived a prognostic signature for each patient based on the individual expression levels of the 12 survival-associated snoRNAs multiplied by their coefficients in the multivariate Cox analysis: prognostic signature=(SNORD114–11 * (−0.168)+SNORD114–14 * (0.201)–SNORD114–15 * (0.229)+SNORD114–9 * (0.543)–SNORA55 * (0.198)–SNORA60 * (0.192)–SNORD88A * (0.167)–SNORD69 * (0.314)–SNORD20 * (0.226)+U49A * (0.432)–SNORD51 * (0.335)+U74 * (0.514) (Figure 3). Patients were separated into high- and low-risk groups by the median value of the prognostic signature. K-M survival plots indicated that patients in the high- and low-risk groups had distinct clinical outcomes (HR=2.500, 95% CI: 1.828–3.420, P<0.001; Figure 4A). The AUC value of the ROC curve was 0.719. The threshold was 2000 days (Figure 4B). The AJCC TNM stage appears to be a moderate prognostic indicator for clinical outcome predicting (HR=2.155, 95% CI: 1.553–2.993, P<0.001; Figure 4C). The AUC value of the TNM stage was 0.636 (Figure 4D). We integrated the clinical factors and risk score to create a composite nomogram based on the results of the multivariate Cox regression analyses to predict 1-year, 2-year, and 3-year OS. (Figure 5). We then submitted clinical parameters and risk scores in multivariate Cox analysis. Notably, the prognostic signature remained an independent significant prognostic risk factor (HR=3.300, 95% CI: 2.203–4.943, P<0.001) (Table 4).
MOLECULAR FUNCTION OF PROGNOSTIC SIGNATURE:
Genes that were significantly correlated with 12 survival-associated snoRNAs were obtained from SNORic based on Spearman correlation analysis. The network was conducted to display the relationships between snoRNAs and genes (Figure 6). Red lines indicate positive correlation relationships while blue lines indicate negative correlation relationships. SnoRNAs-related genes were further submitted to gene functional enrichment analysis. For the biological process, the extracellular structure organization, the extracellular matrix organization, and skeletal system development were the commonly enriched categories (Figure 7A). For the cellular component ontology, the enriched categories were correlated with proteinaceous extracellular matrix, endoplasmic reticulum lumen, and contractile fiber (Figure 7B). With regards to the molecular function, the snoRNA-related genes mainly showed enrichment in cell adhesion molecule binding, actin binding, and sulfur compound binding (Figure 7C). The disease ontology suggested that these genes were enriched in several types of cancer (Figure 8A). Kyoto Encyclopedia of Genes and Genomes (KEGG) analysis revealed significant pathways with these genes (Figure 8B). “Focal adhesion” was the most significant of the enriched terms.
Discussion
Here, we performed a systematic analysis of the snoRNAs and identified a risk score based on the expression profiles of 12 survival-associated snoRNAs in BLCA patients based on 366 clinical cases. Our study resulted in the following: (1) the identification of 58 prognostic relevant snoRNAs; (2) the development of a twelve-snoRNAs-based risk score classifier that predicts OS in BLCA; (3) a pathway analyses that revealed the molecular characteristics of the risk score; and (4) the construction of a nomogram to leverage the complementary value of molecular and clinical factors.
We first identified several snoRNAs that were correlated with the clinical outcomes of BLCA. Given the stable nature of snoRNAs in the human body, these snoRNAs have inherent advantages for use as molecular biomarkers [25–27]. Regrettably, few studies have assessed snoRNAs as diagnostic and prognostic tools for BLCA. This is mainly due to the conventional prejudice that snoRNAs mainly function to modify, mature, and stabilize rRNAs [28]. Furthermore, high-throughput DNA sequencing techniques help to identify cancer-specific snoRNAs [29]. Hence, our group comprehensively analyzed the clinical significance and potential molecular characteristics of snoRNAs to identify the diagnostic and prognostic biomarkers in BLCA.
An important goal of the present study was the construction of risk scores based on the expression pattern of snoRNAs to create a risk stratification model for the practice of precision medicine. Previously, studies involved several molecules, including mRNA expression, copy number variation, DNA methylation, lncRNAs, and miRNAs. For example, Liu et al. proposed a clinical multidimensional transcriptome signature for survival predictions of patients with BLCA [30]. Aberrant DNA methylation can provide reliable biomarkers in the prediction of clinical outcomes of common urological cancers [31]. Several studies have explored the prognostic value of non-coding RNAs in developing prognostic signatures for BLCA [32,33]. We constructed the risk score model with the expectation that it would be applicable to clinical management from the perspectives of snoRNAs. On the one hand, studies that refer to the clinical significance of snoRNAs are limited. The present study provides novel insights into the clinical value and molecular mechanisms of snoRNAs in BLCA. On the other hand, we optimized the prognostic model and considered both molecular and clinical features. The snoRNAs possess the possibility and feasibility as biomarkers and therapeutic targets in clinical use. Aberrant snoRNAs expression was found in many cancers, and the expression level was correlated with diagnosis, classification of subtypes, and patient survival. Moreover, snoRNAs were stably expressed and detectable in body fluids, including blood plasma, serum, and urine of cancer patients, indicating that snoRNAs have the potential to serve as biomarkers in clinical use. Urine testing is very important for early diagnosis of BCLA. Since snoRNAs are stably expressed and detectable in urine, snoRNAs detection in urine might be useful for BLCA diagnosis and prognosis prediction [34–36].
It is interesting to explore the biological characteristics reflected by the risk score. Nowadays, the cellular regulatory roles of snoRNAs in cancers are widely understood. Hence, we selected snoRNAs-related genes to explore the molecular characteristics of snoRNAs in BLCA. We found that prognostic snoRNAs are mainly involved in several signal transduction pathways, such as focal adhesion, extracellular matrix (ECM)-receptor interaction, and peroxisome proliferator-activated receptor (PPAR) signaling pathway. In these findings, the risk score could reflect the cell-cell interaction status of BLCA. Notably, focal adhesion was the most significant KEGG pathway in the pathway functional enrichment analysis. In biological activity, focal adhesion is a sub-cellular regulatory structure that mediates mechanical force and regulatory signals transmitted between the ECM and an interacting cell [37]. The focal adhesion pathway interacts closely with other indispensable oncogenic pathways and is actively involved in the progression of cancers [38,39]. Interestingly, inhibitors focused on the focal adhesion pathway could be effective anti-tumor targets [40]. The biological characteristics of the risk score are correlated with cell-cell interactions, which indicated that these snoRNAs actively participate in the progression and/or metastasis of BLCA, although the results need to be further explored.
Conclusions
In summary, the development of a prognostic signature, as defined by expression profiles of 12 survival-associated snoRNAs, could be an excellent predictor of the clinical outcome of BLCA patients. However, studies of other independent cohorts are required to validate these findings. There is no evidence that the prognostic signature can predict prognosis in patients who received adjuvant therapies after surgical resection. In the present study, the process involves mechanisms that should be validated by
Figures
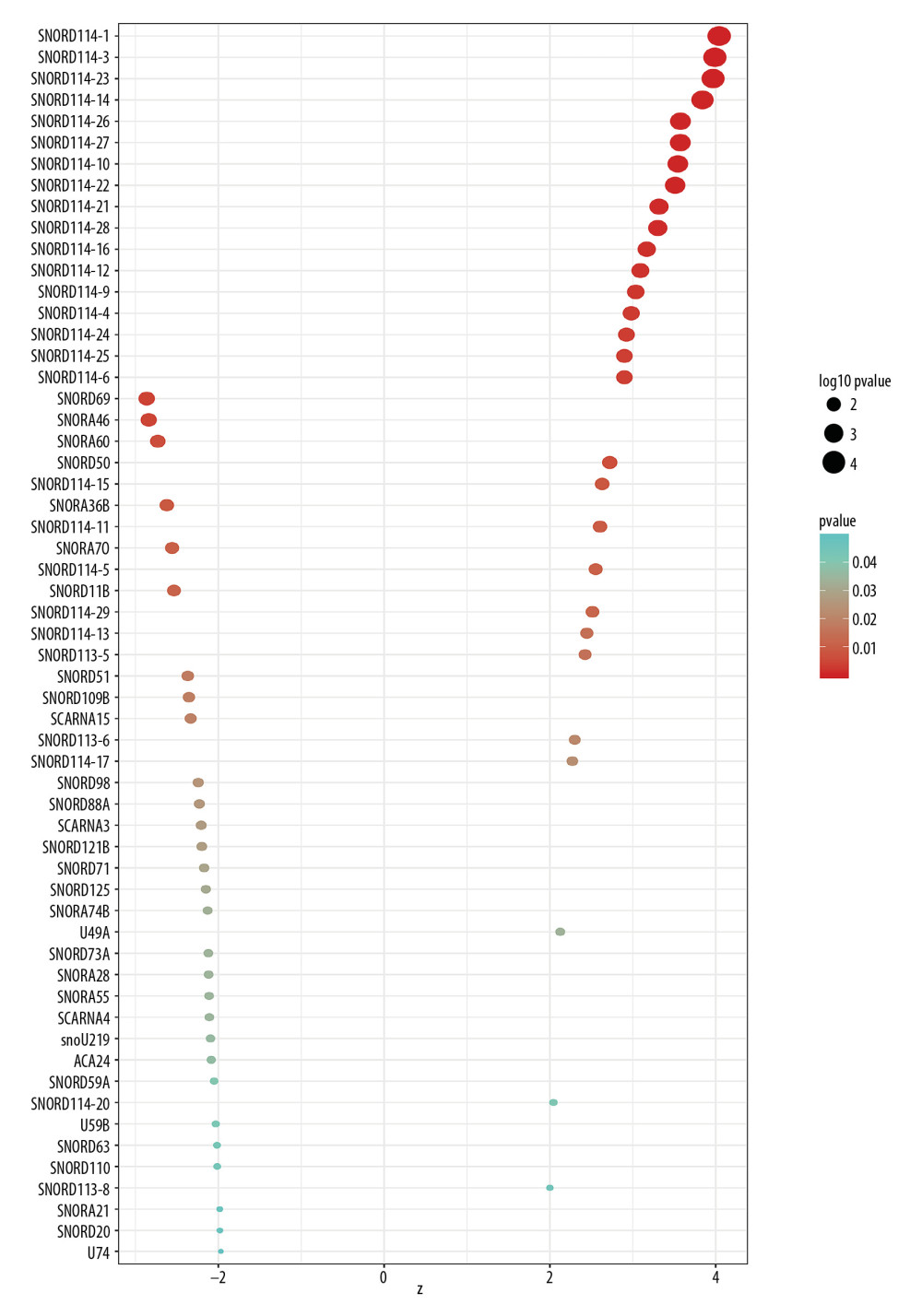 Figure 1. Survival-associated snoRNAs in BLCA. X-axis represents the Z-score of the snoRNAs in univariate Cox analysis and Y-axis represents survival-associated snoRNAs. Thresholds are p<0.05 and |Z-score| >1.8
Figure 1. Survival-associated snoRNAs in BLCA. X-axis represents the Z-score of the snoRNAs in univariate Cox analysis and Y-axis represents survival-associated snoRNAs. Thresholds are p<0.05 and |Z-score| >1.8 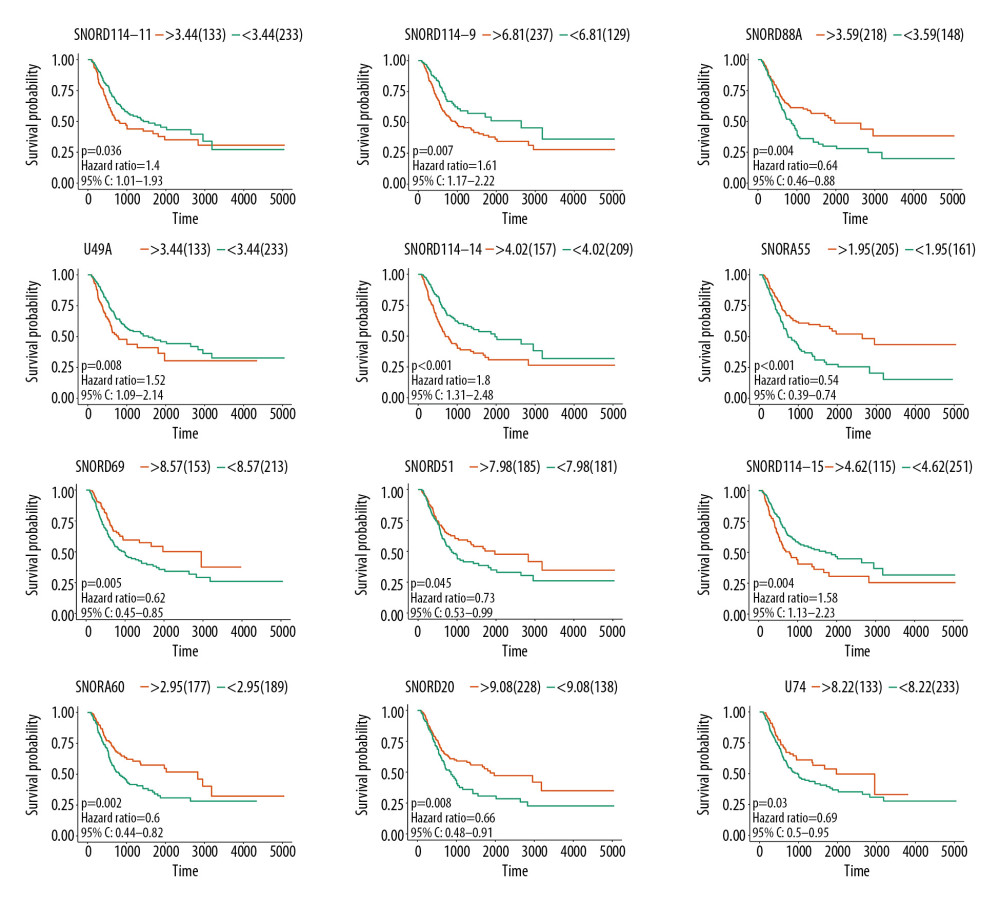 Figure 2. K-M plots prognostic snoRNAs included in prognostic signature.
Figure 2. K-M plots prognostic snoRNAs included in prognostic signature. 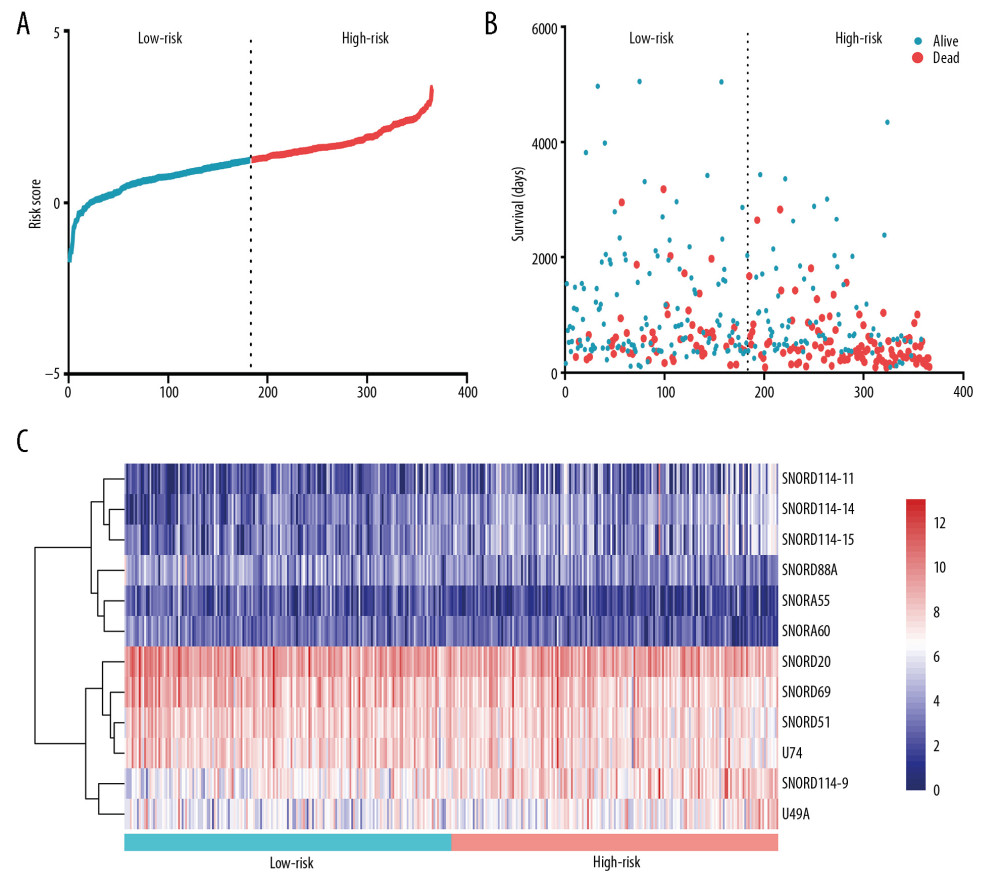 Figure 3. Prognostic signature constructed based on 12 snoRNAs. (A) The risk score assigned to each patient; (B) Survival status of BLCA patients in high- and low-risk group; and (C) The expression pattern of included snoRNAs.
Figure 3. Prognostic signature constructed based on 12 snoRNAs. (A) The risk score assigned to each patient; (B) Survival status of BLCA patients in high- and low-risk group; and (C) The expression pattern of included snoRNAs. 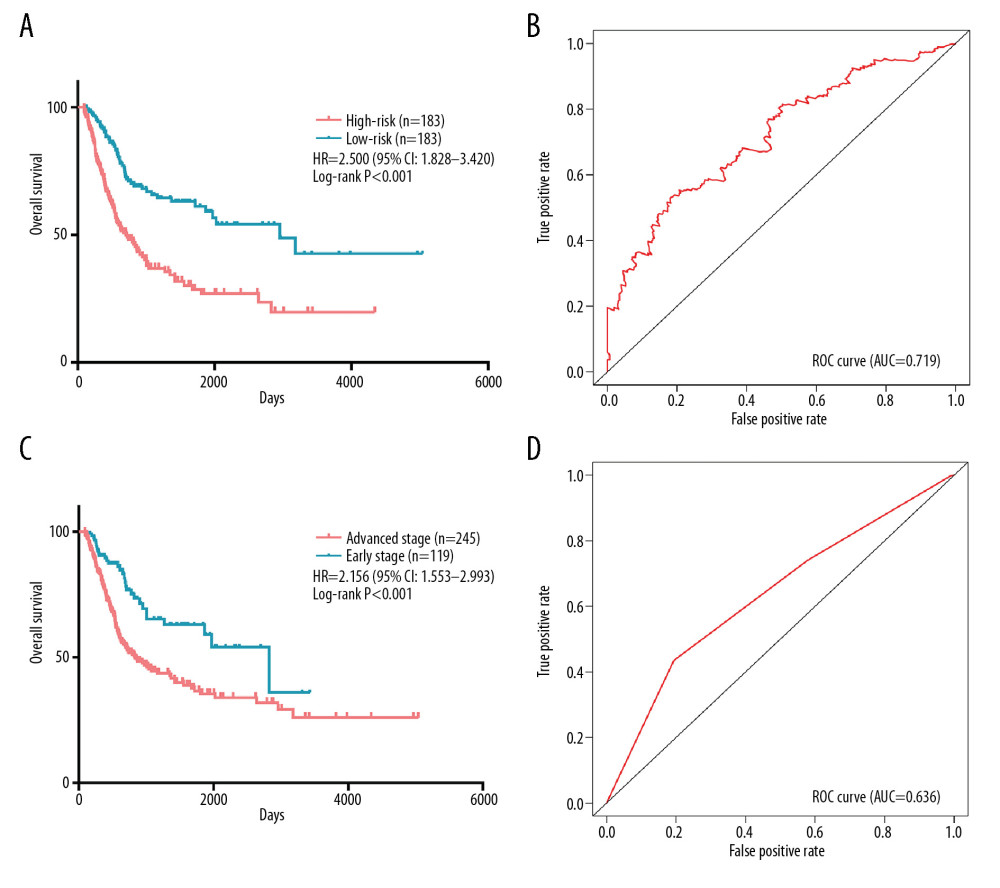 Figure 4. Comparation of snoRNA-based prognostic signature and AJCC_TNM stage in predicting the clinical outcome of BLCA patients. (A) K-M survival plots indicated that patients in the high-risk group tended to have poor clinical outcomes; (B) ROC curves with AUCs of prognostic predictors built by snoRNAs in BLCA; (C) K-M survival plots indicated that patients in advanced stage tended to have poor clinical outcomes; and (D) ROC curves with AUCs of AJCC_TNM stage.
Figure 4. Comparation of snoRNA-based prognostic signature and AJCC_TNM stage in predicting the clinical outcome of BLCA patients. (A) K-M survival plots indicated that patients in the high-risk group tended to have poor clinical outcomes; (B) ROC curves with AUCs of prognostic predictors built by snoRNAs in BLCA; (C) K-M survival plots indicated that patients in advanced stage tended to have poor clinical outcomes; and (D) ROC curves with AUCs of AJCC_TNM stage. 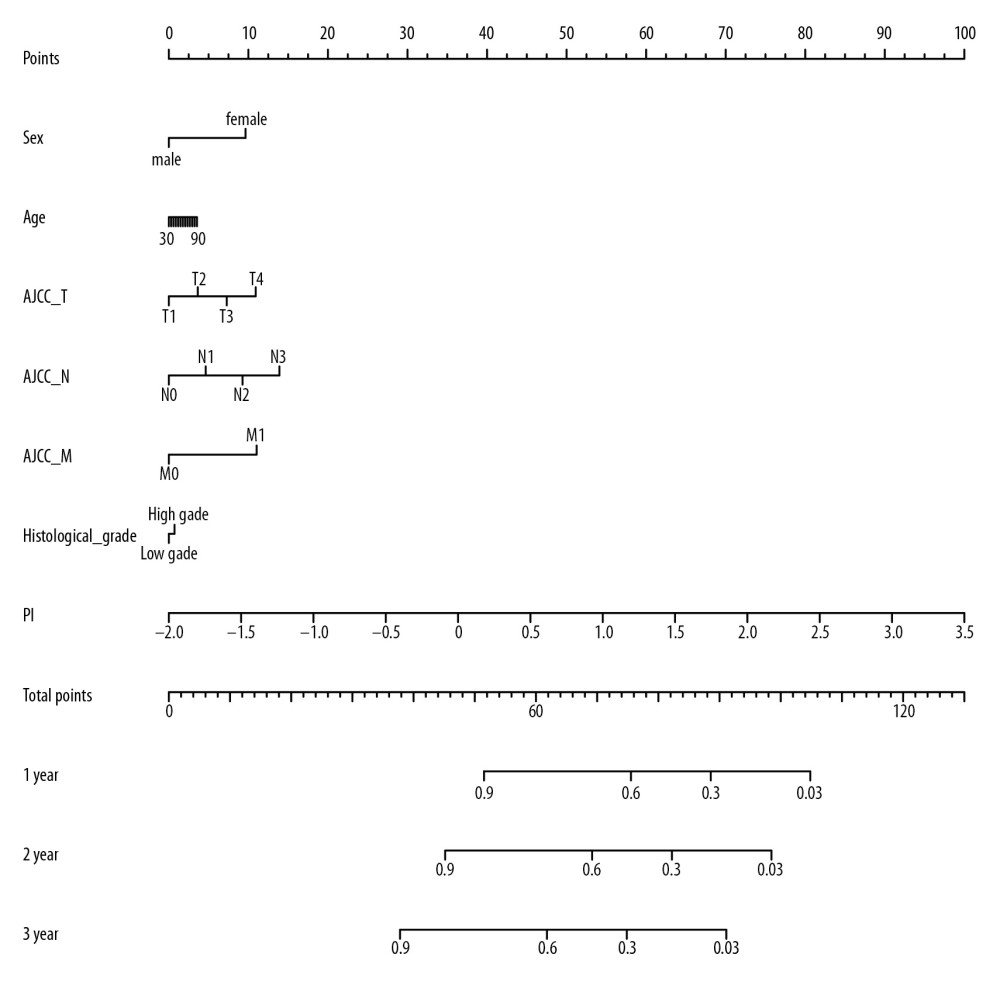 Figure 5. Nomogram of BLCA patients.
Figure 5. Nomogram of BLCA patients. 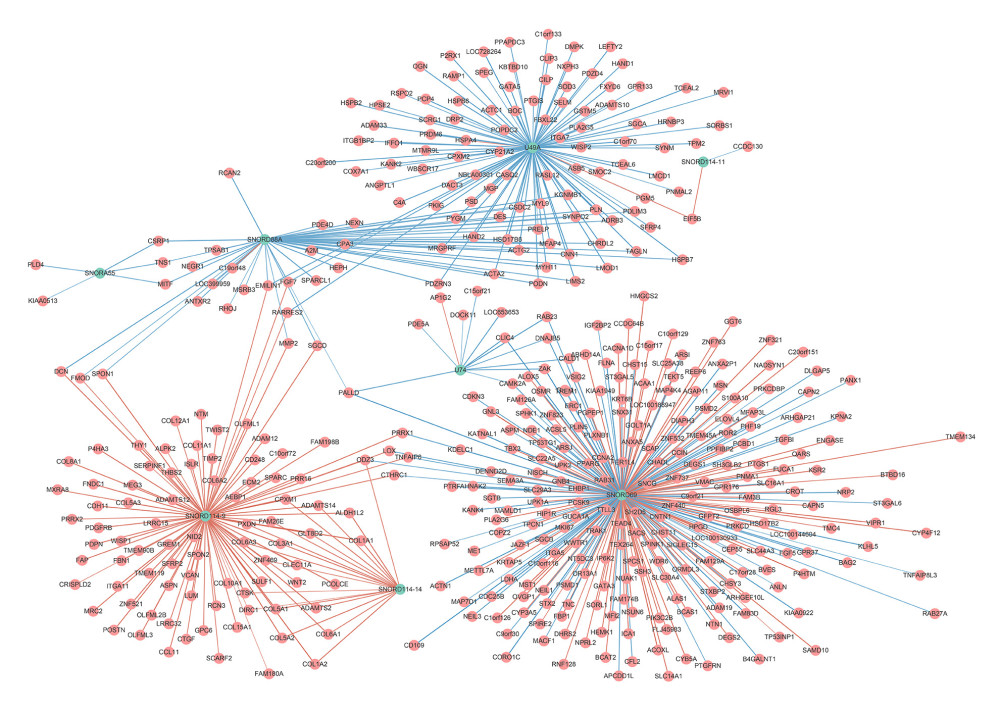 Figure 6. Correlation network of included snoRNAs and mRNAs. Expression of survival-associated snoRNAs (green dots) were positively (red line)/negatively (blue line) correlated with the expression level of mRNAs (red dots).
Figure 6. Correlation network of included snoRNAs and mRNAs. Expression of survival-associated snoRNAs (green dots) were positively (red line)/negatively (blue line) correlated with the expression level of mRNAs (red dots). 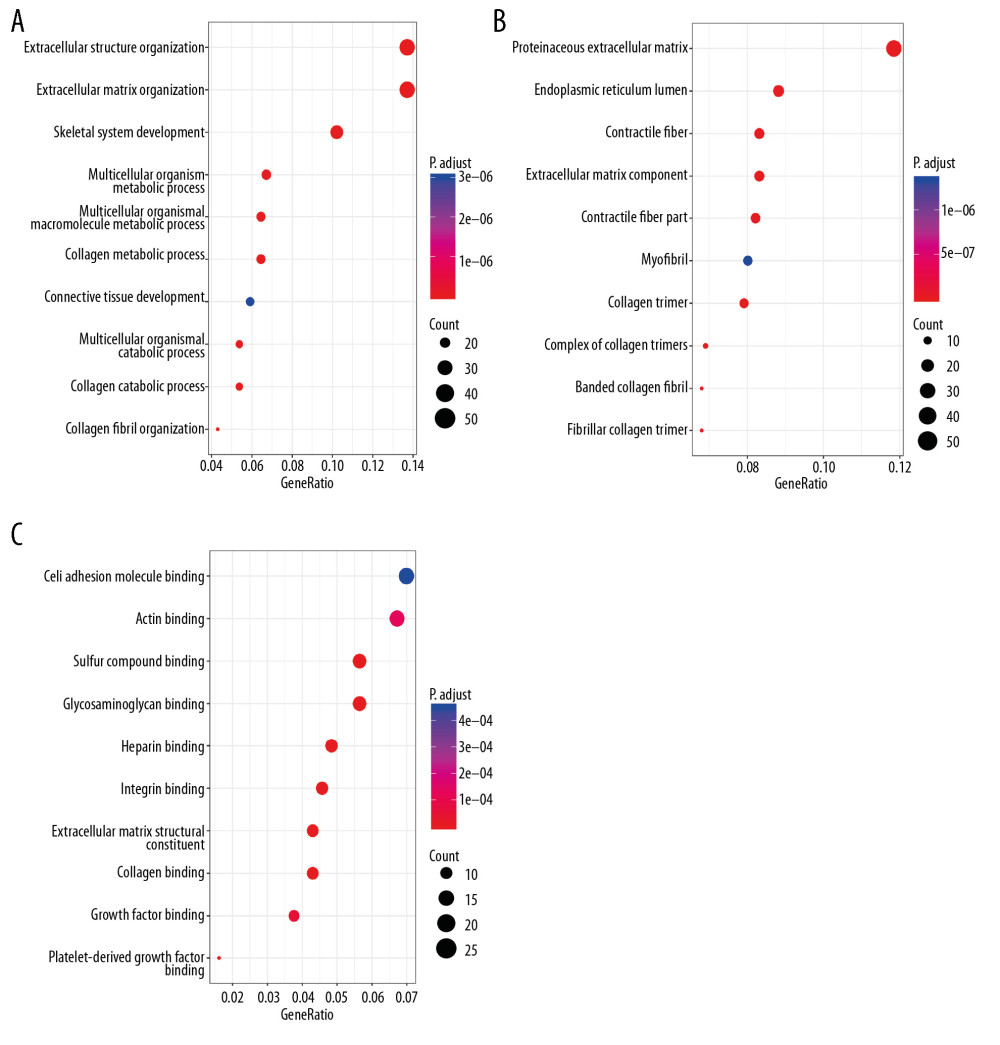 Figure 7. Gene ontology of prognostic signature-related genes. (A) Biological process; (B) Cellular component; (C) Molecular function.
Figure 7. Gene ontology of prognostic signature-related genes. (A) Biological process; (B) Cellular component; (C) Molecular function. 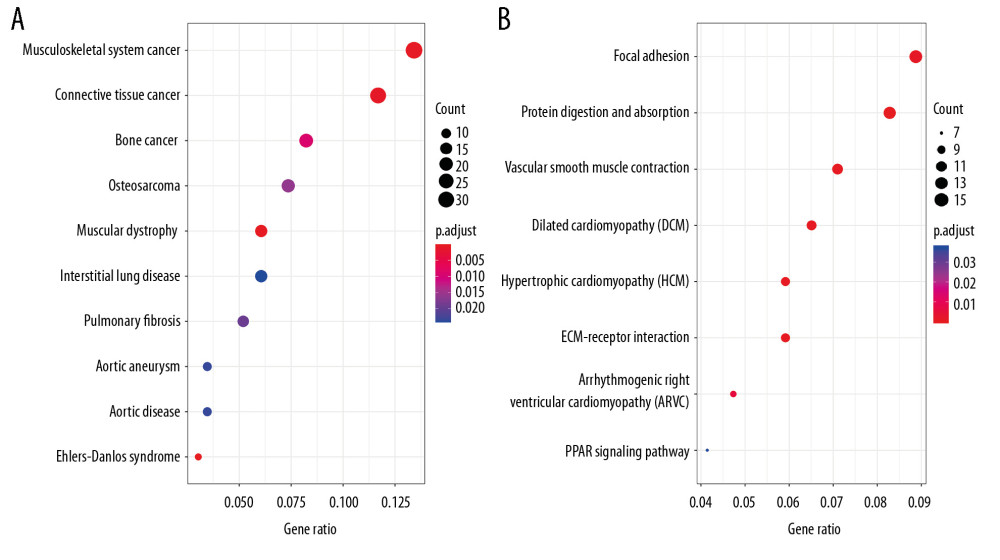 Figure 8. Disease ontology and Kyoto Encyclopedia of Genes and Genomes pathway of prognostic signature-related genes. (A) Disease ontology; (B) Kyoto Encyclopedia of Genes and Genomes pathway.
Figure 8. Disease ontology and Kyoto Encyclopedia of Genes and Genomes pathway of prognostic signature-related genes. (A) Disease ontology; (B) Kyoto Encyclopedia of Genes and Genomes pathway. References
1. Siegel RL, Miller KD, Jemal A, Cancer statistics, 2020: Cancer J Clin, 2020; 70; 7-30
2. Prasad SM, Decastro GJ, Steinberg GD, Urothelial carcinoma of the bladder: Definition, treatment and future efforts: Nat Rev Urol, 2011; 8; 631-42
3. Mo Q, Nikolos F, Chen F, Prognostic power of a tumor differentiation gene signature for bladder urothelial carcinomas: J Natl Cancer Inst, 2018; 110; 448-59
4. Chu J, Li N, Li F, A risk score staging system based on the expression of seven genes predicts the outcome of bladder cancer: Oncol Lett, 2018; 16; 2091-96
5. Liu CP, Zhang JH, Zheng SC, A novel clinical multidimensional transcriptome signature predicts prognosis in bladder cancer: Oncol Rep, 2018; 40; 2826-35
6. Wu S, Zheng J, Li Y, Development and validation of an MRI-based radiomics signature for the preoperative prediction of lymph node metastasis in bladder cancer: EBioMedicine, 2018; 34; 76-84
7. Scheller T, Hofmann R, Hegele A, Sex-related differences in urothelial cell carcinoma of the bladder in Germany: Cancer Manag Res, 2019; 11; 309-16
8. Chen Z, Zhan Y, Chi J, Using microRNAs as novel predictors of urologic cancer survival: An integrated analysis: EBioMedicine, 2018; 34; 94-107
9. Hussain SA, Palmer DH, Syn WK, Gene expression profiling in bladder cancer identifies potential therapeutic targets: Int J Oncol, 2017; 50; 1147-59
10. Ponting CP, Oliver PL, Reik W, Evolution and functions of long noncoding RNAs: Cell, 2009; 136; 629-41
11. Djebali S, Davis CA, Merkel A, Landscape of transcription in human cells: Nature, 2012; 489; 101-8
12. Liu K, Mao X, Chen Y, Regulatory role of long non-coding RNAs during reproductive disease: Am J Transl Res, 2018; 10; 1-12
13. Yu W, Li D, Ding X, LINC00702 suppresses proliferation and invasion in non-small cell lung cancer through regulating miR-510/PTEN axis: Aging (Albany NY), 2019; 11; 1471-85
14. Qi Y, Wang X, Kong X, Expression signatures and roles of microRNAs in inflammatory breast cancer: Cancer Cell Int, 2019; 19; 23
15. Liu KS, Pan F, Mao XD, Biological functions of circular RNAs and their roles in occurrence of reproduction and gynecological diseases: Am J Transl Res, 2019; 11; 1-15
16. Su M, Xiao Y, Ma J, Long non-coding RNAs in esophageal cancer: Molecular mechanisms, functions, and potential applications: J Hematol Oncol, 2018; 11; 118
17. Huarte M, The emerging role of lncRNAs in cancer: Nat Med, 2015; 21; 1253-61
18. Williams GT, Farzaneh F, Are snoRNAs and snoRNA host genes new players in cancer?: Nat Rev Cancer, 2012; 12; 84-88
19. Robertson AG, Kim J, Al-Ahmadie H, Comprehensive molecular characterization of muscle-invasive bladder cancer: Cell, 2017; 171; 540-56.e25
20. Peng D, Ge G, Xu Z, Diagnostic and prognostic biomarkers of common urological cancers based on aberrant DNA methylation: Epigenomics, 2018; 10; 1189-99
21. Shivakumar M, Lee Y, Bang L, Identification of epigenetic interactions between miRNA and DNA methylation associated with gene expression as potential prognostic markers in bladder cancer: BMC Med Genomics, 2017; 10; 30
22. Gong J, Li Y, Liu CJ, A Pan-cancer analysis of the expression and clinical relevance of small nucleolar RNAs in human cancer: Cell Rep, 2017; 21; 1968-81
23. Liu J, Lichtenberg T, Hoadley KA, An integrated TCGA pan-cancer clinical data resource to drive high-quality survival outcome analytics: Cell, 2018; 173; 400-16.e11
24. Vallone C, Rigon G, Gulia C, Non-coding RNAs and endometrial cancer: Genes (Basel), 2018; 9; 187
25. Chamorro-Petronacci C, Perez-Sayáns M, Padín-Iruegas ME, Differential expression of snoRNAs in oral squamous cell carcinomas: New potential diagnostic markers: J Enzyme Inhib Med Chem, 2018; 33; 424-27
26. Umu SU, Langseth H, Bucher-Johannessen C, A comprehensive profile of circulating RNAs in human serum: RNA Biol, 2018; 15; 242-50
27. Umu SU, Langseth H, Bucher-Johannessen C, A comprehensive profile of circulating RNAs in human serum: RNA Biol, 2018; 15; 242-50
28. Gumienny R, Jedlinski DJ, Schmidt A, High-throughput identification of C/D box snoRNA targets with CLIP and RiboMeth-seq: Nucleic Acids Res, 2017; 45; 2341-53
29. Liu CP, Zhang JH, Zheng SC, A novel clinical multidimensional transcriptome signature predicts prognosis in bladder cancer: Oncol Rep, 2018; 40; 2826-35
30. Peng D, Ge G, Xu Z, Diagnostic and prognostic biomarkers of common urological cancers based on aberrant DNA methylation: Epigenomics, 2018; 10; 1189-99
31. Peng F, Li H, Xiao H, Identification of a three-miRNA signature as a novel potential prognostic biomarker in patients with bladder cancer: Oncotarget, 2017; 8; 105553-60
32. Bao Z, Zhang W, Dong D, A potential prognostic lncRNA signature for predicting survival in patients with bladder urothelial carcinoma: Oncotarget, 2017; 8; 10485-97
33. Chen CS, Alonso JL, Ostuni E, Cell shape provides global control of focal adhesion assembly: Biochem Biophys Res Commun, 2003; 307; 355-61
34. Liang J, Wen J, Huang Z, Small nucleolar RNAs: Insight into their function in cancer: Front Oncol, 2019; 9; 587
35. Thorenoor N, Slaby O, Small nucleolar RNAs functioning and potential roles in cancer: Tumour Biol, 2015; 36(1); 41-53
36. Wang X, Xu M, Yan Y, Identification of eight small nucleolar RNAs as survival biomarkers and their clinical significance in gastric cancer: Front Oncol, 2019; 9; 788
37. Tai YL, Chen LC, Shen TL, Emerging roles of focal adhesion kinase in cancer: Biomed Res Int, 2015; 2015 690690
38. Kong DB, Chen F, Sima N, Focal adhesion kinases crucially regulate TGFb-induced migration and invasion of bladder cancer cells via Src kinase and E-cadherin: Onco Targets Ther, 2017; 10; 1783-92
39. Alfieri R, Giovannetti E, Bonelli M, Cavazzoni A, New treatment opportunities in phosphatase and tensin homolog (PTEN)-deficient tumors: Focus on PTEN/focal adhesion kinase pathway: Front Oncol, 2017; 7; 170
40. Mousson A, Sick E, Carl P, Targeting focal adhesion kinase using inhibitors of protein-protein interactions: Cancers (Basel), 2018; 10; 278
Figures
 Figure 1. Survival-associated snoRNAs in BLCA. X-axis represents the Z-score of the snoRNAs in univariate Cox analysis and Y-axis represents survival-associated snoRNAs. Thresholds are p<0.05 and |Z-score| >1.8
Figure 1. Survival-associated snoRNAs in BLCA. X-axis represents the Z-score of the snoRNAs in univariate Cox analysis and Y-axis represents survival-associated snoRNAs. Thresholds are p<0.05 and |Z-score| >1.8 Figure 2. K-M plots prognostic snoRNAs included in prognostic signature.
Figure 2. K-M plots prognostic snoRNAs included in prognostic signature. Figure 3. Prognostic signature constructed based on 12 snoRNAs. (A) The risk score assigned to each patient; (B) Survival status of BLCA patients in high- and low-risk group; and (C) The expression pattern of included snoRNAs.
Figure 3. Prognostic signature constructed based on 12 snoRNAs. (A) The risk score assigned to each patient; (B) Survival status of BLCA patients in high- and low-risk group; and (C) The expression pattern of included snoRNAs. Figure 4. Comparation of snoRNA-based prognostic signature and AJCC_TNM stage in predicting the clinical outcome of BLCA patients. (A) K-M survival plots indicated that patients in the high-risk group tended to have poor clinical outcomes; (B) ROC curves with AUCs of prognostic predictors built by snoRNAs in BLCA; (C) K-M survival plots indicated that patients in advanced stage tended to have poor clinical outcomes; and (D) ROC curves with AUCs of AJCC_TNM stage.
Figure 4. Comparation of snoRNA-based prognostic signature and AJCC_TNM stage in predicting the clinical outcome of BLCA patients. (A) K-M survival plots indicated that patients in the high-risk group tended to have poor clinical outcomes; (B) ROC curves with AUCs of prognostic predictors built by snoRNAs in BLCA; (C) K-M survival plots indicated that patients in advanced stage tended to have poor clinical outcomes; and (D) ROC curves with AUCs of AJCC_TNM stage. Figure 5. Nomogram of BLCA patients.
Figure 5. Nomogram of BLCA patients. Figure 6. Correlation network of included snoRNAs and mRNAs. Expression of survival-associated snoRNAs (green dots) were positively (red line)/negatively (blue line) correlated with the expression level of mRNAs (red dots).
Figure 6. Correlation network of included snoRNAs and mRNAs. Expression of survival-associated snoRNAs (green dots) were positively (red line)/negatively (blue line) correlated with the expression level of mRNAs (red dots). Figure 7. Gene ontology of prognostic signature-related genes. (A) Biological process; (B) Cellular component; (C) Molecular function.
Figure 7. Gene ontology of prognostic signature-related genes. (A) Biological process; (B) Cellular component; (C) Molecular function. Figure 8. Disease ontology and Kyoto Encyclopedia of Genes and Genomes pathway of prognostic signature-related genes. (A) Disease ontology; (B) Kyoto Encyclopedia of Genes and Genomes pathway.
Figure 8. Disease ontology and Kyoto Encyclopedia of Genes and Genomes pathway of prognostic signature-related genes. (A) Disease ontology; (B) Kyoto Encyclopedia of Genes and Genomes pathway. Tables
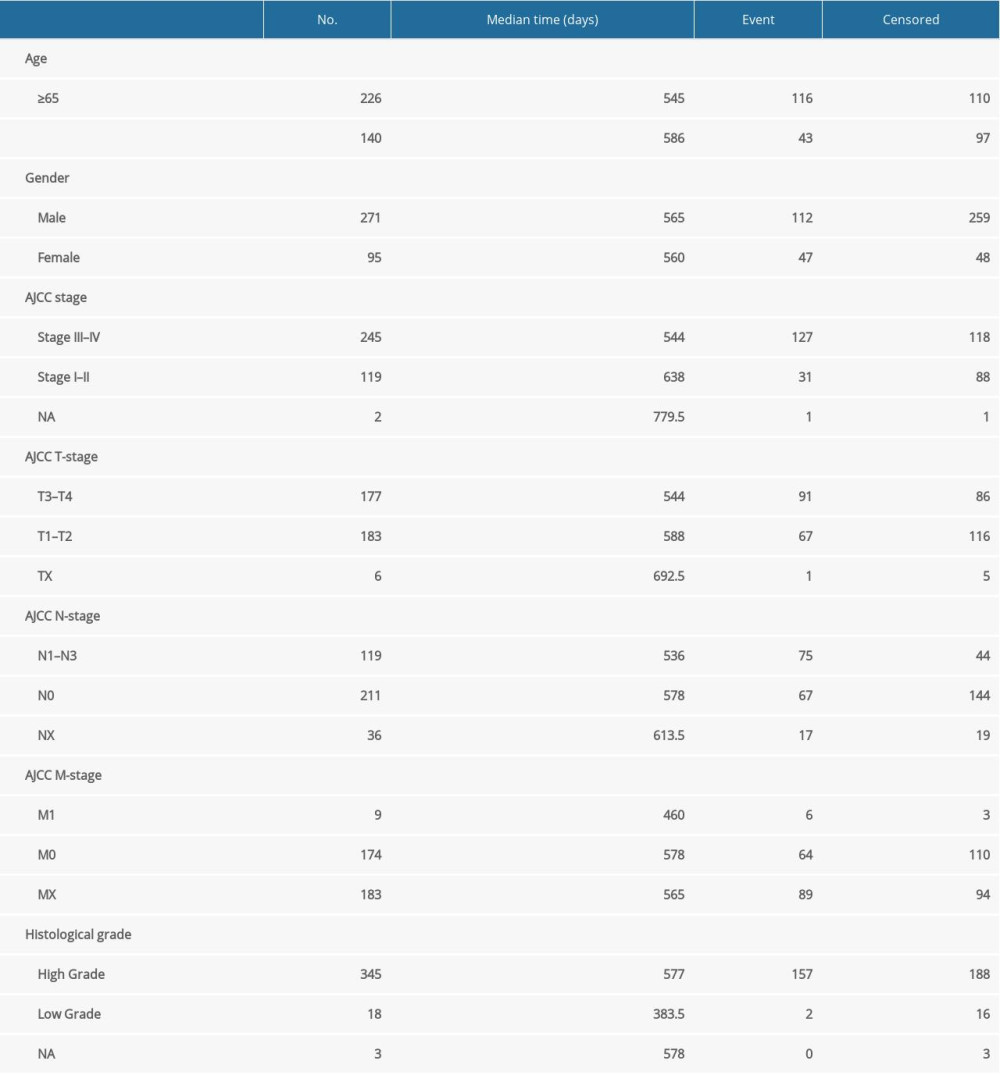 Table 1. Clinical information of included BLCA patients.
Table 1. Clinical information of included BLCA patients.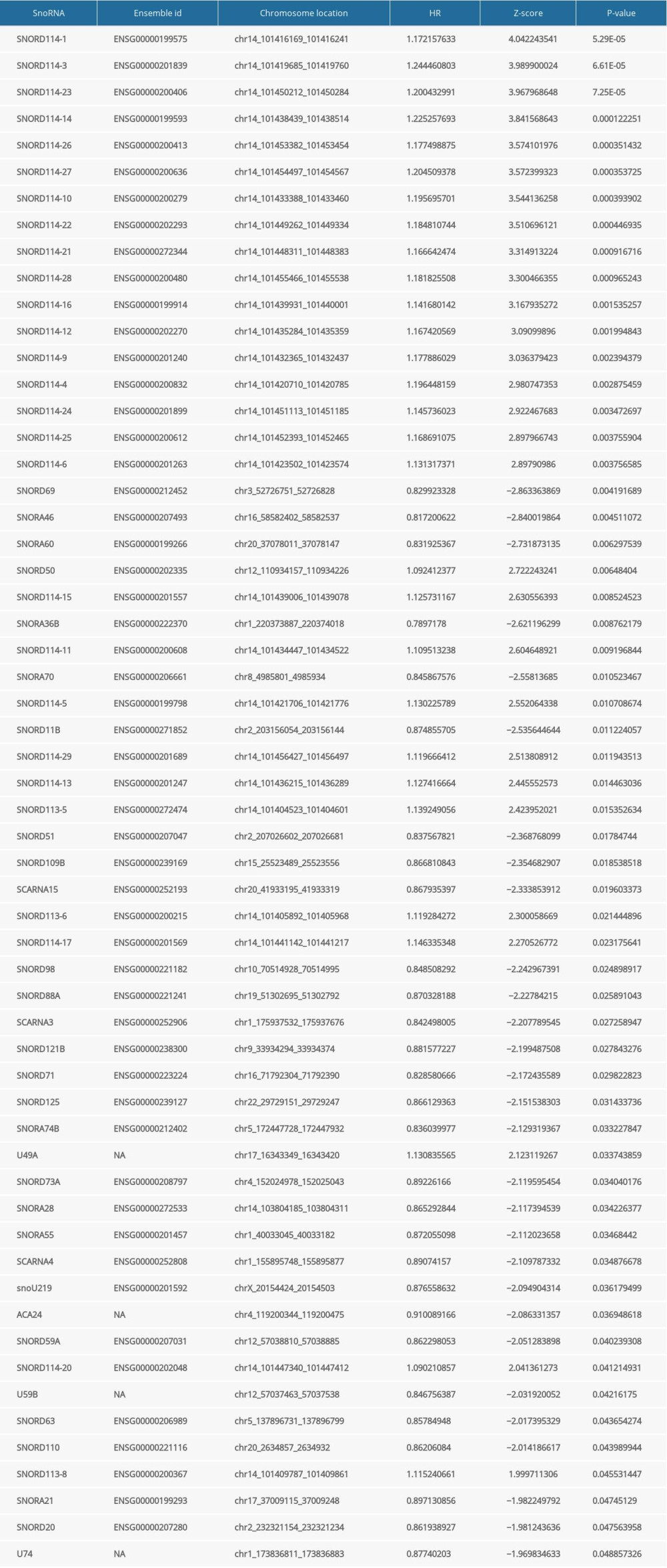 Table 2. General characteristics of prognosis-related snoRNAs in BLCA.
Table 2. General characteristics of prognosis-related snoRNAs in BLCA.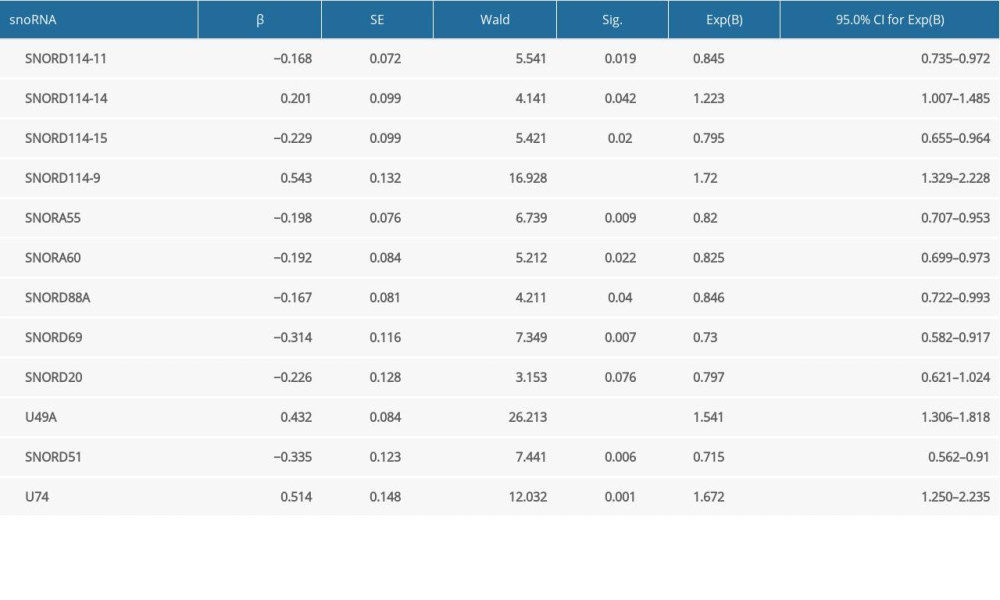 Table 3. The results of multivariate analysis selection.
Table 3. The results of multivariate analysis selection.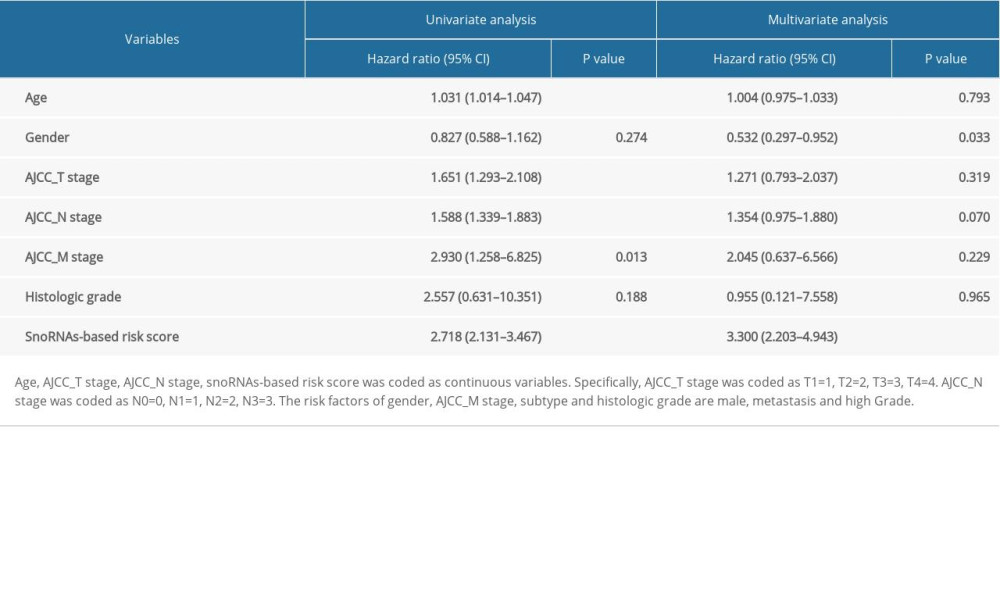 Table 4. Multivariate Cox analysis of OS in BLCA patients of TCGA.
Table 4. Multivariate Cox analysis of OS in BLCA patients of TCGA. Table 1. Clinical information of included BLCA patients.
Table 1. Clinical information of included BLCA patients. Table 2. General characteristics of prognosis-related snoRNAs in BLCA.
Table 2. General characteristics of prognosis-related snoRNAs in BLCA. Table 3. The results of multivariate analysis selection.
Table 3. The results of multivariate analysis selection. Table 4. Multivariate Cox analysis of OS in BLCA patients of TCGA.
Table 4. Multivariate Cox analysis of OS in BLCA patients of TCGA. In Press
Clinical Research
Institutional and Regional Variations in Access to Clinical Trials and Next-Generation Sequencing in Turkis...Med Sci Monit In Press; DOI: 10.12659/MSM.951027
Clinical Research
Low-Intensity Blood Flow-Restricted Multi-Joint Exercise Improves Muscle Function in Patients With Patellof...Med Sci Monit In Press; DOI: 10.12659/MSM.950516
Review article
Musculoskeletal Ultrasound and MRI in the Evaluation of Chemotherapy-Induced Peripheral Neuropathy: A ReviewMed Sci Monit In Press; DOI: 10.12659/MSM.951283
Clinical Research
Sensory Processing, Dissociation, and Affective Symptoms in Misophonia: A Cross-Sectional Study of 35 AdultsMed Sci Monit In Press; DOI: 10.12659/MSM.950938
Most Viewed Current Articles
17 Jan 2024 : Review article 10,187,196
Vaccination Guidelines for Pregnant Women: Addressing COVID-19 and the Omicron VariantDOI :10.12659/MSM.942799
Med Sci Monit 2024; 30:e942799
13 Nov 2021 : Clinical Research 3,708,487
Acceptance of COVID-19 Vaccination and Its Associated Factors Among Cancer Patients Attending the Oncology ...DOI :10.12659/MSM.932788
Med Sci Monit 2021; 27:e932788
14 Dec 2022 : Clinical Research 2,341,643
Prevalence and Variability of Allergen-Specific Immunoglobulin E in Patients with Elevated Tryptase LevelsDOI :10.12659/MSM.937990
Med Sci Monit 2022; 28:e937990
16 May 2023 : Clinical Research 706,524
Electrophysiological Testing for an Auditory Processing Disorder and Reading Performance in 54 School Stude...DOI :10.12659/MSM.940387
Med Sci Monit 2023; 29:e940387








